Abstract
Background: Systemic lupus erythematosus (SLE) is a heterogeneous autoimmune disease predominantly affecting young individuals; however, its late-onset manifestation poses distinct clinical and diagnostic challenges. Methods: This report describes the case of a 93-year-old patient who presented in the Emergency Department with exertional dyspnea, lower limb edema, fatiguability, diffuse abdominal pain, predominantly in the hypogastric region, and loss of appetite. Results: Based on the clinical examination, laboratory tests, and imagistic investigations, we excluded the most common etiologies of edema (decompensated chronic heart failure, glomerular nephropathy/chronic kidney disease, decompensated vascular cirrhosis, hypothyroidism, and hypoproteinemia). Further diagnostic evaluation revealed elevated levels of anti-nuclear antibodies and anti-dsDNA antibodies, along with reduced complement levels, indicating active SLE as the underlying cause of the patient’s edema. During hospitalization, the patient received corticosteroid therapy and, after discharge, was referred to the Rheumatology Department for further treatment. Conclusions: In elderly patients, late-onset SLE exhibits distinct clinical manifestations compared to its early-onset counterpart, likely due to age-related alterations in immune system function.
1. Introduction
Systemic lupus erythematosus (SLE) is a complex autoimmune disease, associated with autoantibodies, that leads to immune reactions and a wide range of clinical manifestations [1]. Usually, SLE involves the integumentary, renal, musculoskeletal, and hematologic systems and is diagnosed in young women between 20 and 40 years old [2]. In contrast, late-onset SLE, defined as disease onset after the age of 50, presents with a distinct clinical phenotype, with reduced mucocutaneous and renal involvement but a higher incidence of serositis, including pleuritis and pericarditis [3,4]. Additionally, the male-to-female ratio in late-onset SLE is lower than in younger patients and has a prevalence of less than 20% in Caucasian populations [5]. The nonspecific presentation of late-onset SLE frequently leads to diagnostic delays or misdiagnosis, as clinical manifestations may be erroneously attributed to age-related physiological changes [6].
We report the case of a female patient in whom generalized edema was the initial manifestation of SLE.
2. Case Presentation
A 93-year-old female patient was admitted to the Internal Medicine Clinic with a constellation of polymorphic symptoms. The symptomatology had insidiously developed over two months prior hospitalization, initially manifesting as diffuse abdominal pain, predominantly in the hypogastric region, loss of appetite, and fatigue, followed by exertional dyspnea and lower limb edema.
The patient’s medical history included arterial hypertension and hyperlipidemia, for which, at the time of the admission, she was receiving treatment with a beta-blocker, an anti-platelet agent, a dihydropyridine calcium channel blocker, and a statin.
Upon clinical examination, the patient was afebrile but with a moderately altered general status. She exhibited dyspnea on moderate exertion, cutaneous pallor, and symmetrical bilateral lower limb pitting edema. Cardiac auscultation revealed rhythmic but faint cardiac sounds, with a heart rate of 55 beats per minute and a blood pressure value of 145/70 mmHg. Peripheral pulses were weak bilaterally, likely secondary to edema. Notably, there were no clinical signs indicative of chronic venous insufficiency. On examination of the respiratory system, percussion of the thorax revealed dullness, and vesicular murmur in the lower third of both lung fields was absent. Oxygen saturation (SpO2) was 95% on room air. Abdominal examination revealed diffuse tenderness upon deep palpation, particularly in the hypogastric region.
Based on the patient’s history and the clinical examination, we identified the presence of edema and suggestive elements for pleural and pericardial effusions. Considering the clinical findings, our differential diagnosis included chronic decompensated heart failure, glomerular nephropathy/chronic kidney disease, decompensated liver cirrhosis, hypothyroidism, and hypoproteinemia. A comprehensive diagnostic workup, including laboratory investigations, functional assessments, and imaging studies, was initiated to determine the underlying etiology of the patient’s edema and pleural effusions.
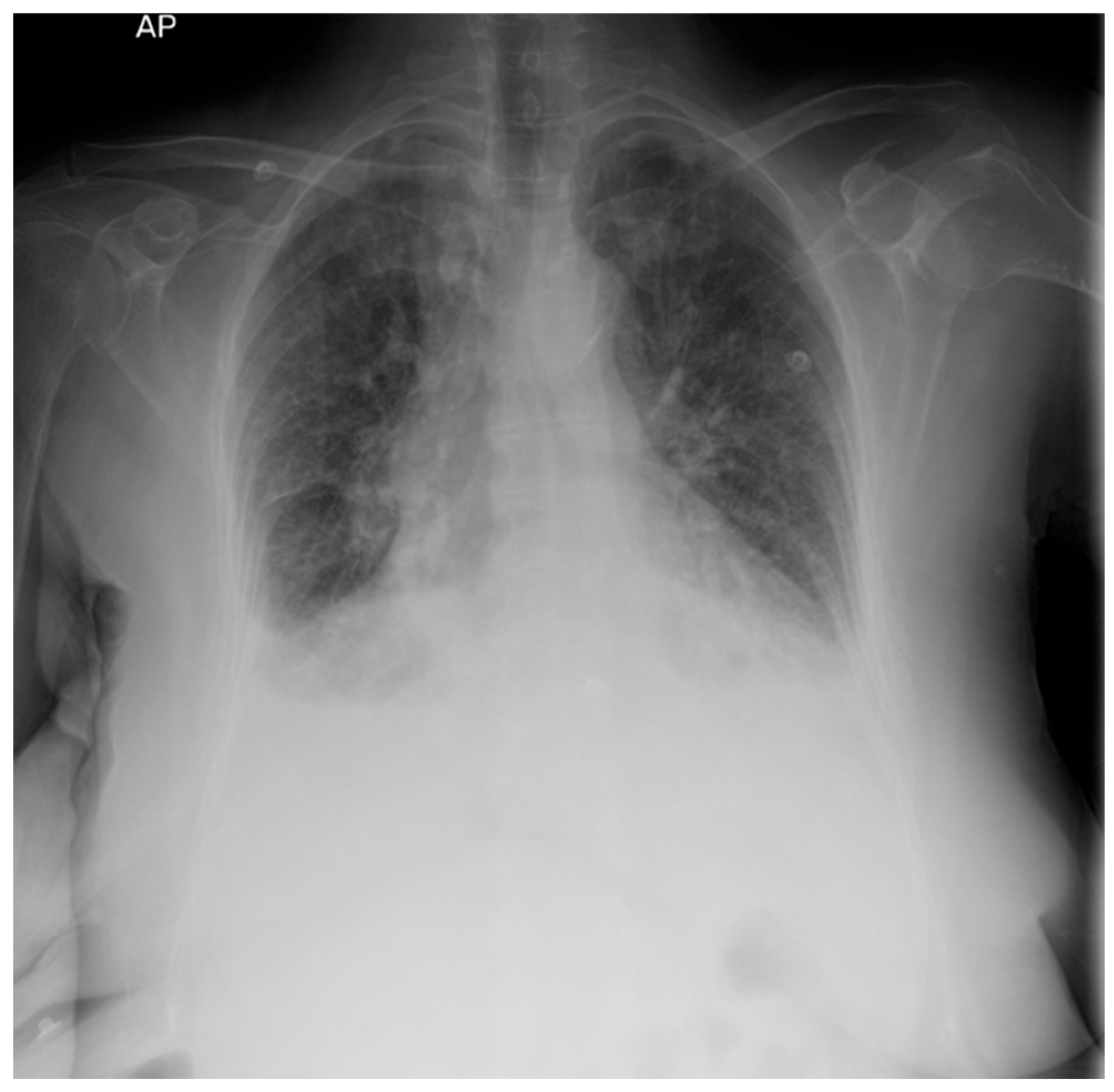
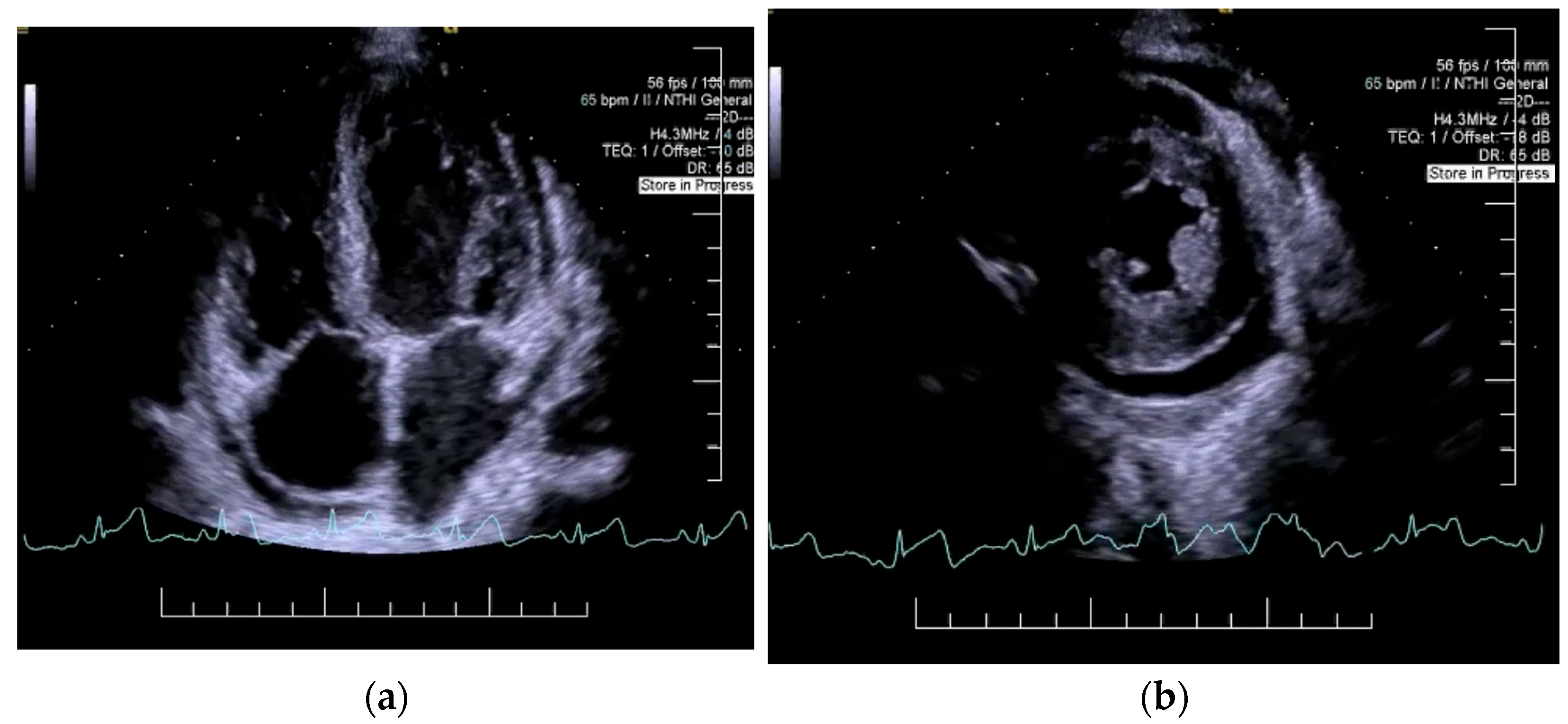
Decompensated chronic heart failure was initially suspected due to the clinical presentation of exertional dyspnea, symmetric lower limb edema, and cardiopulmonary features suggestive of pleural and pericardial effusions. However, laboratory investigations revealed an NT-proBNP level of 418 pg/mL, well below the age-specific cut-off of 1800 pg/mL, reducing the likelihood of heart failure as the primary etiology. Electrocardiographic evaluation demonstrated sinus rhythm with a heart rate of 65 beats per minute and microvoltages in the unipolar limb leads. Chest X-ray identified bilateral pleural effusion and aortic atherosclerosis (Figure 1). H2FPEF score was 1, attributed solely to the patient’s advanced age. To achieve a more definitive diagnosis, transthoracic echocardiography was performed, which revealed a circumferential pericardial effusion (9–11 mm), left ventricular hypertrophy, impaired relaxation-type diastolic dysfunction, and a left ventricular ejection fraction (LVEF) of 50% (Figure 2). Aortic sclerosis was also noted. These findings, along with laboratory tests, established the diagnosis of chronic heart failure, but excluded decompensated heart failure as the underlying cause of the patient’s edema.
Figure 1.
Chest X-ray: bilateral pleural effusion.
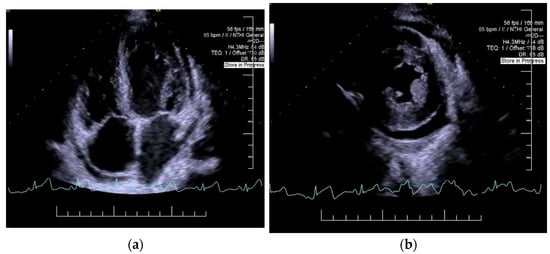
Figure 2.
Echocardiography: left ventricular hypertrophy, impaired relaxation-type 1 diastolic dysfunction, (a) left ventricular ejection fraction of 50%, (b) circumferential pericardial effusion (9–11 mm).
Renal ultrasound demonstrated symmetrically reduced kidney dimensions with otherwise normal morphology, and no evidence of urinary tract obstruction.
Decompensated vascular cirrhosis was also explored as a potential etiology, due to the patient’s fatigue and edema, but hepatic function tests were in normal ranges, without any biochemical elements of hepatocellular injury, cholestasis, hepatic insufficiency, or mesenchymal inflammation. Serological assays for hepatitis B and C were negative. Additionally, abdominal ultrasonography demonstrated normal hepatic and splenic morphology, with no signs of portal or splenic vein dilation and no ascites, further excluding cirrhosis as a contributing factor.
Hypothyroidism, another potential cause of generalized edema and serous effusions, was also considered. The presence of low-voltage electrocardiographic findings and bilateral pleural effusion prompted thyroid function testing, which yielded results within the normal range, thereby excluding hypothyroidism as an underlying cause of the patient’s clinical presentation.
The patient’s history of reduced oral intake, abdominal pain, and bowel movement disturbances raised concerns regarding protein-caloric malnutrition. However, there were no clinical signs of malnutrition, and laboratory investigations showed normal protein levels. Furthermore, following fluid redistribution and decongestion, the patient’s body mass index (BMI) remained within normal limits.
To further elucidate the etiology of the patient’s symptoms, particularly in the context of their advanced age and prior medical history, mesenteric ischemia and acute diverticulitis were considered as potential causes of abdominal pain. An abdominal X-ray was performed to exclude acute surgical emergencies such as intestinal obstruction or perforation. Additionally, laboratory analysis revealed mild normochromic, normocytic anemia, and a positive fecal occult blood test, suggestive of occult gastrointestinal hemorrhage.
Metabolic risk factors, including fasting plasma glucose, HbA1c, and lipid panel, were all within normal ranges.
Based on the presence of pleural and pericardial effusions, we conducted a series of investigations to establish the etiology of polyserositis.
Common differentials for polyserositis include malignancies (hematologic, non-Hodgkin lymphoma, breast, bronchopulmonary, gastric, pelvic, or colonic cancers), infections (Hepatitis B virus, Hepatitis C virus, Cytomegalovirus, Epstein–Barr virus, Coxsackie), autoimmune diseases (SLE, rheumatoid arthritis, adult-onset Still’s disease), and idiopathic cases. Considering the patient’s symptoms, including symmetric pitting edema, abdominal pain, and blood in stool, we suspected intra-abdominal venous or lymphatic obstruction, possibly due to a colonic neoplasm. Serological tumoral markers were elevated, with a notable increase in carbohydrate antigen 19-9 (CA 19-9), typically associated with pancreatic, colonic, or hepatobiliary neoplasms but also increased in autoimmune disease.
Imaging studies, including contrast-enhanced computed tomography (CT) of the chest, abdomen, and pelvis, were performed after the patient declined colonoscopy. Additionally, diagnostic thoracentesis was not feasible due to the minimal volume of pleural effusion. The CT scan confirmed bilateral pleural effusion, pericardial fluid, calcified atheromatous disease of the aorto-iliac vessels, and a 75% stenosis at the celiac trunk origin. Diverticula were observed in the sigmoid and descending colon, but without signs of complications.
Maintaining the suspicion of autoimmune polyserositis, we proceeded with testing for autoimmune causes. Rheumatoid factor was within normal limits, ruling out seroegative rheumatoid arthritis. Anti-nuclear antibodies (ANA) and anti-dsDNA antibodies were significantly elevated: over threefold the normal limit for ANA and over fivefold for anti-dsDNA antibodies, confirming the diagnosis of systemic lupus erythematosus (SLE). Complement levels (C3 and C4) were decreased, indicating active disease.
In the biological assessment, iron deficiency anemia was identified, with decreased serum levels of hemoglobin (11.5 g/dL), iron (17 μg/mL), and ferritin (38 ng/mL). Considering an autoimmune disease as the possible etiology of the edema, the anemia could have been hemolytic. However, the absence of reticulocytosis, along with normal serum levels of indirect bilirubin and lactate dehydrogenase, effectively ruled out hemolysis. Also, inflammatory markers were within normal ranges. Therefore, even if the patient presented with an active disease, the anemia was interpreted as the result of iron deficiency, probably due to the low intake of iron or gastrointestinal bleeding from the diverticula.
The primary therapeutic objectives during hospitalization were clinical stabilization through decongestive therapy, optimization of cardiovascular management, and initiation of targeted treatment for SLE. Loop diuretics (furosemide) were initially administered intravenously and subsequently transitioned to oral administration at a daily dose of 20 mg. SGLT-2 inhibitors (empagliflozin 10 mg daily) were administered to improve heart failure outcomes. Due to the presence of celiac trunk arterial stenosis and an increased cardiovascular risk profile, statin therapy (atorvastatin 40 mg daily) and antiplatelet therapy (acetylsalicylic acid 75 mg daily) were prescribed. Oral iron supplementation was initiated, with good gastrointestinal tolerance. Targeted immunosuppressive therapy for the SLE included intravenous methylprednisolone pulses (125 mg per day for three consecutive days), followed by oral prednisone (5 mg per day) as maintenance therapy, pending reassessment in the Rheumatology Clinic for treatment optimization. After treatment initiation, the patient showed signs of improvement: edema reduction, pain resolution, and absence of electrolyte imbalances. Imaging confirmed a slight reduction in pleural and pericardial effusions. Throughout hospitalization, renal and hepatic function parameters remained stable, and blood pressure and heart rate were within normal limits. Additionally, the patient demonstrated favorable gastrointestinal tolerance to corticosteroid therapy.
Upon discharge, the patient was advised to adhere to a low-sodium, fiber-rich diet with adequate hydration. A referral was made to the Rheumatology Department for treatment adjustment and continued management of SLE.
At the one-month follow-up, the patient exhibited marked clinical improvement, with complete resolution of lower limb edema and normalization of hemoglobin and serum iron levels. Imaging studies further confirmed a reduction in pericardial effusion.
The case highlights the multifaceted challenges of managing polyserositis in a 93-year-old patient with multiple associated comorbidities. After systematically evaluating the most common etiologies of systemic fluid retention in elderly patients, an autoimmune disease was considered a plausible underlying cause. Following the European Alliance of Associations for Rheumatology (EULAR)/American College of Rheumatology (ACR) classification criteria, the diagnosis of SLE was established. Given the potential renal implications of SLE, the patient’s renal function was assessed in accordance with the Kidney Disease: Improving Global Outcomes (KDIGO) 2024 Clinical Practice Guideline for the Management of Lupus Nephritis. At the time of evaluation, there was no evidence of SLE-induced glomerulopathy. Additionally, disease activity was quantified using the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI), with the patient scoring 6 points, indicative of active disease.
Following confirmation of the diagnosis, renal function assessment, and disease activity evaluation, glucocorticosteroid therapy was initiated in accordance with the 2023 EULAR/ACR guidelines for SLE management to achieve rapid symptomatic relief. The uniqueness of this case is underscored by the late-onset presentation of SLE, its association with multiple age-related comorbidities, and the patient’s favorable response to immunosuppressive therapy. Notably, this patient represents the oldest documented case of SLE in the literature to date. The diagnostic and therapeutic approach posed a significant challenge in clinical practice due to the atypical symptomatology, the broad differential diagnosis of polyserositis in this age group, and the potential for adverse effects related to immunosuppressive therapy.
3. Discussions
3.1. Epidemiology
SLE is a complex disease that can affect multiple organ systems, primarily due to autoimmune abnormalities [7,8]. In elderly patients, SLE often presents differently compared to in younger individuals, with a typically lower overall disease activity. This phenomenon can lead to diagnostic challenges, as many of the symptoms are often attributed to the natural aging process, or senescence, rather than an underlying autoimmune condition [6,9]. Regarding the prevalence of SLE in the elderly, the available evidence is limited. Late-onset, defined as after 50 years old, SLE has an incidence of less than 20%, with most of the patients being women, but further epidemiological studies are required to establish prevalences in patients older than 80 years old [3,10].
For the purposes of our article, we selected the age threshold of 70 years, as this delineates the transition into the geriatric age group, a period marked by frailty and the need for specialized medical care. This age limit also aligns with definitions in the literature that distinguish between late- and very-late-onset SLE [11].
Systemic lupus erythematosus (SLE) is rare in the elderly, with distinct features such as equal gender distribution, insidious onset, and relatively benign course, leading to delayed diagnosis. Clinically, it primarily involves arthritis, serositis, myositis, pulmonary fibrosis, and Sjögren’s syndrome. Serologically, there is less hypocomplementemia and more frequent rheumatoid factor compared to in their younger counterparts. The disease is effectively managed with NSAIDs, hydroxychloroquine, low-to-moderate glucocorticoids, azathioprine, and methotrexate, without the need for mycophenolate mofetil, cyclophosphamide, or biologic agents. Despite a milder course, mortality is higher due to comorbidities, infections, and treatment complications [11,12].
In Table 1 are summarized all the clinical and biological characteristics of patients with late-onset SLE reported in the literature.

Table 1.
Clinical and biological characteristics of patients with late-onset SLE.
3.2. Pathophysiology
The primary mechanism of tissue damage in SLE is the development of auto-antibodies and the formation of circulating immune complexes, which are produced by inflammation brought on by dysregulation of immune system responses [2,41]. Inflammation and the adaptive immune system are triggered when processes such as complement activation or phagocytosis are disrupted [42]. In elderly patients, immunosenescence triggers mechanisms such as thymic involution, modifications in T-and B-cell functions, and decreased immune responses, leading to infections, chronic diseases, autoimmune diseases, or cancers [43].
3.2.1. Apoptosis and Toll-like Receptors (TLRs)
Apoptosis is the physiological process by which phagocytes clear the cells programmed for death, preventing the accumulation of autoantigens [41]. Loss of self-tolerance appears when antigen-presenting cells (APCs) activate B and T cells and leads to the formation of autoantigens, stimulated by nucleoprotein complexes from late apoptotic cells that are not cleared by phagocytes [44,45]. Toll-like receptors (TLRs) serve as a bridge between the innate and adaptive immune systems. In SLE, TLR7 and TLR9 are particularly significant because they interact with DNA and RNA particles, which helps produce autoantibodies [46]. TLR activation, interferon regulatory factor 3 (IRF-3) activation, and nuclear factor kappa-light-chain-enhancer (NF-κB) activation all promote the production of type I interferon, which, in turn, stimulates T-cell proliferation and the generation of interferon-gamma (IFN-γ) [47,48].
3.2.2. Innate Immune System
Neutrophils
In SLE, neutrophils exhibit impaired phagocytic activity, reduced production of reactive oxygen species (ROS), and increased formation of neutrophil extracellular traps (NETs). Abnormalities in ROS production contribute to the alteration of apoptosis and promote the generation of NETs, which subsequently trigger type I IFN production [41,49]. In elderly patients, despite elevated neutrophil counts, their functional capacity is compromised due to a diminished ability to form NETs and impaired chemotaxis [50,51].
NET formation (NETosis)
The immune response is influenced by NETs, which are structures that hold DNA, antigens, and other residues. The overproduction of NETs in SLE patients boosts the immune system and the production of autoantibodies [2,52,53].
Natural killer cells (NK cells)
Reduced NK cells seen in SLE are likely caused by elevated IFN-α levels or NK cell migration in target organs, most frequently the kidney. IFN-γ and tumor necrosis factor-alpha (TNF-α) are released when NK cells are activated, which promotes the development of immature NK cells and keeps the inflammation ongoing [41,54].
Macrophages
By generating pro-inflammatory cytokines like TNF-α, IFN-γ, IL-1β, and IL-12, as well as anti-inflammatory cytokines like IL-10 and IL-4, activated macrophages (Mf) trigger inflammatory and immune-modulatory responses [41]. The presence of Mf in the renal tissue leads to lupus nephritis [45].
Basophils
In addition to interacting with B cells by triggering their activation and the production of IgE, basophils have the ability to differentiate T cells into the Th2 type, which stimulates the production of inflammatory cytokines such as IL-4 and IL-6 [55].
Dendritic cells
By using cytokines and HLA-II molecules, dendritic cells (DCs) serve as antigen-presenting cells that link the innate and adaptive immune systems [41]. They can activate B cells and T cells, as well as produce IFN-α, which can result in the development of autoantibodies In patients with SLE, DCs modulate the immune response induced by immune complexes via TLRs [2,56].
3.2.3. Adaptive Immune System
T cells
Follicular helper T cells (Tfh) contribute to B- and T-cell differentiation, autoantibodies, and the production of IL-21 and IL-4, cytokines that stimulate the germinal center B cells subgroup, which produces memory cells [56]. T-cell dysregulation and autoantibody production are caused by elevated levels of IL-12, IL-23, or transforming growth factor-beta (TGF-β), and levels of anti-dsDNA are directly connected with circulating Tfh [45,57].
Through the production of IL-17, Th-17 cells can stimulate inflammation by activating B cells and DCs, resulting in a pro-inflammatory state and elevated auto-antibodies [41,58].
Regulatory T cells (Tregs) exert a suppressive role in SLE; however, reduced levels of Tregs are associated with disease exacerbation. γδT cells can activate T cells and produce cytokines like IFN-γ, TNF-α, IL-10, IL-4, and IL-17, which lead to antibody formation [41,59,60].
B cells
Pro-inflammatory cytokines (TNF-α, IFN-γ, IL-2) activate BAFF, the key factor in the activation of B cells. Being a therapeutic target in SLE, high levels of BAFF are correlated with autoantibodies and SLE activity [45]. Tregs are activated by IL-5 and TGF-β, produced by regulatory B lymphocytes (Breg). Through TNF-α, IL-1, IL-6, IL-8, and inducible nitric oxide synthase (iNOS), anti-dsDNA autoantibodies cause and sustain inflammation [41].
3.2.4. Complement System
The development of SLE is aided by the dysregulation of the complement system, which is a component of the innate immune system, and results in the improper removal of apoptotic debris. Through a variety of mechanisms, the complement system aids in the phagocytosis of antigen–antibody complexes. C1q antibodies are formed by mutations in the C1QA and C1QB genes, conformational alterations of C1q, or the presence of agents with molecular mimicry [61,62]. Lack of C4 causes B cells to proliferate and raises the number of autoantibodies as C4’s primary function is to suppress autoreactive B cells [63,64].
3.2.5. Steroid Hormones
Steroid hormones are classified as sex hormones (androgen, estrogen, and progesterone) and corticosteroids (glucocorticoids and mineralocorticoids), all influencing the immune system [41].
The primary corticosteroid, cortisol, exerts anti-inflammatory effects through several mechanisms, including NF-κB activity inhibition, pro-inflammatory T-cell apoptosis, Treg cell survival, B-cell activity inhibition, proliferation, and antibody formation [65,66].
Aldosterone, the main mineralocorticoid, plays an inflammatory role through increasing NF-κB activity, DC activation, and B- and T-cell proliferation, leading to organ damage [67].
Sex hormones progesterone and androgen have a protective role against SLE, and estrogen contributes to SLE development by interfering with the immune system. Estrogen increases the production of IFN-α and contributes to the survival of autoreactive B cells and the production of IgG antibodies against ds-DNA. High levels of cortisol lead to Th2 cell activation and NETs formation. Progesterone reduces the production of IFN-α, contributes to the production of IgM antibodies, and decreases levels of IgG autoantibodies, and prevents formation of NETs. Androgens decrease the IgG autoantibody production and increase the TGF-β levels and NETosis [41,68,69].
3.2.6. Physiopathology Particularities in Elderly Patients
Age-related immune system alterations in older patients include a pro-inflammatory state marked by a rise in cytokine production (TNF-α, IL-6, IL-1b, IL-18, CRP) and a decrease in cellular immunological responses [51,70]. These alterations could be explained by the inability of the innate immune system to recognize the pathogens and the inability of the adaptive immune system to produce antibodies, mechanisms that exacerbate and maintain the inadequate inflammatory response [43,71].
Alteration of the hematopoietic stem cells contributes to modifications in immune cell functions and composition, resulting in reduced T-cell activation and proliferation due to the decreased level of IL-7 production in the thymus [43].
Also, T cells are the most affected by aging. Loss of the CD28 receptor results in chronic inflammation through the production of IFN-γ, TNF-α, IL-8, granzyme B, and perforin [51,60]. In elderly patients, an elevated number of Tregs is observed, which may lead to altered memory cell response and the development of autoimmune diseases, potentially due to impaired Treg function [51]. Switching from the Th1 (cell-mediated) to the Th2 (humoral) response, along with the decreased production of naïve T cells, increased level of CD8+ T cells, and the loss of TCR sensitivity, enhances inflammation and the improper immune response [72].
Autoimmune response mediated by B cells is driven by the decreased production of naïve B cells and altered specificity of BCR, along with increased mature B cells, leading to a decline in humoral response [72,73,74].
In the elderly, myeloid DCs lose their antigen-presenting ability, fail to activate naïve T cells, and diminish the production of IL-12. Concurrently, plasmacytoid DCs lessen the production of type I and III IFN [72]. NF-kB and macrophage activation increase the IL-6 and TNF-α production, maintaining the low-grade inflammation and contributing to senescence [71]. Neutrophils’ phagocytosis ability is compromised even if their level is normal or increased, amplifying the inflammatory response [75,76].
Also, decreased estrogen levels induce lymphopenia due to the reactive oxygen species that induce DNA damage, predisposing women to autoimmune diseases [77] through epigenetic mechanisms and environmental factors described as predisposing ones. Estrogens generally enhance humoral autoimmunity and stimulate the T-cell response typical of lupus, leading to increased production of cytokines such as IL-4, IL-5, IL-10, and IL-13 [78,79]. The overlapping mechanisms lead not just to autoimmune component overexpression but also to excessive ROS formation, exacerbating SLE and affecting short- and long-term prognosis [80,81,82].
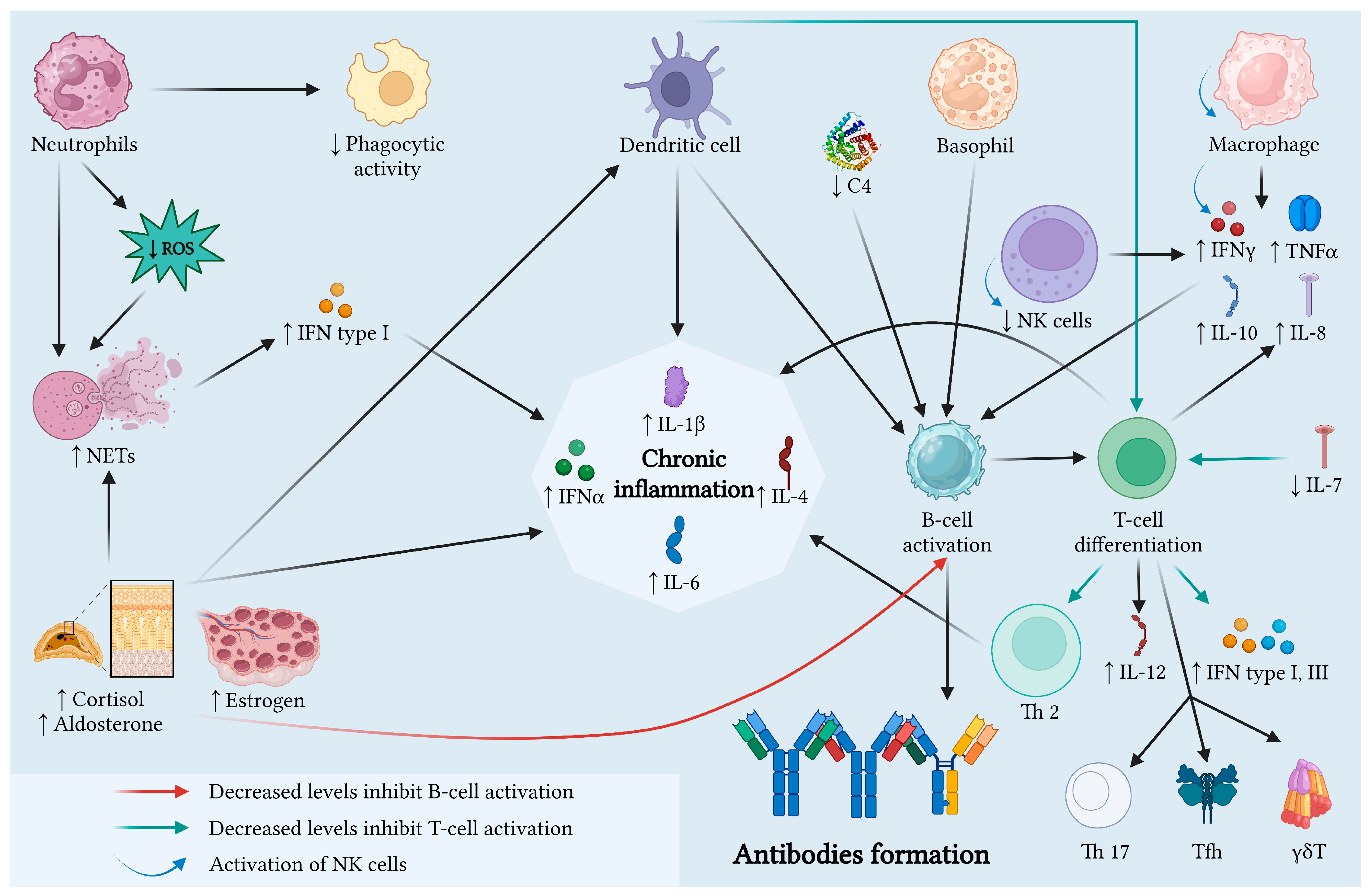
Figure 3 summarizes the physiopathological mechanisms in late-onset SLE explained above.
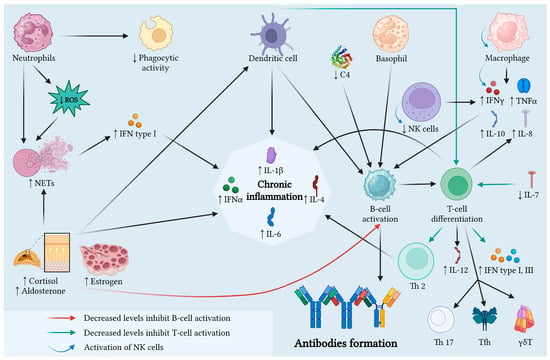
Figure 3.
Physiopathological mechanisms in late-onset SLE. In the elderly, immunosenescence induces low-grade inflammation and maintains the alteration in the immune system. The pro-inflammatory state and the decrease in cellular immunological responses, due to the alteration in immune system components’ function, could explain the clinical manifestations, such as serositis or fibrosis. Legend: C4, complement component 4; IFN, interferon; IL, interleukin; NETs, neutrophil extracellular traps; NK, natural killer cells; ROS, reactive oxygen species; TNF, tumor necrosis factor. Created with BioRender.com.
3.3. Diagnosis
3.3.1. Diagnosis Criteria
The diagnosis of SLE is based on a combination of clinical findings and laboratory tests organized through standardized criteria such as the ACR-1997, SLICC-2012, and EULAR/ACR-2019, which use a scoring system that considers the various manifestations of the disease. Currently, the EULAR/ACR-2019 criteria are preferred for diagnosing SLE as they were developed to increase the sensitivity of the SLICC-2012 criteria while maintaining the specificity of both the ACR-1997 and SLICC-2012 criteria. Having been validated in the adult population in 2020, these criteria have a sensitivity of 92% [83].
The diagnostic complexity of SLE is largely attributable to its marked heterogeneity. The absence of pathognomonic clinical features, coupled with its potential to affect any organ system, often contributes to delayed recognition. Diagnosis remains primarily clinical, supported by serological abnormalities. A high degree of clinical vigilance is warranted in patients exhibiting multisystemic involvement suggestive of SLE [83].
Over time, three main classification systems have been developed to characterize the clinical and immunological features of SLE: the ACR-1997, the SLICC-2012, and the more recent EULAR/ACR-2019 criteria. Although these classification criteria facilitate consistency in patient selection for clinical research, they are not intended for diagnostic purposes. Nevertheless, in routine clinical practice, they are frequently applied to guide the assessment of suspected cases [84].
Currently, the EULAR/ACR-2019 criteria are preferred for classifying SLE as they were developed to increase the sensitivity of the SLICC-2012 criteria while maintaining the specificity of both the ACR-1997 and SLICC-2012 criteria. Having been validated in the adult population in 2020, these criteria have a sensitivity of 92% [85].
3.3.2. Characteristics of SLE in the Elderly Patient
Clinically, SLE in the elderly mainly manifests as serositis, myositis, pulmonary fibrosis, and Sjögren’s syndrome. As is summarized in Table 1, the most common manifestations in late-onset SLE include pleural effusion, lower limb edema, ascites, pericardial effusion, and arthralgia. Photosensitive lesions are less common in elderly patients, though not entirely absent. Serologically, elderly patients typically exhibit less hypocomplementemia and a higher prevalence of rheumatoid factor compared to younger individuals. The immunological alterations associated with aging may help explain the clinical differences observed between young and elderly patients [86].
In elderly patients, differential diagnosis for SLE should include other autoimmune diseases, such as rheumatoid arthritis, Raynaud syndrome, idiopathic thrombocytopenic purpura, polymyalgia rheumatica, or drug-induced lupus. In other autoimmune diseases, the diagnosis is based on clinical and biological modifications, but in lupus-like syndrome, in addition to the presence of clinical manifestations, patients could exhibit anti-nuclear antibodies, without any other immune serological alteration [87].
The management of elderly-onset SLE typically involves a conservative approach, with nonsteroidal anti-inflammatory drugs (NSAIDs), hydroxychloroquine, and low-to-moderate doses of glucocorticoids being the mainstay of treatment. Immunosuppressive agents such as azathioprine and methotrexate are commonly used, while mycophenolate mofetil, cyclophosphamide, and biologic agents are rarely required due to the generally milder course of the disease in this population. Despite a more indolent disease progression, elderly patients with SLE have higher mortality rates, primarily due to comorbidities, infections, and complications associated with treatment [11].
3.4. Management
The management goal in patients with SLE is remission or low disease activity. Treatment options are listed in Figure 4.

Figure 4.
Treatment of SLE—EULAR 2023 recommendations for the management of SLE. GC = glucocorticosteroids; im = intramuscular; iv = intravenous; po = per os; SLEDAI = Systemic Lupus Erythematosus Disease Activity Index.
In patients with SLE, regardless of the severity of the disease, hydroxychloroquine (HCQ) is the first-line treatment, even if the most important complication is retinopathy. The use of glucocorticosteroids is recommended for rapid symptom relief, but long-term use can lead to complications. There are multiple specimens for GC administrations, the main goal being to reduce the dosages to ≤5 mg Prednisone per day or stopping the administration [88].
When HCQ or GCs alone are insufficient to control disease activity, immunosuppressive drugs should be taken into consideration. The second line for mild forms of the condition is methotrexate (MTX); however, Azathioprine (AZA) should be considered in place of MTX if the patient is female and of childbearing age. Mycophenolate mofetil, cyclophosphamide, and rituximab are therapy choices for individuals with severe forms of the condition, while calcineurin inhibitors or mycophenolate mofetil may be administered to those with intermediate forms in addition to MTX and AZA. Immunosuppressive therapy with monoclonal antibodies such as B-lymphocyte stimulator-specific inhibitor (belimumab) and type 1 interferon receptor antibody (anifrolumab) are first-line treatment specimens in patients with severe disease but without renal involvement [88].
The management of late-onset SLE needs a multidisciplinary approach, considering the increased prevalence of comorbidities such as cardiovascular disease and infections, which can complicate treatment regimens [89,90]. The use of GCs and other immunesuppressive agents must be balanced against the risks associated with these therapies in older populations [91,92].
The age and general health of the patient must be carefully taken into account when treating late-onset SLE, especially when lupus nephritis is present. Research shows that individuals with late-onset lupus nephritis frequently receive less aggressive therapy than those with early-onset lupus, with immunosuppressive medications such as cyclo-phosphamide being significantly underutilized [93,94,95].
Clinically, late-onset SLE is characterized by milder symptoms, less renal involvement, and a lower total disease activity than early-onset cases [91,93]. Nevertheless, despite these variations, late-onset SLE is linked to a higher risk of mortality, which is frequently attributed to treatment-related problems and comorbidities rather than the illness itself [89].
3.5. Prognosis
Although elderly patients with late-onset SLE tend to have lower disease activity and less severe organ involvement, they are at higher risk for comorbidities that can exacerbate disease progression and lead to cumulative organ damage. Age at SLE onset is considered a significant risk factor for both mortality and morbidity due to accelerated atherosclerosis associated with chronic inflammation, long-term corticosteroid use, cardiovascular diseases, and osteoporosis [96].
Elderly patients with SLE often have pre-existing comorbid conditions such as hypertension, cardiovascular diseases, venous thrombosis, and pulmonary embolism, which can contribute to cumulative organ damage before the diagnosis of SLE [97]. These conditions, usually present at the time of SLE onset, can worsen the patient’s overall health and lead to further complications.
4. Conclusions
SLE is a heterogeneous autoimmune disorder that typically manifests between the second and fourth decades of life, predominantly in females. Late-onset SLE, defined as disease onset after the age of 50, can present diagnostic challenges due to the nonspecific nature of its manifestations, which may overlap with other chronic conditions commonly observed in elderly patients.
As previously discussed, patients with late-onset SLE often present with edema (including peripheral edema, pleural or pericardial effusion, and ascites), with less frequent involvement of joints or the characteristic malar rash. In these cases, when clinical symptoms cannot be attributed to other conditions, primary care physicians should consider an autoimmune etiology, and referral to a Rheumatology or Internal Medicine Clinic is warranted for further evaluation.
While the management guidelines for SLE do not specifically address the nuances of treatment in the elderly due to a lack of dedicated studies, it is crucial to exercise caution in these patients. Special attention should be paid to dose adjustments and close monitoring for adverse effects should be performed, particularly given the increased susceptibility to side effects in older populations.
The degree of cumulative organ damage is greater in patients with late-onset SLE, which is associated with a more complex risk profile due to advanced age. Aggressive prevention strategies are recommended for late-onset SLE patients to reduce the risk of cardiovascular diseases and osteoporosis. Patients such as those presented should benefit from a proactive treatment strategy and careful monitoring to prevent severe complications and improve their long-term quality of life.
Author Contributions
Conceptualization, A.C.; methodology, R.-E.A. and V.Ș.; formal analysis, E.-D.G.; investigation, B.C.M., O.S. and C.-M.L.; resources, M.C. and A.-D.D.; data curation, A.C., A.B. and E.-D.G.; writing—original draft preparation, R.-E.A., D.C.S. and A.A.; writing—review and editing, C.L. and A.S.; visualization, V.Ș. and R.-E.A.; supervision, L.Ș.; project administration, A.C. and L.Ș. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Upon admission to our hospital, all patients must sign an informed consent form regarding their agreement in clinical research and medical training. Consequently, ethical approval from the ethics commission is unnecessary for case publication.
Informed Consent Statement
Informed consent forms for participation were obtained from the patients.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Aringer, M.; Costenbader, K.; Daikh, D.; Brinks, R.; Mosca, M.; Ramsey-Goldman, R.; Smolen, J.S.; Wofsy, D.; Boumpas, D.T.; Kamen, D.L.; et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Ann. Rheum. Dis. 2019, 78, 1151–1159. [Google Scholar] [CrossRef]
- Arnaud, L.; Chasset, F.; Martin, T. Immunopathogenesis of systemic lupus erythematosus: An update. Autoimmun. Rev. 2024, 23, 103648. [Google Scholar] [CrossRef] [PubMed]
- So, C.; Imai, R.; Tomishima, Y.; Nishimura, N. Bilateral Pleuritis as the Initial Symptom of Systemic Lupus Erythematosus: A Case Series and Literature Review. Intern. Med. 2019, 58, 1617–1620. [Google Scholar] [CrossRef] [PubMed]
- Ikushima, H.; Mitsutake, A.; Hideyama, T.; Sato, T.; Katsumata, J.; Seki, T.; Maekawa, R.; Kishida, Y.; Shiio, Y. Severe pleuritis and pericarditis associated with very-late-onset systemic lupus erythematosus. J. Gen. Fam. Med. 2018, 19, 53–56. [Google Scholar] [CrossRef]
- Frutos, A.R.; Holgado, S.; Bergé, A.S.; Casas, I.; Olivé, A.; López-Longo, F.J.; Calvo-Alén, J.; Galindo, M.; Fernández-Nebro, A.; Pego-Reigosa, J.M.; et al. Late-onset versus early-onset systemic lupus: Characteristics and outcome in a national multicentre register (RELESSER). Rheumatology 2021, 60, 1793–1803. [Google Scholar] [CrossRef]
- Altier, J.; Oates, J.; Ward, C. Polymyalgia Rheumatica or Late Onset Lupus? A Case Report. J. Investig. Med. High Impact Case Rep. 2022, 10, 23247096221089493. [Google Scholar] [CrossRef]
- Hasegawa, T.; Nishikawa, K.; Ohjino, Y.; Sano, C.; Ohta, R. A Case of Late-Onset Systemic Lupus Erythematosus With Systemic Symptoms Leading to Multiple Organ Failure. Cureus 2023, 15, e46428. [Google Scholar] [CrossRef]
- Ameer, M.A.; Chaudhry, H.; Mushtaq, J.; Khan, O.S.; Babar, M.; Hashim, T.; Zeb, S.; Tariq, M.A.; Patlolla, S.R.; Ali, J.; et al. An Overview of Systemic Lupus Erythematosus (SLE) Pathogenesis, Classification, and Management. Cureus 2022, 14, e30330. [Google Scholar] [CrossRef]
- Fujita, Y.; Fukui, S.; Ishida, M.; Endo, Y.; Tsuji, S.; Takatani, A.; Igawa, T.; Shimizu, T.; Umeda, M.; Sumiyoshi, R.; et al. Reversible Cognitive Dysfunction in Elderly-onset Systemic Lupus Erythematosus, Successfully Treated with Aggressive Immunosuppressive Therapy. Intern. Med. 2018, 57, 3025–3028. [Google Scholar] [CrossRef]
- Hammami, S.; Chaabane, N.; Mahmoudi, H.; Bdioui, F.; Saffar, H. Late-onset systemic lupus erythematosus-associated primary biliary cirrhosis. Int. Arch. Med. 2013, 6, 3. [Google Scholar] [CrossRef]
- Viveiros, L.; Neves, A.; Gouveia, T.; Isenberg, D.A. A large cohort comparison of very late-onset systemic lupus erythematosus with younger-onset patients. Clin. Exp. Rheumatol. 2024, 42, 1480–1486. [Google Scholar] [CrossRef] [PubMed]
- Celińska-Löwenhoff, M.; Musiał, J. Late-onset systemic lupus erythematosus: Clinical manifestations, course, and prognosis. Pol. Arch. Med. Wewn. 2015, 125, 497–499. [Google Scholar]
- Kim, E.J.; DeSimone, M.S.; Shi, C.R. Subacute cutaneous lupus erythematosus following osimertinib therapy for non-small cell lung cancer: A case report. JAAD Case Rep. 2024, 53, 30–33. [Google Scholar] [PubMed]
- Mrabet, S.; Boukadida, R.; Sidina, E.; Zellama, D.; Achour, A. Late-Onset Familial Systemic Lupus Erythematosus With Lupus Nephritis in an 82-Year-Old Male: A Case Report and Literature Review. Am. J. Mens. Health 2024, 18, 15579883241293781. [Google Scholar] [PubMed]
- Tsuji, N.; Inoguchi, N.; Sakai, T.; Furumuro, T.; Takaya, K. Unilateral Pleural Effusion as an Initial Manifestation of Systemic Lupus Erythematosus in a Patient of Advanced Age. Cureus 2024, 16, e63327. [Google Scholar] [CrossRef]
- Rodriguez-Perez, N. Systemic Lupus Erythematosus in a Nonagenarian Woman: A Case Report. Cureus 2024, 16, e69812. [Google Scholar]
- Kioi, Y.; Yorifuji, H.; Higami, Y.; Katada, Y. Serositis and lymphopenia are common features of systemic lupus erythematosus following SARS-CoV-2 infection: A case report and literature review. Int. J. Rheum. Dis. 2023, 26, 2267–2271. [Google Scholar]
- Chao, Y.H.; Chen, H.Y. Rare cause of ascites and pleural effusion: The first case report and literature review of pseudo-pseudo Meig’s syndrome in Taiwan. J. Formos. Med. Assoc. 2022, 121, 2633–2638. [Google Scholar]
- Helali, W.; El Euch, M.; Sassi, C.; Kefi, A.; Ben Hamida, F.; Ben Abdelghani, K.; Turki, S.; Abderrahim, E. Fulminant lupus pneumonitis complicating systemic lupus erythematosus in the elderly. Clin. Case Rep. 2023, 11, e6966. [Google Scholar]
- Xu, Z.-G.; Li, W.-L.; Wang, X.; Zhang, S.-Y.; Zhang, Y.-W.; Wei, X.; Li, C.-D.; Zeng, P.; Luan, S.-D. Systemic lupus erythematosus and antineutrophil cytoplasmic antibody-associated vasculitis overlap syndrome in a 77-year-old man: A case report. World J. Clin. Cases 2021, 9, 707–713. [Google Scholar]
- Kuroda, K.; Itagane, M.; Kinjo, M. Late-Onset Systemic Lupus Erythematosus Associated with Autoimmune Hemolytic Anemia and Sixth Cranial Nerve Palsy. Am. J. Case Rep. 2021, 22, e932959. [Google Scholar] [CrossRef] [PubMed]
- Constantinescu, A.; Cobilinschi, C.; Gradinaru, E.; Saulescu, I.; Ionescu, R. Features of Late-Onset Systemic Lupus Erythematosus. Rom. J. Rheumatol. 2021, 30, 121–124. [Google Scholar] [CrossRef]
- Bao, D.; Tan, Y.; Yu, X.; Wang, B.; Wang, H.; Xu, R.; Zhou, F.; Zhao, M. Case Report: A Rare Case of Lupus Nephritis Associated With Mantle Cell Lymphoma. Front. Med. 2021, 8, 759279. [Google Scholar]
- Tay, H.B.; Angkodjojo, S.; Tay, Z.E.A.; Tan, P.H.; Tan, Y.D. Angioimmunoblastic T-cell Lymphoma: A Mimic for Lupus. Eur. J. Case Rep. Intern. Med. 2020, 7, 001734. [Google Scholar]
- Rezazadegan, R.; Koushk-Jalali, B.; Kuntz, T.; Oellig, F.; Tigges, C.; Kreuter, A. Paraneoplastischer subakut kutaner Lupus erythematodes. Z. Rheumatol. 2021, 80, 189–193. [Google Scholar] [CrossRef]
- Bonometti, R.; Sacchi, M.C.; Stobbione, P.; Lauritano, E.C.; Tamiazzo, S.; Marchegiani, A.; Novara, E.; Molinaro, E.; Benedetti, I.; Massone, L.; et al. The first case of systemic lupus erythematosus (SLE) triggered by COVID-19 infection. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 9695–9697. [Google Scholar]
- de Montjoye, S.; Boland, B.; Van Raemdonck, J.; Houssiau, F.A. Very Late-Onset Systemic Lupus Erythematosus as Unusual Cause of Reversible Functional and Cognitive Impairments in an Octogenarian Patient. Eur. J. Case Rep. Intern. Med. 2020, 7, 001570. [Google Scholar]
- Kalenchic, T.I.; Didenko, N.S.; Kabak, S.L. Pulmonary Manifestations of Late-Onset Systemic Lupus Erythematosus: A Case Report. Revmatologiia 2019, 4, 63–66. [Google Scholar]
- D’Andréa, A.; Peillet, D.L.; Serratrice, C.; Petignat, P.A.; Prendki, V.; Reny, J.L.; Serratrice, J. Diagnosis of systemic lupus erythematosus by presence of Hargraves cells in eosinophilic pleural effusion: Case report. Medicine 2018, 97, e12871. [Google Scholar]
- Jatwani, S.; Handa, R.; Jatwani, K.; Chugh, K. Bronchiolitis obliterans organising pneumonia as an initial manifestation in a patient with systemic lupus erythematosus: A rare presentation. BMJ Case Rep. 2018, 2018, bcr2017224094. [Google Scholar] [CrossRef]
- Arai, H.; Hayashi, H.; Ogata, S.; Uto, K.; Saegusa, J.; Takahashi, K.; Koide, S.; Inaguma, D.; Hasegawa, M.; Yuzawa, Y. Progression of immunoglobulin G4-related disease to systematic lupus erythematosus after gastric cancer surgery: A case report. Medicine 2018, 97, e13545. [Google Scholar] [CrossRef] [PubMed]
- Shirai, H.; Shoda, H.; Kobayashi, S.; Fujio, K.; Setoguchi, K.; Yamamoto, K. Two cases of very elderly onset male lupus patient; the characteristics and sex differences of elderly onset systemic lupus erythematosus patients. Mod. Rheumatol. Case 2017, 1, 84–88. [Google Scholar] [CrossRef]
- Boddu, P.; Nadiri, M.; Malik, O. Diffuse Bullous Eruptions in an Elderly Woman: Late-Onset Bullous Systemic Lupus Erythematosus. Case Rep. Dermatol. 2016, 8, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, Y.; Toyoshima, M.; Akiyama, N.; Suda, T. Combined Pulmonary Fibrosis and Emphysema Preceding Lupus Pleuritis. Intern. Med. 2016, 55, 783–786. [Google Scholar] [CrossRef][Green Version]
- Hammami, S.; Bdioui, F.; Ouaz, A.; Loghmari, H.; Mahjoub, S.; Saffar, H. Successful treatment of massive ascites due to lupus peritonitis with hydroxychloroquine in old- onset lupus erythematosus. Pan Afr. Med. J. 2014, 18, 165. [Google Scholar] [CrossRef]
- Aguirre, H.D.; López, A.F.P.; Aponte Monsalve, J.E.; Estupiñan, M.F. Lupus eritematoso sistémico en el anciano: Una presentación atípica de una enfermedad común. Rev. Colomb. Reumatol. 2014, 21, 104–108. [Google Scholar] [CrossRef]
- Chebbi, W.; Berriche, O. Pulmonary fibrosis revealing systemic lupus erythematosus in an elderly subject. Pan Afr. Med. J. 2014, 18, 311. [Google Scholar]
- Iyoda, M.; Suzuki, H.; Ashikaga, E.; Nagai, H.; Kuroki, A.; Shibata, T.; Kitazawa, K.; Akizawa, T. Elderly onset systemic lupus erythematosus (SLE) presenting with disseminated intravascular coagulation (DIC). Clin. Rheumatol. 2008, 27, S15–S18. [Google Scholar] [CrossRef]
- Ito, H.; Nanamiya, W.; Kuroda, N.; Inoue, M.; Sasaoka, A.; Chijiwa, T.; Nishiya, K.; Hashimoto, K.; Nakagawa, O. Chronic lupus peritonitis with massive ascites at elderly onset: Case report and review of the literature. Intern. Med. 2002, 41, 1056–1061. [Google Scholar] [CrossRef][Green Version]
- Aharon, A.; Zandman-Goddard, G.; Shoenfeld, Y. Autoimmune Multiorgan Involvement in Elderly Men Is It SLE? Clin. Rheumatol. 1994, 13, 631–634. [Google Scholar] [CrossRef]
- Bolouri, N.; Akhtari, M.; Farhadi, E.; Mansouri, R.; Faezi, S.T.; Jamshidi, A.; Mahmoudi, M. Role of the innate and adaptive immune responses in the pathogenesis of systemic lupus erythematosus. Inflamm. Res. 2022, 71, 537–554. [Google Scholar] [CrossRef] [PubMed]
- Mathian, A.; Felten, R.; Alarcon-Riquelme, M.E.; Psarras, A.; Mertz, P.; Chasset, F.; Vital, E.M.; Arnaud, L. Type 1 interferons: A target for immune-mediated inflammatory diseases (IMIDs). Jt. Bone Spine 2024, 91, 105627. [Google Scholar] [CrossRef] [PubMed]
- Goyani, P.; Christodoulou, R.; Vassiliou, E. Immunosenescence: Aging and Immune System Decline. Vaccines 2024, 12, 1314. [Google Scholar] [CrossRef] [PubMed]
- Liphaus, B.L.; Kiss, M.H. The role of apoptosis proteins and complement components in the etiopathogenesis of systemic lupus erythematosus. Clinics 2010, 65, 327–333. [Google Scholar] [CrossRef]
- Tsokos, G.C.; Lo, M.S.; Costa Reis, P.; Sullivan, K.E. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat. Rev. Rheumatol. 2016, 12, 716–730. [Google Scholar] [CrossRef]
- Lorenz, G.; Lech, M.; Anders, H.J. Toll-like receptor activation in the pathogenesis of lupus nephritis. Clin. Immunol. 2017, 185, 86–94. [Google Scholar] [CrossRef]
- Murayama, G.; Chiba, A.; Kuga, T.; Makiyama, A.; Yamaji, K.; Tamura, N.; Miyake, S. Inhibition of mTOR suppresses IFNα production and the STING pathway in monocytes from systemic lupus erythematosus patients. Rheumatology 2020, 59, 2992–3002. [Google Scholar] [CrossRef]
- Kato, Y.; Park, J.; Takamatsu, H.; Konaka, H.; Aoki, W.; Aburaya, S.; Ueda, M.; Nishide, M.; Koyama, S.; Hayama, Y.; et al. Apoptosis-derived membrane vesicles drive the cGAS-STING pathway and enhance type I IFN production in systemic lupus erythematosus. Ann. Rheum. Dis. 2018, 77, 1507–1515. [Google Scholar] [CrossRef]
- de Bont, C.M.; Boelens, W.C.; Pruijn, G.J.M. NETosis, complement, and coagulation: A triangular relationship. Cell Mol. Immunol. 2019, 16, 19–27. [Google Scholar] [CrossRef]
- Tseng, C.W.; Liu, G.Y. Expanding roles of neutrophils in aging hosts. Curr. Opin. Immunol. 2014, 29, 43–48. [Google Scholar] [CrossRef]
- Ray, D.; Yung, R. Immune Senescence, Epigenetics and Autoimmunity. Clin. Immunol. 2018, 196, 59–63. [Google Scholar]
- Lood, C.; Blanco, L.P.; Purmalek, M.M.; Carmona-Rivera, C.; De Ravin, S.S.; Smith, C.K.; Malech, H.L.; A Ledbetter, J.; Elkon, K.B.; Kaplan, M.J. Neutrophil extracellular traps enriched in oxidized mitochondrial DNA are interferogenic and contribute to lupus-like disease. Nat. Med. 2016, 22, 146–153. [Google Scholar] [CrossRef]
- Wang, T.; Mei, Y.; Li, Z. Research Progress on Regulatory B Cells in Systemic Lupus Erythematosus. BioMed Res. Int. 2019, 2019, 7948687. [Google Scholar]
- Hudspeth, K.L.; Shu, W.; Wang, J.; Rahman, S.; A Smith, M.; Casey, K.A.; Stephens, G.L.; A Sanjuan, M.; Team, A.M.; A Hasni, S.; et al. NK Cell Phenotype and Proliferation in Systemic Lupus Erythematosus. J. Immunol. 2016, 196, 194.5. [Google Scholar]
- Dossybayeva, K.; Abdukhakimova, D.; Poddighe, D. Basophils and Systemic Lupus Erythematosus in Murine Models and Human Patients. Biology 2020, 9, 308. [Google Scholar] [CrossRef]
- Soni, C.; Reizis, B. Self-DNA at the Epicenter of SLE: Immunogenic Forms, Regulation, and Effects. Front. Immunol. 2019, 10, 1601. [Google Scholar] [CrossRef]
- Men, K.; Chen, Y.; Zhang, J.; Wei, D. The evaluation of cellular immune function in elderly patients with systemic lupus erythematosus. Korean J. Intern. Med. 2019, 34, 932–937. [Google Scholar]
- Suárez-Fueyo, A.; Bradley, S.J.; Tsokos, G.C. T Cells in Systemic Lupus Erythematosus. Curr. Opin. Immunol. 2016, 43, 32–38. [Google Scholar]
- Wu, M.; Yang, J.; Li, X.; Chen, J. The Role of γδ T Cells in Systemic Lupus Erythematosus. J. Immunol. Res. 2016, 2016, 2932531. [Google Scholar] [CrossRef]
- Lu, Y.; Ruan, Y.; Hong, P.; Rui, K.; Liu, Q.; Wang, S.; Cui, D. T-cell senescence: A crucial player in autoimmune diseases. Clin. Immunol. 2023, 248, 109202. [Google Scholar]
- Hristova, M.H.; Stoyanova, V.S. Autoantibodies against Complement Components in Systemic Lupus Erythematosus—Role in the Pathogenesis and Clinical Manifestations. Lupus 2017, 26, 1550–1555. [Google Scholar]
- Pickering, M.C.; Botto, M. Are anti-C1q antibodies different from other SLE autoantibodies? Nat. Rev. Rheumatol. 2010, 6, 490–493. [Google Scholar]
- Leffler, J.; Bengtsson, A.A.; Blom, A.M. The complement system in systemic lupus erythematosus: An update. Ann. Rheum. Dis. 2014, 73, 1601–1606. [Google Scholar]
- Ramsey-Goldman, R.; Li, J.; Dervieux, T.; Alexander, R.V. Cell-bound complement activation products in SLE. Lupus Sci. Med. 2017, 4, e000236. [Google Scholar]
- Baschant, U.; Tuckermann, J. The role of the glucocorticoid receptor in inflammation and immunity. J. Steroid Biochem. Mol. Biol. 2010, 120, 69–75. [Google Scholar] [CrossRef]
- Zen, M.; Canova, M.; Campana, C.; Bettio, S.; Nalotto, L.; Rampudda, M.; Ramonda, R.; Iaccarino, L.; Doria, A. The kaleidoscope of glucorticoid effects on immune system. Autoimmun. Rev. 2011, 10, 305–310. [Google Scholar]
- Ferreira, N.S.; Tostes, R.C.; Paradis, P.; Schiffrin, E.L. Aldosterone, Inflammation, Immune System, and Hypertension. Am. J. Hypertens. 2021, 34, 15–27. [Google Scholar] [CrossRef]
- Bereshchenko, O.; Bruscoli, S.; Riccardi, C. Glucocorticoids, Sex Hormones, and Immunity. Front. Immunol. 2018, 9, 1332. [Google Scholar]
- Pan, Q.; Chen, X.; Liao, S.; Chen, X.; Zhao, C.; Xu, Y.-Z.; Liu, H.-F. Updated advances of linking psychosocial factors and sex hormones with systemic lupus erythematosus susceptibility and development. PeerJ 2019, 7, e7179. [Google Scholar]
- Alberro, A.; Iribarren-Lopez, A.; Sáenz-Cuesta, M.; Matheu, A.; Vergara, I.; Otaegui, D. Inflammaging markers characteristic of advanced age show similar levels with frailty and dependency. Sci. Rep. 2021, 11, 4358. [Google Scholar]
- Cisneros, B.; García-Aguirre, I.; Unzueta, J.; Arrieta-Cruz, I.; González-Morales, O.; Domínguez-Larrieta, J.M.; Tamez-González, A.; Leyva-Gómez, G.; Magaña, J.J. Immune system modulation in aging: Molecular mechanisms and therapeutic targets. Front. Immunol. 2022, 13, 1059173. [Google Scholar] [CrossRef]
- Montoya-Ortiz, G. Immunosenescence, Aging, and Systemic Lupus Erythematous. Autoimmune Dis. 2013, 2013, 267078. [Google Scholar] [CrossRef]
- Gibson, K.L.; Wu, Y.; Barnett, Y.; Duggan, O.; Vaughan, R.; Kondeatis, E.; Nilsson, B.; Wikby, A.; Kipling, D.; Dunn-Walters, D.K. B-cell diversity decreases in old age and is correlated with poor health status. Aging Cell 2009, 8, 18–25. [Google Scholar] [CrossRef]
- Buffa, S.; Bulati, M.; Pellicanò, M.; Dunn-Walters, D.K.; Wu, Y.-C.; Candore, G.; Vitello, S.; Caruso, C.; Colonna-Romano, G. B cell immunosenescence: Different features of naive and memory B cells in elderly. Biogerontology 2011, 12, 473–483. [Google Scholar] [CrossRef]
- Uhl, B.; Vadlau, Y.; Zuchtriegel, G.; Nekolla, K.; Sharaf, K.; Gaertner, F.; Massberg, S.; Krombach, F.; Reichel, C.A. Aged neutrophils contribute to the first line of defense in the acute inflammatory response. Blood 2016, 128, 2327–2337. [Google Scholar] [CrossRef]
- Gullotta, G.S.; De Feo, D.; Friebel, E.; Semerano, A.; Scotti, G.M.; Bergamaschi, A.; Butti, E.; Brambilla, E.; Genchi, A.; Capotondo, A.; et al. Age-induced alterations of granulopoiesis generate atypical neutrophils that aggravate stroke pathology. Nat. Immunol. 2023, 24, 925–940. [Google Scholar] [CrossRef]
- Gubbels Bupp, M.R.; Potluri, T.; Fink, A.L.; Klein, S.L. The Confluence of Sex Hormones and Aging on Immunity. Front. Immunol. 2018, 9, 1269. [Google Scholar] [CrossRef]
- Costenbader, K.H.; Gay, S.; Alarcón-Riquelme, M.E.; Iaccarino, L.; Doria, A. Genes, epigenetic regulation and environmental factors: Which is the most relevant in developing autoimmune diseases? Autoimmun. Rev. 2012, 11, 604–609. [Google Scholar] [CrossRef]
- Cutolo, M.; Sulli, A.; Capellino, S.; Villaggio, B.; Montagna, P.; Seriolo, B.; Straub, R.H. Sex hormones influence on the immune system: Basic and clinical aspects in autoimmunity. Lupus 2004, 13, 635–638. [Google Scholar] [CrossRef]
- Fairweather, D.; Beetler, D.J.; McCabe, E.J.; Lieberman, S.M. Mechanisms underlying sex differences in autoimmunity. J. Clin. Investig. 2024, 134, e180076. [Google Scholar] [CrossRef]
- Richardson, B. Reprint of The Interaction between Environmental Triggers and Epigenetics in Autoimmunity. Clin. Immunol 2018, 196, 72–76. [Google Scholar] [CrossRef]
- Merrheim, J.; Villegas, J.; Van Wassenhove, J.; Khansa, R.; Berrih-Aknin, S.; le Panse, R.; Dragin, N. Estrogen, estrogen-like molecules and autoimmune diseases. Autoimmun. Rev. 2020, 19, 102468. [Google Scholar] [CrossRef]
- Schilirò, D.; Silvagni, E.; Ciribè, B.; Fattorini, F.; Maccarrone, V.; Elefante, E.; Signorini, V.; Zucchi, D.; Cardelli, C.; Bortoluzzi, A.; et al. Systemic Lupus Erythematosus: One Year in Review 2024. Clin. Exp. Rheumatol. 2024, 42, 583–592. [Google Scholar]
- Fanouriakis, A.; Tziolos, N.; Bertsias, G.; Boumpas, D.T. Update on the Diagnosis and Management of Systemic Lupus Erythematosus. Ann. Rheum. Dis. 2020, 80, 14–25. [Google Scholar]
- Suda, M.; Kishimoto, M.; Ohde, S.; Okada, M. Validation of the 2019 ACR/EULAR classification criteria of systemic lupus erythematosus in 100 Japanese patients: A real-world setting analysis. Clin. Rheumatol. 2020, 39, 1823–1827. [Google Scholar] [CrossRef]
- Piga, M.; Tselios, K.; Viveiros, L.; Chessa, E.; Neves, A.; Urowitz, M.B.; Isenberg, D. Clinical patterns of disease: From early systemic lupus erythematosus to late-onset disease. Best Pract. Res. Clin. Rheumatol. 2023, 37, 101938. [Google Scholar] [CrossRef]
- Rovenský, J.; Tuchyňová, A. Systemic lupus erythematosus in the elderly. Autoimmun. Rev. 2008, 7, 235–239. [Google Scholar]
- Fanouriakis, A.; Kostopoulou, M.; Andersen, J.; Aringer, M.; Arnaud, L.; Bae, S.-C.; Boletis, J.; Bruce, I.N.; Cervera, R.; Doria, A.; et al. EULAR recommendations for the management of systemic lupus erythematosus: 2023 update. Ann. Rheum. Dis. 2024, 83, 15–29. [Google Scholar]
- Xu, Y.X.; Tan, Y.; Yu, F.; Zhao, M.H. Late onset lupus nephritis in Chinese patients: Classified by the 2003 International Society of Nephrology and Renal Pathology Society system. Lupus 2011, 20, 801–808. [Google Scholar]
- Kutky, M.; Aloudat, S. Late-Onset Systemic Lupus Erythematosus with Lupus Nephritis in a 74-Year-Old Male: A Brief Case and Review. Can. J. Kidney Health Dis. 2018, 5, 2054358118793397. [Google Scholar] [CrossRef]
- Aljohani, R.; Gladman, D.D.; Su, J.; Urowitz, M.B. Disease evolution in late-onset and early-onset systemic lupus erythematosus. Lupus 2017, 26, 1190–1196. [Google Scholar] [PubMed]
- Wilhelmus, S.; Bajema, I.M.; Bertsias, G.K.; Boumpas, D.T.; Gordon, C.; Lightstone, L.; Tesar, V.; Jayne, D.R. Lupus nephritis management guidelines compared. Nephrol. Dial. Transplant. 2016, 31, 904–913. [Google Scholar] [PubMed]
- Catoggio, L.J.; Soriano, E.R.; Imamura, P.M.; Wojdyla, D.; Jacobelli, S.; Massardo, L.; Díaz, R.C.; Guibert-Toledano, M.; Alvarellos, A.; Saurit, V.; et al. Late-onset systemic lupus erythematosus in Latin Americans: A distinct subgroup? Lupus 2015, 24, 788–795. [Google Scholar]
- Ahn, S.S.; Yoo, J.; Jung, S.M.; Song, J.J.; Park, Y.B.; Lee, S.W. Comparison of clinical features and outcomes between patients with early and delayed lupus nephritis. BMC Nephrol. 2020, 21, 258. [Google Scholar] [CrossRef]
- Delfino, J.; Dos Santos, T.A.F.G.; Skare, T.L. Comparison of lupus patients with early and late onset nephritis: A study in 71 patients from a single referral center. Adv. Rheumatol. 2020, 60, 5. [Google Scholar]
- Zucchi, D.; Cardelli, C.; Elefante, E.; Tani, C.; Mosca, M. Treat-to-Target in Systemic Lupus Erythematosus: Reality or Pipe Dream. J. Clin. Med. 2023, 12, 3348. [Google Scholar] [CrossRef]
- Tomic-Lucic, A.; Petrovic, R.; Radak-Perovic, M.; Milovanovic, D.; Milovanovic, J.; Zivanovic, S.; Pantovic, S.; Veselinovic, M. Late-onset systemic lupus erythematosus: Clinical features, course, and prognosis. Clin. Rheumatol. 2013, 32, 1053–1058. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).