Synergistic Effect of Polyethylene Glycol and Lactic Acid on Handling Properties and Antibacterial Efficacy of Premixed Calcium Silicate Cement
Abstract
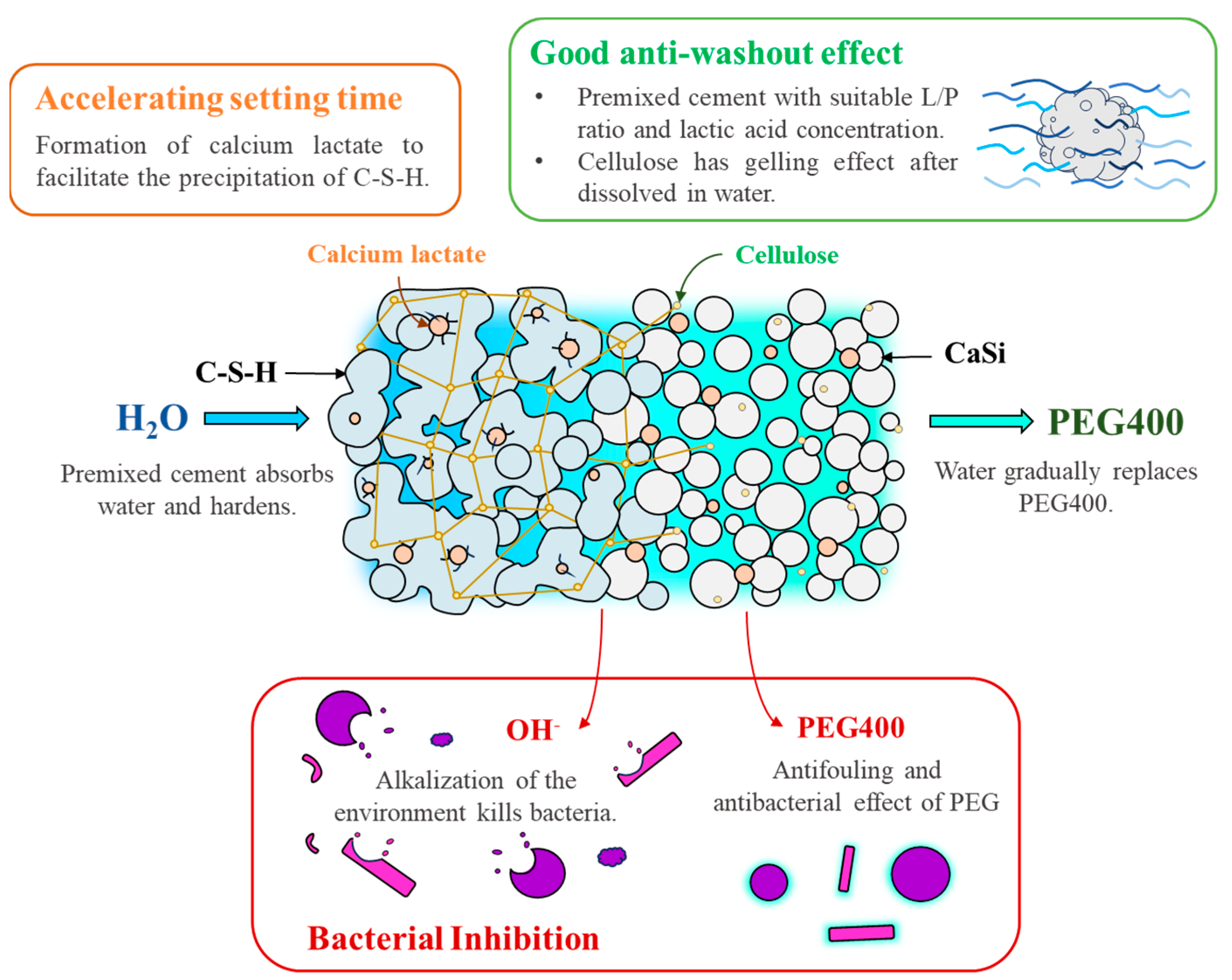
1. Introduction
2. Materials and Methods
2.1. Preparation of Casi Powder
2.2. Preparation of Premixed Paste and Cement
2.3. Phase Composition and Surface Morphology
2.4. Setting Time
2.5. Diametral Tensile Strength
2.6. pH Value
2.7. Anti-Washout Ability of Paste
2.8. Cytotoxicity
2.9. Bacteriostasis
2.10. Statistical Analysis
3. Results
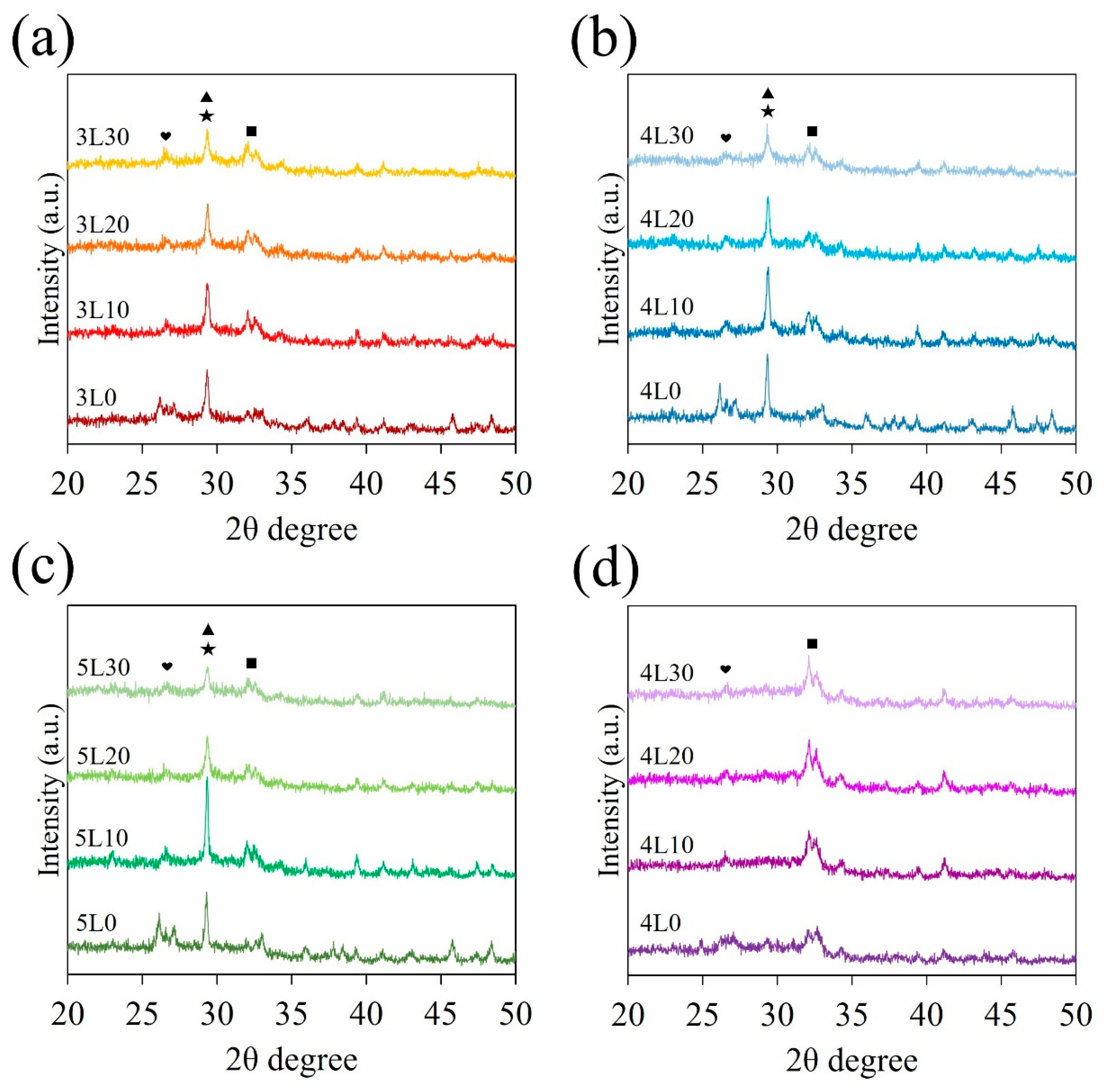
3.1. Phase Composition
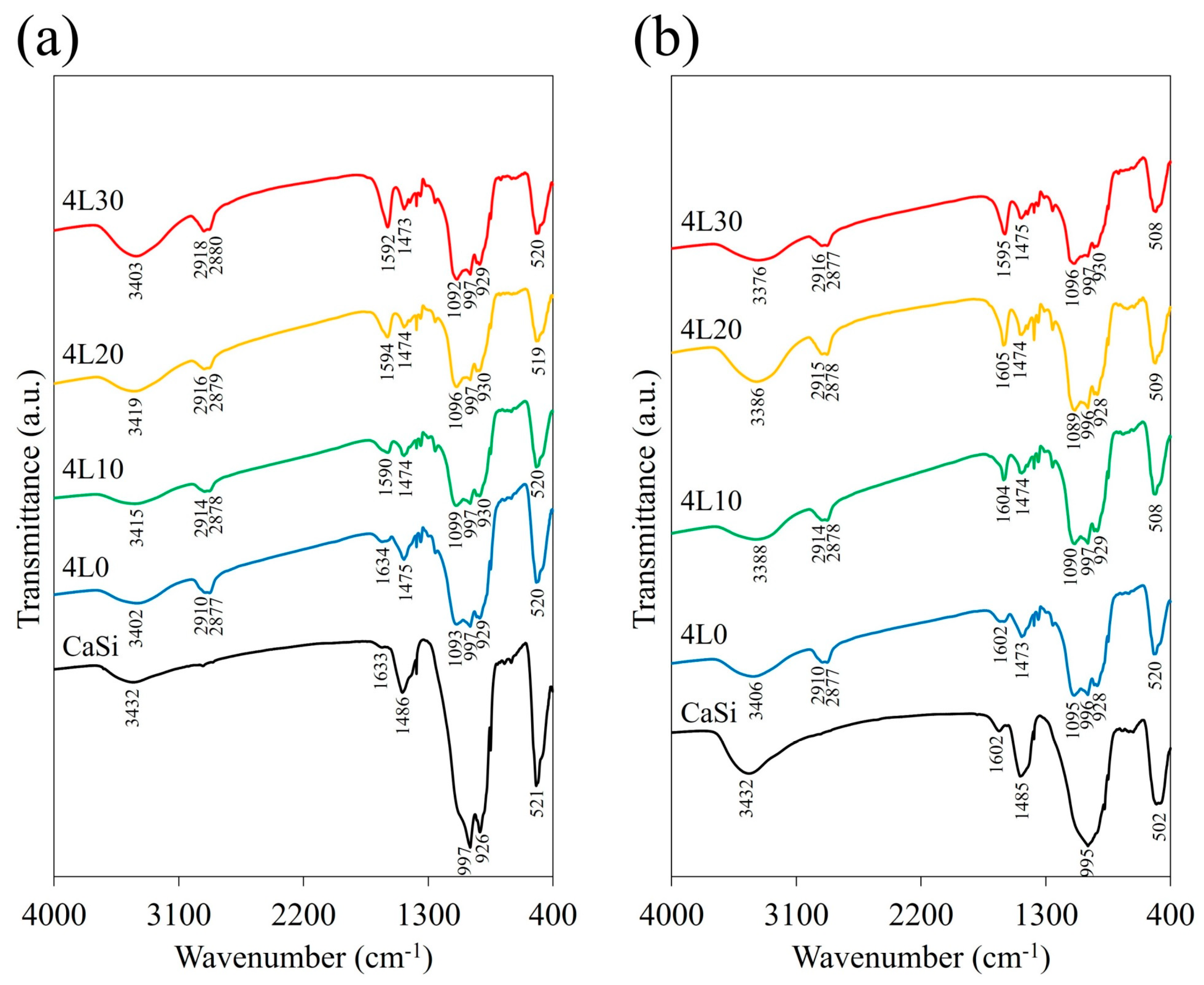
3.2. Surface Morphology
3.3. Setting Time
3.4. Diametral Tensile Strength
3.5. pH Value
3.6. Anti-Washout Ability
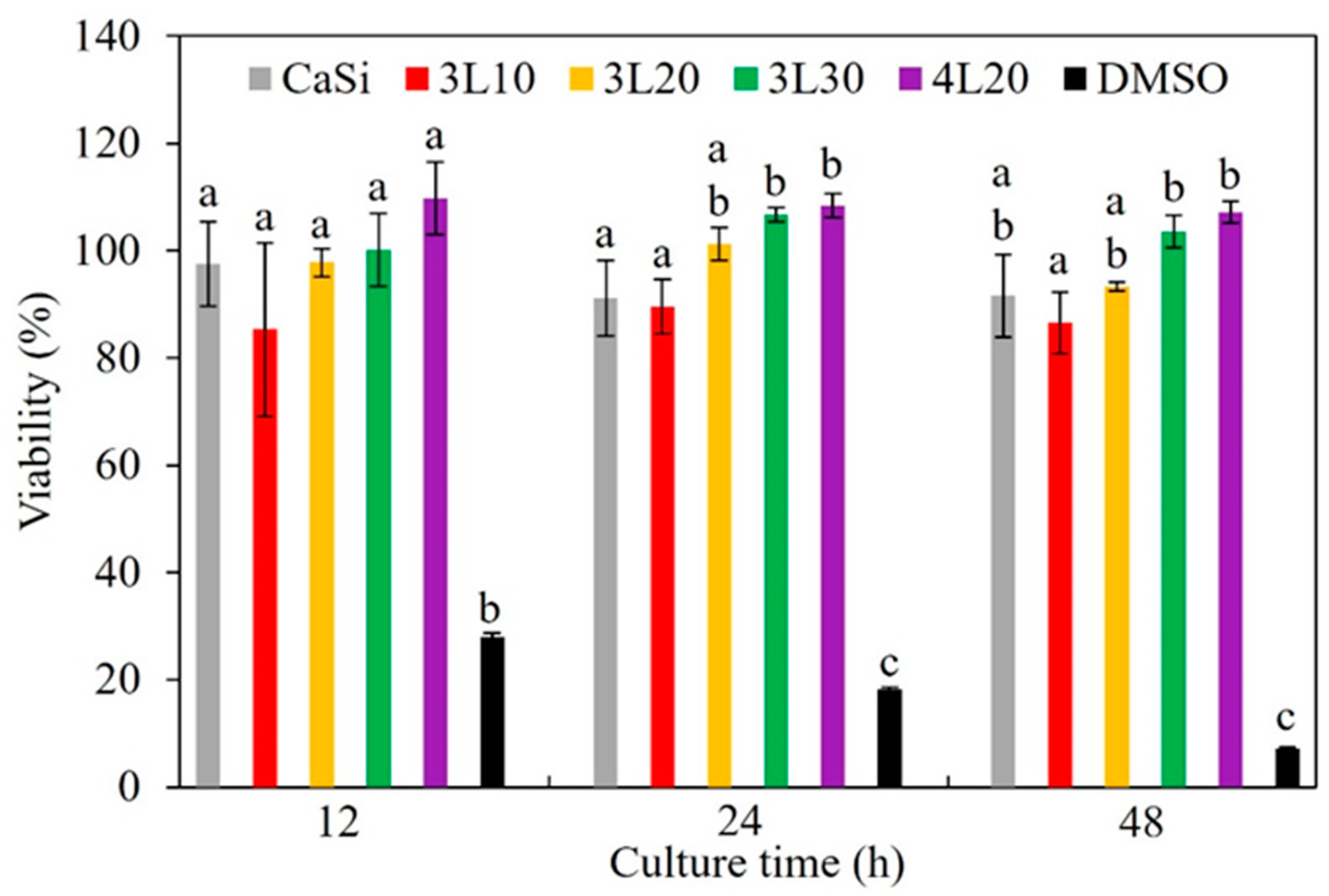
3.7. Cytotoxicity
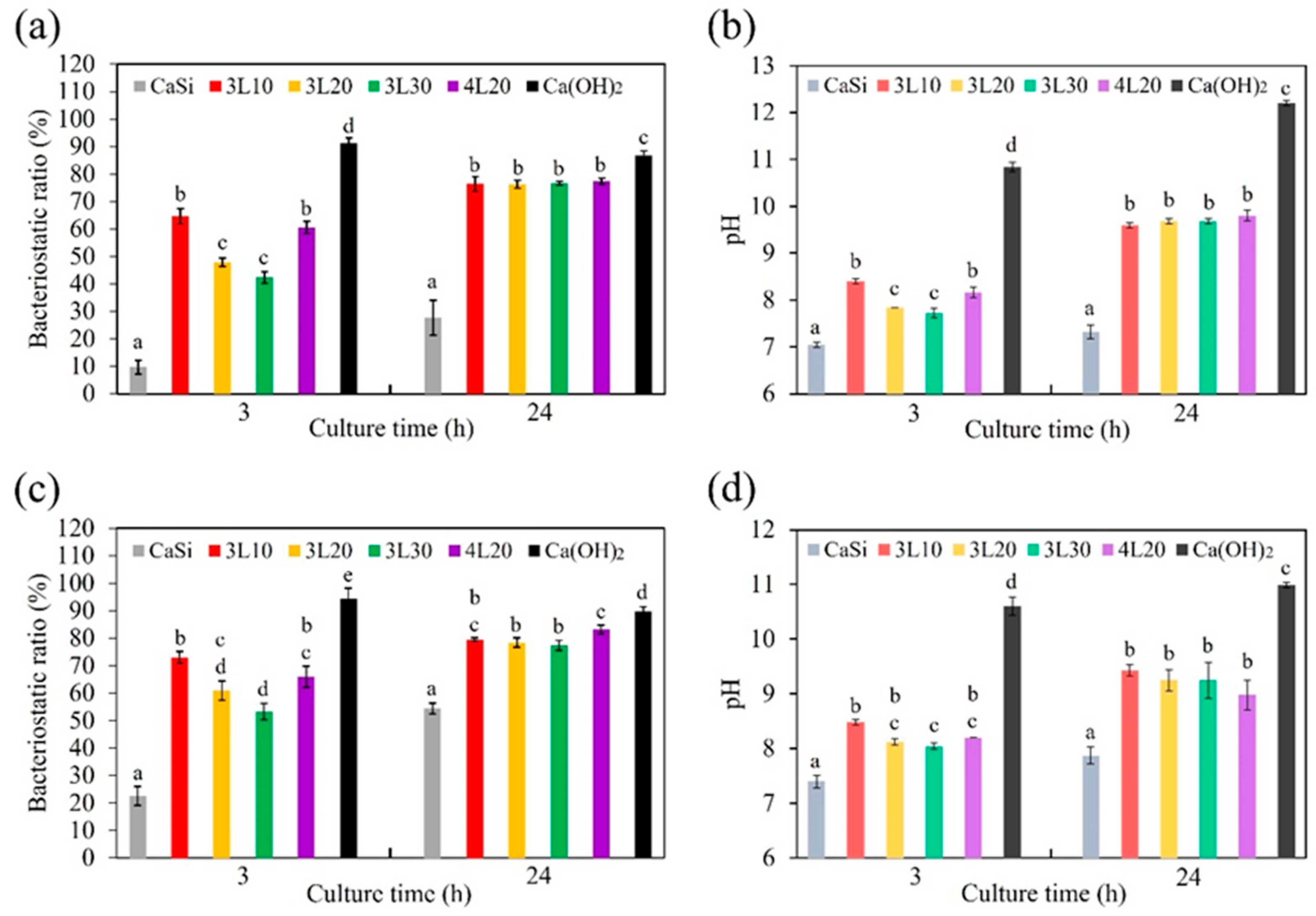
3.8. Anti-Bacterial Property
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Takagi, S.; Chow, L.C.; Hirayama, S.; Sugawara, A. Premixed calcium-phosphate cement pastes. J. Biomed. Mater. Res. 2003, 67, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, A.; Asaoka, K.; Ding, S.J. Calcium phosphate-based cements: Clinical needs and recent progress. J. Mater. Chem. B 2013, 1, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Carey, L.E.; Simon, C.G., Jr.; Takagi, S.; Chow, L.C. Premixed calcium phosphate cements: Synthesis, physical properties, and cell cytotoxicity. Dent. Mater. 2007, 23, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.; Rössler, S.; Lemm, M.; Ruhnow, M.; Nies, B. Properties of injectable ready-to-use calcium phosphate cement based on water-immiscible liquid. Acta Biomater. 2013, 9, 6199–6207. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Ma, P.W.; Zhang, L.L.; Yin, Y.J.; Yao, K.D.; Zhang, F.J.; Zhang, Y.D.; Li, X.L.; Nie, W. β-TCP/MCPM-based premixed calcium phosphate cements. Acta Biomater. 2009, 5, 3165–3177. [Google Scholar] [CrossRef] [PubMed]
- Irbe, Z.; Loca, D. Soluble phosphate salts as setting aids for premixed calcium phosphate bone cement pastes. Ceram. Int. 2021, 47, 24012–24019. [Google Scholar] [CrossRef]
- Lode, A.; Heiss, C.; Knapp, G.; Thomas, J.; Nies, B.; Gelinsky, M.; Schumacher, M. Strontium-modified premixed calcium phosphate cements for the therapy of osteoporotic bone defects. Acta Biomater. 2018, 65, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yu, S.; Zhang, D.; Zhang, W.; Zhang, H.; Zou, J.; Mao, Z.; Yuan, Y.; Gao, C.; Liu, R. Impact of Antifouling PEG Layer on the Performance of Functional Peptides in Regulating Cell Behaviors. J. Am. Chem. Soc. 2019, 141, 16772–16780. [Google Scholar] [CrossRef] [PubMed]
- Leppäranta, O.; Vaahtio, M.; Peltola, T.; Zhang, D.; Hupa, L.; Hupa, M.; Ylänen, H.; Salonen, J.I.; Viljanen, M.K.; Eerola, E. Antibacterial effect of bioactive glasses on clinically important anaerobic bacteria in vitro. J. Mater. Sci. Mater. Med. 2008, 19, 547–551. [Google Scholar] [CrossRef]
- Huang, S.C.; Wu, B.C.; Ding, S.J. Stem cell differentiation-induced calcium silicate cement with bacteriostatic activity. J. Mater. Chem. B 2015, 3, 570–580. [Google Scholar] [CrossRef]
- Hachem, C.E.; Chedid, J.C.A.; Nehme, W.; Kaloustian, M.K.; Ghosn, N.; Sahnouni, H.; Mancino, D.; Haikel, Y.; Kharouf, N. Physicochemical and Antibacterial Properties of Conventional and Two Premixed Root Canal Filling Materials in Primary Teeth. J. Funct. Biomater. 2022, 13, 177. [Google Scholar] [CrossRef] [PubMed]
- Koutroulis, A.; Valen, H.; Ørstavik, D.; Kapralos, V.; Camilleri, J.; Sunde, P.T. Antibacterial Activity of Root Repair Cements in Contact with Dentin—An Ex Vivo Study. J. Funct. Biomater. 2023, 14, 511. [Google Scholar] [CrossRef] [PubMed]
- Wu, I.T.; Chu, Y.H.; Huang, Y.R.; Chen, C.C.; Ding, S.J. Antibacterial ability and osteogenic activity of polyphenols-tailored calcium silicate bone cement. J. Mater. Chem. B 2022, 10, 4640–4649. [Google Scholar] [CrossRef] [PubMed]
- Chiang, T.Y.; Ding, S.J. Comparative physicochemical and biocompatible properties of radiopaque dicalcium silicate cement and mineral trioxide aggregate. J. Endod. 2010, 36, 1683–1687. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhou, Y.; Xia, L.; Zhao, C.; Chen, L.; Yi, D.; Chang, J.; Huang, L.; Zheng, X.; Zhu, H.; et al. Fabrication of nano-structured calcium silicate coatings with enhanced stability, bioactivity and osteogenic and angiogenic activity. Colloids Surf. B Biointerfaces 2015, 126, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Hakki, S.S.; Bozkurt, S.B.; Hakki, E.E.; Belli, S. Effects of Mineral Trioxide Aggregate on Cell Survival, Gene Expression Associated with Mineralized Tissues, and Biomineralization of Cementoblasts. J. Endod. 2009, 35, 513–519. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Taddei, P.; Modena, E.; Siboni, F.; Prati, C. Biointeractivity-related versus chemi/physisorption-related apatite precursor-forming ability of current root end filling materials. J. Biomed. Mater. Res. 2013, 101, 1107–1328. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.; Lu, H.; Chen, L.; Lin, X.; Huang, Y.; Dai, K.; Naoki, K.; Chen, G.; Chang, J. Silicate bioceramics induce angiogenesis during bone regeneration. Acta Biomater. 2012, 8, 341–349. [Google Scholar] [CrossRef]
- Wu, I.T.; Kao, P.F.; Huang, Y.R.; Ding, S.J. In vitro and in vivo osteogenesis of gelatin-modified calcium silicate cement with washout resistance. Mater. Sci. Eng. C 2020, 117, 111297. [Google Scholar] [CrossRef]
- Yousefi-Koma, A.A.; Assadian, H.; Mohaghegh, S.; Nokhbatolfoghahaei, H. Comparative Biocompatibility and Odonto-/Osteogenesis Effects of Hydraulic Calcium Silicate-Based Cements in Simulated Direct and Indirect Approaches for Regenerative Endodontic Treatments: A Systematic Review. J. Funct. Biomater. 2023, 14, 446. [Google Scholar] [CrossRef]
- Kato, G.; Araújo, R.; Rodrigues, C.; Gomes, P.S.; Grenho, L.; Fernandes, M.H. Ex vivo osteogenesis induced by calcium silicate-based cement extracts. J. Funct. Biomater. 2023, 14, 314. [Google Scholar] [CrossRef] [PubMed]
- Edanami, N.; Takenaka, S.; Ibn Belal, R.S.; Yoshiba, K.; Takahara, S.; Yoshiba, N.; Ohkura, N.; Noiri, Y. In vivo assessment of the apatite-forming ability of new-generation hydraulic calcium silicate cements using a rat subcutaneous implantation model. J. Funct. Biomater. 2023, 14, 213. [Google Scholar] [CrossRef] [PubMed]
- Persson, C.; Engqvist, H. Premixed calcium silicate cement for endodontic applications: Injectability, setting time and radiopacity. Biomatter 2011, 1, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xu, C.; Wang, X.; Dou, Y.; Huan, Z.; Chang, J. Fast setting tricalcium silicate/magnesium phosphate premixed cement for root canal filling. Ceram. Int. 2018, 44, 3015–3023. [Google Scholar] [CrossRef]
- Wu, M.; Wang, T.; Zhang, Y. Premixed tricalcium silicate/sodium phosphate dibasic cements for root canal filling. Mater. Chem. Phys. 2021, 257, 123682. [Google Scholar] [CrossRef]
- Kastiukas, G.; Zhou, X.; Castro-Gomes, J.; Huang, S.; Saafi, M. Effects of lactic and citric acid on early-age engineering properties of Portland/calcium aluminate blended cements. Constr. Build. Mater. 2015, 101, 389–395. [Google Scholar] [CrossRef]
- Singh, N.B.; Prabha, S.; Singh, A.K. Effect of lactic acid on the hydration of portland cement. Cem. Concr. Res. 1986, 16, 545–553. [Google Scholar] [CrossRef]
- Oktay, B.; Eroglu, G.Ö.; Demir, S.; Kuruca, S.E.; Apohan, N.K. Poly(lactic acid) nanofibers containing phosphorylcholine grafts for transdermal drug delivery systems. Mater. Today Sustain. 2022, 18, 100132. [Google Scholar] [CrossRef]
- Nam, H.C.; Park, W.H. Eco-friendly poly(lactic acid) microbeads for cosmetics via melt electrospraying. Int. J. Biol. Macromol. 2020, 157, 734–742. [Google Scholar] [CrossRef]
- Chen, C.C.; Ho, C.C.; Chen, C.H.D.; Ding, S.J. Physicochemical Properties of Calcium Silicate Cements for Endodontic Treatment. J. Endod. 2009, 35, 1288–1291. [Google Scholar] [CrossRef]
- Lin, M.C.; Chen, C.C.; Wu, I.T.; Ding, S.J. Enhanced antibacterial activity of calcium silicate-based hybrid cements for bone repair. Mater. Sci. Eng. C 2020, 110, 110727. [Google Scholar] [CrossRef] [PubMed]
- El-Didamony, H.; Sharara, A.M.; Helmy, I.M.; Abd El-Aleem, S. Hydration characteristics of β-C2S in the presence of some accelerators. Cem. Concr. Res. 1996, 26, 1179–1187. [Google Scholar] [CrossRef]
- Ahmed, M.J.; Schollbach, K.; van der Laan, S.; Florea, M.; Brouwers, H.J.H. A quantitative analysis of dicalcium silicate synthesized via different sol-gel methods. Mater. Des. 2022, 213, 110329. [Google Scholar] [CrossRef]
- Meiszterics, A.; Rosta, L.; Peterlik, H.; Rohonczy, J.; Kubuki, S.; Henits, P.; Sinkó, K. Structural characterization of gel-derived calcium silicate systems. J. Phys. Chem. A 2010, 114, 10403–10411. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.K.; Ding, S.J. Acid-resistant calcium silicate-based composite implants with high-strength as load-bearing bone graft substitutes and fracture fixation devices. J. Mech. Behav. Biomed. Mater. 2016, 62, 366–383. [Google Scholar] [CrossRef] [PubMed]
- Alwan, A.F.; Khalaf, H.I. Synthesis of polylactic acid using Zn powder under microwave irradiation. IOP Conf. Ser. Mater. Sci. Eng. 2019, 571, 012085. [Google Scholar] [CrossRef]
- Devi, P.N.; Sathiyabama, J.; Rajendran, S. Study of surface morphology and inhibition efficiency of mild steel in simulated concrete pore solution by lactic acid–Zn2+ system. Int. J. Corros. Scale Inhib. 2017, 6, 18–31. [Google Scholar]
- Hidayat, S.; Ardiaksa, P.; Riveli, N.; Rahayu, I. Synthesis and characterization of carboxymethyl cellulose (CMC) from salak-fruit seeds as anode binder for lithium-ion battery. J. Phys. Conf. Ser. 2018, 1080, 012017. [Google Scholar] [CrossRef]
- Kharouf, N.; Sauro, S.; Eid, A.; Zghal, J.; Jmal, H.; Seck, A.; Macaluso, V.; Addiego, F.; Inchingolo, F.; Affolter-Zbaraszczuk, C.; et al. Physicochemical and Mechanical Properties of Premixed Calcium Silicate and Resin Sealers. J. Funct. Biomater. 2023, 14, 9. [Google Scholar] [CrossRef]
- Zamparini, F.; Spinelli, A.; Cardinali, F.; Ausiello, P.; Gandolfi, M.G.; Prati, C. The Use of Premixed Calcium Silicate Bioceramic Sealer with Warm Carrier-Based Technique: A 2-Year Study for Patients Treated in a Master Program. J. Funct. Biomater. 2023, 14, 164. [Google Scholar] [CrossRef]
- Tsunematsu, S.; Inoue, K.; Kimura, K.; Yamada, H. Improvement of acid resistance of calcium silicate hydrate by thermal treatment. Cem. Concr. Res. 2004, 34, 717–720. [Google Scholar] [CrossRef]
- Khairoun, I.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Limited compliance of some apatitic calcium phosphate bone cements with clinical requirements. J. Mater. Sci. Mater. Med. 1998, 9, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Gou, Z.; Chang, J.; Zhai, W.; Wang, J. Study on the self-setting property and the in vitro bioactivity of β-Ca2SiO4. J. Biomed. Mater. Res. 2005, 73B, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Bresciani, E.; Barata, T.J.E.; Fagundes, T.C.; Adachi, A.; Terrin, M.M.; Navarro, M.F.L. Compressive and diametral tensile strength of glass ionomer cement. J. Appl. Oral Sci. 2004, 12, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Ilie, N.; Hilton, T.J.; Heintze, S.D.; Hickel, R.; Watts, D.C.; Silikas, N.; Stansbury, J.W.; Cadenaro, M.; Ferracane, J.L. Academy of dental materials guidance—Resin composites: Part I—Mechanical properties. Dent. Mater. 2017, 33, 880–894. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.T.; Li, X.; Chen, F.Y.; Wang, Y.J.; Chen, Z.; Ding, S.J.; Zhang, L. Experimental pulp-capping agent radiopaque dicalcium silicate cement facilitates dentinogenesis. Front. Mater. 2021, 8, 738430. [Google Scholar] [CrossRef]
- Huang, Y.R.; Wu, I.T.; Chen, C.C.; Ding, S.J. In vitro comparisons of microscale and nanoscale calcium silicate particles. J. Mater. Chem. B 2020, 8, 6034–6047. [Google Scholar] [CrossRef] [PubMed]
- Jaenisch, M.; Guder, C.; Ossendorff, R.; Randau, T.; Gravius, S.; Wirtz, D.C.; Strauss, A.C.; Schildberg, F.A. In vitro biocompatibility of the novel ceramic composite baghdadite for defect augmentation in revision total hip arthroplasty. J. Funct. Biomater. 2023, 14, 517. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Jiang, L.; Wen, P.; Ye, Y.; Zhang, Y.; Ding, H.; Luo, J.; Xu, L.; Zen, K.; Zhou, Y.; et al. Tubule-derived lactate is required for fibroblast activation in acute kidney injury. Am. J. Physiol. Renal. Physiol. 2020, 318, F689–F701. [Google Scholar] [CrossRef]
- Lampe, K.J.; Namba, R.M.; Silverman, T.R.; Bjugstad, K.B.; Mahoney, M.J. Impact of lactic acid on cell proliferation and free radical-induced cell death in monolayer cultures of neural precursor cells. Biotechnol. Bioeng. 2009, 103, 1214–1223. [Google Scholar] [CrossRef]
- Nalawade, T.M.; Bhat, K.; Sogi, S.H.P. Bactericidal activity of propylene glycol, glycerine, polyethylene glycol 400, and polyethylene glycol 1000 against selected microorganisms. J. Int. Soc. Prev. Community Dent. 2015, 5, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Munir, T.; Imran, M.; Muzammil, S.; Hussain, A.A.; Alam, M.F.; Mahmood, A.; Sohail, A.; Atif, M.; Shafeeq, S.; Afzal, M. Antimicrobial activities of polyethylene glycol and citric acid coated graphene oxide-NPs synthesized via Hummer’s method. Arab. J. Chem. 2022, 15, 104075. [Google Scholar] [CrossRef]
- Sánchez-Clemente, R.; Igeño, M.I.; Población, A.G.; Guijo, M.I.; Merchán, F.; Blasco, R. Study of pH Changes in Media during Bacterial Growth of Several Environmental Strains. Proceedings 2018, 2, 1297. [Google Scholar] [CrossRef]
- Razmi, N.; Lazouskaya, M.; Pajcin, I.; Petrovic, B.; Grahovac, J.; Simic, M.; Willander, M.; Nur, O.; Stojanovic, G.M. Monitoring the effect of pH on the growth of pathogenic bacteria using electrical impedance spectroscopy. Results Eng. 2023, 20, 101425. [Google Scholar] [CrossRef]



| Sample Code | L/P | Liquid | Setting Time (min) | DTS (MPa) | pH Value | |
|---|---|---|---|---|---|---|
| PEG (vol%) | LA (vol%) | |||||
| 3L0 3L10 3L20 3L30 | 0.3 | 100 | 0 | 6.6 ± 0.9 a,b | 0.04 ± 0.00 a,b | 10.3 ± 0.2 a,b |
| 90 | 10 | 5.4 ± 0.5 a,b | 0.10 ± 0.02 b | 9.7 ± 0.3 a,b,c | ||
| 80 | 20 | 2.8 ± 0.4 b | 0.26 ± 0.04 c | 9.6 ± 0.2 a,b,c | ||
| 70 | 30 | 1.8 ± 1.1 b | 0.37 ± 0.05 d | 9.4 ± 0.3 b,c | ||
| 4L0 4L10 4L20 4L30 | 0.4 | 100 | 0 | N/A | 0.02 ± 0.00 a | 10.5 ± 0.5 a |
| 90 | 10 | 94.0 ± 5.5 c | 0.08 ± 0.02 a,b | 9.8 ± 0.3 a,b,c | ||
| 80 | 20 | 11.2 ± 3.0 a,d | 0.20 ± 0.02 c | 9.6 ± 0.1 a,b,c | ||
| 70 | 30 | 2.8 ± 0.4 b | 0.40 ± 0.03 d | 9.2 ± 0.3 c | ||
| 5L0 5L10 5L20 5L30 | 0.5 | 100 | 0 | N/A | 0.01 ± 0.00 a | 10.4 ± 0.1 a,b, |
| 90 | 10 | N/A | 0.08 ± 0.01 a,b | 9.5 ± 0.1 a,b,c | ||
| 80 | 20 | 94.0 ± 5.5 c | 0.19 ± 0.01 c | 9.4 ± 0.2 b,c | ||
| 70 | 30 | 14.4 ± 0.9 d | 0.39 ± 0.05 d | 9.1 ± 0.4 c | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-H.; Wu, I.-T.; Chen, C.-C.; Ding, S.-J. Synergistic Effect of Polyethylene Glycol and Lactic Acid on Handling Properties and Antibacterial Efficacy of Premixed Calcium Silicate Cement. J. Funct. Biomater. 2024, 15, 187. https://doi.org/10.3390/jfb15070187
Huang Y-H, Wu I-T, Chen C-C, Ding S-J. Synergistic Effect of Polyethylene Glycol and Lactic Acid on Handling Properties and Antibacterial Efficacy of Premixed Calcium Silicate Cement. Journal of Functional Biomaterials. 2024; 15(7):187. https://doi.org/10.3390/jfb15070187
Chicago/Turabian StyleHuang, Yi-Huei, I-Ting Wu, Chun-Cheng Chen, and Shinn-Jyh Ding. 2024. "Synergistic Effect of Polyethylene Glycol and Lactic Acid on Handling Properties and Antibacterial Efficacy of Premixed Calcium Silicate Cement" Journal of Functional Biomaterials 15, no. 7: 187. https://doi.org/10.3390/jfb15070187
APA StyleHuang, Y.-H., Wu, I.-T., Chen, C.-C., & Ding, S.-J. (2024). Synergistic Effect of Polyethylene Glycol and Lactic Acid on Handling Properties and Antibacterial Efficacy of Premixed Calcium Silicate Cement. Journal of Functional Biomaterials, 15(7), 187. https://doi.org/10.3390/jfb15070187







