(4Z)-Lachnophyllum Lactone, an Acetylenic Furanone from Conyza bonariensis, Identified for the First Time with Allelopathic Activity against Cuscuta campestris
Abstract
:1. Introduction
2. Materials and Methods
2.1. General Experimental Procedures
2.2. Plant Material
2.3. Plant Extraction for Screening of Allelopathy in Weed Species
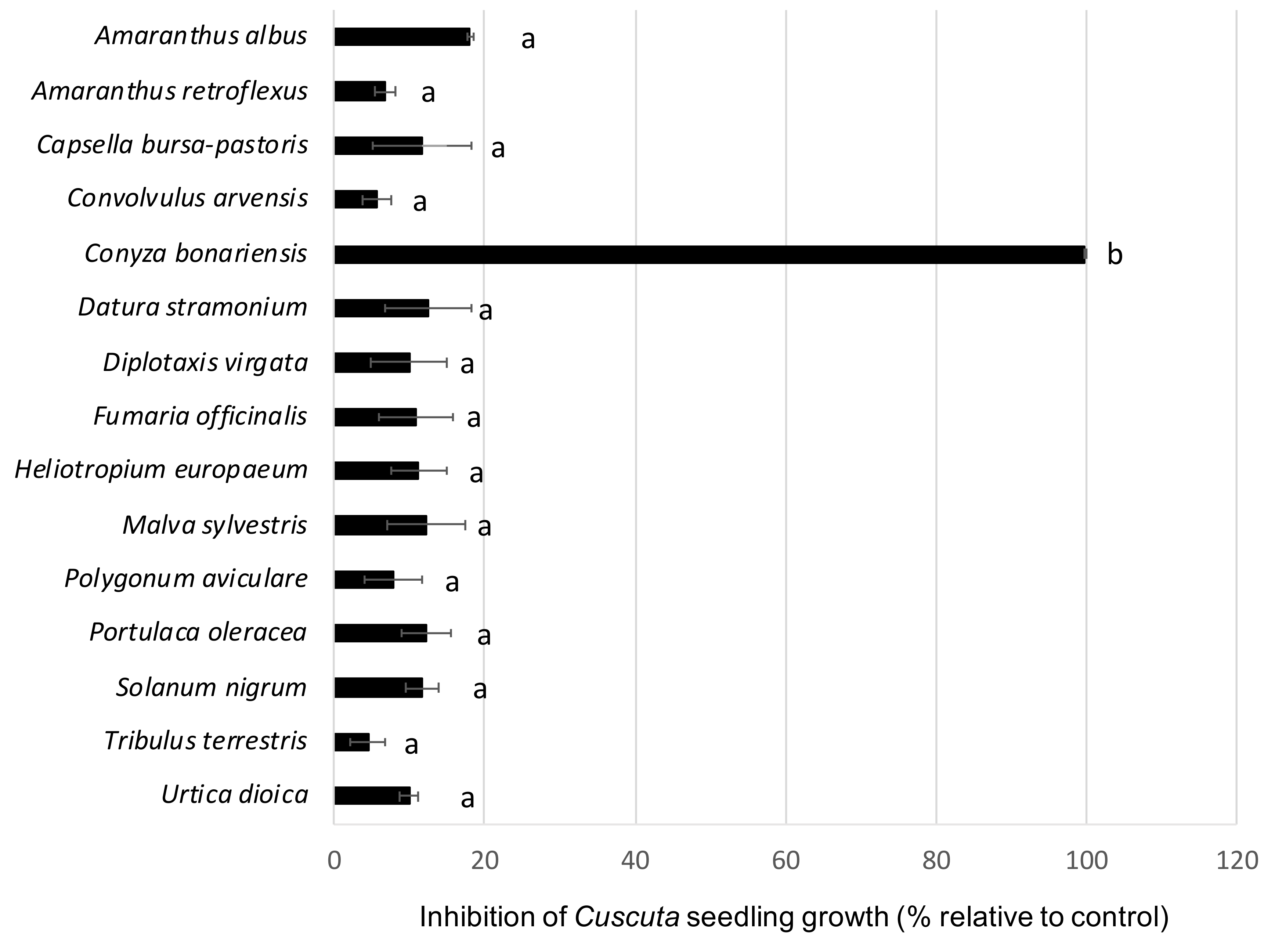
2.4. In Vitro Experiments for Screening of Allelopathy against Cuscuta Seedling Growth
2.5. Pot Experiments for Validation of Conyza Allelopathic Activity
2.6. Isolation and Identification of (4Z)-Lachnophyllum Lactone from Conyza Bonariensis Extracts
2.7. Bioassays against Cuscuta Seedling Growth for Identification of (4Z)-Lachnophyllum Lactone Phytotoxic Activity
2.8. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nickrent, D.L.; Malécot, V.; Vidal-Russell, R.; Der, J.P. A revised classification of Santalales. Taxon 2010, 59, 538–558. [Google Scholar] [CrossRef]
- Westwood, J.H.; Yoder, J.I.; Timko, M.P.; de Pamphilis, C.W. The evolution of parasitism in plants. Trends Plant Sci. 2010, 15, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Dawson, J.H.; Musselman, L.J.; Wolswinkel, P.; Dörr, I. Biology and control of Cuscuta. Rev. Weed Sci. 1994, 6, 265–317. [Google Scholar]
- Nickrent, D.L. Parasitic angiosperms: How often and how many? Taxon 2020, 69, 5–27. [Google Scholar] [CrossRef]
- Christensen, N.M.; Dörr, I.; Hansen, M.; van der Kooij, T.A.W.; Schulz, A. Development of Cuscuta species on a partially incompatible host: Induction of xylem transfer cells. Protoplasma 2003, 220, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Dörr, I. The haustorium of Cuscuta—New structural results. In Proceedings of the Fourth International Symposium on Parasitic Flowering Plants, Marburg, Germany, 2–7 August 1987; Weber, H.C., Forstreuter, W., Eds.; Phillips-Universitat: Marburg, Germany, 1987; pp. 163–170. [Google Scholar]
- Goldwasser, Y.; Miryamchik, H.; Sibony, M.; Rubin, B. Detection of resistant chickpea (Cicer arietinum) genotypes to Cuscuta campestris (field dodder). Weed Res. 2012, 52, 122–130. [Google Scholar] [CrossRef]
- Córdoba, E.M.; Fernández-Aparicio, M.; González-Verdejo, C.I.; López-Grau, C.; Muñoz-Muñoz, M.V.; Nadal, S. Search for Resistant Genotypes to Cuscuta campestris Infection in two legume species, Vicia sativa and Vicia ervilia. Plants 2021, 10, 738. [Google Scholar] [CrossRef]
- Nadler-Hassar, T.; Shaner, D.L.; Nissen, S.; Westra, P.; Rubin, B. Are herbicide-resistant crops the answer to controlling Cuscuta? Pest Manag. Sci. 2009, 65, 811–816. [Google Scholar] [CrossRef]
- Cimmino, A.; Fernández-Aparicio, M.; Andolfi, A.; Basso, S.; Rubiales, D.; Evidente, A. Effect of fungal and plant metabolites on broomrapes (Orobanche and Phelipanche spp.) seed germination and radicle growth. J. Agric. Food Chem. 2014, 62, 10485–10492. [Google Scholar] [CrossRef]
- Fernández-Aparicio, M.; Cimmino, A.; Evidente, A.; Rubiales, D. Inhibition of Orobanche crenata seed germination and radicle growth by allelochemicals identified in cereals. J. Agric. Food Chem. 2013, 61, 9797–9803. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Aparicio, M.; Delavault, P.; Timko, M.P. Management of infection by parasitic weeds: A Review. Plants 2020, 9, 1184. [Google Scholar] [CrossRef] [PubMed]
- Dayan, F.E.; Duke, S.O. Natural compounds as next-generation herbicides. Plant Physiol. 2014, 166, 1090–1105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macías, F.A.; Molinillo, J.M.; Varela, R.M.; Galindo, J.C. Allelopathy-a natural alternative for weed control. Pest Manag. Sci. 2007, 63, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Westwood, J.H.; Charudattan, R.; Duke, S.O.; Fennimore, S.A.; Marrone, P.; Slaughter, D.C.; Swanton, C.; Zollinger, R. Weed Management in 2050: Perspectives on the Future of Weed Science. Weed Sci. 2018, 66, 275–285. [Google Scholar] [CrossRef] [Green Version]
- Cimmino, A.; Masi, M.; Rubiales, D.; Evidente, A.; Fernández-Aparicio, M. Allelopathy for parasitic plant management. Nat. Prod. Commun. 2018, 13, 1934578X1801300307. [Google Scholar] [CrossRef] [Green Version]
- Holm, L.; Doll, J.; Holm, E.; Pancho, J.; Herberger, J. World Weeds: Natural Histories and Distribution; John Wiley and Sons: New York, NY, USA, 1997; pp. 226–235. [Google Scholar]
- González-Torralva, F.; Cruz-Hipolito, H.; Bastida, F.; Mülleder, N.; Smeda, R.J.; De Prado, R. Differential susceptibility to glyphosate among the Conyza weed species in Spain. J. Agric. Food Chem. 2010, 58, 4361–4366. [Google Scholar] [CrossRef]
- Urbano, J.M.; Borrego, A.; Torres, V.; Leon, J.M.; Jimenez, C.; Dinelli, G.; Barnes, J. Glyphosate-resistant hairy fleabane (Conyza bonariensis) in Spain. Weed Technol. 2007, 21, 396–401. [Google Scholar] [CrossRef]
- Heap, I. International Survey of Herbicide Resistant Weeds. Available online: www.weedscience.com. (accessed on 10 February 2022).
- Djurdjević, L.; Mitrović, M.; Gajić, G.; Jarić, S.; Kostić, O.; Oberan, L.; Pavlović, P. An allelopathic investigation of the domination of the introduced invasive Conyza canadensis L. Flora 2011, 206, 921–927. [Google Scholar] [CrossRef]
- Gao, X.; Li, M.; Gao, Z.; Zhang, H.; Sun, Z. Allelopathic effects of Conyza canadesis the germination and growth of wheat, sorghum, cucumber, rape and radish. Alellopathy J. 2009, 23, 287–296. [Google Scholar]
- Hu, G.; Zhang, Z.H. Aqueous tissue extracts of Conyza canadensis inhibit the germination and shoot growth of three native herbs with no autotoxic effects. Planta Daninha 2013, 31, 805–811. [Google Scholar] [CrossRef] [Green Version]
- Shaukat, S.S.; Munir, N.; Siddiqui, I.A. Allelopathic responses of Conyza canadensis (L.) Cronquist: A cosmopolitan weed. Asian J. Plant Sci. 2003, 14, 1034–1039. [Google Scholar]
- Gaertner, E.E. Studies of seed germination, seed identification, and host relationship in Dodders, Cuscuta spp. Mem. Cornell Agric. Exp. Stn. 1950, 294, 1–56. [Google Scholar]
- Queiroz, S.C.N.; Cantrell, C.L.; Duke, S.O.; Wedge, D.E.; Nandula, V.K.; Moraes, R.M.; Cerdeira, A.L. Bioassay-directed isolation and identification of phytotoxic and fungitoxic acetylenes from Conyza canadensis. J. Agric. Food Chem. 2012, 60, 5893–5898. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Aparicio, M.; Sillero, J.C.; Rubiales, D. Resistance to broomrape species (Orobanche spp.) in common vetch (Vicia sativa L.). Crop Prot. 2009, 28, 7–12. [Google Scholar] [CrossRef]
- Heide-Jørgensen, H.S. Parasitic Flowering Plants; Brill Leiden: Leiden, The Netherlands, 2008. [Google Scholar]
- Holmes, M.G.; Smith, H. The function of phytochrome in plants growing in the natural environment. Nature 1975, 254, 512. [Google Scholar] [CrossRef]
- Orr, G.L.; Haidar, M.A.; Orr, D.A. Small seed dodder (Cuscuta planiflora) phototropism toward far-red when in white light. Weed Sci. 1996, 44, 233–240. [Google Scholar] [CrossRef]
- Runyon, J.B.; Mescher, M.C.; De Moraes, C.M. Volatile chemical cues guide host location and host selection by parasitic plants. Science 2006, 313, 1964–1967. [Google Scholar] [CrossRef] [Green Version]
- Dörr, I. Feinstruktur intrazellular wachsender Cuscuta-Hyphen. Protoplasma 1969, 67, 123–137. [Google Scholar] [CrossRef]
- Haidar, M.A.; Orr, G.L.; Westra, P. Effects of light and mechanical stimulation on coiling and pre-haustoria formation in Cuscuta spp. Weed Res. 1997, 37, 219–228. [Google Scholar] [CrossRef]
- Hegenauer, V.; Körner, M.; Albert, M. Plants under stress by parasitic plants. Curr. Opin. Plant Biol. 2017, 38, 34–41. [Google Scholar] [CrossRef]
- Vaughn, K.C. Attachment of the parasitic weed dodder to the host. Protoplasma 2002, 219, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Orkic, I.; Stefanic, E.; Antunovic, S.; Zima, D.; Kovacevic, V.; Stefanic, I.; Dimic, D. Host range of field dodder (Cuscuta campestris Yuncker) in sugar beet fields: Example from Northeastern Croatia. Listy Cukrov. Reparske 2019, 135, 198–203. [Google Scholar]
- Nawamaki, T.; Sakakibara, T.; Ohta, K. Isolation and identification of lachnophyllum lactone and osthol as repellents against a sea snail. Agric. Biol. Chem. 1979, 43, 1603–1604. [Google Scholar]
- Rivera, A.P.; Arancibia, L.; Castillo, M. Clerodane diterpenoids and acetylenic lactones from Baccharris paniculata. J. Nat. Prod. 1989, 52, 433–435. [Google Scholar] [CrossRef]
- Cheng, W.; Zhu, C.; Xu, W.; Fan, X.; Yang, Y.; Li, Y.; Chen, X.; Wang, W.; Shi, J. Chemical constituents of the bark of Machilus wangchiana and their biological activities. J. Nat. Prod. 2009, 72, 2145–2152. [Google Scholar] [CrossRef]
- Masi, M.; Di Lecce, R.; Marsico, G.; Linaldeddu, B.T.; Maddau, L.; Superchi, S.; Evidente, A. Pinofuranoxins A and B, bioactive trisubstituted furanones produced by the invasive pathogen Diplodia sapinea. J. Nat. Prod. 2021, 84, 2600–2605. [Google Scholar] [CrossRef]
- Takeda, K.I.; Sakurawi, K.; Ishii, H. Components of the Lauraceae family—I: New lactonic compounds from Litsea japoncia. Tetrahedron 1972, 28, 3757–3766. [Google Scholar] [CrossRef]
- Bohlmann, F.; Knauf, W.; King, R.M.; Robinson, H. Ein neues diterpen und weitere inhaltsstoffe aus Baccharisarten. Phytochemistry 1979, 18, 1011–1014. [Google Scholar] [CrossRef]
- Terao, D.; Queiroz, S.C.N.; Maia, A.D.H.N. Bioactive compound isolated from Conyza canadensis combined with physical treatments for the control of green mould in Orange. J. Phytopathol. 2022, 170, 158–166. [Google Scholar] [CrossRef]
- Vidari, G.; Abdo, S.; Gilardoni, G.; Ciapessoni, A.; Gusmeroli, M.; Zanoni, G. Fungitoxic metabolites from Erigeron apiculatus. Fitoterapia 2006, 77, 318–320. [Google Scholar] [CrossRef]
- Cimmino, A.; Masi, M.; Evidente, M.; Superchi, S.; Evidente, A. Fungal phytotoxins with potential herbicidal activity: Chemical and biological characterization. Nat. Prod. Rep. 2015, 32, 1629–1653. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Aparicio, M.; Cimmino, A.; Soriano, G.; Masi, M.; Vilariño, S.; Evidente, A. Assessment of weed root extracts for allelopathic activity against Orobanche and Phelipanche species. Phytopathol. Mediterr. 2021, 60, 455–466. [Google Scholar] [CrossRef]
- Soriano, G.; Fernández-Aparicio, M.; Masi, M.; Vilariño-Rodríguez, S.; Cimmino, A. Complex mixture of arvensic acids isolated from Convolvulus arvensis roots identified as inhibitors of radicle growth of broomrape weeds. Agriculture 2022, 12, 585. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Aparicio, M.; Soriano, G.; Masi, M.; Carretero, P.; Vilariño-Rodríguez, S.; Cimmino, A. (4Z)-Lachnophyllum Lactone, an Acetylenic Furanone from Conyza bonariensis, Identified for the First Time with Allelopathic Activity against Cuscuta campestris. Agriculture 2022, 12, 790. https://doi.org/10.3390/agriculture12060790
Fernández-Aparicio M, Soriano G, Masi M, Carretero P, Vilariño-Rodríguez S, Cimmino A. (4Z)-Lachnophyllum Lactone, an Acetylenic Furanone from Conyza bonariensis, Identified for the First Time with Allelopathic Activity against Cuscuta campestris. Agriculture. 2022; 12(6):790. https://doi.org/10.3390/agriculture12060790
Chicago/Turabian StyleFernández-Aparicio, Mónica, Gabriele Soriano, Marco Masi, Pilar Carretero, Susana Vilariño-Rodríguez, and Alessio Cimmino. 2022. "(4Z)-Lachnophyllum Lactone, an Acetylenic Furanone from Conyza bonariensis, Identified for the First Time with Allelopathic Activity against Cuscuta campestris" Agriculture 12, no. 6: 790. https://doi.org/10.3390/agriculture12060790








