A Microfluidic Platform with an Embedded Miniaturized Electrochemical Sensor for On-Chip Plasma Extraction Followed by In Situ High-Sensitivity C-Reactive Protein (hs-CRP) Detection
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagent and Sample Preparation
2.2. Microchannel Fabrication and Assembly
2.3. Instrument Setup
3. Results and Discussion
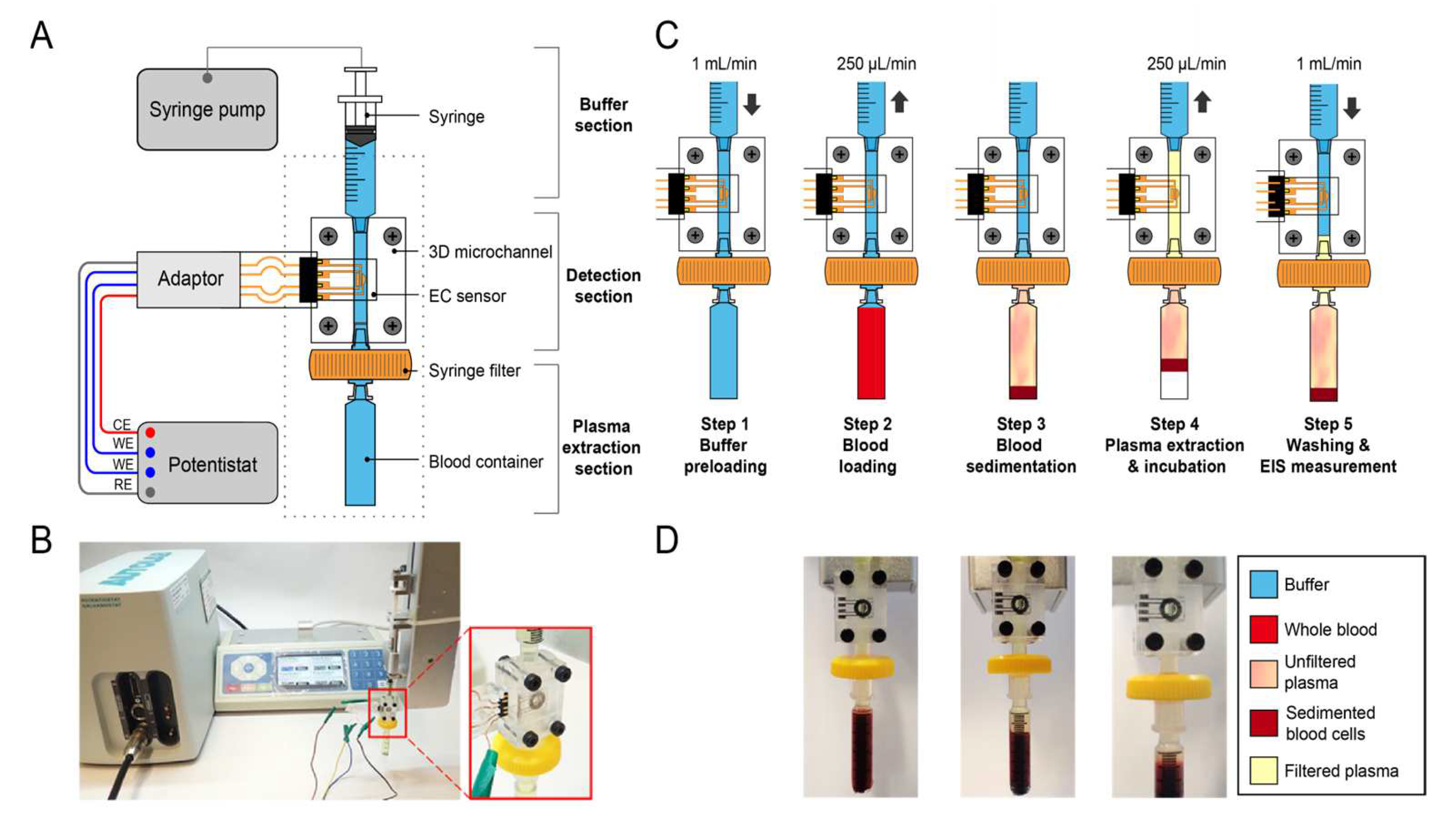
3.1. Operation Protocol of the Microfluidic Platform
3.2. Plasma Extraction Efficiency
3.3. The Electrochemical (EC) Sensor Functionalization Protocol
3.4. Specificity Test
3.5. CRP Detection
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Sohn, E. Diagnosis: Frontiers in blood testing. Nature 2017, 549, S16–S18. [Google Scholar] [CrossRef] [PubMed]
- Dixon, L.R. The Complete Blood Count: Physiologic Basis and Clinical Usage. J. Périnat. Neonatal Nurs. 1997, 11, 1–18. [Google Scholar] [CrossRef] [PubMed]
- George-Gay, B.; Parker, K. Understanding the complete blood count with differential. J. PeriAnesthesia Nurs. 2003, 18, 96–117. [Google Scholar] [CrossRef] [PubMed]
- Fiehn, O.; Kind, T. Metabolite Profiling in Blood Plasma. In Metabolomics; Humana Press: Totowa, NJ, USA, 2007; pp. 3–17. [Google Scholar] [CrossRef]
- Klouche, M.; Schröder, U. Rapid methods for diagnosis of bloodstream infections. Clin. Chem. Lab. Med. 2008, 46, 888–908. [Google Scholar] [CrossRef] [PubMed]
- Fenollar, F.; Raoult, D. Molecular diagnosis of bloodstream infections caused by non-cultivable bacteria. Int. J. Antimicrob. Agents 2007, 30, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Finn, C.; Parrett, C.J.; Dhume, K.; Hwang, J.H.; Sidhom, D.; Strutt, T.M.; Sip, Y.Y.L.; McKinstry, K.K.; Huo, Q. A Rapid Blood Test to Determine the Active Status and Duration of Acute Viral Infection. ACS Infect. Dis. 2017, 3, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Mach, A.J.; Adeyiga, O.B.; Di Carlo, D. Microfluidic sample preparation for diagnostic cytopathology. Lab Chip 2012, 13, 1011–1026. [Google Scholar] [CrossRef] [Green Version]
- Tripathi, S.; Kumar, Y.V.B.V.; Prabhakar, A.; Joshi, S.S.; Agrawal, A. Passive blood plasma separation at the microscale: A review of design principles and microdevices. J. Micromechanics Microeng. 2015, 25, 083001. [Google Scholar] [CrossRef]
- Mielczarek, W.S.; Obaje, E.A.; Bachmann, T.T.; Kersaudy-Kerhoas, M. Microfluidic blood plasma separation for medical diagnostics: Is it worth it? Lab Chip 2016, 16, 3441–3448. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kang, T.G.; Chen, Y.; Kim, S. Plasma separation from blood: The ‘lab-on-a-chip’ approach. Crit. Rev. Biomed. Eng. 2009, 37, 517–529. [Google Scholar] [CrossRef]
- Kersaudy-Kerhoas, M.; Sollier, E. Micro-scale blood plasma separation: From acoustophoresis to egg-beaters. Lab Chip 2013, 13, 3323–3346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.; Liao, S.-C.; Song, J.; Mauk, M.G.; Li, X.; Wu, G.; Ge, D.; Greenberg, R.M.; Yang, S.; Bau, H.H. A high-efficiency superhydrophobic plasma separator. Lab Chip 2015, 16, 553–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, S.; Lin, H.K.; Lu, B.; Williams, A.; Datar, R.; Cote, R.J.; Tai, Y.-C. 3D microfilter device for viable circulating tumor cell (CTC) enrichment from blood. Biomed. Microdevices 2010, 13, 203–213. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Mauk, M.; Gross, R.; Bushman, F.D.; Edelstein, P.H.; Collman, R.G.; Bau, H.H. Membrane-Based, Sedimentation-Assisted Plasma Separator for Point-of-Care Applications. Anal. Chem. 2013, 85, 10463–10470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, X.; Jiang, H.; Zhang, X.; Li, K.; Ge, S.; Xia, N.; Mauk, M.G. A plasma separator with a multifunctional deformable chamber equipped with a porous membrane for point-of-care diagnostics. Analysis 2020, 145, 6138–6147. [Google Scholar] [CrossRef] [PubMed]
- Piliarik, M.; Bocková, M.; Homola, J. Surface plasmon resonance biosensor for parallelized detection of protein biomarkers in diluted blood plasma. Biosens. Bioelectron. 2010, 26, 1656–1661. [Google Scholar] [CrossRef]
- Makowski, M.S.; Ivanisevic, A. Molecular Analysis of Blood with Micro-/Nanoscale Field-Effect-Transistor Biosensors. Small 2011, 7, 1863–1875. [Google Scholar] [CrossRef] [Green Version]
- Kuan, D.-H.; Wang, I.-S.; Lin, J.-R.; Yang, C.-H.; Huang, C.-H.; Lin, Y.-H.; Lin, C.-T.; Huang, N.-T. A microfluidic device integrating dual CMOS polysilicon nanowire sensors for on-chip whole blood processing and simultaneous detection of multiple analytes. Lab Chip 2016, 16, 3105–3113. [Google Scholar] [CrossRef]
- Enomoto, J.; Matharu, Z.; Revzin, A. Chapter Six—Electrochemical Biosensors for On-Chip Detection of Oxidative Stress from Cells. In Methods in Enzymology; Cadenas, E., Packer, L., Eds.; Academic Press: Cambridge, MA, USA, 2013; Volume 526, pp. 107–121. [Google Scholar]
- Wang, L.; Zhang, Y.; Cheng, C.; Liu, X.; Jiang, H.; Wang, X. Highly Sensitive Electrochemical Biosensor for Evaluation of Oxidative Stress Based on the Nanointerface of Graphene Nanocomposites Blended with Gold, Fe3O4, and Platinum Nanoparticles. ACS Appl. Mater. Interfaces 2015, 7, 18441–18449. [Google Scholar] [CrossRef]
- Noiphung, J.; Songjaroen, T.; Dungchai, W.; Henry, C.S.; Chailapakul, O.; Laiwattanapaisal, W. Electrochemical detection of glucose from whole blood using paper-based microfluidic devices. Anal. Chim. Acta 2013, 788, 39–45. [Google Scholar] [CrossRef]
- Wu, H.; Shi, C.; Zhu, Q.; Li, Y.; Xu, Z.; Wei, C.; Chen, D.; Huang, X. Capillary-driven blood separation and in-situ electrochemical detection based on 3D conductive gradient hollow fiber membrane. Biosens. Bioelectron. 2020, 171, 112722. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Fan, Y.; Cheng, Y.; Yang, J. An electrochemical Lab-on-a-CD system for parallel whole blood analysis. Lab Chip 2013, 13, 2634–2640. [Google Scholar] [CrossRef] [PubMed]
- Chikkaveeraiah, B.V.; Mani, V.; Patel, V.; Gutkind, J.S.; Rusling, J.F. Microfluidic electrochemical immunoarray for ultrasensitive detection of two cancer biomarker proteins in serum. Biosens. Bioelectron. 2011, 26, 4477–4483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swensen, J.S.; Xiao, Y.; Ferguson, B.S.; Lubin, A.A.; Lai, R.Y.; Heeger, A.J.; Plaxco, K.W.; Soh, H.T. Continuous, Real-Time Monitoring of Cocaine in Undiluted Blood Serum via a Microfluidic, Electrochemical Aptamer-Based Sensor. J. Am. Chem. Soc. 2009, 131, 4262–4266. [Google Scholar] [CrossRef] [Green Version]
- Thangamuthu, M.; Santschi, C.; Martin, O.J.F. Label-Free Electrochemical Immunoassay for C-Reactive Protein. Biosensors 2018, 8, 34. [Google Scholar] [CrossRef] [Green Version]
- Chinnadayyala, S.R.; Park, J.; Kim, Y.H.; Choi, S.H.; Lee, S.-M.; Cho, W.W.; Lee, G.-Y.; Pyun, J.-C.; Cho, S. Electrochemical Detection of C-Reactive Protein in Human Serum Based on Self-Assembled Monolayer-Modified Interdigitated Wave-Shaped Electrode. Sensors 2019, 19, 5560. [Google Scholar] [CrossRef] [Green Version]
- Avan, A.; Tavakoly Sany, S.B.; Ghayour-Mobarhan, M.; Rahimi, H.R.; Tajfard, M.; Ferns, G. Serum C-reactive protein in the prediction of cardiovascular diseases: Overview of the latest clinical studies and public health practice. J. Cell. Physiol. 2018, 233, 8508–8525. [Google Scholar] [CrossRef]
- Li, K.; Wei, P.; Qin, Y.; Wei, Y. Is C-reactive protein a marker of obstructive sleep apnea? A meta-analysis. Medicine 2017, 96, e6850. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.J.; Choi, K.M.; Lee, S.K.; Yoon, D.W.; Lee, S.G.; Shin, C. Obstructive Sleep Apnea Is Associated with Elevated High Sensitivity C-Reactive Protein Levels Independent of Obesity: Korean Genome and Epidemiology Study. PLoS ONE 2016, 11, e0163017. [Google Scholar] [CrossRef]
- Nadeem, R.; Molnar, J.; Madbouly, E.M.; Nida, M.; Aggarwal, S.; Sajid, H.; Naseem, J.; Loomba, R. Serum Inflammatory Markers in Obstructive Sleep Apnea: A Meta-Analysis. J. Clin. Sleep Med. 2013, 9, 1003–1012. [Google Scholar] [CrossRef] [Green Version]
- Shamsuzzaman, A.S.; Winnicki, M.; Lanfranchi, P.; Wolk, R.; Kara, T.; Accurso, V.; Somers, V.K. Elevated C-Reactive Protein in Patients with Obstructive Sleep Apnea. Circulation 2002, 105, 2462–2464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sapp, P.A.; Riley, T.M.; Tindall, A.M.; Sullivan, V.K.; Johnston, E.A.; Petersen, K.S.; Kris-Etherton, P.M. Chapter 22—Nutrition and Atherosclerotic Cardiovascular Disease. In Present Knowledge in Nutrition, 11th ed.; Marriott, B.P., Birt, D.F., Stallings, V.A., Yates, A.A., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 393–411. [Google Scholar]
- Xu, X.; Makaraviciute, A.; Pettersson, J.; Zhang, S.-L.; Nyholm, L.; Zhang, Z. Revisiting the factors influencing gold electrodes prepared using cyclic voltammetry. Sens. Actuators B Chem. 2018, 283, 146–153. [Google Scholar] [CrossRef]
- Lin, M. Blood groups and transfusion medicine in Taiwan. J. Formos. Med. Assoc. Taiwan Yi Zhi 1997, 96, 933–942. [Google Scholar] [PubMed]
- Agrawal, A.; Bhattacharya, P.; Kamath, S.; Mehta, N.; Tiwari, A.K.; Tulsiani, S.; Wankhede, R. ABO and Rh (D) group distribution and gene frequency; the first multicentric study in India. Asian J. Transfus. Sci. 2014, 8, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, E.S.; A Lerman, M. How to use the erythrocyte sedimentation rate in paediatrics. Arch. Dis. Child. Educ. Pract. Ed. 2014, 100, 30–36. [Google Scholar] [CrossRef] [Green Version]
- Kuan, D.-H.; Wu, C.-C.; Su, W.-Y.; Huang, N.-T. A Microfluidic Device for Simultaneous Extraction of Plasma, Red Blood Cells, and On-Chip White Blood Cell Trapping. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Le, H.; Cho, S. Deciphering the Disaggregation Mechanism of Amyloid Beta Aggregate by 4-(2-Hydroxyethyl)-1-Piperazinepropanesulfonic Acid Using Electrochemical Impedance Spectroscopy. Sensors 2021, 21, 788. [Google Scholar] [CrossRef]
- Guerrero, S.; Martínez-García, G.; Serafín, V.; Agüí, L.; Yáñez-Sedeño, P.; Pingarrón, J.M. Electrochemical immunosensor for sensitive determination of the anorexigen peptide YY at grafted reduced graphene oxide electrode platforms. Analysis 2015, 140, 7527–7533. [Google Scholar] [CrossRef]
- Luna, D.M.; Avelino, K.Y.; Cordeiro, M.T.; Andrade, C.A.; Oliveira, M.D. Electrochemical immunosensor for dengue virus serotypes based on 4-mercaptobenzoic acid modified gold nanoparticles on self-assembled cysteine monolayers. Sens. Actuators B Chem. 2015, 220, 565–572. [Google Scholar] [CrossRef]
- Cheng, I.-F.; Yang, H.-L.; Chung, C.-C.; Chang, H.-C. A rapid electrochemical biosensor based on an AC electrokinetics enhanced immuno-reaction. Analysis 2013, 138, 4656–4662. [Google Scholar] [CrossRef]
- Chinnadayyala, S.R.; Cho, S. Electrochemical Immunosensor for the Early Detection of Rheumatoid Arthritis Biomarker: Anti-Cyclic Citrullinated Peptide Antibody in Human Serum Based on Avidin-Biotin System. Sensors 2020, 21, 124. [Google Scholar] [CrossRef] [PubMed]
- Geng, P.; Zhang, X.; Meng, W.; Wang, Q.; Zhang, W.; Jin, L.; Feng, Z.; Wu, Z. Self-assembled monolayers-based immunosensor for detection of Escherichia coli using electrochemical impedance spectroscopy. Electrochimica Acta 2008, 53, 4663–4668. [Google Scholar] [CrossRef]
- Stanković, V.; Đurđić, S.; Ognjanović, M.; Antić, B.; Kalcher, K.; Mutić, J.; Stanković, D.M. Anti-human albumin monoclonal antibody immobilized on EDC-NHS functionalized carboxylic graphene/AuNPs composite as promising electrochemical HSA immunosensor. J. Electroanal. Chem. 2020, 860, 113928. [Google Scholar] [CrossRef]
- Verbakel, J.Y.; Aertgeerts, B.; Lemiengre, M.; De Sutter, A.; A Bullens, D.M.; Buntinx, F. Analytical accuracy and user-friendliness of the Afinion point-of-care CRP test. J. Clin. Pathol. 2013, 67, 83–86. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, Z.-X.; Wu, C.-C.; Huang, N.-T. A Microfluidic Platform with an Embedded Miniaturized Electrochemical Sensor for On-Chip Plasma Extraction Followed by In Situ High-Sensitivity C-Reactive Protein (hs-CRP) Detection. Biosensors 2022, 12, 1163. https://doi.org/10.3390/bios12121163
Lai Z-X, Wu C-C, Huang N-T. A Microfluidic Platform with an Embedded Miniaturized Electrochemical Sensor for On-Chip Plasma Extraction Followed by In Situ High-Sensitivity C-Reactive Protein (hs-CRP) Detection. Biosensors. 2022; 12(12):1163. https://doi.org/10.3390/bios12121163
Chicago/Turabian StyleLai, Zhi-Xuan, Chia-Chien Wu, and Nien-Tsu Huang. 2022. "A Microfluidic Platform with an Embedded Miniaturized Electrochemical Sensor for On-Chip Plasma Extraction Followed by In Situ High-Sensitivity C-Reactive Protein (hs-CRP) Detection" Biosensors 12, no. 12: 1163. https://doi.org/10.3390/bios12121163




