Bacillus aryabhattai Mitigates the Effects of Salt and Water Stress on the Agronomic Performance of Maize under an Agroecological System
Abstract
1. Introduction
2. Material and Methods
2.1. Location and Characterisation of the Experimental Area
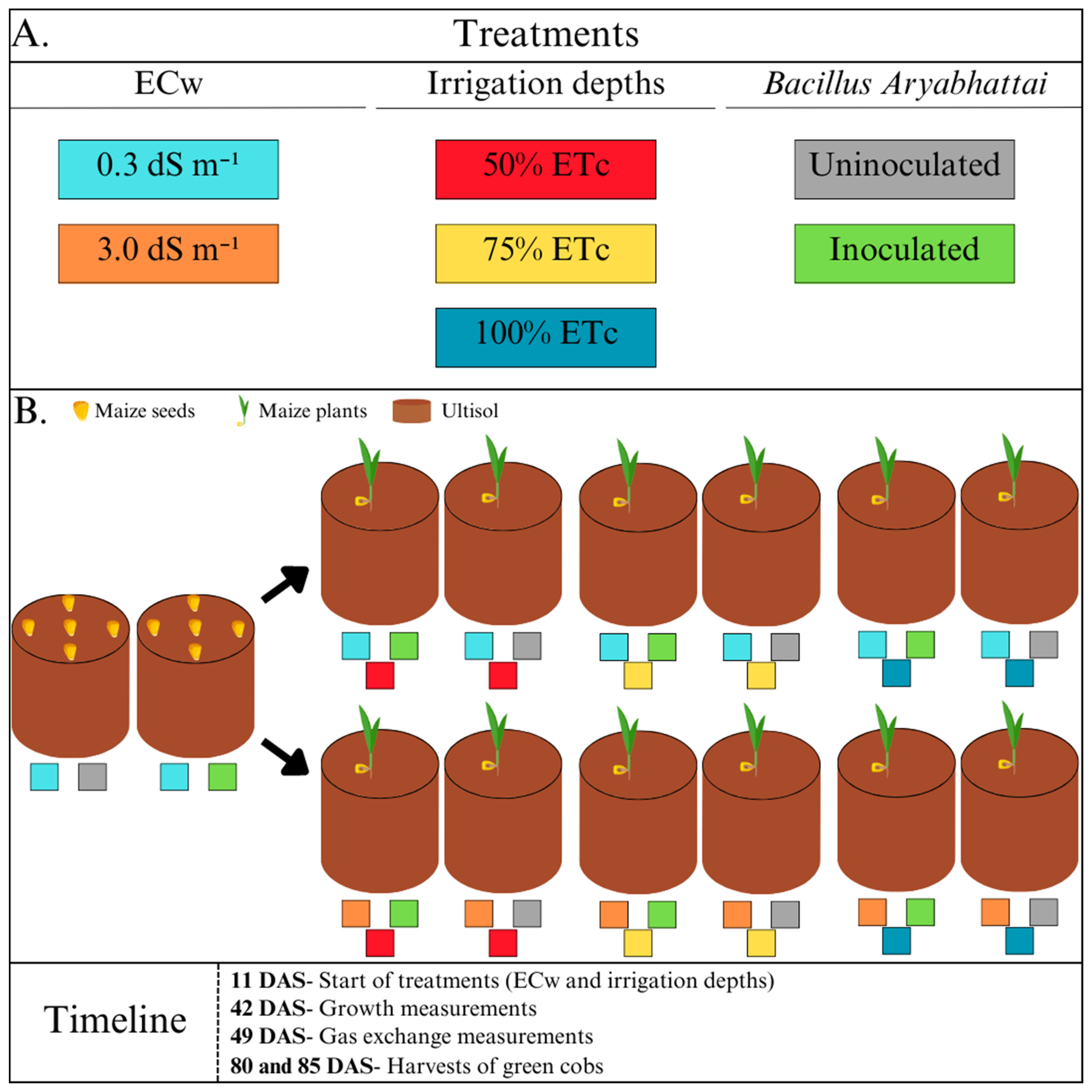
2.2. Experimental Design and Treatments
2.3. Irrigation Management
2.4. Agroecological Maize Production System (Plant Material, Inoculation, and Fertilisation)
2.5. Variables under Analysis
2.5.1. Growth
2.5.2. Leaf Gas Exchange
2.5.3. Yield
2.6. Data Analysis
3. Results and Discussion
3.1. Growth
3.2. Leaf Gas Exchange
3.3. Yield
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fornasieri Filho, D. Manual da Cultura do Milho; FUNEP: Jaboticabal, Brazil, 2007; 576p. [Google Scholar]
- Dantas Junior, E.E.; Chaves, L.H.G.; Fernandes, J.D. Lâminas de irrigação localizada e adubação potássica na produção de milho verde, em condições semiáridas. Rev. Espac. 2016, 37, 1–9. [Google Scholar]
- Lopes, J.R.F.; Dantas, M.P.; Ferreira, F.E.P. Identificação da influência da pluviometria no rendimento do milho no semiárido brasileiro. Rev. Bras. Agric. Irrig. 2019, 13, 3610–3618. [Google Scholar] [CrossRef]
- CONAB–Companhia Nacional de Abastecimento. Ministério da Agricultura, Pecuária e Abastecimento. Safra Brasileira de Grãos: Boletim de Grãos 2019/2020. Available online: https://portaldeinformacoes.conab.gov.br/safra-estimativa-de-evolucao-graos.html (accessed on 5 March 2023).
- Song, L.; Jin, J.; He, J. Effects of Severe Water Stress on Maize Growth Processes in the Field. Sustainability 2019, 11, 5086. [Google Scholar] [CrossRef]
- Sah, R.P.; Chakraborty, M.; Prasad, K.; Pandit, M.; Tudu, V.K.; Chakravarty, M.K.; Narayan, S.C.; Rana, M.; Moharana, D. Impact of water deficit stress in maize: Phenology and yield components. Sci. Rep. 2020, 10, 2944. [Google Scholar] [CrossRef]
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 1985; p. 174. [Google Scholar]
- SUDENE–Superintendência do Desenvolvimento do Nordeste. Nova Delimitação do Semiárido. 2017. Available online: http://antigo.sudene.gov.br/images/arquivos/semiarido/arquivos/Rela%C3%A7%C3%A3o_de_Munic%C3%Adpios_Semi%C3%A1rido.pdf (accessed on 9 January 2023).
- Frizzone, J.A.; Lima, S.C.R.V.; Lacerda, C.F.; Mateos, L. Socio-Economic Indexes for Water Use in Irrigation in a Representative Basin of the Tropical Semiarid Region. Water 2021, 13, 2643. [Google Scholar] [CrossRef]
- Marengo, J.A.; Bernasconi, M. Regional differences in aridity/drought conditions over Northeast Brazil: Present state and future projections. Clim. Change 2015, 129, 103–115. [Google Scholar] [CrossRef]
- Cavalcante Júnior, R.G.; Freitas, M.A.V.; Silva, N.F.; Azevedo Filho, F.R. Sustainable groundwater exploitation aiming at the reduction of water vulnerability in the Brazilian semi-arid region. Energies 2019, 12, 904. [Google Scholar] [CrossRef]
- Lessa, C.I.N.; de Lacerda, C.F.; Cajazeiras, C.C.D.A.; Neves, A.L.R.; Lopes, F.B.; Silva, A.O.D.; Sousa, H.C.; Gheyi, H.R.; Nogueira, R.D.S.; Lima, S.C.R.V.; et al. Potential of Brackish Groundwater for Different Biosaline Agriculture Systems in the Brazilian Semi-Arid Region. Agriculture 2023, 13, 550. [Google Scholar] [CrossRef]
- Rajabi Dehnavi, A.; Zahedi, M.; Ludwiczak, A.; Cardenas Perez, S.; Piernik, A. Effect of Salinity on Seed Germination and Seedling Development of Sorghum (Sorghum bicolor (L.) Moench) Genotypes. Agronomy 2020, 10, 859. [Google Scholar] [CrossRef]
- Isayenkov, S.V.; Maathuis, F.J.M. Plant salinity stress: Many unanswered questions remain. Front. Plant Sci. 2019, 10, 80. [Google Scholar] [CrossRef]
- Islam, A.T.M.T.; Koedsuk, T.; Ullah, H.; Tisarum, R.; Jenweerawat, S.; Cha-um, S.; Datta, A. Salt tolerance of hybrid baby corn genotypes in relation to growth, yield, physiological, and biochemical characters. S. Afr. J. Bot. 2022, 147, 808–819. [Google Scholar] [CrossRef]
- Lacerda, C.F.; Ferreira, J.F.S.; Suarez, D.L.; Freitas, E.D.; Liu, X.; Ribeiro, A.A. Evidence of nitrogen and potassium losses in soil columns cultivated with maize under salt stress. Rev. Bras. Eng. Agrícola Ambient. 2018, 22, 553–557. [Google Scholar] [CrossRef]
- Sousa, H.C.; Sousa, G.G.; Lessa, C.I.N.; Lima, A.F.S.; Ribeiro, R.M.R.; Costa, F.H.R. Growth and gas exchange of corn under salt stress and nitrogen doses. Rev. Bras. Eng. Agrícola Ambient. 2021, 25, 174–181. [Google Scholar] [CrossRef]
- Stadnik, B.; Tobiasz-Salach, R.; Mazurek, M. Effect of silicon on oat salinity tolerance: Analysis of the egipegenetic and physiological response of plants. Agriculture 2023, 13, 81. [Google Scholar] [CrossRef]
- Sousa, H.C.; Sousa, G.G.D.; Cambissa, P.B.C.; Lessa, C.I.N.; Goes, G.F.; Silva, F.D.B.D.; Abreu, F.D.S.; Viana, T.V.D.A. Gas exchange and growth of zucchini crop subjected to salt and water stress. Rev. Bras. Eng. Agrícola Ambient. 2022, 26, 815–822. [Google Scholar] [CrossRef]
- Goes, G.F.; Sousa, G.G.; Santos, S.O.; Silva Júnior, F.B.; Ceita, E.D.A.R.; Leite, K.N. Produtividade da cultura do amendoim sob diferentes supressões da irrigação com água salina. Irriga 2021, 26, 210–220. [Google Scholar] [CrossRef]
- Shilev, S. Plant-Growth-Promoting Bacteria Mitigating Soil Salinity Stress in Plants. Appl. Sci. 2020, 10, 7326. [Google Scholar] [CrossRef]
- Armanhi, J.S.L.; Souza, R.S.C.; Biazotti, B.B.; Yassitepe, J.E.D.C.T.; Arruda, P. Modulating drought stress response of maize by a synthetic bacterial community. Front. Microbiol. 2021, 12, 747541. [Google Scholar] [CrossRef]
- Poudel, M.; Mendes, R.; Costa, L.A.S.; Bueno, C.G.; Meng, Y.; Folimonova, S.Y.; Garrett, K.A.; Martins, S.J. The role of plant-associated bacteria, fungi, and viruses in drought stress mitigation. Front. Microbiol. 2021, 12, 743512. [Google Scholar] [CrossRef]
- Kavamura, V.N.; Santos, S.N.; Silva, J.L.; Parma, M.M.; Ávila, L.A.; Visconti, A.; Zucchi, T.D.; TaketanI, R.G.; Andreote, F.D.; Melo, I.S.D. Screening of Brazilian cacti rhizobacteria for plant growth promotion under drought. Microbiol. Res. 2013, 168, 183–191. [Google Scholar] [CrossRef]
- Niu, X.; Song, L.; Xiao, Y.; Ge, W. Drought-tolerant plant growth-promoting rhizobacteria associated with foxtail millet in a semi-arid agroecosystem and their potential in alleviating drought stress. Front. Microbiol. 2018, 8, 2580. [Google Scholar] [CrossRef] [PubMed]
- Sousa, S.M.; De Oliveira, C.A.; Andrade, D.L.; Carvalho, C.G.D.; Ribeiro, V.P.; Pastina, M.M.; Marriel, I.E.; Lana, U.G.D.P.; Gomes, E.A.P. Tropical Bacillus Strains Inoculation Enhances Maize Root Surface Area, Dry Weight, Nutrient Uptake and Grain Yield. J. Plant Growth Regul. 2021, 40, 867–877. [Google Scholar] [CrossRef]
- May, A.; Santos, M.D.S.; Silva, E.H.F.M.D.; Viana, R.D.S.; Vieira Junior, N.A.; Ramos, N.P.; Melo, I.S.D. Effect of Bacillus aryabhattai on the initial establishment of pre-sprouted seedlings of sugarcane varieties. Res. Soc. Dev. 2021, 10, 1–9. [Google Scholar] [CrossRef]
- Bittencourt, P.P.; Alves, A.F.; Ferreira, M.B.; da Silva Irineu, L.E.S.; Pinto, V.B.; Olivares, F.L. Mechanisms and Applications of Bacterial Inoculants in Plant Drought Stress Tolerance. Microorganisms 2023, 11, 502. [Google Scholar] [CrossRef]
- Etesami, H.; Beattie, G.A. Mining halophytes for plant growth-promoting halotolerant bacteria to enhance the salinity tolerance of non-halophytic crops. Front. Microbiol. 2018, 9, 148. [Google Scholar] [CrossRef]
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Gonçalves, J.L.M.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorol. Z. 2013, 22, 711–728. [Google Scholar] [CrossRef]
- Teixeira, P.C.; Donagemma, G.K.; Fontana, A.; Teixeira, W. Manual de Métodos de Análise de Solo, 3rd ed.; Embrapa: Brasília, Brazil, 2017; p. 573. [Google Scholar]
- de Souza, L.S.B.; de Moura, M.S.B.; Sediyama, G.C.; da Silva, T.G.F. Requerimento hídrico e coeficiente de cultura do milho e feijão-caupi em sistemas exclusivo e consorciado. Rev. Caatinga 2015, 28, 151–160. [Google Scholar] [CrossRef]
- Medeiros, J.F. Qualidade da água de Irrigação Utilizada nas Propriedades Assistidas pelo “GAT” nos Estados do RN, PB, CE e avaliação da Salinidade dos Solos. Master’s Thesis, Universidade Federal da Paraíba, Campina Grande, Brazil, 1992. Available online: http://dspace.sti.ufcg.edu.br:8080/jspui/handle/riufcg/2896 (accessed on 8 January 2023).
- Silva, F.C. Manual de Análises Químicas de Solos, Plantas e Fertilizantes; Embrapa Comunicação para Transferência de Tecnologia: Brasília, Brazil, 1999; p. 370. [Google Scholar]
- Richards, L.A. Diagnosis and Improvement of Saline and Alkali Soils; US Department of Agriculture: Washington, DC, USA, 1954; p. 160. [Google Scholar]
- Kavamura, V.N.; Santos, S.N.; Taketani, R.G.; Vasconcellos, R.L.; Melo, I.S. Draft genome sequence of plant growth-promoting drought-tolerant Bacillus sp. strain CMAA 1363 isolated from the Brazilian Caatinga biome. Genome Announc. 2017, 5, e01534-16. [Google Scholar] [CrossRef]
- Fernandes, V.L.B. Recomendações de Adubação e Calagem para o Estado do Ceará; UFC: Fortaleza, Brazil, 1993; p. 248. [Google Scholar]
- Silva, F.D.A.S.; Azevedo, C.A.V.D. The Assistat Software Version 7.7 and its use in the analysis of experimental data. Afr. J. Agric. Res. 2016, 11, 3733–3740. [Google Scholar]
- Bulgarelli, D.; Schlaeppi, K.; Spaepen, S.; Van Themaat, E.V.L.; Schulze-Lefert, P. Structure and functions of the bacterial microbiota of plants. Annu. Rev. Plant Biol. 2013, 64, 807–838. [Google Scholar] [CrossRef]
- Kavamura, V.N. Bactérias Associadas às Cactáceas da Caatinga: Promoção de Crescimento de Plantas sob Estresse Hídrico. Ph.D. Thesis, University of São Paulo, Piracicaba, Brazil, 2012. Available online: http://www.teses.usp.br/teses/disponiveis/11/11138/tde-25102012-095956/ (accessed on 10 February 2023).
- Oliveira, E.J.; Melo, H.C.D.E.; Trindade, K.L.; Guedes, T.D.E.M.; Sousa, C.M. Morphophysiology and yield of green corn cultivated under different water depths and nitrogen doses in the cerrado conditions of Goiás, Brazil. Res. Soc. Dev. 2020, 9, e6179108857. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E.; Moller, I.M.; Murphy, A. Fisiologia e Desenvolvimento Vegetal, 6th ed.; Artmed: Porto Alegre, Brazil, 2017; 858p. [Google Scholar]
- Irmak, S.; Mohammed, A.T.; Kukal, M.S. Maize response to coupled irrigation and nitrogen fertilisation under center pivot, subsurface drip and surface (furrow) irrigation: Growth, development and productivity. Agric. Water Manag. 2020, 263, 107457. [Google Scholar] [CrossRef]
- Qiu, Y.; Fan, Y.; Chen, Y.; Hao, X.; Li, S.; Kang, S. Response of dry matter and water use efficiency of alfalfa to water and salinity stress in arid and semiarid regions of Northwest China. Agric. Water Manag. 2021, 254, 106934. [Google Scholar] [CrossRef]
- Lin, Y.; Wattes, D.B.; Kloepper, J.W.; Feng, Y.; Torbert, H.A. Influence of plant growth-promoting rhizobacteria on corn growth under drought stress. Commun. Soil Sci. Plant Anal. 2019, 51, 250–264. [Google Scholar] [CrossRef]
- Ricardi, M.; Rosa, H.A. Desenvolvimento inicial do milho submetido a estresse salino. Rev. Cultiv. Saber 2018, 1, 174–184. [Google Scholar]
- Sá, F.; Ferreira Neto, M.; Lima, Y.; Paiva, E.; Prata, R.; Lacerda, C.; Brito, M. Growth, gas exchange and photochemical efficiency of the cowpea bean under salt stress and phosphorus fertilisation. Comum. Sci. 2018, 9, 668–679. [Google Scholar] [CrossRef]
- de Souza, M.V.; de Sousa, G.G.; da Silva Sales, J.R.; da Costa Freire, M.H.; da Silva, G.L.; de Araújo Viana, T.V. Água salina e biofertilisantes de esterco bovino e caprino na salinidade do solo, crescimento e fisiologia da fava. Rev. Bras. Cienc. Agrar. 2019, 14, 340–349. [Google Scholar]
- Oliveira, F.D.; Medeiros, J.F.; Cunha, R.C.; Souza, M.W.; Lima, L.A. Uso de bioestimulante como agente amenizador do estresse salino na cultura do milho pipoca. Cienc. Agron. 2016, 47, 307–315. [Google Scholar]
- Naseem, H.; Bano, A. Role of plant growth-promoting rhizobacteria and their exopolysaccharide in drought tolerance of maize. J. Plant Interact. 2014, 9, 689–701. [Google Scholar] [CrossRef]
- Prado, R.M. Nutrição de Plantas, 2nd ed.; UNESP: São Paulo, Brazil, 2020; 414p. [Google Scholar]
- Jaghdani, S.J.; Janhns, P.; Trankner, M. The impact of magnesium deficiency on photosynthesis and photoprotection in Spinacia oleracea. Plant Stress 2021, 2, 1–11. [Google Scholar] [CrossRef]
- Lima, A.F.; Santos, M.F.; Oliveira, M.L.; Sousa, G.G.; Mendes Filho, P.F.; Luz, L.N. Physiological responses of inoculated and uninoculated peanuts under saline stress. Rev. Ambiente Água 2021, 16, e2643. [Google Scholar] [CrossRef]
- Cao, M.; Narayanan, M.; Shi, X.; Chen, X.; Li, Z.; Ma, Y. Optimistic contributions of plant growth-promoting bacteria for sustainable agriculture and climate stress alleviation. Environ. Res. 2023, 217, 114924. [Google Scholar] [CrossRef] [PubMed]
- Vishnupradeep, R.; Bruno, L.B.; Taj, Z.; Karthik, C.; Challabathula, D.; Kumar, A.; Freitas, H.; Rajkumar, M. Plant growth promoting bacteria improve growth and phytostabilization potential of Zea mays under chromium and drought stress by altering photosynthetic and antioxidant responses. Environ. Technol. Innov. 2022, 25, 102154. [Google Scholar] [CrossRef]
- Mishra, P.; Mishra, J.; Arora, N.K. Plant growth promoting bacteria for combating salinity stress in plants–Recent developments and prospects: A review. Microbiol. Res. 2021, 252, 126861. [Google Scholar] [CrossRef] [PubMed]
- Sales, J.R.; Magalhâes, C.L.; Freitas, A.G.; Goes, G.F.; Sousa, H.C.; Sousa, G.G. Índices fisiológicos de quiabeiro irrigado com água salina sob adubação organomineral. Rev. Bras. Eng. Agrícola Ambient. 2021, 25, 466–471. [Google Scholar] [CrossRef]
- Liao, Q.; Gu, S.; Kang, S.; Du, T.; Tong, L.; Wood, J.D.E.; Ding, R. Mild water and salt stress improve water use efficiency by decreasing stomatal conductance via osmotic adjustment in field maize. Sci. Total Environ. 2022, 805, 150364. [Google Scholar] [CrossRef]
- Rodrigues, V.D.S.; Sousa, G.G.D.; Soares, S.D.C.; Leite, K.N.; Ceita, E.D.; Sousa, J.T.M.D. Gas exchanges and mineral content of corn crops irrigated with saline water. Rev. Ceres 2021, 68, 453–459. [Google Scholar] [CrossRef]
- Nascimento, F.N.; Bastos, E.A.; Cardoso, M.J.; Júnior, A.S.A.; Ribeiro, V.Q. Parâmetros fisiológicos e produtividade de espigas verdes de milho sob diferentes lâminas de irrigação. Rev. Bras. Milho Sorgo 2015, 14, 167–181. [Google Scholar] [CrossRef]
- Hessini, K.; Issaoui, K.; Ferchichi, S.; Saif, T.; Abdelly, C.; Siddique, K.H.M.; Cruz, C. Interactive effects of salinity and nitrogen forms on plant growth, photosynthesis and osmotic adjustment in maize. Plant Physiol. Biochem. 2019, 139, 171–178. [Google Scholar] [CrossRef]
- Abdel Latef, A.A.H.; Abu Alhmad, M.F.; Kordrostami, M.; Abo-Baker, A.B.A.E.; Zakir, A. Inoculation with Azospirillum lipoferum or Azotobacter chroococcum Reinforces Maize Growth by Improving Physiological Activities Under Saline Conditions. J. Plant Growth Regul. 2020, 39, 1293–1306. [Google Scholar] [CrossRef]
- Cavalcante, E.S.; Lacerda, C.F.; Mesquita, R.O.; de Melo, A.S.; da Silva Ferreira, J.F.; dos Santos Teixeira, A.; Lima, S.C.R.V.; da Silva Sales, J.R.; de Souza Silva, J.; Gheyi, H.R. Supplemental Irrigation with Brackish Water Improves Carbon Assimilation and Water Use Efficiency in Maize under Tropical Dryland Conditions. Agriculture 2022, 12, 544. [Google Scholar] [CrossRef]
- Costa-Gutierrez, S.B.; Raimondo, E.E.; Lami, M.J.; Vincent, P.A.; Espinosa-Urgel, M.; Cristóbal, R.E. Inoculation of Pseudomonas mutant strains can improve growth of soybean and corn plants in soils under salt stress. Rhizosphere 2020, 16, 100255. [Google Scholar] [CrossRef]
- Shukla, P.S.; Agarwal, P.K.; Jha, B. Improved salinity tolerance of Arachis hypogaea (L.) by the interaction of halotolerant plant-growth-promoting rhizobacteria. J. Plant Growth Regul. 2012, 31, 195–206. [Google Scholar] [CrossRef]
- Cavallet, L.E.; Pessoa, A.C.S.; Helmich, J.J.; Helmich, P.R.; Ost, C.F. Produtividade do milho em resposta à aplicação de nitrogênio e inoculação das sementes com Azospirillum spp. Rev. Bras. Eng. Agric. Ambient. 2000, 4, 129–132. [Google Scholar] [CrossRef]
- Freire, M.H.; Viana, T.V.; Sousa, G.G.; Azevedo, B.M.; Sousa, H.C.; Goes, G.F.; Lessa, C.I.; da Silva, F.D. Organic fertilisation and salt stress on the agronomic performance of maize crop. Rev. Bras. Eng. Agrícola Ambient. 2022, 26, 848–854. [Google Scholar] [CrossRef]
- Marrocos, S.T.P.; Junior, J.N.; Granjeiro, L.C.; Ambrosio, M.M.D.Q.; Da Cunha, A.P.A. Composição química e microbiológica de biofertilisantes em diferentes tempos de decomposição. Rev. Caatinga 2012, 25, 34–43. [Google Scholar]
- Kumar, M.; Mishra, S.; Dixit, V.; Kumar, M.; Agarwal, L.; Chauhan, P.S.; Nautiyal, C.S. Synergistic effect of Pseudomonas putida and Bacillus amyloliquefaciens ameliorates drought stress in chickpea (Cicer arietinum L.). Plant Signal. Behav. 2016, 11, e1071004. [Google Scholar] [CrossRef]
- EMBRAPA-Empresa Brasileira de Pesquisa Agropecuária. Boletim de Pesquisa e Desenvolvimento. Isolamento e Potencial Uso de Bactérias do Gênero Bacillus na Promoção de Crescimento de Plantas em Condições de Déficit Hídrico. 2019. Available online: https://www.infoteca.cnptia.embrapa.br/infoteca/bitstream/doc/1114738/1/bol192.pdf (accessed on 1 March 2023).
- Qurashi, A.W.; Sabri, A.N. Bacterial exopolysaccharide and biofilm formation stimulate chickpea growth and soil aggregation under salt stress. Braz. J. Microbiol. 2012, 43, 1183–1191. [Google Scholar] [CrossRef]
- Araújo, V.L.V.P.; Lira Júnior, M.A.; Souza Júnior, V.S.; Araújo Filho, J.C.; Fracetto, F.J.C.; Andreote, F.D.; Pereira, A.P.A.; Mendes Júnior, J.P.; Barros, F.M.R.; Fracetto, G.G.M. Bacteria fromm tropical semiarid temporary ponds promote maize growth under water stress. Microbiol. Res. 2020, 24, 126564. [Google Scholar] [CrossRef]
- Kumar, V.; Raghuvanshi, N.; Pandey, A.K.; Kumar, A.; Thoday-Kennedy, E.; Kant, S. Role of halotolerant plant growth-promoting rhizobacteria in mitigating salinity stress: Recent advances and possibilities. Agriculture 2023, 13, 168. [Google Scholar] [CrossRef]
- Rodrigues, V.S.; Bezerra, F.M.L.; Sousa, G.G.; Fiusa, J.N.; Viana, T.V.A. Yield of maize crop irrigated with saline waters. Rev. Bras. Eng. Agric. Ambient. 2020, 24, 101–105. [Google Scholar] [CrossRef]
- Arora, N.K.; Tewari, S.; Singh, R. Multifaceted plant-associated microbes and their mechanisms diminish the concept of direct and indirect PGPRs. In Plant-Microbe Symbiosis: Fundamentals and Advances; Arora, N.K., Ed.; Springer: New Delhi, India, 2013; pp. 411–449. [Google Scholar]
- Cavalcante Filho, H.A.; Helcias, R.P.; Pereira, R.G.; Cavalcante, M.; Medeiros, P.V.Q.; Andrade, A.R.S. Desempenho de cultivares de milho para produção de milho verde, minimilho e forragem. Rev. Ciências Agroveterinárias 2022, 21, 449–455. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X.; Simunek, J.; Shi, H.; Chen, N.; Hu, Q. Optimizing drip irrigation with alternate use of fresh and brackish waters by analyzing salt stress: The experimental and simulation approaches. Soil Tillage Res. 2022, 219, 105355. [Google Scholar] [CrossRef]
- Mazzuchelli, R.C.L.; Sossai, B.F.; Araujo, F.F. Inoculação de Bacillus subtilis e Azospirillum brasilense na cultura do milho. Colloq. Agrar. 2014, 10, 40–47. [Google Scholar] [CrossRef]
- Silva, G.C.; Schmitz, R.; Silva, L.C.; Carpanini, G.G.; Magalhães, R.C. Desempenho de cultivares para pro-dução de milho verde na agricultura familiar do sul de Roraima. Rev. Bras. Milho Sorgo 2015, 14, 273–282. [Google Scholar] [CrossRef]













| pH | OM | N | C | P | Ca | Mg | Na | Al | H + Al | K | ECse | ESP | C/N | V | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2O | g kg−1 | mg kg−1 | cmolc dm−3 | dS m−1 | % | % | |||||||||
| 5.6 | 11.59 | 0.71 | 6.72 | 20 | 3.20 | 2.60 | 0.07 | 0.35 | 2.15 | 0.17 | 0.76 | 1 | 9 | 74 | |
| SD (g cm−3) | CS | FS | Silt | Clay | Textural Classification | ||||||||||
| Bulk | Particle | g kg−1 | |||||||||||||
| 1.31 | 2.61 | 507 | 283 | 133 | 77 | Loamy Sand | |||||||||
| ECw (dS m−1) | ETc (%) | Total Depth Applied (mm) | |
|---|---|---|---|
| Uninoculated | Inoculated | ||
| 0.3 | 50 | 260.4 | 260.4 |
| 75 | 390.6 | 390.6 | |
| 100 | 520.8 | 520.8 | |
| 3.0 | 50 | 260.4 | 260.4 |
| 75 | 390.6 | 390.6 | |
| 100 | 520.8 | 520.8 | |
| ECw | Ca2+ | Mg2+ | K+ | Na+ | Cl− | HCO3− | pH | CE | SAR | Classification 1 |
|---|---|---|---|---|---|---|---|---|---|---|
| dS m−1 | mmolc L−1 | mmol L−1 | in H2O | dS m−1 | (mmolc L−1)0.5 | |||||
| 0.3 | 0.6 | 1.4 | 0.2 | 0.4 | 2.5 | 0.1 | 6.9 | 0.3 | 0.4 | C2S1 |
| 3.0 | 6.33 | 7.64 | 2.0 | 15.6 | 25 | 1.0 | 7.79 | 3.0 | 5.9 | C4S2 |
| Organic Source | N | P | K+ | Ca2+ | Mg2+ |
|---|---|---|---|---|---|
| g L−1 | |||||
| Cattle manure | 0.96 | 0.47 | 0.59 | 1.10 | 0.25 |
| Cattle biofertiliser | 0.82 | 1.4 | 1.0 | 2.5 | 0.75 |
| Source of Variation | DF | Mean Square | |||
|---|---|---|---|---|---|
| PH | NL | SD | LA | ||
| Blocks | 5 | 29.67 ns | 1.95 ns | 1.53 ns | 207.38 ns |
| ECw | 1 | 0.06 ns | 2.60 ns | 87.96 ** | 5613.37 * |
| Residual (ECw) | 5 | 27.97 | 0.61 | 2.74 | 412.05 |
| Irrigation depths (ID) | 2 | 21.30 ns | 0.40 ns | 56.08 ** | 10,546.35 ** |
| Residual (ID) | 20 | 15.37 | 0.43 | 2.49 | 974.55 |
| Inoculation (INOC) | 1 | 9.56 ns | 0.33 ns | 35.25 ** | 10,360.73 ** |
| Residual (INOC) | 30 | 16.57 | 0.54 | 3.05 | 1161.23 |
| ECw × ID | 2 | 160.75 ** | 0.25 ns | 1.87 ns | 2864.11 ns |
| ECw × INOC | 1 | 149.91 ** | 0.004 ns | 0.0007 * | 2064.59 ns |
| ID × INOC | 2 | 0.24 * | 0.16 ns | 0.27 ns | 5769.978 * |
| ECw × ID × INOC | 2 | 70.49 * | 1.42 ns | 4.56 ns | 654.36 ns |
| CV (%)—Ecw | 5.46 | 9.49 | 12.68 | 5.47 | |
| CV (%)—ID | 4.05 | 7.97 | 12.08 | 8.41 | |
| CV (%)—INOC | 4.20 | 8.95 | 13.37 | 9.18 | |
| Source of Variation | DF | Mean Square | ||||||
|---|---|---|---|---|---|---|---|---|
| A | gs | Ci | E | RCI | LT | WUEi | ||
| Blocks | 5 | 9.03 ns | 0.67 ns | 798.40 ns | 0.43 ** | 10.05 ns | 3.67 ns | 0.006 * |
| ECw | 1 | 361.19 ** | 0.15 ns | 4504.68 * | 36.83 ** | 191.12 ** | 74.72 ** | 11.07 ** |
| Residual (ECw) | 5 | 7.25 | 0.16 | 303.63 | 0.00 ** | 2.94 | 0.61 | 0.28 |
| Irrigation depths (ID) | 2 | 2.14 ns | 0.26 ns | 315.25 ns | 0.25 ns | 92.35 * | 0.77 * | 0.22 ns |
| Residual (ID) | 20 | 8.90 | 0.14 | 101.83 | 0.11 | 23.43 | 0.15 | 0.15 |
| Inoculation (INOC) | 1 | 2.13 ns | 3.60 ** | 336.02 ns | 0.11 ns | 39.45 ns | 0.04 * | 0.01 ns |
| Residual (INOC) | 30 | 3.13 | 0.18 | 110.47 | 0.16 | 9.78 | 0.02 | 0.21 |
| ECw × ID | 2 | 2.86 ns | 1.46 ** | 9.75 ns | 0.58 * | 66.17 ns | 0.67 * | 0.01 ns |
| ECw × INOC | 1 | 6.97 * | 2.48 ** | 595.02 * | 0.47 ns | 0.09 ns | 0.04 ns | 0.008 ns |
| ID × INOC | 2 | 1.88 ns | 0.04 ns | 234.33 ns | 0.27 ns | 4.97 ns | 0.10 * | 0.006 ns |
| ECw × ID × INOC | 2 | 0.78 ns | 2.57 ** | 110.47 ns | 0.18 ns | 0.87 ns | 0.02 ns | 0.27 ns |
| CV (%)—ECw | 11.20 | 11.85 | 6.34 | 0.77 | 5.54 | 2.59 | 9.60 | |
| CV (%)—ID | 12.41 | 10.04 | 3.67 | 7.88 | 15.62 | 1.32 | 7.03 | |
| CV (%)—INOC | 7.37 | 14.05 | 3.82 | 9.11 | 10.09 | 0.50 | 8.31 | |
| Source of Variation | DF | Mean Square | |||
|---|---|---|---|---|---|
| EL | ED | EYWS | EYWoS | ||
| Blocks | 5 | 3.09 ns | 17.63 ns | 6,772,473.13 ns | 3,162,178.48 ns |
| ECw | 1 | 5.15 ns | 9.90 ns | 40,862,788.82 ** | 14,079,648.00 ** |
| Residual (ECw) | 5 | 1.69 | 4.58 | 1,711,897.76 | 820,739.41 |
| Irrigation depths (ID) | 2 | 1.35 ns | 5.27 ns | 3,121,961.40 ns | 1,275,678.96 * |
| Residual (ID) | 20 | 1.49 | 3.63 | 1,464,975.55 | 351,695.38 |
| Inoculation (INOC) | 1 | 0.14 ns | 0.38 ns | 3,533,704.78 ** | 450,274.80 * |
| Residual (INOC) | 30 | 0.86 | 1.96 | 363,835.20 | 309,105.44 |
| ECw × ID | 2 | 0.34 ns | 1.57 ns | 5,385,095.95 * | 1,002,466.33 ns |
| ECw × INOC | 1 | 3.78 * | 2.51 ns | 1,257,793.16 ns | 69,497.37 ns |
| ID × INOC | 2 | 6.11 ** | 1.65 ns | 956,622.93 ns | 225,383.24 ns |
| ECw × ID × INOC | 2 | 1.27 ns | 5.31 ns | 1,678,902.68 * | 1,118,325.91 * |
| CV (%)—ECw | 10.92 | 6.24 | 29.91 | 29.74 | |
| CV (%)—ID | 10.24 | 5.56 | 27.67 | 19.47 | |
| CV (%)—INOC | 7.80 | 4.08 | 13.79 | 18.25 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castelo Sousa, H.; Gomes de Sousa, G.; de Araújo Viana, T.V.; Prudêncio de Araújo Pereira, A.; Nojosa Lessa, C.I.; Pires de Souza, M.V.; da Silva Guilherme, J.M.; Ferreira Goes, G.; da Silveira Alves, F.G.; Primola Gomes, S.; et al. Bacillus aryabhattai Mitigates the Effects of Salt and Water Stress on the Agronomic Performance of Maize under an Agroecological System. Agriculture 2023, 13, 1150. https://doi.org/10.3390/agriculture13061150
Castelo Sousa H, Gomes de Sousa G, de Araújo Viana TV, Prudêncio de Araújo Pereira A, Nojosa Lessa CI, Pires de Souza MV, da Silva Guilherme JM, Ferreira Goes G, da Silveira Alves FG, Primola Gomes S, et al. Bacillus aryabhattai Mitigates the Effects of Salt and Water Stress on the Agronomic Performance of Maize under an Agroecological System. Agriculture. 2023; 13(6):1150. https://doi.org/10.3390/agriculture13061150
Chicago/Turabian StyleCastelo Sousa, Henderson, Geocleber Gomes de Sousa, Thales Vinícius de Araújo Viana, Arthur Prudêncio de Araújo Pereira, Carla Ingryd Nojosa Lessa, Maria Vanessa Pires de Souza, José Marcelo da Silva Guilherme, Geovana Ferreira Goes, Francisco Gleyson da Silveira Alves, Silas Primola Gomes, and et al. 2023. "Bacillus aryabhattai Mitigates the Effects of Salt and Water Stress on the Agronomic Performance of Maize under an Agroecological System" Agriculture 13, no. 6: 1150. https://doi.org/10.3390/agriculture13061150
APA StyleCastelo Sousa, H., Gomes de Sousa, G., de Araújo Viana, T. V., Prudêncio de Araújo Pereira, A., Nojosa Lessa, C. I., Pires de Souza, M. V., da Silva Guilherme, J. M., Ferreira Goes, G., da Silveira Alves, F. G., Primola Gomes, S., & Barbosa da Silva, F. D. (2023). Bacillus aryabhattai Mitigates the Effects of Salt and Water Stress on the Agronomic Performance of Maize under an Agroecological System. Agriculture, 13(6), 1150. https://doi.org/10.3390/agriculture13061150








