Abstract
Antibiotic stewardship in urologic reconstruction is critically important, as many patients will require indwelling catheters for days to weeks following surgery and thus are at risk of both developing catheter-associated urinary tract infections (CAUTI) as well as multi-drug resistant (MDR) uropathogens. Accordingly, limiting antibiotic use, when safe, should help reduce antibiotic resistance and the prevalence of MDR organisms. However, there is significant heterogeneity in how antibiotics are prescribed to patients who need indwelling urethral catheters post-operatively. We performed a literature review to determine if there are benefits in the use of antibiotics for various clinical scenarios that require post-operative indwelling catheters for greater than 24 h. In general, for patients undergoing prostatectomy, transurethral resection of the prostate, and/or urethroplasty, antibiotic administration may be limited without increased risk of CAUTI. However, more work is needed to identify optimal antibiotic regimens for these and alternative urologic procedures, whether certain sub-populations benefit from longer courses of antibiotics, and effective non-antibiotic or non-systemic therapies.
1. Introduction
Antibiotic stewardship is a challenging but important consideration in public health and in the care of individual patient. Urologists are routinely tasked with decisions regarding antibiotic selection and duration for both acute infection as well as prophylaxis. Though routinely delivered with the intent of net benefit, there is a great deal of heterogeneity in the paradigms surrounding the use of antibiotic prophylaxis across providers, institutions, and regions at large [1,2,3,4].
Unique and particularly relevant to the urologist are antibiotic practices surrounding indwelling catheterization via any drainage tube that is inserted into the urinary bladder through the urethra or suprapubic region and connected to a closed collection system. There are multiple indications for the use of indwelling catheters, including management of urinary retention, careful monitoring of urine output in critically ill patients, urinary decompression, and support for healing following lower urinary tract procedures. Depending on the indication, indwelling urethral catheters may remain in situ post-operatively for days to weeks at a time.
Indwelling urethral catheters, though necessary in many post-operative settings, are long recognized as nidi for urinary tract infection (UTI). Nevertheless, there is considerable variability in the administration of prophylactic antibiotics to patients with indwelling urethral catheters following urological procedures [1,2,3]. Specifically, certain providers prescribe continuous antibiotics for the duration of catheterization, others prescribe antibiotics for 24–72 h surrounding catheter removal, and some will not prescribe antibiotics at all [1].
The rationale for the use of prophylactic antibiotics in the setting of catheterization after lower urinary tract reconstruction is to decrease the likelihood of symptomatic infection and/or to reduce the likelihood of bacteriuria and associated debris, which may result in catheter obstruction. Catheter-associated urinary tract infections (CAUTI) contribute significantly to healthcare costs at an estimated $115 million to $1.82 billion annually [5]. In most cases, post-operative CAUTI results in mild discomfort for the patient. However, more severe consequences of infection can include severe bladder spasms, incontinence, and sepsis. In the most dreaded scenario, a patient may experience poor catheter drainage secondary to an infection, which can compromise complex surgical repairs, resulting in significant morbidity.
There remain no urologic guidelines that recommend routine utilization of prophylactic antibiotics during a period of prolonged catheterization. Continuous prophylactic antibiotics are not explicitly recommended in the setting of prolonged catheterization due to a lack of evidence that true infections are prevented [6,7,8]. Accordingly, there are knowledge gaps and inconsistencies within the existing literature, societal guidelines, and common practice surrounding the use of antibiotic prophylaxis for post-procedural prolonged indwelling urethral catheterization. It is, therefore, worthwhile to identify if there are circumstances in which antibiotic prophylaxis for catheterization in these settings may be warranted. Should these circumstances exist, then unfavorable infectious outcomes may be prevented with the administration of antibiotics. If not, then antibiotic stewardship may improve by increasing provider confidence in not prescribing antibiotics, thus reducing the development of multi-drug resistant (MDR) organisms.
Comprehensive Cochrane reviews from 2012 and 2013 on antibiotic prophylaxis for short-term bladder drainage in adults [9] and on catheter policies for long-term bladder drainage [10] were conflicted regarding the benefit of antibiotic prophylaxis in the setting of postoperative indwelling catheterization, as are existing societal guidelines [6,7,8,11]. Given the lack of consensus or comprehensive review specific to the post urological surgical setting, we explore the use of post-procedural prophylactic antibiotics in the setting of post-surgical indwelling urethral catheters across the spectrum of urologic lower urinary tract procedures. Specifically, we wish to review current guidelines and existing evidence on the impact of antibiotic prophylaxis with post-surgical indwelling catheterization of >24 h on rates of lower urinary tract infection/cystitis, febrile UTI, and catheter obstruction.
1.1. The Association between Indwelling Catheters, Bacteriuria, and Infection
Urethral catheters are indwelling foreign bodies associated with the colonization of microorganisms (bacteria and fungus), which is temporal in nature [2]. While most patients with short-term indwelling catheters do not acquire colonized bacteria in their urine, virtually all patients will eventually demonstrate bacteria in their urine if catheterization continues for a prolonged interval [8,12,13,14]. Each day that an indwelling catheter is maintained, the risk of bacterial colonization increases by 5% to 10% [15]. Common bacterial and fungal colonizers of urethral catheters include Escherichia coli, Enterococcus spp., and Candida spp. [7,16].
Catheters are also vulnerable to the development of biofilm, which are matrix-enclosed microbial accretions that adhere to biological or non-biological surfaces [17,18]. One of the more worrying complications of bacterial colonization, particularly in the setting of urologic reconstruction, includes urinary obstruction. Crystalline biofilms, particularly those formed by Proteus mirabilis, can aggregate and occlude the lumen of a catheter [18]. With the poor flow of urine caused by a blocked catheter, a patient is vulnerable to the development of severe urinary infection, bacteremia, and breakdown of any surgical repairs of the urinary tract.
Approximately 75–90% of patients with catheter-associated bacteriuria (CAB) will not develop an inflammatory response or other signs or symptoms to suggest infection [7,19,20]. Although commonly asymptomatic and subclinical, CAB may precede CAUTI and related complications [13,14,21]. Table 1 summarizes the definitions of asymptomatic bacteriuria (ASB), CAB, UTI without a catheter, and CAUTI according to the Infectious Diseases Society of America (ISDA), European Association of Urology (EUA), Centers for Disease Control and Prevention (CDC), and American Urological Association (AUA) [7,8,12,22,23,24,25]. CAUTIs are undoubtedly a significant problem on a global scale, and they have been associated with increased morbidity, mortality, hospital cost, and length of stay for hospitalized patients [19,20,21,26,27]. UTIs are the most common type of healthcare-associated infection, accounting for more than 30% of infections reported by acute care hospitals [26,28]. However, the variability in definitions of UTIs across societies highlights the challenges in identifying infection—both in clinical practice as well as an outcome in research settings. To this end, there are many studies that use the term CAUTI when referring to cases of CAB [7,8], and we would venture that ambiguity also extends to the postoperative setting. Heterogeneity in clinical and research settings presents issues in collating data and assessing the quality of evidence [26].

Table 1.
Societal definitions of asymptomatic bacteriuria and urinary tract infections with and without catheters.
1.2. Societal Recommendations and Guidelines on Antibiotic Prophylaxis in Urology
By definition, antimicrobial prophylaxis is preventive in nature: it may be considered primary prevention (prevention of initial infection) or secondary prevention (prevention of the recurrence or reactivation of an infection) [29]. Antibiotic prophylaxis poses a double-edged sword. While its systemic use can, in certain situations, limit the catheter colonization of microorganisms and the development of infections, it can also drive the development of MDR organisms [8,12,13,30]. Antibiotic use also carries a risk of adverse side effects and increased medical costs. Thus, a delicate balance must be achieved in order to derive net benefit. Experts in urology and infectious diseases have synthesized available evidence and generated guidelines regarding the use of antimicrobial prophylaxis in the context of indwelling catheterization. Table 2 summarizes relevant guideline recommendations from the AUA, ISDA, EAU, and CDC. Notably absent are specific recommendations regarding the use of continuous antibiotic prophylaxis (CAP) for postoperative indwelling catheters after lower urinary tract reconstruction.

Table 2.
Summary of guideline recommendations regarding the use of continuous or peri-removal antibiotic prophylaxis with indwelling urinary catheters.
This is most likely related to a relative lack of evidence and relevance as compared to indwelling catheters in other settings (e.g., temporary catheterization in acutely ill patients or patients undergoing non-urologic surgery). The AUA recommends antibiotic prophylaxis at the time of postoperative catheter removal in certain settings [11], whereas EAU guidelines recommend against antibiotic prophylaxis in this setting given the relative societal risks as compared to the likelihood of individual patient benefit [8].
2. Materials and Methods
2.1. Aim/Purpose
As outlined previously, the available societal guidelines from the AUA, EAU, IDSA, and CDC do not explicitly recommend the routine use of continuous prophylactic antibiotics to prevent CAB or CAUTI in patients with indwelling urethral catheters. Across these groups, there are conflicting recommendations for or against antibiotic prophylaxis at the time of catheter removal or exchange based largely on patient factors.
With this review, we aim to explore if there are clinical scenarios in which the use of antibiotics in patients with temporary indwelling urinary catheters remaining longer than 24 h is of benefit following urologic procedures. We then summarize the findings for each clinical scenario and highlight our recommendations (in bolded text). We adhered to standards as outlined by Baethge et al. to optimize review quality and relevance [31].
2.2. Review Framework
2.2.1. Types of Studies
We evaluated studies centered on antibiotic prophylaxis for post-operative catheterization in patients who underwent urologic procedures with an anticipated duration of catheterization > 24 h. We included randomized controlled trials, prospective descriptive studies, retrospective quasi-experimental studies, and surveys.
2.2.2. Participants
We included studies with adults (age ≥ 18 years) requiring post-operative indwelling urethral catheterization (regardless of catheter size, design, or material) following urologic procedures, including radical prostatectomy, transurethral resection of the prostate, holmium laser enucleation of the prostate (HoLEP), transurethral resection of bladder tumor, bladder diverticulectomy, partial cystectomy, urethral diverticulectomy, and urethroplasty.
2.2.3. Interventions and Comparisons
Interventions considered were continuous antibiotic prophylaxis for the duration of catheterization, antibiotic prophylaxis at the time of catheter removal only, and no antibiotic prophylaxis.
2.2.4. Article Search
We performed an electronic search to identify relevant studies from PubMed. We limited our search to full-text articles that were available in the English language. No automation screening tools were used.
The search terms utilized were:
(antibiotic prophylaxis)
AND
(urinary catheter)
AND
(prostatectomy) or (RALP) or (transurethral resection of the prostate) or (TURP) or (holmium laser enucleation of the prostate) or (HoLEP) or (transurethral resection of bladder tumor) or (TURBT) or (bladder diverticulectomy) or (partial cystectomy) or (urethral diverticulectomy) or (urethroplasty).
Authors FM and CC independently assessed all titles and abstracts identified by the search for relevance to the topic. Where there was any possibility that the study might be included, the full paper was obtained and reviewed. Where discordance existed, a consensus was reached by FM, CC, and JC. The date of the most recent search of the register for this review was 11/25/2022.
2.2.5. Outcomes
Outcomes of interest included symptomatic lower urinary tract infection/cystitis, fever, sepsis, catheter malfunction, and cultures with MDR bacteria. The presence of these outcomes was based on their study-specific definitions rather than pre-existing criteria set by the authors of this review.
3. Results
Figure 1 outlines the identification and exclusion of studies for this literature review (See Figure 1, inspired by Page et al. [32]). In our search of the PubMed database, we identified six studies for robotic prostatectomy (1 prospective descriptive study [33], three retrospective cohort studies [34,35,36], and two prospective randomized controlled trials [37,38]), four studies for transurethral resection of the prostate (4 prospective randomized controlled trials [39,40,41,42]), and six studies for urethroplasty (2 cross-sectional surveys [2,3], three retrospective descriptive studies [43,44,45], and one prospective controlled trial [46]).
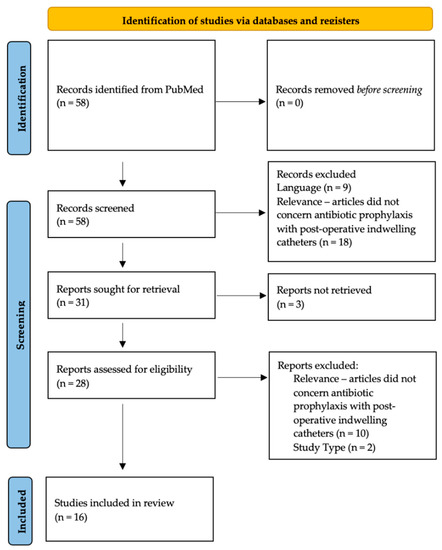
Figure 1.
Identification and exclusion of studies for this literature review.
In our PubMed search, we did not find relevant articles concerning antibiotic prophylaxis for indwelling urethral catheterization following holmium laser enucleation of the prostate (HoLEP), transurethral resection of bladder tumor, bladder diverticulectomy, partial cystectomy, or urethral diverticulectomy.
3.1. Radical Prostatectomy
Patients undergoing radical prostatectomy require temporary use of an indwelling urinary catheter after surgery for primary healing of the vesicourethral anastomosis. Typically, the catheter remains in place for six to fourteen days, depending on the surgeon’s preference.
A 2013 study prospectively examined urine cultures obtained from patients who underwent radical prostatectomy immediately prior to catheter removal on postoperative day (POD) 10. All patients received prophylactic antibiotics surrounding catheter removal with oral ciprofloxacin beginning the night before catheter removal and continuing afterward for a total of seven days. Of 334 patients, 83 (25%) had positive cultures with organisms > 1000 CFU/mL, of which 7% were resistant to ciprofloxacin. The authors concluded that a substantial proportion of prostatectomy patients have positive urine cultures at the time of catheter removal despite the administration of prophylactic fluoroquinolone antibiotics. However, outcomes were favorable overall when culture-specific oral antibiotic therapy was initiated. Nevertheless, the benefits of antibiotic administration in the setting of this study were difficult to assess without a control arm [33].
Shin and colleagues compared infectious outcomes after radical prostatectomy between two different antibiotic protocols. Specifically, 153 patients were administered a cephalosporin for fewer than 2 days, and 160 patients received a cephalosporin for more than 2 days. The presence of bacteriuria was examined at the time of catheter removal on POD 14. The researchers reported that the overall incidence of bacteriuria was 51% post-operatively and significantly higher in patients receiving the shorter antibiotic protocol compared to the longer protocol (57% vs. 45%). However, the incidence of fever was not significantly different between the two groups [34].
Pinochet and colleagues retrospectively examined rates of symptomatic UTI in patients who underwent radical prostatectomy by one of two surgeons. One surgeon routinely prescribed a 3-day course of ciprofloxacin prophylaxis starting the day before catheter removal on POD 11; the other surgeon prescribed no antibiotics prior to catheter removal, which was performed on POD 7. Despite a longer catheterization, the group of patients who received antibiotics was observed to develop fewer UTIs in their post-operative course (3.1 vs. 7.3%). Fever was observed in 11 patients in the non-antibiotic group (2.4%); there were no fevers in patients receiving antibiotic prophylaxis. Based on their statistical analysis, the number needed to treat to prevent one UTI was estimated at 24, and to prevent one case of febrile UTI was 91 [35].
While these studies suggest there may be an advantage to the routine use of antibiotic regimens of longer duration, other studies have not. A 2017 study examined rates of CAUTI following a change of practice at their institution from a protocol of a prolonged course of prophylactic antibiotics following radical prostatectomy (perioperative cephalosporin and aminoglycoside followed by oral quinolones until catheter removal) to perioperative prophylaxis only. No significant difference in the incidence of CAUTI was noted [36].
Several studies have also examined administering antibiotics at the time of catheter removal only. In 2019, Berrondo and associates performed a prospective randomized controlled trial to evaluate the role of antibiotic prophylaxis prior to urinary catheter removal after radical prostatectomy in preventing UTI. The 167 patients were randomized to receive antibiotic prophylaxis (consisting of ciprofloxacin the evening before catheter removal and the morning of catheter removal) or to receive no antibiotics. Overall, eight (4.8%) patients developed symptomatic UTIs within 6 weeks of urinary catheter removal. No significant difference in the rate of UTI between the control group and the antibiotic prophylaxis group (5.95% vs. 6.02%) was observed [37]. Another randomized control from Ehdaie and colleagues assessed a 3-day course of antibiotics (their historic protocol) to a 1-day course at the time of catheter removal. Over 3 years, a total of 824 patients were randomized to either treatment. The authors found zero UTI (0%) in the 1-day regimen and three UTI (0.7%) in the 3-day regimen and accordingly declared the 1-day regimen to be non-inferior [38].
When taken together, these studies suggest that among patients after radical prostatectomy, a peri-operative dose of antibiotics only or a peri-operative dose of antibiotics plus a one-day course around catheter removal for patients may provide adequate prophylaxis against UTI while reducing the duration of antibiotics prescribed.
Although patients undergoing radical prostatectomy undergo lower urinary tract reconstruction, catheter duration is typically less than 1 week, all patients have male anatomy, and patients are considerably less likely to enter surgery with indwelling hardware or recurrent infections than many other patients undergoing lower urinary tract reconstruction for other indications. Therefore, it is not known if these conclusions can be extrapolated to patients undergoing procedures such as bladder diverticulectomy, partial cystectomy, or fistula repair.
3.2. Transurethral Resection of the Prostate (TURP)
Transurethral Resection of the Prostate (TURP) is an endoscopic procedure commonly performed to help relieve bladder outlet obstruction. In general, the literature centers on a comparison of perioperative prophylactic antibiotics versus no perioperative prophylaxis rather than antibiotic prophylaxis, throughout the duration of post-operative indwelling catheterization.
In 1994, Raz and associates randomized 101 patients undergoing TURP to either receive a prophylactic antibiotic regimen consisting of a single dose of ceftriaxone at the time of surgery and another dose at the time of catheter removal (3 to 4 days post-operatively) or no prophylactic antibiotics. Over the entire 28-day study period, bacteriuria appeared in six ceftriaxone-treated patients and in 20 control patients. Three ceftriaxone-treated patients developed symptoms of UTI requiring additional antibiotics versus 14 control patients. Moreover, the incidence and duration of fever were shorter in the patients treated with ceftriaxone, supporting the routine use of periprocedural and peri-catheter removal antibiotics [39].
A 1996 RCT by Hall and colleagues evaluated the prevention of UTI after TURP following various lengths of prophylaxis with a quinolone antibiotic (fleroxacin). The authors compared the efficacy of (1) a single perioperative oral dose, (2) a single perioperative intravenous (IV) dose, and (3) an initial perioperative IV dose followed by a daily oral dose until removal of the urinary catheter for up to 6 days. Only one patient developed a UTI (single dose IV group), which occurred 22 days postoperatively. There were no instances of urosepsis, nor was there a significant difference in rates of fever between groups. The researchers concluded that a single oral dose of a fluoroquinolone agent is adequate prophylaxis for patients undergoing TURP [40]. More recently, Jayanth and colleagues randomized patients at a single center undergoing TURP to receive a one-day or three-day course of IV amikacin as prophylaxis and evaluated the rate of bacteriuria as the primary outcome. All patients had their catheters removed on POD 3, and a midstream urine culture was obtained the following day. They found no significant difference between groups in rates of bacteriuria and symptomatic UTI up to 3 weeks after surgery. The rates of antibiotic resistance, however, were significantly greater in the group which received 3 days of antibiotics. The authors concluded that a one-day regimen is non-inferior with respect to bacteriuria and symptomatic UTI, with the added advantage of lower rates of antibiotic resistance [41].
Interestingly, a study by Conn and colleagues found no benefit from the use of any prophylactic antibiotics at the time of TURP. The 200 patients were randomized to receive a prophylactic antibiotic regimen consisting of a single dose of cephradine at the time of surgery and another dose at the time of catheter removal (3 to 4 days post-operatively), or to receive no prophylactic antibiotics at all. Between the two groups, there was no significant difference in rates of fever or UTI (defined as urine culture growth of >100,000 CFU of a single organism). The authors concluded that the short-term antibiotic regimen was not of benefit to patients in this setting [42].
Overall, investigations into the use of antibiotics in patients with indwelling catheters after TURP tend to support minimizing antibiotic use in patients without preoperative infection to a single perioperative dose and possibly a single prophylactic dose at the time of catheter removal.
These findings are likely to be applicable to other endoscopic outlet reduction procedures, such as water jet ablation, photo vaporization, and laser enucleation. However, with improved hemostasis, catheter duration and manipulation may be minimized, potentially limiting infectious risk.
3.3. Urethroplasty
Given the typically longer indwelling catheter time utilized after urethroplasty, the practice patterns of reconstructive urologists in postoperative antibiotic utilization range widely. McDonald and colleagues administered a survey regarding antimicrobial practice patterns to 34 international members of the Society of Genitourinary Reconstructive Surgeons (GURS) who commonly perform urethroplasty. 18 to 24% of respondents continue intravenous antimicrobials for longer than 24 h post-operatively. 61% administer oral antimicrobials until postoperative catheter removal, which can occur anywhere between 2 and 4 weeks), and the majority give additional antimicrobials at catheter removal [2]. A more recent online survey examining perioperative management of anterior urethroplasty patients was administered to GURS members in 2019, with 142 members responding. The majority (72.2%) of respondents reported continuing oral antimicrobials until catheter removal [3].
Manjunath et al. retrospectively examined close to 400 patients who underwent urethroplasty by a single surgeon from 2000 to 2012. All patients received preoperative antibiotic prophylaxis and postoperative prophylaxis for 30 days or until catheter removal. The investigators identified 102 (25.6%) positive urine cultures (defined as >1000 cfu/mL of an organism) within 30 days of urethroplasty—cultures were collected if there was a concern for UTI (e.g., spasms, fever). There were no significant differences in stricture recurrence (p = 0.36) or wound complications (p = 0.42) between patients who had a positive and negative urine culture. On adjusted analysis, positive urine cultures (hazard ratio 1.0, 95% confidence interval 0.6–1.8, p = 0.88) were not associated with stricture recurrence. The rates of catheter malfunction were not reported [43].
Several studies have assessed the impact of antibiotic duration. Baas and colleagues performed a retrospective review of patients who underwent urethroplasty from September 2017 to March 2020 by a single surgeon, where patients in group 1 (n = 60) received extended postoperative antibiotics for 3 weeks until catheter removal, and patients in group 2 (n = 60) received antibiotics for 3 days around catheter removal. They defined a UTI as a positive urine culture or reported lower urinary tract symptoms/fevers treated with empiric antibiotics. There was no significant difference in UTI (6.7% vs. 11.7%; p = 0.529) or wound infection rates (3.3% vs. 1.7%; p = 0.999) between the two groups [44]. Another study by Kim and colleagues also did not appreciate a difference in complications with extended antibiotics after urethroplasty. In a multi-institutional prospective study, 30-day post-operative infectious complications were evaluated in 900 patients undergoing urethroplasty or perineal urethrostomy at one of 11 centers over 2 years. Patients in the first year (cohort A) received a prolonged course of daily oral antibiotics until catheter removal, whereas those in the second year (cohort B) received antibiotics only on the day of catheter removal. They found that the rate of postoperative urinary tract infection and wound infection within 30 days was 5.1% (6.7% for cohort A vs. 3.9% for cohort B, p = 0.064) and 3.9% (4.1% for cohort A vs. 3.7% for cohort B, p = 0.772), respectively. Given these findings and concerns about the overprescribing of antibiotics, they did not recommend prolonged antibiotic use after urethroplasty [46].
A recent single-center study examined the infectious outcomes associated with the implementation of a post-urethroplasty antimicrobial administration protocol. All 81 patients were treated with intravenous antimicrobial agents until POD 2. Antibiotics were then resumed the day before the urethrogram was performed 2 to 3 weeks postoperatively. Antibiotics were then continued for another 3 to 4 days after the urethrogram. With this protocol, they found a symptomatic UTI rate of 2.5%; however, a significant limitation was that there was no control group for comparison [45].
In the setting of a negative pre-operative urine culture, existing data do not support prolonged use of antibiotics in patients with indwelling catheters who have undergone urethroplasty, nor antibiotics on the day of catheter removal only, although this is common practice. Our literature review identified considerable variability in practice, likely related to a deficient evidence base. Additional work, preferably randomized prospective studies, is needed to assess for antibiotic protocols that limit symptomatic UTI and catheter malfunction in both men and women undergoing urethral reconstruction without unnecessary doses. A summary of all articles reviewed is presented in Table 3.

Table 3.
Summary of Articles Reviewed.
4. Discussion
Decision-making regarding the use of antibiotics in the context of lower urinary tract reconstruction continues to be based on a combination of limited scientific evidence, personal experience, and inherited dogma, and accordingly, there is considerable heterogeneity in clinical practice. As it pertains to the use of antibiotics, optimizing an individual patient’s surgical outcomes and minimizing morbidity may conflict with the same goals in public health. All reconstructive urologists have received a call from a postoperative patient with an indwelling catheter reporting increased spasms, burning, debris, or gross hematuria and wondered, first and foremost, if this could have been prevented before calling in an empiric prescription for a course of antibiotics. If that urologist routinely prescribes a daily antibiotic with catheters in place, he or she may decide that daily antibiotics make little difference and stop prescribing them. On the other hand, if that urologist does not routinely prescribe antibiotics, he or she may decide to start doing so indiscriminately. Awareness of the growing crisis of MDR organisms appropriately adds to the conflict between and within urologic surgeons.
With this review, we aimed to add confidence and simplicity to clinical decision-making surrounding antibiotics and postoperative catheter management. However, what we found is that high-quality data are relatively lacking for all but the most common procedures (e.g., TURP, radical prostatectomy) and completely absent for others (e.g., bladder diverticulectomy, urethral diverticulectomy). Furthermore, no subpopulations have been identified as a group that would benefit from additional antibiotic prophylaxis. However, this is more a story of “absence of evidence” rather than “evidence of absence.”
Societal guidelines are helpful but ultimately only as good as the data on which they are based, which is, unfortunately, limited. The AUA Best Practice Statement [11] and EAU guidelines [8] may be expected to be the most relevant to antibiotic management following urologic reconstructive surgery. However, neither addresses the scenario of “precious” postoperative catheters whose malfunction could result in failed surgical repairs. Furthermore, AUA and EAU societal recommendations are not in agreement. The AUA recommends antibiotic prophylaxis at the time of catheter removal for patients at high risk of consequences from bacteremia (e.g., immunocompromised, recent joint replacement). On the other hand, the EAU weakly recommends that antibiotic prophylaxis not be administered at the time of catheter removal, balancing societal risks and patient benefits, and strongly recommends that antibiotics not be administered with indwelling catheters. The IDSA, whose experts may be most acutely aware of the dangers of the rising prevalence of MDR pathogens, recommend antibiotics to prevent infection associated with the presence, placement, or removal of catheters [7,47].
It would be reasonable to extrapolate societal recommendations to urologic reconstruction and recommend against continuous antibiotic administration with indwelling catheters and against any or at least routine use of antibiotics at the time of catheter removal. However, a close review of references on which societal recommendations are based finds that they may come from meta-analyses encompassing a fairly heterogeneous group of non-urologic procedures and even non-urinary drains [8,11]. This speaks to a relative lack of evidence specific to the field of urology and particularly urologic reconstruction. That said, we have found that for TURP, radical prostatectomy, and urethroplasty, the existing albeit limited data support societal recommendations to avoid antibiotics in the typical patient. Unfortunately, we found no randomized urethroplasty studies and no studies at all specifically evaluating antibiotic use in other forms of lower urinary tract reconstruction, such as bladder diverticulectomy or urethral diverticulectomy.
Importantly, the outcomes of existing studies center around the presence of bacteriuria or the development of symptomatic infection and do not address more surgically relevant outcomes of catheter malfunction and associated morbidity. Prevention of bacteriuria is likely not possible [7,12,14,15,19,20] and, in many patients, irrelevant. Furthermore, symptomatic infection, while uncomfortable, may not impact outcomes sufficiently to justify more widespread use of antibiotics. Urosepsis is an unfortunate but rare complication, but catheter malfunction is not; it is perhaps just harder to identify and its impact more challenging to assess. We would therefore encourage procedure-specific, prospective, randomized studies in reconstructive urology that assess the role of daily antibiotic prophylaxis and peri-catheter antibiotic administration with outcomes that include not only asymptomatic bacteriuria and symptomatic infection but also measures of catheter-associated discomfort, gross hematuria, and catheter malfunction requiring irrigation, exchange, and/or early removal.
What is also apparent is the need for non-antibiotic strategies to prevent postoperative CAUTIs and catheter malfunction. In the setting of chronic and short-term catheterization, these strategies have included antibiotic and non-antibiotic bladder irrigations [48,49,50], non-antibiotic supplements [51,52,53,54], alternative catheter materials and coatings [55,56,57,58,59,60,61,62,63,64], and microbiome alteration [65,66,67]. Table 4 summarizes these alternatives to systemic antimicrobial prophylaxis. Future studies will hopefully add to this armamentarium and identify which are beneficial to a particular subset of patients—and which add cost without sufficient benefit.

Table 4.
Alternative approaches to systemic antimicrobial prophylaxis for the prevention of UTI and CAUTI.
Limitations of our manuscript are those inherent to literature reviews, including potential author bias and failure to identify and therefore include studies relevant to the topic. We aimed to mitigate these risks with multi-author identification and review of studies for inclusion and broad search criteria. In addition, this review is limited by the relative quality of the literature, requiring liberal inclusion criteria (i.e., not limiting studies to randomized controlled trials). Nevertheless, we believe this review has value in highlighting both important findings that inform clinical decision-making and areas where deficient study warrants future research.
5. Conclusions
Antibiotic stewardship demands limiting the use of antibiotics in patients undergoing urologic procedures who require indwelling catheterization for >24 h. Limited data in patients following TURP, radical prostatectomy, and urethroplasty suggests that, in general, avoidance of continuous prophylaxis does not result in a greater risk of symptomatic infection. A single dose of antibiotics at the time of catheter removal may be warranted in some patients to prevent infection. However, this population has yet to be defined. Opportunities for improvement in future study design include additional outcome variables that improve our understanding of the impact of catheter malfunction on outcomes and the impact of antibiotics on catheter malfunction. Given the concerning rise in the prevalence of MDR pathogens and the association of MDR organisms with systemic antibiotic administration, the development of non-antibiotic strategies is paramount.
Author Contributions
Literature search, acquisition of data, and analysis: F.M. and C.C.; Writing of draft: F.M., C.C., J.S. and J.A.C.; Concept and design: J.A.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were generated in the writing of this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wazait, H.; van der Meullen, J.; Patel, H.; Brown, C.; Gadgil, S.; Miller, R.; Kelsey, M.; Emberton, M. Antibiotics on urethral catheter withdrawal: A hit and miss affair. J. Hosp. Infect. 2004, 58, 297–302. [Google Scholar] [CrossRef] [PubMed]
- McDonald, M.L.; Buckley, J. Antimicrobial Practice Patterns for Urethroplasty: Opportunity for Improved Stewardship. Urology 2016, 94, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Hoare, D.T.; Doiron, R.C.; Rourke, K.F. Determining Perioperative Practice Patterns in Urethroplasty: A Survey of Genitourinary Reconstructive Surgeons. Urology 2021, 156, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Köves, B.; Tenke, P.; Tandogdu, Z.; Cai, T.; Bogenhard, F.; Wullt, B.; Naber, K.; Bartoletti, R.; Cek, M.; Kulchavenya, E.; et al. Transurethral Resection of the Prostate: Are We Following the Guidelines?—Outcomes from the Global Prevalence of Infections in Urology (GPIU) Study. J. Chemother. 2019, 31, 15–22. [Google Scholar] [CrossRef]
- Umscheid, C.A.; Mitchell, M.D.; Doshi, J.A.; Agarwal, R.; Williams, K.; Brennan, P.J. Estimating the Proportion of Healthcare-Associated Infections That Are Reasonably Preventable and the Related Mortality and Costs. Infect. Control. Hosp. Epidemiol. 2011, 32, 101–114. [Google Scholar] [CrossRef]
- Gould, C.V.; Umscheid, C.A.; Agarwal, R.K.; Kuntz, G.; Pegues, D.A. Healthcare Infection Control Practices Advisory Committee. Guideline for prevention of catheter-associated urinary tract infections 2009. Infect. Control. Hosp. Epidemiol. 2010, 31, 319–326. [Google Scholar] [CrossRef]
- Hooton, T.M.; Bradley, S.F.; Cardenas, D.D.; Colgan, R.; Geerlings, S.E.; Rice, J.C.; Saint, S.; Schaeffer, A.J.; Tambayh, P.A.; Tenke, P.; et al. Diagnosis, prevention, and treatment of catheter-aassociated urinary tract infection in adults: 2009 international clinical practice guidelines from the infectious diseases society of America. Clin. Infect. Dis. 2010, 50, 625–663. [Google Scholar] [CrossRef]
- Bonkat, G.; Bartoletti, R.; Bruyère, F.; Cai, T.; Geerlings, S.E.; Köves, B.; Schubert, S.; Pilatz, A.; Veeratterapillay, R.; Wagenlehner, F.; et al. EAU Guidelines on Urological Infections. Available online: https://uroweb.org/guidelines/urological-infections (accessed on 31 December 2022).
- Lusardi, G.; Lipp, A.; Shaw, C. Antibiotic prophylaxis for short-term catheter bladder drainage in adults. Cochrane Database Syst. Rev. 2013, 2013, CD005428. [Google Scholar] [CrossRef]
- Niël-Weise, B.S.; van den Broek, P.J.; da Silva, E.M.; Silva, L.A. Urinary catheter policies for long-term bladder drainage. Cochrane Database Syst. Rev. 2012, 2012, CD004201. [Google Scholar] [CrossRef]
- Lightner, D.J.; Wymer, K.; Sanchez, J.; Kavoussi, L. Best Practice Statement on Urologic Procedures and Antimicrobial Prophylaxis. J. Urol. 2020, 203, 351–356. [Google Scholar] [CrossRef]
- E Nicolle, L.; Gupta, K.; Bradley, S.F.; Colgan, R.; DeMuri, G.P.; Drekonja, D.; O Eckert, L.; E Geerlings, S.; Köves, B.; Hooton, T.M.; et al. Clinical practice guideline for the management of asymptomatic bacteriuria: 2019 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2019, 68, e83–e110. [Google Scholar] [CrossRef] [PubMed]
- Bonkat, G.; Widmer, A.F.; Rieken, M.; Van Der Merwe, A.; Braissant, O.; Müller, G.; Wyler, S.; Frei, R.; Gasser, T.C.; Bachmann, A. Microbial biofilm formation and catheter-associated bacteriuria in patients with suprapubic catheterisation. World J. Urol. 2013, 31, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Tambyah, P.A. Catheter-associated urinary tract infections: Diagnosis and prophylaxis. Int. J. Antimicrob. Agents 2004, 24, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Sedor, J.; Mulholland, S.G. Hospital-acquired urinary tract infections associated with the indwelling catheter. Urol. Clin. N. Am. 1999, 26, 821–828. [Google Scholar] [CrossRef]
- Hidron, A.; Edwards, J.; Patel, J.; Horan, T. Update: Antimicrobial-resistant pathogens associated with healthcare-associated infections: Annual summary of data reported to the National Healthcare. Infect. Control. Hosp. 2008, 29, 996–1011. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Stickler, D.J. Bacterial biofilms in patients with indwelling urinary catheters. Nat. Rev. Endocrinol. 2008, 5, 598–608. [Google Scholar] [CrossRef]
- Saint, S. Clinical and economic consequences of nosocomial catheter-related bacteriuria. Am. J. Infect. Control. 2000, 28, 68–75. [Google Scholar] [CrossRef]
- Tambyah, P.A.; Maki, D.G. Catheter-associated urinary tract infection is rarely symptomatic: A prospective study of 1,497 catheterized patients. Arch. Intern. Med. 2000, 160, 678–682. [Google Scholar] [CrossRef]
- Platt, R.; Polk, B.F.; Murdock, B.; Rosner, B. Mortality Associated with Nosocomial Urinary-Tract Infection. N. Engl. J. Med. 1982, 307, 637–642. [Google Scholar] [CrossRef]
- Healthcare-associated Infections (HAI). Preventing HAIs: Urine Culture Stewardship. CDC. Available online: https://www.cdc.gov/hai/prevent/cauti/indwelling/overview.html#anchor_1554900744. (accessed on 31 December 2022).
- National Healthcare Safety Network. Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CAUTI] and Non-Catheter-Associated Urinary Tract Infection [UTI]) Events. CDC. Available online: https://www.cdc.gov/nhsn/pdfs/pscmanual/7psccauticurrent.pdf (accessed on 31 December 2022).
- Anger, J.; Lee, U.; Ackerman, A.L.; Chou, R.; Chughtai, B.; Clemens, J.Q.; Hickling, D.; Kapoor, A.; Kenton, K.S.; Kaufman, M.R.; et al. Recurrent Uncomplicated Urinary Tract Infections in Women: AUA/CUA/SUFU Guideline. J. Urol. 2019, 202, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Lane, G.; Powell, C.R. AUA Core Curriculum: Urinary Tract Infection (Adult). AUA. 2022. Available online: https://university.auanet.org/core/core.cfm?sectionID=92&ct=99a81fed6e38ad2f04e4b0e04028f76aed79ba47dbecfb7a06a5bf4014c951ca9e5444e2e354e37ca2eae0d1a96a0750452c4d0298b9ff2fd5b0c917263f1fbb&ct=7ffaf6ad57e97a99a898c3c75a0fa59fef30890ae25b5460f00fe50b9d9399321e49fd5e62ad89cce232106e0d82b356cfdfad647adb0d7e79c710bb928be21e#ref_4391 (accessed on 31 December 2022).
- Umscheid, C.A.; Agarwal, R.K.; Brennan, P.J. Updating the guideline development methodology of the Healthcare Infection Control Practices Advisory Committee (HICPAC). Am. J. Infect. Control. 2010, 38, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Givens, C.D.; Wenzel, R.P. Catheter-associated urinary tract infections in surgical patients: A controlled study on the excess morbidity and costs. J. Urol. 1980, 124, 646–648. [Google Scholar] [CrossRef]
- Klevens, R.M.; Edwards, J.R.; Richards, C.L., Jr.; Horan, T.C.; Gaynes, R.P.; Pollock, D.A.; Cardo, D.M. Estimating health care-associated infections and deaths in U.S. Hospitals, 2002. Public Health Rep. 2007, 122, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Enzler, M.J.; Berbari, E.; Osmon, D.R. Antimicrobial prophylaxis in adults. Mayo Clin. Proc. 2011, 86, 686–701. [Google Scholar] [CrossRef]
- Tenke, P.; Kovacs, B.; Bjerklund Johansen, T.E.; Matsumoto, T.; Tambyah, P.A.; Naber, K.G. European and Asian guidelines on management and prevention of catheter-associated urinary tract infections. Int. J. Antimicrob. Agents 2008, 31 (Suppl. S1), S68–S78. [Google Scholar] [CrossRef]
- Baethge, C.; Goldbeck-Wood, S.; Mertens, S. SANRA—A scale for the quality assessment of narrative review articles. Res. Integr. Peer Rev. 2019, 4, 5. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Banks, J.A.; McGuire, B.B.; Loeb, S.; Shrestha, S.; Helfand, B.T.; Catalona, W.J. Bacteriuria and antibiotic resistance in catheter urine specimens following radical prostatectomy. Urol. Oncol. Semin. Orig. Investig. 2013, 31, 1049–1053. [Google Scholar] [CrossRef]
- Shin, B.; Chung, H.S.; Hwang, E.C.; Jung, S.; Kwon, D.D. Antibiotic prophylaxis in radical prostatectomy: Comparison of 2-day and more than 2-day prophylaxis. J. Korean Med. Sci. 2017, 32, 1009–1015. [Google Scholar] [CrossRef]
- Pinochet, R.; Nogueira, L.; Cronin, A.M.; Katz, D.; Rabbani, F.; Guillonneau, B.; Touijer, K. Role of short-term antibiotic therapy at the moment of catheter removal after laparoscopic radical prostatectomy. Urol. Int. 2010, 85, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Haifler, M.; Mor, Y.; Dotan, Z.; Ramon, J.; Zilberman, D.E. Prophylactic antibiotic treatment following laparoscopic robot-assisted radical prostatectomy for the prevention of catheter-associated urinary tract infections: Did the AUA guidelines make a difference? J. Robot. Surg. 2017, 11, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Berrondo, C.; Feng, C.; Kukreja, J.B.; Messing, E.M.; Joseph, J. Antibiotic prophylaxis at the time of catheter removal after radical prostatectomy: A prospective randomized clinical trial. Urol. Oncol. Semin. Orig. Investig. 2019, 37, 181.e7–181.e14. [Google Scholar] [CrossRef] [PubMed]
- Ehdaie, B.; Jibara, G.; Sjoberg, D.D.; Laudone, V.; Eastham, J.; Touijer, K.; Scardino, P.; Donahue, T.; Goh, A.; Vickers, A. The Duration of Antibiotics Prophylaxis at the Time of Catheter Removal after Radical Prostatectomy: Clinically Integrated, Cluster, Randomized Trial. J. Urol. 2021, 206, 662–668. [Google Scholar] [CrossRef]
- Raz, R.; Elhanan, G.; Almog, D.; Shental, J. The use of ceftriaxone in the prevention of urinary tract infection in patients undergoing transurethral resection of the prostate (TUR-P). Infection 1994, 22, 347–349. [Google Scholar] [CrossRef]
- Hall, J.; Christiansen, K.; England, P.; Low, A.; McRae, P.; Mander, J.; Taylor, T. Antibiotic prophylaxis for patients undergoing transurethral resection of the prostate. Urology 1996, 47, 852–856. [Google Scholar] [CrossRef]
- Chandrasingh, J.; Jayanth, S.T.; Sahni, R.D.; Mukha, R.P.; Kumar, S.; Devasia, A.; Kekre, N.S. Efficacy of 1 versus 3 days of intravenous amikacin as a prophylaxis for patients undergoing transurethral resection of the prostate: A prospective randomized trial. Indian J. Urol. 2021, 37, 133–139. [Google Scholar] [CrossRef]
- Conn, I.G.; Moffat, L.E.F. Short-term cephradine prophylaxis in elective transurethral prostatectomy. J. Hosp. Infect. 1988, 11, 373–375. [Google Scholar] [CrossRef]
- Manjunath, A.; Chen, L.; Welty, L.J.; Wong, V.J.; Amarasekera, C.; Gonzalez, C.M.; Hofer, M.D. Antibiotic prophylaxis after urethroplasty may offer no benefit. World J. Urol. 2020, 38, 1295–1301. [Google Scholar] [CrossRef]
- Baas, W.; Parker, A.; Radadia, K.; Ogawa, S.; Vetter, J.; Paradis, A.; Meyer, M.; Murphy, G. Antibiotic Duration After Urethroplasty: An Attempt at Improving Antibiotic Stewardship. Urology 2021, 158, 228–231. [Google Scholar] [CrossRef]
- Hanasaki, T.; Kanematsu, A.; Yamamoto, S. Proactive discontinuation of postoperative antibiotic prophylaxis after urethroplasty. Int. J. Urol. 2022, 29, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Cheng, K.C.; Alsikafi, N.F.; Breyer, B.N.; Broghammer, J.A.; Elliott, S.P.; Erickson, B.A.; Myers, J.B.; Smith, T.G.; Vanni, A.J.; et al. Minimizing Antibiotic Use in Urethral Reconstruction. J. Urol. 2022, 208, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Bratzler, D.W.; Dellinger, E.P.; Olsen, K.M.; Perl, T.M.; Auwaerter, P.G.; Bolon, M.K.; Fish, D.N.; Napolitano, L.M.; Sawyer, R.G.; Slain, D.; et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am. J. Health-Syst. Pharm. 2013, 70, 195–283. [Google Scholar] [CrossRef]
- Andretta, E.; Longo, R.; Balladelli, M.; Sgarabotto, C.; Sgarabotto, D. Intravesical Gentamicin: An Option for Therapy and Prophylaxis against Recurrent UTIs and Resistant Bacteria in Neurogenic Bladder Patients on Intermittent Catheterization. Antibiotics 2022, 11, 1335. [Google Scholar] [CrossRef] [PubMed]
- Cox, L.; He, C.; Bevins, J.; Clemens, J.Q.; Stoffel, J.T.; Cameron, A.P. Gentamicin bladder instillations decrease symptomatic urinary tract infections in neurogenic bladder patients on intermittent catheterization. Can. Urol. Assoc. J. 2017, 11, E350–E354. [Google Scholar] [CrossRef] [PubMed]
- Pietropaolo, A.; Jones, P.; Moors, M.; Birch, B.; Somani, B.K. Use and effectiveness of antimicrobial intravesical treatment for prophylaxis and treatment of recurrent urinary tract infections (UTIs): A systematic review. Curr. Urol. Rep. 2018, 19. [Google Scholar] [CrossRef]
- Cotellese, R.; Ledda, A.; Belcaro, G.; Cesarone, M.R.; Scipione, C.; Scipione, V.; Dugall, M.; Feragalli, B.; Riva, A.; Allegrini, P.; et al. Anthocran® Phytosome®: Prevention of Recurring Urinary Infections and Symptoms after Catheterization. J. Diet. Suppl. 2021, 20, 55–67. [Google Scholar] [CrossRef]
- Lee, B.; Haran, M.J.; Hunt, L.M.; Simpson, J.M.; Marial, O.; Rutkowski, S.B.; Middleton, J.; Kotsiou, G.; Tudehope, M.; Cameron, I.D. Spinal-injured neuropathic bladder antisepsis (SINBA) trial. Spinal Cord 2007, 45, 542–550. [Google Scholar] [CrossRef]
- Falagas, M.E.; Betsi, G.I.; Tokas, T.; Athanasiou, S. Probiotics for Prevention of Recurrent Urinary Tract Infections in Women. Drugs 2006, 66, 1253–1261. [Google Scholar] [CrossRef]
- Toh, S.-L.; Lee, B.B.; Ryan, S.; Simpson, J.M.; Clezy, K.; Bossa, L.; Rice, S.A.; Marial, O.; Weber, G.H.; Kaur, J.; et al. Probiotics [LGG-BB12 or RC14-GR1] versus placebo as prophylaxis for urinary tract infection in persons with spinal cord injury [ProSCIUTTU]: A randomised controlled trial. Spinal Cord 2019, 57, 550–561. [Google Scholar] [CrossRef]
- Lam, T.B.L.; Omar, M.I.; Fisher, E.; Gillies, K.; Maclennan, S. Types of indwelling urethral catheters for short-term catheterisation in hospitalised adults. Cochrane Database Syst. Rev. 2014, 2014, CD004013. [Google Scholar] [CrossRef] [PubMed]
- Talja, M.; Korpela, A.; Järvi, K. Comparison of Urethral Reaction to Full Silicone, Hydrogen-coated and Siliconised Latex Catheters. Br. J. Urol. 1990, 66, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Kazmierska, K.A.; Thompson, R.; Morris, N.; Long, A.; Ciach, T. In vitro multicompartmental bladder model for assessing blockage of urinary catheters: Effect of hydrogel coating on dynamics of proteus mirabilis growth. Urology 2010, 76, e15–e20. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, S.; Keatch, R.; Corner, G.; Nabi, G.; Murdoch, S.; Davidson, F.; Zhao, Q. In-vitro antibacterial and anti-encrustation performance of silver-polytetrafluoroethylene nanocomposite coated urinary catheters. J. Hosp. Infect. 2019, 103, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Pickard, R.; Lam, T.; MacLennan, G.; Starr, K.; Kilonzo, M.; McPherson, G.; Gillies, K.; McDonald, A.; Walton, K.; Buckley, B.; et al. Types of urethral catheter for reducing symptomatic urinary tract infections in hospitalised adults requiring short-term catheterisation: Multicentre randomised controlled trial and economic evaluation of antimicrobial- and antisepticimpregnated urethral catheters (the CATHETER trial). Health Technol. Assess. 2012, 16, 1–197. [Google Scholar] [CrossRef] [PubMed]
- Kunin, C.M.; Chin, Q.F.; Chambers, S. Formation of encrustations on indwelling urinary catheters in the elderly: A comparison of different types of catheter materials in “blockers” and “nonblockers” . J. Urol. 1987, 138, 899–902. [Google Scholar] [CrossRef]
- Karchmer, T.B.; Giannetta, E.T.; Muto, C.A.; Strain, B.A.; Farr, B.M. A randomized crossover study of silver-coated urinary catheters in hospitalized patients. Arch. Intern. Med. 2000, 160, 3294–3298. [Google Scholar] [CrossRef]
- Lai, K.K.; Fontecchio, S.A. Use of silver-hydrogel urinary catheters on the incidence of catheter-associated urinary tract infections in hospitalized patients. Am. J. Infect. Control. 2002, 30, 221–225. [Google Scholar] [CrossRef]
- Bonfill, X.; Rigau, D.; Esteban-Fuertes, M.; Barrera-Chacón, J.M.; Jáuregui-Abrisqueta, M.L.; Salvador, S.; Alemán-Sánchez, C.M.; Borau, A.; Bea-Muñoz, M.; Hidalgo, B.; et al. Efficacy and safety of urinary catheters with silver alloy coating in patients with spinal cord injury: A multicentric pragmatic randomized controlled trial. The ESCALE trial. Spine J. 2017, 17, 1650–1657. [Google Scholar] [CrossRef]
- Bonfill, X.; Rigau, D.; Esteban-Fuertes, M.; Barrera-Chacón, J.M.; Jáuregui-Abrisqueta, M.L.; Salvador, S.; Alemán-Sánchez, C.M.; Borau, A.; Bea-Muñoz, M.; Hidalgo, B.; et al. Bioengineered phytomolecules-capped silver nanoparticles using Carissa carandas leaf extract to embed on to urinary catheter to combat UTI pathogens. PLoS ONE 2021, 16, e0256748. [Google Scholar] [CrossRef]
- Thomas-White, K.J.; Gao, X.; Lin, H.; Fok, C.S.; Ghanayem, K.; Mueller, E.R.; Dong, Q.; Brubaker, L.; Wolfe, A.J. Urinary microbes and postoperative urinary tract infection risk in urogynecologic surgical patients. Int. Urogynecol. J. 2018, 29, 1797–1805. [Google Scholar] [CrossRef] [PubMed]
- Groah, S.L.; Pérez-Losada, M.; Caldovic, L.; Ljungberg, I.H.; Sprague, B.M.; Castro-Nallar, E.; Chandel, N.J.; Hsieh, M.H.; Pohl, H. Redefining Healthy Urine: A Cross-Sectional Exploratory Metagenomic Study of People With and Without Bladder Dysfunction. J. Urol. 2016, 196, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Darouiche, R.O.; Green, B.G.; Donovan, W.H.; Chen, D.; Schwartz, M.; Merritt, J.; Mendez, M.; Hull, R.A. Multicenter randomized controlled trial of bacterial interference for prevention of urinary tract infection in patients with neurogenic bladder. Urology 2011, 78, 341–346. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).