Thermochromic Smart Windows Assisted by Photothermal Nanomaterials
Abstract
:1. Introduction
2. Thermochromic Materials for Smart Windows
3. Common Photothermal Conversion Materials
3.1. Carbon-Based Nanomaterials
3.2. Noble Metal Nanoparticles
3.3. Semiconductor Nanomaterials
3.3.1. Metal Oxide
3.3.2. Metal Sulfide
3.4. Others
4. Conclusions and Perspectives
- (1)
- Improvement of morphology (size and shape) of photothermal materials.
- (2)
- Composites of different kinds of photothermal materials.
- (3)
- Construction of bionic anti-reflection structure.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- China Building Energy Efficiency. China building energy consumption annual report 2020. Build. Energy Effic. 2021, 49, 1–6. [Google Scholar]
- Xu, F.; Cao, X.; Luo, H.; Jin, P. Recent advances in VO2-based thermochromic composites for smart windows. J. Mater. Chem. C 2018, 6, 1903–1919. [Google Scholar] [CrossRef]
- Cao, X.; Chang, T.; Shao, Z.; Xu, F.; Luo, H.; Jin, P. Challenges and opportunities toward real application of VO2-based smart glazing. Matter 2020, 2, 862–881. [Google Scholar] [CrossRef]
- Zou, X.; Ji, H.; Zhao, Y.; Lu, M.; Tao, J.; Tang, P.; Liu, B.; Yu, X.; Mao, Y. Research Progress of Photo-/Electro-Driven Thermochromic Smart Windows. Nanomaterials 2021, 11, 3335. [Google Scholar] [CrossRef]
- Tao, X.; Liu, D.; Yu, J.; Cheng, H. Reversible metal electrodeposition devices: An emerging approach to effective light modulation and thermal management. Adv. Opt. Mater. 2021, 9, 2001847. [Google Scholar] [CrossRef]
- Luo, L.; Liang, Y.; Feng, Y. Recent Progress on Preparation Strategies of Liquid Crystal Smart Windows. Crystals 2022, 12, 1426. [Google Scholar] [CrossRef]
- Zhou, Y.; Dong, X.; Mi, Y.; Fan, F.; Xu, Q.; Zhao, H.; Wang, S.; Long, Y. Hydrogel smart windows. J. Mater. Chem. A 2020, 8, 10007–10025. [Google Scholar] [CrossRef]
- Hamberg, I.; Granqvist, C. Evaporated Sn-doped In2O3 films: Basic optical properties and applications to energy-efficient windows. J. Appl. Phys. 1986, 60, 123–160. [Google Scholar] [CrossRef]
- Jing, T. Selective Reflection of Cholesteric Liquid Crystals. Sci. Insights 2022, 40, 515–517. [Google Scholar] [CrossRef]
- Ming, Y.; Sun, Y.; Liu, X. Optical evaluation of a Smart Transparent Insulation Material for window application. Energy Convers. Manag. X 2022, 100315. [Google Scholar] [CrossRef]
- Huang, Z.; Chen, S.; Lv, C.; Huang, Y.; Lai, J. Infrared characteristics of VO2 thin films for smart window and laser protection applications. Appl. Phys. Lett. 2012, 101, 191905. [Google Scholar] [CrossRef]
- Rui, S.; Lin, Y.; Junhui, H.; Jie, L. Thermochromic smart coatings. Prog. Chem. 2019, 31, 1712. [Google Scholar]
- Cui, Y.; Ke, Y.; Liu, C. Thermochromic VO2 for energy-efficient smart windows. Joule 2018, 2, 1707–1746. [Google Scholar] [CrossRef] [Green Version]
- Barimah, E.; Boontan, A.; Steenson, D. Infrared optical properties modulation of VO2 thin film fabricated by ultrafast pulsed laser deposition for thermochromic smart window applications. Sci. Rep. 2022, 12, 1–10. [Google Scholar] [CrossRef]
- Ren, H.; Hassna, O.; Li, J.; Arigong, B. A patterned phase-changing vanadium dioxide film stacking with VO2 nanoparticle matrix for high performance energy-efficient smart window applications. Appl. Phys. Lett. 2021, 118, 051901. [Google Scholar] [CrossRef]
- Riapanitra, A.; Asakura, Y.; Yin, S. One-step hydrothermal synthesis and thermochromic properties of chlorine-doped VO2(M) for smart window application. Funct. Mater. Lett. 2020, 13, 1951008. [Google Scholar] [CrossRef]
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C.; Yang, L.; Ren, D. Infrared thermochromic properties of monoclinic VO2 nanopowders using a malic acid-assisted hydrothermal method for adaptive camouflage. RSC Adv. 2017, 7, 5189–5194. [Google Scholar] [CrossRef] [Green Version]
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C. Inkjet printing of vanadium dioxide nanoparticles for smart windows. J. Mater. Chem. C 2018, 6, 2424–2429. [Google Scholar] [CrossRef]
- Ke, Y.; Zhou, C.; Zhou, Y.; Wang, S.; Chan, S.H.; Long, Y. Emerging thermal-responsive materials and integrated techniques targeting the energy-efficient smart window application. Adv. Funct. Mater. 2018, 28, 1800113. [Google Scholar] [CrossRef]
- Markov, P.; Marvel, R.E.; Conley, H.J.; Miller, K.J.; Haglund, R.F., Jr.; Weiss, S.M. Optically monitored electrical switching in VO2. ACS Photonics 2015, 2, 1175–1182. [Google Scholar] [CrossRef]
- Ji, H.; Liu, D.; Cheng, H.; Tao, Y. Large area infrared thermochromic VO2 nanoparticle films prepared by inkjet printing technology. Sol. Energy Mater. Sol. Cells 2019, 194, 235–243. [Google Scholar] [CrossRef]
- Ji, H.; Liu, D.; Cheng, H. Infrared optical modulation characteristics of W-doped VO2(M) nanoparticles in the MWIR and LWIR regions. Mater. Sci. Semicond. Process. 2020, 119, 105141. [Google Scholar] [CrossRef]
- Ji, H.; Liu, D.; Cheng, H.; Zhang, C.; Yang, L. Vanadium dioxide nanopowders with tunable emissivity for adaptive infrared camouflage in both thermal atmospheric windows. Sol. Energy Mater. Sol. Cells 2018, 175, 96–101. [Google Scholar] [CrossRef]
- Ji, H.; Liu, D.; Zhang, C.; Cheng, H. VO2/ZnS core-shell nanoparticle for the adaptive infrared camouflage application with modified color and enhanced oxidation resistance. Sol. Energy Mater. Sol. Cells 2018, 176, 1–8. [Google Scholar] [CrossRef]
- Meng, W.; Gao, Y.; Hu, X.; Tan, L.; Li, L.; Zhou, G.; Jiang, L. Photothermal Dual Passively Driven Liquid Crystal Smart Window. ACS Appl. Mater. Interfaces 2022, 14, 28301–28309. [Google Scholar] [CrossRef] [PubMed]
- Meng, C.; Chen, E.; Wang, L.; Tang, S.; Tseng, M.; Guo, J.; Kwok, H. Color-switchable liquid crystal smart window with multi-layered light guiding structures. Opt. Express 2019, 27, 13098–13107. [Google Scholar] [CrossRef]
- Kragt, A.; Loonen, R.; Broer, D.; Debije, M.; Schenning, A. ‘Smart’ light-reflective windows based on temperature responsive twisted nematic liquid crystal polymers. J. Polym. Sci. 2021, 59, 1278–1284. [Google Scholar] [CrossRef]
- Oh, S.; Nam, S.; Kim, S.; Yoon, T.; Kim, W. Self-regulation of infrared using a liquid crystal mixture doped with push–pull azobenzene for energy-saving smart windows. ACS Appl. Mater. Interfaces 2021, 13, 5028–5033. [Google Scholar] [CrossRef]
- Li, K.; Meng, S.; Xia, S.; Ren, X.; Gao, G. Durable and controllable smart windows based on thermochromic hydrogels. ACS Appl. Mater. Interfaces 2020, 12, 42193–42201. [Google Scholar] [CrossRef]
- Xiao, X.; Shi, D.; Yang, Z.; Yu, Q.; Kaneko, D.; Chen, M. Near infrared-sensitive smart windows from Au nanorod–polymer hybrid photonic hydrogels. New J. Chem. 2021, 45, 4016–4023. [Google Scholar] [CrossRef]
- Zhang, L.; Xia, H.; Xia, F.; Du, Y.; Wu, Y.; Gao, Y. Energy-saving smart windows with HPC/PAA hybrid hydrogels as thermochromic materials. ACS Appl. Energy Mater. 2021, 4, 9783–9791. [Google Scholar] [CrossRef]
- Tian, J.; Peng, H.; Du, X.; Wang, H.; Cheng, X.; Du, Z. Hybrid thermochromic microgels based on UCNPs/PNIPAm hydrogel for smart window with enhanced solar modulation. J. Alloy. Compd. 2021, 858, 157725. [Google Scholar] [CrossRef]
- Pelton, R. Poly (N-isopropylacrylamide)(PNIPAM) is never hydrophobic. J. Colloid Interface Sci. 2010, 348, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Sherry, L.; Jin, R.; Mirkin, C.; Schatz, G.; Van Duyne, R. Localized surface plasmon resonance spectroscopy of single silver triangular nanoprisms. Nano Lett. 2006, 6, 2060–2065. [Google Scholar] [CrossRef]
- Agrawal, A.; Cho, S.; Zandi, O.; Ghosh, S.; Johns, R.; Milliron, D. Localized surface plasmon resonance in semiconductor nanocrystals. Chem. Rev. 2018, 118, 3121–3207. [Google Scholar] [CrossRef]
- Kang, Z.; Lee, S. Carbon dots: Advances in nanocarbon applications. Nanoscale 2019, 11, 19214–19224. [Google Scholar] [CrossRef]
- Mintz, K.; Bartoli, M.; Rovere, M.; Zhou, Y.; Hettiarachchi, S.; Paudyal, S.; Leblanc, R. A deep investigation into the structure of carbon dots. Carbon 2021, 173, 433–447. [Google Scholar] [CrossRef]
- Dideikin, A.; Vul’, A. Graphene oxide and derivatives: The place in graphene family. Front. Phys. 2019, 6, 149. [Google Scholar] [CrossRef]
- Wang, Y.; Li, S.; Yang, H.; Luo, J. Progress in the functional modification of graphene/graphene oxide: A review. RSC Adv. 2020, 10, 15328–15345. [Google Scholar]
- Farjadian, F.; Abbaspour, S.; Sadatlu, M.; Mirkiani, S.; Ghasemi, A.; Hoseini-Ghahfarokhi, M.; Hamblin, M. Recent developments in graphene and graphene oxide: Properties, synthesis, and modifications: A review. ChemistrySelect 2020, 5, 10200–10219. [Google Scholar]
- Kim, D.; Lee, E.; Lee, H.S.; Yoon, J. Energy efficient glazing for adaptive solar control fabricated with photothermotropic hydrogels containing graphene oxide. Sci. Rep. 2015, 5, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.; Lu, Y.; Peng, J.; Chen, J.; Yu, S. Photothermally sensitive poly (N-isopropylacrylamide)/graphene oxide nanocomposite hydrogels as remote light-controlled liquid microvalves. Adv. Funct. Mater. 2012, 22, 4017–4022. [Google Scholar] [CrossRef]
- Lee, E.; Kim, D.; Yoon, J. Stepwise activation of switchable glazing by compositional gradient of copolymers. ACS Appl. Mater. Interfaces 2016, 8, 26359–26364. [Google Scholar] [CrossRef] [PubMed]
- NIWAER, A.E.A.; Wang, H.; Meng, X.; He, X.; Yang, S.; Yu, H.; Zuo, F. Preparation and preliminary application of PNIPAM/laponite/graphene oxide near-infrared light responsive hydrogels. Fine Chem. 2021, 3, 6. [Google Scholar]
- Zhou, T.; Zhou, Q.; Hua, Y.; Huang, C.; Li, L.; LÜ, W. Preparation and properties of tungsten-doped vanadium dioxide/graphene composites. Fine Chem. 2020, 37, 1574–1579. [Google Scholar]
- Chang, Q.; Shen, Z.; Guo, Z.; Xue, C.; Li, N.; Yang, J.; Hu, S. Hydroxypropylmethyl cellulose modified with carbon dots exhibits light-responsive and reversible optical switching. ACS Appl. Mater. Interfaces 2021, 13, 12375–12382. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Lu, C.; Jiang, Y.; Tang, H.; Yang, X. Ag/ZnO/graphene oxide heterostructure for the removal of rhodamine B by the synergistic adsorption–degradation effects. Ceram. Int. 2015, 41, 4231–4237. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, X.; Zhao, K.; Dong, Y.; Yang, W.; Liu, J.; Li, D. Assembly of gold nanorods with L-cysteine reduced graphene oxide for highly efficient NIR-triggered photothermal therapy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 266, 120458. [Google Scholar] [CrossRef]
- Kim, D.; Lee, H.S.; Yoon, J. Remote control of volume phase transition of hydrogels containing graphene oxide by visible light irradiation. RSC Adv. 2014, 4, 25379–25383. [Google Scholar] [CrossRef]
- Chen, H.; Shao, L.; Ming, T.; Sun, Z.; Zhao, C.; Yang, B.; Wang, J. Understanding the photothermal conversion efficiency of gold nanocrystals. Small 2010, 6, 2272–2280. [Google Scholar] [CrossRef]
- Kim, H.; Lee, D. Near-infrared-responsive cancer photothermal and photodynamic therapy using gold nanoparticles. Polymers 2018, 10, 961. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; He, Y.; Huang, J.; Zhu, J. Synthesis and solar photo-thermal conversion of Au, Ag, and Au-Ag blended plasmonic nanoparticles. Energy Convers. Manag. 2016, 127, 293–300. [Google Scholar] [CrossRef]
- Xiao, J.; Fan, S.; Wang, F.; Sun, L.; Zheng, X.; Yan, C. Porous Pd nanoparticles with high photothermal conversion efficiency for efficient ablation of cancer cells. Nanoscale 2014, 6, 4345–4351. [Google Scholar] [CrossRef]
- Tang, S.; Chen, M.; Zheng, N. Multifunctional ultrasmall Pd nanosheets for enhanced near-infrared photothermal therapy and chemotherapy of cancer. Nano Res. 2015, 8, 165–174. [Google Scholar] [CrossRef]
- Jain, P.K.; Huang, X.; El-Sayed, I.H.; El-Sayed, M.A. Noble metals on the nanoscale: Optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem. Res. 2008, 41, 1578–1586. [Google Scholar] [CrossRef]
- Cao, D.; Xu, C.; Lu, W.; Qin, C.; Cheng, S. Sunlight-Driven Photo-Thermochromic Smart Windows. Sol. RRL 2018, 2, 1700219. [Google Scholar] [CrossRef]
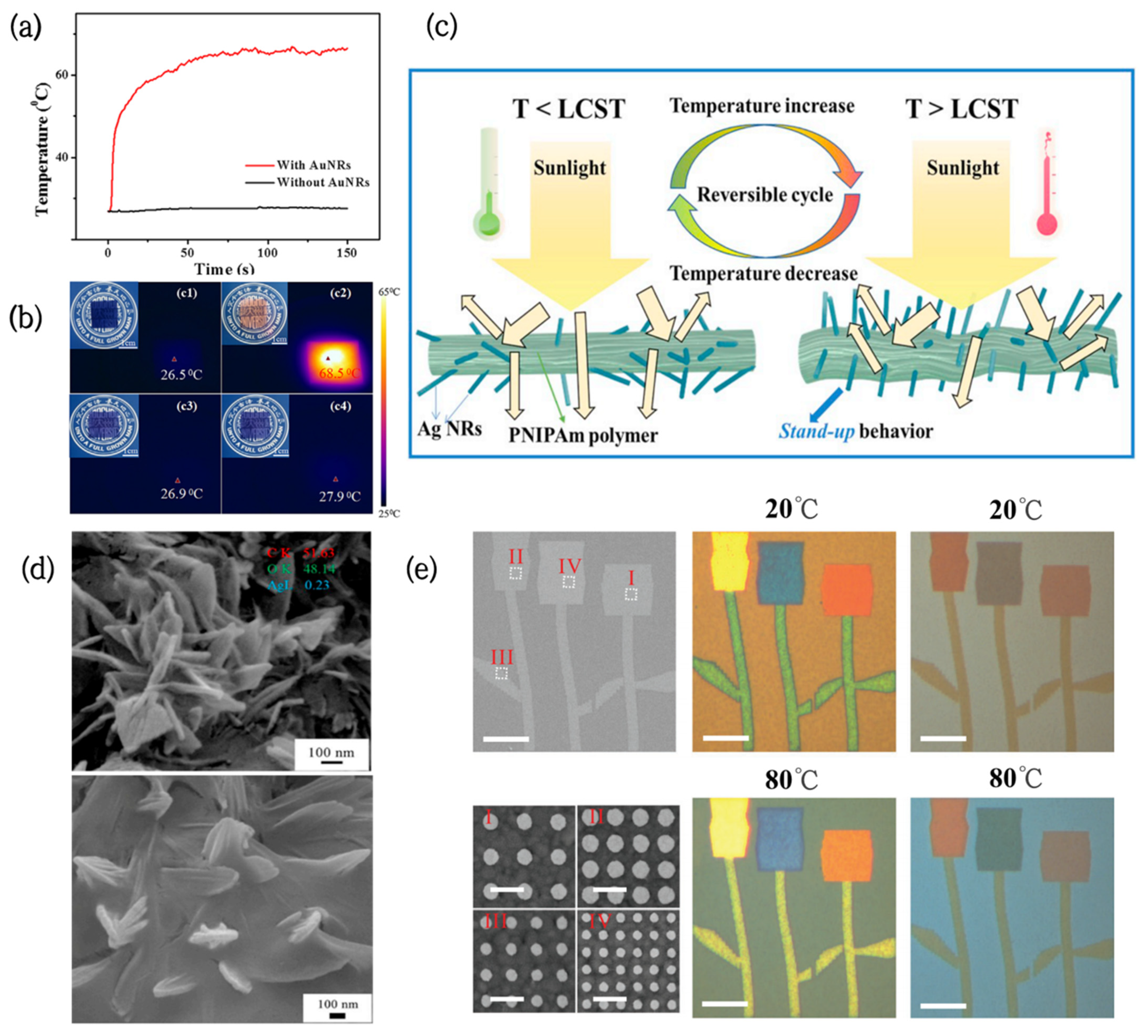
- Wei, G.; Yang, D.; Zhang, T.; Yue, X.; Qiu, F. Thermal-responsive PNIPAm-acrylic/Ag NRs hybrid hydrogel with atmospheric window full-wavelength thermal management for smart windows. Sol. Energy Mater. Sol. Cells 2020, 206, 110336. [Google Scholar] [CrossRef]
- Shu, F.Z.; Yu, F.F.; Peng, R.W.; Zhu, Y.Y.; Xiong, B.; Fan, R.H.; Wang, Z.H.; Liu, Y.; Wang, M. Dynamic plasmonic color generation based on phase transition of vanadium dioxide. Adv. Opt. Mater. 2018, 6, 1700939. [Google Scholar] [CrossRef]
- Zhou, J.; Dong, X.; Zha, L. Research Progress of Gold Nanorods Based Intelligent Composite Nanogels. Polym. Bull. 2015, 6, 1–9. [Google Scholar]
- Liang, J.; Song, X.; Li, P.; Zhou, L.; Guo, J. Localized surface plasmon resonance tunability of VO2/Au/VO2 composite nanotriangle sandwich array. Mater. Res. Express 2019, 6, 0850f0856. [Google Scholar] [CrossRef]
- Dong, X.; Liu, X.; Zhou, J.; Zhang, L.; Lin, D.; Zha, L. Fabrication and characterization of thermo-responsive composite microgels based on Au@Ag nanorod as core. Chem. Res. Appl. 2015, 27, 836–841. [Google Scholar]
- Li, M.; Liu, D.; Cheng, H.; Peng, L.; Zu, M. Manipulating metals for adaptive thermal camouflage. Sci. Adv. 2020, 6, eaba3494. [Google Scholar] [CrossRef] [PubMed]
- Haruta, M.; Daté, M. Advances in the catalysis of Au nanoparticles. Appl. Catal. A Gen. 2001, 222, 427–437. [Google Scholar] [CrossRef]
- Irshad, M.; Arshad, N.; Wang, X. Nanoenabled photothermal materials for clean water production. Glob. Chall. 2021, 5, 2000055. [Google Scholar] [CrossRef]
- Chang, M.; Hou, Z.; Wang, M.; Yang, C.; Wang, R.; Li, F.; Jun, L. Single-Atom Pd Nanozyme for Ferroptosis-Boosted Mild-Temperature Photothermal Therapy. Angew. Chem. 2021, 133, 202101924. [Google Scholar] [CrossRef]
- Blemker, M.; Gibbs, S.; Raulerson, E.; Milliron, D.; Roberts, S. Modulation of the visible absorption and reflection profiles of ITO nanocrystal thin films by plasmon excitation. ACS Photonics 2020, 7, 1188–1196. [Google Scholar] [CrossRef]
- Wei, W.; Hong, R.; Jing, M.; Shao, W.; Tao, C.; Zhang, D. Thickness-dependent surface plasmon resonance of ITO nanoparticles for ITO/In-Sn bilayer structure. Nanotechnology 2017, 29, 015705. [Google Scholar] [CrossRef]
- Kalanur, S.; Seo, H. Synthesis of CuxS thin films with tunable localized surface plasmon resonances. ChemistrySelect 2018, 3, 5920–5926. [Google Scholar] [CrossRef]
- Song, G.; Han, L.; Zou, W.; Xiao, Z.; Huang, X.; Qin, Z.; Hu, J. A novel photothermal nanocrystals of Cu7S4 hollow structure for efficient ablation of cancer cells. Nano-Micro Lett. 2014, 6, 169–177. [Google Scholar] [CrossRef]
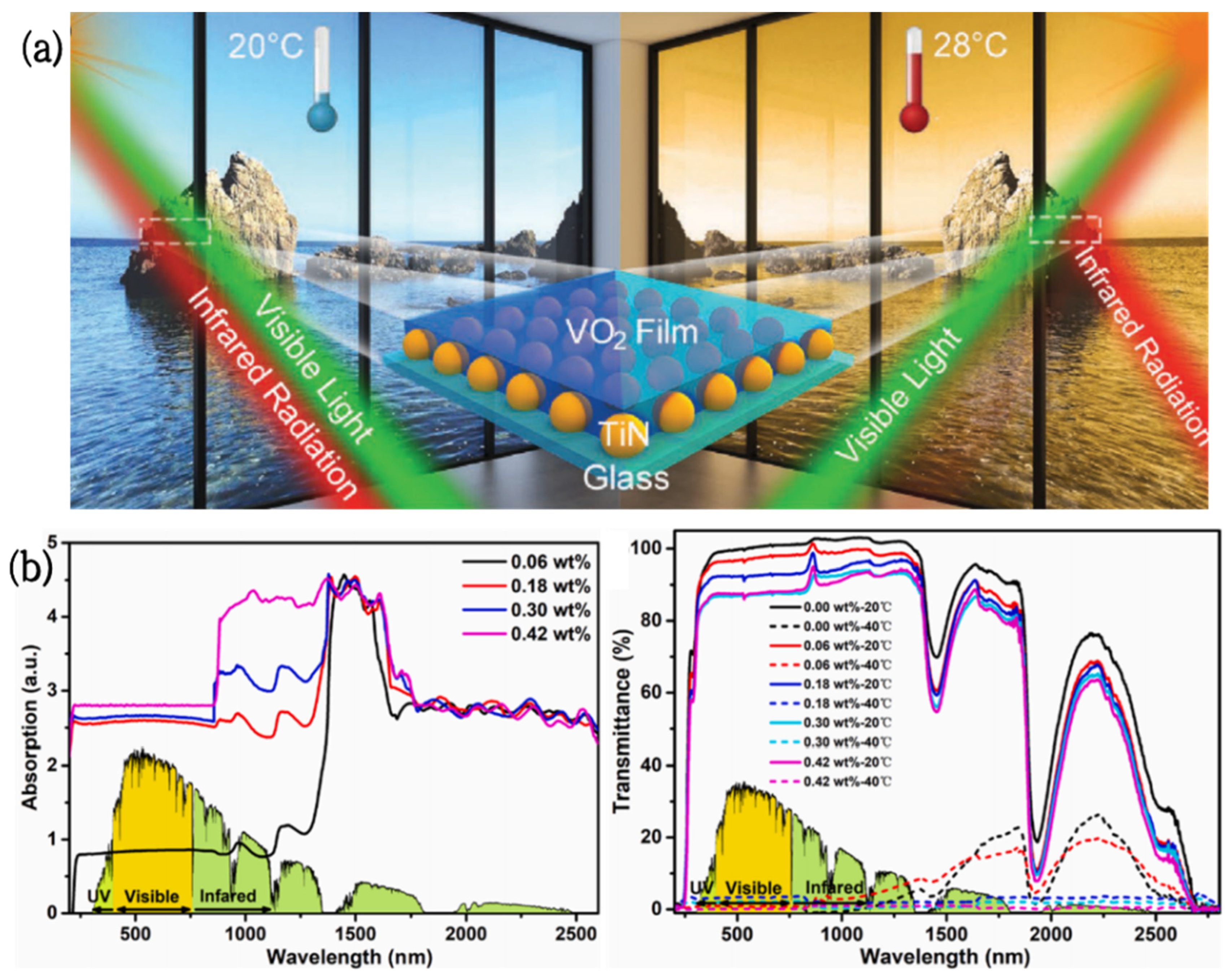
- Zhang, J.; Chen, T.; Li, X.; Liu, Y.; Liu, Y.; Yang, H. Investigation of localized surface plasmon resonance of TiN nanoparticles in TiNxOy thin films. Opt. Mater. Express 2016, 6, 2422–2433. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, G.; Wang, M.; Yu, W.; Zeng, J.; Yu, X.; Li, Q. Dual plasmonic Au/TiN nanofluids for efficient solar photothermal conversion. Sol. Energy 2019, 184, 240–248. [Google Scholar] [CrossRef]
- Lee, H.Y.; Cai, Y.; Bi, S.; Liang, Y.N.; Song, Y.; Hu, X.M. A Dual-Responsive Nanocomposite toward Climate-Adaptable Solar Modulation for Energy-Saving Smart Windows. Acs Appl. Mater. Interfaces 2017, 9, 6054. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, S.; Hu, X.; Jiang, J.; Wang, L. Sunlight-Induced Photo-Thermochromic Supramolecular Nanocomposite Hydrogel Film for Energylogaving Smart Window. Sol. RRL 2018, 2, 1800204. [Google Scholar] [CrossRef]
- Wu, M.; Shi, Y.; Li, R.; Wang, P. Spectrally selective smart window with high near-infrared light shielding and controllable visible light transmittance. ACS Appl. Mater. Interfaces 2018, 10, 39819–39827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Zhang, R.; Xu, L.; Li, J.; Yang, L.; Yang, Y.; Bolshakov, A.; Zhu, J. Visible and infrared optical modulation of PSLC smart films doped with ATO nanoparticles. Dalton Trans. 2021, 50, 10033–10040. [Google Scholar] [CrossRef]
- Liang, X.; Guo, S.; Chen, M.; Li, C.; Wang, Q.; Zou, C.; Zhang, C.; Zhang, L.; Guo, S.; Yang, H. A temperature and electric field-responsive flexible smart film with full broadband optical modulation. Mater. Horiz. 2017, 4, 878–884. [Google Scholar] [CrossRef]
- Liang, X.; Guo, C.; Chen, M.; Guo, S.; Zhang, L.; Li, F.; Guo, S.; Yang, H. A roll-to-roll process for multi-responsive soft-matter composite films containing CsxWO3 nanorods for energy-efficient smart window applications. Nanoscale Horiz. 2017, 2, 319–325. [Google Scholar] [CrossRef]
- Ding, B.; Shi, M.; Chen, F.; Zhou, R.; Deng, M.; Wang, M.; Chen, H. Shape-controlled syntheses of PbS submicro-/nano-crystals via hydrothermal method. J. Cryst. Growth 2009, 311, 1533–1538. [Google Scholar] [CrossRef]
- Chou, S.; Kaehr, B.; Kim, J.; Foley, B.; De, M.; Hopkins, P.; Dravid, V. Chemically exfoliated MoS2 as near-infrared photothermal agents. Angew. Chem. Int. Ed. 2013, 52, 4160–4164. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; He, Y.; Hu, Y.; Jin, G.; Jiang, B.; Huang, Y. Photothermal-conversion-enhanced photocatalytic activity of flower-like CuS superparticles under solar light irradiation. Sol. Energy 2018, 170, 586–593. [Google Scholar] [CrossRef]
- Xi, M.; Xu, L.; Li, N.; Zhang, S.; Wang, Z. Plasmonic Cu27S24 nanocages for novel solar photothermal nanoink and nanofilm. Nano Res. 2022, 15, 3161–3169. [Google Scholar] [CrossRef]
- Ji, Q. Design of Nanomaterials System for Smart Thermal Controlling. Master’s Thesis, Zhengzhou University, Zhengzhou, China, 2014. [Google Scholar]
- Zhang, J.; Du, P.; Xu, D.; Li, Y.; Peng, W.; Zhang, G.; Zhang, F.; Fan, X. Near-infrared responsive MoS2/poly (N-isopropylacrylamide) hydrogels for remote light-controlled microvalves. Ind. Eng. Chem. Res. 2016, 55, 4526–4531. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, L. Smart window based on Cu7S4/hydrogel composites with fast photothermal response. Sol. Energy Mater. Sol. Cells 2019, 202, 110109. [Google Scholar] [CrossRef]
- Zhang, C.; Yan, C.; Xue, Z.; Yu, W.; Wang, T. Shape-Controlled Synthesis of High-Quality Cu7S4 Nanocrystals for Efficient Light-Induced Water Evaporation. Small 2016, 12, 5320–5328. [Google Scholar] [CrossRef]
- Patsalas, P.; Kalfagiannis, N.; Kassavetis, S. Optical properties and plasmonic performance of titanium nitride. Materials 2015, 8, 3128–3154. [Google Scholar] [CrossRef] [Green Version]
- Gschwend, P.; Conti, S.; Kaech, A.; Maake, C.; Pratsinis, S. Silica-coated TiN particles for killing cancer cells. ACS Appl. Mater. Interfaces 2019, 11, 22550–22560. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Fu, Q.; Wei, H.; Yao, A. TiN nanoparticles: Synthesis and application as near-infrared photothermal agents for cancer therapy. J. Mater. Sci. 2019, 54, 5743–5756. [Google Scholar] [CrossRef]
- Hao, Q.; Li, W.; Xu, H.; Wang, J.; Yin, Y.; Wang, H.; Ma, L.; Ma, F.; Jiang, X.; Schmidt, O. VO2/TiN plasmonic thermochromic smart coatings for room-temperature applications. Adv. Mater. 2018, 30, 1705421. [Google Scholar] [CrossRef]
- Yang, L.; Tong, R.; Wang, Z.; Xia, H. Polydopamine Particle-Filled Shape-Memory Polyurethane Composites with Fast Near-Infrared Light Responsibility. ChemPhysChem 2018, 19, 2052–2057. [Google Scholar] [CrossRef]
- Song, M.; Wang, Y.; Liang, X.; Zhang, X.; Zhang, S.; Li, B. Functional materials with self-healing properties: A review. Soft Matter. 2019, 15, 6615–6625. [Google Scholar] [CrossRef]
- Tian, J.; Gu, J.; Peng, H.; Wang, H.; Du, Z.; Cheng, X.; Du, X. Sunlight-driven photo-thermochromic hybrid hydrogel with fast responsiveness and durability for energy efficient smart windows. Compos. Part A Appl. Sci. Manuf. 2021, 149, 106538. [Google Scholar] [CrossRef]
- Deng, C. Graphene oxide Aggregate Modification and Photothermal Complex Properties of Phase Change Materials. Master’s Thesis, East China University of Technology, Fuzhou, China, 2021. [Google Scholar]
- Lebepe, T.; Oluwafemi, O. Thermal and Medium Stability Study of Polyvidone-Modified Graphene Oxide-Coated Gold Nanorods with High Photothermal Efficiency. Nanomaterials 2022, 12, 3382. [Google Scholar] [CrossRef]
- Shan, G.; Xu, S.; Shi, W. Measuring the thermal conversion efficiency of gold nanoparticle solution. Phys. Exp. 2018, 38, 10–15. [Google Scholar]
- Balitskii, O.; Mashkov, O.; Barabash, A. Ligand Tuning of Localized Surface Plasmon Resonances in Antimony-Doped Tin Oxide Nanocrystals. Nanomaterials 2022, 12, 3469. [Google Scholar] [CrossRef]
- Huang, L.; Tang, H.; Bai, Y. Preparation of monodispersed Cs0.33WO3 nanocrystals by mist chemical vapor deposition for near-infrared shielding application. Nanomaterials 2020, 10, 2295. [Google Scholar] [CrossRef]
- Tian, Q.; Jiang, F.; Zou, R.; Liu, Q.; Hu, J. Hydrophilic Cu9S5 nanocrystals: A photothermal agent with a 25.7% heat conversion efficiency for photothermal ablation of cancer cells in vivo. Acs Nano 2011, 5, 9761. [Google Scholar] [CrossRef]
- Cui, J.; Jiang, R.; Xu, S.; Hu, G.; Wang, L. Cu7S4 nanosuperlattices with greatly enhanced photothermal efficiency. Small 2015, 11, 4183–4190. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, G.; He, F.; Chen, L.; Huang, Y. Au@Cu7S4 yolk–shell nanoparticles as a 980 nm laser-driven photothermal agent with a heat conversion efficiency of 63%. RSC Adv. 2015, 5, 87903–87907. [Google Scholar] [CrossRef]
- Kaur, M.; Ishii, S.; Shinde, S.L.; Nagao, T. All-ceramic microfibrous solar steam generator: TiN plasmonic nanoparticle-loaded transparent microfibers. ACS Sustain. Chem. Eng. 2017, 5, 8523–8528. [Google Scholar] [CrossRef]
- Li, Y.; Wang, D. Application of polydopamine in tumor nano-drug delivery. Chin. J. Pharm. 2021, 19, 81–86. [Google Scholar]
- Boden, S.; Bagnall, D. Optimization of moth-eye antireflection schemes for silicon solar cells. Prog. Photovolt. Res. Appl. 2010, 18, 195–203. [Google Scholar] [CrossRef]



| Type | Power of Excitation Light Source | Efficiency | Reference |
|---|---|---|---|
| GO NSs | Simulated sunlight (0.1 W/cm2) | 63.2% | [93] |
| CDs | AM1.5 Standard sunlight (0.1 W/cm2) | 23.46% | [46] |
| Au NRs | 808 nm laser (1.27 W/cm2) | 39.2% | [94] |
| Au STs | 808 nm laser (2 W/cm2) | 42% | [95] |
| ATO Hollow ball | 1064 nm laser (-) | 30.69% | [96] |
| Cs0.33WO3 | 980 nm laser (1.66 W/cm2) | 23.1% | [97] |
| Cu9S5 NCs | 980 nm laser (0.51 W/cm2) | 25.7% | [98] |
| Cu7S4 NPs | 808 nm NIR laser (1 W/cm2) | 56.32% | [99] |
| Cu7S4 Nano Superlattice | 808 nm NIR laser (1 W/cm2) | 65.7% | [99] |
| Au@Cu7S4 Yolk–shell NPs | 980 nm laser (0.51 W/cm2) | 63% | [100] |
| TiN | 808 nm NIR laser (1.5 W/cm2) | 38.6% | [101] |
| PDAPs | 808 nm NIR laser (2 W/cm2) | 40% | [102] |
| Thermochromic Material | Photothermal Material | Tlum,l | Tlum,h | ΔTsol | TC | Reference |
|---|---|---|---|---|---|---|
| HPMC | - | 98.94% | 89.32% | 6.52% | 40°C | [46] |
| PNIPAM | - | 73.2% | 48.58% | 26.88% | 31.4°C | [58] |
| VO2 | - | 47% | 52% | ΔTNIR = 51% | 68°C | [86] |
| PNIPAM | GO | 97.8% | 3.0% | - | 32.5°C | [41] |
| HPMC | CDs | 82.11% | 3.50% | 65.55% | 45°C | [46] |
| HPMC | Au NCs | 85.4% | 3.5% | 62.6% | 40°C | [56] |
| VO2 | Au | 74.1% | 66.6% | 13.2% | 50°C | [60] |
| PNIPAM | Ag NRs | 61.36% | 3.43% | 59.24% | 32.9°C | [58] |
| PNIPAM (10%Sb) | ATO NPs | 83.0% | 55.3% | 35.7% | 33.6°C | [72] |
| PNIPAM | ATO NPs | 77.2% | 18.6% | 56.1% | 32.1°C | [73] |
| PSLC | ATO NPs | 78.5% | 10% | - | - | [74] |
| PDLC-PSLC | ITO NCs | 78% | 1.5% | - | 28.3°C | [75] |
| PAM-PNIPAM | CsxWO3 | 78.2% | 45.3% | - | 30°C | [76] |
| PT-LCs | CsxWO3 NRs | 67% | 1.5% | - | 30.5°C | [77] |
| PNIPAM | Cu7S4 NPs | 79% | 41.1% | 31.2% | 32°C | [84] |
| VO2 | TiN NPs | 49% | 51% | ΔTNIR = 48% | 28°C | [85] |
| PNIPAM | PDAPs | 92.58% | 3.41% | 88.9% | 33°C | [92] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Ji, H.; Lu, M.; Tao, J.; Ou, Y.; Wang, Y.; Chen, Y.; Huang, Y.; Wang, J.; Mao, Y. Thermochromic Smart Windows Assisted by Photothermal Nanomaterials. Nanomaterials 2022, 12, 3865. https://doi.org/10.3390/nano12213865
Zhao Y, Ji H, Lu M, Tao J, Ou Y, Wang Y, Chen Y, Huang Y, Wang J, Mao Y. Thermochromic Smart Windows Assisted by Photothermal Nanomaterials. Nanomaterials. 2022; 12(21):3865. https://doi.org/10.3390/nano12213865
Chicago/Turabian StyleZhao, Yong, Haining Ji, Mingying Lu, Jundong Tao, Yangyong Ou, Yi Wang, Yongxing Chen, Yan Huang, Junlong Wang, and Yuliang Mao. 2022. "Thermochromic Smart Windows Assisted by Photothermal Nanomaterials" Nanomaterials 12, no. 21: 3865. https://doi.org/10.3390/nano12213865
APA StyleZhao, Y., Ji, H., Lu, M., Tao, J., Ou, Y., Wang, Y., Chen, Y., Huang, Y., Wang, J., & Mao, Y. (2022). Thermochromic Smart Windows Assisted by Photothermal Nanomaterials. Nanomaterials, 12(21), 3865. https://doi.org/10.3390/nano12213865






