Abstract
Chronic rhinosinusitis with nasal polyps (CRSwNP) is an inflammatory disease of the nose and paranasal sinuses characterized by the presence of nasal polyps. The symptoms produced by the presence of nasal polyps such as nasal obstruction, nasal discharge, facial pain, headache, and loss of smell cause a worsening in the quality of life of patients. The source of the nasal polyps remains unclear, although it seems to be due to a chronic inflammation process in the sinonasal mucosa. Fibroblasts, the main cells in connective tissue, are intimately involved in the inflammation processes of various diseases; to this end, we carried out a systematic review to evaluate their inflammatory role in nasal polyps. Thus, we evaluated the main cytokines produced by nasal polyp-derived fibroblasts (NPDF) to assess their involvement in the production of nasal polyps and their involvement in different inflammatory pathways. The results of the review highlight the inflammatory role of NPDF through the secretion of various cytokines involved in the T1, T2, and T3 inflammatory pathways, as well as the ability of NPDF to be stimulated by a multitude of substances. With these findings, the fibroblast is positioned as a new potential therapeutic target in the treatment of CRSwNP.
1. Introduction
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a subtype of chronic rhinosinusitis (CRS) that affects 18–20% of all patients with CRS and 1 to 4% of the world population [1,2]. Patients suffering from this condition are mainly adult males with a 2:1 preponderance, the disease being rare during childhood, where it is usually found in association with cystic fibrosis [3]. The quality of life of patients suffering from CRSwNP is greatly impaired. It is essentially due to the discomfort caused by the main symptoms, such as difficulty breathing and constant runny nose, but also due to the indirect effects of symptoms such as loss of smell, taste, headaches, changes in sleep patterns, daytime sleepiness, as well as decreased intellectual capacity [2].
The main phenotypic characteristic of these patients is the growth of nasal polyps generally bilateral at the level of the middle meatus, although they can also be found in the paranasal sinuses [4]. Macroscopically, nasal polyps are bright yellowish-grey, shiny, translucent masses composed of gelatinous inflammatory material, and they have a benign behaviour. Histologically, they are described as an oedematous myxoid stroma, infiltrated mainly by eosinophils covered by a respiratory epithelium that frequently presents hyperplasia or squamous metaplasia [5]. Therefore, in the inflammatory infiltrate of nasal polyps, fibroblasts appear to be one of the most abundant cells in polypoid tissues (47%) followed by eosinophils (20%) [6], and other inflammatory cell types, such as neutrophils, mast cells, lymphocytes, dendritic cells, plasma cells, and ILC2s [7,8,9,10].
The cause of the development of these polypoid lesions remains unclear, although advances in the understanding of the pathophysiology of the disease suggest that they are the product of a chronic inflammatory mechanism. There are several theories as to why this sustained inflammation would take place, such as an alteration of the mucosal barrier, the presence of microorganisms (such as bacteria, fungi, or viruses), the production of superantigens, or the presence of allergens [11]. Until recently, the inflammatory profile leading to CRSwNP was believed to be mediated almost exclusively by a Th2 response. Nevertheless, recent studies have shown that the inflammatory response is more complex and heterogeneous, with the involvement of three different inflammatory endotypes T1, T2, and T3 [2]. These endotypes are characterized by the secretion of various cytokines depending on the participation of Th1, Th2, and Th17 helper cells. Therefore, IFN-γ secretion is predominant in the T1 endotype, while the T2 endotype cytokines are characterized by IL-4, IL-5, and IL-13 cytokines, as well as eosinophilia, and the T3 endotype is associated with IL-17cytokine expression and neutrophilia [12]. Currently, many of these cytokines are used as therapeutic targets against certain CRSwNPs, such as IL-4 with dupilumab or IL-5 with mepolizumab.
Fibroblasts are the main cell type found in connective tissue and are mainly known for their production of the extracellular matrix (ECM). However, they are not only involved in the production of the ECM but are also intimately involved in inflammation due to their capacity to secrete cytokines, adipokines, and growth factors [13]. Thus, these mesenchymal cells appear to be directly involved in the innate host defence and have been linked to various inflammatory diseases such as rheumatoid arthritis [14], inflammatory bowel disease [15], lung inflammation [16], or nasal polyposis [17]. However, fibroblasts differ according to the anatomical site, varying their function, proliferation, and production of substances according to the requirements of the tissue environment [18]. Thus, we evaluated the main cytokines produced by nasal-polyp-derived fibroblasts (NPDF) to assess their involvement in the production of nasal polyps. This systematic review was designed with the aim of investigating the main cytokines secreted by NPDF, and their involvement in different inflammatory pathways.
2. Methods
A systematic review of the literature was carried out to study the main cytokines produced by nasal polyp fibroblasts.
2.1. Research Questions and Eligibility Criteria
With this systematic review, we aimed to answer the following research question: What are the cytokines involved in the inflammatory response produced by fibroblasts in patients with nasal polyps? This question was defined in terms of the PICO format. The criteria for the inclusion/exclusion of studies are shown in Table 1. The types of studies included were clinical trials, prospective, and retrospective studies published in peer-reviewed journals. Clinical cases and articles dating before 2000 were excluded. We also excluded articles that evaluated the inhibition of a cytokine or inflammatory pathway. The search was last updated in May 2022. Only studies published in English language were included.

Table 1.
Detailed criteria for inclusion/exclusion of studies.
2.2. Search Strategy
We followed the Preferred Reporting Items for Systematic Reviews and Meta- Analysis (PRISMA) [19], and the search was carried out in the following databases: Pubmed (Medline), SCOPUS, EMBASE, Web of Science, and the Cochrane Library. The search terms used were as index terms or free-text words and the complete strategy was as follows: ((“nose polyp” OR “nasal cavity polyp” OR “nasal polyp” OR “nasal polyposis” OR “nasal polyps” OR “nose polyp” OR “nose polyposis” OR “polyp of the nasal cavity” OR “polyp of the nose” OR “polyp, nose” OR “polyposis nasi” OR “polyposis of the nose” OR “chronic rhinosinusitis with nasal polyp”) AND (“fibroblast” OR “fibroblast cell” OR “fibroblasts” OR “primary fibroblast” OR “primary fibroblasts”) AND (“inflammation” OR “inflammation reaction” OR “inflammation response” OR “inflammatory condition” OR “inflammatory reaction” OR “inflammatory response” OR “reaction, inflammation” OR “response, inflammatory” OR “cytokine” OR “cytokines” OR “interleukin”)).
2.3. Data Collection
Two reviewers (JPG, SSG) independently screened the references based on the title and abstract followed by eligibility at the full-text level. In case of disagreements between reviewers when reviewing abstracts, articles were included in the full-text review phase for a final evaluation. The references of all selected articles were also manually reviewed to identify any potentially omitted publications.
2.4. Data Analysis
Results are discussed in narrative and table format.
3. Results
3.1. Search Results
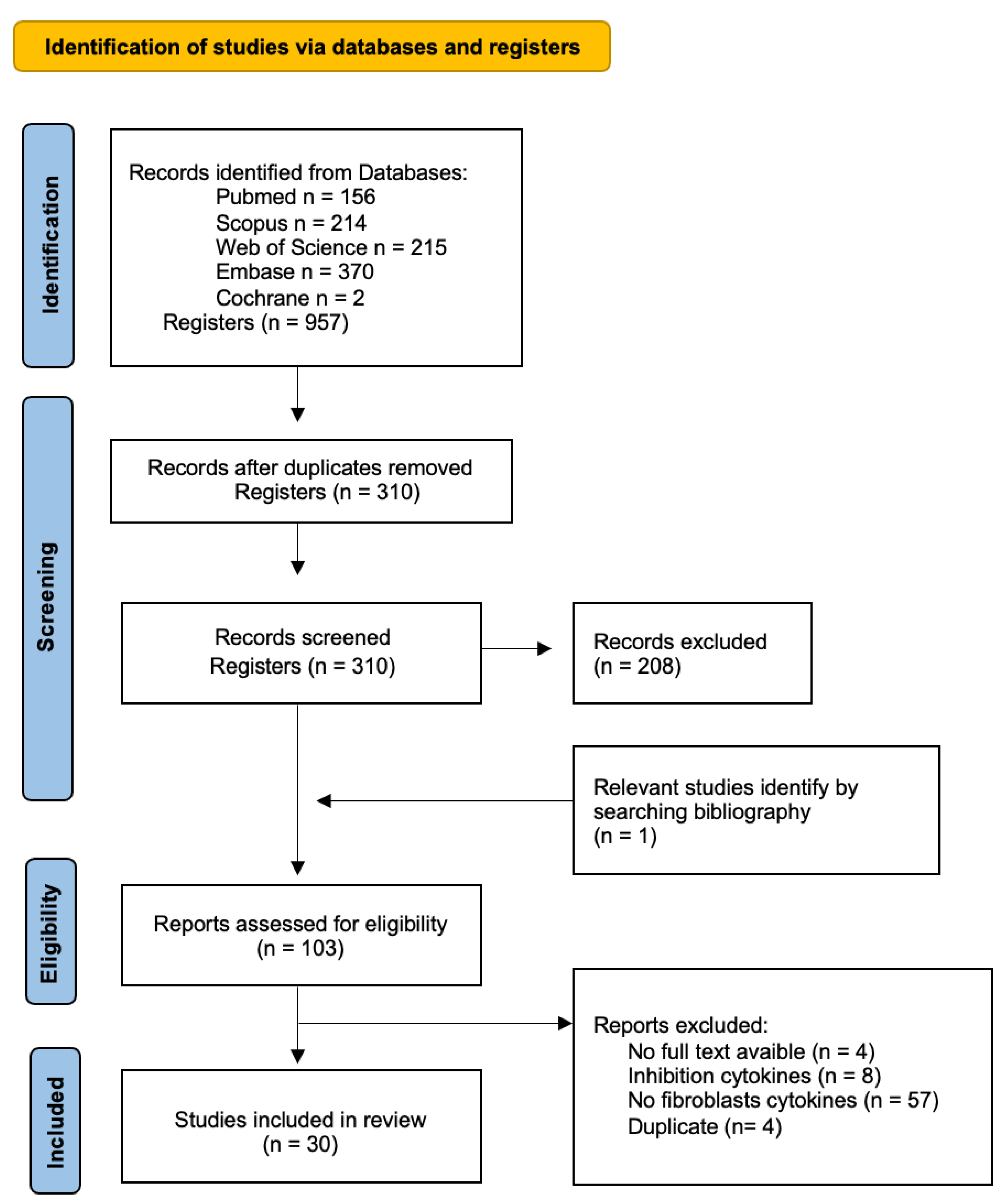
The initial search generated a total of 957 articles: 156 in Pubmed, 214 in Scopus, 215 in Web of Science, 370 in Embase, and 2 in Cochrane. Duplicate references found in different databases were removed. Next, we screened by title and abstract, including 102 articles for the full-text analysis. This analysis included a total of 30 articles to review. The flow chart of the literature search and selection process is shown in Figure 1. The baseline characteristics of the 30 articles included are presented in Table 2.
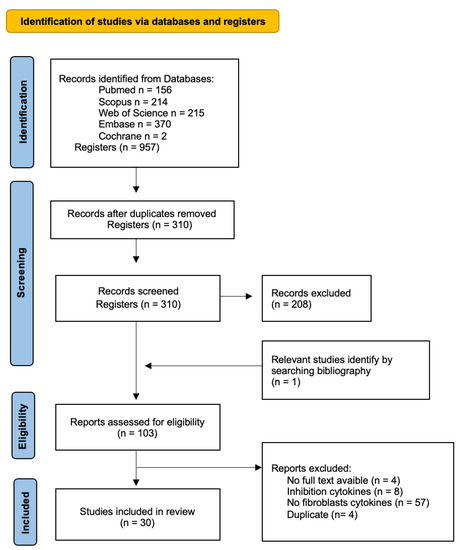
Figure 1.
Flow diagram of the identification, screening, and inclusion of studies within the systematic review.

Table 2.
Summary of studies investigating cytokines produced by NPDF.
3.2. NPDF Cytokines
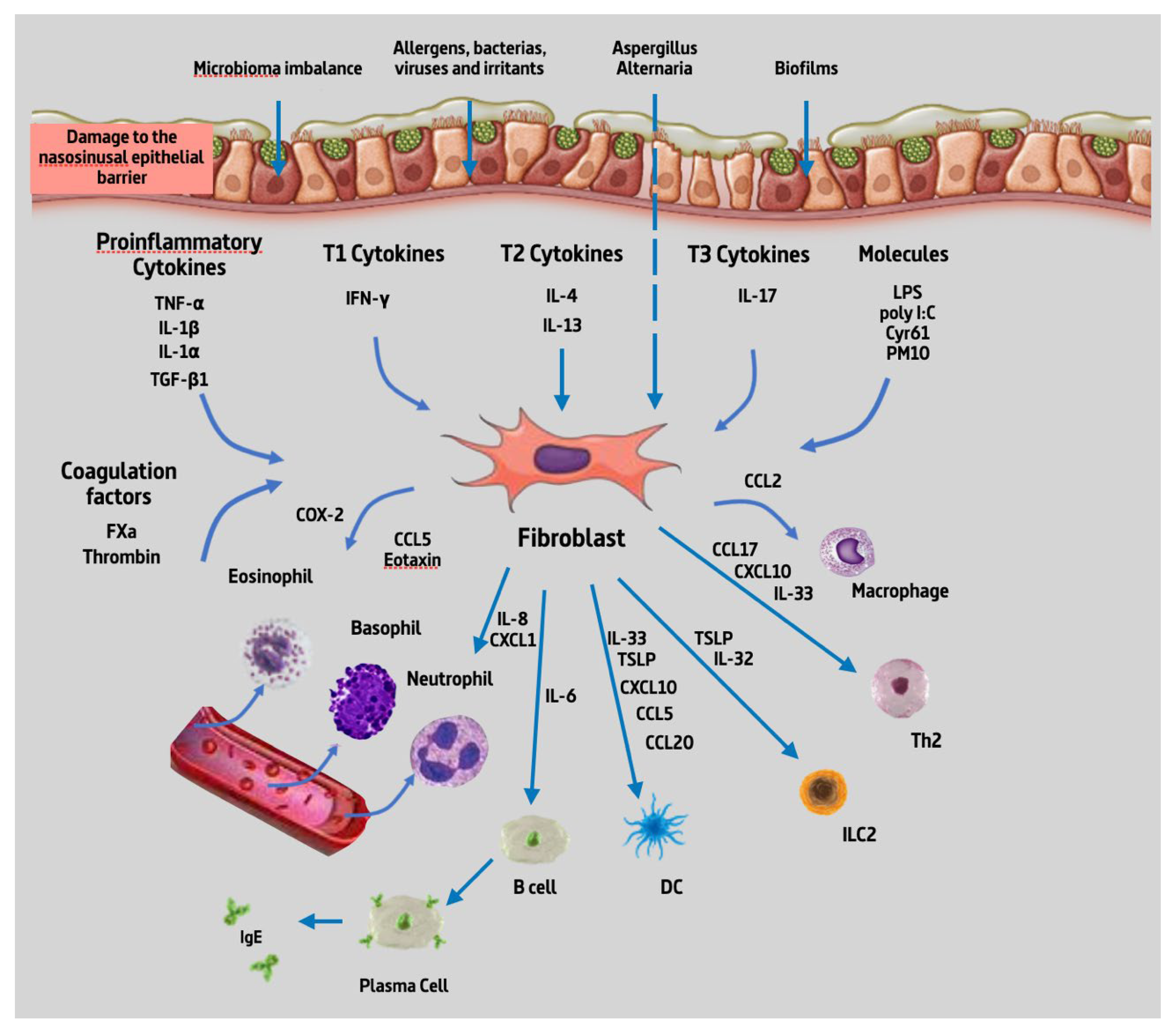
From the 32 articles reviewed, we found that NPDF could produce 19 different types of cytokines upon stimulation with about 22 different types of substances. Given the high number of cytokines produced by NPDF and substances capable of producing a stimulation, they are summarized in Figure 2 below.
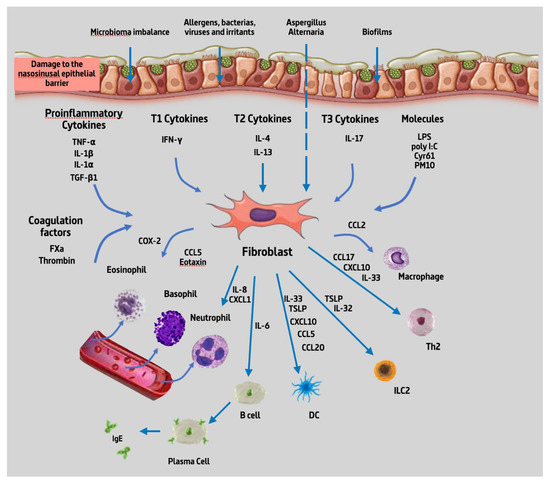
Figure 2.
Summary of cytokines produced by NPDF. Abbreviations: TNF-α, tumour necrosis factor alpha; IL-1β, interleukin 1 beta; IL-1⍺, interleukin 1 alpha; TGF- β, transforming growth factor beta; IFN-γ, interferon gamma; IL-4, interleukin 4; IL-13, interleukin 13; IL-17, interleukin 17; LPS, lipopolysaccharide; poly IC, polyinosinic:polycytidylic acid; Cyr61, cysteine-rich angiogenic inducer 61; PM10, particular matter smaller than 10 µm; FXa, coagulation factor Xa; COX-2, cyclooxygenase-2; CCL5, chemokine (C-C motif) ligand 5; IL-8, interleukin 8; CXCL1, C-X-C motif chemokine ligand 1; IL-6, interleukin 6; IL-32, interleukin 32; IL-33, interleukin 33; TSLP, thymic stromal lymphopoietin; CXCL10, C-X-C motif chemokine ligand 10; CCL20, chemokine (C-C motif) ligand 20, CCL17, chemokine (C-C motif) ligand 17; CCL2, chemokine (C-C motif) ligand 2; B cell, B lymphocytes; DC, dendritic cell; ILC2, type 2 innate lymphoid cells; Th2, T helper 2 lymphocytes; IgE, immunoglobulin E.
4. Discussion
Fibroblasts are present in all tissues and play a fundamental role in processes such as wound healing, ECM remodelling, and the modulation of a tissue-specific immune response [20]. They represent the main type of mesenchymal cells and have the ability to receive signals from the epithelial barrier upon damage. At the same time, fibroblasts can emit signals to activate innate immune cells [21]. In fact, NPDF themselves, as well as other fibroblasts of other tissues, appear to act as sentinels of tissue-specific innate immune responses by producing inflammatory mediators such as cytokines, chemokines, antimicrobial peptides, and growth factors in response to various substances [22]. NPDF actively participate in epithelial remodelling and proliferation, while modulating ECM through a proinflammatory transcriptional programme with resistance to apoptotic signalling, which may explain the maintenance of inflammatory signalling in the production of nasal polyps [23].
4.1. Chemokine C-C Motif Ligand 5 (CCL5)
NPDFs are capable of expressing the cytokine CCL5, also known as RANTES, upon stimulation with proinflammatory cytokines such as TNF-⍺, IL-1β, and IL-4 [24,25,26]. CCL5 (RANTES) is a chemoattractant protein of various types of inflammatory cells, such as monocytes, memory T cells, basophils, macrophages, mast cells, and eosinophils [27]. In CRSwNP, RANTES has been proposed to act as a strong attractant of eosinophil infiltration [28]. However, Yoshikufu et al. [26] found no differences in RANTES expression levels in eosinophil-rich nasal polyp versus noneosinophilic nasal polyp, indicating that increased RANTES secretion may not be associated with eosinophilic infiltration in nasal polyps. The transcriptional upregulation of RANTES is mediated by nuclear factor κB (NFκB) following the activation of TLR1/TLR2, TLR3, and NOD1 [29]. These fast intracellularly acting pathways are generally present in the innate response of the immune system generally after contact with pathogen-associated molecular patterns (PAMPs), suggesting that NPDF may be involved in the initial response to epithelial damage.
4.2. Eotaxins
Eotaxins are a family of eosinophil recruitment chemokines that carry out their activity through CCR3 receptors [30]. In humans, there are three types of eotaxins: CCL11 (eotaxin-1), CCL24 (eotaxin-2), and CCL26 (eotaxin-3). Terada et al. [31] reported that nasal mucosa fibroblasts were the major source of eotaxins in the nasal mucosa. Nasal fibroblasts are capable of producing at least CCL11 (eotaxin-1) and CCL26 (eotaxin-3) but do not appear capable of secreting CCL24 (eotaxin-2) [32]. NPDF produces CCL11 (eotaxin-1) nonconstitutively upon IL-4 stimulation or costimulation with lipopolysaccharide (LPS) and IL-4 [33,34]. Furthermore, CCL11 (eotaxin-1) appears to be more upregulated in patients with eosinophilic nasal polyps than in those with noneosinophilic polyps following IL-4 stimulation or co-stimulation with TNF-α and IL-4 [26]. On the other hand, Shimizu et al. [35] also verified the production of eotaxin-1 after induction with activated coagulation factors such as thrombin or FXa, which highlights the role of coagulation factors in CRSwNP. Recently, it has been shown that NPDF can produce CCL26 (eotaxin-3) after stimulation with IL-4, IL-13, and leptin [36]. NPDF eotaxin production participates not only in the recruitment of eosinophils, with a subsequent secretion of cytokines, but also in the leakage of plasma into polypoid tissue with the recruitment of other substances such as leptin or coagulation factors, which may favour the development of nasal polyps.
4.3. Interleukin-4 (IL-4)
IL-4 is a cytokine that promotes the differentiation of Th2 lymphocytes and B lymphocytes [37]. In patients with CRSwNP, IL-4 is normally elevated and is associated with its involvement in the loss of epithelial integrity by decreasing the expression of occluding epithelial tight junction proteins and zonula occludens [38]. The production of IL-4 by NPDF was demonstrated by Lee et al. [39] after a stimulation of NPDF with particulate matter <10 μm (PM10). This IL-4 production after PM places air pollution as a risk factor for the development of CRSwNP [40,41]. Steinke et al. [34] found that NPDF contained IL-4 receptors (IL-4R) and that their activation caused the upregulation of TGF-1, IL-6, CCL11 (eotaxin-1), and CCL13 (MCP-4). This upregulation occurred in constitutively expressed cytokines, except for CCL11 (eotaxin-1), which did not appear to be constitutively expressed, probably due to the need for eosinophilic regulation by NPDF. This migration of eosinophils can be promoted by a single stimulation, with IL-4 secreting CCL13 (MCP-4) [42], or by combined stimulations of IL-4 with TNF-⍺, LPS, and TFG-β1 [26,33].
However, many other constitutive cytokines are not activated by IL-4 such as IL-6, IL-8, CCL5 (RANTES), CCL13 (MCP-4), and GM-CSF. Furthermore, IL-4 may have an inhibitory role in the secretion of some interleukins by NPDF, such as the inhibition of IL-11 [34]. This inhibition of interleukins, such as IL-11, may be crucial in maintaining the inflammatory state in nasal polyps, as IL-11 has been shown to attenuate inflammation in diseases such as rheumatoid arthritis or inflammatory bowel diseases [43,44].
4.4. Interleukin-6 (IL-6) and (IL-8)
IL-6 and IL-8 production is generally increased in CRSwNP [45,46], as well as other inflammatory airway diseases such as asthma and COPD [47,48]. IL6- and IL-8 have as their main function neutrophil chemotaxis. In addition to this shared function, IL-6 is also involved in increasing epithelial cell proliferation [49], while IL-8 is involved in angiogenesis [50]. NPDF are capable of producing these cytokines together or separately after stimulation with various substances. Liu et al. [51] found that the NPDF secretion of IL-6 after stimulation with TNF-⍺ and IL-4 played a role in the pathogenesis of nasal polyps by modulating the immune response and ECM secretion via cycloxygenase-2 (COX-2). Cho et al. [52] confirmed that the COX-2 pathway was important for the production of IL-6 and IL-8 by stimulating NPDF with prostaglandin E2 (PGE2), a metabolite of COX-2, proving that its activation depended on the Akt and the NFκB pathways. The expression of IL-6 and IL-8 by NPDF also occurs after stimulation with cellular products. LPS is capable of producing IL-6 and IL-8 secretion through the PI3K/Akt pathway [53]. Alternaria stimulus, a saprophytic fungus, also produces the NPDF secretion of IL-6 and IL-8 through pattern recognition receptors (PRR), receptors present in the innate immune system for the recognition of microbial molecules [54]. Peripheral blood mononuclear cells (PBMC) stimulated with the house dust mite Dermatophagoides pteronyssinus also produce an increase in IL-6 and IL-8 [55].
Shun et al. [56] demonstrated the production of IL-8 and VEGF by NPDF under hypoxic conditions, leading to angiogenesis and the neutrophil infiltration of nasal polyps. This IL-8 production by NPDF can also occur after stimulation with enzymes of the coagulation cascade, such as factor Xa or thrombin, thus indicating that the coagulation system is an inducer of cytokine production in nasal polyps [35] and is probably involved in the accumulation of extracellular fluid required for polyp formation.
4.5. Interleukin-13 (IL-13)
IL-13 is a cytokine whose main function is to regulate monocyte and B cell function [37]. In addition to these main functions in patients with CRPScPNS, it has been implicated in the disruption of the epithelial barrier [57], goblet cell hyperplasia [58], and eosinophil recruitment [33]. The expression of IL-13 by NPDF has not been previously reported, but it appears capable of its recognition when expressing the alpha IL-13 receptor (IL-13R⍺) [59]. The IL-13R⍺1 subunit appears to be constitutively expressed, while the IL-13R⍺2 subunit needs to be previously stimulated with TNF-⍺ and/or IL-4 to be upregulated. The synergistic combination of these proinflammatory cytokines (TNF-⍺ and IL-4) induces the upregulation of IL-13R⍺2 but not IL-13R⍺1. IL-13R⍺2 has a high affinity for IL-13 binding and appears to act as a decoy receptor that inhibits STAT6, the canonical IL-13 activation pathway [60,61]. Additionally, IL-13R⍺2 also inhibits the action of IL-4 because this cytokine shares with IL-13 the IL-4 type II receptor for cell signalling [62]. These inhibitory mechanisms may be part of a cellular regulatory feedback in the face of exaggerated inflammatory stimuli by dysregulated IL-4 and IL-13 signalling.
4.6. Interleukin-17 (IL-17)
The T3 inflammatory pathway is dominated by Th17 and ILC3 cells, which characteristically produce IL-17A and IL-22 [63,64]. IL-17 is involved in autoimmune inflammation processes and allergic reactions [65]. This signalling pathway mainly produces a neutrophil recruitment, and according to the study by Cheng et al. [66], NPDF, upon stimulation by TNF-α, are able to produce increased levels of IL-17A via the PI3K/Akt pathway, thus indicating that NPDF may participate in some way in the T3 inflammatory pathway by recruiting neutrophils in cases of CRSwNP without eosinophilia. In addition to the ability to secrete IL-17A, this cytokine can also activate NPDF to produce IL-8 and CXCL1. NPDF secretion of these two cytokines after IL-17 stimulation is also correlated with neutrophil chemotaxis, which in turn would lead to an increased IL-17 secretion, accumulating more neutrophils in the nasal mucosa, therefore causing a chronic inflammatory loop [67,68]. Homma et al. [69] confirmed the IL-8 and CXCL1 production after stimulation with IL-17, but they also proved that the stimulation with IL-17 also produced IL-6, IL-9, MCP-1 (CCL2), G-CSF, cytokines involved in the attraction of eosinophils.
The other main interleukin in the T3 pathway is IL-22, which is a fundamental mediator of inflammation after tissue damage, activating the epithelial cells of many organs, including fibroblastic cells [70]. This interleukin has other functions such as mucus production, protective function against pathogens, wound healing, and tissue regeneration that may be deregulated in chronic inflammation [71]. However, in our review, we could not find any study analysing NPDF stimulation with this cytokine or reporting IL-22 production by NPDF.
Although endotyping has proven to be pivotal in the management of chronic inflammation of CRSwNP, the actual clinical presentation may comprise a spectrum with overlapped inflammatory responses. Indeed, Hao et al. [72] found that IL-17A production correlated in turn with IFN-γ production, which highlights the existence of a synergistic effect between the T1 and T3 inflammatory response in the pathogenesis of certain patients with CRSwNP.
4.7. Interleukin-32 (IL-32)
IL-32 is a proinflammatory cytokine produced by monocytes, T cells, natural killer cells, dendritic cells, endothelial and epithelial cells, and fibroblasts [73,74]. IL-32 expression is induced by numerous cytokines such as TNF-α, IL-1β, IL-12, and IL-18 [75,76]. Keswani et al. [77] found that IL-32 mRNA was significantly increased in nasal polyp tissue from patients with CRSwNP compared to uncinate tissue from the same patients and healthy controls. Similarly, Soyka et al. [78] also demonstrated that IL-32 mRNA expression levels were significantly increased in nasal polyp compared to control tissues. Cho et al. [79] demonstrated that NPDF could induce IL-32 production when stimulated by LPS, and this effect was mediated by the TLR4/JNK/AKT/CREB pathway.
Interleukin-33 (IL-33) is a cytokine that belongs to the IL-1 superfamily group. It can be expressed by various cell types such as mast cells, macrophages, dendritic cells, fibroblasts, osteoblasts, epithelial, and endothelial cells [80] and induces many immune cells such as ILC2, T helper cells, mast cells, eosinophils, dendritic cells, and basophils [81]. It has been described as being involved in allergic and nonallergic inflammation via the IL-33/ST2 pathway, being considered an alarmin following tissue damage [82]. Recently, Lee et al. [39] described the ability of NPDF to secrete IL-33 after PM10 stimuli, suggesting that NPDF may be one of the first cells to participate in the inflammatory response of the nasal mucosa of patients with nasal polyps.
4.8. CXC Motif Chemokine Ligand 10 (CXCL10)
CXCL10, also known as interferon gamma-induced protein 10 (IP-10), is a cytokine secreted in response primarily to IFN-γ by cells such as monocytes, fibroblasts, and endothelial cells [83]. CXCL10 can function as a chemoattractant for T cells, monocytes, NK cells, and dendritic cells, in addition to its role in cell adhesion functions, antitumor activity, angiogenesis, and inhibition of bone marrow colonies [83,84]. Yoshikawa et al. [85] found that CXCL10 expression by NPDF was upregulated by poly IC from CRS patients with asthma and this induced a Th1 cell infiltration into nasal polyp tissues. This activation of the T1 inflammatory pathway by viral immunostimulatory particles appears to be inhibited by bacteria such as S. aureus, which appears to downregulate IP-10 production and prevent Th1 cell recruitment [86]. Recently, Nam et al. [87] pointed to elevated levels of IkappaB kinase (IKK), in particular IKKε, as the trigger for severe eosinophilic inflammation with CXCL10 overproduction.
4.9. Chemokine C-C Motif Ligand 2 (CCL2)
CCL2 is a small cytokine that belongs to the CC chemokine family [88]. Its main function is to recruit monocytes/macrophages into tissues; hence, it is also called monocyte chemoattractant protein 1 (MCP1). NPDF can produce CCL2 after the activation of TNF-⍺ mainly through the B-Raf/MEK/ERK pathway, although also with the participation of transcriptional factors such as c-Fos and AP-1 [89,90]. The macrophage-stimulating potential of CCL2 is correlated with COX-2 expression, which is, in turn, associated with the development of polypoid lesions, such as nasal polyps [91] or colonic polyps [92].
4.10. Chemokine C-C Motif Ligand 17 (CCL17)
CCL17, also called thymus and activation-regulated chemokine (TARC), is a cytokine belonging to the CC chemokine family and is associated with type 2 immune responses by inducing T cell chemotaxis through its interaction with the CCR4 chemokine receptor [93]. NPDF can produce CCL17 (TARC) through the combined stimulation of IL-4 and LPS [94]. Nonaka et al. [95] demonstrated that NPDF did not induce CCL17 (TARC) production after individual stimulation with TLR ligands. However, a combined stimulation of different TLRs, such as TLR2, 3, 4, 5 ligands, together with IL-4, induced an increase in CCL17 (TARC) secretion. The same author found that the stimulation of NPDF with poly IC, poly IC and TNF-⍺, poly IC and IL-4, or IL-4 and TNF-⍺ induced a significant increase in the expression of TARC. Furthermore, the simultaneous exposure of cells to poly IC, IL-4, and TNF-⍺ induced an even higher increase in the expression of CCL17 (TARC) [96,97]. In view of these findings, IL-4 seems fundamental in the induction of CCL17 (TARC) expression by NPDF and in the regulation of T2 cell chemotaxis.
4.11. Chemokine C-C Motif Ligand 20 (CCL20)
Chemokine ligand 20, also known as macrophage inflammatory protein-3 (MIP-3α), is a chemokine that attracts lymphocytes and to a lesser extent neutrophils [98]. It interacts with the CCR6 receptor, a property shared with antimicrobial beta-defensins [99]. It is produced by dendritic cells (DC), monocytes, granulocytes, T cells, B cells, epithelial cells, endothelial cells, and synovial fibroblasts [75,100], and its production is induced by LPS, TNF-⍺, and IFN-γ and deregulated by IL-10 [101]. NPDF are capable of producing CCL20 (MIP-3α) upon stimulation with cytokines such as IL-1β and TNF-α, but also by TLR2, 3, 4, and 5 ligands, suggesting the ability of NPDF to recruit dendritic cells [6]. In addition, IL-17 has also been shown to act synergistically with TNF-α in inducing CCL20 (MIP-3α) production by NPDF. Thus, the involvement of Th17 cells in the pathophysiology of nasal polyps is emphasised [102].
4.12. Thymic Stromal Lymphopoietin (TSLP)
Thymic stromal lymphopoietin (TSLP) is an epithelial cell-derived cytokine implicated in the initiation and persistence of inflammatory pathways. It is released predominantly by ciliated epithelial cells, mast cells, macrophages, and endothelial cells [103,104]. NPDF can secrete TSLP after synergistic stimulation with TNF-α and Th2 cytokines (IL-4 and IL-13). On the other hand, stimulation with Th1 cytokines, such as IFN-γ, can inhibit TSLP production, indicating its regulatory role in dendritic cell activation [105,106]. Furthermore, NPDF can produce TSLP after the activation of TLR2 by fungi such as Alternaria, which could also facilitate the development and exacerbation of type 2 nasal inflammation in patients with CRSwNP [107]. Consequently, the production of TSLP by fibroblasts suggests that these cells may play a role in the development and regulation of Th2 inflammation [108].
4.13. Overall Effects of Cytokines Produced by NPDF in CRSwNP
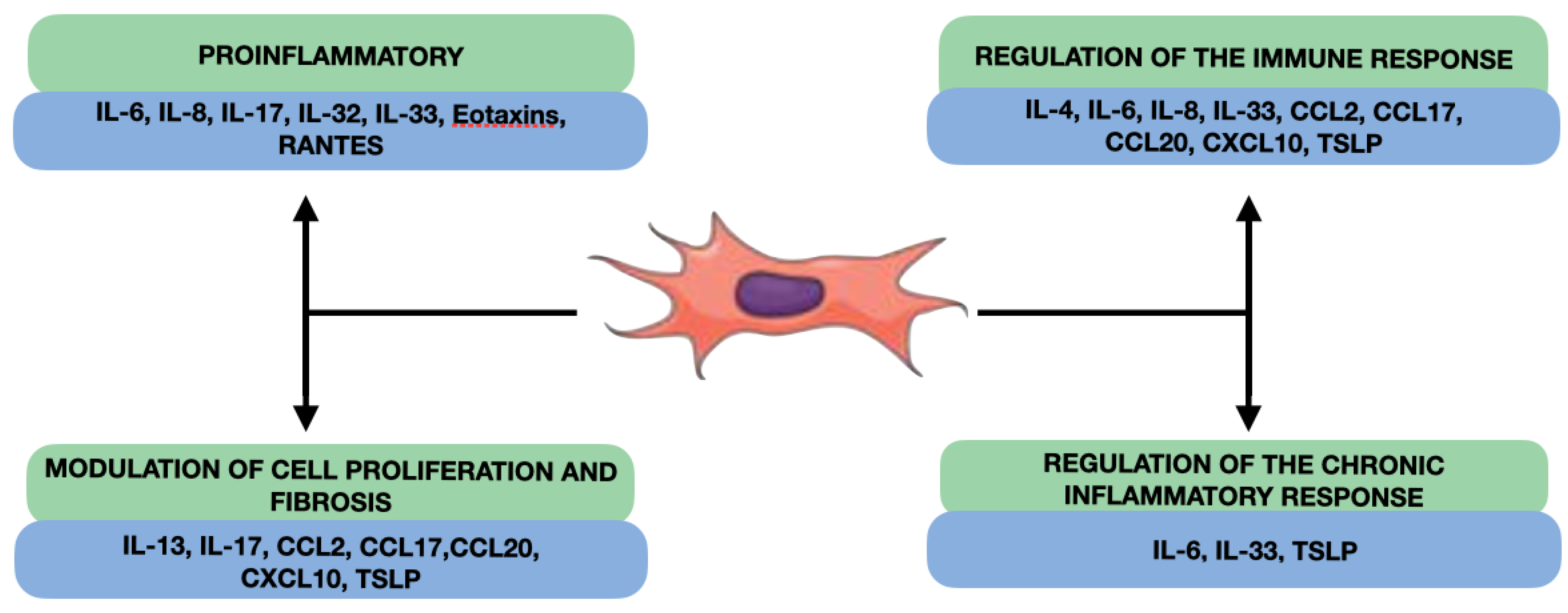
Our review reveals that fibroblast-produced interleukins in nasal polyps can have several effects on the inflammation and pathogenesis of CRSwNP, such as the promotion of inflammation, the regulation of the immune response, the modulation of cell proliferation and fibrosis, and the participation in the regulation of the chronic inflammatory response (Figure 3). NPDF can secrete proinflammatory interleukins, such as IL-6 and IL-8, which can act as proinflammatory mediators in the nasal cavity [109]. Furthermore, NPDF-produced interleukins can also interact with the cells of the local immune system and inflammatory cells and modulate the immune response in nasal polyps (Figure 2). Some interleukins produced by fibroblasts, such as TGF-β, and IL-4, or platelet-derived growth factor (PDGF) may have profibrogenic effects, stimulating cell proliferation and ECM production, such as collagen and proteoglycans [110,111]. This may contribute to the formation of fibrous tissue characteristic of nasal polyps and to the remodelling of connective tissue. Finally, the NPDF-produced interleukins can also be involved in the regulation of the chronic inflammatory response in nasal polyps, interacting with other cells and inflammatory mediators. For example, IL-6 may act as a proinflammatory and proangiogenic mediator in nasal polyps, contributing to chronic inflammation and polyp growth [112].
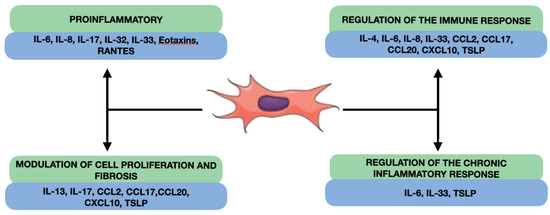
Figure 3.
Overall effects of cytokines produced by NDPF.
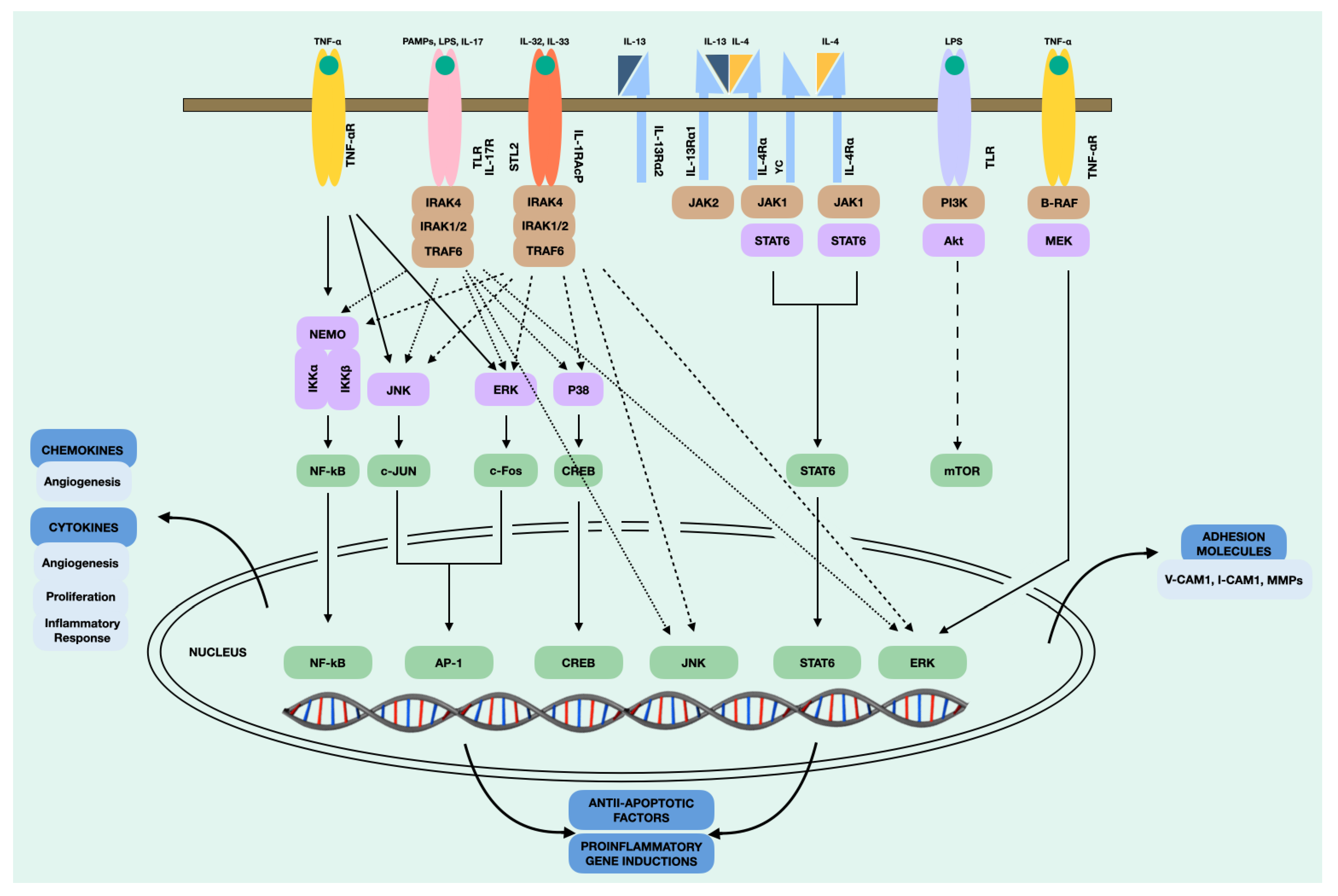
In addition, NPDF, upon stimulation, trigger intracellular signals that lead to the subsequent secretion of cytokines. This process involves a series of biochemical and molecular events that allow the transmission of the signal through the cell, eventually leading to a specific cellular response. It is important to note that intracellular signalling is a highly regulated and complex process that can vary depending on the cell type, the type of signal, and the cellular context. In our case, we found numerous intracellular signalling pathways in the NPDF after stimulation. The main intracellular signalling pathways that take place in the NPDF are shown in Figure 4. The NF-kB pathway is involved in the induction of inflammatory cytokines, such as RANTES, and the activation of chronic inflammation [113]. The JAK1/STAT6 pathway is stimulated primarily by IL-4 and IL-13, which produce the Th2 immune response and macrophage activation [114]. On the other hand, MAPK/ERK is a complex pathway that participates in the proliferation and cell cycle [115], which, in the case of NPDF, appears to be activated by TNF-α for CCL2 production. Recent studies such as that of Nemec et al. [116] demonstrated that a structurally guided chemical–genetic approach could be rationally applied to target kinase receptors. Therefore, we believe that further targeted studies are needed on these intracellular signalling pathways that may be susceptible to inhibition.
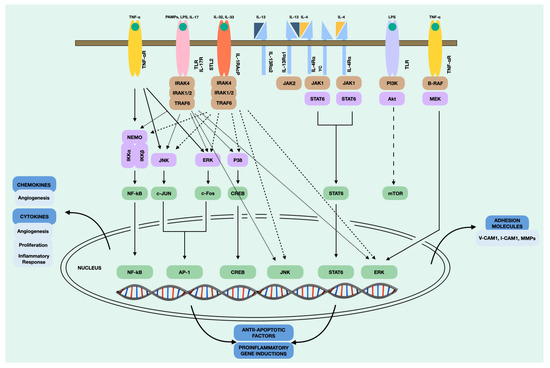
Figure 4.
Intracellular signalling pathways of the NDPF.
5. Limitations
This review has a few limitations. However, we believe that due to the search criteria used, since the articles included only evaluated cytokine production after fibroblast stimulation, we cannot verify the inhibitory role that certain substances can play in fibroblast activity, as well as their constitutive production without prior stimulation, as described by Homma et al. [69]. Additionally, the production of other cytokines not included in the review may have been described in articles prior to 2000 excluded from our study.
6. Future Directions
Currently, therapeutic targets in CRSwNP focus on various interleukins such as IL-4, IL-5, and IL-13 produced by Th2 lymphocytes or immunoglobulins such as IgE, produced by B lymphocytes or plasma cells [117,118,119]. The purpose of this review was to evaluate the inflammatory role of NPDF as a major source of cytokine production in the matrix of nasal polyps. Indeed, NPDF produces a great diversity of cytokines that act by activating different inflammatory cells, being directly or indirectly involved in all three inflammatory endotypes T1, T2, and T3. Therefore, we believe that it is interesting to evaluate the role of NPDF as a possible therapeutic target. Future research is needed to determine whether fibroblast inhibition can block nasal polyp production or produce less aggressive CRSwNP.
In addition, we believe it is of interest to evaluate the different behaviours of the fibroblast within other locations of the nasal cavity such as the floor of the nasal fossa, where nasal polyps do not develop [120,121].
Author Contributions
Conceptualization, J.P.-G. and R.M.-L.; writing—original draft preparation, J.P.-G., C.P.-G., R.M.-L. and S.S.-G., writing—review and editing, J.P.-G., C.P.-G., R.M.-L., J.M.-S., J.P.-P., J.L.M.-B. and S.S.-G.; supervision, J.P.-P., J.L.M.-B. and S.S.-G. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by grant PIGE-0367-2019 and RH-0048-2021 of the “Consejería de Salud y Familias, Junta de Andalucía”, and PAIDI-POSTDOC-2021 Spain of the “Consejería de Transformación Económica, Industria, Conocimiento y Universidades de la Junta de Andalucía”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The study did not report any data.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Stevens, W.W.; Schleimer, R.P.; Kern, R.C. Chronic Rhinosinusitis with Nasal Polyps. J. Allergy Clin. Immunol. Pract. 2016, 4, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.J.; Lund, V.J.; Hopkins, C.; Hellings, P.W.; Kern, R.; Reitsma, S.; Toppila-Salmi, S.; Bernal-Sprekelsen, M.; Mullol, J.; Alobid, I.; et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology 2020, 58, 1–464. [Google Scholar] [CrossRef] [PubMed]
- Newton, J.R.; Ah-See, K.W. A review of nasal polyposis. Ther. Clin. Risk Manag. 2008, 4, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Scadding, G.K.; Durham, S.R.; Mirakian, R.; Jones, N.S.; Drake-Lee, A.B.; Ryan, D.; Dixon, T.A.; Huber, P.A.J.; Nasser, S.M. BSACI guidelines for the management of rhinosinusitis and nasal polyposis. Clin. Exp. Allergy 2008, 38, 260–275. [Google Scholar] [CrossRef] [PubMed]
- Tos, M.; Mogensen, C. Pathogenesis of nasal polyps. Rhinology 1977, 15, 87–95. [Google Scholar] [PubMed]
- Nonaka, M.; Ogihara, N.; Fukumoto, A.; Sakanushi, A.; Kusama, K.; Pawankar, R.; Yagi, T. Nasal polyp fibroblasts produce MIP-3alpha in response to toll-like receptor ligands and cytokine stimulation. Rhinology 2010, 48, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, M.; Pawankar, R.; Fukumoto, A.; Ogihara, N.; Sakanushi, A.; Yagi, T. Distinct role for nasal fibroblasts in initiation of the eosinophilic inflammatory response. Clin. Exp. Allergy Rev. 2005, 5, 77–80. [Google Scholar] [CrossRef]
- Bernstein, J.M.; Gorfien, J.; Noble, B.; Yankaskas, J.R. Nasal polyposis: Immunohistochemistry and bioelectrical findings (a hypothesis for the development of nasal polyps). J. Allergy Clin. Immunol. 1997, 99, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Shiozawa, A.; Ono, N.; Kusunoki, T.; Hirotsu, M.; Homma, H.; Saitoh, T.; Murata, J. Subclassification of chronic rhinosinusitis with nasal polyp based on eosinophil and neutrophil. Laryngoscope 2013, 123, E1–E9. [Google Scholar] [CrossRef]
- Ho, J.; Bailey, M.; Zaunders, J.; Mrad, N.; Sacks, R.; Sewell, W.; Harvey, R.J. Cellular comparison of sinus mucosa vs polyp tissue from a single sinus cavity in chronic rhinosinusitis. Int. Forum Allergy Rhinol. 2015, 5, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.J.; Lund, V.J.; Mullol, J.; Bachert, C.; Alobid, I.; Baroody, F.; Cohen, N.; Cervin, A.; Douglas, R.; Gevaert, P.; et al. EPOS 2012: European position paper on rhinosinusitis and nasal polyps 2012. A summary for otorhinolaryngologists. Rhinology 2012, 50, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Klingler, A.I.; Stevens, W.W.; Tan, B.K.; Peters, A.T.; Poposki, J.A.; Grammer, L.C.; Welch, K.C.; Smith, S.S.; Conley, D.B.; Kern, R.C.; et al. Mechanisms and biomarkers of inflammatory endotypes in chronic rhinosinusitis without nasal polyps. J. Allergy Clin. Immunol. 2021, 147, 1306–1317. [Google Scholar] [CrossRef] [PubMed]
- Cavagnero, K.J.; Gallo, R.L. Essential immune functions of fibroblasts in innate host defense. Front. Immunol. 2022, 13, 1058862. [Google Scholar] [CrossRef] [PubMed]
- Croft, A.P.; Campos, J.; Jansen, K.; Turner, J.D.; Marshall, J.; Attar, M.; Savary, L.; Wehmeyer, C.; Naylor, A.J.; Kemble, S.; et al. Distinct fibroblast subsets drive inflammation and damage in arthritis. Nature 2019, 570, 246–251. [Google Scholar] [CrossRef]
- Kinchen, J.; Chen, H.H.; Parikh, K.; Antanaviciute, A.; Jagielowicz, M.; Fawkner-Corbett, D.; Ashley, N.; Cubitt, L.; Mellado-Gomez, E.; Attar, M.; et al. Structural Remodeling of the Human Colonic Mesenchyme in Inflammatory Bowel Disease. Cell 2018, 175, 372–386.e17. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Liu, L.; Roth, M.; Tian, J.; He, Q.; Zhong, B.; Bao, R.; Lan, X.; Jiang, C.; Sun, J.; et al. PRMT1 Upregulated by Epithelial Proinflammatory Cytokines Participates in COX2 Expression in Fibroblasts and Chronic Antigen-Induced Pulmonary Inflammation. J. Immunol. 2015, 195, 298–306. [Google Scholar] [CrossRef]
- Park, S.-K.; Jin, Y.-D.; Park, Y.-K.; Yeon, S.-H.; Xu, J.; Han, R.-N.; Rha, K.-S.; Kim, Y.-M. IL-25-induced activation of nasal fibroblast and its association with the remodeling of chronic rhinosinusitis with nasal polyposis. PLoS ONE 2017, 3, e0181806. [Google Scholar] [CrossRef]
- Van Linthout, S.; Miteva, K.; Tschope, C. Crosstalk between fibroblasts and inflammatory cells. Cardiovasc. Res. 2014, 102, 258–269. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Wei, K.; Nguyen, H.N.; Brenner, M.B. Fibroblast pathology in inflammatory diseases. J. Clin. Investig. 2021, 131, e149538. [Google Scholar] [CrossRef]
- Nowarski, R.; Jackson, R.; Flavell, R.A. The Stromal Intervention: Regulation of Immunity and Inflammation at the Epithelial-Mesenchymal Barrier. Cell 2017, 168, 362–375. [Google Scholar] [CrossRef] [PubMed]
- Bautista-Hernández, L.A.; Gómez-Olivares, J.L.; Buentello-Volante, B.; Bautista-de Lucio, V.M. Fibroblasts: The unknown sentinels eliciting immune responses against microorganisms. Eur. J. Microbiol. Immunol. 2017, 7, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Porras-González, C.; Palacios-García, J.M.; Sánchez-Gómez, S.; Maza-Solano, J.M.; Alba, G.; Sánchez-Margalet, V.; Palorames, O.; Del Cuvillo, A.; Cordero-Varela, J.A.; Moreno-Luna, R.; et al. Transcriptional analysis of nasal polyps fibroblasts reveals a new source of pro-inflammatory signaling in CRSwNP. Rhinology 2023, 61, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Saji, F.; Nonaka, M.; Pawankar, R. Expression of RANTES by IL-1 beta and TNF-alpha stimulated nasal polyp fibroblasts. Auris Nasus Larynx 2000, 27, 247–252. [Google Scholar] [CrossRef]
- Yamada, T.; Fujieda, S.; Yanagi, S.; Yamamura, H.; Inatome, R.; Yamamoto, H.; Igawa, H.; Saito, H. IL-1 induced chemokine production through the association of Syk with TNF receptor-associated factor-6 in nasal fibroblast lines. J. Immunol. 2001, 167, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Yoshifuku, K.; Matsune, S.; Ohori, J.; Sagara, Y.; Fukuiwa, T.; Kurono, Y. IL-4 and TNF-alpha increased the secretion of eotaxin from cultured fibroblasts of nasal polyps with eosinophil infiltration. Rhinology 2007, 45, 235–241. [Google Scholar] [PubMed]
- Bishara, N. The Use of Biomarkers for Detection of Early- and Late-Onset Neonatal Sepsis. In Hematology, Immunology and Infectious Disease: Neonatology Questions and Controversies; Elsevier: Amsterdam, The Netherlands, 2012; pp. 303–315. [Google Scholar] [CrossRef]
- Meyer, J.E.; Bartels, J.; Görögh, T.; Sticherling, M.; Rudack, C.; Ross, D.A.; Maune, S. The role of RANTES in nasal polyposis. Am. J. Rhinol. 2005, 19, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Bérubé, J.; Bourdon, C.; Yao, Y.; Rousseau, S. Distinct intracellular signaling pathways control the synthesis of IL-8 and RANTES in TLR1/TLR2, TLR3 or NOD1 activated human airway epithelial cells. Cell. Signal. 2009, 21, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Erin, E.M.; Williams, T.J.; Barnes, P.J.; Hansel, T.T. Eotaxin receptor (CCR3) antagonism in asthma and allergic disease. Curr. Drug Targets Inflamm. Allergy 2002, 1, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Terada, N.; Hamano, N.; Nomura, T.; Numata, T.; Hirai, K.; Nakajima, T.; Yamada, H.; Yoshie, O.; Ito, I.; Konno, A. Interleukin-13 and tumour necrosis factor-alpha synergistically induce eotaxin production in human nasal fibroblasts. Clin. Exp. Allergy 2000, 30, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, D.; Meyer, J.; Pods, R.; Pethe, W.; Hedderich, J.; Schmidt, C.; Maune, S. Endothelial and epithelial expression of eotaxin-2 (CCL24) in nasal polyps. Int. Arch. Allergy Immunol. 2006, 140, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, M.; Pawankar, R.; Fukumoto, A.; Ogihara, N.; Sakanushi, A.; Yagi, T. Induction of eotaxin production by interleukin-4, interleukin-13 and lipopolysaccharide by nasal fibroblasts. Clin. Exp. Allergy 2004, 34, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Steinke, J.W.; Crouse, C.D.; Bradley, D.; Hise, K.; Lynch, K.; Kountakis, S.E.; Borish, L. Characterization of Interleukin-4-Stimulated Nasal Polyp Fibroblasts. Am. J. Respir. Cell Mol. Biol. 2004, 30, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, S.; Tojima, I.; Takezawa, K.; Matsumoto, K.; Kouzaki, H.; Shimizu, T. Thrombin and Activated coagulation factor x stimulate the release of cytokines and fibronectin from Nasal polyp fibroblasts via proteaseactivated receptors. Am. J. Rhinol. Allergy 2017, 31, e13–e18. [Google Scholar] [CrossRef] [PubMed]
- Imoto, Y.; Ueki, S.; Kato, Y.; Yoshida, K.; Morikawa, T.; Kimura, Y.; Kidoguchi, M.; Tsutsumiuchi, T.; Koyama, K.; Adachi, N.; et al. Elevated Serum Leptin Levels in Patients with Eosinophilic Chronic Rhinosinusitis. Front. Pharmacol. 2022, 12, 793607. [Google Scholar] [CrossRef]
- Izuhara, K.; Arima, K.; Yasunaga, S. IL-4 and IL-13: Their pathological roles in allergic diseases and their potential in developing new therapies. Curr. Drug Targets Inflamm. Allergy 2002, 1, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Soyka, M.B.; Wawrzyniak, P.; Eiwegger, T.; Holzmann, D.; Treis, A.; Wanke, K.; Kast, J.I.; Akdis, C.A. Defective epithelial barrier in chronic rhinosinusitis: The regulation of tight junctions by IFN-γ and IL-4. J. Allergy Clin. Immunol. 2012, 130, 1087–1096.e10. [Google Scholar] [CrossRef]
- Lee, H.J.; Kim, D.K. Effect of Airborne Particulate Matter on the Immunologic Characteristics of Chronic Rhinosinusitis with Nasal Polyps. Int. J. Mol. Sci. 2022, 23, 1018. [Google Scholar] [CrossRef]
- Leland, E.M.; Zhang, Z.; Kelly, K.M.; Ramanathan, M. Role of Environmental Air Pollution in Chronic Rhinosinusitis. Curr. Allergy Asthma Rep. 2021, 21, 42. [Google Scholar] [CrossRef]
- Park, M.; Lee, J.S.; Park, M.K. The Effects of Air Pollutants on the Prevalence of Common Ear, Nose, and Throat Diseases in South Korea: A National Population-Based Study. Clin. Exp. Otorhinolaryngol. 2019, 12, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, M.; Fukumoto, A.; Ogihara, N.; Pawankar, R.; Sakanushi, A.; Yagi, T. Expression of MCP-4 by TLR ligand-stimulated nasal polyp fibroblasts. Acta Oto-Laryngol. 2007, 127, 1304–1309. [Google Scholar] [CrossRef] [PubMed]
- Bamba, S.; Andoh, A.; Yasui, H.; Makino, J.; Kim, S.; Fujiyama, Y. Regulation of IL-11 expression in intestinal myofibroblasts: Role of c-Jun AP-1- and MAPK-dependent pathways. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, G529–G538. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zhao, R.; Chen, H.; Jia, P.; Bao, L.; Tang, H. Bornyl acetate has an anti-inflammatory effect in human chondrocytes via induction of IL-11. IUBMB Life 2014, 66, 854–859. [Google Scholar] [CrossRef]
- Allen, J.S.; Eisma, R.; Leonard, G.; Lafreniere, D.; Kreutzer, D. Interleukin-8 expression in human nasal polyps. Otolaryngol. Head. Neck Surg. 1997, 117, 535–541. [Google Scholar] [PubMed]
- Peters, A.T.; Kato, A.; Zhang, N.; Conley, D.B.; Suh, L.; Tancowny, B.; Carter, D.; Carr, T.; Radtke, M.; Hulse, K.E.; et al. Evidence for altered activity of the IL-6 pathway in chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2010, 125, 397–403.e10. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Sun, S.; Zhang, Y.; Tang, P.; Lv, C.; Ma, H.; Yu, Y.; Xu, S.; Deng, Z. Serum IL-8 and VEGFA are Two Promising Diagnostic Biomarkers of Asthma-COPD Overlap Syndrome. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Rincon, M.; Irvin, C.G. Role of IL-6 in asthma and other inflammatory pulmonary diseases. Int. J. Biol. Sci. 2012, 8, 1281–1290. [Google Scholar] [CrossRef] [PubMed]
- Bequignon, E.; Mangin, D.; Bécaud, J.; Pasquier, J.; Angely, C.; Bottier, M.; Escudier, E.; Isabey, D.; Filoche, M.; Louis, B.; et al. Pathogenesis of chronic rhinosinusitis with nasal polyps: Role of IL-6 in airway epithelial cell dysfunction. J. Transl. Med. 2020, 18, 136. [Google Scholar] [CrossRef] [PubMed]
- Fousek, K.; Horn, L.A.; Palena, C. Interleukin-8: A chemokine at the intersection of cancer plasticity, angiogenesis, and immune suppression. Pharmacol. Ther. 2021, 219, 107692. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.M.; Hong, C.Y.; Shun, C.T.; Hsiao, T.Y.; Wang, C.C.; Wang, J.S.; Hsiao, M.; Lin, S.K. Inducible cyclooxygenase and interleukin 6 gene expressions in nasal polyp fibroblasts: Possible implication in the pathogenesis of nasal polyposis. Arch. Otolaryngol.-Head Neck Surg. 2002, 128, 945–951. [Google Scholar] [CrossRef]
- Cho, J.S.; Han, I.H.; Lee, H.R.; Lee, H.M. Prostaglandin E2 Induces IL-6 and IL-8 Production by the EP Receptors/Akt/NF-kappa B Pathways in Nasal Polyp-Derived Fibroblasts. Allergy Asthma Immunol. Res. 2014, 6, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.S.; Kang, J.H.; Um, J.Y.; Han, I.H.; Park, I.H.; Lee, H.M. Lipopolysaccharide induces pro-inflammatory cytokines and MMP production via TLR4 in nasal polyp-derived fibroblast and organ culture. PLoS ONE 2014, 9, e90683. [Google Scholar] [CrossRef]
- Shin, S.H.; Ye, M.K.; Kim, Y.H.; Kim, J.K. Role of TLRs in the production of chemical mediators in nasal polyp fibroblasts by fungi. Auris Nasus Larynx 2016, 43, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Olsson, S.; Cagnoni, F.; Dignetti, P.; Melioli, G.; Canonica, G.W. Low concentrations of cytokines produced by allergen-stimulated peripheral blood mononuclear cells have potent effects on nasal polyp-derived fibroblasts. Clin. Exp. Immunol. 2003, 132, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Shun, C.T.; Lin, S.K.; Hong, C.Y.; Huang, H.M.; Liu, C.M. Hypoxia induces cysteine-rich 61, vascular endothelial growth factor, and interleukin-8 expressions in human nasal polyp fibroblasts: An implication of neutrophils in the pathogenesis of nasal polyposis. Am. J. Rhinol. Allergy 2011, 25, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Wise, S.K.; Laury, A.M.; Katz, E.H.; Den Beste, K.A.; Parkos, C.A.; Nusrat, A. Interleukin-4 and interleukin-13 compromise the sinonasal epithelial barrier and perturb intercellular junction protein expression. Int. Forum Allergy Rhinol. 2014, 4, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Vatrella, A.; Fabozzi, I.; Calabrese, C.; Maselli, R.; Pelaia, G. Dupilumab: A novel treatment for asthma. J. Asthma Allergy 2014, 7, 123–130. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Nakajima, T.; Tsukidate, T.; Matsumoto, K.; Iida, M.; Otori, N.; Haruna, S.-I.; Moriyama, H.; Saito, H. TNF-alpha and IL-4 regulate expression of IL-13 receptor alpha2 on human fibroblasts. Biochem. Biophys. Res. Commun. 2003, 312, 1248–1255. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, S.O.; Sharma, P.; Harbor, P.C.; Aman, M.J.; Vogelbaum, M.A.; Haque, S.J. IL-13R(alpha)2, a decoy receptor for IL-13 acts as an inhibitor of IL-4-dependent signal transduction in glioblastoma cells. Cancer Res. 2002, 62, 1103–1109. [Google Scholar]
- Jiang, H.; Harris, M.B.; Rothman, P. IL-4/IL-13 signaling beyond JAK/STAT. J. Allergy Clin. Immunol. 2000, 105 Pt 1, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Murata, T.; Taguchi, J.; Puri, R.K.; Mohri, H. Sharing of receptor subunits and signal transduction pathway between the IL-4 and IL-13 receptor system. Int. J. Hematol. 1999, 69, 13–20. [Google Scholar] [PubMed]
- Fuchs, A.; Colonna, M. Innate lymphoid cells in homeostasis, infection, chronic inflammation and tumors of the gastrointestinal tract. Curr. Opin. Gastroenterol. 2013, 29, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, W.; Kolls, J.K.; Zheng, Y. The biological functions of T helper 17 cell effector cytokines in inflammation. Immunity 2008, 28, 454–467. [Google Scholar] [CrossRef]
- Pappu, R.; Ramirez-Carrozzi, V.; Sambandam, A. The interleukin-17 cytokine family: Critical players in host defence and inflammatory diseases: IL-17 cytokine family. Immunology 2011, 134, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.J.; Zhou, M.L.; Liu, Y.C.; Zhou, S.H. Roles Played by the PI3K/Akt/HIF-1 alpha Pathway and IL-17A in the Chinese Subtype of Chronic Sinusitis with Nasal Polyps. Mediat. Inflamm. 2022, 2022, 8609590. [Google Scholar] [CrossRef]
- Niu, Y.Z.; Gong, G.Q.; Chen, S.; Chen, J.J.; Kong, W.J.; Wang, Y.J. Effects of IL-17 on expression of GRO-alpha and IL-8 in fibroblasts from nasal polyps. J. Huazhong Univ. Sci. Technol.-Med. Sci. 2014, 34, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Rudack, C.; Sachse, F.; Alberty, J. Primary role of growth-related oncogene-alpha and granulocyte chemotactic protein-2 as neutrophil chemoattractants in chronic rhinosinusitis. Clin. Exp. Allergy 2006, 36, 748–759. [Google Scholar] [CrossRef]
- Homma, H.; Kamiya, K.; Kusunoki, T.; Ikeda, K. Multiplex analyses of cytokine and chemokine release from the cultured fibroblast of nasal polyps: The effect of IL-17A. Acta Oto-Laryngol. 2013, 133, 1065–1072. [Google Scholar] [CrossRef]
- Wolk, K.; Witte, E.; Wallace, E.; Döcke, W.-D.; Kunz, S.; Asadullah, K.; Volk, H.-D.; Sterry, W.; Sabat, R. IL-22 regulates the expression of genes responsible for antimicrobial defense, cellular differentiation, and mobility in keratinocytes: A potential role in psoriasis. Eur. J. Immunol. 2006, 36, 1309–1323. [Google Scholar] [CrossRef]
- Arshad, T.; Mansur, F.; Palek, R.; Manzoor, S.; Liska, V. A Double Edged Sword Role of Interleukin-22 in Wound Healing and Tissue Regeneration. Front. Immunol. 2020, 11, 2148. [Google Scholar] [CrossRef]
- Hao, D.; Wu, Y.; Li, P.; Li, C.; Jiang, T.; Zhang, Q.; Liu, S.; Shi, L. An Integrated Analysis of Inflammatory Endotypes and Clinical Characteristics in Chronic Rhinosinusitis with Nasal Polyps. JIR 2022, 15, 5557–5565. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A.; Kim, S.H. IL-32, a novel cytokine with a possible role in disease. Ann. Rheum. Dis. 2006, 65 (Suppl. S3), iii61–iii64. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Han, S.Y.; Azam, T.; Yoon, D.Y.; Dinarello, C.A. Interleukin-32: A cytokine and inducer of TNFalpha. Immunity 2005, 22, 131–142. [Google Scholar] [PubMed]
- Shoda, H.; Fujio, K.; Yamaguchi, Y.; Okamoto, A.; Sawada, T.; Kochi, Y.; Yamamoto, K. Interactions between IL-32 and tumor necrosis factor alpha contribute to the exacerbation of immune-inflammatory diseases. Arthritis Res. Ther. 2006, 8, R166. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Bae, S.; Kang, Y.; Yoon, D.; Bai, X.; Chan, E.D.; Azam, T.; Dinarello, C.A.; Lee, S.; Her, E.; et al. Suppressing IL-32 in monocytes impairs the induction of the proinflammatory cytokines TNFalpha and IL-1beta. Cytokine 2010, 49, 171–176. [Google Scholar] [CrossRef]
- Keswani, A.; Chustz, R.T.; Suh, L.; Carter, R.; Peters, A.T.; Tan, B.K.; Chandra, R.; Kim, S.H.; Azam, T.; Dinarello, C.A. Differential expression of interleukin-32 in chronic rhinosinusitis with and without nasal polyps. Allergy 2012, 67, 25–32. [Google Scholar] [CrossRef]
- Soyka, M.B.; Treis, A.; Eiwegger, T.; Menz, G.; Zhang, S.; Holzmann, D.; Akdis, C.A.; Meyer, N. Regulation and expression of IL-32 in chronic rhinosinusitis. Allergy 2012, 67, 790–798. [Google Scholar] [CrossRef]
- Cho, J.S.; Kim, J.A.; Park, J.H.; Park, I.H.; Han, I.H.; Lee, H.M. Toll-like receptor 4-mediated expression of interleukin-32 via the c-Jun N-terminal kinase/protein kinase B/cyclic adenosine monophosphate response element binding protein pathway in chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2016, 6, 1020–1028. [Google Scholar] [CrossRef]
- Mirchandani, A.S.; Salmond, R.J.; Liew, F.Y. Interleukin-33 and the function of innate lymphoid cells. Trends Immunol. 2012, 33, 389–396. [Google Scholar] [CrossRef]
- Cayrol, C.; Girard, J.P. IL-33: An alarmin cytokine with crucial roles in innate immunity, inflammation and allergy. Curr. Opin. Immunol. 2014, 31, 31–37. [Google Scholar] [CrossRef]
- Cayrol, C.; Girard, J.P. Interleukin-33 (IL-33): A nuclear cytokine from the IL-1 family. Immunol. Rev. 2018, 281, 154–168. [Google Scholar] [CrossRef]
- Angiolillo, A.L.; Sgadari, C.; Taub, D.D.; Liao, F.; Farber, J.M.; Maheshwari, S.; Kleinman, H.K.; Reaman, G.H.; Tosato, G. Human interferon-inducible protein 10 is a potent inhibitor of angiogenesis in vivo. J. Exp. Med. 1995, 182, 155–162. [Google Scholar] [CrossRef]
- Dufour, J.H.; Dziejman, M.; Liu, M.T.; Leung, J.H.; Lane, T.E.; Luster, A.D. IFN-γ-Inducible Protein 10 (IP-10; CXCL10)-Deficient Mice Reveal a Role for IP-10 in Effector T Cell Generation and Trafficking. J. Immunol. 2002, 168, 3195–3204. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Wada, K.; Yoshimura, T.; Asaka, D.; Okada, N.; Matsumoto, K.; Moriyama, H. Increased CXCL10 expression in nasal fibroblasts from patients with refractory chronic rhinosinusitis and asthma. Allergol. Int. 2013, 62, 495–502. [Google Scholar] [CrossRef]
- Li, Z.; Levast, B.; Madrenas, J. Staphylococcus aureus Downregulates IP-10 Production and Prevents Th1 Cell Recruitment. J. Immunol. 2017, 198, 1865–1874. [Google Scholar] [CrossRef]
- Nam, Y.R.; Lee, K.J.; Lee, H.; Joo, C.H. CXCL10 production induced by high levels of IKKε in nasal airway epithelial cells in the setting of chronic inflammation. Biochem. Biophys. Res. Commun. 2019, 514, 607–612. [Google Scholar] [CrossRef]
- Evers, T.M.J.; Sheikhhassani, V.; Haks, M.C.; Storm, C.; Ottenhoff, T.H.M.; Mashaghi, A. Single-cell analysis reveals chemokine-mediated differential regulation of monocyte mechanics. iScience 2022, 25, 103555. [Google Scholar] [CrossRef]
- Shun, C.T.; Lin, S.K.; Hong, C.Y.; Kok, S.H.; Juan, Y.H.; Wang, C.C.; Hsu, M.C.; Liu, C.M. CC chemokine ligand 2 gene expression in nasal polyp fibroblasts: Possible implication in the pathogenesis of nasal polyposis. Ann. Otol. Rhinol. Laryngol. 2005, 114, 879–885. [Google Scholar] [CrossRef]
- Lin, S.-K.; Kok, S.-H.; Shun, C.-T.; Hong, C.-Y.; Wang, C.-C.; Hsu, M.-C.; Liu, C.-M. Tumor necrosis factor-alpha stimulates the expression of C-C chemokine ligand 2 gene in fibroblasts from the human nasal polyp through the pathways of mitogen-activated protein kinase. Am. J. Rhinol. 2007, 21, 251–255. [Google Scholar] [CrossRef]
- Pujols, L.; Mullol, J.; Alobid, I.; Roca-Ferrer, J.; Xaubet, A.; Picado, C. Dynamics of COX-2 in nasal mucosa and nasal polyps from aspirin-tolerant and aspirin-intolerant patients with asthma. J. Allergy Clin. Immunol. 2004, 114, 814–819. [Google Scholar] [CrossRef]
- Tanaka, S.; Tatsuguchi, A.; Futagami, S.; Gudis, K.; Wada, K.; Seo, T.; Mitsui, K.; Yonezawa, M.; Nagata, K.; Fujimori, S.; et al. Monocyte chemoattractant protein 1 and macrophage cyclooxygenase 2 expression in colonic adenoma. Gut 2006, 55, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Imai, T.; Baba, M.; Nishimura, M.; Kakizaki, M.; Takagi, S.; Yoshie, O. The T cell-directed CC chemokine TARC is a highly specific biological ligand for CC chemokine receptor 4. J. Biol. Chem. 1997, 272, 15036–15042. [Google Scholar] [CrossRef] [PubMed]
- Fukumoto, A.; Nonaka, M.; Ogihara, N.; Pawankar, R. Induction of TARC production by lipopolysaccharide and interleukin-4 in nasal fibroblasts. Int. Arch. Allergy Immunol. 2008, 145, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, M.; Ogihara, N.; Fukumoto, A.; Sakanushi, A.; Pawankar, R.; Yagi, T. Toll-like receptor 2, 3, 4, 5 ligands and interleukin-4 synergistically induce TARC production in nasal polyp fibroblasts. Auris Nasus Larynx 2008, 35, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, M.; Ogihara, N.; Fukumoto, A.; Sakanushi, A.; Kusama, K.; Pawankar, R.; Yagi, T. Combined stimulation with Poly(I:C), TNF-alpha and Th2 cytokines induces TARC production by human fibroblasts from the nose, bronchioles and lungs. Int. Arch. Allergy Immunol. 2010, 152, 327–341. [Google Scholar] [CrossRef]
- Nonaka, M.; Ogihara, N.; Fukumoto, A.; Sakanushi, A.; Pawankar, R.; Yagi, T. Combined stimulation of nasal polyp fibroblasts with poly IC, interleukin 4, and tumor necrosis factor α potently induces production of thymus- and activation-regulated chemokine. Arch. Otolaryngol.-Head. Neck Surg. 2008, 134, 630–635. [Google Scholar] [CrossRef]
- Hieshima, K.; Imai, T.; Opdenakker, G.; Van Damme, J.; Kusuda, J.; Tei, H.; Sakaki, Y.; Takatsuki, K.; Miura, R.; Yoshie, O.; et al. Molecular cloning of a novel human CC chemokine liver and activation-regulated chemokine (LARC) expressed in liver. Chemotactic activity for lymphocytes and gene localization on chromosome 2. J Biol Chem. 1997, 272, 5846–5853. [Google Scholar] [CrossRef]
- Schutyser, E.; Struyf, S.; Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 2003, 14, 409–426. [Google Scholar] [CrossRef]
- van Tongeren, J.; Reinartz, S.M.; Fokkens, W.J.; de Jong, E.C.; van Drunen, C.M. Interactions between epithelial cells and dendritic cells in airway immune responses: Lessons from allergic airway disease. Allergy 2008, 63, 1124–1135. [Google Scholar] [CrossRef]
- Schutyser, E.; Struyf, S.; Menten, P.; Lenaerts, J.-P.; Conings, R.; Put, W.; Wuyts, A.; Proost, P.; Van Damme, J. Regulated production and molecular diversity of human liver and activation-regulated chemokine/macrophage inflammatory protein-3 alpha from normal and transformed cells. J. Immunol. 2000, 165, 4470–4477. [Google Scholar] [CrossRef]
- Nonaka, M.; Ogihara, N.; Fukumoto, A.; Sakanushi, A.; Kusama, K.; Pawankar, R.; Yagi, T. Synergistic Induction of Macrophage Inflammatory Protein-3 alpha/CCL20 Production by Interleukin-17A and Tumor Necrosis Factor-alpha in Nasal Polyp Fibroblasts. World Allergy Organ. J. 2009, 2, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, S.F.; Artis, D. Sensing the outside world: TSLP regulates barrier immunity. Nat. Immunol. 2010, 11, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.N.; Kohanski, M.A.; Maina, I.W.; Workman, A.D.; Herbert, D.R.; Cohen, N.A. Sentinels at the wall: Epithelial-derived cytokines serve as triggers of upper airway type 2 inflammation: Epithelial cytokines in upper airway. Int. Forum Allergy Rhinol. 2019, 9, 93–99. [Google Scholar] [CrossRef]
- Nonaka, M.; Fukumoto, A.; Ogihara, N.; Sakanushi, A.; Pawankar, R.; Yagi, T. Synergistic induction of thymic stromal lymphopoietin by tumor necrosis factor alpha and Th2 cytokine in nasal polyp fibroblasts. Am. J. Rhinol. Allergy 2010, 24, e14–e18. [Google Scholar] [CrossRef]
- Pan, J.; Zhang, M.; Wang, J.; Wang, Q.; Xia, D.; Sun, W.; Zhang, L.; Yu, H.; Liu, Y.; Cao, X. Interferon-gamma is an autocrine mediator for dendritic cell maturation. Immunol. Lett. 2004, 94, 141–151. [Google Scholar] [CrossRef]
- Shin, S.H.; Kim, Y.H.; Jin, H.S.; Kang, S.H. Alternaria Induces Production of Thymic Stromal Lymphopoietin in Nasal Fibroblasts Through Toll-like Receptor 2. Allergy Asthma Immunol. Res. 2016, 8, 63–68. [Google Scholar] [CrossRef]
- Kabata, H.; Moro, K.; Koyasu, S. The group 2 innate lymphoid cell (ILC2) regulatory network and its underlying mechanisms. Immunol. Rev. 2018, 286, 37–52. [Google Scholar] [CrossRef]
- Rostkowska-Nadolska, B.; Latocha, M.; Gawron, W.; Kutner, A.; Bochnia, M. The influence of calcitriol and tacalcitol on proliferation of fibroblasts cultured from nasal polyps. Adv. Clin. Exp. Med. 2007, 16, 213–219. [Google Scholar]
- Carsuzaa, F.; Béquignon, É.; Bainaud, M.; Jégou, J.F.; Dufour, X.; Lecron, J.C.; Favot, L. Oncostatin M Counteracts the Fibrotic Effects of TGF-β1 and IL-4 on Nasal-Polyp-Derived Fibroblasts: A Control of Fibrosis in Chronic Rhinosinusitis with Nasal Polyps? Int. J. Mol. Sci. 2022, 23, 6308. [Google Scholar] [CrossRef]
- Carsuzaa, F.; Béquignon, É.; Dufour, X.; de Bonnecaze, G.; Lecron, J.C.; Favot, L. Cytokine Signature and Involvement in Chronic Rhinosinusitis with Nasal Polyps. IJMS 2021, 23, 417. [Google Scholar] [CrossRef]
- Park, S.A.; Park, I.H.; Cho, J.S.; Moon, Y.M.; Lee, S.H.; Kim, T.H.; Lee, S.H.; Lee, H.M. Effect of [6]-Gingerol on Myofibroblast Differentiation in Transforming Growth Factor Beta 1–Induced Nasal polyp–Derived Fibroblasts. Am. J. Rhinol. Allergy 2012, 26, 97–103. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Yan, D.; Wang, H.W.; Bowman, R.L.; Joyce, J.A. STAT3 and STAT6 Signaling Pathways Synergize to Promote Cathepsin Secretion from Macrophages via IRE1α Activation. Cell Rep. 2016, 16, 2914–2927. [Google Scholar] [CrossRef]
- McCain, J. The MAPK (ERK) Pathway: Investigational Combinations for the Treatment Of BRAF-Mutated Metastatic Melanoma. Pharm. Ther. 2013, 38, 96–108. [Google Scholar]
- Nemec, C.M.; Singh, A.K.; Ali, A.; Tseng, S.C.; Syal, K.; Ringelberg, K.J.; Ho, Y.H.; Hintermair, C.; Ahmad, M.F.; Kar, R.K.; et al. Noncanonical CTD kinases regulate RNA polymerase II in a gene-class-specific manner. Nat. Chem. Biol. 2019, 15, 123–131. [Google Scholar] [CrossRef]
- Hong, C.J.; Tsang, A.C.; Quinn, J.G.; Bonaparte, J.P.; Stevens, A.; Kilty, S.J. Anti-IgE monoclonal antibody therapy for the treatment of chronic rhinosinusitis: A systematic review. Syst. Rev. 2015, 4, 166. [Google Scholar] [CrossRef]
- Franzese, C.B. The Role of Biologics in the Treatment of Nasal Polyps. Immunol. Allergy Clin. 2020, 40, 295–302. [Google Scholar] [CrossRef]
- Patel, G.B.; Peters, A.T. The Role of Biologics in Chronic Rhinosinusitis with Nasal Polyps. Ear Nose Throat J. 2021, 100, 44–47. [Google Scholar] [CrossRef]
- Moreno-Luna, R.; González-García, J.; Palacios-García, J.; Maza-Solano, J.M.; del Cuvillo Bernal, A.; Sánchez-Gómez, S. Usefulness of endonasal mucoplasty in the surgical treatment of chronic rhinosinusitis with nasal polyps. Prospective study [Utilidad de la mucoplastia endonasal en el tratamiento quirúrgico de la rinosinusitis crónica con pólipos nasales. Estudio prospectivo]. Acta Otorrinolaringol. Esp. 2021, 72, 256–261. [Google Scholar]
- Moreno-Luna, R.; Martin-Jimenez, D.I.; Callejon-Leblic, M.A.; Gonzalez-Garcia, J.; Maza-Solano, J.M.; Porras-Gonzalez, C.; Del Cuvillo-Bernal, A.; Sanchez-Gomez, S. Usefulness of bilateral mucoplasty plus reboot surgery in severe type-2 chronic rhinosinusitis with nasal polyps. Rhinology 2022, 60, 368–376. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).