Abstract
Background: Data are scarce regarding the incidence, reasons, potential risk factors, and long-term outcomes of reintervention after thoracic endovascular aortic repair (TEVAR) in patients with uncomplicated type B aortic dissection (TBAD). Methods: Between January 2010 and December 2020, 238 patients with uncomplicated TBAD who received TEVAR were analyzed retrospectively. The clinical baseline data, aorta anatomy, dissection characteristics, and details of the TEVAR procedure were evaluated and compared. A competing-risk regression model was used to estimate the cumulative incidences of reintervention. The multivariate Cox model was used to identify the independent risk factors. Results: The mean follow-up time was 68.6 months. A total of 27 (11.3%) cases of reintervention were observed. The competing-risk analyses showed that the 1-, 3-, and 5-year cumulative incidences of reintervention were 5.07%, 7.08%, and 14.0%, respectively. Reasons for reintervention included endoleak (25.9%), aneurysmal dilation (22.2%), retrograde type A aortic dissection (18.5%), distal stent-graft-induced new entry and false lumen expansion (18.5%), and dissection progression and/or malperfusion (14.8%). Multivariable Cox analysis demonstrated that a larger initial maximal aortic diameter (Hazard ratio [HR], 1.75; 95% Confidence interval [CI], 1.13–2.69, p = 0.011) and increased proximal landing zone oversizing (HR, 1.07; 95% CI, 1.01–1.47, p = 0.033) were the significant risk factors for reintervention. Long-term survival rates were comparable between patients with or without reintervention (p = 0.915). Conclusions: Reintervention after TEVAR in patients with uncomplicated TBAD is not uncommon. A larger initial maximal aortic diameter and excessive proximal landing zone oversizing are associated with the second intervention. Reintervention does not significantly affect long-term survival.
1. Introduction
Aortic dissection is a catastrophic cardiovascular disease. Thoracic endovascular aortic repair (TEVAR) has altered the management algorithm of type B aortic dissection (TBAD) since its introduction by Dake et al. in 1994 [1]. With lower mortality rates and fewer complications, TEVAR has replaced traditional open surgery as the treatment of choice for patients with complicated TBAD [2]. For uncomplicated TBAD, the preferred treatment is currently under debate. Some guidelines recommend the best medical treatment (BMT) to control blood pressure and slow the heart rate as first-line therapy [3,4]. However, published evidence suggests that TEVAR results in significantly better long-term survival and aortic remodeling than BMT in patients with uncomplicated TBAD [5,6,7,8].
Regrettably, TEVAR is not a once-and-for-all solution. The long-term benefits of TEVAR may be attenuated or even offset by postoperative adverse events, such as endoleak, stroke, retrograde type A aortic dissection (RTAD), stent-graft migration, distal stent-graft-induced new entry (dSINE), and aneurysmal degeneration. These are the biggest obstacles to TEVAR becoming a first-line treatment for uncomplicated TBAD. In most situations, these complications need reintervention to save lives or to achieve better aortic remodeling.
In the literature published to date, some studies have addressed the issue of reintervention after TEVAR [9,10,11,12]. However, the majority of these are based on populations with complicated TBAD, and/or mixed cases of dissection and aneurysm, or involve a short follow-up period. The purpose of this study was to evaluate the incidence, reasons, potential risk factors, and long-term outcomes of reintervention in patients with uncomplicated TBAD.
2. Materials and Methods
2.1. Study Population
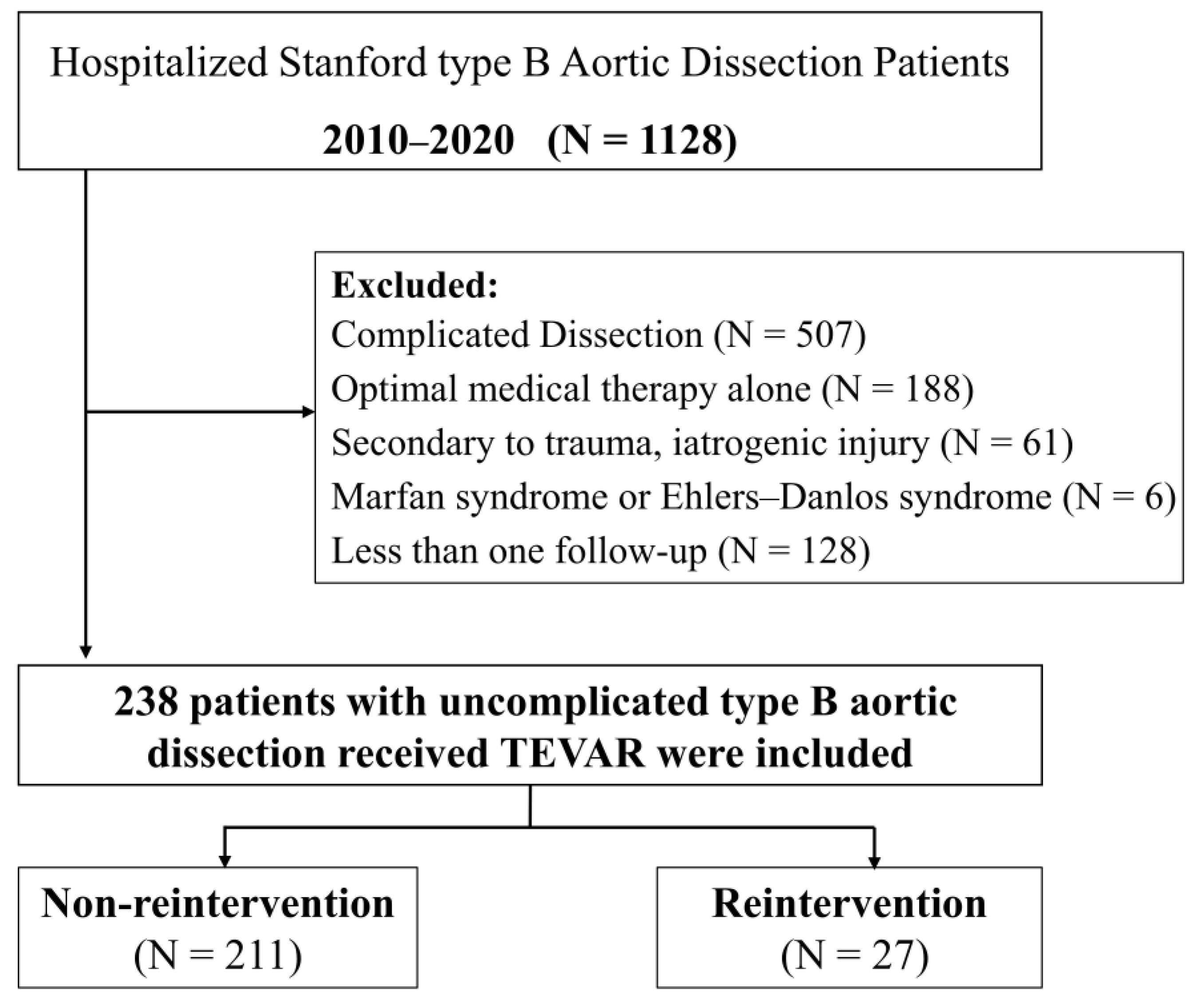
Between January 2010 and December 2020, 1128 patients were diagnosed with TBAD at the Union Hospital of Huazhong University of Science and Technology. The local ethical review board approved the study, and informed consent was waived because of its retrospective nature. Patients with TBAD confirmed by arterial computed tomography angiography (CTA) and who were treated with TEVAR were included. Uncomplicated TBAD was defined as dissection without rupture, malperfusion syndrome, refractory pain, or rapid aortic expansion at onset or during a patient’s presentation at the hospital [13]. Finally, 238 patients diagnosed with uncomplicated TBAD who received TEVAR were enrolled in the study. The flowchart of patient selection is shown in Figure 1.
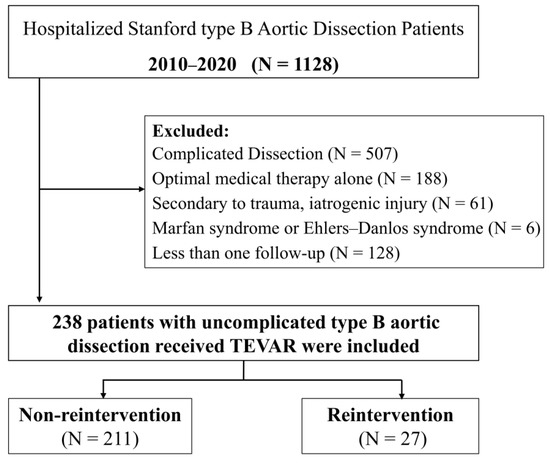
Figure 1.
Flowchart of patient selection.
2.2. TEVAR Procedure
Indications for TEVAR are usually considered to include an initial maximum aortic diameter ≥40 mm, an initial false lumen diameter ≥22 mm, a primary entry tear diameter ≥10 mm, and a patent or partially thrombosed false lumen [4,14,15]. The need for TEVAR and its timing were discussed among the surgical decision-making team in all cases.
Details of the TEVAR procedure were described in a previous study [16]. The procedure was typically performed in a hybrid operating room under local or general anesthesia by experienced endovascular and vascular surgeons, cardiologists, or interventional radiologists. The size of each thoracic stent graft was selected based on the diameter of the normal aorta proximal to the dissection. Proximal oversizing was usually chosen from a range of 1–10%, and the distal diameter was left to the discretion of the surgeon. We do not routinely administer antithrombotic therapy postoperatively.
2.3. Data Collection and Follow-Up
Baseline clinical data and details about TEVAR were collected retrospectively by searching the electronic medical record system. The anatomical features of the aorta were evaluated and measured in preoperative CTA images based on the centerline of flow in the Syngo.via post-processing workstation (Siemens Healthcare, Forchheim, Germany). Postoperative aortic-related events were evaluated using follow-up CTA images, which were taken routinely at 1-, 3-, 6-, and 12-month intervals after the primary TEVAR procedure, and yearly thereafter. Details of reintervention were obtained from patient readmission records. The follow-up period for the present study ended on 30 June 2022.
2.4. Endpoints and Definitions
The outcomes were the incidence, reasons, potential risk factors, and outcomes of reintervention after primary TEVAR during follow-up. Reintervention was defined as a second intervention for the management of complications after the primary TEVAR, not including the second-stage operation [17]. The coverage ratio of the thoracic aorta was defined as the ratio of the length of thoracic aortic stent coverage to the total length of the thoracic aorta. Aneurysmal degeneration was defined as an increase in the total aortic diameter of >5.5 cm or growth of ≥0.5 cm in a 6-month period [18].
2.5. Statistical Analysis
Continuous variables were expressed as the mean ± standard deviation or the median with the interquartile range, whereas categorical variables were presented as the frequency and percentages. Statistical comparisons were made with the χ2 or Fisher exact tests for categorical variables and Student’s t or Mann–Whitney U tests for continuous variables. The Cox proportional hazards model was used to identify risk factors for reintervention. p < 0.1 in the univariate analysis was selected for entry into the multivariable regression analysis. Reinterventions were evaluated by a competing-risk analysis with mortality as the competing event, and between-group differences were assessed using the Fine–Gray test for estimates of cumulative incidences. All statistical tests were two-sided, and the significance level was <0.05. Statistical analyses were performed using R 4.1.2 (R Development Core Team, Vienna, Austria).
3. Results
3.1. Baseline Characteristics
In total, 238 patients with uncomplicated TBAD treated with TEVAR were included in this study; among them, 27 (11.3%) patients received reintervention during the follow-up. The baseline characteristics are listed in Table 1. The mean age was 52.6 ± 10.9 years in the non-intervention group and 54.4 ± 9.1 years in the intervention group (p = 0.397). Hypertensive patients accounted for 65.4% of the total in the non-reintervention group and 66.7% in the reintervention group (p = 0.896).

Table 1.
Baseline covariate distribution.
3.2. Anatomical Features and TEVAR Procedure
Anatomical features of the thoracic aorta and details about the TEVAR procedure are listed in Table 2. The median initial maximal aortic diameter was 3.6 cm in the non-reintervention group and 3.5 cm in the reintervention group (p = 0.142). The mean oversizing at the proximal landing zone was 4.57 ± 4.26% in the non-reintervention group and 6.68 ± 6.20% in the reintervention group (p = 0.067). There were no significant differences in the stent graft length, coverage ratio of thoracic aorta, or proximal landing zone between the two groups.

Table 2.
Anatomical features of the thoracic aorta and details of the TEVAR procedure.
Technical success was achieved in all patients by the primary TEVAR procedure. A total of 269 stent grafts were implanted in the thoracic aortas of 238 patients. Among them, 209 patients (87.8%) received one stent graft, 27 (11.3%) received two stent grafts, and 2 (0.8%) received three stent grafts. Valiant (Medtronic, Inc, Minneapolis, MN, USA) was the most used brand at 46.1% (n = 124), followed by Ankura (Lifetech Scientific, Shenzhen, China) at 24.5% (n = 66).
3.3. In-Hospital and 30-Day Outcomes
In-hospital and 30-day outcomes are reported in Table 3. One patient in the non-reintervention group died because of a sudden stroke and RTAD 22 days after primary TEVAR. The difference in the 30-day mortality rate between the two groups was not statistically significant (p = 1.000). The total numbers of in-hospital and 30-day adverse events were comparable between the two groups (7.6% vs. 18.5%, p = 0.072). However, the incidence of RTAD was significantly higher in the reintervention group than in the non-reintervention group (7.4% vs. 0.5%, p = 0.035). Two patients in the reintervention group developed RTAD, both of whom underwent total arch replacement. Type I endoleaks were the most common adverse events, with a total of eleven type I endoleaks observed during the first 30 days, nine of which were mild and did not require reintervention.

Table 3.
In-hospital and 30-day outcomes.
3.4. Incidence and Reasons
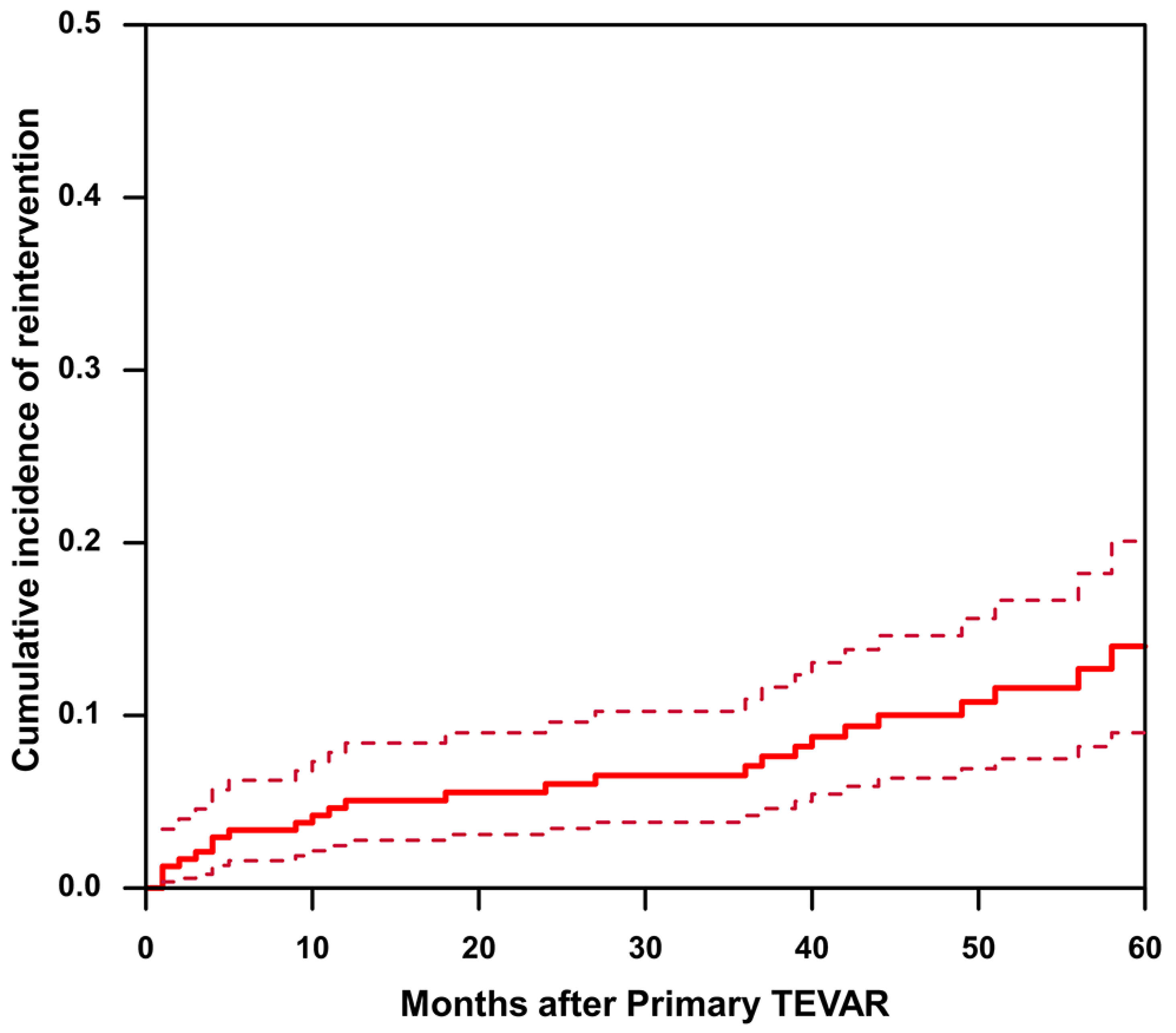
The mean follow-up time was 63.7 ± 25.9 months in the non-reintervention group and 73.4 ± 25.8 months in the reintervention group (p = 0.069). A total of 27 (11.3%) cases of reintervention were observed during the follow-up period. Considering death as a competing risk, cumulative incidence estimates suggest that the 1-, 3-, and 5-year cumulative incidences of reintervention were 5.07%, 7.08%, and 14.0%, respectively (Figure 2). The reasons for reinterventions, as well as their type and timing, are listed in Table 4. Reasons for reintervention included endoleak (25.9%), RTAD (18.5%), dSINE and false lumen expansion (18.5%), aneurysmal dilation (non-dSINE causes) (22.2%), and dissection progression and/or malperfusion (14.8%).
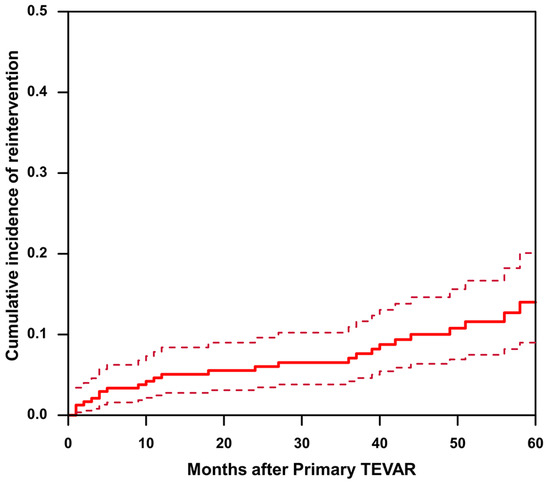
Figure 2.
The cumulative incidence of reintervention after TEVAR in patients with uncomplicated type B aortic dissection. The solid line represents cumulative incidence curve. The dashed line represents the 95% confidence interval of the cumulative incidence.

Table 4.
Indications and descriptions of reinterventions.
3.5. Risk Factors and Outcome of Reintervention
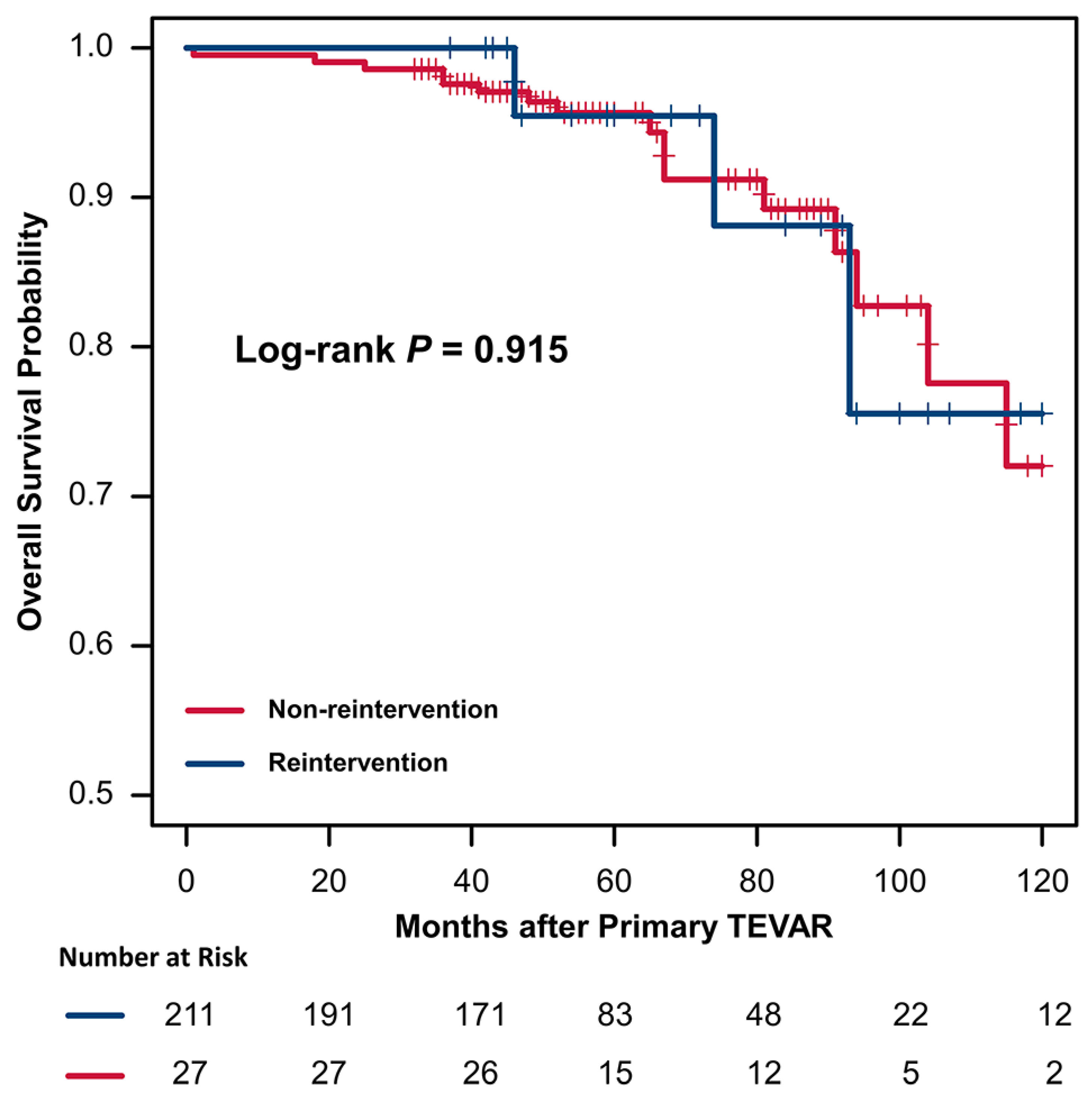
The results of the Cox hazard regression analyses are summarized in Table 5. Univariate analysis and multivariable Cox hazard regression analysis demonstrated that a larger maximal aortic diameter (hazard ratio [HR], 1.75; 95% confidence interval [CI], 1.13–2.69, p = 0.011) and increased proximal landing zone oversizing (HR, 1.07; 95% CI, 1.01–1.47, p = 0.033) were the significant risk factors for reintervention. Kaplan–Meier survival analysis showed that 1-, 3-, 5-, and 10-year overall survival rates in the intervention group were 100%, 100%, 95.5%, and 75.5%, respectively, with corresponding figures of 99.5%, 97.6%, 95.7%, and 72.0% for the non-intervention group (Figure 3). A log-rank test showed that the presence or absence of reintervention had no significant effect on overall survival (p = 0.915).

Table 5.
Univariate and multivariate Cox hazard analysis of reintervention.
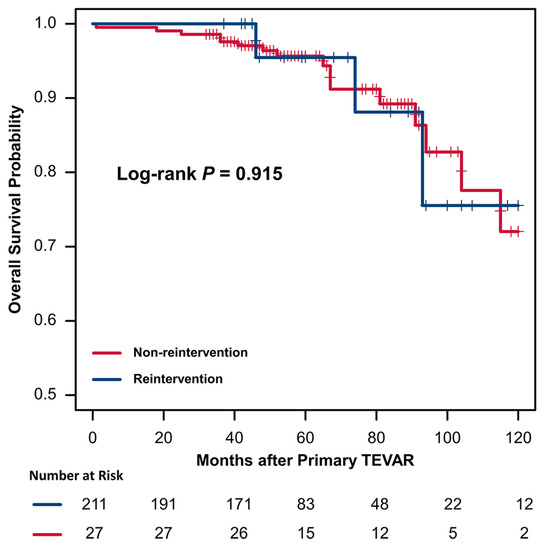
Figure 3.
Kaplan–Meier curves of overall survival in patients with or without reintervention after TEVAR.
4. Discussion
The present study is one of the first to use a large cohort to report the incidence, reasons, risk factors, and outcomes of reintervention after TEVAR in a subset of uncomplicated TBAD cases with an average follow-up of more than 5 years. To objectively assess the incidence, a competing-risk model with all-cause mortality as a competing event was used to determine the cumulative incidence of reintervention. To explore potential risk factors, we considered multiple variables including clinical baseline data, aorta anatomy, dissection characteristics, and details of the TEVAR procedure.
In a previously published meta-analysis of 27 studies involving 2403 patients with aortic dissection, the pooled overall incidence of reintervention was 15.0% during 33.7 months of follow-up [17]. In the present study, 11.3% of patients required reintervention and the 3- and 5-year cumulative incidences of reintervention were 7.08% and 14.0%. These results were lower than the results reported in the above systematic review. A possible explanation is that this study only focused on uncomplicated TBAD, whereas the data reported in the literature mostly concern mixed cases of all dissections and/or aneurysms. Uncomplicated TBAD may be less severe in pathological processes than complicated TBAD and aneurysms.
In this study, the reasons for reintervention in descending order were as follows: Endoleak, aneurysmal dilation (non-dSINE causes), RTAD, dSINE and false lumen expansion, and dissection progression and/or malperfusion. This finding is consistent with the results of a previous study [17]. Interestingly, the incidence of these indications did not differ much but was close. Endoleak, especially type I endoleak, is still the most common reason for reintervention after TEVAR. Nozdrzykowski et al. [19] also reported type I endoleak as the most frequent indication for a secondary procedure (26.8%; n = 15/56). The use of scallop, fenestration, and chimney techniques, as well as steep aortic arches, too-short landing zones, and oversizing (too large or too small), have all been reported as causative factors for type I endoleak [10,11,20]. In the present study, of the six cases of endoleak that underwent reintervention, two cases progressed after 30 days of detection, and four cases were newly developed during follow-up. Timely intervention (e.g., cuff, candy-plug, coil and/or plug embolization, and STABILISE techniques) is necessary when persistent moderate or severe endoleaks are found, and all endoleaks in this study achieved good long-term outcomes after reintervention. Aneurysmal dilation is a chronic process that may be difficult to detail as intrinsic factors of the disease may be responsible. The length of the covered segment seems to play a major role. Some studies have reported that covering the entire thoracic aorta with stents can prevent the occurrence of aneurysmal dilation and promote aortic remodeling [21,22].
RTAD and dSINE share the same pathogenic mechanism, but RTAD is more dangerous and often requires open surgical repair. Although there are many possible reasons for RTAD after TEVAR, including excessive oversizing, proximal bare stents, acute dissection or connective tissue disorders, and inappropriate landing zones [11,23,24,25], excessive oversizing remains the most recognized risk factor in many studies. In the present study, multivariable regression analysis demonstrated that excessive proximal landing zone oversizing is a significant risk factor for reintervention, and this is consistent with the findings of previous studies [10,26]. Excessive oversizing can not only lead to RTAD but may also cause endoleak and dSINE [10,27]. Although the potential relationship between oversizing and reintervention has not been elucidated, the selection of oversizing is important. At present, the optimal oversizing for TBAD remains controversial. In our experience, an oversizing of approximately 5% is appropriate for TEVAR treatment of uncomplicated TBAD. In an integrated data study, Canaud et al. indicated that when oversizing was greater than 9%, the relative risk of RTAD increased by 14% for each percentage-point increase [25]. No in-stent thrombosis or stent folding was observed in this study. This also confirmed that it is difficult to form a thrombus in the thoracic aortic stent after TEVAR, and oversizing within 10% can effectively prevent the occurrence of stent folding [28].
The initial maximal aortic diameter is considered to be a predictor of poor prognosis in patients with uncomplicated TBAD treated with medical therapy alone. Early TEVAR intervention can thus be recommended for these high-risk patients. Interestingly, the present study showed that, even with TEVAR treatment, a larger initial maximal aortic diameter remained a significant risk factor for reintervention. Giles et al. also reported that a larger maximal aortic diameter at presentation was associated with secondary aortic intervention after TEVAR for aortic dissection [26]. Therefore, whether patients with larger initial maximal aortic diameters should decline open surgery or receive TEVAR with lifelong close surveillance is an issue that needs to be explored in future studies.
This study has some limitations. First, it is a single-center retrospective study, and potential selection bias is difficult to avoid in such research. Second, there are no standard guidelines for TEVAR and subsequent secondary interventions in uncomplicated TBAD, so our empirical decisions on TEVAR and reinterventions may have potentially influenced outcomes of uncomplicated TBAD. Third, although we tried to incorporate many features to find potential risk factors for reintervention, the findings reported here may yet represent only the tip of the iceberg.
5. Conclusions
The incidence of reintervention after TEVAR in patients with uncomplicated TBAD is not low. Larger initial maximal aortic diameter and excessive proximal landing zone oversizing were associated with reintervention after TEVAR. Multiple pathological factors contributed to reintervention. However, reintervention did not significantly affect long-term survival.
Author Contributions
Conceptualization, L.C. and D.X.; methodology, D.X.; software, L.C. and S.Z.; validation, L.C., D.X. and S.Z.; formal analysis, L.C.; investigation, D.X.; resources, S.Z.; data curation, D.X.; writing—original draft preparation, L.C. and D.X.; writing—review and editing, L.C. and D.X.; visualization, D.X.; supervision, X.W.; project administration, C.Z. and X.W.; funding acquisition, C.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the National Natural Science Foundation of China (81873919).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and the local ethical review board approved the study, and informed consent was waived for its retrospective nature.
Informed Consent Statement
Each general practitioner obtained the consent of his patients to process his data anonymously.
Data Availability Statement
The data and/or related materials of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dake, M.D.; Miller, D.C.; Semba, C.P.; Mitchell, R.S.; Walker, P.J.; Liddell, R.P. Transluminal placement of endovascular stent-grafts for the treatment of descending thoracic aortic aneurysms. N. Engl. J. Med. 1994, 331, 1729–1734. [Google Scholar] [CrossRef]
- Riambau, V.; Bockler, D.; Brunkwall, J.; Cao, P.; Chiesa, R.; Coppi, G.; Czerny, M.; Fraedrich, G.; Haulon, S.; Jacobs, M.J.; et al. Editor’s choice—Management of descending thoracic aorta diseases: Clinical practice guidelines of the european society for vascular surgery (esvs). Eur. J. Vasc. Endovasc. Surg. 2017, 53, 4–52. [Google Scholar] [CrossRef] [PubMed]
- Erbel, R.; Aboyans, V.; Boileau, C.; Bossone, E.; Di Bartolomeo, R.; Eggebrecht, H.; Evangelista, A.; Falk, V.; Frank, H.; Gaemperli, O.; et al. 2014 esc guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The task force for the diagnosis and treatment of aortic diseases of the european society of cardiology (esc). Eur. Heart J. 2014, 35, 2873–2926. [Google Scholar] [PubMed]
- MacGillivray, T.E.; Gleason, T.G.; Patel, H.J.; Aldea, G.S.; Bavaria, J.E.; Beaver, T.M.; Chen, E.P.; Czerny, M.; Estrera, A.L.; Firestone, S.; et al. The society of thoracic surgeons/american association for thoracic surgery clinical practice guidelines on the management of type b aortic dissection. Ann. Thorac. Surg. 2022, 113, 1073–1092. [Google Scholar] [CrossRef]
- Nienaber, C.A.; Kische, S.; Rousseau, H.; Eggebrecht, H.; Rehders, T.C.; Kundt, G.; Glass, A.; Scheinert, D.; Czerny, M.; Kleinfeldt, T.; et al. Endovascular repair of type b aortic dissection: Long-term results of the randomized investigation of stent grafts in aortic dissection trial. Circ. Cardiovasc. Interv. 2013, 6, 407–416. [Google Scholar] [CrossRef]
- Brunkwall, J.; Kasprzak, P.; Verhoeven, E.; Heijmen, R.; Taylor, P.; Alric, P.; Canaud, L.; Janotta, M.; Raithel, D.; Malina, M.; et al. Endovascular repair of acute uncomplicated aortic type b dissection promotes aortic remodelling: 1-year results of the adsorb trial. Eur. J. Vasc. Endovasc. Surg. 2014, 48, 285–291. [Google Scholar] [CrossRef]
- Qin, Y.L.; Wang, F.; Li, T.X.; Ding, W.; Deng, G.; Xie, B.; Teng, G.J. Endovascular repair compared with medical management of patients with uncomplicated type b acute aortic dissection. J. Am. Coll. Cardiol. 2016, 67, 2835–2842. [Google Scholar] [CrossRef]
- Weissler, E.H.; Osazuwa-Peters, O.L.; Greiner, M.A.; Hardy, N.C.; Kougias, P.; O’Brien, S.M.; Mark, D.B.; Jones, W.S.; Secemsky, E.A.; Vekstein, A.M.; et al. Initial Thoracic Endovascular Aortic Repair vs Medical Therapy for Acute Uncomplicated Type B Aortic Dissection. JAMA Cardiol. 2023, 8, 44–53. [Google Scholar] [CrossRef]
- Dumfarth, J.; Michel, M.; Schmidli, J.; Sodeck, G.; Ehrlich, M.; Grimm, M.; Carrel, T.; Czerny, M. Mechanisms of failure and outcome of secondary surgical interventions after thoracic endovascular aortic repair (tevar). Ann. Thorac. Surg. 2011, 91, 1141–1146. [Google Scholar] [CrossRef] [PubMed]
- Faure, E.M.; Canaud, L.; Agostini, C.; Shaub, R.; Böge, G.; Marty-ané, C.; Alric, P. Reintervention after thoracic endovascular aortic repair of complicated aortic dissection. J. Vasc. Surg. 2014, 59, 327–333. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, J.; Lu, Q.; Zhao, Z.; Bao, J.; Jing, Z. Potential risk factors of re-intervention after endovascular repair for type b aortic dissections. Catheter. Cardiovasc. Interv. 2015, 86, E1–E10. [Google Scholar] [CrossRef] [PubMed]
- Alhussaini, M.; Arnaoutakis, G.J.; Scali, S.T.; Giles, K.A.; Fatima, J.; Back, M.; Arnaoutakis, D.; Jeng, E.I.; Martin, T.D.; Neal, D.; et al. Impact of secondary aortic interventions after thoracic endovascular aortic repair on long-term survival. Ann. Thorac. Surg. 2020, 110, 27–38. [Google Scholar] [CrossRef]
- Trimarchi, S.; Eagle, K.A.; Nienaber, C.A.; Pyeritz, R.E.; Jonker, F.H.; Suzuki, T.; O’Gara, P.T.; Hutchinson, S.J.; Rampoldi, V.; Grassi, V.; et al. Importance of refractory pain and hypertension in acute type b aortic dissection insights from the international registry of acute aortic dissection (irad). Circulation 2010, 122, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.T.; Evangelista, A.; Nienaber, C.A.; Myrmel, T.; Meinhardt, G.; Cooper, J.V.; Smith, D.E.; Suzuki, T.; Fattori, R.; Llovet, A.; et al. Partial thrombosis of the false lumen in patients with acute type b aortic dissection. N. Engl. J. Med. 2007, 357, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Evangelista, A.; Salas, A.; Ribera, A.; Ferreira-González, I.; Cuellar, H.; Pineda, V.; González-Alujas, T.; Bijnens, B.; Permanyer-Miralda, G.; Garcia-Dorado, D. Long-term outcome of aortic dissection with patent false lumen: Predictive role of entry tear size and location. Circulation 2012, 125, 3133–3141. [Google Scholar] [CrossRef] [PubMed]
- Xiang, D.; Kan, X.; Liang, H.; Xiong, B.; Liang, B.; Wang, L.; Zheng, C. Comparison of mid-term outcomes of endovascular repair and medical management in patients with acute uncomplicated type b aortic dissection. J. Thorac. Cardiovasc. Surg. 2021, 162, 26–36.e1. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, Z.; Chen, Y.; Sun, Y.; Bao, J.; Jing, Z.; Zhou, J. Reintervention after endovascular repair for aortic dissection: A systematic review and meta-analysis. J. Thorac. Cardiovasc. Surg. 2016, 152, 1279–1288.e3. [Google Scholar] [CrossRef]
- Schwartz, S.I.; Durham, C.; Clouse, W.D.; Patel, V.I.; Lancaster, R.T.; Cambria, R.P.; Conrad, M.F. Predictors of late aortic intervention in patients with medically treated type b aortic dissection. J. Vasc. Surg. 2018, 67, 78–84. [Google Scholar] [CrossRef]
- Nozdrzykowski, M.; Luehr, M.; Garbade, J.; Schmidt, A.; Leontyev, S.; Misfeld, M.; Mohr, F.-W.; Etz, C.D. Outcomes of secondary procedures after primary thoracic endovascular aortic repair. Eur. J. Cardio-Thorac. 2016, 49, 770–777. [Google Scholar] [CrossRef]
- Marrocco-Trischitta, M.M.; de Beaufort, H.W.; Secchi, F.; van Bakel, T.M.; Ranucci, M.; Van Herwaarden, J.A.; Moll, F.L.; Trimarchi, S. A geometric reappraisal of proximal landing zones for thoracic endovascular aortic repair according to aortic arch types. J. Vasc. Surg. 2017, 65, 1584–1590. [Google Scholar] [CrossRef]
- Lou, X.; Duwayri, Y.M.; Jordan, W.D., Jr.; Chen, E.P.; Veeraswamy, R.K.; Leshnower, B.G. The safety and efficacy of extended tevar in acute type b aortic dissection. Ann. Thorac. Surg. 2020, 110, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Leshnower, B.G. Permissive hypertension and selective cerebrospinal fluid drainage for extended tevar for acute type b dissection: Reply. Ann. Thorac. Surg. 2020, 110, 1435–1436. [Google Scholar] [CrossRef]
- Dong, Z.H.; Fu, W.G.; Wang, Y.Q.; Guo, D.Q.; Xu, X.; Ji, Y.; Chen, B.; Jiang, J.H.; Yang, J.; Shi, Z.Y.; et al. Retrograde type a aortic dissection after endovascular stent graft placement for treatment of type b dissection. Circulation 2009, 119, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.B.; Andersen, N.D.; Bhattacharya, S.D.; Scheer, E.; Piccini, J.P.; McCann, R.L.; Hughes, G.C. Retrograde ascending aortic dissection as an early complication of thoracic endovascular aortic repair. J. Vasc. Surg. 2012, 55, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Canaud, L.; Ozdemir, B.A.; Patterson, B.O.; Holt, P.J.; Loftus, I.M.; Thompson, M.M. Retrograde aortic dissection after thoracic endovascular aortic repair. Ann. Surg. 2014, 260, 389–395. [Google Scholar] [CrossRef]
- Giles, K.A.; Beck, A.W.; Lala, S.; Patterson, S.; Back, M.; Fatima, J.; Arnaoutakis, D.J.; Arnaoutakis, G.J.; Beaver, T.M.; Berceli, S.A.; et al. Implications of secondary aortic intervention after thoracic endovascular aortic repair for acute and chronic type b dissection. J. Vasc. Surg. 2019, 69, 1367–1378. [Google Scholar] [CrossRef]
- Jang, H.; Kim, M.D.; Kim, G.M.; Won, J.Y.; Ko, Y.G.; Choi, D.; Joo, H.-C.; Lee, D.Y. Risk factors for stent graft-induced new entry after thoracic endovascular aortic repair for stanford type b aortic dissection. J. Vasc. Surg. 2017, 65, 676–685. [Google Scholar] [CrossRef] [PubMed]
- Kasirajan, K.; Dake, M.D.; Lumsden, A.; Bavaria, J.; Makaroun, M.S. Incidence and outcomes after infolding or collapse of thoracic stent grafts. J. Vasc. Surg. 2012, 55, 652–658. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).