Abstract
Pregnancy-related acute kidney injury (pAKI), preeclampsia (PE), and the hypertensive disorders of pregnancy are closely related conditions, which are, in turn, frequently linked to pre-existing and often non-diagnosed chronic kidney disease (CKD). The current literature and research mainly underline the effects of pregnancy complications on the offspring; this review strongly emphasizes the maternal health as well. These conditions not only negatively affect pregnancy outcomes, but have a relevant effect on the future health of affected mothers and their children. Therefore, dedicated diagnostic and follow-up programs are needed, for optimizing materno-foetal health and reducing the impact of pregnancy-related problems in the mothers and in the new generations. This narrative review, performed on the occasion of the 2018 World Kidney Day dedicated to women’s health, focuses on three aspects of the problem. Firstly, the risk of AKI in the hypertensive disorders of pregnancy (the risk is the highest in developing countries; however PE is the main cause of pregnancy related AKI worldwide). Secondly, the effect of AKI and the hypertensive disorders of pregnancy on the development of CKD in the mother and offspring: long-term risks are increased; the entity and the trajectories are still unknown. Thirdly, the role of CKD in the pathogenesis of AKI and the hypertensive disorders of pregnancy: CKD is a major risk factor and the most important element in the differential diagnosis; pregnancy is a precious occasion for early diagnosis of CKD. Higher awareness on the importance of AKI in pregnancy is needed to improve short and long term outcomes in mothers and children.
1. Introduction
Acute kidney injury (AKI) is still an enormous unsolved health-care problem worldwide [1,2,3]. Pregnancy-related AKI (p-AKI) is one of the most common causes of acute kidney injury in young women: it has not disappeared in high-income countries and is the leading cause of AKI in women in the developing world [1,2,3,4,5,6,7,8,9,10,11,12,13]. Even though in the past AKI was generally considered to be an all-or-nothing situation, in which complete reversal was the rule in surviving patients, this condition is now known to be associated with future risk for chronic kidney disease (CKD), hypertension, and cardiovascular diseases [14,15,16,17,18].
This narrative review, performed on the occasion of World Kidney Day 2018, dedicated to women and kidney diseases, will discuss the complex relationship between preeclampsia (PE), CKD and AKI, in order to identify fields for future intervention and research, and enable us to improve these fundamental aspects of women’s health.
2. Pregnancy-Related AKI and Its Relationship with the Hypertensive Disorders of Pregnancy
Pregnancy is a physiological situation involving the risk of death. The risk varies widely: it is modulated not only by genetic background and environmental factors, but also by country and individual income, and disempowered women in low-income countries have the highest rate of death from pregnancy-related causes, among which AKI plays a major role [13,19,20,21]. These women are not only economically disempowered, they are socially and educationally disempowered as well, leading to their inability to make informed health choices and, thus, avoid adverse outcomes.
According to Women Aid International, the probability that a pregnant African woman will die from pregnancy-related complications has been reported to be as high as 1:20, in sharp contrast with 1:2000 in the developed world. The main causes are haemorrhage, sepsis, preeclampsia-eclampsia and septic abortion, all of which also cause of AKI [13]. According to the World Health Organisation, the Maternal Mortality Ratio is 12/100,000 in developed regions and 239/100,000 in developing ones [14]. As a consequence, the pattern of p-AKI can serve as an indirect, but interesting marker of the quality of health-care delivery in pregnancy (Table 1).

Table 1.
A general, but non-exhaustive classification of the main causes of pregnancy-related AKI, in the early and late phases of pregnancy.
The causes of p-AKI vary from one country to another, and laws on abortion and assisted fertilization are important factors as well. Septic abortion after an illegal procedure is the leading cause of early p-AKI in countries where legal abortions are not available, while PE after assisted fertilization (e.g., donation in particular) is now becoming a potential cause of p-AKI or of subsequent renal damage in developed countries [22,23,24,25,26,27,28].
As will be further discussed, the role of undiagnosed CKD, presenting as AKI in pregnancy is probably higher in developing countries, thus posing difficult clinical and ethical problems, in particular with regard to the start of chronic dialysis in pregnancy [6,29].
An in-depth discussion of all the causes of p-AKI is beyond the scope of this review, as it touches too many fields of medicine (nephrology, urology, infectious diseases, haematology, and immunology) and involves too many sensitive issues, such as availability and access to health care or legalisation on abortion. Many of these issues are addressed in the International Society of Nephrology’s wide-ranging “0 by 25” project, aimed at avoiding all preventable deaths due to AKI worldwide by 2025 [1,2,3].
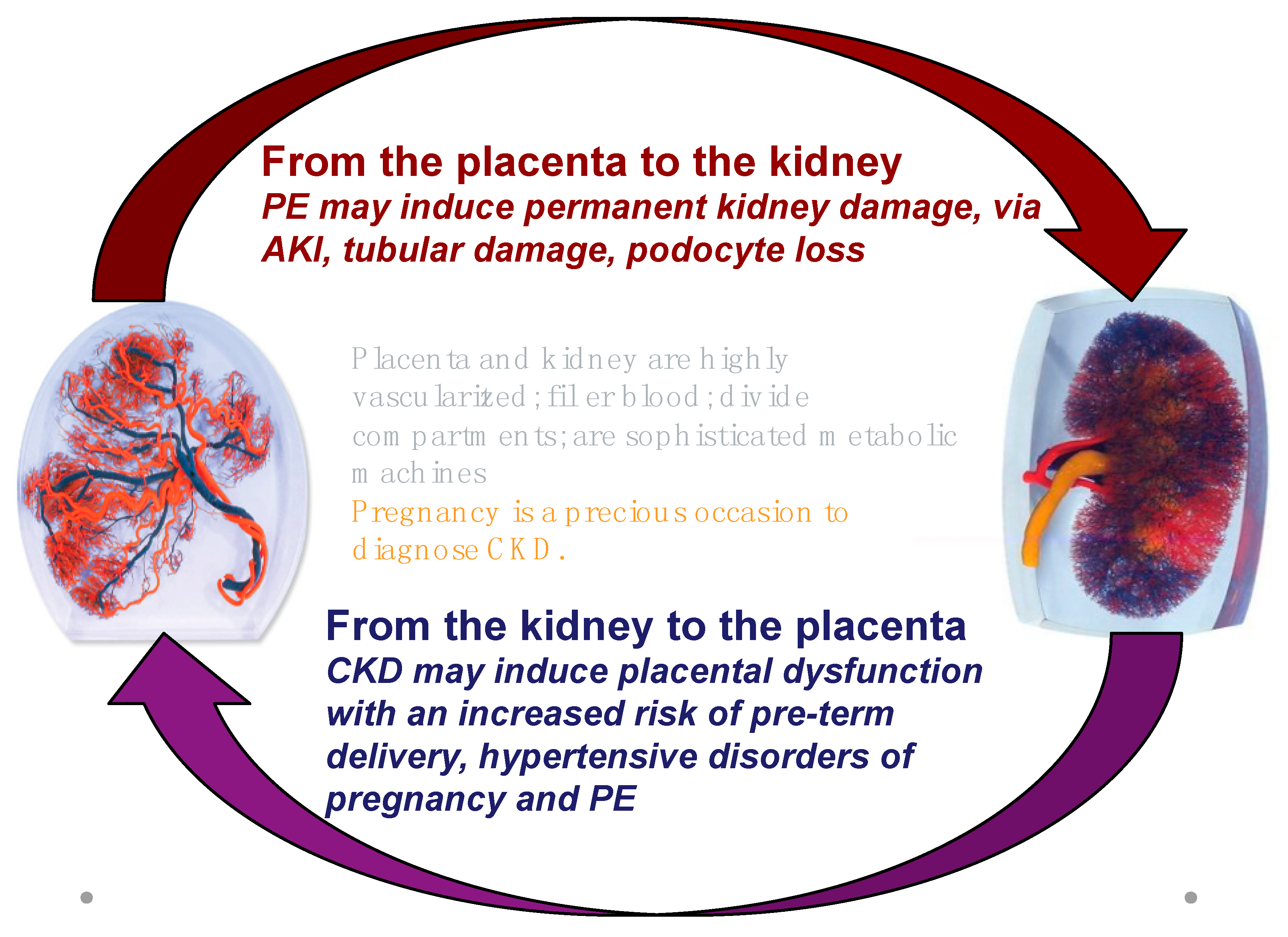
Therefore, while referring to the project for all issues of general importance, we will focus on the multifaceted relationship between kidney and placenta: CKD is a risk factor for PE; most of the risk factors for PE are also risk factors for CKD (diabetes, immunologic diseases, hypertension, obesity, and the metabolic syndrome); PE is a risk factor for the development of CKD later in life (Figure 1 and Figure 2).
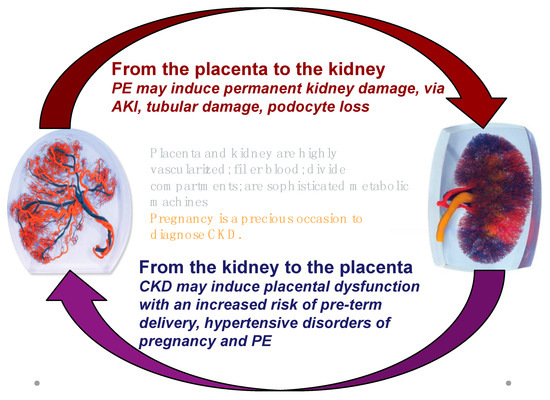
Figure 1.
The kidney-placenta crosstalk in pregnancy (modified from the World Kidney Day 2018 editorial).
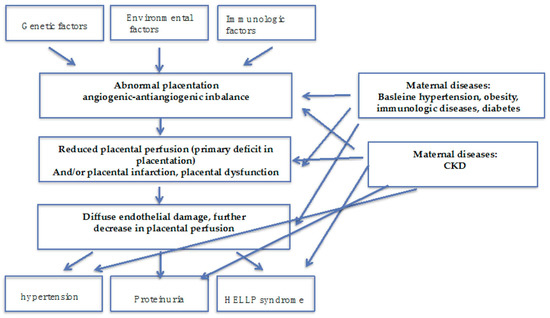
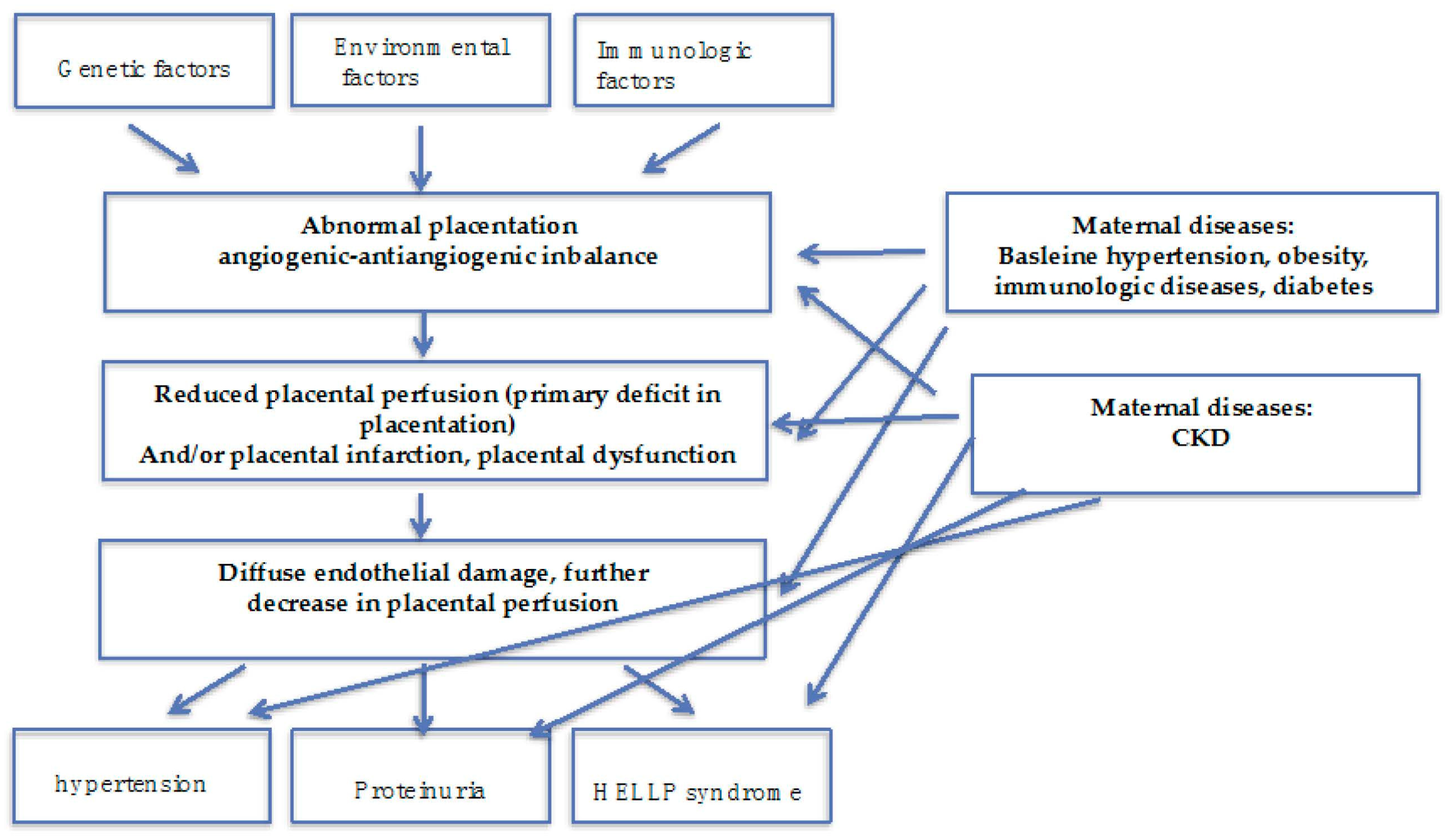
Figure 2.
Preeclampsia is a multifaceted syndrome: some pathophysiology insights. PE may be result from a deficit in placentation, in the absence of maternal diseases; the presence of maternal diseases, mainly affecting the kidney (hypertension, diabetes, immunologic diseases) and of CKD may act at different levels modulating onset and severity of the “PE syndrome”.
The following issues will be therefore discussed: (a) The risk of AKI in the hypertensive disorders of pregnancy; (b) the effect of AKI and the hypertensive disorders of pregnancy on the development of CKD in the mother and in the offspring; (c) the role of CKD in the pathogenesis of AKI and the hypertensive disorders of pregnancy (the latter point will be further developed in a twin review on CKD and pregnancy).
3. The Risk of AKI in the Hypertensive Disorders of Pregnancy
The hypertensive disorders of pregnancy encompass a wide range of conditions of different severity, impact on kidney function and on future health [30,31,32,33,34,35]. To some extent their incidence varies according to how the conditions are defined, and to whether “superimposed” disorders are included. Overall, preeclampsia (PE) accounts for about one third of all hypertensive disorders, with a prevalence ranging from about 2%, considering only the cases developing in the absence of predisposing factors (“low-risk” pregnancy), to over 5% in unselected populations [30,31,32,33,34,35]. The modifications in the definitions over time may partially impair comparisons of historic data [30,31,32,33,34,35]. The classifications are however different, in particular for severity, as one condition (mild PE) can develop into another (severe PE); furthermore, while early PE is often severe, and is usually associated with altered biomarkers, in keeping with a primary defect of placentation, late PE may be severe and life threatening (Table 2).

Table 2.
Preeclampsia: focus on classifications and definitions.
Similar considerations apply to the hypertensive disorders of pregnancy, ranging from less than 10% to over 15% of pregnancies, according to whether the following disorders are included: chronic pre-conception hypertension; HELLP syndrome, the acronym for haemolysis-low platelets elevated liver enzymes, and intrauterine growth restriction (IUGR) [36,37,38,39,40]. In fact, the relationship between PE, pregnancy-induced hypertension (PIH), and HELLP is not fully clear and other conditions, such as intrauterine growth restriction, probably share a common pathogenesis [40,41,42,43,44,45].
Whatever the causes, even if proteinuria, which is one of the hallmarks of PE, is usually considered as a marker of glomerular involvement, and podocyte damage is regarded as a main pathogenetic pathway, tubular damage is probably more common than previously considered [46]. In PE and all the kidney structures may be affected, ultimately resulting in p-AKI, the common pathway of other pregnancy- related noxae, such as sepsis and shock [47,48,49,50] (Table 3, Figure 3). Once more, socioeconomic status is a major component of the risk of both incidence and severity [51,52,53].

Table 3.
Kidney involvement in pregnancy: pathologic findings.
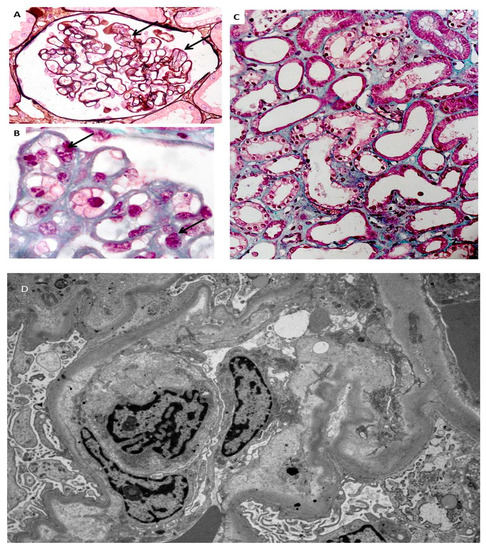
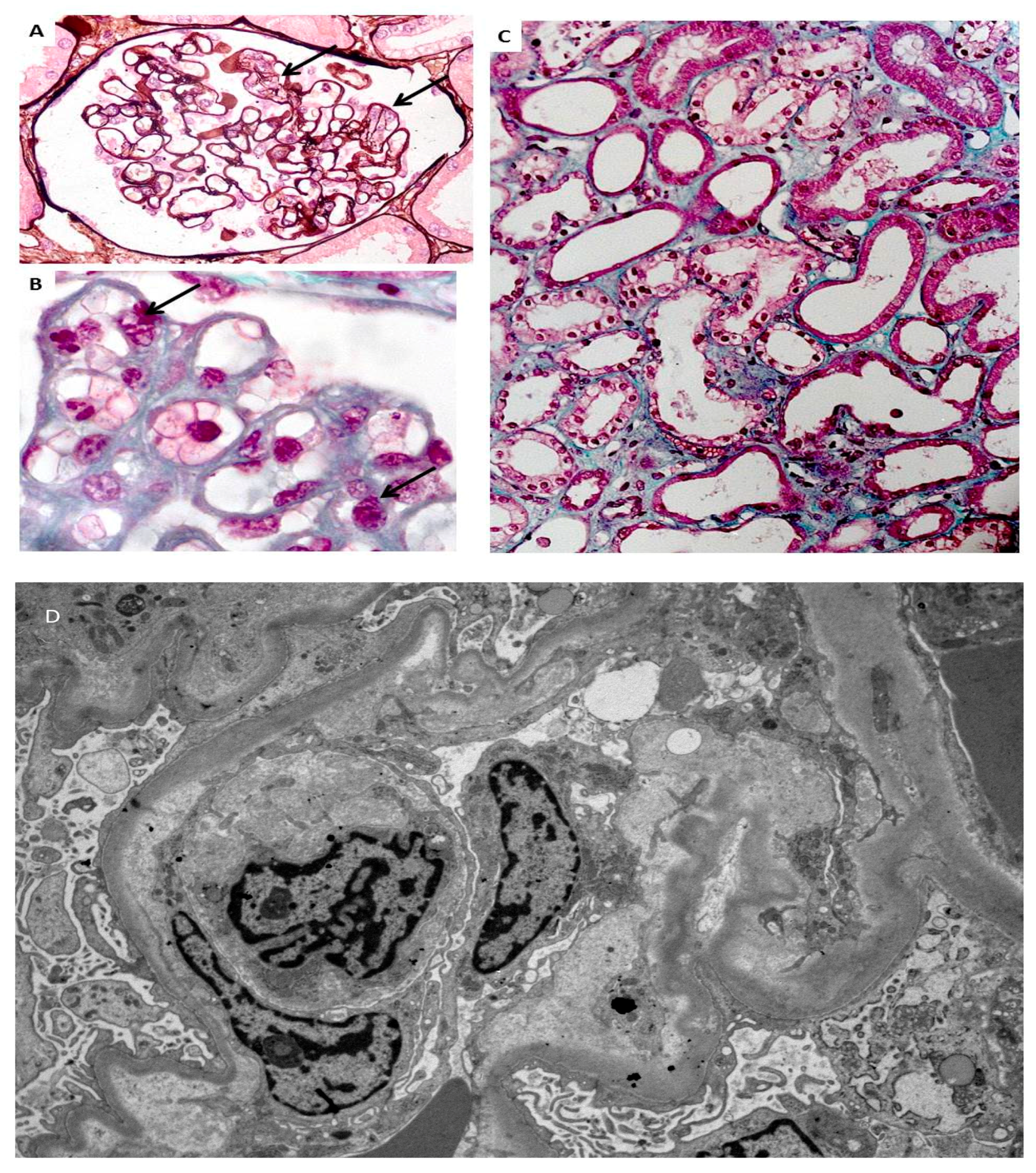
Figure 3.
Endotheliosis is defined by endothelial changes in renal glomeruli, combining swollen endothelial cells leading to narrowed capillary lumen (A,B). Foci of acute tubular necrosis are present (C). Electronic microscopy shows endothelial changes (D) (modified from [47], with permission of the authors).
While our understanding of pathogenic mechanisms is improving, we have to acknowledge that our knowledge of the incidence of these complications of the hypertensive disorders of pregnancy is still severely limited, in particular in middle- and low-income countries, a gap which is all the more important, given the estimation of a higher incidence of both AKI and the hypertensive disorders of pregnancy in these settings [54,55].
4. The Effect of AKI and the Hypertensive Disorders of Pregnancy on the Development of CKD in Mother and Child
AKI, PE and CKD are intrinsically related, and kidney damage may represent the common link (Figure 1). A large body of evidence is accumulating on the effect of all kinds of AKI on the future development of CKD, and, in a circular spiral of risk, also affecting the hypertensive disorders of pregnancy [15,16,17,18,56,57,58,59,60].
Evidence of the long-term effect of p-AKI on the future development of CKD is limited. However, there is a growing number of studies addressed to describing the relationship between PE and the hypertensive disorders of pregnancy and the development of cardiovascular diseases and CKD [61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87]. The data from the literature are somewhat conflicting, as a result of the heterogeneity of the studies and the differences in the definitions they adopt. None of the available studies is prospective. Within these limits, previous PE is associated with a short-term risk of albuminuria and long-term risk of end-stage renal disease (ESRD) [64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87]. Further research on this topic is needed to make it possible to tailor long- term interventions.
It is likely that p-AKI increases the risk of CKD, which is already higher after PE, for reasons still to be fully elucidated; as previously mentioned, as podocyte loss is a hallmark of PE, this suggests the cause is permanent glomerular damage. Endotheliosis sometimes heralds glomerulosclerosis; tubular and vascular damage may co-exist (Table 3, Figure 1, Figure 2 and Figure 3). A large recent study provides additional evidence linking AKI, PE and CKD and suggests that previous AKI is a risk factor for PE and the hypertensive disorders of pregnancy [56]. Conversely, the limit between “physiologic and pathologic” pregnancies is not fully clear, and a consistent overlap exists for pathologic findings [88,89]. For this reason, the position of the Italian Society of Nephrology (SIN), the only one so far made available by a nephrology society, is in favour of long-term follow-up of PE patients [90]; it is the only society to explicitly support this approach, based on current data. Adoption of the practice of tracking individuals longitudinally should be possible in high-income countries, especially those with electronic medical records, and this should be encouraged. In addition to maternal risks, PE is associated with intrauterine and perinatal death, pre-term delivery, and restricted intrauterine growth, the last two of which result in “small babies” (Table 4) [30,31,32].

Table 4.
“Small babies”: focus on classifications and definitions.
Small babies and preterm babies have a highly increased risk of neurological deficits (a risk that is inversely proportional to gestational age), and post-natal complications, in particular those of septic origin, with further potential long-term detrimental effects [91,92,93,94,95,96,97,98,99,100,101,102]. The risks are higher in low- and mid-income countries, since survival and deficit-free survival depends to a large extent on providing intensive postnatal care [101,102,103,104,105,106,107,108].
Since kidney development is completed in the last phases of pregnancy, delayed, insufficient kidney growth, resulting in a low nephron number is the basis of the increased risk of CKD in adulthood; besides this, CKD, small, and preterm babies are at risk for the development of diabetes, metabolic syndrome, and cardiovascular diseases in adulthood [100,101,102,109,110,111,112]. The question of whether all preterm babies or only/mainly babies that are small for gestational age are at increased risk for kidney and metabolic diseases is still open, and its clarification will be useful in targeting future interventions. Interestingly, delivery of a small for gestational age baby is also a marker of risk of future CKD and cardiovascular diseases in the mother [113,114,115].
5. The Role of CKD in the Pathogenesis of AKI and Hypertensive Disorders of Pregnancy
Starting in its early stages, in which it is often asymptomatic, CKD is a risk factor for PE, the hypertensive disorders of pregnancy, and preterm delivery; conversely, PE may be the first sign of undiagnosed CKD [6,90,116,117,118,119,120].
CKD and PE share many signs and symptoms (hypertension, proteinuria and increases in serum creatinine); while differential diagnosis may be difficult, it is not impossible during pregnancy. When signs and symptoms are correctly interpreted, and follow-up is continued after delivery, pregnancy can be a precious occasion to diagnose CKD [6,90,116,117,118,119,120,121,122,123,124].
The differential diagnosis can be pragmatically supported by the distinction between placental and maternal preeclampsia, the first of which is linked to a primary defect in placentation, and associated with high levels of anti-angiogenic factors and by a deep imbalance between angiogenic and anti-angiogenic factors, with impaired utero-placental flows, while the latter is a secondary effect exerted by maternal diseases, and can be expected to display a lesser and later angiogenic/non-angiogenic imbalance, together with relatively well-preserved utero-placental flows [121,123,124,125,126,127].
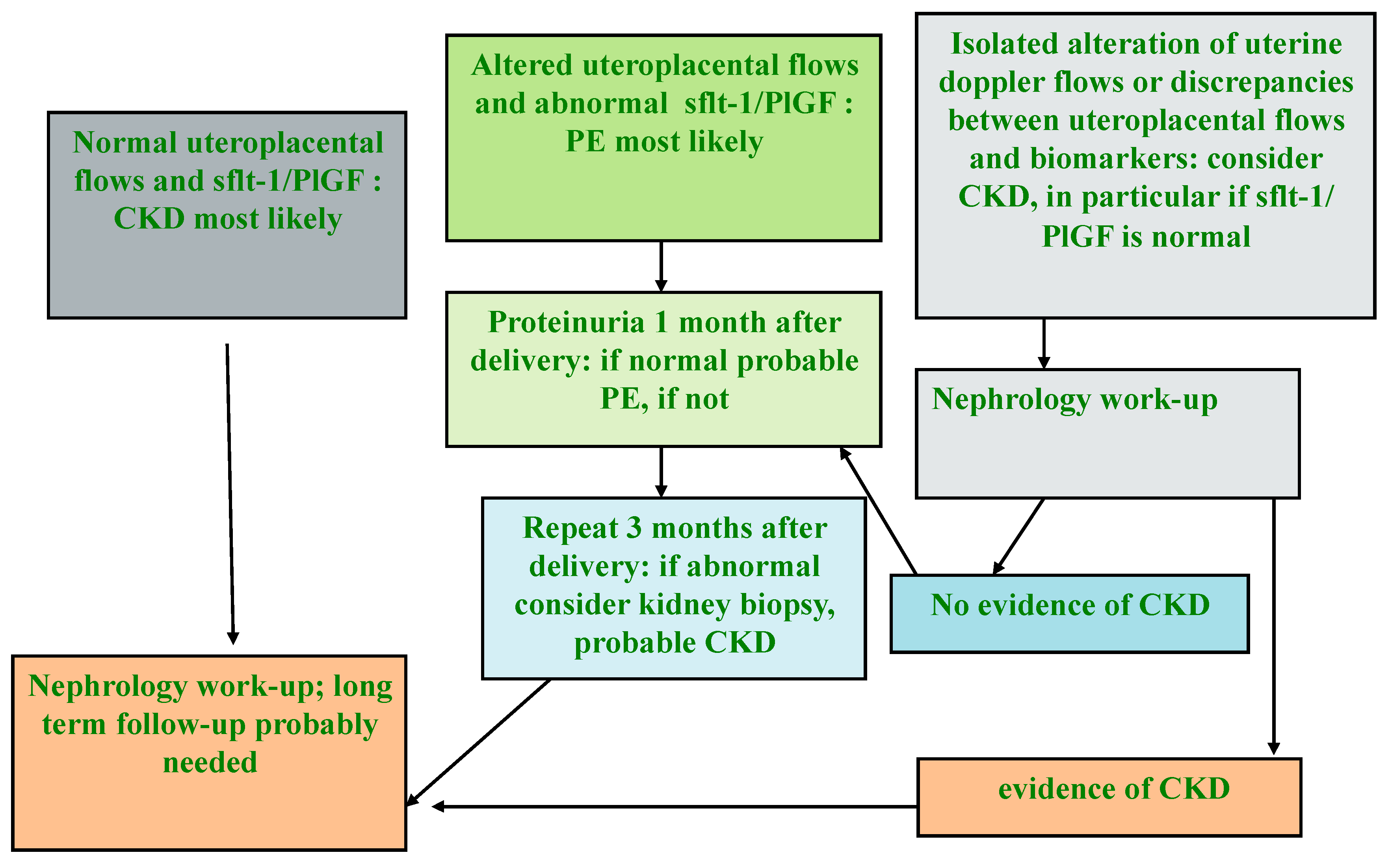
It is conceivable that this Manichean differentiation does not capture all the important issues in the pathogenesis of PE, and that a “mixed” pathogenesis may frequently be present. However, the analysis of circulating biomarkers, namely Soluble fms-like tyrosine kinase- (sFlt-1) and placental growth factor (PlGF), the two that have most often been studied in detail), and of utero-placental flows, where available (Doppler study is economically less demanding, and can be performed rapidly at the bedside) can support the differential diagnosis between CKD and PE and help guide post-pregnancy follow-up (Figure 4) [123,124,125,126,127].
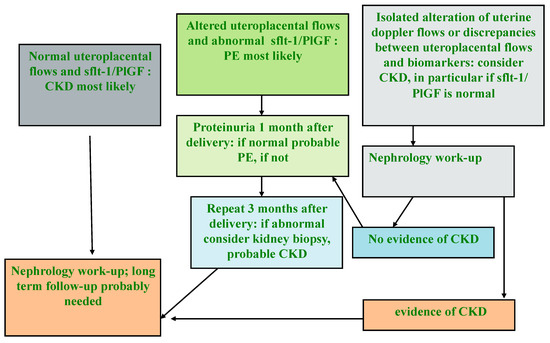
Figure 4.
Flow chart for the differential diagnosis between CKD and PE (modified by the Authors from [90]).
Moreover, because making a differential diagnosis between acute damage and chronic kidney disease (AKI vs CKD) may be difficult during pregnancy, it is important to carry out extensive kidney evaluation after delivery, once the physiological changes related to pregnancy have been resolved (Figure 4).
Unfortunately, these intentions may be impossible to put into practice in situations where they are in contrast with the harsh reality of insufficient care and inadequate attention to the early diagnosis of CKD. Long-term studies are needed to establish the link between the different clinical and subtle-subclinical changes observed in formerly preeclamptic women, as an ongoing investment in health care for the young.
6. The Role of Immunologic Diseases in the Pathogenesis of the Hypertensive Disorders of Pregnancy and p-AKI
Among the immunologic diseases, systemic lupus erythematosus (SLE) principally affects women of childbearing age, and is the most recognized risk factor for the hypertensive disorders of pregnancy and p-AKI [128,129,130,131].
The most common feature of kidney damage in SLE is lupus nephritis (LN), characterised by glomerular involvement, but interstitial and vascular lesions also contribute to CKD. Patients affected by SLE share with other CKD patients the traditional risk factors for adverse pregnancy outcomes, but they have also specific risks of p-AKI including lupus flares, preeclampsia, HELLP syndrome, and thrombotic events. The major predictors for acute pregnancy-related complications are Class III and IV lupus nephritis, a previous history of renal flares, longer disease, hypocomplementemia, antiphospholipid syndrome (APS), and the presence of antiphospholipid antibodies [132,133,134,135,136]. In this context, high titres and triple positivity for antiphospholipid antibodies, previous thrombosis and the presence of a lupus anticoagulant are associated with a higher risk of severe maternal and foetal complications, both in primary and secondary APS, and in catastrophic APS (CAPS) the most severe form of the syndrome [136,137,138,139,140,141,142]. Conversely, the occurrence of the HELLP syndrome in a patient with APS should raise the suspicion that CAPS will shortly be manifested [139,141,142,143].
In addition to CAPS, thrombotic thrombocytopenic purpura and atypical haemolytic-uremic syndrome comprise the continuum of pregnancy-related thrombotic microangiopathies (TMA), associated with high maternal and foetal morbidity and mortality, including p-AKI [139,144,145,146,147]. Pregnancy, itself a procoagulant state, is a trigger for thrombotic thrombocytopenic purpura, especially in the setting of ADAMTS-13 deficiency. A deficiency of ADAMTS-13 is responsible for most cases of thrombotic thrombocytopenic purpura, generally occurring in the second and third trimester of pregnancy. Pregnancy also induces complement activation and is a trigger for atypical haemolytic uremic syndrome, associated with complement dysregulation, which mainly occurs post partum [139,144,145,146,147].
After delivery, inflammation, the release of foetal cells in the maternal circulation, infections, and haemorrhage can lead to activation of the alternative complement pathway, which, in the absence of effective regulatory mechanisms, may induce postpartum atypical haemolytic-uremic syndrome (aHUS); complement dysregulation was also found to be associated with the HELLP syndrome, which shares several features with pregnancy-associated TMA [148,149,150,151,152]. The new frontiers of treatment with eculizumab make diagnosis of pivotal importance for allowing timely treatment [149,150,151,153].
7. Conclusions: The Need for Research and Clinical Intervention
Research is increasingly being done on the short- and long-term effects of p-AKI and the hypertensive disorders of pregnancy on the health of mothers and children, and studies are generally in agreement in highlighting an increased risk for both [60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87]. However, study designs and definitions are heterogeneous and there is need for establishing and validating a common lexicon in this regard, to allow integration of the available data and, more importantly, make possible a detailed, comparable set of information for the future studies.
While information on the short-term effects of p-AKI and the hypertensive disorders of pregnancy is available in differently resourced settings, data on the long-term effects are limited to highly-resourced countries, and even then tend to be sparse and non-uniform, thus underlining the need for more research in the fragile populations that are most susceptible both to p-AKI and other kidney diseases.
While the new generation of biomarkers of preeclampsia and related disorders facilitates better understanding of the pathogenesis and prognosis of the hypertensive disorders of pregnancy, there is a need for reliable, simple and inexpensive prognostic markers of the effects of p-AKI and the hypertensive disorders of pregnancy on future health. Clinical care of p-AKI, including dialysis care, should be a priority, as maternal health is fundamental not only for the patient, but also for her family and society [1,2,3].
Treatment for p-AKI and the hypertensive disorders of pregnancy should not be discontinued at delivery or shortly afterwards, and long-term programs should be established at least for women with evidence of chronic kidney disease. Children born to p-AKI mothers, born pre-term or small for gestational age should be followed up to optimize prognosis. All these are ambitious tasks. It will be up to the nephrology community to ensure that they are not seen as being overambitious.
Author Contributions
Conceptualization, G.B.P., A.L., E.Z., R.A., M.I.H., B.C., M.A., Z.-H.L., G.A., A.O.G.; Writing—Original Draft Preparation, G.B.P., R.A., G.C.; Writing—Review & Editing, G.B.P., A.L., E.Z., R.A., M.I.H., B.C., M.A., Z.-H.L., G.A., A.O.G.; Supervision, G.C., P.K.T.L., G.G.-G.
Acknowledgments
Authors want to thank Susan Finnel for her careful language editing.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Mehta, R.L.; Cerdá, J.; Burdmann, E.A.; Tonelli, M.; García-García, G.; Jha, V. International Society of Nephrology's 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): A human rights case for nephrology. Lancet 2015, 385, 2616–2643. [Google Scholar] [CrossRef]
- Macedo, E.; Mehta, R.L. Preventing Acute Kidney Injury. Crit. Care Clin. 2015, 31, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Perico, N.; Remuzzi, G. Prevention programs for chronic kidney disease in low-income countries. Intern. Emerg. Med. 2016, 11, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Jim, B.; Garovic, V.D. Acute Kidney Injury in Pregnancy. Semin. Nephrol. 2017, 37, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, F.; Deltombe, C. Pregnancy-related acute kidney injury in high income countries: Still a critical issue. J. Nephrol. 2017, 30, 767–771. [Google Scholar] [CrossRef] [PubMed]
- Ibarra-Hernández, M.; Orozco-Guillén, O.A.; de la Alcantar-Vallín, M.L.; Garrido-Roldan, R.; Jiménez-Alvarado, M.P.; Castro, K.B.; Villa-Villagrana, F.; Borbolla, M.; Gallardo-Gaona, J.M.; García-García, G.; et al. Acute kidney injury in pregnancy and the role of underlying CKD: A point of view from México. J. Nephrol. 2017, 30, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Prakash, J.; Pant, P.; Prakash, S.; Sivasankar, M.; Vohra, R.; Doley, P.K.; Pandey, L.K.; Singh, U. Changing picture of acute kidney injury in pregnancy: Study of 259 cases over a period of 33 years. Indian J. Nephrol. 2016, 26, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Ahmed, M.M.; Kedir, S.; Bekele, D. Clinical profile and outcome of patients with acute kidney injury requiring dialysis–An experience from a haemodialysis unit in a developing country. BMC Nephrol. 2016, 17, 91. [Google Scholar] [CrossRef] [PubMed]
- Bentata, Y.; Housni, B.; Mimouni, A.; Azzouzi, A.; Abouqal, R. Acute kidney injury related to pregnancy in developing countries: Etiology and risk factors in an intensive care unit. J. Nephrol. 2012, 25, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Jha, V.; Chugh, K.S. Community-acquired acute kidney injury in Asia. Semin. Nephrol. 2008, 28, 330–347. [Google Scholar] [CrossRef] [PubMed]
- Naicker, S.; Aboud, O.; Gharbi, M.B. Epidemiology of acute kidney injury in Africa. Semin. Nephrol. 2008, 28, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, R.; Yu, L.; Younes-Ibrahim, M.; Schor, N.; Burdmann, E.A. Epidemiology of acute kidney injury in Latin America. Semin. Nephrol. 2008, 28, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Cerdá, J.; Bagga, A.; Kher, V.; Chakravarthi, R.M. The contrasting characteristics of acute kidney injury in developed and developing countries. Nat. Clin. Pract. Nephrol. 2008, 4, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Trends in Maternal Mortality: 1990 to 2015. Available online: http://www.who.int/reproductivehealth/publications/monitoring/maternal-mortality-2015/en/ (accessed on 15 July 2018).
- Parr, S.K.; Matheny, M.E.; Abdel-Kader, K.; Greevy, R.A.; Bian, A., Jr.; Fly, J.; Chen, G.; Speroff, T.; Hung, A.M.; Ikizler, T.A.; et al. Acute kidney injury is a risk factor for subsequent proteinuria. Kidney Int. 2018, 93, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Hsu, R.K.; Hsu, C.Y. The Role of Acute Kidney Injury in Chronic Kidney Disease. Semin. Nephrol. 2016, 36, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Mammen, C.; Al Abbas, A.; Skippen, P.; Nadel, H.; Levine, D.; Collet, J.P.; Matsell, D.G. Long-term risk of CKD in children surviving episodes of acute kidney injury in the intensive care unit: A prospective cohort study. Am. J. Kidney Dis. 2012, 59, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Silver, S.A.; Siew, E.D. Follow-up Care in Acute Kidney Injury: Lost in Transition. Adv. Chronic Kidney Dis. 2017, 24, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ma, X.; Zheng, J.; Liu, X.; Yan, T. Pregnancy outcomes in patients with acute kidney injury during pregnancy: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2017, 17, 235. [Google Scholar] [CrossRef] [PubMed]
- Oladapo, O.T.; Adetoro, O.O.; Ekele, B.A.; Chama, C.; Etuk, S.J.; Aboyeji, A.P.; Onah, H.E.; Abasiattai, A.M.; Adamu, A.N.; et al. Nigeria Near-miss and Maternal Death Surveillance Network. When getting there is not enough: A nationwide cross-sectional study of 998 maternal deaths and 1451 near-misses in public tertiary hospitals in a low-income country. BJOG 2016, 123, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Theilen, L.H.; Fraser, A.; Hollingshaus, M.S.; Schliep, K.C.; Varner, M.W.; Smith, K.R.; Esplin, M.S. All-Cause and Cause-Specific Mortality After Hypertensive Disease of Pregnancy. Obstet. Gynecol. 2016, 128, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Acharya, A. Management of Acute Kidney Injury in Pregnancy for the Obstetrician. Obstet. Gynecol. Clin. N. Am. 2016, 43, 747–765. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, R.; Rosa-Diez, G.; Ferreiro, A.; Greloni, G.; Yu, L.; Younes-Ibrahim, M.; Burdmann, E.A. Acute kidney injury in Latin America: A view on renal replacement therapy resources. Nephrol. Dial. Transplant. 2014, 29, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Nwoko, R.; Plecas, D.; Garovic, V.D. Acute kidney injury in the pregnant patient. Clin. Nephrol. 2012, 78, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Srinil, S.; Panaput, T. Acute kidney injury complicating septic unsafe abortion: Clinical course and treatment outcomes of 44 cases. J. Obstet. Gynaecol. Res. 2011, 37, 1525–1531. [Google Scholar] [CrossRef] [PubMed]
- Sabban, H.; Zakhari, A.; Patenaude, V.; Tulandi, T.; Abenhaim, H.A. Obstetrical and perinatal morbidity and mortality among in-vitro fertilization pregnancies: A population-based study. Arch. Gynecol. Obstet. 2017, 296, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Storgaard, M.; Loft, A.; Bergh, C.; Wennerholm, U.B.; Söderström-Anttila, V.; Romundstad, L.B.; Aittomaki, K.; Oldereid, N.; Forman, J.; Pinborg, A. Obstetric and neonatal complications in pregnancies conceived after oocyte donation: A systematic review and meta-analysis. BJOG 2017, 124, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, A.; García, D.; Rodríguez, A.; Vassena, R.; Figueras, F.; Vernaeve, V. Is oocyte donation a risk factor for preeclampsia? A systematic review and meta-analysis. J. Assist. Reprod. Genet. 2016, 33, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Orozco-Guillen, A.; Merodio-Anguiano, R.; Iyune-Cojab, E.; Piccoli, G.B.; Rodriguez-Bosh, M. A baby and a kidney neoplasia: A question of priorities. Kidney Int. 2016, 90, 1133. [Google Scholar] [CrossRef] [PubMed]
- Mol, B.W.; Roberts, C.T.; Thangaratinam, S.; Magee, L.A.; de Groot, C.J.; Hofmeyr, G.J. Pre-eclampsia. Lancet 2016, 387, 999–1011. [Google Scholar] [CrossRef]
- Brown, M.A.; Magee, L.A.; Kenny, L.C.; Karumanchi, S.A.; McCarthy, F.P.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Ishaku, S. The hypertensive disorders of pregnancy: ISSHP classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2018, 23, 24–43. [Google Scholar]
- Tranquilli, A.L.; Dekker, G.; Magee, L.; Roberts, J.; Sibai, B.M.; Steyn, W.; Zeeman, G.G.; Brown, M.A. The classification, diagnosis and management of the hypertensive disorders of pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens. 2014, 4, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.A.; Lindheimer, M.D.; de Swiet, M.; van Assche, A.; Moutquin, J.M. The classification and diagnosis of the hypertensive disorders of pregnancy: Statement from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Hypertens. Pregnancy 2001, 20. [Google Scholar] [CrossRef] [PubMed]
- Kallela, J.; Jääskeläinen, T.; Kortelainen, E.; Heinonen, S.; Kajantie, E.; Kere, J.; Kivinen, K.; Pouta, A.; Laivuori, H. The diagnosis of pre-eclampsia using two revised classifications in the Finnish Pre-eclampsia Consortium (FINNPEC) cohort. BMC Pregnancy Childbirth 2016, 16, 221. [Google Scholar] [CrossRef] [PubMed]
- Tranquilli, A.L.; Brown, M.A.; Zeeman, G.G.; Dekker, G.; Sibai, B.M. The definition of severe and early-onset preeclampsia. Statements from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Pregnancy Hypertens. 2013, 3, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Helewa, M.E.; Burrows, R.F.; Smith, J.; Williams, K.; Brain, P.; Rabkin, S.W. Report of the Canadian Hypertension Society Consensus Conference: 1. Definitions, evaluation and classification of hypertensive disorders in pregnancy. CMAJ 1997, 157, 715–725. [Google Scholar] [PubMed]
- Ferrazzi, E.; Stampalija, T.; Monasta, L.; di Martino, D.; Vonck, S.; Gyselaers, W. Maternal hemodynamics: A method to classify hypertensive disorders of pregnancy. Am. J. Obstet. Gynecol. 2018, 218, 124. [Google Scholar] [CrossRef] [PubMed]
- Sjaus, A.; McKeen, D.M.; George, R.B. Hypertensive disorders of pregnancy. Can. J. Anaesth. 2016, 63, 1075–1097. [Google Scholar] [CrossRef] [PubMed]
- Magee, L.A.; Pels, A.; Helewa, M.; Rey, E.; von Dadelszen, P. The hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 643–657. [Google Scholar] [CrossRef] [PubMed]
- Wagner, S.J.; Barac, S.; Garovic, V.D. Hypertensive pregnancy disorders: Current concepts. J. Clin. Hypertens. (Greenwich) 2007, 9, 560–566. [Google Scholar] [CrossRef]
- Muñoz-Hernández, R.; Medrano-Campillo, P.; Miranda, M.L.; Macher, H.C.; Praena-Fernández, J.M.; Vallejo-Vaz, A.J.; Dominguez-Simeon, M.J.; Moreno-Luna, R.; Stiefel, P. Total and Fetal Circulating Cell-Free DNA, Angiogenic, and Antiangiogenic Factors in Preeclampsia and HELLP Syndrome. Am. J. Hypertens. 2017, 30, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Schaarschmidt, W.; Rana, S.; Stepan, H. The course of angiogenic factors in early-vs. late-onset preeclampsia and HELLP syndrome. J. Perinat. Med. 2013, 41, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Lind Malte, A.; Uldbjerg, N.; Wright, D.; Tørring, N. Prediction of severe pre-eclampsia/HELLP syndrome by combination of sFlt-1, CT-pro-ET-1 and blood pressure: Exploratory study. Ultrasound Obstet. Gynecol. 2018, 51, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Jim, B.; Karumanchi, S.A. Preeclampsia: Pathogenesis, Prevention, and Long-term Complications. Semin. Nephrol. 2017, 37, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Verlohren, S.; Perschel, F.H.; Thilaganathan, B.; Dröge, L.A.; Henrich, W.; Busjahn, A.; Khalil, A. Angiogenic Markers and Cardiovascular Indices in the Prediction of Hypertensive Disorders of Pregnancy. Hypertension 2017, 69, 1192–1197. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Fierro, M.L.; Hernández-Delgadillo, G.P.; Flores-Morales, V.; Cardenas-Vargas, E.; Mercado-Reyes, M.; Rodriguez-Sanchez, I.P.; Delgado-Enciso, I.; Galván-Tejada, C.E.; Galván-Tejada, J.I.; Celaya-Padilla, J.M. Current model systems for the study of preeclampsia. Exp. Biol. Med. (Maywood) 2018, 243, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Villie, P.; Dommergues, M.; Brocheriou, I.; Piccoli, G.B.; Tourret, J.; Hertig, A. Why kidneys fail post-partum: A tubulocentric viewpoint. J. Nephrol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Wagner, S.J.; Craici, I.M.; Grande, J.P.; Garovic, V.D. From placenta to podocyte: Vascular and podocyte pathophysiology in preeclampsia. Clin. Nephrol. 2012, 78, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Henao, D.E.; Saleem, M.A.; Cadavid, A.P. Glomerular disturbances in preeclampsia: Disruption between glomerular endothelium and podocyte symbiosis. Hypertens Pregnancy 2010, 29, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, M.; Yahata, K.; Ohno, S.; Koga, K.; Kikuchi, Y.; Hasegawa, C.; Seta, K.; Kuwahara, T.; Sugawara, A. Glomerular endotheliosis in a pregnant woman with severe gestational proteinuria. Intern. Med. 2013, 52, 1943–1947. [Google Scholar] [CrossRef] [PubMed]
- Von Dadelszen, P.; Magee, L.A. Preventing deaths due to the hypertensive disorders of pregnancy. Best. Pract. Res. Clin. Obstet. Gynaecol. 2016, 36, 83–102. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, I.R.; Silva, W.B.; Cerqueira, G.S.; Novo, N.F.; Almeida, F.A.; Novo, J.L. Maternal and fetal outcome in women with hypertensive disorders of pregnancy: The impact of prenatal care. Ther. Adv. Cardiovasc. Dis. 2015, 9, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Von Dadelszen, P.; Ansermino, J.M.; Dumont, G.; Hofmeyr, G.J.; Magee, L.A.; Mathai, M.; Sawchuck, D.; Teela, K.; Donnay, F.; Roberts, J.M. Improving maternal and perinatal outcomes in the hypertensive disorders of pregnancy: A vision of a community-focused approach. Int. J. Gynaecol. Obstet. 2012, 119, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Cerdá, J.; Mohan, S.; Garcia-Garcia, G.; Jha, V.; Samavedam, S.; Gowrishankar, S.; Bagga, A.; Chakravarthi, R.; Mehta, R. Acute Kidney Injury Recognition in Low- and Middle-Income Countries. Kidney Int. Rep. 2017, 2, 530–543. [Google Scholar] [CrossRef] [PubMed]
- Kashani, K.; Macedo, E.; Burdmann, E.A.; Hooi, L.S.; Khullar, D.; Bagga, A.; Chakravarthi, R.; Mehta, R. Acute Kidney Injury Risk Assessment: Differences and Similarities Between Resource-Limited and Resource-Rich Countries. Kidney Int. Rep. 2017, 2, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Tangren, J.S.; Powe, C.E.; Ankers, E.; Ecker, J.; Bramham, K.; Hladunewich, M.A.; Karumanchi, S.A.; Thadhani, R. Pregnancy Outcomes after Clinical Recovery from AKI. J. Am. Soc. Nephrol. 2017, 28, 1566–1574. [Google Scholar] [CrossRef] [PubMed]
- Chawla, L.S.; Kimmel, P.L. Acute kidney injury and chronic kidney disease: An integrated clinical syndrome. Kidney Int. 2012, 82, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Kaballo, M.A.; Elsayed, M.E.; Stack, A.G. Linking acute kidney injury to chronic kidney disease: The missing links. J. Nephrol. 2017, 30, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Pannu, N. Bidirectional relationships between acute kidney injury and chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2013, 22, 351–356. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Wei, Q.; Liu, J.; Yi, M.; Liu, Y.; Liu, H.; Sun, L.; Peng, Y.; Liu, F.; Venkatachalam, M.A.; et al. AKI on CKD: Heightened injury, suppressed repair, and the underlying mechanisms. Kidney Int. 2017, 92, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.C.; Best, K.E.; Pearce, M.S.; Waugh, J.; Robson, S.C.; Bell, R. Cardiovascular disease risk in women with pre-eclampsia: Systematic review and meta-analysis. Eur. J. Epidemiol. 2013, 28, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, L.; Casas, J.P.; Hingorani, A.D.; Williams, D.J. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: Systematic review and meta-analysis. BMJ 2007, 335, 974. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.; Dunford, J.; Mehran, R.; Robson, S.; Kunadian, V. Pre-eclampsia and future cardiovascular risk among women: A review. J. Am. Coll. Cardiol. 2014, 63, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Haththotuwa, R.; Kwok, C.S.; Babu, A.; Kotronias, R.A.; Rushton, C.; Zaman, A.; Fryer, A.A.; Kadam, U.; Chew-Graham, C.A.; et al. Preeclampsia and Future Cardiovascular Health: A Systematic Review and Meta-Analysis. Circ. Cardiovasc. Qual. Outcomes 2017, 10, e003497. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.D.; Han, Z.; Walsh, M.W.; Gerstein, H.C.; Devereaux, P.J. Kidney disease after preeclampsia: A systematic review and meta-analysis. Am. J. Kidney. Dis. 2010, 55, 1026–1039. [Google Scholar] [CrossRef] [PubMed]
- Paauw, N.D.; van der Graaf, A.M.; Bozoglan, R.; van der Ham, D.P.; Navis, G.; Gansevoort, R.T.; Groen, H.; Lely, A.T. Kidney Function After a Hypertensive Disorder of Pregnancy: A Longitudinal Study. Am. J. Kidney Dis. 2017, 71, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Van Balen Lopes, V.A.; Spaan, J.J.; Cornelis, T.; Heidema, W.M.; Scholten, R.R.; Spaanderman, M.E.A. Endothelial and kidney function in women with a history of preeclampsia and healthy parous controls: A case control study. Microvasc. Res. 2017, 116, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Tooher, J.; Thornton, C.; Makris, A.; Ogle, R.; Korda, A.; Hennessy, A. All Hypertensive Disorders of Pregnancy Increase the Risk of Future Cardiovascular Disease. Hypertension 2017, 70, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Bokslag, A.; Teunissen, P.W.; Franssen, C.; van Kesteren, F.; Kamp, O.; Ganzevoort, W.; Paulus, W.J.; de Groot, C.J.M. Effect of early-onset preeclampsia on cardiovascular risk in the fifth decade of life. Am. J. Obstet. Gynecol. 2017, 216, 523. [Google Scholar] [CrossRef] [PubMed]
- Paauw, N.D.; Joles, J.A.; Drost, J.T.; Verhaar, M.C.; Franx, A.; Navis, G.; Maas, A.H.; Lely, A.T. High-Normal Estimated Glomerular Filtration Rate in Early-Onset Preeclamptic Women 10 Years Postpartum. Hypertension 2016, 68, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Ayansina, D.; Black, C.; Hall, S.J.; Marks, A.; Millar, C.; Prescott, G.J.; Wilde, K.; Bhattacharya, S. Long term effects of gestational hypertension and pre-eclampsia on kidney function: Record linkage study. Pregnancy Hypertens. 2016, 6, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Kessous, R.; Shoham-Vardi, I.; Pariente, G.; Sergienko, R.; Sheiner, E. Long-term maternal atherosclerotic morbidity in women with pre-eclampsia. Heart 2015, 101, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.C.; Chen, S.H.; Ho, C.H.; Liang, F.W.; Chu, C.C.; Wang, H.Y.; Lu, Y.H. End-stage renal disease after hypertensive disorders in pregnancy. Am. J. Obstet. Gynecol. 2014, 210, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, M.K.; Hallan, S.; Svarstad, E.; Vikse, B.E. Preeclampsia and prevalence of microalbuminuria 10 years later. Clin. J. Am. Soc. Nephrol. 2013, 8, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Männistö, T.; Mendola, P.; Vääräsmäki, M.; Järvelin, M.R.; Hartikainen, A.L.; Pouta, A.; Suvanto, E. Elevated blood pressure in pregnancy and subsequent chronic disease risk. Circulation 2013, 127, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.K.; Muo, C.H.; Chang, Y.C.; Liang, C.C.; Chang, C.T.; Lin, S.Y.; Yen, T.H.; Chuang, F.R.; Chen, P.C.; Huang, C.C.; et al. Association between hypertensive disorders during pregnancy and end-stage renal disease: A population-based study. CMAJ 2013, 185, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Irgens, L.M.; Karumanchi, S.A.; Thadhani, R.; Reisæter, A.V.; Skjærven, R. Familial factors in the association between preeclampsia and later ESRD. Clin. J. Am. Soc. Nephrol. 2012, 7, 1819–1826. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.D.; Yusuf, S.; Walsh, M.W.; Lonn, E.; Teo, K.; Anand, S.S.; Pogue, J.; Islam, S.; Devereaux, P.J.; Gerstein, H.C. Increased cardiovascular risk after pre-eclampsia in women with dysglycaemia. Diabet. Med. 2012, 30, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Prescott, G.J.; Iversen, L.; Campbell, D.M.; Smith, W.C.; Hannaford, P.C. Hypertensive disorders of pregnancy and future health and mortality: A record linkage study. Pregnancy Hypertens. 2011, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Shahbazian, N.; Shahbazian, H.; Ehsanpour, A.; Aref, A.; Gharibzadeh, S. Hypertension and microalbuminuria 5 years after pregnancies complicated by pre-eclampsia. Iran. J. Kidney Dis. 2011, 5, 324–327. [Google Scholar] [PubMed]
- Vikse, B.E.; Hallan, S.; Bostad, L.; Leivestad, T.; Iversen, B.M. Previous preeclampsia and risk for progression of biopsy-verified kidney disease to end-stage renal disease. Nephrol. Dial. Transplant. 2010, 25, 3289–3296. [Google Scholar] [CrossRef] [PubMed]
- Sandvik, M.K.; Iversen, B.M.; Irgens, L.M.; Skjaerven, R.; Leivestad, T.; Søfteland, E.; Vikse, B.E. Are adverse pregnancy outcomes risk factors for development of end-stage renal disease in women with diabetes? Nephrol. Dial. Transplant. 2010, 25, 3600–3607. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Irgens, L.M.; Leivestad, T.; Skjaerven, R.; Iversen, B.M. Preeclampsia and the risk of end-stage renal disease. N. Engl. J. Med. 2008, 359, 800–809. [Google Scholar] [CrossRef] [PubMed]
- Lampinen, K.H.; Rönnback, M.; Groop, P.H.; Kaaja, R.J. Renal and vascular function in women with previous preeclampsia: A comparison of low- and high-degree proteinuria. Kidney Int. 2006, 70, 1818–1822. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Irgens, L.M.; Bostad, L.; Iversen, B.M. Adverse perinatal outcome and later kidney biopsy in the mother. J. Am. Soc. Nephrol. 2006, 17, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Shammas, A.G.; Maayah, J.F. Hypertension and its relation to renal function 10 years after pregnancy complicated by pre-eclampsia and pregnancy induced hypertension. Saudi Med. J. 2000, 21, 190–192. [Google Scholar] [PubMed]
- Kattah, A.G.; Scantlebury, D.C.; Agarwal, S.; Mielke, M.M.; Rocca, W.A.; Weaver, A.L.; Vaughan, L.E.; Miller, V.M.; Weissgerber, T.L.; White, W.; et al. Preeclampsia and ESRD: The Role of Shared Risk Factors. Am. J. Kidney Dis. 2017, 69, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Wide-Swensson, D.; Strevens, H.; Willner, J. Antepartum percutaneous renal biopsy. Int. J. Gynaecol. Obstet. 2007, 98, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Shiiki, H.; Dohi, K.; Hanatani, M.; Fujii, Y.; Sanai, H.; Ichijo, M.; Shimamoto, I.; Ishikawa, H.; Watanabe, T. Focal and segmental glomerulosclerosis in preeclamptic patients with nephrotic syndrome. Am. J. Nephrol. 1990, 10, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Cabiddu, G.; Castellino, S.; Gernone, G.; Santoro, D.; Moroni, G.; Spotti, D.; Giacchino, F.; Attini, R.; Limardo, M.; et al. A best practice position statement on the role of the nephrologist in the prevention and follow-up of preeclampsia: The Italian study group on kidney and pregnancy. J. Nephrol. 2017, 30, 307–317. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, N.; Wright, D.; Poon, L.C.; Rolnik, D.L.; Syngelaki, A.; de Alvarado, M.; Carbone, I.F.; Dutemeyer, V.; Fiolna, M.; Frick, A. Multicenter screening for pre-eclampsia by maternal factors and biomarkers at 11–13 weeks’ gestation: Comparison with NICE guidelines and ACOG recommendations. Ultrasound Obstet. Gynecol. 2017, 49, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Von Dadelszen, P.; Payne, B.; Li, J.; Ansermino, J.M.; Broughton Pipkin, F.; Côté, A.M.; Douglas, M.J.; Gruslin, A.; Hutcheon, J.A.; Joseph, K.S.; et al. Prediction of adverse maternal outcomes in pre-eclampsia: Development and validation of the fullPIERS model. Lancet 2011, 377, 219–227. [Google Scholar] [CrossRef]
- Linsell, L.; Malouf, R.; Morris, J.; Kurinczuk, J.J.; Marlow, N. Risk Factor Models for Neurodevelopmental Outcomes in Children Born Very Preterm or With Very Low Birth Weight: A Systematic Review of Methodology and Reporting. Am. J. Epidemiol. 2017, 185, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Guellec, I.; Lapillonne, A.; Marret, S.; Picaud, J.C.; Mitanchez, D.; Charkaluk, M.L.; Fresson, J.; Arnaud, C.; Flamand, C.; Cambonie, G. Effect of Intra- and Extrauterine Growth on Long-Term Neurologic Outcomes of Very Preterm Infants. J. Pediatr. 2016, 175, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.; Hennessy, E.M.; Myles, J.; Johnson, S.J.; Draper, E.S.; Costeloe, K.L.; Marlow, N. Neurological and developmental outcome in extremely preterm children born in England in 1995 and 2006: The EPICure studies. BMJ 2012. [Google Scholar] [CrossRef] [PubMed]
- Guillén, U.; DeMauro, S.; Ma, L.; Zupancic, J.; Roberts, R.; Schmidt, B.; Kirpalani, H. Relationship between attrition and neurodevelopmental impairment rates in extremely preterm infants at 18 to 24 months: A systematic review. Arch. Pediatr. Adolesc. Med. 2012, 166, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Ranke, M.B.; Schweizer, R.; Rodemann, S.M.; Bevot, A.; Martin, D.D.; Goelz, R.; Wollmann, H.A.; Krägeloh-Mann, I.; Binder, G. Schoolchildren born VLBW or VLGA show height-related changes in body composition and muscle function but no evidence of metabolic syndrome risk factors. Results from the NEOLONG study. J. Pediatr. Endocrinol. Metab. 2016, 29, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Castanys-Muñoz, E.; Kennedy, K.; Castañeda-Gutiérrez, E.; Forsyth, S.; Godfrey, K.M.; Koletzko, B.; Ozanne, S.E.; Rueda, R.; Schoemaker, M.; van der Beek, E.M. Systematic review indicates postnatal growth in term infants born small-for-gestational-age being associated with later neurocognitive and metabolic outcomes. Acta Paediatr. 2017, 106, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Ong, K.K.; Kennedy, K.; Castañeda-Gutiérrez, E.; Forsyth, S.; Godfrey, K.M.; Koletzko, B.; Latulippe, M.E.; Ozanne, S.E.; Rueda, R.; Schoemaker, M.H. Postnatal growth in preterm infants and later health outcomes: A systematic review. Acta Paediatr. 2015, 104, 974–986. [Google Scholar] [CrossRef] [PubMed]
- Low Birth Weight and Nephron Number Working Group. The Impact of Kidney Development on the Life Course: A Consensus Document for Action. Nephron Clin. Pract. 2017, 136, 3–49. [Google Scholar]
- Luyckx, V.A.; Bertram, J.F.; Brenner, B.M.; Fall, C.; Hoy, W.E.; Ozanne, S.E.; Vikse, B.E. Effect of fetal and child health on kidney development and long-term risk of hypertension and kidney disease. Lancet 2013, 382, 273–283. [Google Scholar] [CrossRef]
- Luyckx, V.A.; Brenner, B.M. Birth weight, malnutrition and kidney-associated outcomes—A global concern. Nat. Rev. Nephrol. 2015, 11, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Monier, I.; Ancel, P.Y.; Ego, A.; Jarreau, P.H.; Lebeaux, C.; Kaminski, M.; Goffinet, F.; Zeitlin, J. Fetal and neonatal outcomes of preterm infants born before 32 weeks of gestation according to antenatal vs postnatal assessments of restricted growth. Am. J. Obstet. Gynecol. 2017, 216, 516. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.C.; Kozuki, N.; Cousens, S.; Stevens, G.A.; Blencowe, H.; Silveira, M.F.; CHERG Small-for-Gestational-Age-Preterm Birth Working Group. Estimates of burden and consequences of infants born small for gestational age in low and middle income countries with INTERGROWTH-21 standard: Analysis of CHERG datasets. BMJ 2017, 358, 3677. [Google Scholar] [CrossRef] [PubMed]
- Temming, L.A.; Dicke, J.M.; Stout, M.J.; Rampersad, R.M.; Macones, G.A.; Tuuli, M.G.; Cahill, A.G. Early Second-Trimester Fetal Growth Restriction and Adverse Perinatal Outcomes. Obstet. Gynecol. 2017, 130, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Y.; Chughtai, A.A.; Lui, K.; Sullivan, E.A. Morbidity and mortality among very preterm singletons following fertility treatment in Australia and New Zealand, a population cohort study. BMC Pregnancy Childbirth 2017, 17, 50. [Google Scholar] [CrossRef] [PubMed]
- Darmstadt, G.L.; Walker, N.; Lawn, J.E.; Bhutta, Z.A.; Haws, R.A.; Cousens, S. Saving newborn lives in Asia and Africa: Cost and impact of phased scale-up of interventions within the continuum of care. Health Policy Plan 2008, 23, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Das, J.K.; Bahl, R.; Lawn, J.E.; Salam, R.A.; Paul, V.K.; Sankar, M.J.; Blencowe, H.; Rizvi, A.; Chou, V.B.; et al. Can available interventions end preventable deaths in mothers, newborn babies, and stillbirths, and at what cost? Lancet 2014, 384, 347–370. [Google Scholar] [CrossRef]
- Luyckx, V.A. Preterm Birth and its Impact on Renal Health. Semin. Nephrol. 2017, 37, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Abitbol, C.L.; Rodriguez, M.M. The long-term renal and cardiovascular consequences of prematurity. Nat. Rev. Nephrol. 2012, 8, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, M.; Ryan, D.; Black, M.J.; Kent, A.L. Long-term renal consequences of preterm birth. Clin. Perinatol. 2014, 41, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Carmody, J.B.; Charlton, J.R. Short-term gestation, long-term risk: Prematurity and chronic kidney disease. Pediatrics 2013, 131, 1168–1179. [Google Scholar] [CrossRef] [PubMed]
- Almasi, O.; Pariente, G.; Kessous, R.; Sergienko, R.; Sheiner, E. Association between delivery of small-for-gestational-age neonate and long-term maternal chronic kidney disease. J. Matern. Fetal Neonatal Med. 2016, 29, 2861–2864. [Google Scholar] [CrossRef] [PubMed]
- Pariente, G.; Sheiner, E.; Kessous, R.; Michael, S.; Shoham-Vardi, I. Association between delivery of a small-for-gestational-age neonate and long-term maternal cardiovascular morbidity. Int. J. Gynaecol. Obstet. 2013, 123, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Neiger, R. Long-Term Effects of Pregnancy Complications on Maternal Health: A Review. J. Clin. Med. 2017, 6, 76. [Google Scholar] [CrossRef] [PubMed]
- Cabiddu, G.; Castellino, S.; Gernone, G.; Santoro, D.; Moroni, G.; Giannattasio, M.; Gregorini, G.; Giacchino, F.; Attini, R.; Loi, V.; et al. A best practice position statement on pregnancy in chronic kidney disease: The Italian Study Group on Kidney and Pregnancy. J. Nephrol. 2016, 29, 277–303. [Google Scholar] [CrossRef] [PubMed]
- Blom, K.; Odutayo, A.; Bramham, K.; Hladunewich, M.A. Pregnancy and Glomerular Disease: A Systematic Review of the Literature with Management Guidelines. Clin. J. Am. Soc. Nephrol. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Cabiddu, G.; Attini, R.; Vigotti, F.N.; Maxia, S.; Lepori, N.; Tuveri, M.; Massidda, M.; Marchi, C.; Mura, S. Risk of Adverse Pregnancy Outcomes in Women with CKD. J. Am. Soc. Nephrol. 2015, 26, 2011–2022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Ma, X.X.; Hao, L.; Liu, L.J.; Lv, J.C.; Zhang, H. A Systematic Review and Meta-Analysis of Outcomes of Pregnancy in CKD and CKD Outcomes in Pregnancy. Clin. J. Am. Soc. Nephrol. 2015, 10, 1964–1978. [Google Scholar] [CrossRef] [PubMed]
- Alsuwaida, A.; Mousa, D.; Al-Harbi, A.; Alghonaim, M.; Ghareeb, S.; Alrukhaimi, M.N. Impact of early chronic kidney disease on maternal and fetal outcomes of pregnancy. J. Matern. Fetal. Neonatal. Med. 2011, 24, 1432–1436. [Google Scholar] [CrossRef] [PubMed]
- De Jesus, G.R.; de Jesus, N.R.; Levy, R.A.; Klumb, E.M. The use of angiogenic and antiangiogenic factors in the differential diagnosis of pre-eclampsia, antiphospholipid syndrome nephropathy and lupus nephritis. Lupus 2014, 23, 1299–1301. [Google Scholar] [CrossRef] [PubMed]
- Morton, A. Imitators of preeclampsia: A review. Pregnancy Hypertens. 2016, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rolfo, A.; Attini, R.; Tavassoli, E.; Neve, F.V.; Nigra, M.; Cicilano, M.; Nuzzo, A.M.; Giuffrida, D.; Biolcati, M.; Nichelatti, M.; et al. Is It Possible to Differentiate Chronic Kidney Disease and Preeclampsia by means of New and Old Biomarkers? A Prospective Study. Dis. Mark. 2015. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Gaglioti, P.; Attini, R.; Parisi, S.; Bossotti, C.; Olearo, E.; Oberto, M.; Ferraresi, M.; Rolfo, A.; Versino, E.; et al. Pre-eclampsia or chronic kidney disease? The flow hypothesis. Nephrol. Dial. Transplant. 2013, 28, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Zeisler, H.; Llurba, E.; Chantraine, F.; Vatish, M.; Staff, A.C.; Sennström, M.; Olovsson, M.; Brennecke, S.P.; Stepan, H.; Allegranza, D.; et al. Predictive Value of the sFlt-1: PlGF Ratio in Women with Suspected Preeclampsia. N. Engl. J. Med. 2016, 374, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Masuyama, H.; Nobumoto, E.; Okimoto, N.; Inoue, S.; Segawa, T.; Hiramatsu, Y. Superimposed preeclampsia in women with chronic kidney disease. Gynecol. Obstet. Invest. 2012, 74, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Zhu, Z.; Liu, C.; Zhang, Z. Dual-cutoff of sFlt-1/PlGF ratio in the stratification of preeclampsia: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2017, 295, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, E.; Medcalf, K.E.; Park, A.L.; Ray, J.G. Clinical risk factors for pre-eclampsia determined in early pregnancy: Systematic review and meta-analysis of large cohort studies. BMJ 2016, 353, 1753. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Ma, J.; Zhang, W.H.; Di, W. Management and outcomes of pregnancy with or without lupus nephritis: A systematic review and meta-analysis. Ther. Clin. Risk. Manag. 2018, 14, 885–901. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Lai, K.; Yang, Z.; Zeng, K. Systemic lupus erythematosus and risk of preterm birth: A systematic review and meta-analysis of observational studies. Lupus 2017, 26, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Smyth, A.; Oliveira, G.H.; Lahr, B.D.; Bailey, K.R.; Norby, S.M.; Garovic, V.D. A systematic review and meta-analysis of pregnancy outcomes in patients with systemic lupus erythematosus and lupus nephritis. Clin. J. Am. Soc. Nephrol. 2010, 5, 2060–2068. [Google Scholar] [CrossRef] [PubMed]
- Carvalheiras, G.; Vita, P.; Marta, S.; Trovão, R.; Farinha, F.; Braga, J.; Rocha, G.; Almeida, I.; Marinho, A.; Mendonça, T.; et al. Pregnancy and systemic lupus erythematosus: Review of clinical features and outcome of 51 pregnancies at a single institution. Clin. Rev. Allergy Immunol. 2010, 38, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Vellanki, V.S.; Parvathina, S.; Gondi, S.; Yadla, M.; Chenu, K.K.; Vishnubhotla, S. Post-partum bilateral renal cortical necrosis in antiphospholipid syndrome and systemic lupus erythematosus. Saudi J. Kidney Dis. Transpl. 2013, 24, 549–552. [Google Scholar] [PubMed]
- Mizuno, R.; Fujimoto, S.; Fujimoto, T.; Nishino, T.; Shiiki, H.; Hashimoto, T.; Nakamura, S.; Dohi, K. Catastrophic antiphospholipid antibody syndrome in systemic lupus erythematosus: An autopsy case report of a young woman. Intern. Med. 2000, 39, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Moroni, G.; Doria, A.; Giglio, E.; Imbasciati, E.; Tani, C.; Zen, M.; Strigini, F.; Zaina, B.; Tincani, A.; Gatto, M. Maternal outcome in pregnant women with lupus nephritis. A prospective multicenter study. J. Autoimmun. 2016, 74, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Buyon, J.P.; Kim, M.Y.; Guerra, M.M.; Laskin, C.A.; Petri, M.; Lockshin, M.D.; Sammaritano, L.; Branch, D.W.; Porter, T.F.; Sawitzke, A. Predictors of Pregnancy Outcomes in Patients with Lupus: A Cohort Study. Ann. Intern. Med. 2015, 163, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Alchi, B.; Griffiths, M.; Jayne, D. What nephrologists need to know about antiphospholipid syndrome. Nephrol. Dial. Transplant. 2010, 25, 3147–3154. [Google Scholar] [CrossRef] [PubMed]
- Hanouna, G.; Morel, N.; Le Thi Huong, D.; Josselin, L.; Vauthier-Brouzes, D.; Saadoun, D.; Kettaneh, A.; Levesque, K.; Le Guern, V.; Goffinet, F.; et al. Catastrophic antiphospholipid syndrome and pregnancy: An experience of 13 cases. Rheumatology (Oxford) 2013, 52, 1635–1641. [Google Scholar] [CrossRef] [PubMed]
- Dashe, J.S.; Ramin, S.M.; Cunningham, F.G. The long-term consequences of thrombotic microangiopathy (thrombotic thrombocytopenic purpura and hemolytic uremic syndrome) in pregnancy. Obstet. Gynecol. 1998, 91, 662–668. [Google Scholar] [PubMed]
- Silver, R.M. Catastrophic antiphospholipid syndrome and pregnancy. Semin. Perinatol. 2018, 42, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Jim, B. Acute Kidney Injury in Pregnancy: The Changing Landscape for the 21st Century. Kidney Int. Rep. 2018, 3, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Cervera, R.; Serrano, R.; Pons-Estel, G.J.; Ceberio-Hualde, L.; Shoenfeld, Y.; de Ramón, E. Morbidity and mortality in the antiphospholipid syndrome during a 10-year period: A multicentre prospective study of 1000 patients. Ann. Rheum. Dis. 2015, 74, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Pengo, V.; Banzato, A.; Bison, E.; Bracco, A.; Denas, G.; Ruffatti, A. What have we learned about antiphospholipid syndrome from patients and antiphospholipid carrier cohorts? Semin. Thromb. Hemost. 2012, 38, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Ducloy-Bouthors, A.S.; Caron, C.; Subtil, D. Thrombotic thrombocytopenic purpura: Medical and biological monitoring of six pregnancies. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 111, 146–152. [Google Scholar] [CrossRef]
- Sanchez-Luceros, A.; Farias, C.E.; Amaral, M.M. Von Willebrand factor cleaving protease (ADAMTS13) activity in normal non-pregnant women, pregnant and post-delivery women. Thromb. Haemost. 2004, 92, 1320–1326. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, F.; Roumenina, L.; Provot, F.; Sallée, M.; Caillard, S.; Couzi, L. Pregnancy-associated hemolytic uremic syndrome revisited in the era of complement gene mutations. J. Am. Soc. Nephrol. 2010, 21, 859–867. [Google Scholar] [CrossRef] [PubMed]
- Fakhouri, F.; Jablonski, M.; Lepercq, J. Factor H, membrane cofactor protein, and factor I mutations in patients with hemolysis, elevated liver enzymes, and low platelet count syndrome. Blood 2008, 112, 4542–4545. [Google Scholar] [CrossRef] [PubMed]
- Baghli, S.; Abendroth, C.; Farooq, U.; Schaub, J.A. Atypical Presentation of Pregnancy-Related Hemolytic Uremic Syndrome. Am. J. Kidney Dis. 2018, 72, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Barbullushi, M.; Idrizi, A.; Spasovski, G. Familial hemolytic uremic syndrome with occurrence in the postpartum period. Saudi J. Kidney Dis. Transpl. 2017, 28, 1427–1431. [Google Scholar] [CrossRef] [PubMed]
- Gately, R.; San, A.; Kurtkoti, J.; Parnham, A. Life-threatening pregnancy-associated atypical haemolytic uraemic syndrome and its response to eculizumab. Nephrology (Carlton) 2017, 22, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Servais, A.; Devillard, N.; Frémeaux-Bacchi, V.; Hummel, A.; Salomon, L.; Contin-Bordes, C.; Gomer, H.; Legendre, C.; Delmas, Y. Atypical haemolytic uraemic syndrome and pregnancy: Outcome with ongoing eculizumab. Nephrol. Dial. Transplant. 2016, 31, 2122–2130. [Google Scholar] [CrossRef] [PubMed]
- Noris, M.; Bresin, E.; Mele, C.; Remuzzi, G. Genetic Atypical Hemolytic-Uremic Syndrome. In GeneReviews; Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 2007. [Google Scholar]
- Shanmugalingam, R.; Hsu, D.; Makris, A. Pregnancy-induced atypical haemolytic uremic syndrome: A new era with eculizumab. Obstet. Med. 2018, 11, 28–31. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).