Plasma-Sprayed Osseoconductive Hydroxylapatite Coatings for Endoprosthetic Hip Implants: Phase Composition, Microstructure, Properties, and Biomedical Functions
Abstract
1. Introduction
2. Why Hydroxylapatite Coatings?
- Enhanced bone apposition rate by osseoinduction, owing to preferential adsorption of bone growth-supporting factors such as BMPs as well as NCPs such as osteocalcin, osteopontin, sialylated glycoproteins, proteoglycans, and several other hormones, chemokines and cytokines;
- Enhanced bone-bonding ability that provides a strong and continuous interface between bone tissue and implant and thus enables the transmission of compressive as well as (limited) tensile and also some shear forces;
- Variable HAp coating thicknesses between 50 and 250 µm can be selected, dependent on medical requirements;
- The option to apply some novel deposition techniques including SPS and SPPS allows the deposition of coatings with thicknesses << 50 µm;
- Acceleration of the healing process when compared to implants without an osseoconductive coating;
- Supporting attachment of the epithelium in the case of transmucosal dental implants;
- Reducing the health risk of potentially toxic heavy metal ions released from the surface of the metallic implant into the periprosthetic tissue and thus minimizing a possible cytotoxic response, and
- Support available by quality control and standards according to ASTM F1185-03 (2014), ASTM F1044-05 (2017), ASTM F1160 (2014), ISO 13179-2: 2018, and others.
3. A Short History of Calcium Orthophosphate Research
4. Hierarchical Structure of Bone
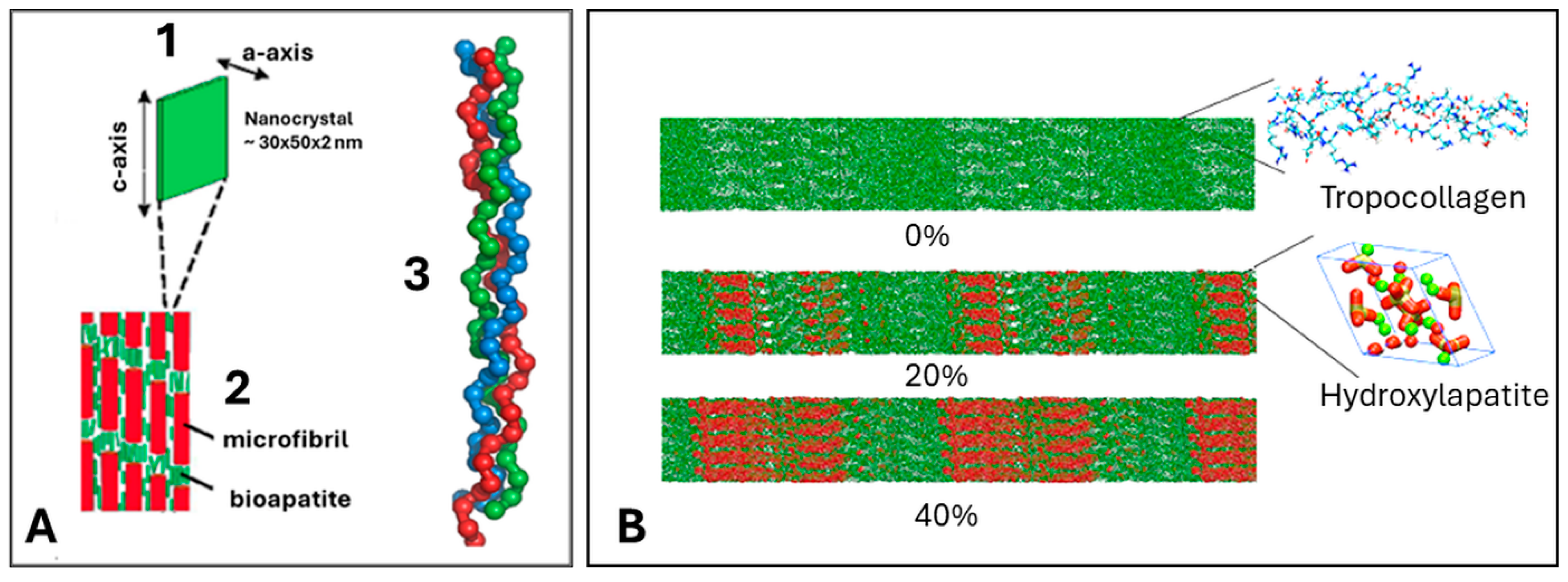
5. Osseoconductive Hydroxylapatite Coatings
5.1. Osseoconduction, Osseoinduction, and Osseointegration
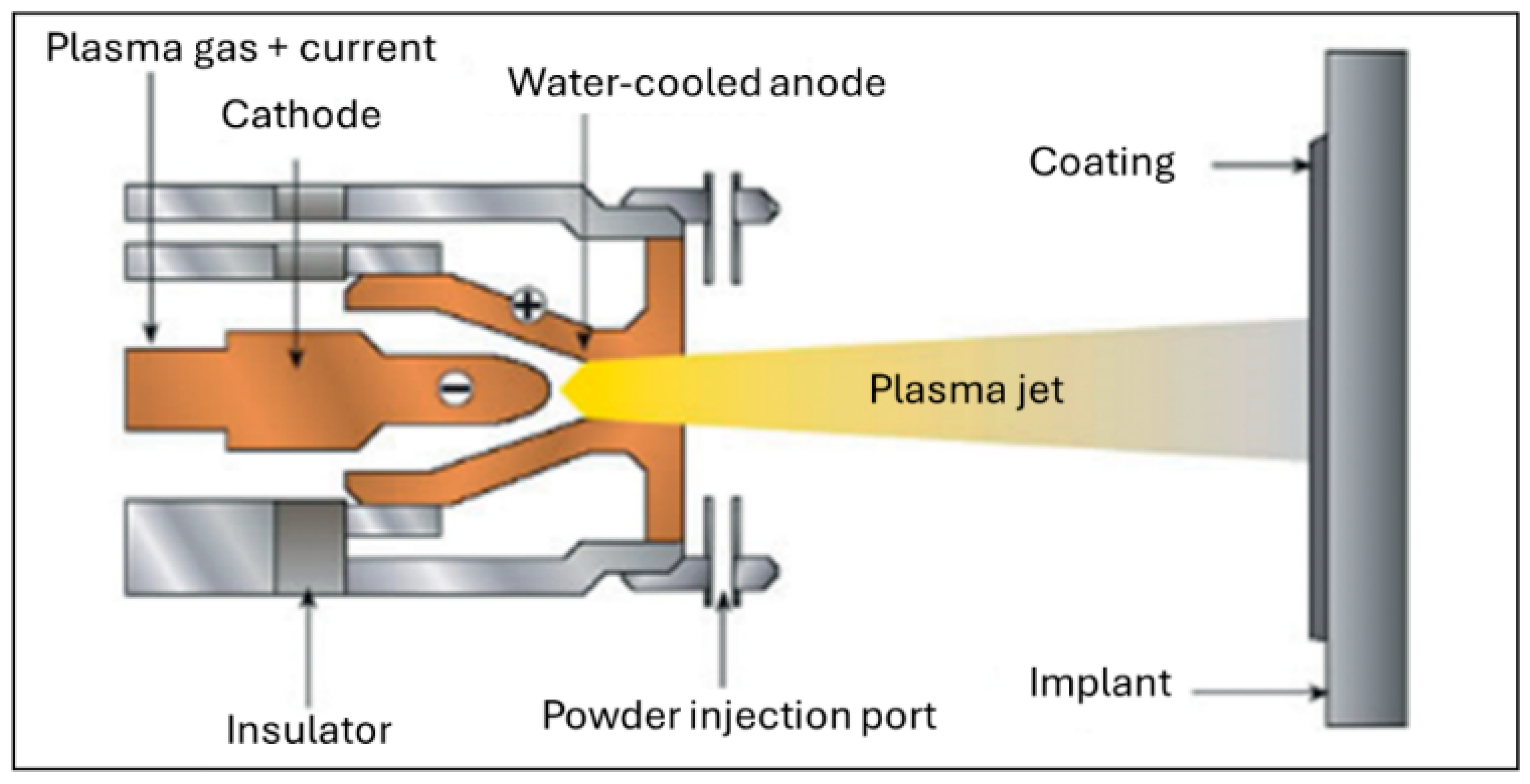
5.2. Deposition Techniques
5.3. Property Requirements and Performance Profile of Hydroxylapatite Coatings
5.3.1. Incongruent Melting and Thermal Decomposition of HAp: Phase Composition
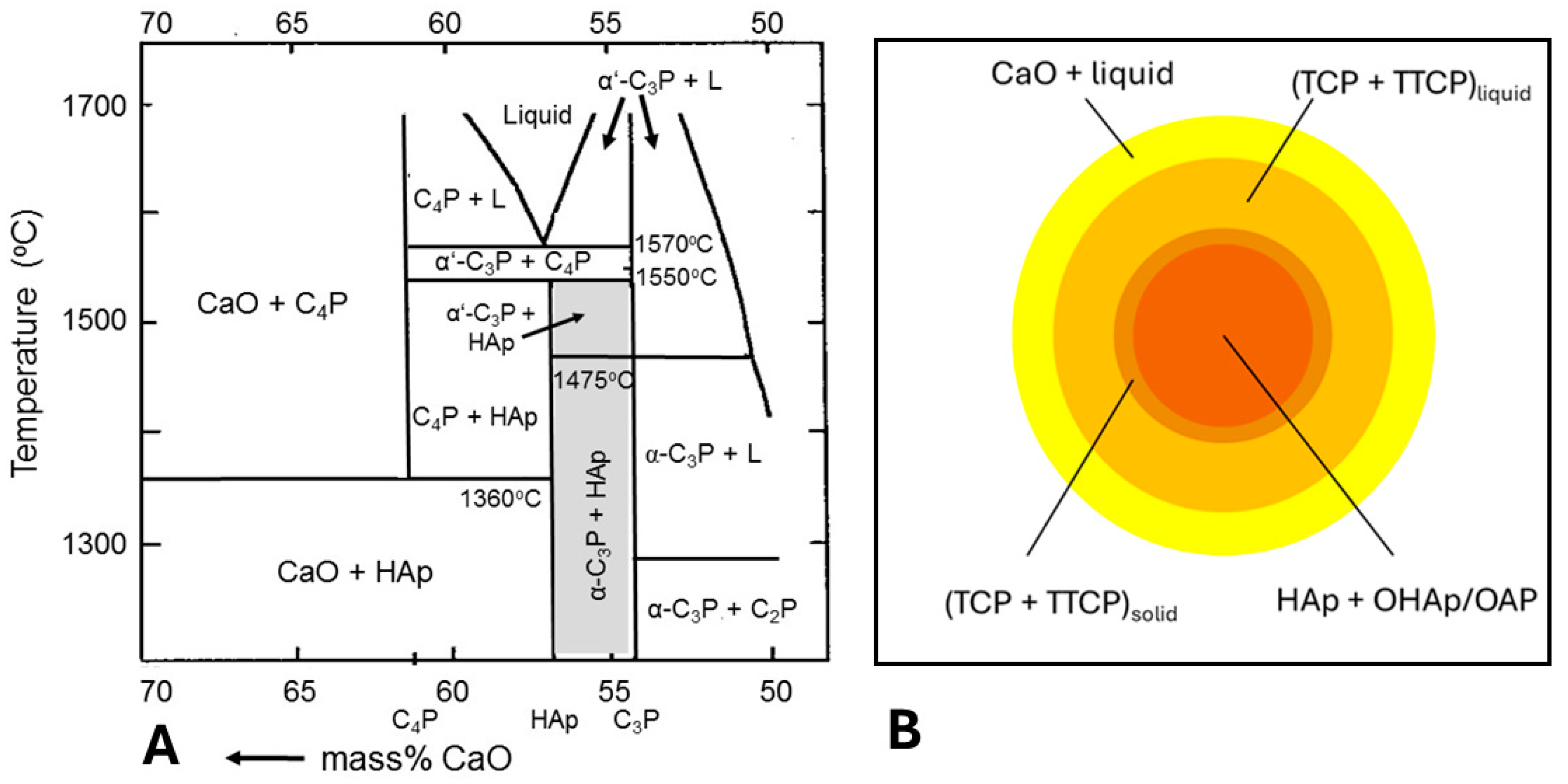
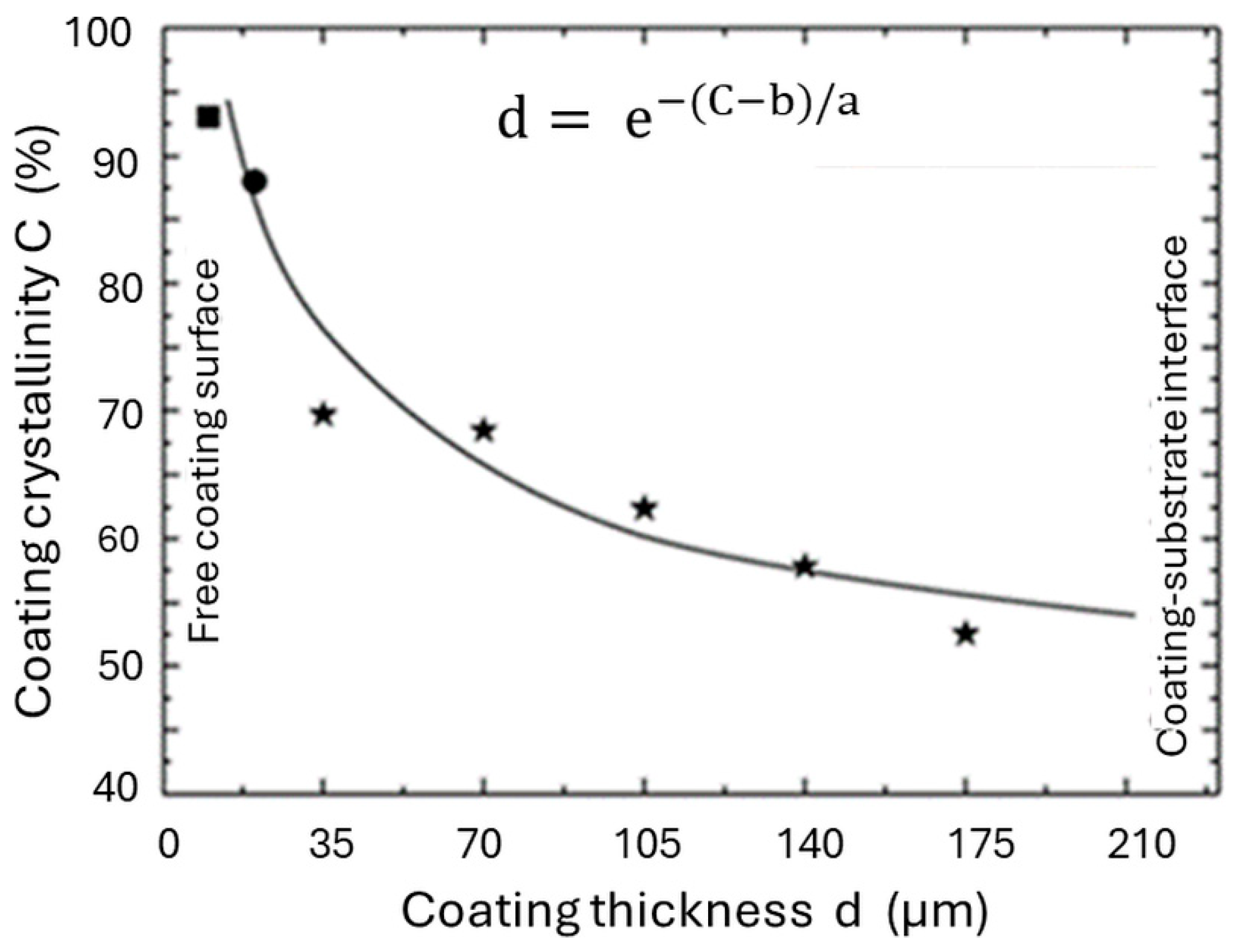
5.3.2. Degree of Crystallinity
- Increase of adhesion to both metal and HAp. For example, a titania bond coat is thought to act as an extension of the native oxide layer on metallic titanium that may interact with HAp to form a thin reaction layer of perovskitic calcium titanate.
- Reduction of the thermal decomposition of HAp by inhibiting the heat flow using a thin titania bond coat film with low thermal conductivity (~1 W/mK) as opposed to a Ti6Al4V substrate (~7 W/mK).
- Reduction of the formation of amorphous phase that forms by a quenching contact immediately at the metal interface. An increase in crystallinity is caused by the thermal barrier function of a bond coat that prolongs solidification time and thus allows the ACP to nucleate apatite and crystallize. Experimental NMR results [50] show that as-sprayed coatings without a bond coat contain only 46 mass% well-ordered HAp at the free coating surface as contrasted with 62 mass% in coatings with a titania bond coat. During incubation for 12 weeks in r-SBF [51], these values increase by dissolving TCP, TTCP, CaO, and ACP phases to 74 mass% and 92 mass%, respectively.
- Reduction of residual coating stresses by reducing the gradient of the coefficient of thermal expansion between the metal substrate and the ceramic overlayer.
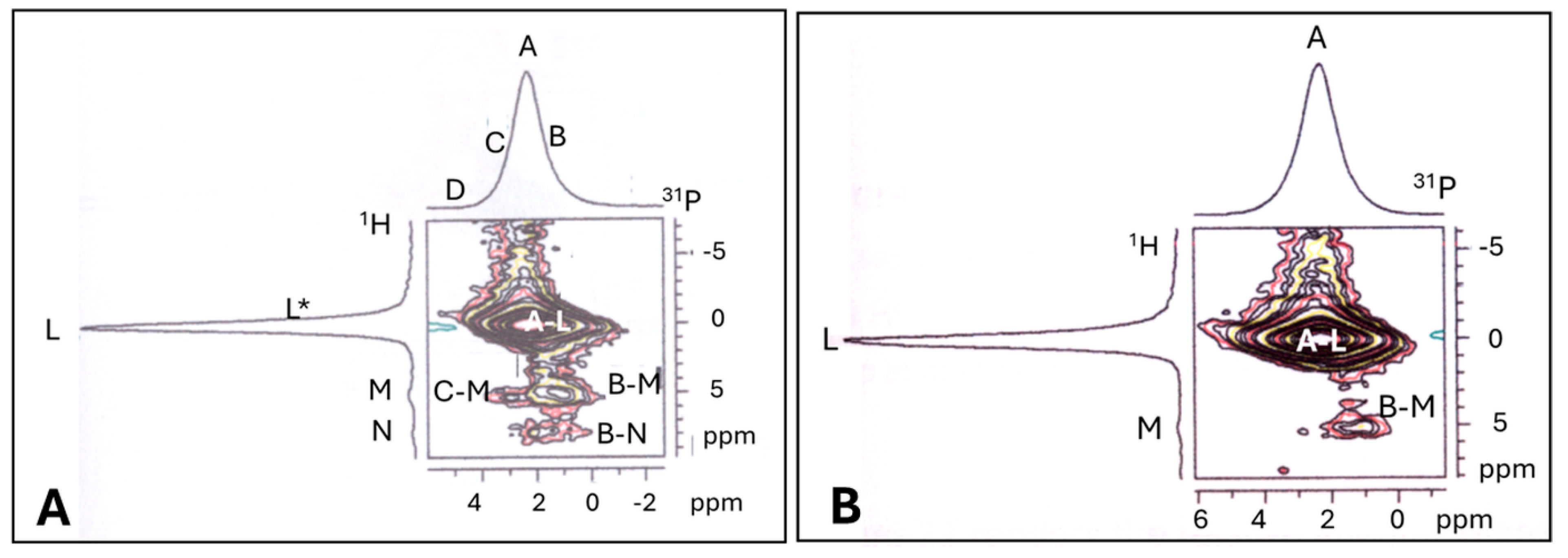
5.3.3. Assessment of Structural Order in Hydroxylapatite Coatings: Raman and NMR Studies
5.3.4. Crystallographic Structure of Hydroxylapatite
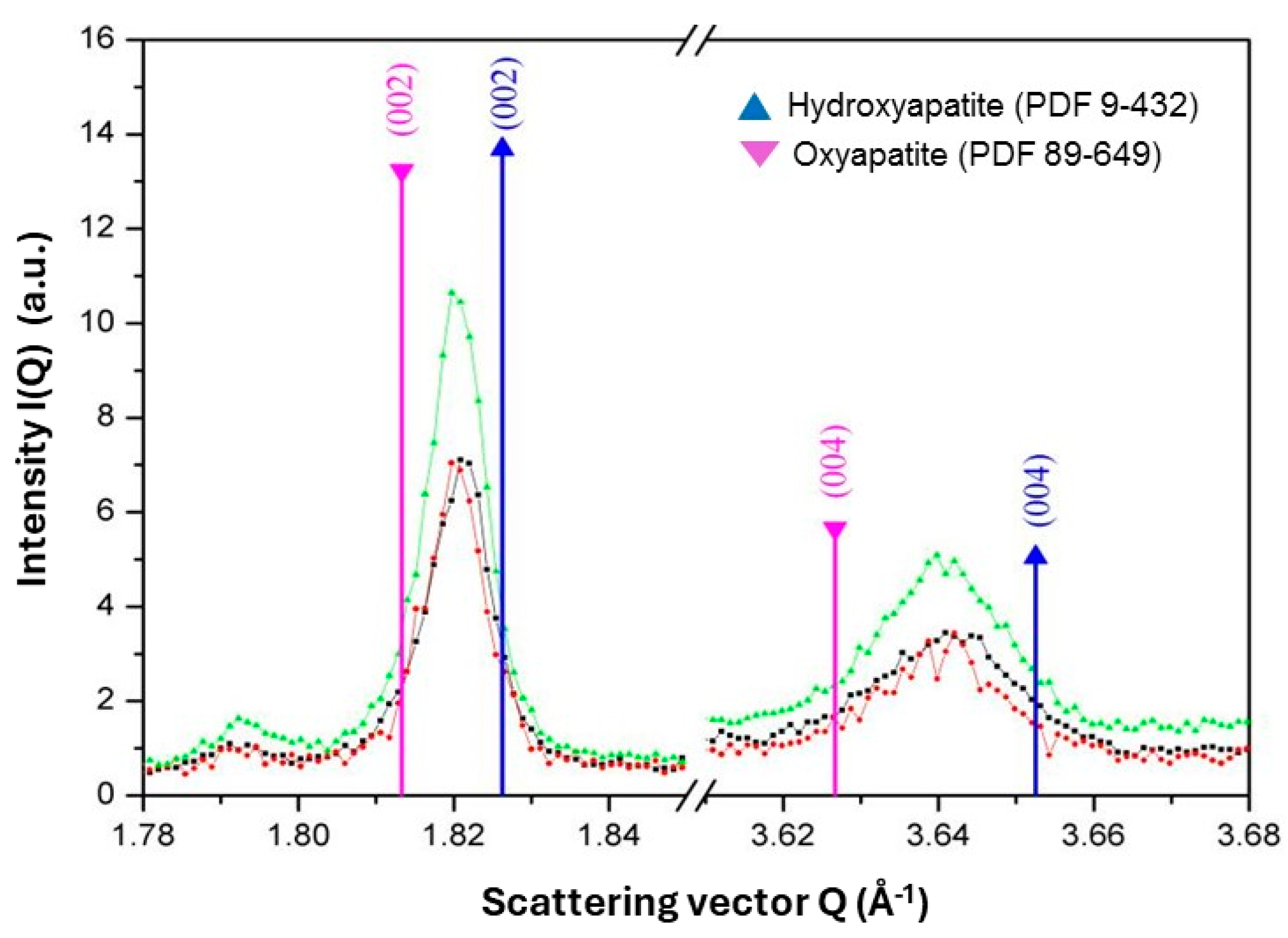
5.3.5. Oxyapatite: Fact or Fiction?
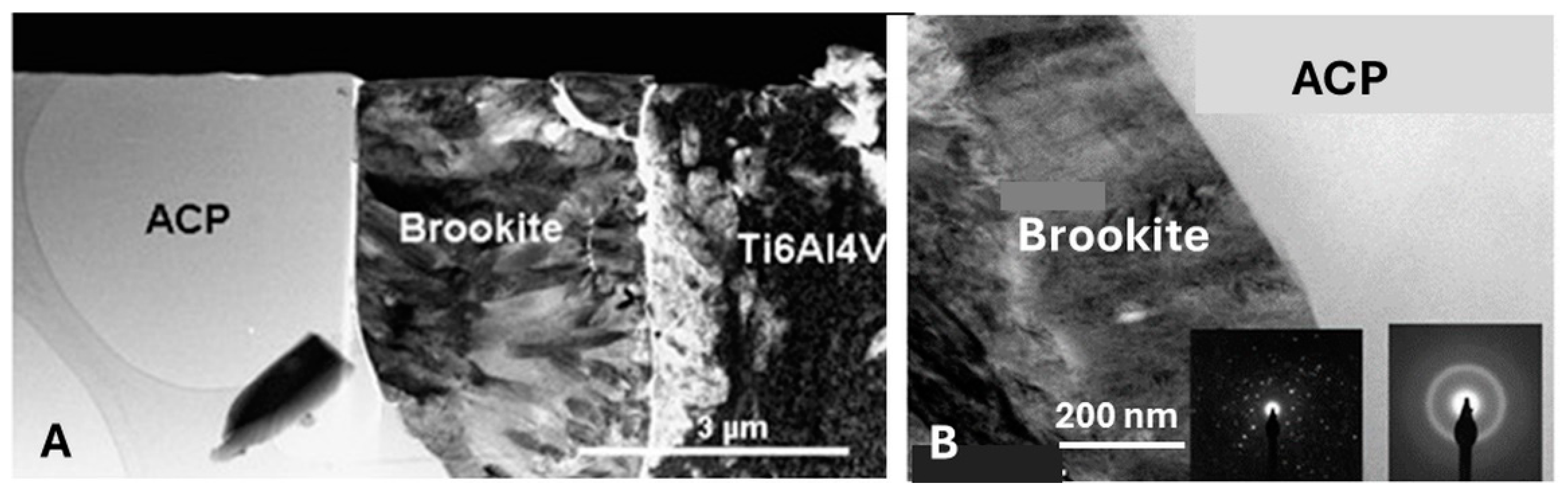
5.3.6. Transformation of Amorphous Calcium Phosphate (ACP)
5.3.7. Coating Porosity, Surface Roughness, and Surface Nanotopography
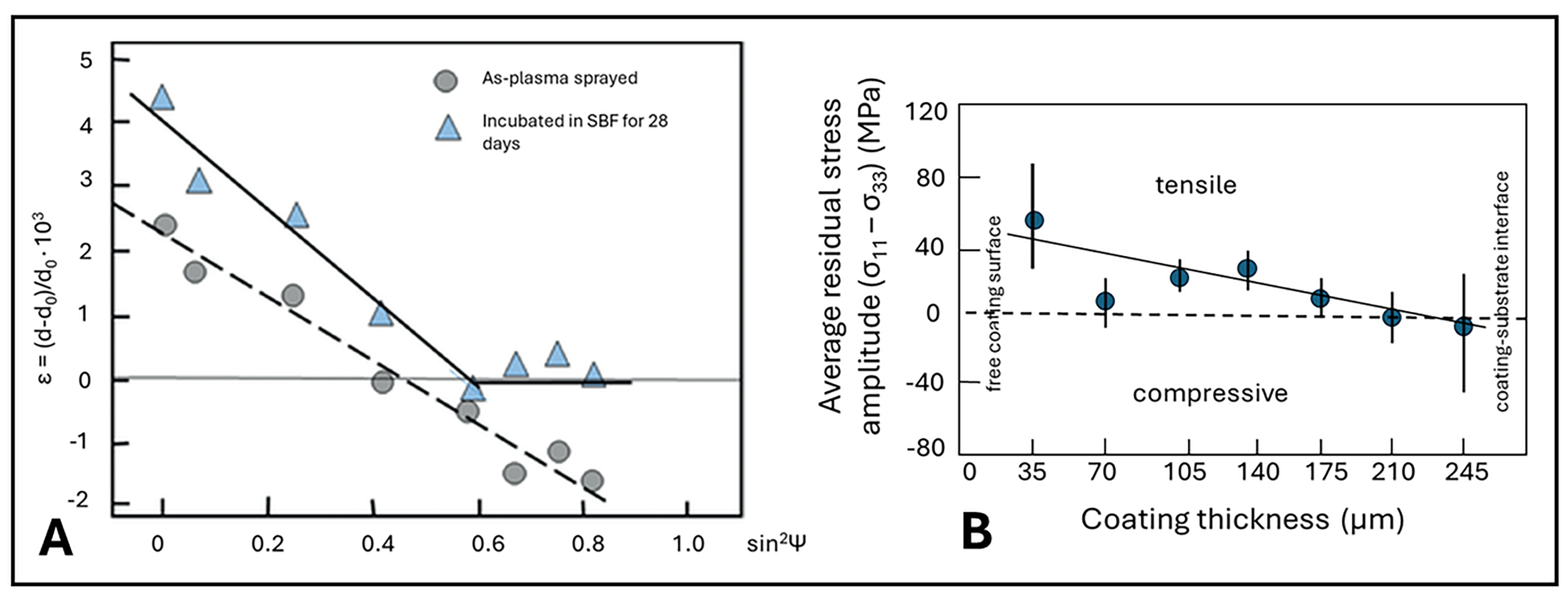
5.3.8. Residual Coating Stresses
5.3.9. Adhesion of Plasma-Sprayed Hydroxylapatite Coatings
5.3.10. Other Implant Surface Functionalization Strategies
6. Concluding Remarks
Funding
Conflicts of Interest
Acronyms
References
- Hegde, V.; Stambough, J.B.; Levine, B.R.; Springer, B.D. Highlights of the American Joint Replacement Registry Annual Report. Arthroplast. Today 2023, 21, 101137. [Google Scholar] [CrossRef]
- Grimberg, A.; Lützner, J.; Melsheimer, O.; Morlock, M.; Steinbrück, A. EPDR Annual Report 2023; EPDR Deutsche Endoprothesenregister gGmbH: Berlin, Germany, 2023; Available online: https://www.eprd.de/en/about-the-eprd/news/article/annual-report-2023 (accessed on 16 March 2024).
- Global Hip Replacement Implants Market Report and Forecast 2023–2031. Available online: https://www.researchandmarkets.com/reports/5797997 (accessed on 16 March 2024).
- Merfort, R.; Maffulli, N.; Hofmann, U.K.; Hildebrand, F.; Simeone, F.; Eschweiler, J.; Migliorini, F. Head, acetabular liner composition, and rate of revision and wear in total hip arthroplasty: A Bayesian network meta-analysis. Sci. Rep. 2023, 13, 2032. [Google Scholar] [CrossRef]
- Heimann, R.B.; Schürmann, N.; Müller, R.T. In vitro and in vivo performance of Ti6Al4V implants with plasma-sprayed osteoconductive hydroxylapatite-bioinert titania bond coat ‘duplex’ systems: An experimental study in sheep. J. Mater. Sci. Mater. Med. 2004, 15, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Itiravivong, P.; Promasa, A.; Laiprasert, T.; Techapongworachai, T.; Kuptnitazsaikul, S.; Thanakit, V.; Heimann, R.B. Comparison of tissue reaction and osteointegration of metal implants between hydroxyapatite/Ti alloy coat: An animal experimental study. J. Medical Assoc. Thail. 2003, 86, S422–S430. [Google Scholar]
- Zhang, D.H.; Chen, Q.; Shi, C.; Chen, M.; Ma, K.; Wan, J.; Liu, R. Dealing with the foreign body response to implanted biomaterials: Strategies and applications of new materials. Adv. Funct. Mater. 2021, 31, 2007226. [Google Scholar] [CrossRef]
- Heimann, R.B.; Lehmann, H.D. Bioceramic Coatings for Medical Implants. Trends and Techniques; Wiley-VCH: Weinheim, Germany, 2015; p. 467. ISBN 978-3-527-334743-9. [Google Scholar]
- Brande, W.T.; Taylor, A.S. Chemistry; Blanchard and Lea: Philadelphia, PA, USA, 1863; p. 696. [Google Scholar]
- Warington, R., Jr. Researches on the phosphates of calcium, and upon the solubility of tricalcic phosphate. J. Chem. Soc. 1866, 19, 296–318. [Google Scholar] [CrossRef]
- Bragg, W.H. Application of the ionisation spectrometer to the determination of the structure of minute crystals. Proc. Phys. Soc. 1921, 33, 222–224. [Google Scholar] [CrossRef]
- Hendricks, S.B.; Hill, W.A.; Jakobs, K.D.; Jefferson, M.E. Structural characteristics of apatite-like substances and composition of phosphate rock and bone as determined from microscopical and X-ray examinations. Ind. Eng. Chem. 1931, 23, 1413–1418. [Google Scholar] [CrossRef]
- Roseberry, H.H.; Hastings, A.B.; Morse, J.K. X-ray analysis of bone and teeth. J. Biol. Chem. 1931, 90, 395–407. [Google Scholar] [CrossRef]
- Trömel, G. Untersuchungen über die Bildung eines halogenfreien Apatits aus basischen Calciumphosphaten. Z. Phys. Chem. A 1932, 158, 422–432. [Google Scholar] [CrossRef]
- Bredig, M.A. Zur Apatitstruktur der anorganischen Knochen- und Zahnsubstanz. Hoppe Seyler’s Z. Physiol. Chem. 1933, 216, 239–243. [Google Scholar] [CrossRef]
- Bredig, M.A.; Franck, H.H.; Füldner, H. Beiträge zur Kenntnis der Kalk-Phosphorsäure-Verbindungen II. Z. Elektrochem. 1933, 39, 959–969. [Google Scholar] [CrossRef]
- De Jong, W.F. La substance minerale dans les os. Recl. Trav. Chim. Pays. Bas. Belg. 1926, 45, 445–448. [Google Scholar] [CrossRef]
- De Leeuw, N.H.; Bowe, J.R.; Rabone, J.A.L. A computational investigation of stoichiometric and calcium-deficient oxy- and hydroxyapatites. Faraday Discuss. 2007, 134, 195–214. [Google Scholar] [CrossRef] [PubMed]
- Alberius Henning, P.; Landa-Canovas, A.; Larsson, A.K.; Lidin, S. The structure of oxyapatite solved by HREM. Acta Cryst. 1999, B55, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Calderin, L.; Stott, M.J.; Rubio, A. Electronic and crystallographic structure of apatites. Phys. Rev. 2003, B67, 134106–134112. [Google Scholar] [CrossRef]
- Gross, K.A.; Pluduma, L. Putting oxyhydroxyapatite into perspective. A pathway to oxyapatite and its application. In Calcium Phosphate. Structure, Synthesis, Properties, and Applications; Heimann, R.B., Ed.; Biochemistry Research Trends Series; Nova Science Publication: New York, NY, USA, 2012; pp. 95–120. ISBN 978-1-62257-299-1. [Google Scholar]
- Heimann, R.B. Characterisation of as-sprayed and incubated hydroxyapatite coatings with high resolution techniques. Mater. Wiss. Werkstofftechn. 2009, 40, 21–30. [Google Scholar]
- Hattori, T.; Iwadate, Y. Hydrothermal preparation of calcium hydroxylapatite powders. J. Am. Ceram. Soc. 1990, 73, 1803–1807. [Google Scholar] [CrossRef]
- Liu, C.; Huang, A.; Shen, W.; Cui, J. Kinetics of hydroxyapatite precipitation at pH 10 and 11. Biomaterials 2001, 23, 301–306. [Google Scholar] [CrossRef]
- Rey, C.; Combes, C.; Drouet, C.; Glimcher, M.J. Bone mineral: Update on chemical composition and structure. Osteopor. Int. 2009, 20, 1013–1021. [Google Scholar] [CrossRef]
- Pasteris, J.D. Structurally incorporated water in bone apatite: A cautionary tale. In Calcium Phosphate. Structure, Synthesis, Properties, and Applications; Heimann, R.B., Ed.; Biochemistry Research Trends Series; Nova Science Publication: New York, NY, USA, 2012; pp. 63–94. ISBN 978-1-62257-299-1. [Google Scholar]
- Heimann, R.B. Materials for Medical Application; Walter de Gruyter GmbH: Berlin, Germany, 2020; p. 615. ISBN 978-3-11-061919-5. [Google Scholar]
- Glimcher, M.J. A basic architectural principle in the organisation of mineralized tissue. Clin. Orthop. 1968, 61, 16–36. [Google Scholar] [CrossRef] [PubMed]
- Florencio-Silva, R.; Rodrigues da Silva Sasso, G.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of bone tissue: Structure, function, and factors that influence bone cells. Biomed. Res. Int. 2015, 2015, 421746. [Google Scholar] [CrossRef] [PubMed]
- Allo, B.A.; Costa, D.O.; Dixon, S.J.; Mequanint, K.; Rizkalla, A.S. Bioactive and biodegradable nanocomposites and hybrid biomaterials for bone regeneration. J. Funct. Mater. 2012, 5, 432–463. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.K.; Gautieri, A.; Chang, S.W.; Buehler, M.J. Molecular mechanics of mineralized collagen fibrils in bone. Nature Comm. 2013, 4, 1724. [Google Scholar] [CrossRef]
- Jarcho, M.; Bolen, C.M.; Thomas, M.B.; Bobick, J.; Kay, J.F.; Doremus, R.H. Hydroxylapatite synthesis and characterization in dense polycrystalline form. J. Mater. Sci. 1976, 11, 2027–2035. [Google Scholar] [CrossRef]
- Ducheyne, P.; Hench, L.L.; Kagan, A.; Martens, M.; Mulier, J.C.; Burssens, A. The effect of hydroxyapatite impregnation on bonding of porous coated implants. J. Biomed. Mater. Res. 1980, 14, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Johansson, C. Osteoinduction, osseoconduction and osseointegration. Eur. Spine J. 2001, 10 (Suppl. S2), S96–S101. [Google Scholar]
- León, B.; Jansen, J.A. Thin Calcium Phosphate Coatings for Medical Implants; Springer: New York, NY, USA, 2009; p. 328. ISBN 978-0-387-77718-4. [Google Scholar]
- Heimann, R.B. Plasma Spray Coating. Principles and Applications, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2008; ISBN 978-3-527-32050-9. [Google Scholar]
- Heimann, R.B. Hydroxylapatite coatings: Applied mineralogy research in the bioceramics field. In Highlights in Applied Mineralogy; Heuss-Aßbichler, S., Amthauer, G., John, M., Eds.; Walter de Gruyter GmbH: Berlin, Germany, 2018; pp. 301–316. ISBN 978-3-11-049122-7. [Google Scholar]
- Heimann, R.B. The nature of plasma spraying. Coatings 2023, 13, 622. [Google Scholar] [CrossRef]
- Sun, L. Thermal spray coatings on orthopedic devices: When and how the FDA reviews your coatings. J. Thermal Spray Technol. 2018, 27, 1280–1290. [Google Scholar] [CrossRef]
- Graßmann, O.; Heimann, R.B. Compositional and microstructural changes of engineered plasma-sprayed hydroxyapatite coatings on Ti6Al4V substrates during incubation in protein-free simulated body fluid. J. Biomed. Mater. Res. 2000, 53, 685–693. [Google Scholar] [CrossRef]
- Heimann, R.B.; Graßmann, O.; Zumbrink, T.; Jennissen, H.P. Biomimetic processes during in vitro leaching of plasma-sprayed hydroxyapatite coatings for endoprosthetic application. Mater. Wiss. Werkstofftechn. 2001, 32, 913–921. [Google Scholar] [CrossRef]
- Dyshlovenko, S.; Pateyron, B.; Pawlowski, L.; Murano, D. Numerical simulation of hydroxyapatite powder behaviour in plasma jet. Surf. Coat. Technol. 2004, 179, 110–117. [Google Scholar] [CrossRef]
- Riboud, P.V. Composition et stabilité des phases a structure d’apatite dans le systeme CaO-P2O5-oxide de Fer-H2O a haute temperature. Ann. Chim. 1973, 8, 381–390. [Google Scholar]
- Frayssinet, P.; Tourenne, F.; Roquet, N.; Conte, P.; Delga, C.; Bonel, G. Comparative biological properties of HA plasma-sprayed coatings having different crystallinities. J. Mater. Sci. Mater. Med. 1994, 5, 11–17. [Google Scholar] [CrossRef]
- De Santis, D.; Guerriero, C.; Nocini, P.F.; Ungersbock, A.; Richards, G.; Gotte, P.; Armato, U. Adult human bone cells from jaw bones cultured on plasma-sprayed or polished surfaces of titanium or hydroxyapatite discs. J. Mater. Sci. Mater. Med. 1996, 7, 21–28. [Google Scholar] [CrossRef]
- Ntsoane, T.P.; Topić, M.; Härting, M.; Heimann, R.B.; Theron, C. Spatial and depth-resolved studies of air plasma-sprayed hydroxyapatite coatings by means of diffraction techniques: Part 1. Surf. Coat. Technol. 2016, 294, 153–163. [Google Scholar] [CrossRef]
- Hesse, C.; Hengst, M.; Kleeberg, R.; Götze, J. Influence of experimental parameters on spatial phase distribution in as-sprayed and incubated hydroxyapatite coatings. J. Mater. Sci. Mater. Med. 2008, 19, 3225–3241. [Google Scholar] [CrossRef]
- Park, E.; Condrate, R.A.; Lee, D.H. Infrared spectral investigation of plasma spray coated hydroxyapatite. Mater. Lett. 1998, 36, 38–43. [Google Scholar] [CrossRef]
- Heimann, R.B. Novel approaches towards design and biofunctionality of plasma-sprayed osteoconductive calcium phosphate coatings for biomedical implants: The concept of bond coats. In Trends in Biomaterials Research; Pannone, P.J., Ed.; Nova Science Publishers Inc.: New York, NY, USA, 2007; pp. 1–80. ISBN 978-1-60021-361-8. [Google Scholar]
- Heimann, R.B.; Tran, H.V.; Hartmann, P. Laser-Raman and nuclear magnetic resonance (NMR) studies on plasma-sprayed hydroxyapatite coatings: Influence of bioinert bond coats on phase composition and resorption kinetics in simulated body fluid. Mater. Wiss. Werkstofftechn. 2003, 34, 1163–1169. [Google Scholar] [CrossRef]
- Kim, H.M.; Miyazaki, M.; Kokubo, T.; Nakamura, T. Revised simulated body fluid. Key Eng. Mater. 2001, 192/195, 47–50. [Google Scholar] [CrossRef]
- Park, E.; Condrate, R.A.; Lee, D.H.; Kociba, K.; Gallagher, P.K. Characterization of hydroxyapatite before and after plasma spraying. J. Mater. Sci. Mater. Med. 2002, 13, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Demnati, I.; Parco, M.; Grossin, D.; Fagoaga, I.; Drouet, C.; Barykin, G.; Combes, C.; Braceras, I.; Goncalves, S.; Rey, C. Hydroxyapatite coating on titanium by a low energy plasma spraying mini-gun. Surf. Coat. Technol. 2012, 206, 2346–2353. [Google Scholar] [CrossRef]
- Demnati, I.; Grossin, D.; Combes, C.; Rey, C. Plasma-sprayed apatite coatings: Review of physical-chemical aspects and their biological consequences. J. Med. Biol. Eng. 2014, 34, 1–7. [Google Scholar] [CrossRef]
- Heimann, R.B.; Vu, T.A. Low-pressure plasma-sprayed (LPPS) bioceramic coatings with improved adhesion strength and resorption resistance. J. Thermal Spray Technol. 1997, 6, 145–149. [Google Scholar] [CrossRef]
- Cusco, R.; Guitian, F.; de Aza, S.; Artus, L. Differentiation between hydroxyapatite and ß-tricalcium phosphate by means of µ-Raman spectroscopy. J. Eur. Ceram. Soc. 1998, 18, 1301–1305. [Google Scholar] [CrossRef]
- Shamray, V.F.; Sirotinkin, V.P.; Kalita, V.I.; Komlev, V.S.; Barinov, S.M.; Fedotov, A.Y.; Gordeev, A.S. Studies of the crystal structure of hydroxyapatite in plasma coating. Surf. Coat. Technol. 2019, 372, 201–208. [Google Scholar] [CrossRef]
- Hartmann, P.; Jäger, C.; Barth, S.; Vogel, J.; Meyer, K. Solid state NMR, X-ray diffraction, and infrared characterization of local structure in heat-treated oxyhydroxylapatite microcrystals: An analogy of the thermal deposition of hydroxyapatite during plasma-spray procedure. J. Solid State Chem. 2001, 160, 460–468. [Google Scholar] [CrossRef]
- Jäger, C.; Welzel, T.; Meyer-Zaika, W.; Epple, M. A solid-state NMR investigation of the structure of nanocrystalline hydroxyapatite. Magn. Reason. Chem. 2006, 44, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.H.V. Investigation into the Thermal Dehydroxylation and Decomposition of Hydroxylapatite during Atmospheric Plasma Spraying: NMR and Raman Spectroscopic Study of as-Sprayed Coatings and Coatings Incubated in Simulated Body Fluid. Ph.D. Thesis, Department of Mineralogy, Technische Universität Bergakademie, Freiberg, Germany, 2004. [Google Scholar]
- Hartmann, P.; Barth, S.; Vogel, J.; Jäger, C. Investigation of structural changes in plasma-sprayed hydroxy/apatite coatings. In Applied Mineralogy in Research, Economy, Technology, Ecology and Culture; Rammlmair, D., Mederer, J., Oberthür, T., Heimann, R.B., Pentinghaus, H., Eds.; A.A. Balkema: Rotterdam, The Netherlands, 2000; Volume 1, pp. 147–150. ISBN 90-5809-164-3. [Google Scholar]
- LeGeros, R.Z. Formation and transformation of calcium phosphates: Relevance to vascular calcification. Z. Kardiol. 2001, 90 (Suppl. S3), 116–124. [Google Scholar] [CrossRef]
- von Euw, S.; Wang, Y.; Laurent, G.; Drouet, C.; Babonneau, F.; Nassif, N.; Azaïs, T. Bone mineral: New insights into its chemical composition. Sci. Rep. 2019, 9, 8456. [Google Scholar] [CrossRef]
- Posner, A.S.; Perloff, A.; Diorio, A.F. Refinement of the hydroxyapatite structure. Acta Cryst. 1958, 11, 308–309. [Google Scholar] [CrossRef]
- Heimann, R.B. Plasma-sprayed hydroxylapatite coatings as biocompatible intermediaries between inorganic implant surfaces and living tissue. J. Thermal Spray Technol. 2018, 27, 1212–1237. [Google Scholar] [CrossRef]
- Laskus, A.; Kolmas, J. Ionic substitution in non-apatitic calcium phosphates. Int. J. Mol. Sci. 2017, 18, 2542. [Google Scholar] [CrossRef] [PubMed]
- Graziani, G.; Boi, M.; Bianchi, M. A review on ionic substitution in hydroxyapatite thin films. Coatings 2018, 8, 269. [Google Scholar] [CrossRef]
- Trombe, J.C.; Montel, C. Sur la préparation del’oxyapatite phospho-calcique. C. R. Acad. Sci. Paris 1971, 273, 462–465. [Google Scholar]
- Takahashi, T.; Tanase, S.; Yamamoto, O. Electrical conductivity of some hydroxyapatites. Electrochim. Acta 1978, 23, 369–373. [Google Scholar] [CrossRef]
- Heimann, R.B.; Wirth, R. Formation and transformation of amorphous calcium phosphates on titanium alloy surfaces during atmospheric plasma spraying and their subsequent in vitro performance. Biomaterials 2006, 27, 823–831. [Google Scholar] [CrossRef]
- Weng, J.; Liu, Q.; Wolke, C.D.; Zhang, D.; de Groot, K. The role of amorphous phase in nucleating bone-like apatite on plasma-sprayed hydroxyapatite coating in simulated body fluid. J. Mater. Sci. Letters 1997, 16, 335–337. [Google Scholar] [CrossRef]
- Cook, S.D.; Thomas, K.A.; Kay, J.F.; Jarcho, M. Hydroxyapatite coated titanium for orthopaedic implant applications. Clin. Orthop. 1988, 232, 225–243. [Google Scholar] [CrossRef]
- Yang, C.Y.; Wang, B.C.; Chang, E.; Wu, J.D. Bond degradation at the plasma-sprayed HA coating/Ti-6Al-4V alloy interface: An in vitro study. J. Mater. Sci. Mater. Med. 1995, 6, 258–265. [Google Scholar] [CrossRef]
- Lemons, J.E. Biodegradation and wear of total joint replacements. In Bone Implant Interface; Cameron, H.U., Ed.; Mosby: Baltimore, Germany, 1994; pp. 307–317. [Google Scholar]
- Wintermantel, E.; Ha, S.W. Biokompatible Werkstoffe und Bauweisen. In Implantate für Medizin und Umwelt; Springer: Berlin/Heidelberg, Germany, 1996; p. 225. [Google Scholar]
- Li, H.; Ma, Y.L.; Zhao, Z.C.; Tian, Y.L. Fatigue behavior of plasma sprayed structural-grade hydroxyapatite coating under simulated body fluid. Surf. Coat. Technol. 2019, 368, 110–118. [Google Scholar] [CrossRef]
- Gross, K.A.; Saber-Samandari, S. Revealing mechanical properties of a suspension plasma sprayed hydroxyapatite coating with nanoindentation. Surf. Coat. Technol. 2009, 203, 2995–2999. [Google Scholar] [CrossRef]
- Wu, F.; Huang, Y.; Song, L.; Liu, X.; Xiao, Y.; Feng, J.; Chen, J. Method for Preparing Porous Hydroxyapatite Coatings by Suspension Plasma Spraying. U.S. Patent 8877283 B2, 4 November 2014. [Google Scholar]
- Reisel, G.; Heimann, R.B. Correlation between surface roughness of plasma-sprayed chromium oxide coatings and powder grain size distribution: A fractal approach. Surf. Coat. Technol. 2004, 185, 215–221. [Google Scholar] [CrossRef]
- Heimann, R.B. On the self-affine fractal geometry of plasma-sprayed surfaces. J. Thermal Spray Technol. 2011, 20, 898–908. [Google Scholar] [CrossRef]
- Gentile, F.; Tirinato, I.; Battista, E.; Causa, F.; Liberale, C.; di Fabrizio, E.M.; Decuzzi, P. Cells preferentially grow on rough substrates. Biomaterials 2010, 31, 7205–7212. [Google Scholar] [CrossRef] [PubMed]
- Gittens, R.A.; Olivares-Navarrete, R.; Schwartz, Z.; Boyan, B.D. Implant osseointegration and the role of microroughness and nanostructures: Lessons for spine implants. Acta Biomater. 2014, 10, 3363–3371. [Google Scholar] [CrossRef]
- Liu, Y.; Rath, B.; Tingart, M.; Eschweiler, J. Role of implant modification in osseointegration: A systematic review. J. Biomed. Mater. Res. 2019, 108, 470–484. [Google Scholar] [CrossRef]
- Dalby, M.J.; McCloy, D.; Robertson, M.; Wilkinson, C.D.; Oreffo, R.O. Osteoprogenitor response to defined topographies with nanoscale depths. Biomaterials 2006, 27, 1306–1315. [Google Scholar] [CrossRef]
- Ross, A.M.; Jiang, Z.; Bastmeyer, M.; Lahann, J. Physical aspects of cell culture substrates: Topography, roughness, and elasticity. Small 2012, 8, 336–355. [Google Scholar] [CrossRef] [PubMed]
- Heimann, R.B.; Kleiman, J.I. Shock-induced growth of superhard materials. In Crystals, Growth, Properties, and Applications; Freyhardt, H.C., Ed.; Springer: Berlin/Heidelberg, Germany, 1988; Volume 11, pp. 1–73. ISBN 3-540-18602-6. [Google Scholar]
- Gill, S.C.; Clyne, T.W. Stress distribution and material response in thermal spraying of metallic and ceramic deposits. Metall. Trans. 1990, B21, 377–385. [Google Scholar] [CrossRef]
- Kuroda, S.; Clyne, T.W. The quenching stress in thermally sprayed coatings. Thin. Solid Film. 1991, 200, 49–66. [Google Scholar] [CrossRef]
- Kuroda, S.; Dendo, T.; Kitahara, S. Quenching stress in plasma sprayed coatings and its correlation with the deposit microstructure. J. Thermal Spray Technol. 1995, 4, 75–84. [Google Scholar] [CrossRef]
- Matejicek, J.; Sampath, S. Intrinsic residual stresses in single splats produced by thermal spray processes. Acta Mater. 2001, 49, 1993–1999. [Google Scholar] [CrossRef]
- Matejicek, J.; Sampath, S. In situ measurement of residual stresses and elastic moduli in thermal sprayed coatings. Part 1: Apparatus and analysis. Acta Mater. 2003, 51, 863–872. [Google Scholar] [CrossRef]
- Stammeier, J.A.; Purgstaller, B.; Hippler, D.; Mavromatis, V.; Dietzel, M. In-situ Raman spectroscopy of amorphous calcium phosphate to crystalline hydroxyapatite transformation. MethodX 2018, 5, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Topíc, M.; Ntsoane, T.; Hüttel, T.; Heimann, R.B. Microstructural characterisation and stress determination in as-plasma sprayed and incubated bioconductive hydroxyapatite coatings. Suf. Coat. Technol. 2006, 201, 3633–3641. [Google Scholar] [CrossRef]
- Heimann, R.B.; Ntsoane, T.; Pineda-Vargas, C.A.; Przybylowicz, W.J.; Topíc, M. Biomimetic formation of hydroxyapatite investigated by analytical techniques with high resolution. J. Mater. Sci. Mater, Med. 2008, 19, 3295–3302. [Google Scholar] [CrossRef] [PubMed]
- Heimann, R.B.; Graßmann, O.; Hempel, M.; Bucher, R.; Härting, M. Phase content, resorption resistance and residual stresses of bioceramic coatings. In Applied Mineralogy in Research, Economy, Technology, Ecology and Culture; Rammlmair, D., Mederer, J., Oberthür, T., Heimann, R.B., Pentinghaus, H., Eds.; A.A. Balkema: Rotterdam, The Netherlands, 2000; Volume 1, pp. 155–158. ISBN 90-5809-164-3. [Google Scholar]
- Geesink, R.G. Hydroxyapatite-coated total hip prostheses two-year clinical and roentgenographic results of 100 cases. Clin. Orthop. Relat. Res. 1990, 261, 39–58. [Google Scholar] [CrossRef]
- ISO 12891-2; Retrieval and Analysis of Surgical Implants. Part 2: Analysis of Retrieved Surgical Implants. International Organization for Standardization: Geneva, Switzerland, 2020.
- ISO 13779-2; Implants for Surgery-Hydroxyapatite. Part 2: Coatings of Hydroxyapatite. International Organization for Standardization: Geneva, Switzerland, 2018.
- Heimann, R.B. Plasma-sprayed hydroxyapatite-based coatings: Chemical, mechanical, microstructural, and biomedical properties. J. Thermal Spray Technol. 2016, 25, 827–850. [Google Scholar] [CrossRef]
- Lacombe, R. Adhesion Measurement Methods: Theory and Practice; CRC Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- Filiaggi, M.J.; Coombs, N.A.; Pilliar, R.M. Characterization of the interface in the plasma-sprayed HApcCoating/Ti-6Al-4V implant system. J. Biomed. Mater. Res. 1991, 25, 1211–1229. [Google Scholar] [CrossRef]
- Webster, T.J.; Ergun, C.; Doremus, R.H.; Lanford, W.A. Increased osteoblast adhesion on titanium-coated hydroxylapatite that forms CaTiO3. J. Biomed. Mater. Res. A 2003, 67, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Zhou, Y.; Jia, D.; Wang, Y. Structure of calcium titanate/titania bioceramic composite coatings on titanium alloy and apatite deposition on their surfaces in a simulated body fluid. Surf. Coat. Technol. 2007, 201, 8715–8722. [Google Scholar] [CrossRef]
- Zhang, B.G.X.; Myers, D.E.; Wallace, G.G.; Brandt, M.; Choong, P.F. Bioactive coatings for orthopaedic implants—recent trends in development of implant coatings. Int. J. Mol. Sci. 2014, 15, 11878–11921. [Google Scholar] [CrossRef]
- Fujibayashi, S.; Neo, M.; Kim, H.M.; Kokubo, T.; Nakamura, T. Osteoinduction of porous bioactive titanium metal. Biomaterials 2004, 25, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.; Akhavan, B.; Wise, S.G.; Bilek, M.M.M. A review of biomimetic surface functionalization for bone-integrating orthopedic implants: Mechanisms, current approaches, and further directions. Progr. Mater. Sci. 2019, 106, 100588. [Google Scholar] [CrossRef]
- Oh, S.; Brammer, K.S.; Li, Y.S.; Teng, D.; Engler, A.J.; Chien, S.; Jin, S. Stem cell fate dictated solely by altered nanotube dimension. Proc. Natl. Acad. Sci. USA 2009, 106, 2130–2135. [Google Scholar] [CrossRef]
- Shimazaki, T.; Miyamoto, H.; Ando, Y.; Noda, I.; Yonekura, Y.; Kawano, S.; Miyazaki, M.; Mawatari, M.; Hotokebuchi, T. In vivo antibacterial and silver-releasing properties of novel thermal sprayed silver-containing hydroxyapatite coating. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 92, 386–389. [Google Scholar] [CrossRef]
- Rabea, E.I.; Badawy, M.E.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: Applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Neut, D.; van de Belt, H.; Stokroos, I.; van Horn, J.R.; van der Mei, H.C.; Busscher, H.J. Biomaterial-associated infection of gentamicin-loaded PMMA beads in orthopaedic revision surgery. J. Antimicrob. Chemother. 2001, 47, 885–891. [Google Scholar] [CrossRef]
- Pezzotti, G.; Boschetto, F.; Ohgitani, E.; Fujita, Y.; Shin-Ya, M.; Adachi, T.; Yamamoto, T.; Kanamura, N.; Marin, E.; Zhu, W.; et al. Mechanism of instantaneous inactivation of SARS-CoV-2 by silicon nitride bioceramics. Mater. Today Bio. 2021, 12, 100144. [Google Scholar] [CrossRef]
- Heimann, R.B. Silicon nitride ceramics: Structure, synthesis, properties, and biomedical applications. Materials 2023, 16, 5142. [Google Scholar] [CrossRef] [PubMed]
- Rammelt, S.; Schulze, E.; Bernhardt, R.; Hanisch, U.; Scharnweber, D.; Worch, H.; Zwipp, H.; Biewener, A. Coating of titanium implants with type-I collagen. J. Orthop. Res. 2004, 22, 1025–1034. [Google Scholar] [CrossRef] [PubMed]
- Bitschnau, A.; Alt, V.; Bohner, F.; Heerich, K.E.; Margesin, E.; Hartmann, S.; Sewing, A.; Meyer, C.; Wenisch, S.; Schnettler, R. Comparison of new bone formation, implant integration, and biocompatibility between RGD-hydroxyapatite and pure hydroxyapatite coating for cementless joint prostheses—An experimental study in rabbits. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 88, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Lobel, K.D.; Hench, L.L. In vitro adsorption and activity of enzymes on reaction layers of bioactive glass substrates. J. Biomed. Mater. Res. 1998, 39, 575–579. [Google Scholar] [CrossRef]
- Mohseni, E.; Zalnezhad, E.; Bushroa, A.R. Comparative investigation on the adhesion of hydroxyapatite coating on Ti-6Al-4V implant: A review paper. Int. J. Adhes. 2014, 48, 238–257. [Google Scholar] [CrossRef]
- Harazawa, K.; Cousins, B.; Roach, P.; Fernandez, A. Modification of the surface nanotopography of implant devices: A translational perspective. Mater. Today Bio. 2021, 12, 100152. [Google Scholar] [CrossRef]
- Joshi, M.U.; Kulkarni, S.P.; Choppadandi, M.; Keerthana, M.; Kapusetti, G. Current state of art smart coatings for orthopedic implants: A comprehensive review. Smart Mater. Med. 2023, 4, 661–679. [Google Scholar] [CrossRef]










| Step 1: | Ca10(PO4)6(OH)2 | → | Ca10(PO4)6(OH)2−xOx□x + xH2O | Oxyhydroxylapatite (OHAp) |
| Step 2: | Ca10(PO4)6(OH)2−xOx□x | → | Ca10(PO4)6Ox□x + (1 − x)H2O | Oxyapatite (OAp) |
| Step 3: | Ca10(PO4)6Ox□x | → | 2 Ca3(PO4)2 + Ca4O(PO4)2 | TCP + TTCP (C3P + C4P) |
| Step 4a: | Ca3(PO4)2 | → | 3 CaO + P2O5 | Stepwise decomposition of TCP and TTCP |
| Step 4b: | Ca4O(PO4)2 | → | 4 CaO + P2O5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heimann, R.B. Plasma-Sprayed Osseoconductive Hydroxylapatite Coatings for Endoprosthetic Hip Implants: Phase Composition, Microstructure, Properties, and Biomedical Functions. Coatings 2024, 14, 787. https://doi.org/10.3390/coatings14070787
Heimann RB. Plasma-Sprayed Osseoconductive Hydroxylapatite Coatings for Endoprosthetic Hip Implants: Phase Composition, Microstructure, Properties, and Biomedical Functions. Coatings. 2024; 14(7):787. https://doi.org/10.3390/coatings14070787
Chicago/Turabian StyleHeimann, Robert B. 2024. "Plasma-Sprayed Osseoconductive Hydroxylapatite Coatings for Endoprosthetic Hip Implants: Phase Composition, Microstructure, Properties, and Biomedical Functions" Coatings 14, no. 7: 787. https://doi.org/10.3390/coatings14070787
APA StyleHeimann, R. B. (2024). Plasma-Sprayed Osseoconductive Hydroxylapatite Coatings for Endoprosthetic Hip Implants: Phase Composition, Microstructure, Properties, and Biomedical Functions. Coatings, 14(7), 787. https://doi.org/10.3390/coatings14070787


.jpg)



