Abstract
For the UV–curable waterborne polyurethane acrylate (WPUA) emulsion, the free carboxylic groups in the molecular chains introduced by hydrophilic monomers are the inherent disadvantage. We prepared UV and aziridine dual–cured WPUA (UV/AZ–WPUA) emulsion that the carboxylic groups can be crosslinked by the trifunctional aziridine crosslinking agent, trimethylolpropane tris(2-methyl-1-aziridinepropionate) (Sac–100). The effects of Sac–100 content on the gel fraction, tensile properties, hardness, wear resistance, water resistance, and surface morphology of the cured films and coatings were investigated. The results show that, with the increase of the Sac–100 content, the gel fraction, tensile strength, elongation at break, and toughness of the films increase first and then decrease and the wear weight loss and water absorption ratio gradually decrease; in addition, Sac–100 also has an effect on the surface roughness of the coatings. Through comprehensive evaluation, it is considered that the optimum Sac–100 content is 4 wt% in UV/AZ–WPUA system. With the optimum Sac–100 content, the gel fraction of the coating film is 89.78 wt%, the tensile strength is 34.58 MPa, the elongation at break is 343.64%, the toughness is 65.08 MJ/m3, the wear loss is 4.7 mg, and the equilibrious water–absorption ratio is 11.63 wt%. Compared with the UV–cured film without Sac–100, the tensile strength, toughness, and hardness of the dual–cured films increase by 155.4%, 129.3%, and 25.7%, respectively, and the water absorption ratio decreases by 27.1%.
Keywords:
UV; Sac–100; waterborne; coating; film; crosslinking; water resistance; wear resistance; toughness 1. Introduction
Both UV–curable and waterborne coatings are major directions in the field of coatings [1]. For UV–curable coatings, photosensitive resins can polymerize quickly by UV irradiation [2]. All components participate in crosslinking and curing and turn into solid films without volatile organic compound (VOC) emission [3,4]. At the same time, water–based coatings are also widely used because of their environmentally friendly features, high energy efficiency, and low toxicity [5]. Waterborne UV–curable coatings have the performance and environmental requirements necessary for industrial applications as protectors of substrates, such as wood, metal, automotive parts, synthetic leather, fabrics, and paper. They can also be applied in the form of ink for flexographic printing packaging materials, magazines, and miscellaneous pieces of printing sectors [6].
The soft and hard segments in the molecular chains as well as a large number of intramolecular or intermolecular hydrogen bonds of polyurethane (PU) lead to its excellent mechanical properties [6,7,8]. In addition, acrylate can be used to improve the water resistance and mechanical properties of water–based polyurethane (WPU), because the incorporation of double bonds contributes to the formation of a crosslinking structure [9]. The water–based polyurethane acrylate (WPUA) can be used as the surface protective coating of various materials to perform the functions of wear resistance, corrosion resistance, high–temperature resistance, and antifouling; it is also widely used as a high–strength adhesive. The WPUA and the UV–curing technology are combined in the UV–WPUA coatings, which have the characteristics of environmental–friendliness and high efficiency in curing [7,10,11]. Xu et al. [12] first prepared the waterborne polyurethane–acrylate (PUA) oligomer based on isophorone diisocyanate (IPDI), polyether polyol (NJ–210), dimethylol propionic acid (DMPA), and hydroxyethyl methyl acrylate (HEMA) via an in situ and anionic self–emulsifying method. The UV–curable polyurethane–acrylate (UV–PUA) was obtained with an oligomer and monomers (BA and TPGDA).
However, the water–based system has the defect of insufficient water resistance [6,11,13]. Aziridine (AZ) can react with the carboxylic groups in the WPUA molecular chains at room temperature to increase the degree of crosslinking and improve the water resistance of the coatings [14,15]. Hu et al. [16] prepared a solvent–free polyurethane–acrylate (PUA) composite emulsion via in situ polymerization and AZ modification. The results show that AZ can improve the mechanical properties; water, solvent, and salt–spray resistance; and anti–corrosion of the film. Lai et al. [17] synthesized curing agents (di– and triaziridinyl compounds, HDDA–AZ, and TMPTA–AZ) from the reaction of aziridine with hexanediol diacrylate and trimethylolpropane triacrylate, respectively, and then a single–component polymer dispersion was obtained by mixing the water–reducible acrylate copolymer, the polyurethane ionomer, and the latent curing agent. The carboxyl anions of waterborne polymers not only stabilize the aqueous polymer dispersions, but also serve the curing site as a latent curing agent in the drying process. However, the strength and hardness of the coating crosslinked only by AZ are poor.
The combination of UV– and AZ–curing in coatings has rarely been reported. In this work, a UV– and AZ–dual–curable waterborne polyurethane acrylate (UV/AZ–WPUA) was prepared. Compared with the UV–curable coating, the tensile strength, wear resistance, and water resistance of UV/AZ–WPUA films are improved through the reaction between aziridine and carboxylic groups during the film formation. The dual–curing technology can be used in the coatings for wooden wares and metal and plastics products.
2. Materials and Methods
2.1. Materials
Isophorone diisocyanate (IPDI) and polybutylene adipate (PBA, polyester glycol with molecular weight of 1000 g/mol and hydroxyl content of 112 mg KOH/g) were purchased from Macklin Biochemical Technology Co., Ltd. (Shanghai, China). The 2,2′-Bis(hydroxymethyl)butyric acid (DMBA), hydroxyethyl acrylate (HEA), triethylamine(TEA), dibutyltin dilaurate (DBTDL), and 2-hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone (photoinitiator, Irgacure 2959) were purchased from Heowns Chemical Co., Ltd. (Tianjin, China). Dipropylene glycol diacrylate (DPGDA) and trimethylolpropane trimethacrylate (TMPTMA) were provided by Tianjiao Chemical Co., Ltd. (Tianjin, China). Deionized water was used as the dispersed phase. The AZ–type crosslinker trimethylolpropane tris(2-methyl-1-aziridinepropionate) (trade name: Sac–100) was purchased from Youen Chemical Co., Ltd. (Shanghai, China).
2.2. Synthesis of UV–WPUA Emulsion
For preparing the PU prepolymer, the molar ratio of the isocyanate groups to hydroxyl groups was set to 1.10, the content of DMBA was 5 wt%, and the end–capping ratio of the polyurethane prepolymer was 90% by HEA; the photoinitiator was added according to double bonds content, and the molar ratio of TEA to DMBA was 1.0.
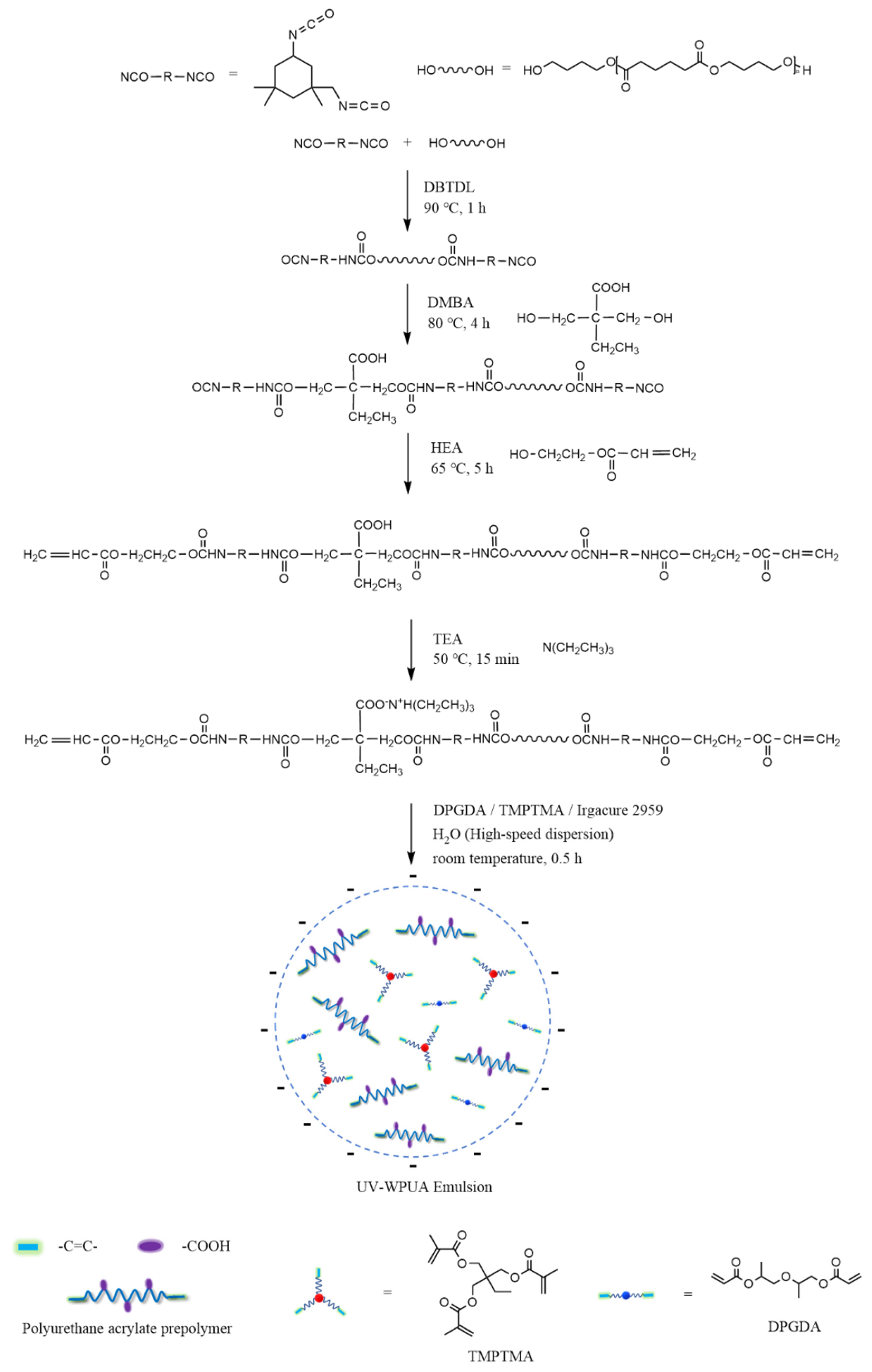
According to the scheme shown in Figure 1, the UV–WPUA emulsion was synthesized in 5 steps.
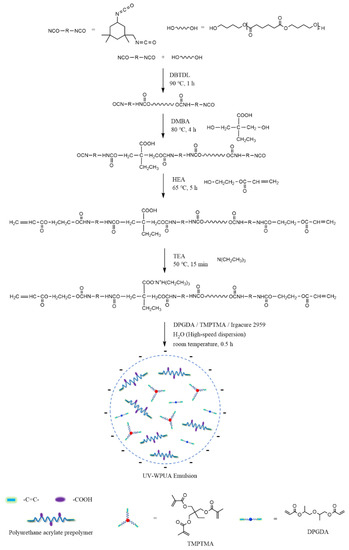
Figure 1.
The preparing process for UV–WPUA emulsion.
- IPDI, PBA, and DBTDL were accurately weighed and fed into a four–necked flask equipped with a mechanical stirrer, a condensation reflux device, a nitrogen conduit, and a thermometer. The NCO–terminated PU prepolymer was obtained after the 1 h reaction at 90 °C.
- DMBA was added, and the reaction was kept at 80 °C for 4 h so that the –NCO–terminated PU prepolymer was extended, and the carboxylic groups was introduced.
- The prepolymer was further reacted with HEA at 65 °C for 5 h; the prepolymer terminated by the unsaturated double bonds was obtained.
- The neutralizer TEA was added at 50 °C and kept stirring for 15 min, then DPGDA, TMPTMA, and Irgacure 2959 were added to the system and mixed evenly.
- The deionized water was added under high–speed stirring at room temperature. The dispersion was vigorously stirred for 0.5 h to obtain a UV–WPUA emulsion with a solid content of about 35 wt%.
2.3. Preparation of UV/AZ–WPUA Dual–Cured Films and Coatings
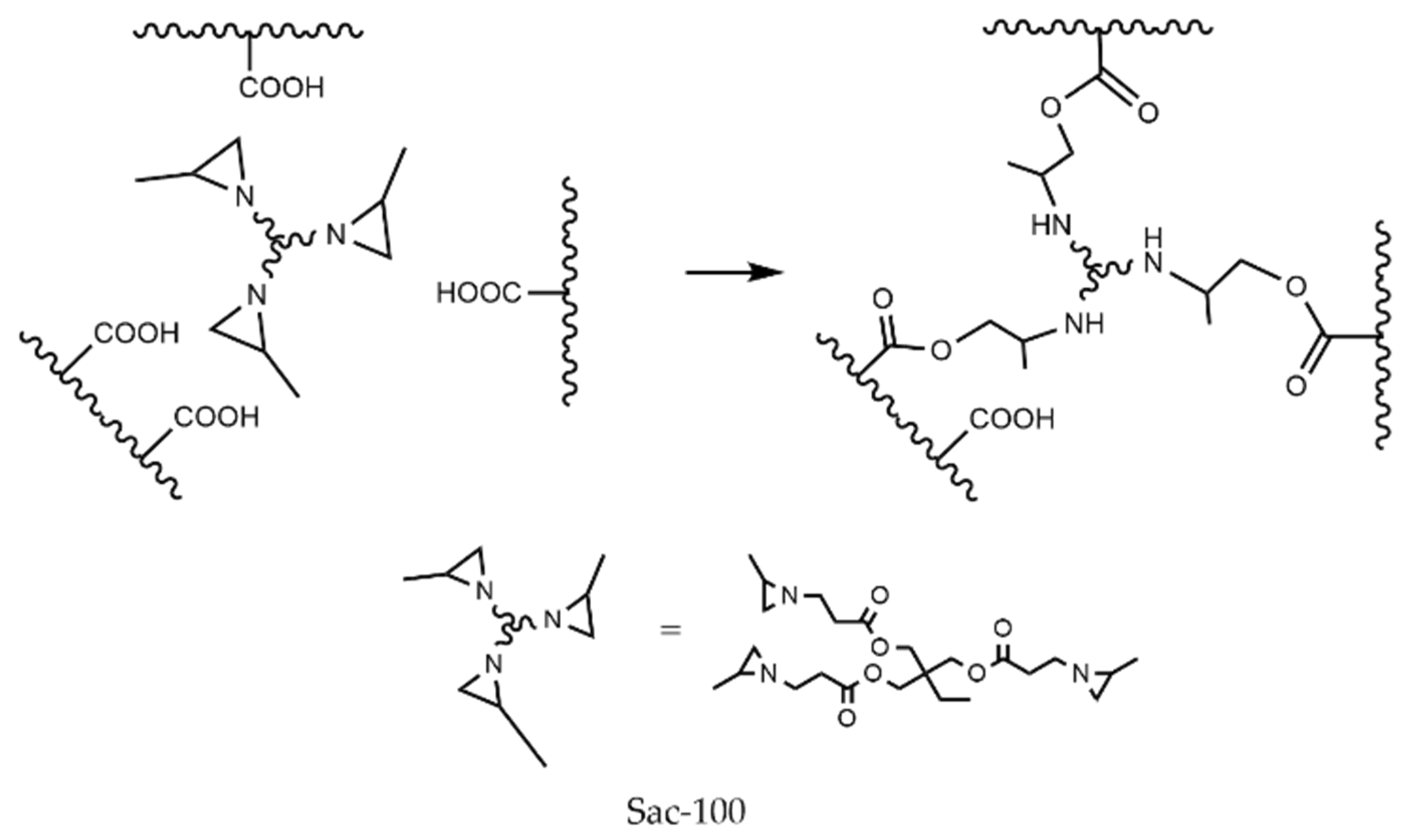
Aziridine can react with carboxylic groups to form amino esters. Figure 2 shows the reaction between Sac–100 and PUA chains with carboxylic groups, which results in the connection of three polymer chains.
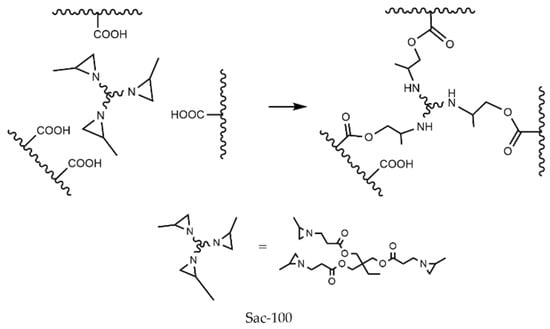
Figure 2.
Crosslinking reaction between Sac–100 and carboxylic groups.
Different amounts of Sac–100 were added to the UV–WPUA emulsion to obtain the UV/AZ–WPUA emulsions by mechanical stirring and vacuum defoaming, which then were coated on stainless steel plates, dried at 45 °C for 5 min to remove moisture and cured by UV irradiation for 60 s to obtain coatings with the thickness of 50 μm. In order to test tensile properties, hardness, and water resistance, UV/AZ–WPUA emulsions were injected into PTFE molds, dried at 45 °C for 12 h to totally remove moisture, and then cured by UV irradiation to obtain transparent films with a thickness of about 0.4 mm. The UV/AZ–WPUA coatings and films were obtained by adding different amounts of Sac–100. The samples with 0 wt%, 2 wt%, 4 wt%, 6 wt%, 8 wt%, 10 wt%, and 12 wt% Sac–100 are represented by the symbols S0, S2, S4, S6, S8, S10, and S12, respectively. The AZ–WPUA (only AZ–curing but without UV–curing) coatings and films (OS2, OS4, OS6, OS8, OS10, OS12) were obtained by adding different amount of Sac–100 (2 wt%, 4 wt%, 6 wt%, 8 wt%, 10 wt% and 12 wt%). The relevant characterizations (tensile properties and water absorption ratio) are only shown in Supplementary Material because the crosslinking degree of coatings and films only cured by Sac–100 is very low.
2.4. Characterization
2.4.1. Fourier Transform Infrared (FTIR) Spectroscopy
The PUA oligomer was obtained by the reaction of IPDI, PBA, and DMBA, which was terminated by HEA and diluted with DPGDA and TMPTMA, which was dissolved in acetone to obtain a homogeneous and clear solution. The UV–cured film (S0), AZ–cured film (OS6), and UV/AZ–cured film (S6) were cut into slices. All the spectra were taken by ATR mode with a Nicolet 6700 infrared spectrometer (Thermo Fisher Scientific Co., Ltd., Waltham, MA, USA) in 1000~4000 cm−1.
2.4.2. Emulsion Properties
The particle–size distribution and zeta potential of the emulsion were measured using a Nano ZS instrument (Malvern Instrument Co., Ltd., Malvern, PA, UK). The emulsion stability was measured using a TG–16 high–speed centrifuge (Xiangyi Instrument Co., Ltd., Xiangtan, China) under 3000 rpm for 15 min. If there were no signs of precipitation after centrifugation, the emulsions could be evaluated with a shelf life longer than six months [18].
2.4.3. Gel Fraction
The 1 × 1 cm2 slices of WPUA films that had been wrapped with filter paper and sealed with steel wire were immersed in acetone with magnetic rotor stirring for 72 h and then were taken out and dried at 60 °C to constant weight. The gel fraction of the films (Gel%) was calculated using the following equation:
where m1, m2, and m3 are the initial weight of the film; the total weight of the film, filter paper, and steel wire; and the constant weight after drying, respectively.
2.4.4. Mechanical Properties
Tensile properties. According to GB/T 528–2009, the WPUA films were cut into dumbbell–shaped samples, which were subjected to tensile tests with the electronic universal testing machine (WDW–T05, Tianchen Testing Machine Manufacturing Co., Ltd., Jinan, China) at a tensile speed of 50 mm·min−1. At least three or more parallel experiments were carried out.
Hardness. The films were placed horizontally on the test table, then Shore D durometers were handled and pressed vertically on the samples until the bottom disc of the durometer was in contact with the sample surface. Each sample was tested from more than 3 positions to calculate the average value as the corresponding hardness value.
2.4.5. Wear Resistance
According to ASTM D4060-19, the coatings were abraded for 500 cycles by Taber Rotary Platform Abrasion Tester Model 1700 (Taber Industry Co., Ltd., North Tonawanda, NY, USA) with a CS–10 grinding wheel at the speed of 60 ± 2 min−1 under the load of 1 kg/wheel, and the weight loss was recorded after abrasion. Three parallel experiments were carried out.
2.4.6. Water Resistance
Water–Absorption Ratio. The 1 × 1 cm2 dried slices cut from the films were immersed in distilled water and then taken out at regular intervals and weighed quickly after wiping the water off the surface. The water–absorption ratio (WA%) of the cured films was calculated by the following equation:
where m1 and m2 are the weights of the film before and after absorbing water, respectively.
Three parallel experiments were carried out for each sample.
Water–Contact Angle. The water–contact angles of coatings were measured at room temperature using a JC2000FM contact–angle measuring instrument (Powereach Digital Technology Equipment Co., Ltd., Shanghai, China), and each sample was tested at 3 different locations.
2.4.7. Atomic Force Microscopy (AFM) Surface Topography
The surface topographies of the curing films were characterized by AFM (NTEGRA Spectra, Bruker, Karlsruhe, Germany) with a scan size of 5 × 5 μm2.
2.4.8. Scanning Electron Microscopy (SEM) Cross–Sectional Morphology
The cross–sectional morphologies of the fractured samples in the tensile test were observed using SEM (S-4800, Hitachi, Tokyo, Japan) at the accelerating voltage of 3 kV.
2.5. Statistical Analysis
Figures were produced using Origin 2022 (Originlab Inc., Northampton, MA, USA). All of the analyzed data were expressed as means ± standard deviation (SD). SPSS version 26 (SPSS Inc., Chicago, IL, USA) software was used to perform statistical analyses. A one-way ANOVA with a Duncan’s t-test was used to evaluate the significant differences (p ≤ 0.05). The letters “abcde…” are used to indicate the significance of the data. If there is the same letter in any two groups of data, there is no significant difference between the two groups of data; otherwise, it indicates that there is significant difference.
3. Results and Discussion
3.1. FTIR Spectrum
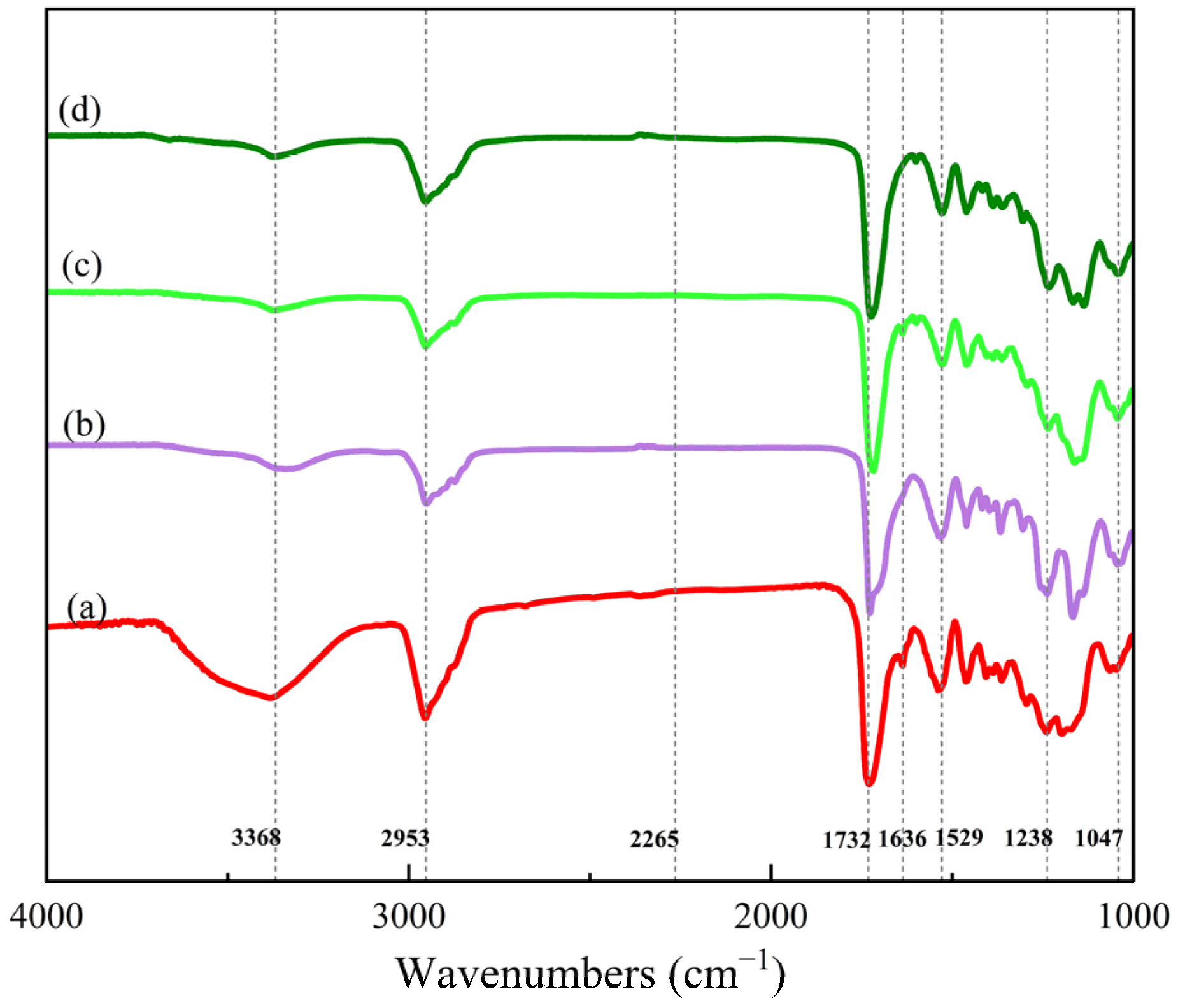
In Figure 3, the spectra of the PUA oligomer, UV–cured film (S0), AZ–cured film (OS6), and UV/AZ–cured film (S6) are illustrated.
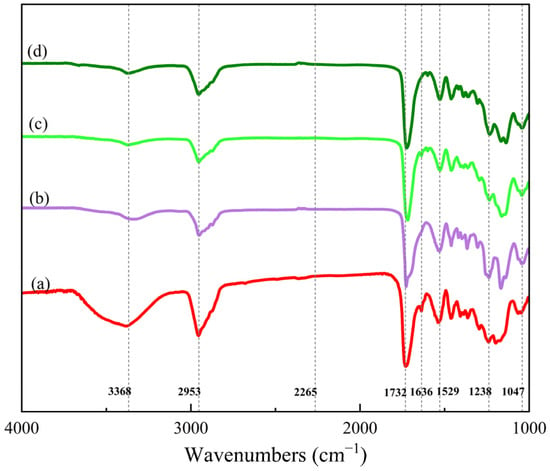
Figure 3.
FTIR spectra of (a) PUA oligomer, (b) UV–cured film, (c) AZ–cured film, and (d) UV/AZ–cured film.
The absorption peaks at 3368 cm−1 (stretching vibration of –N–H), 1529 cm−1 (bending vibration of –N–H), 1732 cm−1 (stretching vibration of C=O), 1047 cm−1, and 1238 cm−1 (symmetric stretching vibration and asymmetric stretching vibration of C–O–C) exist in all spectra, indicating that –NCO reacts with –OH to form carbamate groups. Simultaneously, the absorption peak at 2265 cm−1 (stretching vibration of –NCO) only appears apparently in the IPDI spectrum, which proves a sufficient reaction between –NCO and –OH [19].
The characteristic peak at 1636 cm−1 (stretching vibration of –C=C–) exists in the spectra of the PUA oligomer and the AZ–cured film, while it weakens or disappears in the spectra of the UV–cured film and the UV/AZ–cured film, suggesting that –C=C– has been crosslinked by UV irradiation.
3.2. The Properties of UV–WPUA Emulsion
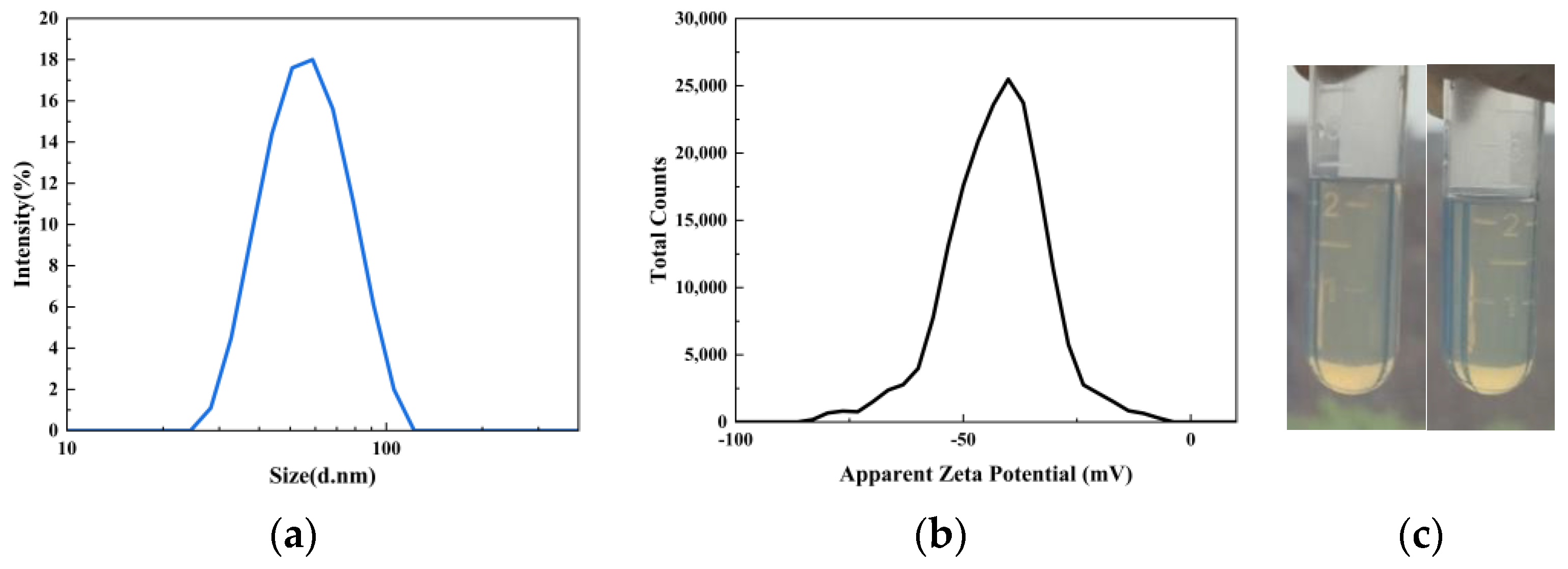
As shown in Figure 4, the average particle size of the UV–WPUA emulsion (S0) is 53.85 nm, and the particle–distribution index (PDI) is 0.061, showing a monodisperse state. If the particles in dispersion have large negative or positive zeta potentials, then they will tend to repel each other, and there is less tendency to flocculate. Particles with zeta potentials more positive than +30 mV or more negative than −30 mV are normally considered stable [20]. The zeta potential of the UV–WPUA emulsion S0 is −41.4 mV, indicating that the surface charge of particles is negative, and the repulsive force among particles prevents the aggregation of particles, so the emulsion has good stability [21,22].
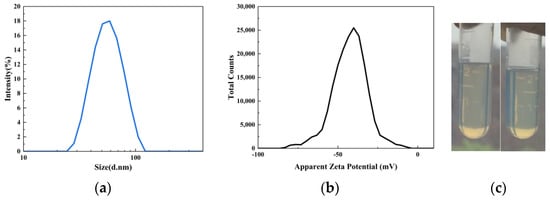
Figure 4.
(a) Particle–size distribution, (b) zeta potential, and (c) comparison before (left) and after (right) centrifugation of UV–WPUA emulsion.
No signs of precipitation were observed when the emulsion was centrifuged at 3000 rpm for 15 min (Figure 4c), indicating that the emulsion can be stored stably for more than six months.
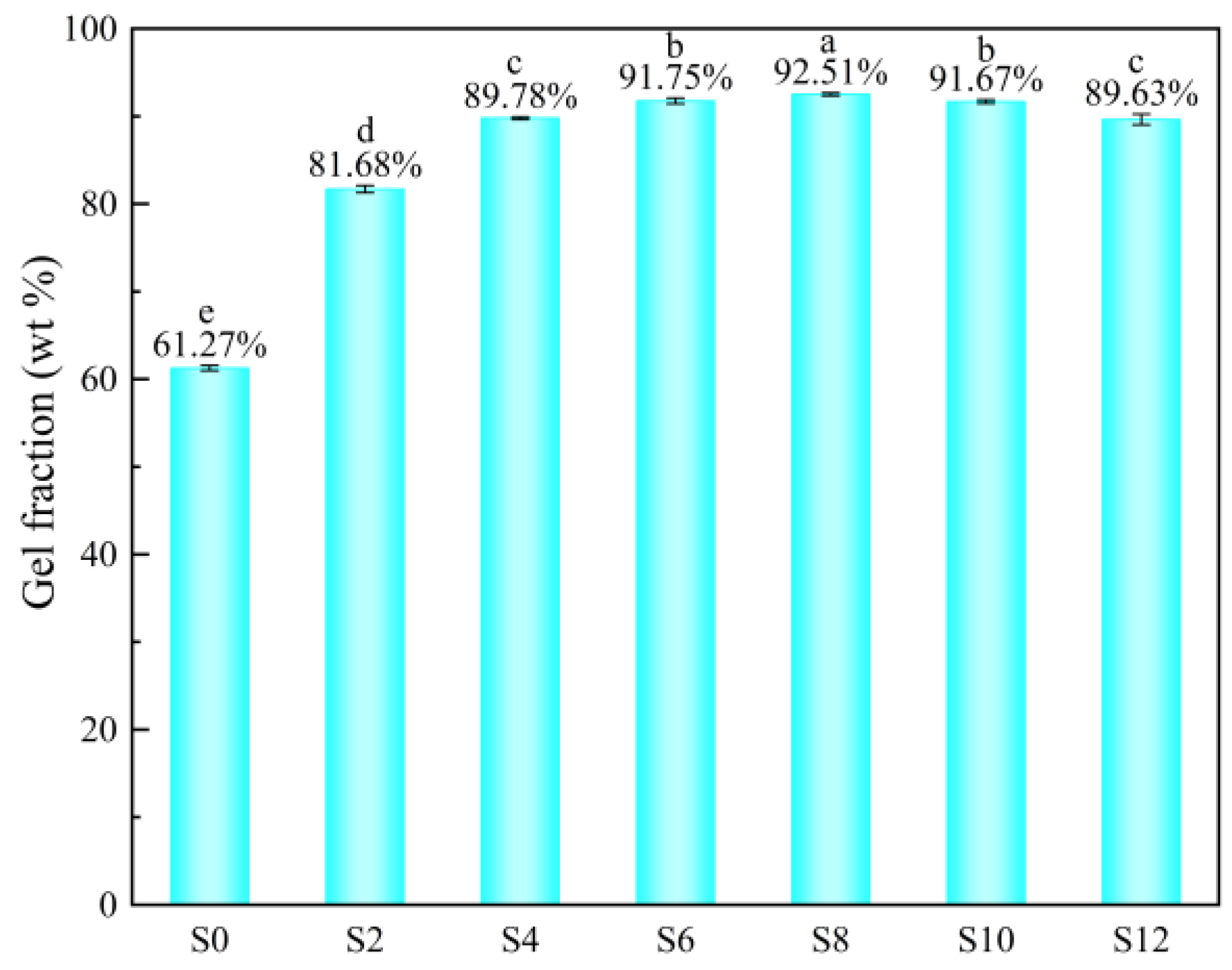
3.3. Gel Fraction
The effect of the Sac–100 content on the gel fraction of WPUA films is shown in Figure 5. With the increase of the Sac–100 content from 0 wt% to 8 wt%, the gel fraction increases from 61.27 wt% to a maximum value of 92.51 wt%, because the crosslinking between the Sac–100 and the carboxylic groups increases with the Sac–100 content. As the Sac–100 content further increases to 12 wt%, the gel fraction decreases slightly to 89.63 wt%, which is due to the limited amount of carboxylic groups in the system. When the Sac–100 content is too high, the excess aziridine will exist in the system in the form of free small molecules, which will lead to the reduction of the crosslinking degree (gel fraction) in the film, thus affecting the solvent resistance of the film. Moreover, if the initial crosslinking degree is too high due to the high Sac–100 content, it also hinders the movement of molecular chains and the crosslinking of double bonds in the subsequent UV irradiation.
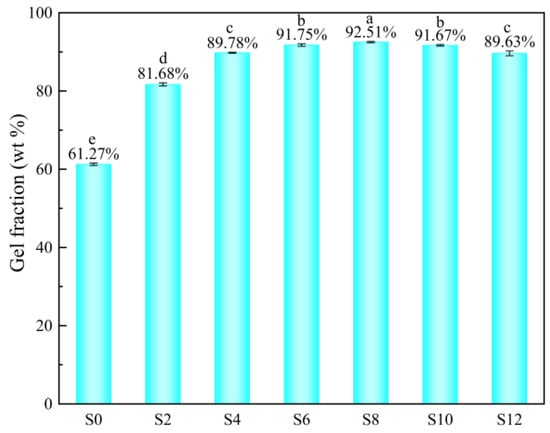
Figure 5.
Gel fraction of WPUA films with different Sac–100 contents.
3.4. Mechanical Characterization
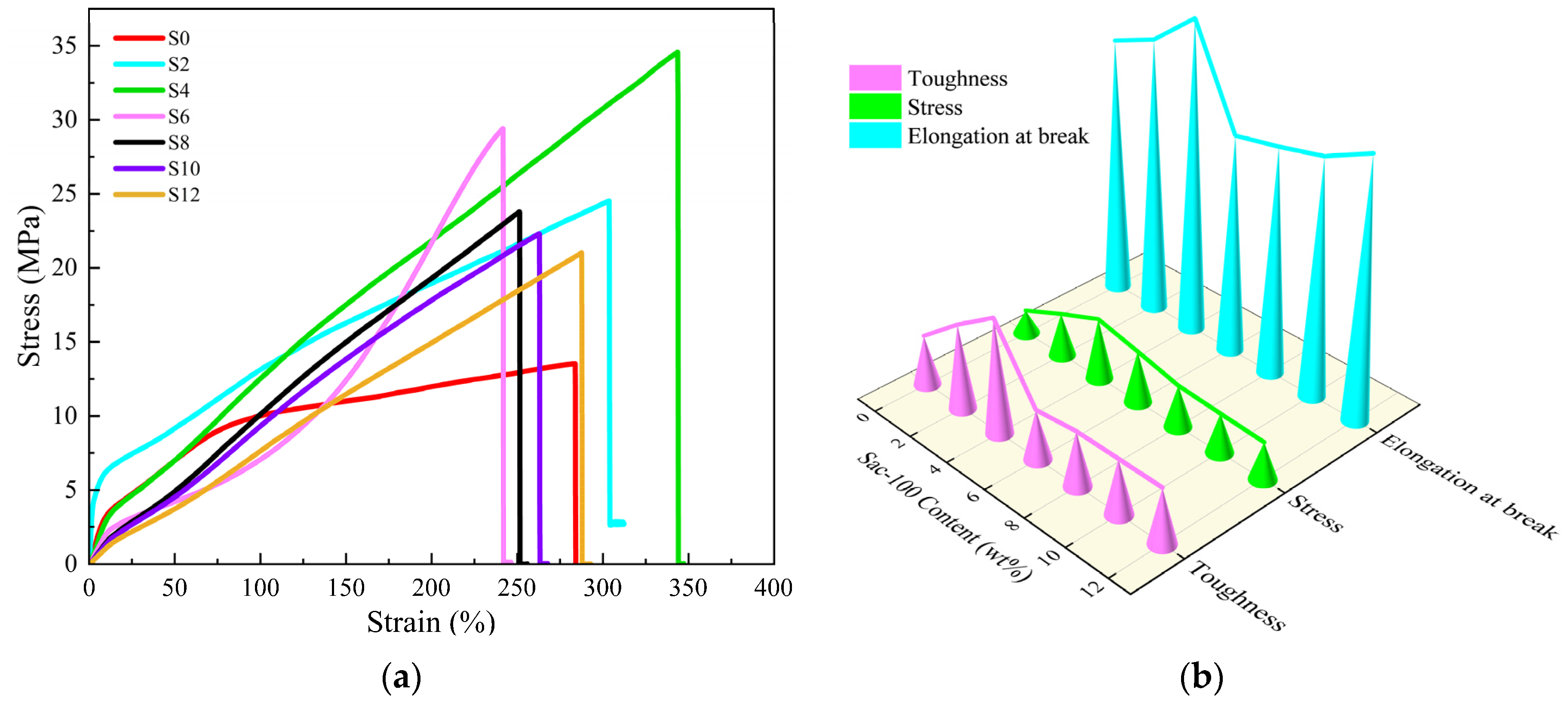
The effects of the Sac–100 content on the tensile properties and hardness of the WPUA films are shown in Table 1 and Figure 6 and Figure 7. The effects of Sac–100 content on the tensile properties of the WPUA films only cured by Sac–100 are shown in Table S1 and Figure S1 of Supplementary Material.

Table 1.
Tensile properties of WPUA films with different Sac–100 contents.
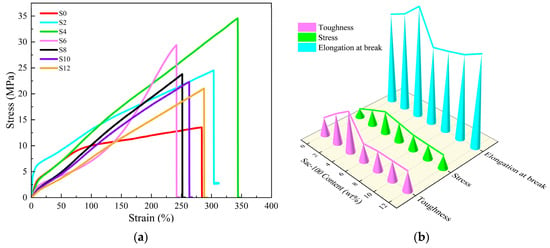
Figure 6.
(a) Stress–strain curves and (b) tensile parameters of WPUA films with different Sac–100 contents.
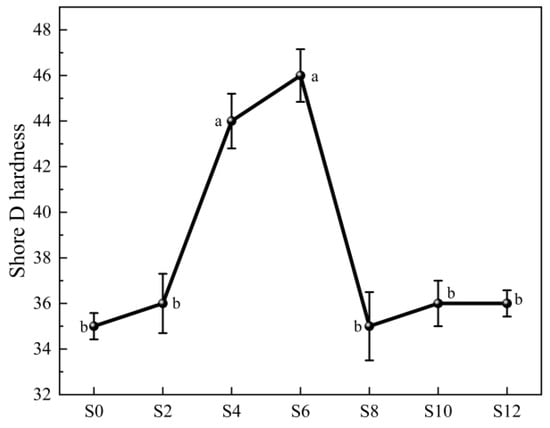
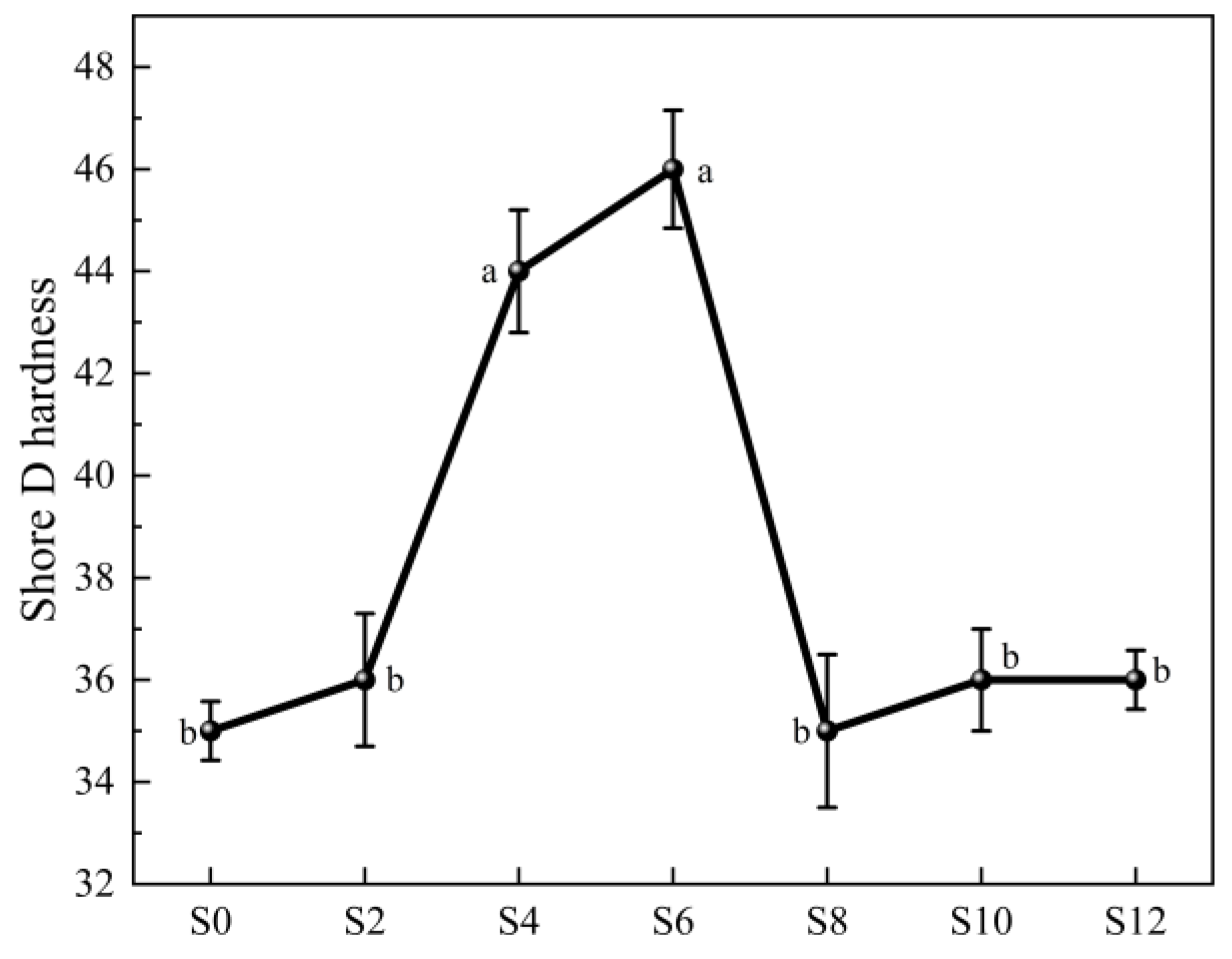
Figure 7.
Shore D hardness of WPUA films with different Sac–100 contents.
With the increase of the Sac–100 content from 0 wt% to 4 wt%, the tensile strength, elongation at break, and toughness increase first and reach their maximum values of 34.58 MPa, 343.64% and 65.08 MJ/m3, respectively. More Sac–100 will lead to decreasing of all three parameters. The results show that the appropriate content of Sac–100 (4 wt%) can simultaneously improve the tensile strength and the elongation at break of the WPUA films, resulting in a toughening effect. However, due to the limited carboxylic groups in the system, when the dosage of Sac–100 exceeds 6 wt%, excessive addition results in the deterioration of the mechanical properties.
In addition, Sac–100 has an optimal content for the hardness of the film (Figure 7). When its dosage is 6 wt%, the shore D hardness of the film reaches its highest value of 46. In fact, the hardness also reflects the crosslinking degree of the cured resins. From the tensile properties and hardness of the film, it can be concluded that the best dosage of Sac–100 is in the range of 4~6 wt%.
The cross–section morphology can also reflect the mechanical properties of materials. As shown in Figure 8, the pit– and stripe–like wavy terrain in the fracture morphology of all samples are characteristics of ductile fracture [23]. When the Sac–100 content is 0 wt%, the tensile cross–section of the WPUA film has a relatively smooth background, despite the existence of wrinkles. When the Sac–100 content increases to 4 wt%, the shell texture is observed in the SEM image, because the crosslinking degree between the connected components increases, and the components are bound to each other when they are broken, indicating that the toughness is improved [24]. As the Sac–100 content increases to 8 wt%, the tensile cross–section becomes slightly smoother, reflecting the decrease in toughness. The result is consistent with the tensile test. In addition, we increased the magnification of SEM to observe the cross-sectional morphology of the samples (see Figure S3).


Figure 8.
SEM images of WPUA films’ tensile cross–section of (a) S0, (b) S4, and (c) S8.
3.5. Wear Resistance
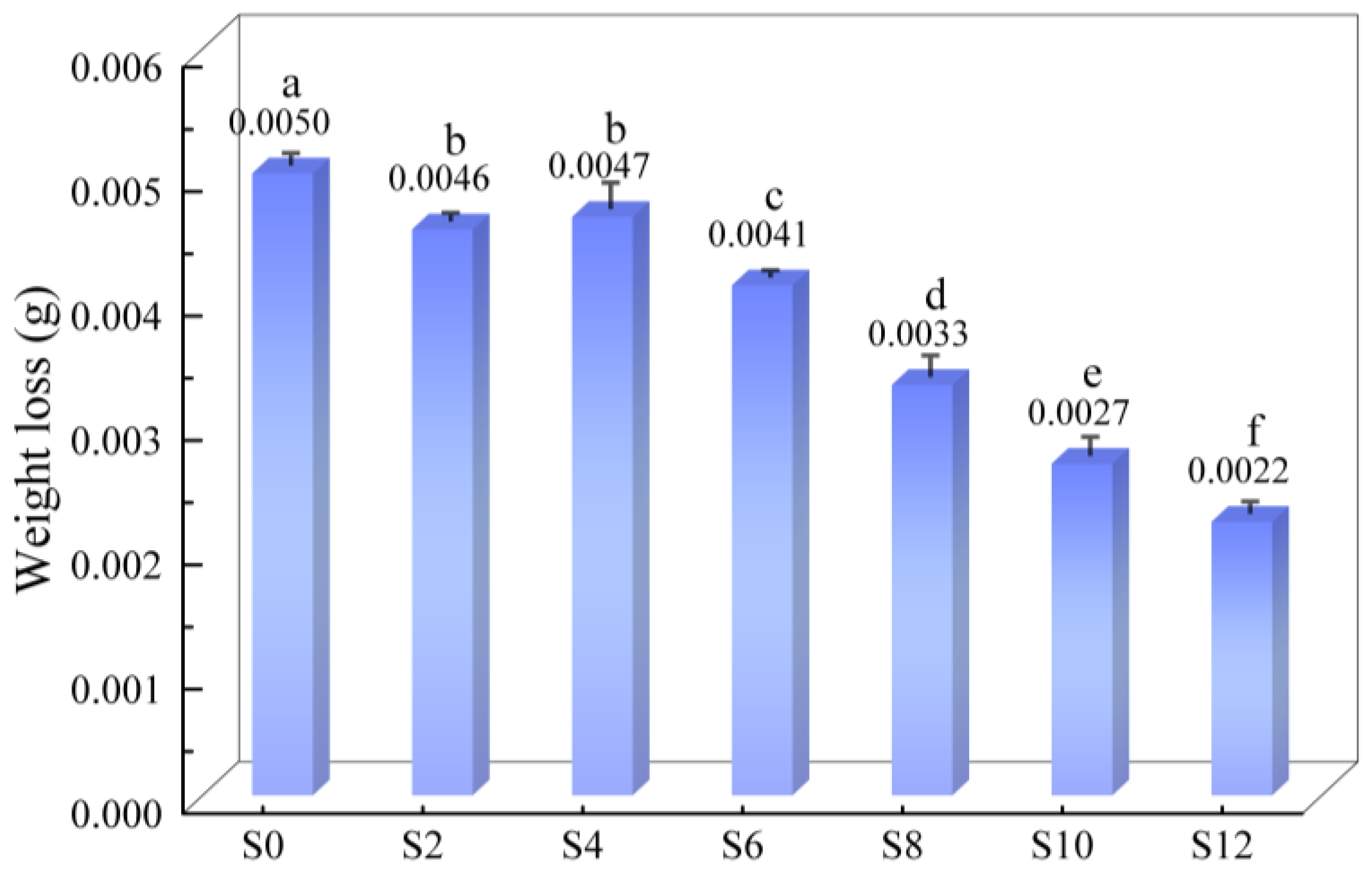
The weight loss of the coatings with different Sac–100 contents in the wear test are shown in Figure 9. As the Sac–100 content increases from 0 wt% to 12 wt%, the weight loss decreases from 5.0 mg to 2.2 mg, and the wear resistance gradually improves, because a large number of Sac–100 molecules interspersed in the crosslinked network endure certain intrinsic stress.
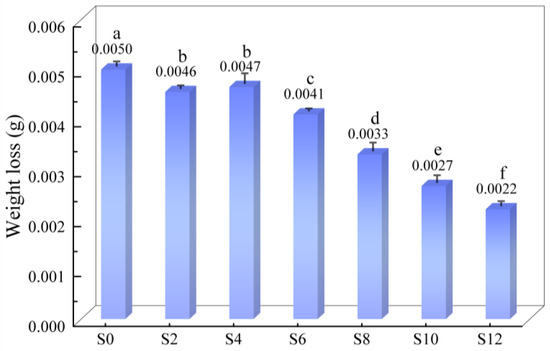
Figure 9.
Weight loss of coatings with different Sac–100 contents in the wear test.
3.6. Water Resistance
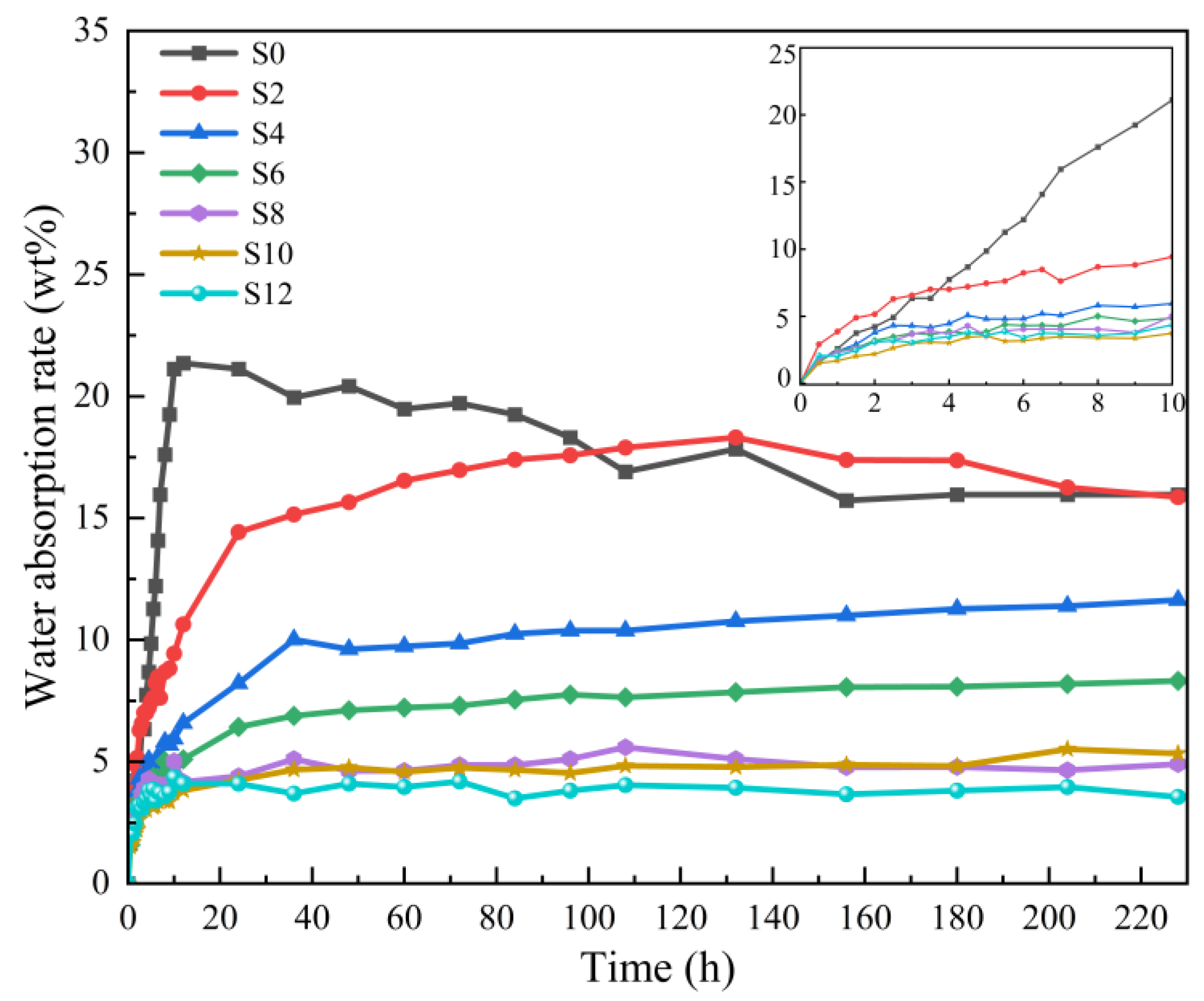
Figure 10 and Table 2 show the effect of the Sac–100 content on the water–absorption ratio of WPUA films. With the increase of the Sac–100 dosage from 0 wt% to 12 wt%, the equilibrious water–absorption ratios decrease from 15.96 wt% to 3.55 wt%, indicating that the water resistance of the films has gradually improved. This is because Sac–100 reacts with carboxylic groups in the WPUA chains to form the crosslinking network, which not only effectively prevents the hydration caused by carboxyl groups, but also increases the crosslinking degree of the cured film, therefore slowing down the penetration of the water molecules [25,26].
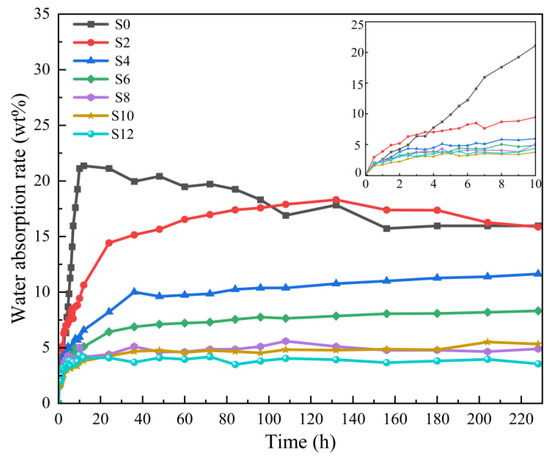
Figure 10.
Water–absorption ratio of WPUA films with different Sac–100 contents.

Table 2.
Equilibrious water–absorption ratio and water–contact–angle data of WPUA films with different Sac–100 contents.
For S0 (without Sac–100), the water–absorption ratio of its UV–cured film decreases after reaching a maximum value. We believe that this is only an illusion. The insufficient crosslinking degree only through the UV–curing (without aziridine–curing) means that the molecular chains are still motional, and the polymer film shows a certain water–absorption–loss elasticity. Moreover, the un–crosslinked oligomer in the film may slowly diffuse into the water while it is soaked in water, resulting in the illusion that the water–absorption ratio is reduced.
With the increase of the Sac–100 content, the equilibrious water–absorption ratios of the WPUA films gradually decrease, indicating that UV– and aziridine– dual curing technology can effectively improve the crosslinking degree of the film, and the aziridine crosslinking is effective in improving the water resistance. When the Sac–100 content is higher than 8%, the equilibrious water–absorption ratios of the cured films decrease slowly, also indicating that more aziridine has little effect on the water resistance of the film.
In addition, the effects of Sac–100 content on the water–absorption ratio of WPUA films only cured by Sac–100 are shown in Figure S2 and Table S2.
We also investigated the effect of the Sac–100 content on the water contact angles of the cured films, and the results are shown in Table 2. It can be seen that the water contact angles of WPUA films with different Sac–100 contents fluctuate in a limited range. In fact, aziridine can improve the crosslinking degree of the cured film and reduce the content of the free carboxylic groups in the film, thereby reducing the equilibrious water–absorption ratio. However, aziridine itself will not change the film polarity, so it will hardly affect the water wettability of the film; in other words, it has little effect on the water contact angle. In addition, during the drying process, the carboxylic groups will be arranged inside the coating, so there is no obvious difference in the external surface characteristics of the coating over a short time. We believe that the equilibrious water absorption ratio represents the long–term water resistance of the film, while the water contact angle reflects the instantaneous water resistance. Although aziridine does not significantly reduce the water contact angle of the cured film, it can effectively prevent the absorption and diffusion of water molecules in the film.
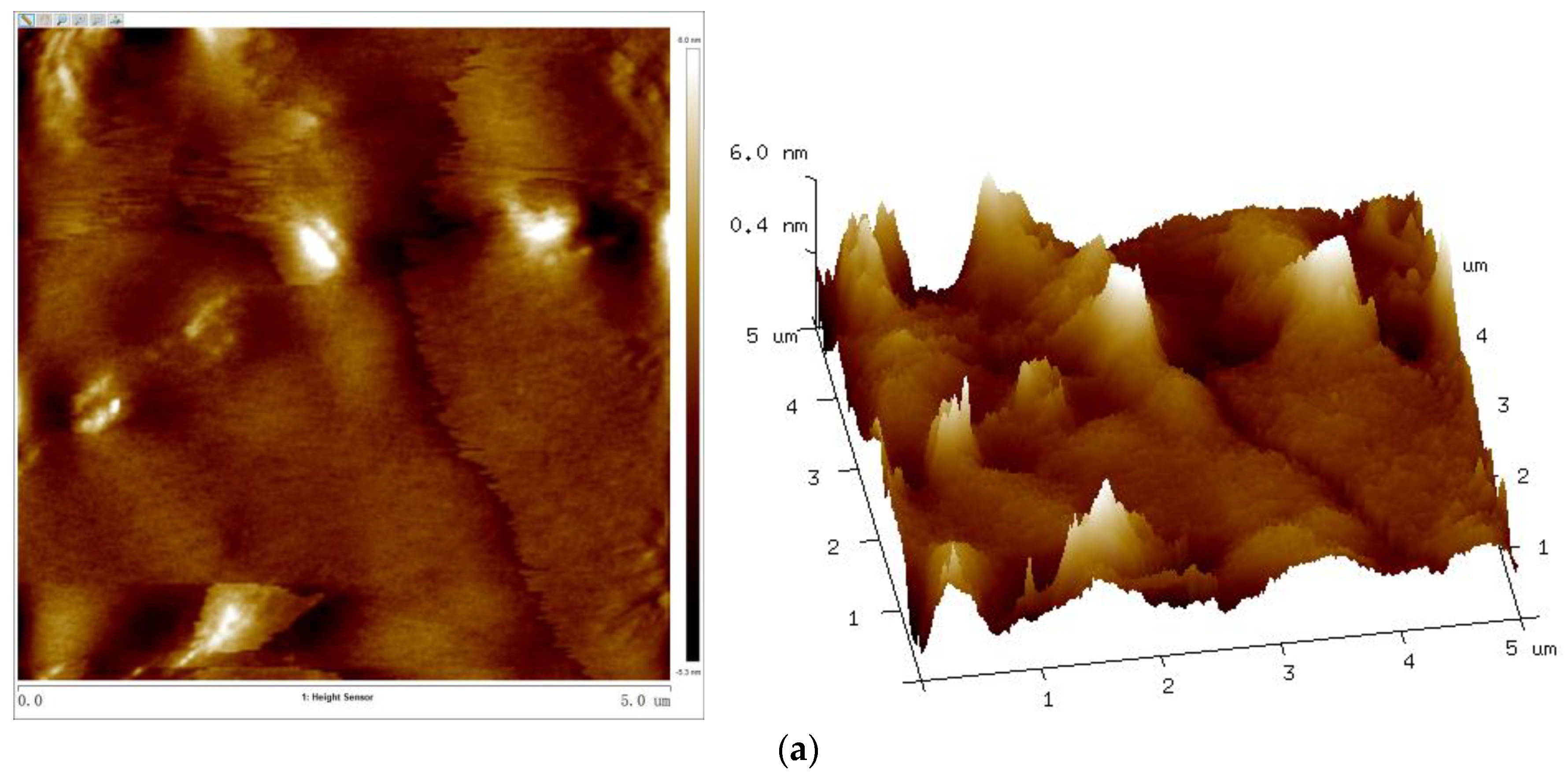
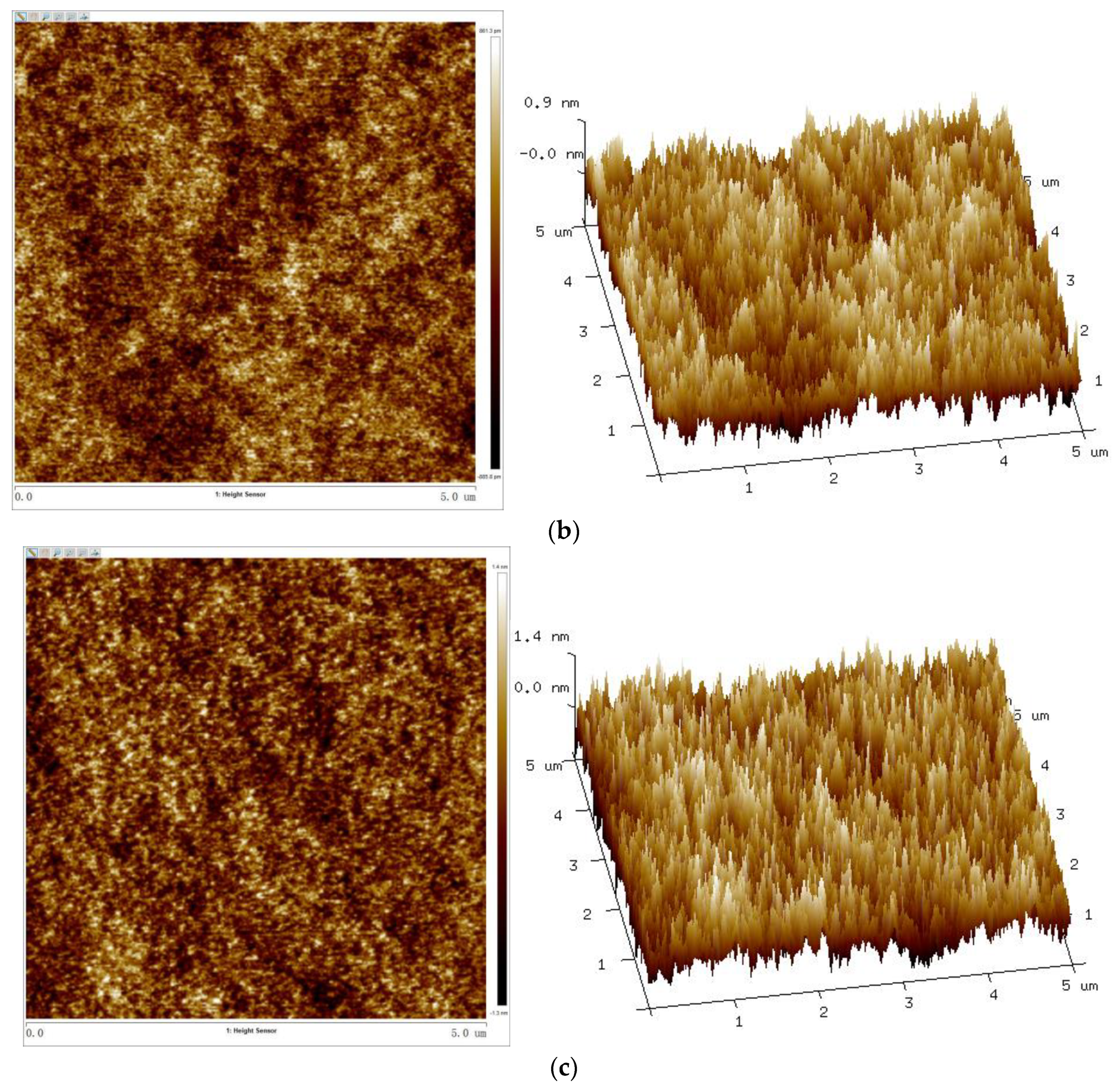
3.7. Surface Topography
Table 3 and Figure 11 show the surface–roughness data and AFM 2D and 3D phase diagrams of coatings with different Sac–100 contents.

Table 3.
Surface–roughness parameters of WPUA coatings with different Sac–100 contents.
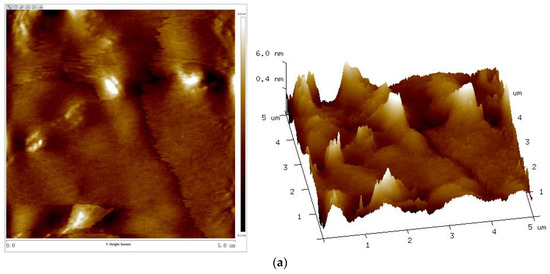
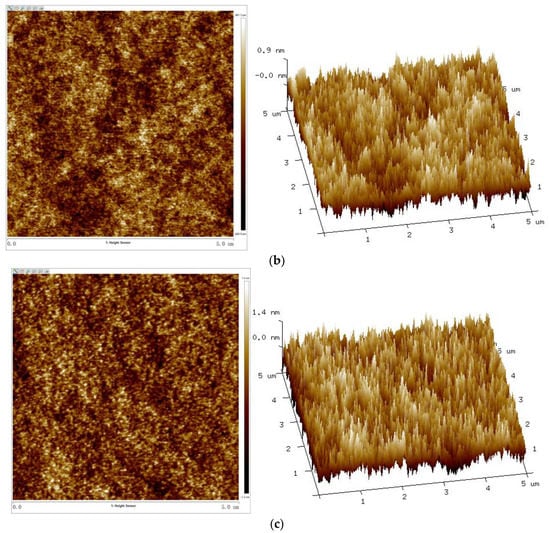
Figure 11.
AFM 2D (left) and 3D (right) phase images of UV–cured coatings of (a) S0, (b) S6, and (c) S12.
With the increase of the Sac–100 content from 0 wt% to 6 wt%, the crosslinking of the cured films becomes denser and tighter, which reduces the degree of phase separation of the WPUA chains and the shrinkage during the UV curing, so all the roughness parameters, including the root mean square roughness (Rq), average roughness (Ra), and maximum roughness (Rmax), decrease. However, when the aziridine content is too high, the excess aziridine cannot crosslink with the limited carboxylic groups but can affect the uniformity and consistency of the cured films. Therefore, from the point of film smoothness, the preferred Sac–100 content is 6 wt%.
The color, thickness and moisture content of the relevant samples are shown in Table S3 of Supplementary Material.
Table 4 shows the differences between this study and other studies of curing methods, as well as some properties of the samples. The results show that the S4 sample has excellent toughness and great relative strength.

Table 4.
Performance comparison across relevant studies.
4. Conclusions
A series of UV/AZ–WPUA films and coatings with different Sac–100 contents were successfully prepared. As the Sac–100 content increases, the gel fraction, tensile strength, elongation at break, toughness, and hardness of films first increase and then decrease, and wear resistance and water resistance are improved. Sac–100 also had an effect on the surface roughness of the coatings. After a comprehensive evaluation, the optimal dosage of Sac–100 is 4 wt%. Compared with the single–UV–cured film without Sac–100, the dual–cured film has higher values of tensile strength and toughness, as well as better wear resistance and water resistance.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/coatings12091293/s1, Figure S1. Stress–strain curves of WPUA films only cured with Sac–100; Table S1. Tensile properties of WPUA films only cured with Sac–100; Table S2. Water–absorption ratio of WPUA films only cured with Sac–100; Figure S2. Water–absorption ratio of WPUA films only cured with Sac–100; Table S3. Physical properties of WPUA films with different Sac–100 contents; Figure S3. SEM images of WPUA films’ tensile fracture section of (a) S0, (b) S4 and (c) S8.
Author Contributions
Conceptualization, R.W. and C.Y.; Data curation, R.W.; Formal analysis, R.W.; Investigation, R.W., X.B., Z.X., J.Z. and F.P.; Methodology, R.W., Z.Z. and C.Y.; Project administration, C.Y.; Resources, C.Y.; Supervision, C.Y.; Validation, Z.Z., X.B. and F.P.; Visualization, R.W. and Z.X.; Writing—original draft, R.W.; Writing—review and editing, X.B. and C.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Liu, F.G.; Liu, A.M.; Tao, W.J.; Yang, Y. Preparation of UV curable organic/inorganic hybrid coatings—A review. Prog. Org. Coat. 2020, 145, 105685. [Google Scholar] [CrossRef]
- Decker, C. Kinetic study and new applications of UV radiation curing. Macromol. Rapid Commun. 2002, 23, 1067–1093. [Google Scholar] [CrossRef]
- Shan, C.L.; Ning, C.; Lou, J.J.; Xu, W.; Zhang, Y. Design and preparation of UV–curable waterborne polyurethane based on novel fluorinated chain extender. Polym. Bull. 2021, 78, 2067–2083. [Google Scholar] [CrossRef]
- Dominika, C.; Barbara, P. Progress in development of UV curable powder coatings. Prog. Org. Coat. 2021, 158, 106355. [Google Scholar] [CrossRef]
- Noreen, A.; Zia, K.M.; Zuber, M.; Tabasum, S.; Saif, M.J. Recent trends in environmentally friendly water–borne polyurethane coatings: A review. Korean J. Chem. Eng. 2016, 33, 388–400. [Google Scholar] [CrossRef]
- Agnol, L.D.; Dias, F.T.G.; Ornaghi Jr, H.L.; Sangermano, M.; Bianchi, O. UV–curable waterborne polyurethane coatings: A state–of–the–art and recent advances review. Prog. Org. Coat. 2021, 154, 106156. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Raju, K.V.S.N. Structural engineering of polyurethane coatings for high performance applications. Prog. Polym. Sci. 2007, 32, 352–418. [Google Scholar] [CrossRef]
- Akindoyo, J.O.; Beg, M.D.H.; Ghazali, S.; Islam, M.R.; Jeyaratnam, N.; Yuvaraj, A.R. Polyurethane types, synthesis and applications—A review. RSC Adv. 2016, 6, 114453–114482. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, H.; Hu, G.; Wang, J.; Xin, Y.; Xiang, C.; Zhou, Y. Excellent water resistance and mechanically robust waterborne polyurethane–acrylate based on dithiol post–chain extension. J. Coat. Technol. Res. 2020, 17, 1065–1074. [Google Scholar] [CrossRef]
- Tennebroek, R.; van der Hoeven-van Casteren, I.; Swaans, R.; van der Slot, S.; Stals, P.J.M.; Tuijtelaars, B.; Koning, C. Water-based polyurethane dispersions. Polym. Int. 2019, 68, 832–842. [Google Scholar] [CrossRef]
- Kim, B.K.; Ahn, B.U.; Lee, M.H.; Lee, S.K. Design and properties of UV cured polyurethane dispersions. Prog. Org. Coat. 2006, 55, 194–200. [Google Scholar] [CrossRef]
- Xu, H.P.; Qiu, F.X.; Wang, Y.Y.; Wu, W.L.; Yang, D.; Guo, Q. UV–curable waterborne polyurethane–acrylate: Preparation, characterization and properties. Prog. Org. Coat. 2012, 73, 47–53. [Google Scholar] [CrossRef]
- Honarkar, H. Waterborne polyurethanes: A review. J. Dispers. Sci. Technol. 2018, 39, 507–516. [Google Scholar] [CrossRef]
- Tillet, G.; Boutevin, B.; Ameduri, B. Chemical reactions of polymer crosslinking and post–crosslinking at room and medium temperature. Prog. Polym. Sci. 2011, 36, 191–217. [Google Scholar] [CrossRef]
- Luo, C.H.; Qu, J.Q.; Chen, H.Q. Crosslinking modification of aqueous polyurethane dispersions and its properties. J. Chem. Eng. Chin. Univ. 2009, 23, 650–654. [Google Scholar]
- Hu, M.M.; Shen, Y.D.; Wang, H.H. Effects of cross–Linking on the mechanical and anti–corrosion properties of aziridine/waterborne polyurethane–acrylate. Polym. Mater. Sci. Eng. 2015, 31, 75–80. [Google Scholar]
- Lai, J.Z.; Ling, H.J.; Chen, G.N.; Yeh, J.T.; Chen, K.N. Polymer hybrids from self–emulsified PU anionomer and water–reducible acrylate copolymer via a postcuring reaction. J. Appl. Polym. Sci. 2003, 90, 3578–3587. [Google Scholar] [CrossRef]
- Yuan, C.D.; Wang, M.Y.; Li, H.T.; Wang, Z.W. Preparation and properties of UV–curable waterborne polyurethane–acrylate emulsion. J. Appl. Polym. Sci. 2017, 134, 45208. [Google Scholar] [CrossRef]
- Sultan, M.; Zia, K.M.; Bhatti, H.N.; Jamil, T.; Hussain, R.; Zuber, M. Modification of cellulosic fiber with polyurethane acrylate copolymers. Part I: Physicochemical properties. Carbohydr. Polym. 2012, 87, 397–404. [Google Scholar] [CrossRef]
- Malvern Panalytical. Zetasizernano Series User Manual MAN0485; Malvern Panalytical: Malvern, UK, 2013; pp. 12–13. [Google Scholar]
- Stachurski, J.; MichaLek, M. The effect of the zeta potential on the stability of a non–polar oil–in–Water emulsion. J. Colloid Interface Sci. 1996, 184, 433–436. [Google Scholar] [CrossRef]
- de Morais, J.M.; dos Santos, O.D.H.; Delicato, T.; da Rocha-Filho, P.A. Characterization and evaluation of electrolyte influence on canola oil/water nano–emulsion. J. Sci. Technol. 2006, 27, 1009–1014. [Google Scholar] [CrossRef]
- Ligon–Auer, S.C.; Schwentenwein, M.; Gorsche, C.; Stampfl, J.; Liska, R. Toughening of photo–curable polymer networks: A review. Polym. Chem. 2016, 7, 257–286. [Google Scholar] [CrossRef]
- Xin, H.; Wang, J.; Yang, J.; Cai, J. Effects of epoxy resin on properties of castor oil based WPUA emulsions. J. Chem. Eng. Chin. Univ. 2019, 33, 1516–1522. [Google Scholar]
- Yang, W.; Du, X.S.; Du, Z.L.; Wang, H.B.; Cheng, X. Effect of crosslinking on the surface free energy and surface reorganization of waterborne fluorinated polyurethane. J. Appl. Polym. Sci. 2019, 136, 47167. [Google Scholar] [CrossRef]
- Bai, C.Y.; Zhang, X.Y.; Dai, J.B.; Zhang, C.Y. Water resistance of the membranes for UV curable waterborne polyurethane dispersions. Prog. Org. Coat. 2007, 59, 331–336. [Google Scholar] [CrossRef]
- Xia, Y.; Larock, R.C. Castor-Oil-Based Waterborne Polyurethane Dispersions Cured with an Aziridine-Based Crosslinker. Macromol. Mater. Eng. 2011, 296, 703–709. [Google Scholar] [CrossRef]
- Zhu, K.; Li, X.; Wang, H.; Fei, G.; Li, J. Properties and paper sizing application of waterborne polyurethanemicroemulsions: Effects of extender, cross-linker, and polyol. J. Appl. Polym. Sci. 2016, 133, 43211. [Google Scholar] [CrossRef]
- Lai, J.Z.; Chang, Y.C.; Yeh, J.T.; Chen, K.N. Single component self-curable aqueous-based PU system with new aziridinyl curing agent. J. Appl. Polym. Sci. 2004, 91, 1997–2007. [Google Scholar] [CrossRef]
- Xu, J.C.; Rong, X.S.; Chi, T.Y.; Wang, M.; Wang, Y.; Yang, D.; Qiu, F. Preparation, characterization of UV-Curable Waterborne Polyurethane–Acrylate and the application in metal iron surface protection. J. Appl. Polym. Sci. 2013, 130, 3142–3152. [Google Scholar] [CrossRef]
- Wang, X.; Cui, Y.; Wang, Y.; Ban, T.; Zhang, Y.; Zhang, J.; Zhu, X. Preparation and characteristics of crosslinked fluorinated acrylate modified waterborne polyurethane for metal protection coating. Prog. Org. Coat. 2021, 158, 106371. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).