Recurrent Pregnancy Loss Etiology, Risk Factors, Diagnosis, and Management. Fresh Look into a Full Box
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Study Selection
3.2. Recurrent Pregnancy Loss Definitions and Terminology
3.3. Epidemiology of Recurrent Pregnancy Loss
3.4. Recurrent Pregnancy Loss Etiology and Risk Factors
3.4.1. Maternal Age
3.4.2. Uterine Factors
3.4.3. Genetic Factors
3.4.4. Endocrine Disorders
3.4.5. Infections
3.4.6. Thrombophilia
3.4.7. Immune Factors
3.4.8. Vitamin D Deficiency
3.4.9. Other Risk Factors
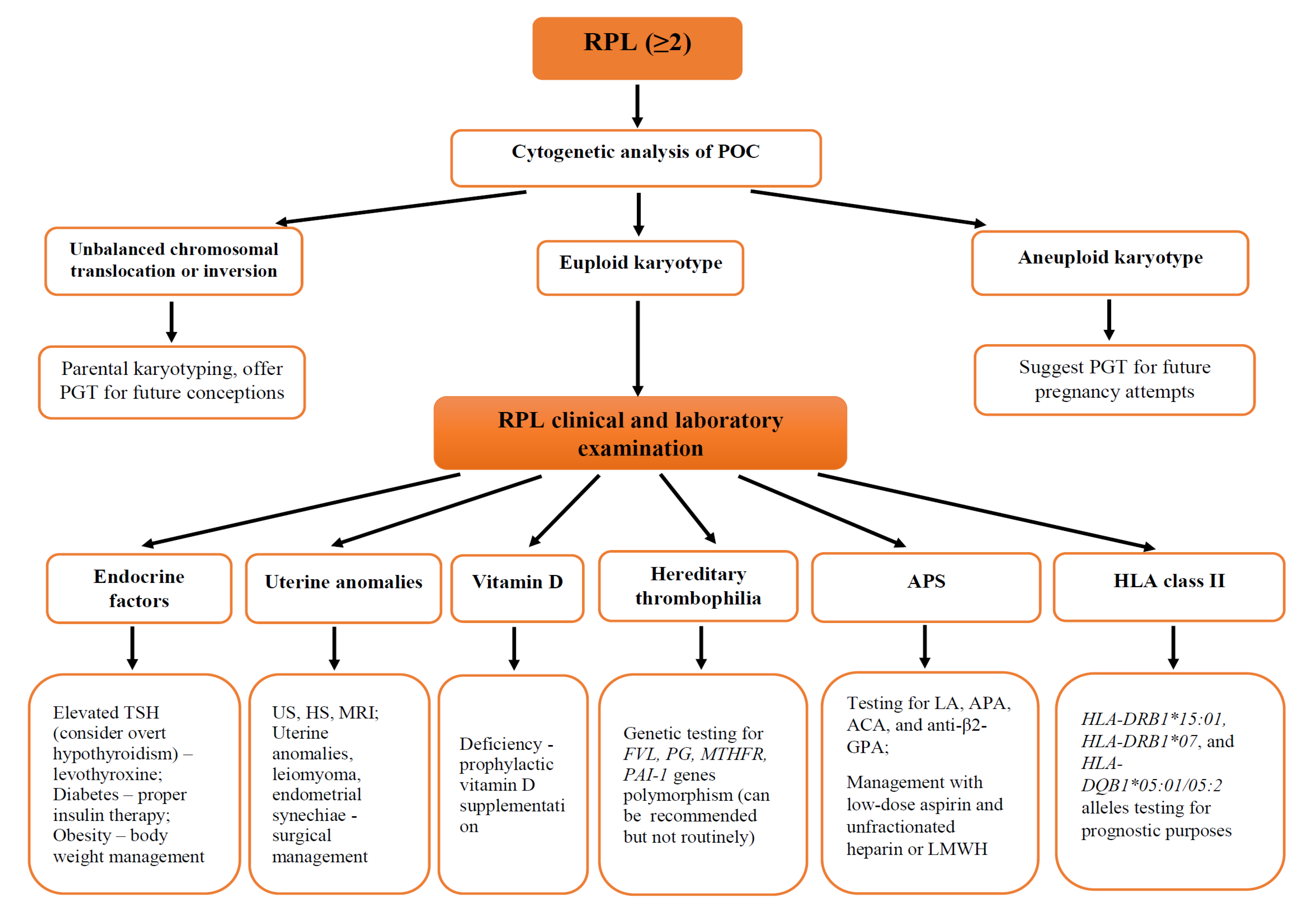
3.5. Diagnostic Approach to Recurrent Pregnancy Loss
3.5.1. Prognostic Tools for Recurrent Pregnancy Loss Prediction
3.5.2. Genetic Factors Identification
3.5.3. Uterine Anomalies Diagnosis
3.5.4. Chronic Endometritis Assessment
3.5.5. Endocrine Factors Evaluation
3.5.6. Thrombophilia Assessment
3.5.7. Immune Factors Evaluation
3.5.8. Vitamin D Levels
3.6. Management of Recurrent Pregnancy Loss
3.6.1. Uterine Anomalies Management
3.6.2. Management of Recurrent Pregnancy Loss Associated with Genetic Factors
3.6.3. Progesterone Therapy
3.6.4. Thyroid Hormone Replacement
3.6.5. Management of Thrombophilia
3.6.6. Treatment of Recurrent Pregnancy Loss Related to Immune Factors
3.6.7. Vitamin D Supplementation
3.7. Psychological Effect of Recurrent Miscarriage
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Quenby, S.; Gallos, I.D.; Dhillon-Smith, R.K.; Podesek, M.; Stephenson, M.D.; Fisher, J.; Brosens, J.J.; Brewin, J.; Ramhorst, R.; Lucas, E.S.; et al. Miscarriage matters: The epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet 2021, 397, 1658–1667. [Google Scholar] [CrossRef] [PubMed]
- Khalife, D.; Ghazeeri, G.; Kutteh, W. Review of current guidelines for recurrent pregnancy loss: New strategies for optimal evaluation of women who may be superfertile. Semin. Perinatol. 2019, 43, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Royal College of Obstetricians & Gynaecologists. The Investigation and Treatment of Couples with Recurrent First-Trimester and Second Trimester Miscarriage; Guideline No. 17; Royal College of Obstetricians & Gynaecologists: London, UK, 2011. [Google Scholar]
- The ESHRE Guideline Group on RPL; Bender Atik, R.; Christiansen, O.B.; Elson, J.; Kolte, A.M.; Lewis, S.; Middeldorp, S.; Nelen, W.; Peramo, B.; Quenby, S.; et al. ESHRE guideline: Recurrent pregnancy loss. Hum. Reprod. Open 2018, 2018, hoy004. [Google Scholar] [CrossRef] [PubMed]
- The ESHRE Guideline Group on RPL; Atik, R.B.; Christiansen, O.B.; Elson, J.; Kolte, A.M.; Lewis, S.; Middeldorp, S.; Mcheik, S.; Peramo, B.; Quenby, S.; et al. ESHRE guideline: Recurrent pregnancy loss: An update in 2022. Hum. Reprod. Open 2022, 2023, hoad002. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: A committee opinion. Fertil. Steril. 2012, 98, 1103–1111. [Google Scholar] [CrossRef]
- Practice Committee of American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: A committee opinion. Fertil. Steril. 2013, 99, 63. [Google Scholar] [CrossRef] [PubMed]
- Practice Committee of the American Society for Reproductive Medicine. Definitions of infertility and recurrent pregnancy loss: A committee opinion. Fertil. Steril. 2020, 113, 533–535. [Google Scholar] [CrossRef]
- Youssef, A.; Vermeulen, N.; Lashley, E.E.L.O.; Goddijn, M.; van der Hoorn, M.L.P. Comparison and appraisal of (inter)national recurrent pregnancy loss guidelines. Reprod. Biomed. Online 2019, 39, 497–503. [Google Scholar] [CrossRef] [Green Version]
- Coomarasamy, A.; Dhillon-Smith, R.K.; Papadopoulou, A.; Al-Memar, M.; Brewin, J.; Abrahams, V.M.; Maheshwari, A.; Christiansen, O.B.; Stephenson, M.D.; Goddijn, M.; et al. Recurrent miscarriage: Evidence to accelerate action. Lancet 2021, 397, 1675–1682. [Google Scholar] [CrossRef]
- La, X.; Wang, W.; Zhang, M.; Liang, L. Definition and Multiple Factors of Recurrent Spontaneous Abortion. Adv. Exp. Med. Biol. 2021, 1300, 231–257. [Google Scholar] [CrossRef]
- Aimagambetova, G.; Issanov, A.; Terzic, S.; Bapayeva, G.; Ukybassova, T.; Baikoshkarova, S.; Aldiyarova, A.; Shauyen, F.; Terzic, M. The effect of psychological distress on IVF outcomes: Reality or speculations? PLoS ONE 2020, 15, e0242024. [Google Scholar] [CrossRef]
- Bapayeva, G.; Aimagambetova, G.; Issanov, A.; Terzic, S.; Ukybassova, T.; Aldiyarova, A.; Utepova, G.; Daribay, Z.; Bekbossinova, G.; Balykov, A.; et al. The Effect of Stress, Anxiety and Depression on In Vitro Fertilization Outcome in Kazakhstani Public Clinical Setting: A Cross-Sectional Study. J. Clin. Med. 2021, 10, 937. [Google Scholar] [CrossRef]
- Issanov, A.; Aimagambetova, G.; Terzic, S.; Bapayeva, G.; Ukybassova, T.; Baikoshkarova, S.; Utepova, G.; Daribay, Z.; Bekbossinova, G.; Balykov, A.; et al. Impact of governmental support to the IVF clinical pregnancy rates: Differences between public and private clinical settings in Kazakhstan—A prospective cohort study. BMJ Open 2022, 12, e049388. [Google Scholar] [CrossRef]
- Li, Y.H.; Marren, A. Recurrent pregnancy loss: A summary of international evidence-based guidelines and practice. Aust. J. Gen. Pract. 2018, 47, 432–436. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Recommended definitions, terminology, and format for statistical tables related to the perinatal period and use of a new certificate for cause of perinatal deaths. Modifications recommended by FIGO as amended 14 October 1976. Acta Obstet. Gynecol. Scand. 1977, 56, 247–253. [Google Scholar]
- Boogaard, E.V.D.; Kaandorp, S.P.; Franssen, M.T.M.; Mol, B.W.J.; Leschot, N.J.; Wouters, C.H.; van der Veen, F.; Korevaar, J.C.; Goddijn, M. Consecutive or non-consecutive recurrent miscarriage: Is there any difference in carrier status? Hum. Reprod. 2010, 25, 1411–1414. [Google Scholar] [CrossRef] [Green Version]
- Dimitriadis, E.; Menkhorst, E.; Saito, S.; Kutteh, W.H.; Brosens, J.J. Recurrent pregnancy loss. Nat. Rev. Dis. Prim. 2020, 6, 98. [Google Scholar] [CrossRef]
- Toth, B.; Würfel, W.; Bohlmann, M.; Zschocke, J.; Rudnik-Schöneborn, S.; Nawroth, F.; Schleußner, E.; Rogenhofer, N.; Wischmann, T.; von Wolff, M.; et al. Recurrent Miscarriage: Diagnostic and Therapeutic Procedures. Guideline of the DGGG, OEGGG and SGGG (S2k-Level, AWMF Registry Number 015/050). Geburtshilfe Frauenheilkd. 2018, 78, 364–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. International Classification of the Diseases, 10th ed.; WHO: Geneva, Switzerland, 2019; Available online: https://icd.who.int/browse10/2019/en#/ (accessed on 20 January 2023).
- Kolte, A.M.; Bernardi, L.A.; Christiansen, O.B.; Quenby, S.; Farquharson, R.G.; Goddijn, M.; Stephenson, M.D.; ESHRE Special Interest Group, Early Pregnancy. Terminology for pregnancy loss prior to viability: A consensus statement from the ESHRE early pregnancy special interest group. Hum. Reprod. 2014, 30, 495–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardinale, C.; Berbis, J.; Chau, C.; Bernard, F.; Arnoux, D.; Fratacci, M.-F.; Boubli, L.; Bretelle, F. Two miscarriages, consecutive or non-consecutive, does it change something? J. Gynecol. Obstet. Hum. Reprod. 2017, 46, 721–725. [Google Scholar] [CrossRef] [PubMed]
- Egerup, P.; Kolte, A.; Larsen, E.; Krog, M.; Nielsen, H.; Christiansen, O. Recurrent pregnancy loss: What is the impact of consecutive versus non-consecutive losses? Hum. Reprod. 2016, 31, 2428–2434. [Google Scholar] [CrossRef] [Green Version]
- Jackson, T.M.; Watkins, E.D. Early pregnancy loss. J. Am. Acad. Physician Assist. 2021, 34, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Dugas, C.; Slane, V.H. Miscarriage. In StatPearls; StatPearls Publishing: Tampa, FL, USA, 27 June 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK532992/ (accessed on 1 January 2023).
- A du Fossé, N.; van der Hoorn, M.-L.P.; van Lith, J.M.M.; le Cessie, S.; Lashley, E.E.L.O. Advanced paternal age is associated with an increased risk of spontaneous miscarriage: A systematic review and meta-analysis. Hum. Reprod. Updat. 2020, 26, 650–669. [Google Scholar] [CrossRef] [PubMed]
- Robinson, G.E. Pregnancy loss. Best Pract. Res. Clin. Rheumatol. 2014, 28, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Arias-Sosa, L.A.; Acosta, I.D.; Lucena-Quevedo, E.; Moreno-Ortiz, H.; Esteban-Pérez, C.; Forero-Castro, M. Genetic and epigenetic variations associated with idiopathic recurrent pregnancy loss. J. Assist. Reprod. Genet. 2018, 35, 355–366. [Google Scholar] [CrossRef]
- Branch, D.W.; Gibson, M.; Silver, R.M. Clinical practice. Recurrent miscarriage. N. Engl. J. Med. 2010, 363, 1740–1747. [Google Scholar] [CrossRef]
- Larsen, E.C.; Christiansen, O.B.; Kolte, A.M.; Macklon, N. New insights into mechanisms behind miscarriage. BMC Med. 2013, 11, 11154. [Google Scholar] [CrossRef] [Green Version]
- Redinger, A.; Nguyen, H. Incomplete Abortions. [Updated 2022 Jun 27]. In StatPearls [Internet]; StatPearls Publishing: Tampa, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559071/ (accessed on 10 January 2023).
- Salmanov, A.G.; Ishchak, O.M.; Shostak, Y.M.; Kozachenko, V.V.; Rud, V.O.; Golyanovskiy, O.V.; Shkorbotun, V.O. Bacterial infection causes of pregnancy loss and prema-ture birth in the women in Ukraine. Wiad. Lek. 2021, 74, 1355–1359. [Google Scholar] [CrossRef]
- Abdukassimova, M.; Kanabekova, P.; Bauyrzhanova, Z.; Ukybassova, T.; Kaldygulova, L.; Imankulova, B.; Aimagambetova, G.; Almawi, W.Y. Association of Human forkhead box protein 3 (FOXP3) gene polymorphisms with idiopathic recurrent pregnancy loss among Kazakhstani women. Gene 2021, 801, 145835. [Google Scholar] [CrossRef]
- Aimagambetova, G.; Hajjej, A.; Malalla, Z.H.; Finan, R.R.; Sarray, S.; Almawi, W.Y. Maternal HLA-DR, HLA-DQ, and HLA-DP loci are linked with altered risk of recurrent pregnancy loss in Lebanese women: A case-control study. Am. J. Reprod. Immunol. 2019, 82, e13173. [Google Scholar] [CrossRef]
- Christiansen, O.; Jauniaux, E.; Farquharson, R.; Exalto, N. Reply: Evidence-based guidelines for the investigation and medical treatment of recurrent miscarriage. Hum. Reprod. 2006, 22, 309–311. [Google Scholar] [CrossRef] [Green Version]
- Stephenson, M.; Awartani, K.; Robinson, W. Cytogenetic analysis of miscarriages from couples with recurrent miscarriage: A case-control study. Hum. Reprod. 2002, 17, 446–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rasmark Roepke, E.; Matthiesen, L.; Rylance, R.; Christiansen, O.B. Is the incidence of recurrent pregnancy loss increasing? A ret-rospective register-based study in Sweden. Acta Obstet. Gynecol. Scand. 2017, 96, 1365–1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, M.M.; Visser, J.; Verburg, H.; Hukkelhoven, C.; Van Lith, J.M.; Bloemenkamp, K.W. Pregnancy before recurrent pregnancy loss more often complicated by post-term birth and perinatal death. Acta Obstet. Gynecol. Scand. 2017, 97, 82–88. [Google Scholar] [CrossRef] [Green Version]
- Huang, T.; Owolabi, T.; Summers, A.M.; Meier, C.; Wyatt, P.R. The identification of risk of spontaneous fetal loss through second-trimester maternal serum screening. Am. J. Obstet. Gynecol. 2005, 193, 395–403. [Google Scholar] [CrossRef]
- Jaslow, C.R.; Carney, J.L.; Kutteh, W.H. Diagnostic factors identified in 1020 women with two versus three or more recurrent pregnancy losses. Fertil. Steril. 2010, 93, 1234–1243. [Google Scholar] [CrossRef]
- Van Dijk, M.M.; Kolte, A.M.; Limpens, J.; Kirk, E.; Quenby, S.; van Wely, M.; Goddijn, M. Recurrent pregnancy loss: Diagnostic workup after two or three pregnancy losses? A systematic review of the literature and meta-analysis. Hum. Reprod. Updat. 2020, 26, 356–367. [Google Scholar] [CrossRef] [Green Version]
- Meuleman, T.; Lashley, L.E.; Dekkers, O.M.; van Lith, J.M.; Claas, F.H.; Bloemenkamp, K.W. HLA associations and HLA sharing in recurrent miscarriage: A systematic review and meta-analysis. Hum. Immunol. 2015, 76, 362–373. [Google Scholar] [CrossRef]
- Diejomaoh, M.F.; Bello, Z.; Al Jassar, W.; Jirous, J.; Karunakaran, K.; Mohammed, A.T. Consecutive successful pregnancies subsequent to intravenous immunoglobulin therapy in a patient with recurrent spontaneous miscarriage. Int. Med. Case Rep. J. 2015, 8, 337–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahine, L.; Lathi, R. Recurrent Pregnancy Loss. Obstet. Gynecol. Clin. N. Am. 2015, 42, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Deng, T.B.; Liao, X.B.; Zhu, S. Recent Advances in Treatment of Recurrent Spontaneous Abortion. Obstet. Gynecol. Surv. 2022, 77, 355–366. [Google Scholar] [CrossRef]
- Nybo Andersen, A.-M.; Wohlfahrt, J.; Christens, P.; Olsen, J.; Melbye, M. Maternal age and fetal loss: Population based register linkage study. BMJ 2000, 320, 1708–1712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, K.; Case, A.; Reproductive Endocrinology and Infertility Committee. Advanced reproductive age and fertility. J. Obstet. Gynaecol. Can. 2011, 33, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Vollenhoven, B.; Hunt, S. Ovarian ageing and the impact on female fertility. F1000Research 2018, 7, 1835. [Google Scholar] [CrossRef] [PubMed]
- Magnus, M.C.; Wilcox, A.J.; Morken, N.-H.; Weinberg, C.; Håberg, S.E. Role of maternal age and pregnancy history in risk of miscarriage: Prospective register based study. BMJ 2019, 364, l869. [Google Scholar] [CrossRef] [Green Version]
- Carbonnel, M.; Pirtea, P.; de Ziegler, D.; Ayoubi, J.M. Uterine factors in recurrent pregnancy losses. Fertil. Steril. 2021, 115, 538–545. [Google Scholar] [CrossRef]
- Ono, S.; Yonezawa, M.; Watanabe, K.; Abe, T.; Mine, K.; Kuwabara, Y.; Akira, S.; Takeshita, T. Retrospective cohort study of the risk factors for secondary infertility following hysteroscopic metroplasty of the uterine septum in women with recurrent pregnancy loss. Reprod. Med. Biol. 2017, 17, 77–81. [Google Scholar] [CrossRef] [Green Version]
- Kolte, A.M.; Steffensen, R.; Christiansen, O.B.; Nielsen, H.S. Maternal HY-restricting HLA class II alleles are associated with poor long-term outcome in recurrent pregnancy loss after a boy. Am. J. Reprod. Immunol. 2016, 76, 400–405. [Google Scholar] [CrossRef]
- Lee, H.A.; Ahn, E.H.; Kim, J.H.; Kim, J.O.; Ryu, C.; Lee, J.Y.; Cho, S.; Lee, W.; Kim, N. Association study of frame-shift and splice variant polymorphisms with risk of idiopathic recurrent pregnancy loss. Mol. Med. Rep. 2018, 18, 2417–2426. [Google Scholar]
- Pereza, N.; Ostojić, S.; Kapović, M.; Peterlin, B. Systematic review and meta-analysis of genetic association studies in idiopathic recurrent spontaneous abortion. Fertil. Steril. 2016, 107, 150–159.e2. [Google Scholar] [CrossRef] [Green Version]
- Bahia, W.; Finan, R.; Al-Mutawa, M.; Haddad, A.; Soua, A.; Janhani, F.; Mahjoub, T.; Almawi, W. Genetic variation in the progesterone receptor gene and susceptibility to recurrent pregnancy loss: A case-control study. BJOG Int. J. Obstet. Gynaecol. 2017, 125, 729–735. [Google Scholar] [CrossRef] [Green Version]
- Dendana, M.; Bahia, W.; Finan, R.R.; Al-Mutawa, M.; Almawi, W.Y. Association of adiponectin gene variants with idiopathic re-current miscarriage according to obesity status: A case-control study. J. Transl. Med. 2018, 16, 76. [Google Scholar] [CrossRef] [Green Version]
- Grimstad, F.; Krieg, S. Immunogenetic contributions to recurrent pregnancy loss. J. Assist. Reprod. Genet. 2016, 33, 833–847. [Google Scholar] [CrossRef] [Green Version]
- Rai, R.; Regan, L. Recurrent miscarriage. Lancet 2006, 368, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, A.E.; Silver, R.M.; LaCoursiere, D.Y.; Porter, T.F.; Branch, D.W. Recurrent Fetal Aneuploidy and Recurrent Miscarriage. Obstet. Gynecol. 2004, 104, 784–788. [Google Scholar] [CrossRef] [PubMed]
- Colley, E.; Hamilton, S.; Smith, P.; Morgan, N.V.; Coomarasamy, A.; Allen, S. Potential genetic causes of miscarriage in euploid pregnancies: A systematic review. Hum. Reprod. Updat. 2019, 25, 452–472. [Google Scholar] [CrossRef] [PubMed]
- Bagislar, S.; Üstüner, I.; Cengiz, B.; Söylemez, F.; Akyerli, C.B.; Ceylaner, S.; Ceylaner, G.; Acar, A.; Özçelik, T.; Üstüner, I. Extremely skewed X-chromosome inactivation patterns in women with recurrent spontaneous abortion. Aust. N. Z. J. Obstet. Gynaecol. 2006, 46, 384–387. [Google Scholar] [CrossRef]
- Almawi, W.Y.; Saldanha, F.L.; Mahmood, N.A.; Al-Zaman, I.; Sater, M.S.; Mustafa, F.E. Relationship between VEGFA polymorphisms and serum VEGF protein levels and recurrent spontaneous miscarriage. Hum. Reprod. 2013, 28, 2628–2635. [Google Scholar] [CrossRef] [Green Version]
- Pereza, N.; Ostojić, S.; Smirčić, A.; Hodžić, A.; Kapović, M.; Peterlin, B. The −2549 insertion/deletion polymorphism in the promoter region of the VEGFA gene in couples with idiopathic recurrent spontaneous abortion. J. Assist. Reprod. Genet. 2015, 32, 1789–1794. [Google Scholar] [CrossRef] [Green Version]
- Negro, R.; Stagnaro-Green, A. Clinical Aspects of Hyperthyroidism, Hypothyroidism, and Thyroid Screening in Pregnancy. Endocr. Pract. 2014, 20, 597–607. [Google Scholar] [CrossRef]
- Dhillon-Smith, R.K.; Middleton, L.J.; Sunner, K.K.; Cheed, V.; Baker, K.; Farrell-Carver, S.; Bender-Atik, R.; Agrawal, R.; Bhatia, K.; Edi-Osagie, E.; et al. Levothyroxine in women with thyroid peroxidase antibodies before conception. N. Engl. J. Med. 2019, 380, 1316–1325. [Google Scholar] [CrossRef] [PubMed]
- Duane, M.; Schliep, K.; Porucznik, C.A.; Najmabadi, S.; Stanford, J.B. Does a short luteal phase correlate with an increased risk of miscarriage? A cohort study. BMC Pregnancy Childbirth 2022, 22, 922. [Google Scholar] [CrossRef] [PubMed]
- Coomarasamy, A.; Devall, A.J.; Brosens, J.J.; Quenby, S.; Stephenson, M.D.; Sierra, S.; Christiansen, O.B.; Small, R.; Brewin, J.; Roberts, T.E.; et al. Micronized vaginal progesterone to prevent miscarriage: A critical evaluation of randomized evidence. Am. J. Obstet. Gynecol. 2020, 223, 167–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavalcante, M.B.; Sarno, M.; Peixoto, A.B.; Júnior, E.A.; Barini, R. Obesity and recurrent miscarriage: A systematic review and meta-analysis. J. Obstet. Gynaecol. Res. 2018, 45, 30–38. [Google Scholar] [CrossRef]
- Eapen, A.; Hayes, E.T.; McQueen, D.B.; Beestrum, M.; Eyck, P.T.; Boots, C. Mean differences in maternal body mass index and recurrent pregnancy loss: A systematic review and meta-analysis of observational studies. Fertil. Steril. 2021, 116, 1341–1348. [Google Scholar] [CrossRef]
- Terzic, M.; Aimagambetova, G.; Terzic, S.; Radunovic, M.; Bapayeva, G.; Laganà, A.S. Periodontal Pathogens and Preterm Birth: Current Knowledge and Further Interventions. Pathogens 2021, 10, 730. [Google Scholar] [CrossRef]
- Baqer, N.N.; Saheb, E.J.; Ahmed, N.S.; Alhadad, N.A.A. The association of IL-3, IL-17A, and IL 27 serum levels with susceptibility to toxoplasmosis in recurrent abortion of Iraqi women. Exp. Parasitol. 2022, 234, 108217. [Google Scholar] [CrossRef]
- McQueen, D.B.; Maniar, K.P.; Hutchinson, A.; Confino, R.; Bernardi, L.; Pavone, M.E. Redefining chronic endometritis: The importance of endometrial stromal changes. Fertil. Steril. 2021, 116, 855–861. [Google Scholar] [CrossRef]
- McQueen, D.B.; Maniar, K.P.; Hutchinson, A.; Confino, R.; Bernardi, L.; Pavone, M.E. Retained pregnancy tissue after miscarriage is associated with high rate of chronic endometritis. J. Obstet. Gynaecol. 2022, 42, 3101–3105. [Google Scholar] [CrossRef]
- Pirtea, P.; Cicinelli, E.; De Nola, R.; de Ziegler, D.; Ayoubi, J.M. Endometrial causes of recurrent pregnancy losses: Endometriosis, adenomyosis, and chronic endometritis. Fertil. Steril. 2021, 115, 546–560. [Google Scholar] [CrossRef]
- Bouet, P.-E.; El Hachem, H.; Monceau, E.; Gariépy, G.; Kadoch, I.-J.; Sylvestre, C. Chronic endometritis in women with recurrent pregnancy loss and recurrent implantation failure: Prevalence and role of office hysteroscopy and immunohistochemistry in diagnosis. Fertil. Steril. 2016, 105, 106–110. [Google Scholar] [CrossRef] [Green Version]
- Shi, Y.; Yamada, H.; Sasagawa, Y.; Tanimura, K.; Deguchi, M. Uterine endometrium microbiota and pregnancy outcome in women with recurrent pregnancy loss. J. Reprod. Immunol. 2022, 152, 103653. [Google Scholar] [CrossRef]
- Moreno, I.; Cicinelli, E.; Garcia-Grau, I.; Gonzalez-Monfort, M.; Bau, D.; Vilella, F.; De Ziegler, D.; Resta, L.; Valbuena, D.; Simon, C. The diagnosis of chronic endometritis in infertile asymptomatic women: A comparative study of histology, microbial cultures, hysteroscopy, and molecular microbiology. Am. J. Obstet. Gynecol. 2018, 218, 602.e1–602.e16. [Google Scholar] [CrossRef] [Green Version]
- Peuranpää, P.; Holster, T.; Saqib, S.; Kalliala, I.; Tiitinen, A.; Salonen, A.; Hautamäki, H. Female reproductive tract microbiota and recurrent pregnancy loss: A nested case-control study. Reprod. Biomed. Online 2022, 45, 1021–1031. [Google Scholar] [CrossRef] [PubMed]
- Masucci, L.; D’ippolito, S.; De Maio, F.; Quaranta, G.; Mazzarella, R.; Bianco, D.M.; Castellani, R.; Inversetti, A.; Sanguinetti, M.; Gasbarrini, A.; et al. Celiac Disease Predisposition and Genital Tract Microbiota in Women Affected by Recurrent Pregnancy Loss. Nutrients 2023, 15, 221. [Google Scholar] [CrossRef]
- Di Biase, A.R.; Marasco, G.; Ravaioli, F.; Dajti, E.; Colecchia, L.; Righi, B.; D’Amico, V.; Festi, D.; Iughetti, L.; Colecchia, A. Gut microbiota signatures and clinical manifestations in celiac disease children at onset: A pilot study. J. Gastroenterol. Hepatol. 2020, 36, 446–454. [Google Scholar] [CrossRef]
- Cao, Y.; Wang, X.; Cao, Z.; Cheng, X. Association of Vitamin D receptor gene TaqI polymorphisms with tuberculosis suscep-tibility: A meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 10187–10203. [Google Scholar]
- Terzic, M.; Aimagambetova, G. Prothrombotic gene polymorphisms and adverse reproductive outcomes in assisted reproductive technology. In Management of Infertility: A Practical Approach; Academic Press: Cambridge, MA, USA, 2023; pp. 55–63. ISBN 978-0-323-89907-9. [Google Scholar] [CrossRef]
- Alecsandru, D.; Klimczak, A.M.; Velasco, J.A.G.; Pirtea, P.; Franasiak, J.M. Immunologic causes and thrombophilia in recurrent pregnancy loss. Fertil. Steril. 2021, 115, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, Y.; Ye, C.; Xing, D.; Wu, R.; Li, F.; Chen, L.; Wang, T. Hereditary thrombophilia and recurrent pregnancy loss: A systematic review and meta-analysis. Hum. Reprod. 2021, 36, 1213–1229. [Google Scholar] [CrossRef] [PubMed]
- Mierla, D.; Szmal, C.; Neagos, D.; Cretu, R.; Stoian, V.; Jardan, D. Association of Prothrombin (A20210G) and Factor V Leiden (A506G) with Recurrent Pregnancy Loss. MAEDICA 2012, 7, 222–226. [Google Scholar] [PubMed]
- Karadağ, C.; Akar, B.; Gönenç, G.; Aslancan, R.; Yılmaz, N.; Çalışkan, E. Aspirin, low molecular weight heparin, or both in preventing pregnancy complications in women with recurrent pregnancy loss and factor V Leiden mutation. J. Matern. Neonatal Med. 2019, 33, 1934–1939. [Google Scholar] [CrossRef]
- Villani, M.; Dentali, F.; Colaizzo, D.; Tiscia, G.L.; Vergura, P.; Petruccelli, T.; Petruzzelli, F.; Ageno, W.; Margaglione, M.; Grandone, E. Pregnancy-related venous thrombosis: Comparison between spontaneous and ART conception in an Italian cohort. BMJ Open 2015, 5, e008213. [Google Scholar] [CrossRef] [Green Version]
- American College of Obstetricians and Gynecologists Women’s Health Care Physicians. ACOG Practice Bulletin No. 138: Inherited thrombophilias in pregnancy. Obstet. Gynecol. 2013, 122, 706–716. [Google Scholar] [CrossRef]
- Sergi, C.; Al Jishi, T.; Walker, M. Factor V Leiden mutation in women with early recurrent pregnancy loss: A meta-analysis and systematic review of the causal association. Arch. Gynecol. Obstet. 2014, 291, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Franco, R.F.; Reitsma, P.H. Genetic risk factors of venous thrombosis. Hum. Genet. 2001, 109, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Barut, M.U.; Bozkurt, M.; Kahraman, M.; Yıldırım, E.; Imirzalioğlu, N.; Kubar, A.; Sak, S.; Ağaçayak, E.; Aksu, T.; Çoksüer, H. Thrombophilia and Recurrent Pregnancy Loss: The Enigma Continues. J. Exp. Clin. Res. 2018, 24, 4288–4294. [Google Scholar] [CrossRef]
- Dossenbach-Glaninger, A.; Van Trotsenburg, M.; Schneider, B.; Oberkanins, C.; Hopmeier, P. ACE I/D polymorphism and recurrent first trimester pregnancy loss: Interaction with SERPINE1 4G/5G and F13 Val34Leu polymorphisms. Br. J. Haematol. 2008, 141, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Kim, J.-H.; Song, G.G.; Choi, S.J. Association of the F13A1 Val34Leu polymorphism and recurrent pregnancy loss: A meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 215, 234–240. [Google Scholar] [CrossRef]
- Dossenbach-Glaninger, A.; van Trotsenburg, M.; Oberkanins, C.; Atamaniuk, J. Risk for Early Pregnancy Loss by Factor XIII Val34Leu: The Impact of Fibrinogen Concentration. J. Clin. Lab. Anal. 2013, 27, 444–449. [Google Scholar] [CrossRef]
- Guo, X.; Yi, H.; Li, T.C.; Wang, Y.; Wang, H.; Chen, X. Role of Vascular Endothelial Growth Factor (VEGF) in Human Embryo Implantation: Clinical Implications. Biomolecules 2021, 11, 253. [Google Scholar] [CrossRef]
- Amirchaghmaghi, E.; Rezaei, A.; Moini, A.; Roghaei, M.A.; Hafezi, M.; Aflatoonian, R. Gene expression analysis of VEGF and its receptors and assessment of its serum level in unexplained recurrent spontaneous abortion. Cell J. 2015, 16, 538–545. [Google Scholar] [CrossRef]
- Xu, X.; Du, C.; Li, H.; Du, J.; Yan, X.; Peng, L.; Li, G.; Chen, Z.J. Association of VEGF genetic polymorphisms with recurrent spon-taneous abortion risk: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0123696. [Google Scholar]
- Liew, S.-C.; Das Gupta, E. Methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism: Epidemiology, metabolism and the associated diseases. Eur. J. Med. Genet. 2015, 58, 1–10. [Google Scholar] [CrossRef]
- Kang, S.; Zhou, J.; Wong, P.; Kowalisyn, J.; Strokosch, G. Intermediate homocysteinaemia: A thermolabile variant of meth-ylenetrahydrofolate reductase. Am. J. Hum. Genet. 1988, 43, 414–421. [Google Scholar]
- Dean, L. Methylenetetrahydrofolate reductase deficiency. In Medical Genetics Summaries; Pratt, V.M., Scott, S.A., Pirmohamed, M., Esquivel, B., Kattman, B.L., Malheiro, A.J., Eds.; National Center for Biotechnology Information (US): Bethesda, MD, USA, 2012. [Google Scholar]
- Kaldygulova, L.; Ukybassova, T.; Aimagambetova, G.; Gaiday, A.; Tussupkaliyev, A. Biological Role of Folic Acid in Pregnancy and Possible Therapeutic Application for the Prevention of Preeclampsia. Biomedicines 2023, 11, 272. [Google Scholar] [CrossRef] [PubMed]
- Akilzhanova, A.; Takamura, N.; Kusano, Y.; Karazhanova, L.; Yamashita, S.; Saito, H.; Aoyagi, K. Association between C677T/MTHFR genotype and homocysteine concentration in a Kazakh population. Asia Pac. J. Clin. Nutr. 2008, 17, 325–329. [Google Scholar] [PubMed]
- Hiraoka, M.; Kagawa, Y. Genetic polymorphisms and folate status. Congenit. Anom. 2017, 57, 142–149. [Google Scholar] [CrossRef] [Green Version]
- Osunkalu, V.; Taiwo, I.; Makwe, C.; Quao, R. Methylene tetrahydrofolate reductase and methionine synthase gene polymorphisms as genetic determinants of pre-eclampsia. Pregnancy Hypertens. 2020, 20, 7–13. [Google Scholar] [CrossRef]
- Ganguly, P.; Alam, S.F. Role of homocysteine in the development of cardiovascular disease. Nutr. J. 2015, 14, 6. [Google Scholar] [CrossRef] [Green Version]
- Gaiday, A.N.; Tussupkaliyev, A.B.T.; Bermagambetova, S.K.; Zhumagulova, S.S.; Sarsembayeva, L.K.; Dossimbetova, M.B.; Daribay, Z.Z. Effect of homocysteine on pregnancy: A systematic review. Chem. Biol. Interact. 2018, 293, 70–76. [Google Scholar] [CrossRef]
- Chen, H.; Yang, X.; Lu, M. Methylenetetrahydrofolate reductase gene polymorphisms and recurrent pregnancy loss in China: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2015, 293, 283–290. [Google Scholar] [CrossRef]
- Dai, C.; Fei, Y.; Li, J.; Shi, Y.; Yang, X. A Novel Review of Homocysteine and Pregnancy Complications. BioMed Res. Int. 2021, 2021, 6652231. [Google Scholar] [CrossRef]
- Santos, T.D.S.; Ieque, A.L.; de Carvalho, H.C.; Sell, A.M.; Lonardoni, M.V.C.; Demarchi, I.G.; de Lima Neto, Q.A.; Teixeira, J.J.V. Antiphospholipid syndrome and recurrent miscarriage: A systematic review and meta-analysis. J. Reprod. Immunol. 2017, 123, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Ornoy, A.; Yacobi, S.; Matalon, S.T.; Blank, M.; Blumenfeld, Z.; Miller, R.K.; Shoenfeld, Y. The effects of antiphospholipid antibodies obtained from women with SLE/APS and associated pregnancy loss on rat embryos and placental explants in culture. Lupus 2003, 12, 573–578. [Google Scholar] [CrossRef]
- Ali, S.; Majid, S.; Ali, N.; Taing, S.; El-Serehy, H.A.; Al-Misned, F.A. Evaluation of etiology and pregnancy outcome in recurrent miscarriage patients. Saudi J. Biol. Sci. 2020, 27, 2809–2817. [Google Scholar] [CrossRef] [PubMed]
- Vomstein, K.; Feil, K.; Strobel, L.; Aulitzky, A.; Hofer-Tollinger, S.; Kuon, R.-J.; Toth, B. Immunological Risk Factors in Recurrent Pregnancy Loss: Guidelines Versus Current State of the Art. J. Clin. Med. 2021, 10, 869. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Chen, D.; Duan, X.; Li, L.; Tang, Y.; Peng, B. The association between antiphospholipid antibodies and late fetal loss: A systematic review and meta-analysis. Acta Obstet. Gynecol. Scand. 2019, 98, 1523–1533. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Shen, X.; Zhou, C.; Wang, M.; Liu, Y.; Zhou, L. Prevention of recurrent miscarriage in women with antiphospholipid syndrome: A systematic review and network meta-analysis. Lupus 2020, 30, 70–79. [Google Scholar] [CrossRef]
- Meuleman, T.; Haasnoot, G.W.; van Lith, J.M.M.; Verduijn, W.; Bloemenkamp, K.W.M.; Claas, F.H.J. Paternal HLA-C is a risk factor in unexplained recurrent miscarriage. Am. J. Reprod. Immunol. 2017, 79, e12797. [Google Scholar] [CrossRef]
- Comba, C.; Bastu, E.; Dural, O.; Yasa, C.; Keskin, G.; Ozsurmeli, M.; Buyru, F.; Serdaroglu, H. Role of inflammatory mediators in patients with recurrent pregnancy loss. Fertil. Steril. 2015, 104, 1467–1474.e1. [Google Scholar] [CrossRef]
- Li, D.; Zheng, L.; Zhao, D.; Xu, Y.; Wang, Y. The Role of Immune Cells in Recurrent Spontaneous Abortion. Reprod. Sci. 2021, 28, 3303–3315. [Google Scholar] [CrossRef] [PubMed]
- Caroline, M.M.; Milner, C.M.; Campbell, R.D. Genetic organization of the human MHC class III region. Front. Biosci. 2001, 6, d914–d926. [Google Scholar] [CrossRef] [Green Version]
- Kuroshli, Z.; Gourabi, H.; Bazrgar, M.; Sanati, M.; Zamani Esteki, M. The Relationship between HLA-G Gene Poly-morphisms and Repeated Implantation Failure in Infertile Couples Undergoing Assisted Reproductive Technique. Iran. J. Allergy Asthma Immunol. 2015, 14, 535–542. [Google Scholar] [PubMed]
- Fan, W.; Huang, Z.; Li, S.; Xiao, Z. The HLA-G 14-bp polymorphism and recurrent implantation failure: A meta-analysis. J. Assist. Reprod. Genet. 2017, 34, 1559–1565. [Google Scholar] [CrossRef] [PubMed]
- Prins, J.R.; Kieffer, T.E.; Scherjon, S.A. Immunomodulators to treat recurrent miscarriage. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 181, 334–337. [Google Scholar] [CrossRef]
- Robertson, S.A.; Jin, M.; Yu, D.; Moldenhauer, L.M.; Davies, M.J.; Hull, M.L.; Norman, R.J. Corticosteroid therapy in assisted reproduction–immune suppression is a faulty premise. Hum. Reprod. 2016, 31, 2164–2173. [Google Scholar] [CrossRef] [Green Version]
- Christiansen, O.B.; Rasmussen, K.L.; Jersild, C.; Grunnet, N. HLA class II alleles confer susceptibility to recurrent fetal losses in Danish women. Tissue Antigens 1994, 44, 225–233. [Google Scholar] [CrossRef]
- Thomsen, C.; Steffensen, R.; Nielsen, H.; Kolte, A.; Krog, M.; Egerup, P.; Larsen, E.; Hviid, T.; Christiansen, O. HLA-DRB1 polymorphism in recurrent pregnancy loss: New evidence for an association to HLA-DRB1*07. J. Reprod. Immunol. 2021, 145, 103308. [Google Scholar] [CrossRef]
- Choo, S.Y. The HLA System: Genetics, Immunology, Clinical Testing, and Clinical Implications. Yonsei Med. J. 2007, 48, 11–23. [Google Scholar] [CrossRef] [Green Version]
- Chighizola, C.B.; Lonati, P.A.; Trespidi, L.; Meroni, P.L.; Tedesco, F. The Complement System in the Pathophysiology of Pregnancy and in Systemic Autoimmune Rheumatic Diseases During Pregnancy. Front. Immunol. 2020, 11, 2084. [Google Scholar] [CrossRef]
- Takakuwa, K.; Hataya, I.; Arakawa, M.; Kikuchi, A.; Higashino, M.; Yasuda, M.; Kurabayashi, T.; Tanaka, K. Possible Susceptibility of the HLA-DPB1*0402 and HLA-DPB1*04 Alleles to Unexplained Recurrent Abortion: Analysis by means of Polymerase Chain Reaction-Restricted Fragment Length Polymorphism Method. Am. J. Reprod. Immunol. 1999, 42, 233–239. [Google Scholar] [CrossRef]
- Kolte, A.M.; Steffensen, R.; Nielsen, H.S.; Hviid, T.V.; Christiansen, O.B. Study of the structure and impact of human leukocyte antigen (HLA)-G-A, HLA-G-B, and HLA-G-DRB1 haplotypes in families with recurrent miscarriage. Hum. Immunol. 2010, 71, 482–488. [Google Scholar] [CrossRef]
- Aruna, M.; Nagaraja, T.; Bhaskar, S.A.; Tarakeswari, S.; Reddy, A.G.; Thangaraj, K.; Singh, L.; Reddy, B.M. Novel alleles of HLA-DQ and -DR loci show association with recurrent miscarriages among South Indian women. Hum. Reprod. 2011, 26, 765–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruse, C.; Steffensen, R.; Varming, K.; Christiansen, O. A study of HLA-DR and -DQ alleles in 588 patients and 562 controls confirms that HLA-DRB1*03 is associated with recurrent miscarriage. Hum. Reprod. 2004, 19, 1215–1221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tafti, F.D.; Zare, F.; Miresmaeili, S.M.; Fesahat, F. Evaluating Vitamin D and foxp3 mRNA levels in women with recurrent spontaneous abortion. JBRA Assist. Reprod. 2021, 26, 232–236. [Google Scholar] [CrossRef]
- Bahia, W.; Zitouni, H.; Kanabekova, P.; Bauyrzhanova, Z.; Shaimardanova, M.; Finan, R.R.; Aimagambetova, G.; Almawi, W.Y. Human forkhead box protein 3 gene variants associated with altered susceptibility to idiopathic recurrent pregnancy loss: A retrospective case-control study. Am. J. Reprod. Immunol. 2022, 88, e13551. [Google Scholar] [CrossRef]
- Hu, X.; Wang, Y.; Mor, G.; Liao, A. Forkhead box P3 is selectively expressed in human trophoblasts and decreased in recurrent pregnancy loss. Placenta 2019, 81, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. (Ed.) Vitamin D: Physiology, Molecular Biology, and Clinical Applications; Springer Science & Business Media: Totowa, NJ, USA, 2010. [Google Scholar]
- Sharif, K.; Sharif, Y.; Watad, A.; Yavne, Y.; Lichtbroun, B.; Bragazzi, N.L.; Amital, H.; Shoenfeld, Y. Vitamin D, autoimmunity and recurrent pregnancy loss: More than an association. Am. J. Reprod. Immunol. 2018, 80, e12991. [Google Scholar] [CrossRef]
- Flood-Nichols, S.K.; Tinnemore, D.; Huang, R.R.; Napolitano, P.G.; Ippolito, D.L. Vitamin D Deficiency in Early Pregnancy. PLoS ONE 2015, 10, e0123763. [Google Scholar] [CrossRef]
- Hollis, W.; Wagner, C.L. Vitamin D supplementation during pregnancy: Improvements in birth outcomes and complications through direct genomic alteration. Mol. Cell. Endocrinol. 2017, 453, 113–130. [Google Scholar] [CrossRef]
- Tamblyn, J.A.; Pilarski, N.S.; Markland, A.D.; Marson, E.J.; Devall, A.; Hewison, M.; Morris, R.K.; Coomarasamy, A. Vitamin D and miscarriage: A systematic review and meta-analysis. Fertil. Steril. 2022, 118, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, C.; Wang, Y.; Zhao, J.; Li, H.; Shen, Q.; Wang, X.; Ni, M.; Ouyang, F.; Vinturache, A.; et al. Relationship of maternal obesity and vitamin D concentrations with fetal growth in early pregnancy. Eur. J. Nutr. 2021, 61, 915–924. [Google Scholar] [CrossRef] [PubMed]
- Von Websky, K.; Hasan, A.A.; Reichetzeder, C.; Tsuprykov, O.; Hocher, B. Impact of vitamin D on pregnancy-related disorders and on offspring outcome. J. Steroid Biochem. Mol. Biol. 2018, 180, 51–64. [Google Scholar] [CrossRef]
- Zhao, H.; Wei, X.; Yang, X. A novel update on vitamin D in recurrent pregnancy loss (Review). Mol. Med. Rep. 2021, 23, 382. [Google Scholar] [CrossRef] [PubMed]
- Javorski, N.; Lima, C.; Silva, L.; Crovella, S.; de Azêvedo Silva, J. Vitamin D receptor (VDR) polymorphisms are associated to spontaneous preterm birth and maternal aspects. Gene 2018, 642, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Krpina, M.G.; Barišić, A.; Peterlin, A.; Tul, N.; Ostojić, S.; Peterlin, B.; Pereza, N. Vitamin D receptor polymorphisms in spontaneous preterm birth: A case-control study. Croat. Med. J. 2020, 61, 338–345. [Google Scholar] [CrossRef]
- Li, N.; Wu, H.; Hang, F.; Zhang, Y.; Li, M. Women with recurrent spontaneous abortion have decreased 25(OH) vitamin D and VDR at the fetal-maternal interface. Braz. J. Med. Biol. Res. 2017, 50, e6527. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.; Wang, L.; Yan, C.; Zhang, X.; Hui, L.; Sheng, Q.; Xue, M.; Yu, X. Decreased expression of the vitamin D receptor in women with recurrent pregnancy loss. Arch. Biochem. Biophys. 2016, 606, 128–133. [Google Scholar] [CrossRef]
- Ota, K.; Dambaeva, S.; Han, A.-R.; Beaman, K.; Gilman-Sachs, A.; Kwak-Kim, J. Vitamin D deficiency may be a risk factor for recurrent pregnancy losses by increasing cellular immunity and autoimmunity. Hum. Reprod. 2013, 29, 208–219. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, D.R.; Braga, A.; Braga, J.; Marinho, A. Recurrent pregnancy loss and vitamin D: A review of the literature. Am. J. Reprod. Immunol. 2018, 80, e13022. [Google Scholar] [CrossRef]
- Tavakoli, M.; Salek-Moghaddam, A.; Jeddi-Tehrani, M.; Talebi, S.; Kazemi-Sefat, G.-E.; Vafaei, S.; Mohammadzadeh, A.; Sheikhhassani, S.; Zarnani, A.-H. Comparable vitamin D3 metabolism in the endometrium of patients with recurrent spontaneous abortion and fertile controls. Mol. Reprod. Dev. 2015, 82, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.L.; Ng, B.K.; Wu, L.L.; Cheah, F.C.; Othman, H.; Ismail, N.A.M. Vitamin D deficiency in pregnancy at term: Risk factors and pregnancy outcomes. Horm. Mol. Biol. Clin. Investig. 2017, 31, 20170005. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Huang, Z.; Xiao, L.; Jiang, X.; Chen, D.; Wei, Y. Meta-analysis of the effect of the maternal vitamin D level on the risk of spontaneous pregnancy loss. Int. J. Gynecol. Obstet. 2017, 138, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.; Gallos, I.; Tobias, A.; Tan, B.; Eapen, A.; Coomarasamy, A. Vitamin D and assisted reproductive treatment outcome: A systematic review and meta-analysis. Hum. Reprod. 2017, 33, 65–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Møller, U.K.; Streym, S.; Heickendorff, L.; Mosekilde, L.; Rejnmark, L. Effects of 25OHD concentrations on chances of pregnancy and pregnancy outcomes: A cohort study in healthy Danish women. Eur. J. Clin. Nutr. 2012, 66, 862–868. [Google Scholar] [CrossRef]
- Griebel, C.P.; Halvorsen, J.; Golemon, T.B.; Day, A.A. Management of Spontaneous Abortion. Am. Fam. Physician 2005, 72, 1243–1250. [Google Scholar]
- Jírová, J.; Michalová, Z.; Beranek, L.; Kotrbová, K.; Zölzer, F. Incidence of spontaneous abortions and congenital anomalies in the vicinity of a uranium processing plant. Cent. Eur. J. Public Health 2020, 28, 44–47. [Google Scholar] [CrossRef]
- Kaur, M.; Sharma, P.; Kaur, R.; Khetarpal, P. Increased incidence of spontaneous abortions on exposure to cadmium and lead: A systematic review and meta-analysis. Gynecol. Endocrinol. 2021, 38, 16–21. [Google Scholar] [CrossRef]
- Ng, K.Y.B.; Cherian, G.; Kermack, A.J.; Bailey, S.; Macklon, N.; Sunkara, S.K.; Cheong, Y. Systematic review and meta-analysis of female lifestyle factors and risk of recurrent pregnancy loss. Sci. Rep. 2021, 11, 7081. [Google Scholar] [CrossRef]
- George, L.; Granath, F.; Johansson, A.L.V.; Olander, B.; Cnattingius, S. Risks of repeated miscarriage. Paediatr. Périnat. Epidemiol. 2006, 20, 119–126. [Google Scholar] [CrossRef]
- Sansone, M.; Zaami, S.; Cetta, L.; Costanzi, F.; Signore, F. Ovotoxicity of smoking and impact on AMH levels: A pilot study. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 5255–5260. [Google Scholar] [CrossRef] [PubMed]
- Stefanidou, E.M.; Caramellino, L.; Patriarca, A.; Menato, G. Maternal caffeine consumption and sine causa recurrent miscarriage. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 158, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Maconochie, N.; Doyle, P.; Prior, S.; Simmons, R. Risk factors for first trimester miscarriage-results from a UK-population-based case-control study. BJOG Int. J. Obstet. Gynaecol. 2007, 114, 170–186. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Newell-Price, J.; Jones, G.; Ledger, W.; Li, T. Relationship between psychological stress and recurrent miscarriage. Reprod. Biomed. Online 2012, 25, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Bashiri, A.; Giliutin, M.; Ziedenberg, H.; Plakht, Y.; Baumfeld, Y. A proposed prognostic prediction tool for a live birth among women with recurrent pregnancy loss. J. Matern. Neonatal Med. 2020, 35, 3736–3742. [Google Scholar] [CrossRef]
- Dai, Y.-F.; Lin, L.-Z.; Lin, N.; He, D.-Q.; Guo, D.-H.; Xue, H.-L.; Li, Y.; Xie, X.; Xu, L.-P.; He, S.-Q. APA scoring system: A novel predictive model based on risk factors of pregnancy loss for recurrent spontaneous abortion patients. J. Obstet. Gynaecol. 2022, 42, 2069–2074. [Google Scholar] [CrossRef]
- Terzic, M.; Aimagambetova, G.; Norton, M.; Della Corte, L.; Marín-Buck, A.; Lisón, J.F.; Amer-Cuenca, J.J.; Zito, G.; Garzon, S.; Caruso, S.; et al. Scoring systems for the evaluation of adnexal masses nature: Current knowledge and clinical applications. J. Obstet. Gynaecol. 2020, 41, 340–347. [Google Scholar] [CrossRef]
- Terzic, M.M.; Aimagambetova, G.; Terzic, S.; Norton, M.; Bapayeva, G.; Garzon, S. Current role of Pipelle endometrial sampling in early diagnosis of endometrial cancer. Transl. Cancer Res. 2020, 9, 7716–7724. [Google Scholar] [CrossRef]
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins. Obstetrics Practice Bulletin No. 118: Antiphospholipid Syndrome. Obstet. Gynecol. 2011, 117, 192–199. [Google Scholar] [CrossRef]
- Mekinian, A.; Cohen, J.; Alijotas-Reig, J.; Carbillon, L.; Nicaise-Roland, P.; Kayem, G.; Daraï, E.; Fain, O.; Bornes, M. Unexplained Recurrent Miscarriage and Recurrent Implantation Failure: Is There a Place for Immunomodulation? Am. J. Reprod. Immunol. 2016, 76, 8–28. [Google Scholar] [CrossRef]
- Williams, S.; Heuberger, R. Outcomes of Vitamin D Supplementation in Adults Who are Deficient and Critically Ill: A Review of the Literature. Am. J. Ther. 2016, 23, e1890–e1902. [Google Scholar] [CrossRef]
- Herrmann, M.; Farrell, C.-J.L.; Pusceddu, I.; Fabregat-Cabello, N.; Cavalier, E. Assessment of vitamin D status—A changing landscape. Clin. Chem. Lab. Med. 2016, 55, 3–26. [Google Scholar] [CrossRef] [Green Version]
- Alves, C.; Rapp, A. Spontaneous Abortion. In StatPearls; StatPearls Publishing: Tampa, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK560521/ (accessed on 1 February 2023).
- Kim, C.; Barnard, S.; Neilson, J.P.; Hickey, M.; Vazquez, J.C.; Dou, L. Medical treatments for incomplete miscarriage. Cochrane Database Syst. Rev. 2017, 1, CD007223. [Google Scholar] [CrossRef] [Green Version]
- Rikken, J.F.W.; Kowalik, C.R.; Emanuel, M.H.; Bongers, M.Y.; Spinder, T.; Jansen, F.W.; Mulders, A.G.M.G.J.; Padmehr, R.; Clark, T.J.; A van Vliet, H.; et al. Septum resection versus expectant management in women with a septate uterus: An international multicentre open-label randomized controlled trial. Hum. Reprod. 2021, 36, 1260–1267. [Google Scholar] [CrossRef]
- Homer, H.A. Modern management of recurrent miscarriage. Aust. N. Z. J. Obstet. Gynaecol. 2018, 59, 36–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yerezhepbayeva, M.; Terzic, M.; Aimagambetova, G.; Crape, B. Comparison of two invasive non-surgical treatment options for uterine myomas: Uterine artery embolization and magnetic resonance guided high intensity focused ultrasound—Systematic review. BMC Women’s Health 2022, 22, 55. [Google Scholar] [CrossRef] [PubMed]
- Haas, D.M.; Hathaway, T.J.; Ramsey, P.S. Progestogen for preventing miscarriage in women with recurrent miscarriage of unclear etiology. Cochrane Database Syst. Rev. 2019, 2019, CD003511. [Google Scholar] [CrossRef] [PubMed]
- Devall, A.J.; Papadopoulou, A.; Podesek, M.; Haas, D.M.; Price, M.J.; Coomarasamy, A.; Gallos, I.D. Progestogens for preventing miscarriage: A network meta-analysis. Cochrane Database Syst. Rev. 2021, 2021, CD013792. [Google Scholar] [CrossRef]
- Arab, H.; Alharbi, A.J.; Oraif, A.; Sagr, E.; Al Madani, H.; Abduljabbar, H.; Bajouh, O.S.; Faden, Y.; Sabr, Y. The Role of Progestogens In Threatened And Idiopathic Recurrent Miscarriage. Int. J. Women’s Health 2019, 11, 589–596. [Google Scholar] [CrossRef] [Green Version]
- Bashiri, A.; Galperin, G.; Zeadna, A.; Baumfeld, Y.; Wainstock, T. Increased Live Birth Rate with Dydrogesterone among Patients with Recurrent Pregnancy Loss Regardless of Other Treatments. J. Clin. Med. 2023, 12, 1967. [Google Scholar] [CrossRef] [PubMed]
- Tetruashvili, N.; Domar, A.; Bashiri, A. Prevention of Pregnancy Loss: Combining Progestogen Treatment and Psychological Support. J. Clin. Med. 2023, 12, 1827. [Google Scholar] [CrossRef]
- Kobayashi, T.; Sugiura, K.; Ojima, T.; Hirai, K.; Morishita, E. Peripartum management of hereditary thrombophilia: Results of primary surveillance in Japan. Int. J. Hematol. 2022, 116, 364–371. [Google Scholar] [CrossRef]
- Jacobson, B.; Rambiritch, V.; Paek, D.; Sayre, T.; Naidoo, P.; Shan, J.; Leisegang, R. Safety and Efficacy of Enoxaparin in Pregnancy: A Systematic Review and Meta-Analysis. Adv. Ther. 2019, 37, 27–40. [Google Scholar] [CrossRef] [Green Version]
- De Jong, P.G.; Kaandorp, S.; Di Nisio, M.; Goddijn, M.; Middeldorp, S. Aspirin and/or heparin for women with unexplained recurrent miscarriage with or without inherited thrombophilia. Cochrane Database Syst. Rev. 2014, 2014, CD004734. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.M.; Khiary, M.; Sherif, L.S.; Hassan, M.; Ragab, A.; Abdelall, I. Low-molecular weight heparin in patients with recurrent early miscarriages of unknown aetiology. J. Obstet. Gynaecol. 2008, 28, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Clark, P.; Walker, I.D.; Langhorne, P.; Crichton, L.; Thomson, A.; Greaves, M.; Whyte, S.; Greer, I.A.; Scottish Pregnancy Intervention Study (SPIN) Collaborators. SPIN (Scottish Pregnancy Intervention) study: A multicenter, randomized controlled trial of low-molecular-weight heparin and low-dose aspirin in women with recurrent miscarriage. Blood 2010, 115, 4162–4167. [Google Scholar] [CrossRef] [Green Version]
- Martinelli, I.; Ruggenenti, P.; Cetin, I.; Pardi, G.; Perna, A.; Vergani, P.; Acaia, B.; Facchinetti, F.; La Sala, G.B.; Bozzo, M.; et al. Heparin in pregnant women with previous placenta-mediated pregnancy complications: A prospective, randomized, multicenter, controlled clinical trial. Blood 2012, 119, 3269–3275. [Google Scholar] [CrossRef] [PubMed]
- Giancotti, A.; La Torre, R.; Spagnuolo, A.; D’Ambrosio, V.; Cerekja, A.; Piazze, J.; Chistolini, A. Efficacy of three different antithrombotic regimens on pregnancy outcome in pregnant women affected by recurrent pregnancy loss. J. Matern. Neonatal Med. 2011, 25, 1191–1194. [Google Scholar] [CrossRef]
- Sammaritano, L.R. Antiphospholipid syndrome. Best Pract. Res. Clin. Rheumatol. 2019, 34, 101463. [Google Scholar] [CrossRef]
- Yamada, H.; Deguchi, M.; Saito, S.; Takeshita, T.; Mitsui, M.; Saito, T.; Nagamatsu, T.; Takakuwa, K.; Nakatsuka, M.; Yoneda, S.; et al. Intravenous immunoglobulin treatment in women with four or more recurrent pregnancy losses: A double-blind, randomised, placebo-controlled trial. Eclinicalmedicine 2022, 50, 101527. [Google Scholar] [CrossRef]
- Serapinas, D.; Boreikaite, E.; Bartkeviciute, A.; Bandzeviciene, R.; Silkunas, M.; Bartkeviciene, D. The importance of folate, vitamins B6 and B12 for the lowering of homocysteine concentrations for patients with recurrent pregnancy loss and MTHFR mutations. Reprod. Toxicol. 2017, 72, 159–163. [Google Scholar] [CrossRef]
- Duckitt, K.; Qureshi, A. Recurrent miscarriage. BMJ Clin. Evid. 2011, 2011, 1409. [Google Scholar]
- Ata, B.; Tan, S.L.; Shehata, F. A systematic review of intravenous immunoglobulin for treatment of unexplained recurrent mis-carriage. Fertil. Steril. 2011, 95, 1080–1085. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Z.M.; Aal, R.M.A.; El Biely, M.M. Role of 1,25-dihydroxyvitamin D (vitamin D3) as immunomodulator in recurrent missed miscarriage. Middle East Fertil. Soc. J. 2013, 18, 171–176. [Google Scholar] [CrossRef]
- Hollis, B.W.; Johnson, D.; Hulsey, T.C.; Ebeling, M.; Wagner, C.L. Vitamin D supplementation during pregnancy: Double-blind, randomized clinical trial of safety and effectiveness. J. Bone Min. Res. 2011, 26, 2341–2357. [Google Scholar] [CrossRef] [Green Version]
- Rafiee, M.; Gharagozloo, M.; Ghahiri, A.; Mehrabian, F.; Maracy, M.R.; Kouhpayeh, S.; Pieper, I.L.; Rezaei, A. Al-tered Th17/Treg Ratio in Recurrent Miscarriage after Treatment with Paternal Lymphocytes and Vitamin D3: A Double-Blind Placebo-Controlled Study. Iran. J. Immunol. 2015, 12, 252–262. [Google Scholar]
- Samimi, M.; Foroozanfard, F.; Amini, F.; Sehat, M. Effect of vitamin D supplemen-tation on unexplained recurrent spontaneous abortion: A double-blind ran-domized controlled trial. Glob. J. Health Sci. 2016, 9, 95. [Google Scholar] [CrossRef]
- Choudhry, U.K. Traditional Practices of Women from India: Pregnancy, Childbirth, and Newborn Care. J. Obstet. Gynecol. Neonatal Nurs. 1997, 26, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Schneiderman, G.; Barrera, M. Family Traditions and Generations. Fam. Community Health 2009, 32, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Elmusharaf, K.; Byrne, E.; O’donovan, D. Social and traditional practices and their implications for family planning: A participatory ethnographic study in Renk, South Sudan. Reprod. Health 2017, 14, 10. [Google Scholar] [CrossRef] [Green Version]
- Fata, S.; Tokat, M.; Bagardi, N.; Yilmaz, B. The traditional practices used by couples with fertility problems, affecting factors, expected benefits, and learning paths: The Turkey Sample. Niger. J. Clin. Pract. 2019, 22, 806–811. [Google Scholar] [CrossRef]
- Issakhanova, A.; Issanov, A.; Ukybassova, T.; Kaldygulova, L.; Marat, A.; Imankulova, B.; Kamzayeva, N.; Almawi, W.Y.; Aimagambetova, G. Depression, Anxiety, and Stress in Kazakhstani Women with Recurrent Pregnancy Loss: A Case–Control Study. J. Clin. Med. 2023, 12, 658. [Google Scholar] [CrossRef]
- Serrano, F.; Lima, M.L. Recurrent miscarriage: Psychological and relational consequences for couples. Psychol. Psychother. Theory Res. Pract. 2006, 79, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Craig, M.; Tata, P.; Regan, L. Psychiatric morbidity among patients with recurrent miscarriage. J. Psychosom. Obstet. Gynecol. 2002, 23, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Catherino, W.H. Stress relief to augment fertility: The pressure mounts. Fertil. Steril. 2011, 95, 2462–2463. [Google Scholar] [CrossRef] [Green Version]
- Tavoli, Z.; Mohammadi, M.; Tavoli, A.; Moini, A.; Effatpanah, M.; Khedmat, L.; Montazeri, A. Quality of life and psychological distress in women with recurrent miscarriage: A comparative study. Health Qual. Life Outcomes 2018, 16, 150. [Google Scholar] [CrossRef] [Green Version]
- He, L.; Wang, T.; Xu, H.; Chen, C.; Liu, Z.; Kang, X.; Zhao, A. Prevalence of depression and anxiety in women with recurrent pregnancy loss and the associated risk factors. Arch. Gynecol. Obstet. 2019, 300, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Meng, Z.; Pei, J.; Qian, L.; Mao, B.; Li, Y.; Li, J.; Dai, Z.; Cao, J.; Zhang, C.; et al. Anxiety and depression are risk factors for recurrent pregnancy loss: A nested case–control study. Health Qual. Life Outcomes 2021, 19, 78. [Google Scholar] [CrossRef]
- Hedegaard, S.; Landersoe, S.K.; Olsen, L.R.; Krog, M.C.; Kolte, A.M.; Nielsen, H.S. Stress and depression among women and men who have experienced recurrent pregnancy loss: Focusing on both sexes. Reprod. Biomed. Online 2021, 42, 1172–1180. [Google Scholar] [CrossRef]
- Kolte, A.; Olsen, L.; Mikkelsen, E.M.; Christiansen, O.; Nielsen, H.S. Depression and emotional stress is highly prevalent among women with recurrent pregnancy loss. Hum. Reprod. 2015, 30, 777–782. [Google Scholar] [CrossRef]
- Campillo, I.S.L.; Meaney, S.; McNamara, K.; O’Donoghue, K. Psychological and support interventions to reduce levels of stress, anxiety or depression on women’s subsequent pregnancy with a history of miscarriage: An empty systematic review. BMJ Open 2017, 7, e017802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jensen, K.H.K.; Krog, M.C.; Koert, E.; Hedegaard, S.; Chonovitsch, M.; Schmidt, L.; Kolte, A.M.; Nielsen, H.S. Meditation and mindfulness reduce perceived stress in women with recurrent pregnancy loss: A randomized controlled trial. Reprod. Biomed. Online 2021, 43, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Koert, E.; Malling, G.M.H.; Sylvest, R.; Krog, M.C.; Kolte, A.M.; Schmidt, L.; Nielsen, H.S. Recurrent pregnancy loss: Couples’ perspectives on their need for treatment, support and follow up. Hum. Reprod. 2018, 34, 291–296. [Google Scholar] [CrossRef] [PubMed]
- A Murphy, F.; Lipp, A.; Powles, D.L. Follow-up for improving psychological well being for women after a miscarriage. Cochrane Database Syst. Rev. 2012, 3, CD008679. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Criteria | Guidelines (Year of Publication) | ||||
|---|---|---|---|---|---|
| WHO (1977) | RCOG (2011) | ASRM (2012/2013) | ESHRE (2017/2022) | DGGG/ÖGGG/SGGG (2018) | |
| Number of RPLs (n) | 3 | 3 | 2 | 2 | 3 |
| Sequence of loss | Not specified | Consecutive | Consecutive | Consecutive and non-consecutive | Consecutive and non-consecutive |
| Type of pregnancy | All pregnancy losses, types are not specified | All pregnancy losses, types are not specified | Clinical pregnancies | Non-visualized pregnancy losses (biochemical pregnancy losses and/or resolved and treated pregnancies of unknown location), clinical pregnancies | All pregnancy losses, types are not specified |
| Evidence of loss | Not specified | Not specified | US or histopathologic examination | Serum or urine β–hCG; US | Not specified |
| Gestational age at pregnancy loss (weeks) | 20 (22) | 24 | The first trimester of pregnancy, gestational weeks not specified | 24 | 24 |
| Fetal weight (grams) | <500 | Not specified | Not specified | Not specified | <500 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turesheva, A.; Aimagambetova, G.; Ukybassova, T.; Marat, A.; Kanabekova, P.; Kaldygulova, L.; Amanzholkyzy, A.; Ryzhkova, S.; Nogay, A.; Khamidullina, Z.; et al. Recurrent Pregnancy Loss Etiology, Risk Factors, Diagnosis, and Management. Fresh Look into a Full Box. J. Clin. Med. 2023, 12, 4074. https://doi.org/10.3390/jcm12124074
Turesheva A, Aimagambetova G, Ukybassova T, Marat A, Kanabekova P, Kaldygulova L, Amanzholkyzy A, Ryzhkova S, Nogay A, Khamidullina Z, et al. Recurrent Pregnancy Loss Etiology, Risk Factors, Diagnosis, and Management. Fresh Look into a Full Box. Journal of Clinical Medicine. 2023; 12(12):4074. https://doi.org/10.3390/jcm12124074
Chicago/Turabian StyleTuresheva, Akbayan, Gulzhanat Aimagambetova, Talshyn Ukybassova, Aizada Marat, Perizat Kanabekova, Lyazzat Kaldygulova, Ainur Amanzholkyzy, Svetlana Ryzhkova, Anastassiya Nogay, Zaituna Khamidullina, and et al. 2023. "Recurrent Pregnancy Loss Etiology, Risk Factors, Diagnosis, and Management. Fresh Look into a Full Box" Journal of Clinical Medicine 12, no. 12: 4074. https://doi.org/10.3390/jcm12124074
APA StyleTuresheva, A., Aimagambetova, G., Ukybassova, T., Marat, A., Kanabekova, P., Kaldygulova, L., Amanzholkyzy, A., Ryzhkova, S., Nogay, A., Khamidullina, Z., Ilmaliyeva, A., Almawi, W. Y., & Atageldiyeva, K. (2023). Recurrent Pregnancy Loss Etiology, Risk Factors, Diagnosis, and Management. Fresh Look into a Full Box. Journal of Clinical Medicine, 12(12), 4074. https://doi.org/10.3390/jcm12124074








