Acute Respiratory Distress Syndrome and Fluid Management: Finding the Perfect Balance
Abstract
1. Introduction
2. Pathophysiology
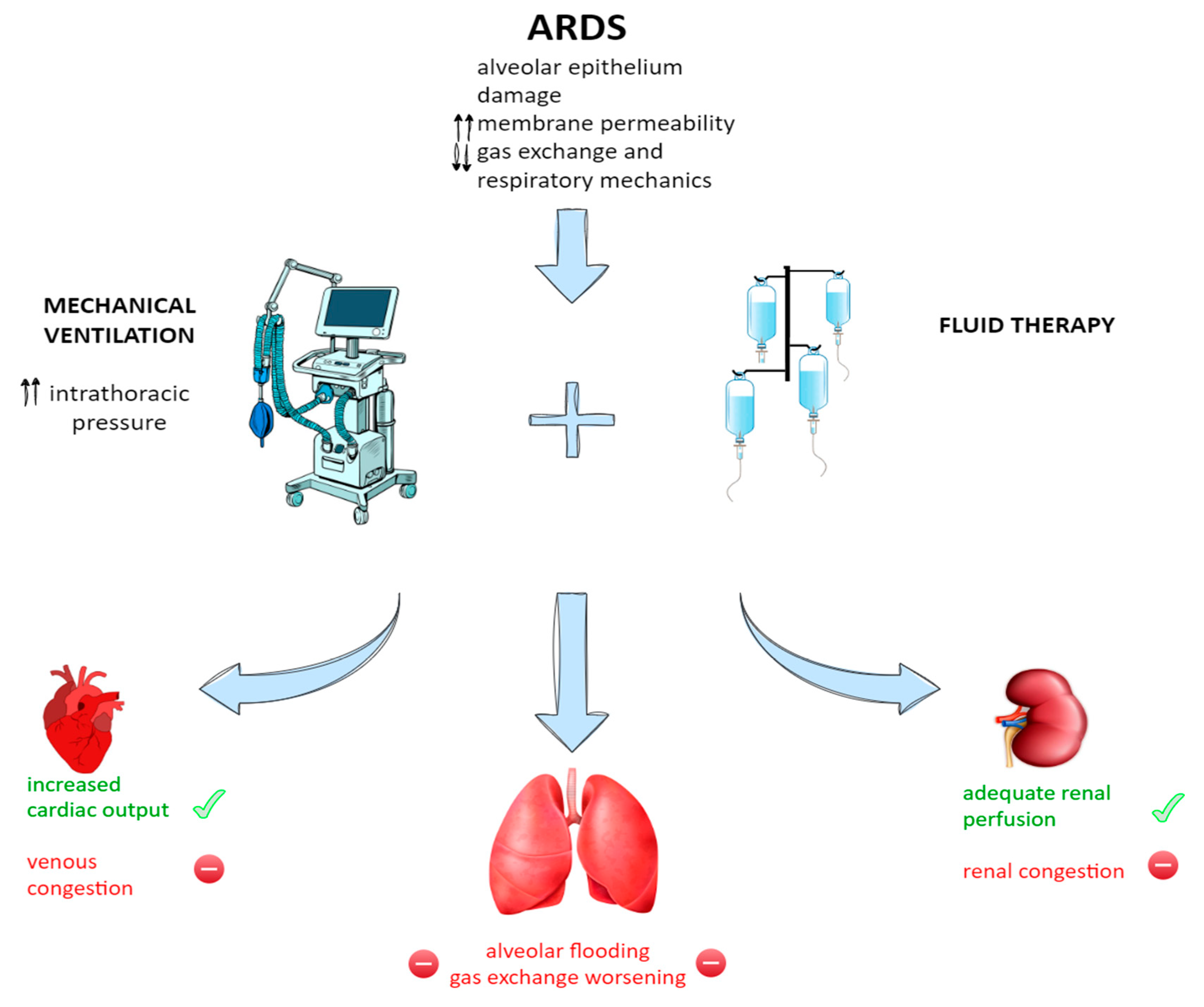
2.1. Impact of Restrictive Versus Liberal Fluid Management on Lung and Distal Organ Damage
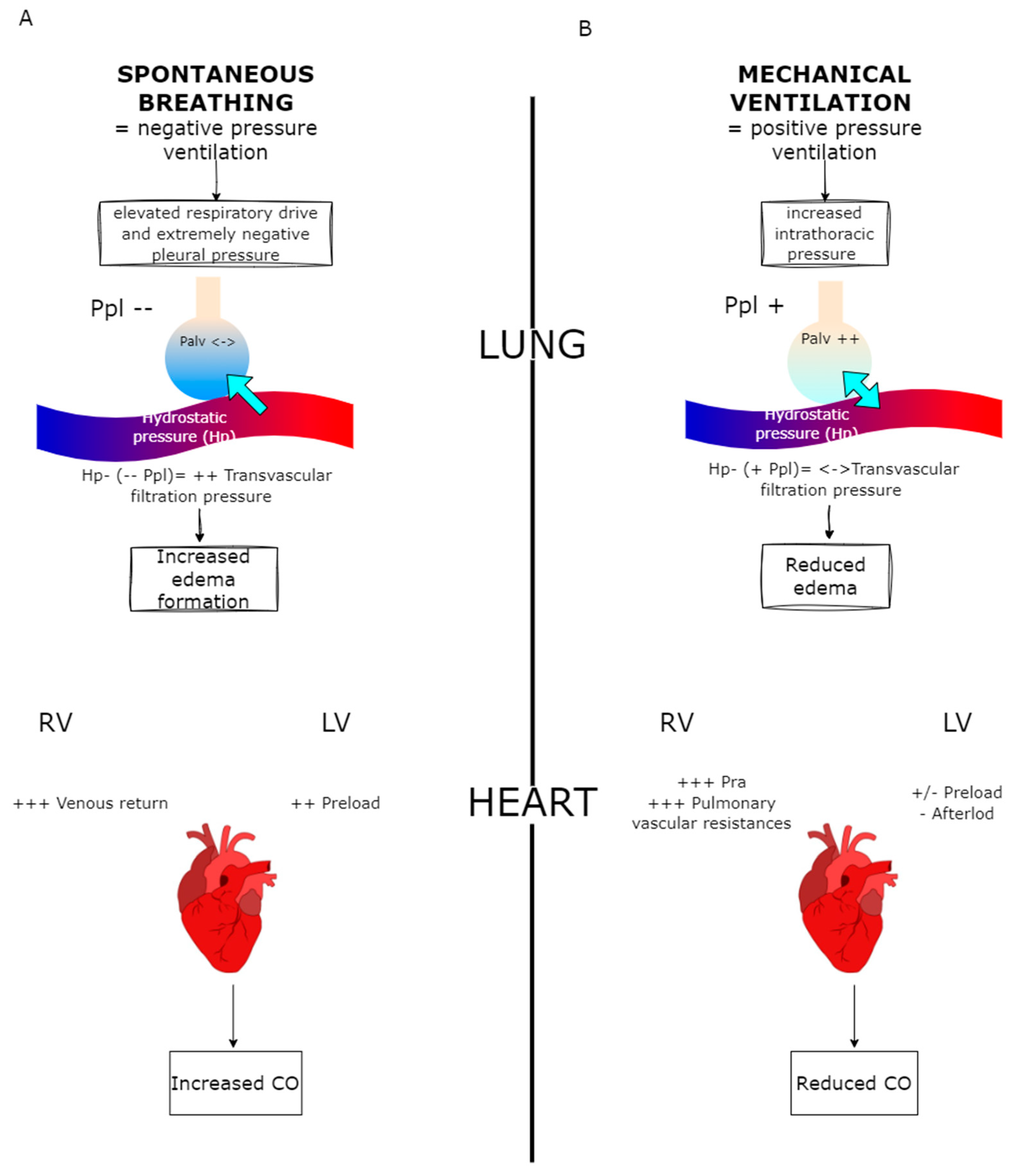
2.2. Heart–Lung Interactions During Spontaneous Breathing and Positive Pressure Ventilation
2.3. The Effects of PEEP
3. Discussion
3.1. Liberal vs. Restrictive Strategies in Clinical Studies
3.2. Practical Management and Fluid Therapy Monitoring
3.3. Fluid Responsiveness
4. Conclusions
5. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ARDS | Acute respiratory distress syndrome |
| ANP | Atrial natriuretic peptide |
| VILI | Ventilator-induced lung injury |
| AKI | Acute kidney injury |
| Pmsf | Mean systemic filling pressure |
| Pra | Right atrial pressure |
| PEEP | Positive end-expiratory pressure |
| RAAS | Renin–angiotensin–aldosterone system |
| EVLW | Extra-vascular lung water |
| SIRS | Systemic inflammatory response syndrome |
| TPTD | Transpulmonary thermodilution |
| CCE | Critical care echocardiography |
| PAC | Pulmonary artery catheter |
| FR | Fluid responsiveness |
| SVV | Stroke volume variation |
| PPV | Pulse pressure variation |
| CO | Cardiac output |
| PLR | Passive leg raising |
| EEO | End-expiratory occlusion test |
| PVPI | Pulmonary vascular permeability index |
| CI | Cardiac index |
| SVC | Superior vena cava |
| IVC | Inferior vena cava |
References
- Matthay, M.A.; Zemans, R.L.; Zimmerman, G.A.; Arabi, Y.M.; Beitler, J.R.; Mercat, A.; Herridge, M.; Randolph, A.G.; Calfee, C.S. Acute respiratory distress syndrome. Nat. Rev. Dis. Primers 2019, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Bos, L.D.J.; Ware, L.B. Acute respiratory distress syndrome: Causes, pathophysiology, and phenotypes. Lancet 2022, 400, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.N.; Haeger, S.M.; Oshima, K.; Yang, Y.; Wallbank, A.M.; Jin, Y.; Lettau, M.; McCaig, L.A.; Wickersham, N.E.; McNeil, J.B.; et al. Alveolar epithelial glycocalyx degradation mediates surfactant dysfunction and contributes to acute respiratory distress syndrome. JCI Insight 2022, 7, e154573. [Google Scholar] [CrossRef]
- Chappell, D.; Jacob, M. Role of the glycocalyx in fluid management: Small things matter. Best. Pr. Res. Clin. Anaesthesiol. 2014, 28, 227–234. [Google Scholar] [CrossRef]
- Chappell, D.; Bruegger, D.; Potzel, J.; Jacob, M.; Brettner, F.; Vogeser, M.; Conzen, P.; Becker, B.F.; Rehm, M. Hypervolemia increases release of atrial natriuretic peptide and shedding of the endothelial glycocalyx. Crit. Care 2014, 18, 538. [Google Scholar] [CrossRef]
- Guidet, B.; Ait-Oufella, H. Fluid resuscitation should respect the endothelial glycocalyx layer. Crit. Care 2014, 18, 707. [Google Scholar] [CrossRef]
- Marjanovic, G.; Villain, C.; Juettner, E.; zur Hausen, A.; Hoeppner, J.; Hopt, U.T.; Drognitz, O.; Obermaier, R. Impact of different crystalloid volume regimes on intestinal anastomotic stability. Ann. Surg. 2009, 249, 181–185. [Google Scholar] [CrossRef]
- Messmer, A.S.; Zingg, C.; Müller, M.; Gerber, J.L.; Schefold, J.C.; Pfortmueller, C.A. Fluid Overload and Mortality in Adult Critical Care Patients-A Systematic Review and Meta-Analysis of Observational Studies. Crit. Care Med. 2020, 48, 1862–1870. [Google Scholar] [CrossRef]
- Carvalho, E.B.; Fonseca, A.C.F.; Magalhães, R.F.; Pinto, E.F.; Samary, C.S.; Antunes, M.A.; Baldavira, C.M.; Silveira, L.K.R.; Teodoro, W.R.; de Abreu, M.G.; et al. Effects of different fluid management on lung and kidney during pressure-controlled and pressure-support ventilation in experimental acute lung injury. Physiol. Rep. 2022, 10, e15429. [Google Scholar] [CrossRef]
- Gattarello, S.; Pozzi, T.; Galizia, M.; Busana, M.; Ghidoni, V.; Catozzi, G.; Donati, B.; Nocera, D.; Giovanazzi, S.; D’Albo, R.; et al. Impact of Fluid Balance on the Development of Lung Injury. Am. J. Respir. Crit. Care Med. 2025, 211, 331–338. [Google Scholar] [CrossRef]
- Slutsky, A.S.; Ranieri, V.M. Ventilator-induced lung injury. N. Engl. J. Med. 2013, 369, 2126–2136. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Crichton, S.; Dixon, A.; Seylanova, N.; Peng, Z.Y.; Ostermann, M. Cumulative fluid accumulation is associated with the development of acute kidney injury and non-recovery of renal function: A retrospective analysis. Crit. Care Lond. Engl. 2019, 23, 392. [Google Scholar] [CrossRef] [PubMed]
- Lankadeva, Y.R.; Kosaka, J.; Iguchi, N.; Evans, R.G.; Booth, L.C.; Bellomo, R.; May, C.N. Effects of Fluid Bolus Therapy on Renal Perfusion, Oxygenation, and Function in Early Experimental Septic Kidney Injury. Crit. Care Med. 2019, 47, e36–e43. [Google Scholar] [CrossRef] [PubMed]
- Kreimeier, U. Pathophysiology of fluid imbalance. Crit. Care Lond. Engl. 2000, 4 (Suppl. 2), S3–S7. [Google Scholar] [CrossRef]
- Bakker, J.; Nijsten, M.W.; Jansen, T.C. Clinical use of lactate monitoring in critically ill patients. Ann. Intensive Care 2013, 3, 12. [Google Scholar] [CrossRef]
- Fuller, B.M.; Dellinger, R.P. Lactate as a hemodynamic marker in the critically ill. Curr. Opin. Crit. Care 2012, 18, 267–272. [Google Scholar] [CrossRef]
- Roy, T.K.; Secomb, T.W. Effects of impaired microvascular flow regulation on metabolism-perfusion matching and organ function. Microcirculation 2021, 28, e12673. [Google Scholar] [CrossRef]
- Cortes-Puentes, G.A.; Oeckler, R.A.; Marini, J.J. Physiology-guided management of hemodynamics in acute respiratory distress syndrome. Ann. Transl. Med. 2018, 6, 353. [Google Scholar] [CrossRef]
- Guyton, A.C.; Lindsey, A.W.; Abernathy, B.; Richardson, T. Venous return at various right atrial pressures and the normal venous return curve. Am. J. Physiol. 1957, 189, 609–615. [Google Scholar] [CrossRef]
- Grübler, M.R.; Wigger, O.; Berger, D.; Blöchlinger, S. Basic concepts of heart-lung interactions during mechanical ventilation. Swiss Med. Wkly. 2017, 147, w14491. [Google Scholar] [CrossRef]
- Mahmood, S.S.; Pinsky, M.R. Heart-lung interactions during mechanical ventilation: The basics. Ann. Transl. Med. 2018, 6, 349. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Torsani, V.; Gomes, S.; De Santis, R.R.; Beraldo, M.A.; Costa, E.L.V.; Tucci, M.R.; Zin, W.A.; Kavanagh, B.P.; Amato, M.B.P. Spontaneous effort causes occult pendelluft during mechanical ventilation. Am. J. Respir. Crit. Care Med. 2013, 188, 1420–1427. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Marini, J.J.; Collino, F.; Maiolo, G.; Rapetti, F.; Tonetti, T.; Vasques, F.; Quintel, M. The future of mechanical ventilation: Lessons from the present and the past. Crit. Care 2017, 21, 183. [Google Scholar] [CrossRef]
- Lansdorp, B.; Hofhuizen, C.; van Lavieren, M.; van Swieten, H.; Lemson, J.; van Putten, M.J.A.M.; van der Hoeven, J.G.; Pickkers, P. Mechanical ventilation-induced intrathoracic pressure distribution and heart-lung interactions. Crit. Care. Med. 2014, 42, 1983–1990. [Google Scholar] [CrossRef]
- de Carvalho, E.B.; Battaglini, D.; Robba, C.; Malbrain, M.L.N.G.; Pelosi, P.; Rocco, P.R.M.; Silva, P.L. Fluid management strategies and their interaction with mechanical ventilation: From experimental studies to clinical practice. Intensive Care Med. Exp. 2023, 11, 44. [Google Scholar] [CrossRef]
- Gattarello, S.; Lombardo, F.; Romitti, F.; D’Albo, R.; Velati, M.; Fratti, I.; Pozzi, T.; Nicolardi, R.; Fioccola, A.; Busana, M.; et al. Determinants of acute kidney injury during high-power mechanical ventilation: Secondary analysis from experimental data. Intensive Care Med. Exp. 2024, 12, 31. [Google Scholar] [CrossRef]
- Annat, G.; Viale, J.P.; Bui Xuan, B.; Hadj Aissa, O.; Benzoni, D.; Vincent, M.; Gharib, C.; Motin, J. Effect of PEEP ventilation on renal function, plasma renin, aldosterone, neurophysins and urinary ADH, and prostaglandins. Anesthesiology 1983, 58, 136–141. [Google Scholar] [CrossRef]
- Beyer, J.; Conzen, P.; Schosser, R.; Messmer, K. The effect of PEEP ventilation on hemodynamics and regional blood flow with special regard to coronary blood flow. Thorac. Cardiovasc. Surg. 1980, 28, 128–132. [Google Scholar] [CrossRef]
- Sha, M.; Saito, Y.; Yokoyama, K.; Sawa, T.; Amaha, K. Effects of continuous positive-pressure ventilation on hepatic blood flow and intrahepatic oxygen delivery in dogs. Crit. Care Med. 1987, 15, 1040–1043. [Google Scholar] [CrossRef]
- Felix, N.S.; Maia, L.A.; Rocha, N.N.; Rodrigues, G.C.; Medeiros, M.; da Silva, L.A.; Baldavira, C.M.; Fernezlian, S.M.; Eher, E.M.; Capelozzi, V.L.; et al. Biological impact of restrictive and liberal fluid strategies at low and high PEEP levels on lung and distal organs in experimental acute respiratory distress syndrome. Front. Physiol. 2022, 13, 992401. [Google Scholar] [CrossRef]
- Rocha, N.N.; Samary, C.S.; Antunes, M.A.; Oliveira, M.V.; Hemerly, M.R.; Santos, P.S.; Capelozzi, V.L.; Cruz, F.F.; Marini, J.J.; Silva, P.L.; et al. The impact of fluid status and decremental PEEP strategy on cardiac function and lung and kidney damage in mild-moderate experimental acute respiratory distress syndrome. Respir. Res. 2021, 22, 214. [Google Scholar] [CrossRef] [PubMed]
- Simmons, R.S.; Berdine, G.G.; Seidenfeld, J.J.; Prihoda, T.J.; Harris, G.D.; Smith, J.D.; Gilbert, T.J.; Mota, E.; Johanson, W.G. Fluid balance and the adult respiratory distress syndrome. Am. Rev. Respir. Dis. 1987, 135, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, H.; Hall, J.; Sznajder, I.; Silverstein, M.; Wood, L. Improved survival in ARDS patients associated with a reduction in pulmonary capillary wedge pressure. Chest 1990, 97, 1176–1180. [Google Scholar] [CrossRef]
- Mitchell, J.P.; Schuller, D.; Calandrino, F.S.; Schuster, D.P. Improved outcome based on fluid management in critically ill patients requiring pulmonary artery catheterization. Am. Rev. Respir. Dis. 1992, 145, 990–998. [Google Scholar] [CrossRef]
- Sakr, Y.; Vincent, J.-L.; Reinhart, K.; Groeneveld, J.; Michalopoulos, A.; Sprung, C.L.; Artigas, A.; Ranieri, V.M. High Tidal Volume and Positive Fluid Balance Are Associated with Worse Outcome in Acute Lung Injury. Chest 2005, 128, 3098–3108. [Google Scholar] [CrossRef]
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann, H.P.; Wheeler, A.P.; Bernard, G.R.; Thompson, B.T.; Hayden, D.; de Boisblanc, B.; Connors, A.F.; Hite, R.D.; Harabin, A.L. Comparison of two fluid-management strategies in acute lung injury. N. Engl. J. Med. 2006, 354, 2564–2575. [Google Scholar] [CrossRef]
- Stewart, R.M.; Park, P.K.; Hunt, J.P.; McIntyre, R.C.; McCarthy, J.; Zarzabal, L.A.; Michalek, J.E.; National Institutes of Health/National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome Clinical Trials Network. Less is more: Improved outcomes in surgical patients with conservative fluid administration and central venous catheter monitoring. J. Am. Coll. Surg. 2009, 208, 725–735, discussion 735–737. [Google Scholar] [CrossRef]
- Liu, K.D.; Thompson, B.T.; Ancukiewicz, M.; Steingrub, J.S.; Douglas, I.S.; Matthay, M.A.; Wright, P.; Peterson, M.W.; Rock, P.; Hyzy, R.C.; et al. Acute kidney injury in patients with acute lung injury: Impact of fluid accumulation on classification of acute kidney injury and associated outcomes. Crit. Care Med. 2011, 39, 2665–2671. [Google Scholar] [CrossRef]
- Grams, M.E.; Estrella, M.M.; Coresh, J.; Brower, R.G.; Liu, K.D. Fluid Balance, Diuretic Use, and Mortality in Acute Kidney Injury. Clin. J. Am. Soc. Nephrol. 2011, 6, 966–973. [Google Scholar] [CrossRef]
- Mikkelsen, M.E.; Christie, J.D.; Lanken, P.N.; Biester, R.C.; Thompson, B.T.; Bellamy, S.L.; Localio, A.R.; Demissie, E.; Hopkins, R.O.; Angus, D.C. The Adult Respiratory Distress Syndrome Cognitive Outcomes Study: Long-Term Neuropsychological Function in Survivors of Acute Lung Injury. Am. J. Respir. Crit. Care Med. 2012, 185, 1307–1315. [Google Scholar] [CrossRef]
- Famous, K.R.; Delucchi, K.; Ware, L.B.; Kangelaris, K.N.; Liu, K.D.; Thompson, B.T.; Calfee, C.S. ARDS Network Acute Respiratory Distress Syndrome Subphenotypes Respond Differently to Randomized Fluid Management Strategy. Am. J. Respir. Crit. Care Med. 2017, 195, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, A.L.; Dechert, R.E.; Park, P.K.; Bartlett, R.H.; NIH NHLBI ARDS Network. Review of A Large Clinical Series: Association of Cumulative Fluid Balance on Outcome in Acute Lung Injury: A Retrospective Review of the ARDSnet Tidal Volume Study Cohort. J. Intensive Care Med. 2009, 24, 35–46. [Google Scholar] [CrossRef] [PubMed]
- van Mourik, N.; Metske, H.A.; Hofstra, J.J.; Binnekade, J.M.; Geerts, B.F.; Schultz, M.J.; Vlaar, A.P.J. Cumulative fluid balance predicts mortality and increases time on mechanical ventilation in ARDS patients: An observational cohort study. PLoS ONE 2019, 14, e0224563. [Google Scholar] [CrossRef] [PubMed]
- Silversides, J.A.; Fitzgerald, E.; Manickavasagam, U.S.; Lapinsky, S.E.; Nisenbaum, R.; Hemmings, N.; Nutt, C.; Trinder, T.J.; Pogson, D.G.; Fan, E.; et al. Deresuscitation of Patients with Iatrogenic Fluid Overload Is Associated with Reduced Mortality in Critical Illness. Crit. Care Med. 2018, 46, 1600–1607. [Google Scholar] [CrossRef]
- Silversides, J.A.; Major, E.; Ferguson, A.J.; Mann, E.E.; McAuley, D.F.; Marshall, J.C.; Blackwood, B.; Fan, E. Conservative fluid management or deresuscitation for patients with sepsis or acute respiratory distress syndrome following the resuscitation phase of critical illness: A systematic review and meta-analysis. Intensive Care Med. 2017, 43, 155–170. [Google Scholar] [CrossRef]
- Martin, G.S.; Mangialardi, R.J.; Wheeler, A.P.; Dupont, W.D.; Morris, J.A.; Bernard, G.R. Albumin and furosemide therapy in hypoproteinemic patients with acute lung injury. Crit. Care Med. 2002, 30, 2175–2182. [Google Scholar] [CrossRef]
- Martin, G.S.; Moss, M.; Wheeler, A.P.; Mealer, M.; Morris, J.A.; Bernard, G.R. A randomized, controlled trial of furosemide with or without albumin in hypoproteinemic patients with acute lung injury. Crit. Care Med. 2005, 33, 1681–1687. [Google Scholar] [CrossRef]
- Callum, J.; Skubas, N.J.; Bathla, A.; Keshavarz, H.; Clark, E.G.; Rochwerg, B.; Fergusson, D.; Arbous, S.; Bauer, S.R.; China, L.; et al. Use of Intravenous Albumin. Chest 2024, 166, 321–338. [Google Scholar] [CrossRef]
- Malbrain, M.L.N.G.; Van Regenmortel, N.; Saugel, B.; De Tavernier, B.; Van Gaal, P.-J.; Joannes-Boyau, O.; Teboul, J.-L.; Rice, T.W.; Mythen, M.; Monnet, X. Principles of fluid management and stewardship in septic shock: It is time to consider the four D’s and the four phases of fluid therapy. Ann. Intensive Care 2018, 8, 66. [Google Scholar] [CrossRef]
- Vignon, P.; Evrard, B.; Asfar, P.; Busana, M.; Calfee, C.S.; Coppola, S.; Demiselle, J.; Geri, G.; Jozwiak, M.; Martin, G.S.; et al. Fluid administration and monitoring in ARDS: Which management? Intensive Care Med. 2020, 46, 2252–2264. [Google Scholar] [CrossRef]
- Jozwiak, M.; Teboul, J.-L. Heart-Lungs interactions: The basics and clinical implications. Ann. Intensive Care 2024, 14, 122. [Google Scholar] [CrossRef] [PubMed]
- Cecconi, M.; De Backer, D.; Antonelli, M.; Beale, R.; Bakker, J.; Hofer, C.; Jaeschke, R.; Mebazaa, A.; Pinsky, M.R.; Teboul, J.L.; et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014, 40, 1795–1815. [Google Scholar] [CrossRef] [PubMed]
- Monnet, X.; Shi, R.; Teboul, J.-L. Prediction of fluid responsiveness. What’s New? Ann. Intensive Care 2022, 12, 46. [Google Scholar] [CrossRef]
- Guérin, L.; Teboul, J.-L.; Persichini, R.; Dres, M.; Richard, C.; Monnet, X. Effects of passive leg raising and volume expansion on mean systemic pressure and venous return in shock in humans. Crit. Care 2015, 19, 411. [Google Scholar] [CrossRef]
- Yonis, H.; Bitker, L.; Aublanc, M.; Perinel Ragey, S.; Riad, Z.; Lissonde, F.; Louf-Durier, A.; Debord, S.; Gobert, F.; Tapponnier, R.; et al. Change in cardiac output during Trendelenburg maneuver is a reliable predictor of fluid responsiveness in patients with acute respiratory distress syndrome in the prone position under protective ventilation. Crit. Care 2017, 21, 295. [Google Scholar] [CrossRef]
- Jozwiak, M.; Monnet, X.; Teboul, J.-L. Prediction of fluid responsiveness in ventilated patients. Ann. Transl. Med. 2018, 6, 352. [Google Scholar] [CrossRef]
- Messina, A.; Dell’Anna, A.; Baggiani, M.; Torrini, F.; Maresca, G.M.; Bennett, V.; Saderi, L.; Sotgiu, G.; Antonelli, M.; Cecconi, M. Functional hemodynamic tests: A systematic review and a metanalysis on the reliability of the end-expiratory occlusion test and of the mini-fluid challenge in predicting fluid responsiveness. Crit. Care 2019, 23, 264. [Google Scholar] [CrossRef]
- Lai, C.; Shi, R.; Beurton, A.; Moretto, F.; Ayed, S.; Fage, N.; Gavelli, F.; Pavot, A.; Dres, M.; Teboul, J.L.; et al. The increase in cardiac output induced by a decrease in positive end-expiratory pressure reliably detects volume responsiveness: The PEEP-test study. Crit. Care 2023, 27, 136. [Google Scholar] [CrossRef]
- Jozwiak, M.; Silva, S.; Persichini, R.; Anguel, N.; Osman, D.; Richard, C.; Teboul, J.-L.; Monnet, X. Extravascular lung water is an independent prognostic factor in patients with acute respiratory distress syndrome. Crit. Care Med. 2013, 41, 472–480. [Google Scholar] [CrossRef]
- Mahjoub, Y.; Pila, C.; Friggeri, A.; Zogheib, E.; Lobjoie, E.; Tinturier, F.; Galy, C.; Slama, M.; Dupont, H. Assessing fluid responsiveness in critically ill patients: False-positive pulse pressure variation is detected by Doppler echocardiographic evaluation of the right ventricle. Crit. Care Med. 2009, 37, 2570–2575. [Google Scholar] [CrossRef]
- Vieillard-Baron, A.; Chergui, K.; Rabiller, A.; Peyrouset, O.; Page, B.; Beauchet, A.; Jardin, F. Superior vena caval collapsibility as a gauge of volume status in ventilated septic patients. Intensive Care Med. 2004, 30, 1734–1739. [Google Scholar] [CrossRef] [PubMed]
- Feissel, M.; Michard, F.; Mangin, I.; Ruyer, O.; Faller, J.P.; Teboul, J.L. Respiratory changes in aortic blood velocity as an indicator of fluid responsiveness in ventilated patients with septic shock. Chest 2001, 119, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Beaubien-Souligny, W.; Rola, P.; Haycock, K.; Bouchard, J.; Lamarche, Y.; Spiegel, R.; Denault, A.Y. Quantifying systemic congestion with Point-Of-Care ultrasound: Development of the venous excess ultrasound grading system. Ultrasound J. 2020, 12, 16. [Google Scholar] [CrossRef]
- Malbrain, M.L.N.G.; Langer, T.; Annane, D.; Gattinoni, L.; Elbers, P.; Hahn, R.G.; De laet, I.; Minini, A.; Wong, A.; Ince, C.; et al. Intravenous fluid therapy in the perioperative and critical care set-ting: Executive summary of the International Fluid Academy (IFA). Ann. Intensive Care 2020, 10, 64. [Google Scholar] [CrossRef]
| Index | Main Diagnostic Threshold | Limitations | CO Monitoring | Spontaneous Breathing | Prone Position |
|---|---|---|---|---|---|
| Tests and Indices Derived from Heart–Lung Interaction | |||||
| PPV/SVV | ≥15% | Vt ≥ 8 mL/Kg PBW False positives: cardiac arrhythmias, RV failure False negatives: IAH, low lung compliance, low Vt |  |  |  |
| Vt challenge | ↑ PPV ≥ 3.5% ↑ SVV ≥ 2.5% | Not reliable in cardiac arrhythmias, IAH |  |  |  |
| EEO test | ↑ CO ≥ 5% | Need for 15 s EEO Not if breathing efforts or intense SB activity |  |  |  |
| PEEP test | ↑ CO ≥ 9% | PEEP ≥ 10 cm H2O Not reliable in: RV failure, IAH |  |  |  |
| IVC distensibility index (dIVC) | dIVC ≥ 18% | False positives: SB activity, RV failure False negatives: IAH, low lung compliance, low Vt |  |  |  |
| SVC collapsibility index (SVC-CI) | ΔSVC ≥ 36% | Need for TEE |  |  |  |
| Test Mimicking a Fluid Challenge | |||||
| PLR | ↑ CO ≥ 10% ↑ VTI ≥ 10% ↓ PPV/SVV ≥ 1–4 points ↑ etCO2 ≥ 2 mmHg | False negatives: IAH or venous compression stockings Not applicable if increased ICP |  |  |  |
| Trendelenburg maneuver | ↑ CO ≥ 8–10% | Not reliable in: IAH Risk of gastric reflux Not applicable if increased ICP |  |  |  |
| Mini–FC | ↑ CO ≥ 5% ↑ VTI ≥ 10% | Fluid infusion (min. 100 mL) |  |  |  |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sbaraini Zernini, I.; Nocera, D.; D’Albo, R.; Tonetti, T. Acute Respiratory Distress Syndrome and Fluid Management: Finding the Perfect Balance. J. Clin. Med. 2025, 14, 2067. https://doi.org/10.3390/jcm14062067
Sbaraini Zernini I, Nocera D, D’Albo R, Tonetti T. Acute Respiratory Distress Syndrome and Fluid Management: Finding the Perfect Balance. Journal of Clinical Medicine. 2025; 14(6):2067. https://doi.org/10.3390/jcm14062067
Chicago/Turabian StyleSbaraini Zernini, Irene, Domenico Nocera, Rosanna D’Albo, and Tommaso Tonetti. 2025. "Acute Respiratory Distress Syndrome and Fluid Management: Finding the Perfect Balance" Journal of Clinical Medicine 14, no. 6: 2067. https://doi.org/10.3390/jcm14062067
APA StyleSbaraini Zernini, I., Nocera, D., D’Albo, R., & Tonetti, T. (2025). Acute Respiratory Distress Syndrome and Fluid Management: Finding the Perfect Balance. Journal of Clinical Medicine, 14(6), 2067. https://doi.org/10.3390/jcm14062067






