In Vitro Assessment of Antifungal and Antibiofilm Efficacy of Commercial Mouthwashes against Candida albicans
Abstract
:1. Introduction
2. Results
2.1. Antimicrobial Activity (MIC/MFC)
2.2. Antibiofilm Activity
3. Discussion
4. Materials and Methods
4.1. Mouthwashes
4.2. Fungal Strains
4.3. Minimal Inhibitory Concentrations (MICs)
4.4. Minimal Fungicidal Concentrations (MFCs)
4.5. MFC/MIC Ratio
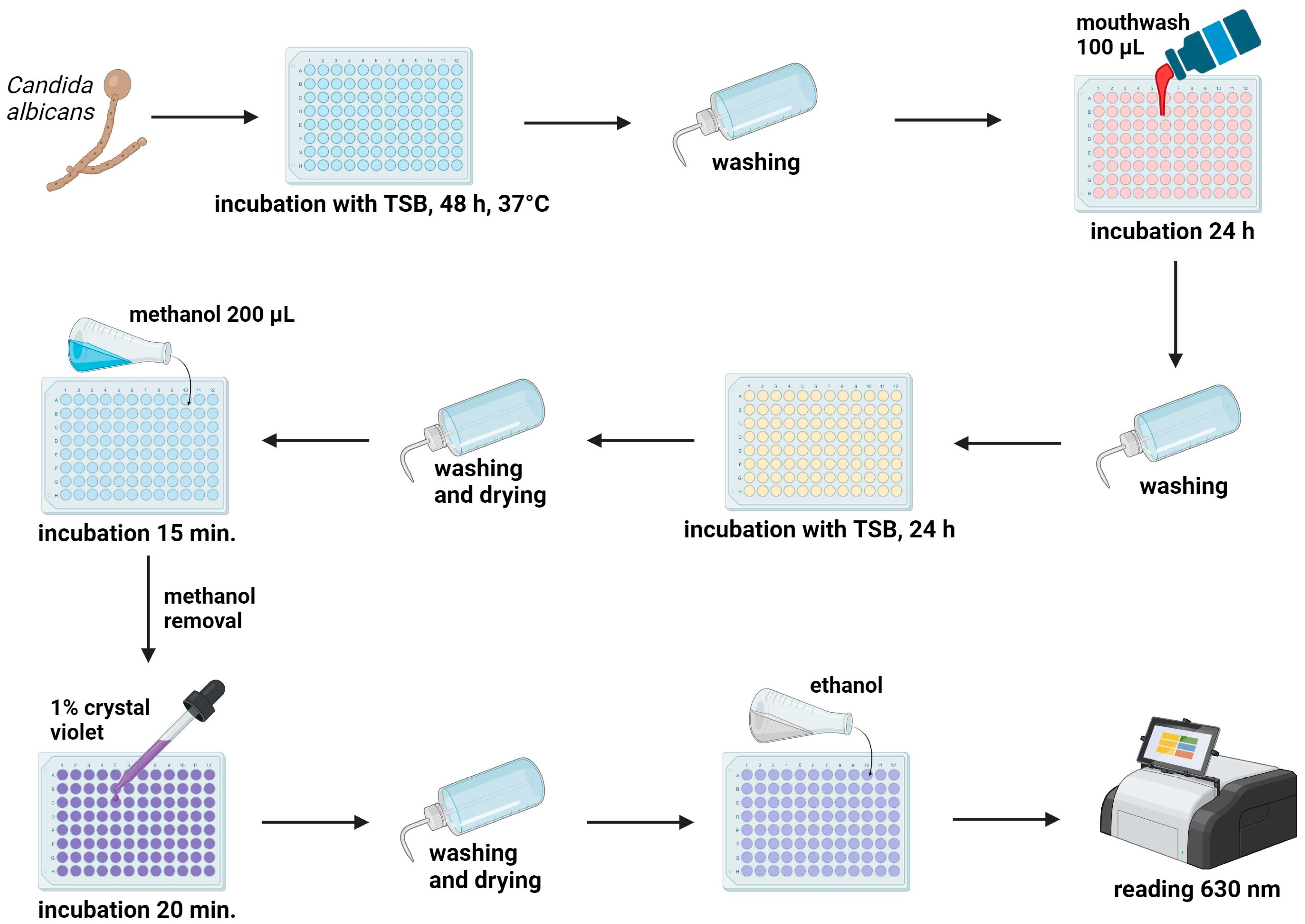
4.6. Anti-Biofilm Activity Test
4.7. Statistics
5. Conclusions
- Among 15 commercial mouthwashes, those containing OCT, CHX, or CHX + CPC demonstrate the most effective activity (MIC, MFC) against Candida albicans. Products with PHMB or BAC also exhibit good antifungal action.
- Mouthwashes containing OCT display the most potent activity against Candida biofilm. Products with CHX, PHMB, BAC, Scutellaria extract, and fluorides + essential oils show a moderate antibiofilm effect.
- Considering the in vitro activity against Candida albicans, it can be inferred that, clinically, mouthwashes containing OCT are likely to offer the highest effectiveness. Meanwhile, products containing CHX, PHMB, or BAC can be considered as promising alternatives.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Talapko, J.; Juzbašić, M.; Matijević, T.; Pustijanac, E.; Bekić, S.; Kotris, I.; Škrlec, I. Candida Albicans-The Virulence Factors and Clinical Manifestations of Infection. J. Fungi 2021, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Drgona, L.; Khachatryan, A.; Stephens, J.; Charbonneau, C.; Kantecki, M.; Haider, S.; Barnes, R. Clinical and Economic Burden of Invasive Fungal Diseases in Europe: Focus on Pre-Emptive and Empirical Treatment of Aspergillus and Candida Species. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 7–21. [Google Scholar] [CrossRef]
- Brown, G.D.; Denning, D.W.; Gow, N.A.R.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden Killers: Human Fungal Infections. Sci. Transl. Med. 2012, 4, 165rv13. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.-Y. Oral Candidosis: Pathophysiology and Best Practice for Diagnosis, Classification, and Successful Management. J. Fungi 2021, 7, 555. [Google Scholar] [CrossRef] [PubMed]
- Grzegocka, K.; Krzyściak, P.; Hille-Padalis, A.; Loster, J.E.; Talaga-Ćwiertnia, K.; Loster, B.W. Candida Prevalence and Oral Hygiene Due to Orthodontic Therapy with Conventional Brackets. BMC Oral Health 2020, 20, 277. [Google Scholar] [CrossRef] [PubMed]
- Martorano-Fernandes, L.; Dornelas-Figueira, L.M.; Marcello-Machado, R.M.; Silva, R.d.B.; Magno, M.B.; Maia, L.C.; Del Bel Cury, A.A. Oral Candidiasis and Denture Stomatitis in Diabetic Patients: Systematic Review and Meta-Analysis. Braz. Oral Res. 2020, 34, e113. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.a.O.; Williams, D.W. Diagnosis and Management of Oral Candidosis. Br. Dent. J. 2017, 223, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.; Seneviratne, C.J.; Koga-Ito, C.Y.; Samaranayake, L.P. Is the Oral Fungal Pathogen Candida Albicans a Cariogen? Oral Dis. 2018, 24, 518–526. [Google Scholar] [CrossRef]
- Jepsen, K.; Falk, W.; Brune, F.; Cosgarea, R.; Fimmers, R.; Bekeredjian-Ding, I.; Jepsen, S. Prevalence and Antibiotic Susceptibility Trends of Selected Enterobacteriaceae, Enterococci, and Candida Albicans in the Subgingival Microbiota of German Periodontitis Patients: A Retrospective Surveillance Study. Antibiotics 2022, 11, 385. [Google Scholar] [CrossRef]
- Stasiewicz, M.; Karpiński, T.M. The Oral Microbiota and Its Role in Carcinogenesis. Semin. Cancer Biol. 2022, 86, 633–642. [Google Scholar] [CrossRef]
- Naglik, J.R.; Gaffen, S.L.; Hube, B. Candidalysin: Discovery and Function in Candida Albicans Infections. Curr. Opin. Microbiol. 2019, 52, 100–109. [Google Scholar] [CrossRef]
- Staniszewska, M. Virulence Factors in Candida Species. Curr. Protein Pept. Sci. 2020, 21, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.; Dos Santos Fontenelle, R.O.; de Brito, E.H.S.; de Morais, S.M. Biofilm of Candida Albicans: Formation, Regulation and Resistance. J. Appl. Microbiol. 2021, 131, 11–22. [Google Scholar] [CrossRef]
- Babalska, Z.Ł.; Korbecka-Paczkowska, M.; Karpiński, T.M. Wound Antiseptics and European Guidelines for Antiseptic Application in Wound Treatment. Pharmaceuticals 2021, 14, 1253. [Google Scholar] [CrossRef]
- Sopata, M.; Jawień, A.; Mrozikiewicz-Rakowska, B.; Augusewicz, Z.; Bakowska, M.; Samson, I.; Gabriel, M.; Grzela, T.; Karpiński, T.; Kuberka, I.; et al. Guidelines for Topical Management in Non-Infected, at Risk of Infection and Infected Wounds-an Overview of Available Antimicrobial Substances Used in the Treatment of Wounds. Recommendations of the Polish Wound Treatment Society. Leczenie. Ran. 2020, 17, 1–21. [Google Scholar] [CrossRef]
- Barbosa, A.H.; Damasceno, J.L.; Casemiro, L.A.; Martins, C.H.G.; Pires, R.H.; Candido, R.C. Susceptibility to Oral Antiseptics and Virulence Factors Ex Vivo Associated with Candida Spp. Isolated from Dental Prostheses. J. Prosthodont. 2019, 28, 398–408. [Google Scholar] [CrossRef] [PubMed]
- Vlachojannis, C.; Chrubasik-Hausmann, S.; Hellwig, E.; Al-Ahmad, A. A Preliminary Investigation on the Antimicrobial Activity of Listerine®, Its Components, and of Mixtures Thereof. Phytother. Res. 2015, 29, 1590–1594. [Google Scholar] [CrossRef]
- Ramage, G.; Jose, A.; Coco, B.; Rajendran, R.; Rautemaa, R.; Murray, C.; Lappin, D.F.; Bagg, J. Commercial Mouthwashes Are More Effective than Azole Antifungals against Candida Albicans Biofilms in Vitro. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 111, 456–460. [Google Scholar] [CrossRef]
- Ardizzoni, A.; Pericolini, E.; Paulone, S.; Orsi, C.F.; Castagnoli, A.; Oliva, I.; Strozzi, E.; Blasi, E. In Vitro Effects of Commercial Mouthwashes on Several Virulence Traits of Candida Albicans, Viridans Streptococci and Enterococcus Faecalis Colonizing the Oral Cavity. PLoS ONE 2018, 13, e0207262. [Google Scholar] [CrossRef]
- Černáková, L.; Jordao, L.; Bujdáková, H. Impact of Farnesol and Corsodyl® on Candida Albicans Forming Dual Biofilm with Streptococcus Mutans. Oral Dis. 2018, 24, 1126–1131. [Google Scholar] [CrossRef]
- Katagiri, H.; Stuck, N.-J.; Arakawa, I.; Nietzsche, S.; Eick, S. In Vitro Activity of Oral Health Care Products on Candida Biofilm Formation. Monogr. Oral Sci. 2021, 29, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Wei, P.; Zhao, C.; He, C.; Yan, Z.; Hua, H. In Vitro Antifungal Effect and Inhibitory Activity on Biofilm Formation of Seven Commercial Mouthwashes. Oral Dis. 2014, 20, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Paulone, S.; Malavasi, G.; Ardizzoni, A.; Orsi, C.F.; Peppoloni, S.; Neglia, R.G.; Blasi, E. Candida Albicans Survival, Growth and Biofilm Formation Are Differently Affected by Mouthwashes: An in Vitro Study. New Microbiol. 2017, 40, 45–52. [Google Scholar]
- Di Lodovico, S.; Dotta, T.C.; Cellini, L.; Iezzi, G.; D’Ercole, S.; Petrini, M. The Antibacterial and Antifungal Capacity of Eight Commercially Available Types of Mouthwash against Oral Microorganisms: An In Vitro Study. Antibiotics 2023, 12, 675. [Google Scholar] [CrossRef] [PubMed]
- Koburger, T.; Hübner, N.-O.; Braun, M.; Siebert, J.; Kramer, A. Standardized Comparison of Antiseptic Efficacy of Triclosan, PVP-Iodine, Octenidine Dihydrochloride, Polyhexanide and Chlorhexidine Digluconate. J. Antimicrob. Chemother. 2010, 65, 1712–1719. [Google Scholar] [CrossRef] [PubMed]
- Tirali, R.E.; Turan, Y.; Akal, N.; Karahan, Z.C. In Vitro Antimicrobial Activity of Several Concentrations of NaOCl and Octenisept in Elimination of Endodontic Pathogens. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2009, 108, e117–e120. [Google Scholar] [CrossRef] [PubMed]
- Moroz, J.; Kurnatowski, P. The in Vitro Activity of Selected Mouthrinses on Standard Strains of Fungi. Ann. Parasitol. 2017, 63, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.L.; Lim, L.Y.; Hammer, K.; Hettiarachchi, D.; Locher, C. A Review of Commonly Used Methodologies for Assessing the Antibacterial Activity of Honey and Honey Products. Antibiotics 2022, 11, 975. [Google Scholar] [CrossRef]
- Krasowski, G.; Junka, A.; Paleczny, J.; Czajkowska, J.; Makomaska-Szaroszyk, E.; Chodaczek, G.; Majkowski, M.; Migdał, P.; Fijałkowski, K.; Kowalska-Krochmal, B.; et al. In Vitro Evaluation of Polihexanide, Octenidine and NaClO/HClO-Based Antiseptics against Biofilm Formed by Wound Pathogens. Membranes 2021, 11, 62. [Google Scholar] [CrossRef]
- de Oliveira, J.R.; Belato, K.K.; de Oliveira, F.E.; Jorge, A.O.C.; Camargo, S.E.A.; de Oliveira, L.D. Mouthwashes: An in Vitro Study of Their Action on Microbial Biofilms and Cytotoxicity to Gingival Fibroblasts. Gen. Dent. 2018, 66, 28–34. [Google Scholar]
- Kollmuss, M.; Tolksdorf, K.; Wuersching, S.N.; Hickel, R.; Huth, K.C. Effect of Polyhexanide as Antiseptic Mouth Rinse against Oral Pathogens in an in Vitro Biofilm Model. Acta Odontol. Scand. 2021, 79, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Dudek-Wicher, R.; Junka, A.F.; Migdał, P.; Korzeniowska-Kowal, A.; Wzorek, A.; Bartoszewicz, M. The Antibiofilm Activity of Selected Substances Used in Oral Health Prophylaxis. BMC Oral Health 2022, 22, 509. [Google Scholar] [CrossRef] [PubMed]
- Alem, M.A.S.; Douglas, L.J. Effects of Aspirin and Other Nonsteroidal Anti-Inflammatory Drugs on Biofilms and Planktonic Cells of Candida Albicans. Antimicrob. Agents Chemother. 2004, 48, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Lyu, X.; Zhao, C.; Yan, Z.-M.; Hua, H. Efficacy of Nystatin for the Treatment of Oral Candidiasis: A Systematic Review and Meta-Analysis. Drug Des. Dev. Ther. 2016, 10, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Sabanceva, E.G.; Dmitrieva, N.A.; Avramenko, E.A.; Ivanova, E.V.; Ezhova, E.G.; Pochivalin, V.P. Evaluation of effectiveness of a mouthwash containing antiseptic octenidine dihydrochloride. Stomatologiia 2021, 100, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Karpiński, T.M.; Szkaradkiewicz, A.K. Chlorhexidine--Pharmaco-Biological Activity and Application. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 1321–1326. [Google Scholar] [PubMed]
- Tartaglia, G.M.; Tadakamadla, S.K.; Connelly, S.T.; Sforza, C.; Martín, C. Adverse Events Associated with Home Use of Mouthrinses: A Systematic Review. Ther. Adv. Drug Saf. 2019, 10, 2042098619854881. [Google Scholar] [CrossRef]
- Lachapelle, J.-M. A Comparison of the Irritant and Allergenic Properties of Antiseptics. Eur. J. Dermatol. 2014, 24, 3–9. [Google Scholar] [CrossRef]
- Kramer, A.; Dissemond, J.; Kim, S.; Willy, C.; Mayer, D.; Papke, R.; Tuchmann, F.; Assadian, O. Consensus on Wound Antisepsis: Update 2018. Ski. Pharmacol. Physiol. 2018, 31, 28–58. [Google Scholar] [CrossRef]
- Weber, J.; Bonn, E.L.; Auer, D.L.; Kirschneck, C.; Buchalla, W.; Scholz, K.J.; Cieplik, F. Preprocedural Mouthwashes for Infection Control in Dentistry—An Update. Clin. Oral Investig. 2023, 27, 33–44. [Google Scholar] [CrossRef]
- Fromm-Dornieden, C.; Rembe, J.-D.; Schäfer, N.; Böhm, J.; Stuermer, E.K. Cetylpyridinium Chloride and Miramistin as Antiseptic Substances in Chronic Wound Management—Prospects and Limitations. J. Med. Microbiol. 2015, 64, 407–414. [Google Scholar] [CrossRef]
- Jockel-Schneider, Y.; Schlagenhauf, U.; Petsos, H.; Rüttermann, S.; Schmidt, J.; Ziebolz, D.; Wehner, C.; Laky, M.; Rott, T.; Noack, M.; et al. Impact of 0.1% Octenidine Mouthwash on Plaque Re-Growth in Healthy Adults: A Multi-Center Phase 3 Randomized Clinical Trial. Clin. Oral Investig. 2021, 25, 4681–4689. [Google Scholar] [CrossRef] [PubMed]
- Karpiński, T.M.; Ożarowski, M.; Seremak-Mrozikiewicz, A.; Wolski, H. Anti-Candida and Antibiofilm Activity of Selected Lamiaceae Essential Oils. Front. Biosci. (Landmark Ed.) 2023, 28, 28. [Google Scholar] [CrossRef] [PubMed]
- Konstantinovitch, K.Y.; Arsene, M.M.J.; Aliya, M.V.; Viktorovna, P.I.; Elena, V.G.; Azova, M.M.; Amira, A.A. Assessment of Antimicrobial Activity of Ethanolic and Aqueous Extracts of Aesculus hippocastanum L. (Horse Chestnut) Bark against Bacteria Isolated from Urine of Patients Diagnosed Positive to Urinary Tract Infections. Front. Biosci. (Schol. Ed.) 2022, 14, 11. [Google Scholar] [CrossRef] [PubMed]
- Długaszewska, J.; Leszczynska, M.; Lenkowski, M.; Tatarska, A.; Pastusiak, T.; Szyfter, W. The Pathophysiological Role of Bacterial Biofilms in Chronic Sinusitis. Eur. Arch. Otorhinolaryngol. 2016, 273, 1989–1994. [Google Scholar] [CrossRef] [PubMed]
- Guinea, J.; Recio, S.; Escribano, P.; Torres-Narbona, M.; Peláez, T.; Sánchez-Carrillo, C.; Rodríguez-Créixems, M.; Bouza, E. Rapid Antifungal Susceptibility Determination for Yeast Isolates by Use of Etest Performed Directly on Blood Samples from Patients with Fungemia. J. Clin. Microbiol. 2010, 48, 2205–2212. [Google Scholar] [CrossRef]
| Mouthwash | MIC (% Concentration of Commercial Product), Mean ± SD (Range) | MFC (% Concentration of Commercial Product), Mean ± SD (Range) | MFC/MIC | MIC of Main Antifungal Compound (in µg/mL), Mean ± SD |
|---|---|---|---|---|
| Fluconazole—control antifungal | - | - | 2.13 ± 2.23 µg/mL | |
| Octenident | 0.10 ± 0.05 (0.05–0.2) | 0.10 ± 0.05 (0.05–0.2) | 1 | 0.5 ± 0.25 µg/mL |
| Octenisept Oral Mono | 0.09 ± 0.04 (0.05–0.2) | 0.09 ± 0.04 (0.05–0.2) | 1 | 0.9 ± 0.4 µg/mL |
| Eludril Classic | 0.12 ± 0.05 (0.05–0.2) | 0.12 ± 0.05 (0.05–0.2) | 1 | 1.1 ± 0.5 µg/mL |
| Corsodyl | 0.12 ± 0.09 (0.05–0.39) | 0.12 ± 0.09 (0.05–0.39) | 1 | 2.4 ± 1.8 µg/mL |
| SeptOralMed | 0.12 ± 0.09 (0.05–0.39) | 0.12 ± 0.09 (0.05–0.39) | 1 | 2.4 ± 1.8 µg/mL |
| Perio Aid Intensive Care | 0.13 ± 0.05 (0.1–0.2) | 0.13 ± 0.05 (0.1–0.2) | 1 | 1.56 ± 0.6 µg/mL CHX, 0.65 ± 0.25 µg/mL CPC |
| Gum Paroex | 0.13 ± 0.06 (0.05–0.2) | 0.13 ± 0.06 (0.05–0.2) | 1 | 1.56 ± 0.6 µg/mL CHX, 0.65 ± 0.25 µg/mL CPC |
| ProntOral | 1.89 ± 0.78 (0.78–3.125) | 1.89 ± 0.78 (0.78–3.125) | 1 | 28.35 ± 11.7 µg/mL |
| Fomukal | 6.38 ± 3.30 (1.56–12.5) | 6.51 ± 3.11 (3.125–12.5) | 1–2 | 6.03 ± 2.23 µg/mL |
| Listerine Total Care | 16.67 ± 7.69 (6.25–25) | 18.23 ± 7.28 (6.25–25) | 1–2 | 36.7 ± 16.9 µg/mL |
| Baikadent mint | 31.25 ± 14.60 (12.5–50) | 33.33 ± 12.31 (25–50) | 1–2 | the inability to calculate |
| Dentosept | 45.83 ± 9.73 (25–50) | 45.83 ± 9.73 (25–50) | 1 | the inability to calculate |
| Meridol Gum Protection | 43.75 ± 11.31 (25–50) | 43.75 ± 11.31 (25–50) | 1 | 109.4 ± 28.3 µg/mL |
| Elmex Sensitive Plus | 47.92 ± 7.22 (25–50) | 47.92 ± 7.22 (25–50) | 1 | 119.8 ± 18.1 µg/mL |
| Glimbax | 70.83 ± 25.75 (50–100) | 75.00 ± 26.11 (50–100) | 1–2 | 524.1 ± 190.6 µg/mL |
| Mouthwashes with: | OCT | CHX and CHX + CPC | PHMB | BAC | F + EO | Olaflur + F | Plant Extracts | Diclofenac |
|---|---|---|---|---|---|---|---|---|
| OCT | - | ns | * | ** | *** | *** | *** | *** |
| CHX and CHX + CPC | ns | - | ns | * | *** | *** | *** | *** |
| PHMB | * | ns | - | ns | ns | * | ns | * |
| BAC | ** | * | ns | - | ns | ns | ns | ns |
| F + EO | *** | *** | ns | ns | - | ns | ns | ns |
| Olaflur + F | *** | *** | * | ns | ns | - | ns | ns |
| Plant extracts | *** | *** | ns | ns | ns | ns | - | ns |
| Diclofenac | *** | *** | * | ns | ns | ns | ns | - |
| Mouthwash | C. albicans Biofilm Reduction, Mean ± SD |
|---|---|
| Octenident | 47.0 ± 10.5 |
| Octenisept Oral Mono | 51.1 ± 13.1 |
| Eludril Classic | 41.7 ± 5.1 |
| Corsodyl | 32.6 ± 5.4 |
| SeptOralMed | 32.1 ± 4.8 |
| Perio Aid Intensive Care | 29.2 ± 5.2 |
| Gum Paroex | 27.6 ± 5.6 |
| ProntOral | 38.6 ± 20.0 |
| Fomukal | 35.7 ± 13.0 |
| Listerine Total Care | 33.2 ± 22.2 |
| Baikadent mint | 35.6 ± 20.8 |
| Dentosept | Rejected due to coloration of the biofilm |
| Meridol Gum Protection | 27.2 ± 20.1 |
| Elmex Sensitive Plus | 28.4 ± 16.5 |
| Glimbax | 26.4 ± 10.9 |
| Mouthwash (Producer) | Main Antimicrobial Components | Other Components |
|---|---|---|
| Octenident® (Schülke & Mayr GmbH, Norderstedt, Germany) | Octenidine HCl (OCT; 500 µg/mL) | Aqua, PEG-40 hydrogenated castor oil, glycerin, aroma, sodium gluconate, sucralose, citric acid, BHT |
| Octenisept Oral Mono® (Schülke & Mayr GmbH, Norderstedt, Germany) | Octenidine dihydrochloride (1000 µg/mL) | Glycerol, sodium gluconate, citric acid, disodium phosphate dihydrate, macrogolglycerol hydroxystearate, sucralose, water, mint flavor |
| Eludril Classic® (Pierre Fabre, Castres, France) | Chlorhexidine digluconate (CHX; 1000 µg/mL) | Glycerin, alcohol, aqua, chlorobutanol, CI 16255, diethylhexyl sodium sulfosuccinate, flavor, limonene, menthol |
| Corsodyl® (GlaxoSmithKline, Brentford, UK) | Chlorhexidine digluconate (2000 µg/mL) | Ethanol, macrogolglycerol hydroxystearate, sorbitol, peppermint oil, water |
| SeptOralMed® (Avec Pharma, Wrocław, Poland) | Chlorhexidine digluconate (2000 µg/mL) | Aqua, glycerin, Peg 40 hydrogenated castor oil, limonene, eugenol, linalool, sodium saccharin |
| Perio Aid Intensive Care® (Dentaid, Barcelona, Spain) | Chlorhexidine digluconate (1200 µg/mL), Cetylpyridinium chloride (CPC; 500 µg/mL) | Aqua, glycerin, propylene glycol, xylitol, PEG-40 hydrogenated castor oil, potassium acesulfame, sodium saccharin, neohesperidin dichalcone, aroma, CI 42090 |
| Gum Paroex® (Sunstar, Etoy, Switzerland) | Chlorhexidine digluconate (1200 µg/mL), Cetylpyridinium chloride (500 µg/mL) | Aqua, glycerin, propylene glycol, PEG-40 hydrogenated castor oil, aroma, sodium citrate, sucralose, citric acid, CI 14720 |
| ProntOral® (B Braun, Melsungen, Germany) | Polyaminopropyl biguanide (Polyhexanide, PHMB; 1500 µg/mL) | Aroma, sodium cyclamate, surfactants, excipients |
| Fomukal® (Vipharm, Ożarów Mazowiecki, Poland) | Benzalkonium chloride (BAC; 125 µg/mL) | Sodium phosphate dibasic, sodium phosphate monobasic, calcium chloride, sodium chloride, water |
| Listerine Total Care® (Johnson & Johnson, New Brunswick, NJ, USA) | Sodium fluoride (220 µg/mL), Eucalyptol, Thymol, Menthol, Alcohol | Aqua, sorbitol, aroma, poloxamer 407, benzoic acid, zinc chloride, aroma, sodium saccharin, methyl salicylate, sodium benzoate, sucralose, propylene glycol, CI 16035, CI 42090 |
| Elmex Sensitive Plus® (Colgate Palmolive, New York, NY, USA) | Olaflur, Potassium fluoride, total fluorine (250 µg/mL) | Aqua, propylene glycol, PEG-40 hydrogenated castor oil, aroma, PVP/dimethylaminoethylmethacrylate polycarbamyl polyglycol ester, saccharin, hydroxyethylcellulose, potassium hydroxide, polyaminopropyl biguanide |
| Meridol Gum Protection® (Colgate Palmolive, New York, NY, USA) | Olaflur, Stannous fluoride, total fluorine (250 µg/mL) | Aqua, xylitol, PVP, PEG-40 hydrogenated castor oil, aroma, sodium saccharin, CI 42051 |
| Baikadent mint® (Herbapol Wrocław, Wrocław, Poland) | Scutellaria baicalensis root extract (concentration data is confidential and unavailable) | Aqua, sorbitol, xylitol, glycerin, PEG-40 hydrogenated castor oil, sodium benzoate, aroma, sodium lauryl sulfate, sodium carbonate, citric acid |
| Dentosept® (Phytopharm, Nowe Miasto nad Wartą, Poland) | Liquid complex extract (910 mg/mL) | A liquid extract from sage leaf (Salviae folium), peppermint herb (Menthae piperitae herba), thyme herb (Thymi herb), chamomile flower (Matricariae flos), oak bark (Quercus cortex), arnica herb (Arnica herba), calamus rhizome (Calami rhizomate), benzocaine |
| Glimbax® (Angelini Pharma, Rome, Italy) | Diclofenac (740 µg/mL) | Choline solution 50%, sorbitol, sodium benzoate, disodium edetate, acesulfame potassium, peach flavor enhancer, mint flavor enhancer, cochineal red (E 124), water |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korbecka-Paczkowska, M.; Karpiński, T.M. In Vitro Assessment of Antifungal and Antibiofilm Efficacy of Commercial Mouthwashes against Candida albicans. Antibiotics 2024, 13, 117. https://doi.org/10.3390/antibiotics13020117
Korbecka-Paczkowska M, Karpiński TM. In Vitro Assessment of Antifungal and Antibiofilm Efficacy of Commercial Mouthwashes against Candida albicans. Antibiotics. 2024; 13(2):117. https://doi.org/10.3390/antibiotics13020117
Chicago/Turabian StyleKorbecka-Paczkowska, Marzena, and Tomasz M. Karpiński. 2024. "In Vitro Assessment of Antifungal and Antibiofilm Efficacy of Commercial Mouthwashes against Candida albicans" Antibiotics 13, no. 2: 117. https://doi.org/10.3390/antibiotics13020117
APA StyleKorbecka-Paczkowska, M., & Karpiński, T. M. (2024). In Vitro Assessment of Antifungal and Antibiofilm Efficacy of Commercial Mouthwashes against Candida albicans. Antibiotics, 13(2), 117. https://doi.org/10.3390/antibiotics13020117








