Valorization of Invasive Plant Extracts against the Bispecies Biofilm Staphylococcus aureus–Candida albicans by a Bioguided Molecular Networking Screening
Abstract
1. Introduction
2. Results and Discussion
2.1. Plant Extracts
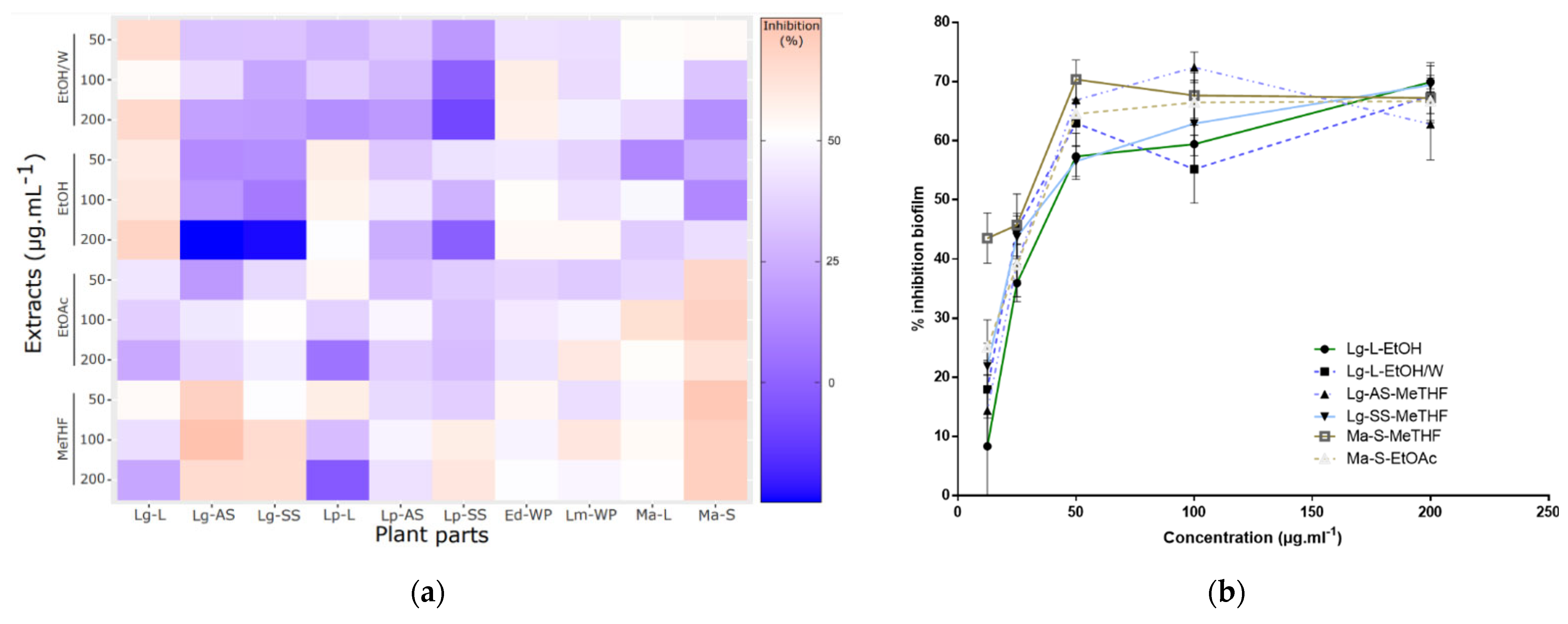
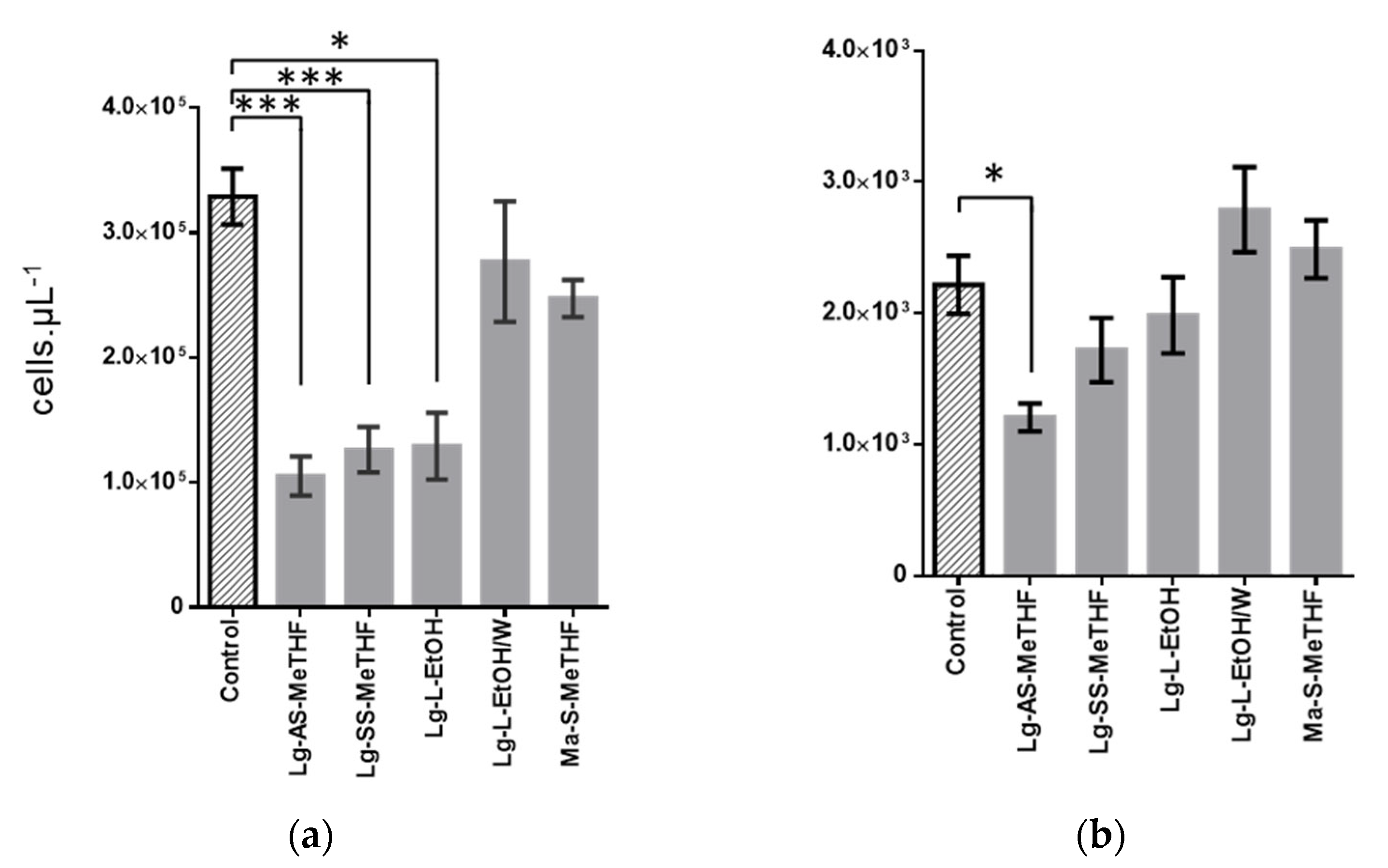
2.2. Antibiofilm Activities Screening
2.3. Characterization of Active Extracts
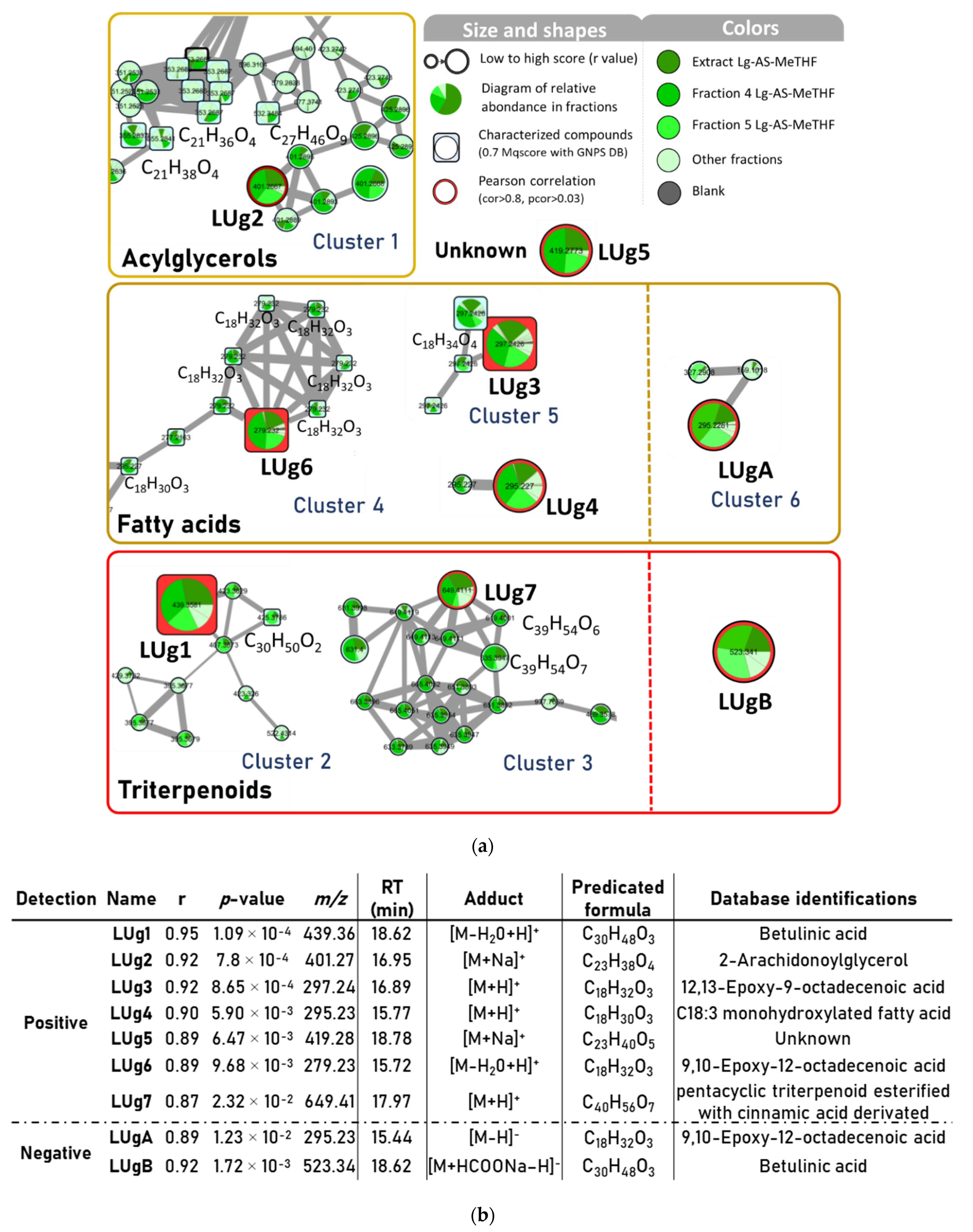
2.4. Bioactive Molecular Networking
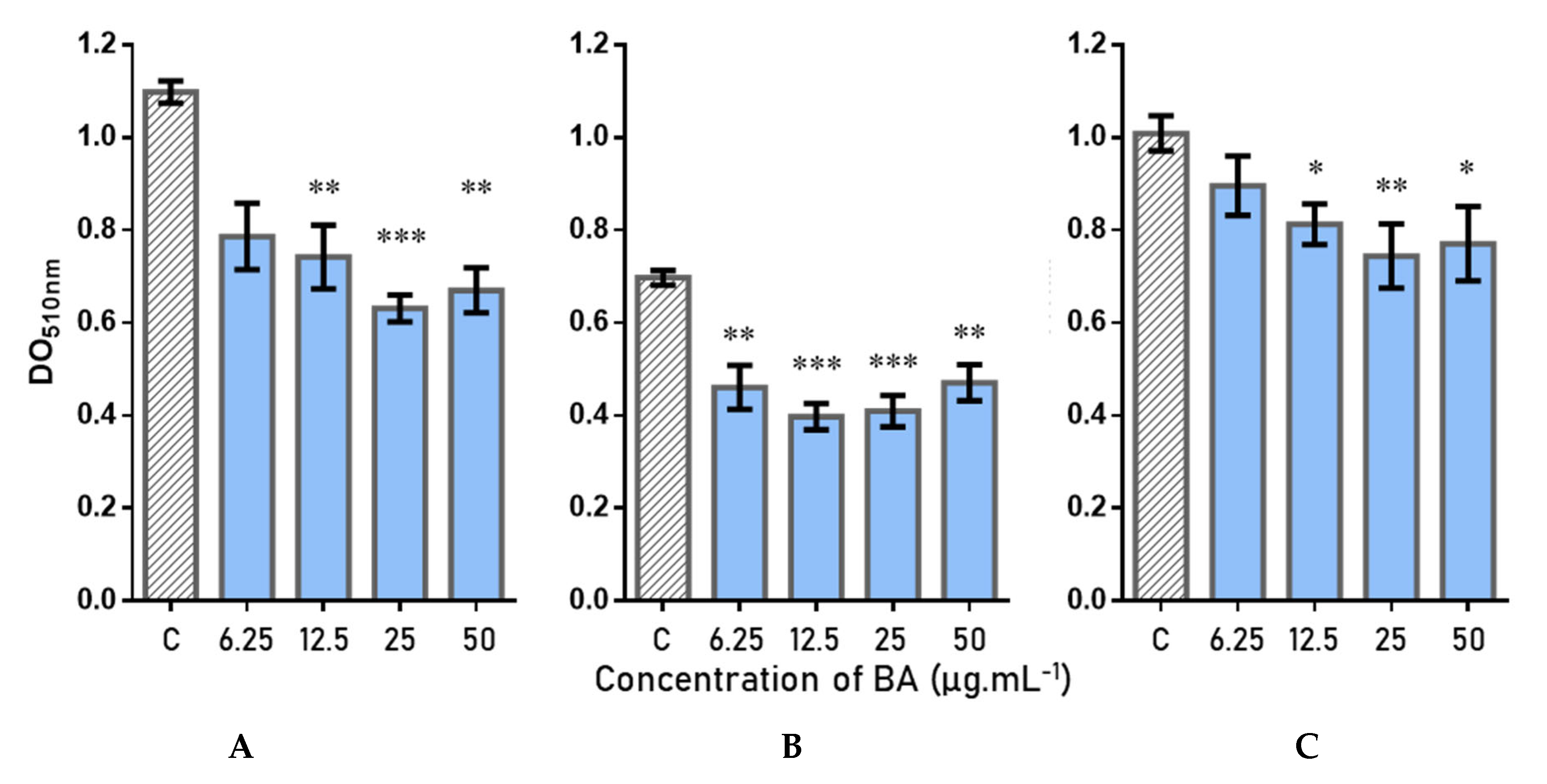
2.5. Antibiofilm Activity and Quantification of Betulinic Acid
3. Materials and Methods
3.1. Plant Material
3.2. Chemicals and Reagents
3.3. Extraction and Fractionation
3.4. HPLC Analysis
3.5. Organisms
3.6. Biofilm Studies
3.6.1. Biofilm Growth
3.6.2. Crystal Violet Staining (CVS) Assay
3.6.3. Flow Cytometry (FCM) Assay
3.6.4. Cryo-Scanning Electron Microscopy
3.7. Molecular Networking
3.7.1. Data-Dependent LC-ESI-HRMS2 Analyses
3.7.2. MZmine 2 Data Preprocessing Parameters
3.7.3. Molecular Networks Analysis
3.8. Annotation and Quantification of Betulinic Acid
3.9. Statistical Analyses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival Mechanisms of Clinically Relevant Microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhao, H.; Wei, Y.; Gan, X.; Ling, Y.; Ying, Y. Retrospective Analysis of Microbial Colonization Patterns in Central Venous Catheters, 2013–2017. J. Healthc. Eng. 2019, 2019, 8632701. [Google Scholar] [CrossRef] [PubMed]
- Mavor, A.L.; Thewes, S.; Hube, B. Systemic Fungal Infections Caused by Candida Species: Epidemiology, Infection Process and Virulence Attributes. Curr. Drug Targets 2005, 6, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Johnson, A.D. Candida albicans Biofilms and Human Disease. Annu. Rev. Microbiol. 2015, 69, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Bougnoux, M.-E.; Diogo, D.; François, N.; Sendid, B.; Veirmeire, S.; Colombel, J.F.; Bouchier, C.; Van Kruiningen, H.; D’Enfert, C.; Poulain, D. Multilocus Sequence Typing Reveals Intrafamilial Transmission and Microevolutions of Candida albicans Isolates from the Human Digestive Tract. J. Clin. Microbiol. 2006, 44, 1810–1820. [Google Scholar] [CrossRef]
- Nett, J.; Andes, D. Candida albicans biofilm development, modeling a host–pathogen interaction. Curr. Opin. Microbiol. 2006, 9, 340–345. [Google Scholar] [CrossRef]
- Van Dyck, K.; Pinto, R.M.; Pully, D.; Van Dijck, P. Microbial Interkingdom Biofilms and the Quest for Novel Therapeutic Strategies. Microorganisms 2021, 9, 412. [Google Scholar] [CrossRef]
- Carolus, H.; Van Dyck, K.; Van Dijck, P. Candida albicans and Staphylococcus Species: A Threatening Two-some. Front. Microbiol. 2019, 10, 2162. [Google Scholar] [CrossRef]
- Eichelberger, K.R.; Cassat, J.E. Metabolic Adaptations During Staphylococcus aureus and Candida albicans Co-Infection. Front. Immunol. 2021, 12, 797550. [Google Scholar] [CrossRef]
- Bouza, E.; Burillo, A.; Muñoz, P.; Guinea, J.; Marín, M.; Rodríguez-Créixems, M. Mixed bloodstream infections involving bacteria and Candida spp. J. Antimicrob. Chemother. 2013, 68, 1881–1888. [Google Scholar] [CrossRef][Green Version]
- Zhong, L.; Zhang, S.; Tang, K.; Zhou, F.; Zheng, C.; Zhang, K.; Cai, J.; Zhou, H.; Wang, Y.; Tian, B.; et al. Clinical characteristics, risk factors and outcomes of mixed Candida albicans/bacterial bloodstream infections. BMC Infect. Dis. 2020, 20, 810. [Google Scholar] [CrossRef]
- Klotz, S.A.; Chasin, B.S.; Powell, B.; Gaur, N.K.; Lipke, P.N. Polymicrobial bloodstream infections involving Candida species: Analysis of patients and review of the literature. Diagn. Microbiol. Infect. Dis. 2007, 59, 401–406. [Google Scholar] [CrossRef]
- Harriott, M.M.; Noverr, M.C. Candida albicans and Staphylococcus aureus Form Polymicrobial Biofilms: Effects on Antimicrobial Resistance. Antimicrob. Agents Chemother. 2009, 53, 3914–3922. [Google Scholar] [CrossRef]
- Schlecht, L.M.; Peters, B.M.; Krom, B.P.; Freiberg, J.A.; Hänsch, G.M.; Filler, S.G.; Jabra-Rizk, M.A.; Shirtliff, M.E. Systemic Staphylococcus aureus infection mediated by Candida albicans hyphal invasion of mucosal tissue. Microbiology 2015, 161, 168–181. [Google Scholar] [CrossRef]
- Todd, O.A.; Fidel, P.L.; Harro, J.M.; Hilliard, J.J.; Tkaczyk, C.; Sellman, B.R.; Noverr, M.C.; Peters, B.M. Candida albicans Augments Staphylococcus aureus Virulence by Engaging the Staphylococcal agr Quorum Sensing System. MBio 2019, 10, e00910-19. [Google Scholar] [CrossRef]
- Moutou, F.; Pastoret, P.P. Defining an invasive species. Rev. Sci. Tech. l’OIE 2010, 29, 37–45. [Google Scholar]
- IUCN. Invasive Alien Species and Climate Change. 2021. Available online: https://iucn.org/resources/issues-brief/invasive-alien-species-and-climate-change (accessed on 3 November 2022).
- Reddy, A.M.; Pratt, P.D.; Grewell, B.J.; Harms, N.E.; Walsh, G.C.; Ndez, M.C.H.; Faltlhauser, A.; Ci-bils-Stewart, X. Biological Control of Invasive Water Primroses, Ludwigia spp., in the United States: A Feasi-bility Assessment. J. Aquat. Plant Manag. 2021, 11, 67–77. [Google Scholar]
- Fan, P.; Marston, A. How Can Phytochemists Benefit from Invasive Plants? Nat. Prod. Commun. 2009, 4, 1407–1416. [Google Scholar] [CrossRef]
- Máximo, P.; Ferreira, L.M.; Branco, P.S.; Lourenço, A. Invasive Plants: Turning Enemies into Value. Molecules 2020, 25, 3529. [Google Scholar] [CrossRef]
- Smida, I.; Sweidan, A.; Souissi, Y.; Rouaud, I.; Sauvager, A.; Torre, F.; Calvert, V.; Le Petit, J.; Tomasi, S. Anti-Acne, Antioxidant and Cytotoxic Properties of Ludwigia Peploides Leaf Extract. Int. J. Pharmacogn. Phytochem. Res. 2018, 10, 271–278. [Google Scholar]
- Smida, I.; Charpy-Roubaud, C.; Cherif, S.Y.; Torre, F.; Audran, G.; Smiti, S.; Le Petit, J. Antibacterial properties of extracts of Ludwigia peploides subsp. montevidensis and Ludwigia grandiflora subsp. hexapetala during their cycle of development. Aquat. Bot. 2015, 121, 39–45. [Google Scholar] [CrossRef]
- Visweswari, G.; Christopher, R.; Rajendra, W.; Visweswari, D.G. Phytochemical Screening of Active Secondary Metabolites Present in Withania somnifera Root: Role in Traditional Medecine. Int. J. Pharm. Sci. Res. 2013, 4, 2770–2776. [Google Scholar]
- Chinou, I. Primary and Secondary Metabolites and Their Biological Activity. In Thin Layer Chromatography in Phytochemistry; CRC Press: Boca Raton, FL, USA, 2008; pp. 60–74. [Google Scholar]
- Sousa, S.N.B.; De Andrade, M.L.; Brito, R.D.V.D.D. Antibiofilm Activity of Natural Products: Promising Strategies for Combating Microbial Biofilms. Ann. Public Health Rep. 2020, 4, 92–99. [Google Scholar] [CrossRef]
- Kim, Y.-G.; Lee, J.-H.; Park, J.G.; Lee, J. Inhibition of Candida albicans and Staphylococcus aureus biofilms by centipede oil and linoleic acid. Biofouling 2020, 36, 126–137. [Google Scholar] [CrossRef]
- Bernard, C.; Renaudeau, N.; Mollichella, M.-L.; Quellard, N.; Girardot, M.; Imbert, C. Cutibacterium acnes protects Candida albicans from the effect of micafungin in biofilms. Int. J. Antimicrob. Agents 2018, 52, 942–946. [Google Scholar] [CrossRef]
- Domingue, G.; Costerton, J.W.; Brown, M.R. Bacterial doubling time modulates the effects of opsonisation and available iron upon interactions between Staphylococcus aureus and human neutrophils. FEMS Immunol. Med. Microbiol. 1996, 16, 223–228. [Google Scholar] [CrossRef]
- Mahto, K.K.; Singh, A.; Khandelwal, N.K.; Bhardwaj, N.; Jha, J.; Prasad, R. An Assessment of Growth Media Enrichment on Lipid Metabolome and the Concurrent Phenotypic Properties of Candida albicans. PLoS ONE 2014, 9, e113664. [Google Scholar] [CrossRef]
- Christensen, G.D.; Simpson, W.A.; Younger, J.J.; Baddour, L.M.; Barrett, F.F.; Melton, D.M.; Beachey, E.H. Adherence of coagulase-negative staphylococci to plastic tissue culture plates: A quantitative model for the adherence of staphylococci to medical devices. J. Clin. Microbiol. 1985, 22, 996–1006. [Google Scholar] [CrossRef]
- Alby, K.; Schaefer, D.; Sherwood, R.K.; Jones, S.; Bennett, R.J. Identification of a Cell Death Pathway in Candida albicans during the Response to Pheromone. Eukaryot. Cell 2010, 9, 1690–1701. [Google Scholar] [CrossRef]
- Nothias, L.-F.; Nothias-Esposito, M.; da Silva, R.; Wang, M.; Protsyuk, I.; Zhang, Z.; Sarvepalli, A.; Leyssen, P.; Touboul, D.; Costa, J.; et al. Bioactivity-Based Molecular Networking for the Discovery of Drug Leads in Natural Product Bioassay-Guided Fractionation. J. Nat. Prod. 2018, 81, 758–767. [Google Scholar] [CrossRef]
- Weber, H. Fatty acid-derived signals in plants. Trends Plant Sci. 2002, 7, 217–224. [Google Scholar] [CrossRef]
- Mitra, S.; Sarkar, N.; Barik, A. Long-chain alkanes and fatty acids from Ludwigia octovalvis weed leaf surface waxes as short-range attractant and ovipositional stimulant to Altica cyanea (Weber) (Coleoptera: Chrysomelidae). Bull. Èntomol. Res. 2017, 107, 391–400. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, Y.-G.; Park, J.G.; Lee, J. Supercritical fluid extracts of Moringa oleifera and their unsaturated fatty acid components inhibit biofilm formation by Staphylococcus aureus. Food Control 2017, 80, 74–82. [Google Scholar] [CrossRef]
- Yuyama, K.; Rohde, M.; Molinari, G.; Stadler, M.; Abraham, W.-R. Unsaturated Fatty Acids Control Biofilm Formation of Staphylococcus aureus and Other Gram-Positive Bacteria. Antibiotics 2020, 9, 788. [Google Scholar] [CrossRef]
- Nigam, S.; Ciccoli, R.; Ivanov, I.; Sczepanski, M.; Deva, R. On Mechanism of Quorum Sensing in Candida albicans by 3(R)-Hydroxy-Tetradecaenoic Acid. Curr. Microbiol. 2010, 62, 55–63. [Google Scholar] [CrossRef]
- Shareck, J.; Nantel, A.; Belhumeur, P. Conjugated Linoleic Acid Inhibits Hyphal Growth in Candida albicans by Modulating Ras1p Cellular Levels and Downregulating TEC1 Expression. Eukaryot. Cell 2011, 10, 565–577. [Google Scholar] [CrossRef]
- Gauthier, C.; Legault, J.; Lavoie, S.; Rondeau-Gagné, S.; Tremblay, S.; Pichette, A. Synthesis of two natural betulinic acid saponins containing α-l-rhamnopyranosyl-(1→2)-α-l-arabinopyranose and their analogues. Tetrahedron 2008, 64, 7386–7399. [Google Scholar] [CrossRef]
- Shilpi, J.A.; Gray, A.I.; Seidel, V. Chemical constituents from Ludwigia adscendens. Biochem. Syst. Ecol. 2010, 38, 106–109. [Google Scholar] [CrossRef]
- Bhattacharya, S.P.; Mitra, A.; Bhattacharya, A.; Sen, A. Quorum quenching activity of pentacyclic triterpenoids leads to inhibition of biofilm formation by Acinetobacter baumannii. Biofouling 2020, 36, 922–937. [Google Scholar] [CrossRef]
- da Silva, G.N.S.; Primon-Barros, M.; Macedo, A.J.; Gnoatto, S.C.B. Triterpene Derivatives as Relevant Scaffold for New Antibiofilm Drugs. Biomolecules 2019, 9, 58. [Google Scholar] [CrossRef]
- Yogeeswari, P.; Sriram, D. Betulinic Acid and Its Derivatives: A Review on their Biological Properties. Curr. Med. Chem. 2005, 12, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Hu, Y.; Yang, Y.; Wang, L.; Yang, X.; Wang, B.; Huang, M. Betulinic Acid Induces ROS-Dependent Apoptosis and S-Phase Arrest by Inhibiting the NF-κB Pathway in Human Multiple Myeloma. Oxid. Med. Cell. Longev. 2019, 2019, 5083158. [Google Scholar] [CrossRef] [PubMed]
- Bocalon, L.G.; Tozatti, M.G.; Januário, A.H.; Pauletti, P.M.; Silva, M.L.A.; Rocha, L.A.; Molina, E.F.; Santos, M.F.C.; Cunha, W.R. Incorporation of Betulinic Acid into Silica-Based Nanoparticles for Controlled Phytochemical Release. Anal. Lett. 2022, 1–15. [Google Scholar] [CrossRef]
- Kerstens, M.; Boulet, G.; Pintelon, I.; Hellings, M.; Voeten, L.; Delputte, P.; Maes, L.; Cos, P. Quantification of Candida albicans by flow cytometry using TO-PRO®-3 iodide as a single-stain viability dye. J. Microbiol. Methods 2013, 92, 189–191. [Google Scholar] [CrossRef]
- Pluskal, T.; Castillo, S.; Villar-Briones, A.; Orešič, M. MZmine 2: Modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinform. 2010, 11, 395. [Google Scholar] [CrossRef]
- Wang, M.; Carver, J.J.; Phelan, V.V.; Sanchez, L.M.; Garg, N.; Peng, Y.; Nguyen, D.D.; Watrous, J.; Kapono, C.A.; Luzzatto-Knaan, T.; et al. Sharing and community curation of mass spectrometry data with Global Natural Products Social Molecular Networking. Nat. Biotechnol. 2016, 34, 828–837. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of Biomolecular Interaction Networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Zhao, G.; Yan, W.; Cao, D. Simultaneous determination of betulin and betulinic acid in white birch bark using RP-HPLC. J. Pharm. Biomed. Anal. 2007, 43, 959–962. [Google Scholar] [CrossRef]
| Plants and Parts 2 Extracted | Extraction Solvents | Total per Plant | |||||
|---|---|---|---|---|---|---|---|
| MeTHF 1 | EtOAc | EtOH | EtOH/W | ||||
| Yield (%) | Yield (%) | Weight (mg) | |||||
| E. densa | WP | 1.09 | 0.58 | 1.39 | 4.59 | 7.67 | 2142 |
| L. major | WP | 1.19 | 0.06 | 0.56 | 2.89 | 4.69 | 940 |
| M. aquaticum | S | 4.85 | 0.95 | 8.52 | 19.30 | 33.64 | 6731 |
| L | 6.59 | 1.05 | 11.09 | 19.75 | 38.49 | 7701 | |
| L. peploides | AS | 2.54 | 0.30 | 3.41 | 16.09 | 22.37 | 4476 |
| SS | 2.18 | 0.19 | 3.07 | 11.42 | 16.87 | 3376 | |
| L | 11.21 | 0.16 | 6.44 | 19.62 | 37.42 | 7496 | |
| L. grandiflora | AS | 2.73 | 0.12 | 2.84 | 13.20 | 18.90 | 3783 |
| SS | 2.35 | 0.13 | 1.48 | 8.63 | 12.60 | 2522 | |
| L | 5.33 | 0.56 | 6.92 | 16.50 | 29.32 | 5873 | |
| Yield | Activity (CVS, 50 µg∙mL−1) | Bioactive Score | |||
|---|---|---|---|---|---|
| (%) | Inhibition (%) | SD (%) | |||
| Lg-AS-MeTHF (238 mg) | F1 | 7.11 | 0 | 20.03 | 0 |
| F2 | 1.50 | 0 | 27.50 | 0 | |
| F3 | 2.59 | 30.82 | 9.89 | 37 | |
| F4 | 1.10 | 63.96 | 6.89 | 100 | |
| F5 | 1.77 | 50.83 | 8.16 | 74 | |
| F6 | 1.87 | 39.19 | 13.63 | 44 | |
| F7 | 14.54 | 25.61 | 12.55 | 23 | |
| Extract | 53.88 | 7.96 | 80 | ||
| Lg-L-EtOH (600 mg) | F1′ | 15.71 | 0 | 21.20 | 0 |
| F2′ | 1.84 | 10.39 | 18.70 | 0 | |
| F3′ | 9.13 | 33.85 | 13.96 | 34 | |
| F4′ | 4.14 | 39.26 | 9.52 | 52 | |
| F5′ | 1.50 | 27.78 | 8.94 | 33 | |
| F6′ | 1.61 | 25.74 | 16.96 | 15 | |
| F7′ | 4.05 | 0 | 24.28 | 0 | |
| F8′ | 1.76 | 20.19 | 12.55 | 13 | |
| F9′ | 7.13 | 15.56 | 20.20 | 0 | |
| Extract | 42.25 | 7.85 | 60 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamion, G.; Aucher, W.; Tardif, C.; Miranda, J.; Rouger, C.; Imbert, C.; Girardot, M. Valorization of Invasive Plant Extracts against the Bispecies Biofilm Staphylococcus aureus–Candida albicans by a Bioguided Molecular Networking Screening. Antibiotics 2022, 11, 1595. https://doi.org/10.3390/antibiotics11111595
Hamion G, Aucher W, Tardif C, Miranda J, Rouger C, Imbert C, Girardot M. Valorization of Invasive Plant Extracts against the Bispecies Biofilm Staphylococcus aureus–Candida albicans by a Bioguided Molecular Networking Screening. Antibiotics. 2022; 11(11):1595. https://doi.org/10.3390/antibiotics11111595
Chicago/Turabian StyleHamion, Guillaume, Willy Aucher, Charles Tardif, Julie Miranda, Caroline Rouger, Christine Imbert, and Marion Girardot. 2022. "Valorization of Invasive Plant Extracts against the Bispecies Biofilm Staphylococcus aureus–Candida albicans by a Bioguided Molecular Networking Screening" Antibiotics 11, no. 11: 1595. https://doi.org/10.3390/antibiotics11111595
APA StyleHamion, G., Aucher, W., Tardif, C., Miranda, J., Rouger, C., Imbert, C., & Girardot, M. (2022). Valorization of Invasive Plant Extracts against the Bispecies Biofilm Staphylococcus aureus–Candida albicans by a Bioguided Molecular Networking Screening. Antibiotics, 11(11), 1595. https://doi.org/10.3390/antibiotics11111595










