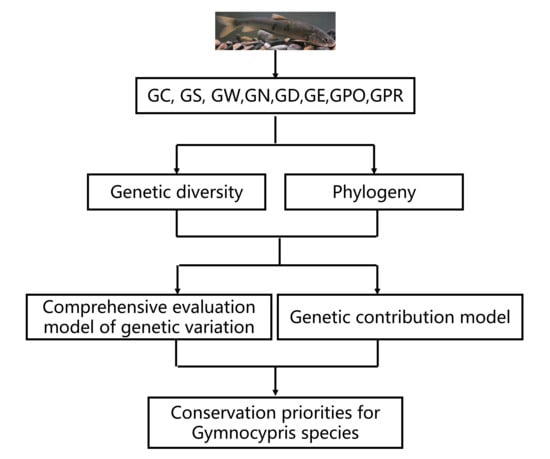
4.1. Assessment of Gymnocypris Species for Priority Conservation on the Basis of Genetic Diversity
The
Gymnocypris species belong to the subfamily Schizothoracinae, are endemic to the QTP river system, are relatively slow-growing, tolerant to high salinity and low temperatures, and have reproductive migratory characteristics rarely found in plateau fishes [
14]. The distribution and evolution of fishes are limited by water systems, making
Gymnocypris species an ideal model for biogeographic studies. Recently, the wild resources of
Gymnocypris species have been declining due to environmental degradation, artificial fishing, and the low recovery capacity of the population itself, so they were listed as endangered on the Red List of Chinese Vertebrates in 2016 [
15]. A previous study has shown that
Gymnocypris species are rich in genetic diversity in terms of morphological characteristics, proteins, genome, and mtDNA, reflecting the exceptional evolutionary history of these plateau fishes and their ability to adapt to the complex environment of the plateau [
1]. To scientifically and effectively conserve the germplasm resources of
Gymnocypris species, it is particularly important to investigate the genetic diversity, population structure, and priority conservation of
Gymnocypris fishes distributed on the QTP.
In this study, the eight populations showed differences in Hd; the GE population had the highest value, followed by the GPR and GPO populations. The GPO population showed the highest Pi and K values, followed by the GE population. In contrast, the GS population showed the lowest Hd, Pi, and K values. Hd, Pi, and K are three representative parameters for genetic diversity assessment, of which Hd is a measure of the uniqueness of a specific haplotype in a population and thus reflects the abundance of haplotypes in a population. Pi and K reflect only the degree of variation in each haplotype within a population [
16]. It is challenging to prioritize conservation on the basis of three separate perspectives. The comprehensive values of genetic diversity that we established using PCA take Hd, Pi, and K into account to judge the level of genetic diversity of different species in an integrated manner. The GPO, GE, and GPR populations had positive Fz
(F1) values, indicating that they have relatively high genetic diversity and, conversely, that the other populations have low genetic diversity (
Figure 2a).
The GPO population is more widely distributed than the other populations in China such as in the Jinsha and Lancang River systems and in Songpan County in the eastern part of the QTP. Therefore, the GPO population is subject to greater disturbance by artificial selection, which may account for its higher genetic diversity; this is consistent with the findings of similar studies [
17]. The GPR population is widely distributed in Qinghai Lake; the GE population is present not only in the Qinghai Lake system but also in the Yellow River system and therefore similarly subject to human intervention, thus allowing these populations to acquire higher genetic diversity [
18]. We found that all three populations (GPO, GE, and GPR) had relatively high H, Nps, Pis, and Svs, which are important intrinsic factors that contribute to high genetic diversity. The GS population is distributed in only the Langtze region of Tibet; the population size is small with little interaction with the outside world [
19], resulting in its low genetic diversity. According to our investigation, the other populations (GC, GW, GN, and GD) are also geographically homogeneous and isolated and have low H, Nps, Pis, and Svs, resulting in low genetic diversity.
The main indicators of the degree of polymorphism in a population are the genetic distance between populations and the population differentiation index, with higher values of genetic distance and population differentiation index representing higher polymorphism in the population [
20]. GPO, GE, and GPR are more genetically distant from the other populations and more genetically differentiated, resulting in a higher haplotype diversity. Interestingly, we found that the genetic distances between the GS and GC populations were lower than those within the GC population. This also indicates a stable GS population structure with very little genetic variation within the species [
21]. Therefore, we infer that there is a large genetic divergence between all populations, except for GC and GS, and the presence of genetic divergence between
Gymnocypris populations makes the recovery of their resources possible. Zhao et al. (2006) studied the structure and genetic diversity of
Gymnocypris species population in Qinghai Lake on the basis of the mitochondrial
Cyt b gene, detected frequent gene exchange and low genetic diversity within the population, and speculated that it may have historically experienced a bottleneck effect [
22]. Similarly, analysis of the mitochondrial D-loop gene revealed that genetic variation in the Qinghai Lake
Gymnocypris species was mainly within populations, with less variations between populations; this suggests that the
Gymnocypris species established a relatively stable pattern of reproductive migration, with greater gene exchange between populations migrating to the same river for mating and less gene exchange between populations migrating to different rivers for breeding [
23].
4.2. Model for Assessing Genetic Contribution on the Basis of Phylogeny and Genetic Diversity
The phylogeny of a species is in part a response to its genetic variation and is part of the study of genetic diversity. Of the four regions sampled in this study, Qinghai Province has the richest distribution of
Gymnocypris species resources (both in terms of species and numbers), largely because of the wide distribution of Qinghai Lake and its water system [
24]. The trend in network distribution showed that domestication events of
Gymnocypris species from clades A and C were mainly located in Qinghai, with a trend for clade A to expand from the Qinghai region to the Tibetan region (
Figure 1a,c). Clade B had two domestication events distributed in the Qinghai and Gansu regions, mainly in the GE population, and the phylogenetic tree analysis also supports the validity of this view. The GE population was found in the upper reaches of the Yellow River; as the Qinghai Lake and Yellow River were connected in the past, previous studies have suggested that the GE and GPR populations are closely related. However, similar studies have concluded that they do not form a separate lineage, that there is a crossover between their individuals, and the GPR population originated from the GE population [
18]. Therefore, it is essential to consider the priority conservation of
Gymnocypris species from different evolutionary clades.
For the genetic diversity calculations, the frequency of occurrence of each locus and haplotype was averaged (i.e., their contribution to genetic diversity was treated equally) and the contribution of certain specific haplotypes determined by some of these specific loci to both the maintenance and enhancement of population diversity was not revealed. Therefore, to integrate the contributions of both genetic variation and genetic distinctiveness within populations, the method proposed by Petite et al. (1998) and optimized and improved by Quan et al. (2020) to assess the conservation priority of species with a genetic contribution model was adopted. Positive values indicate a positive effect, which means that the genetic contribution of the taxon is higher than the average genetic contribution of the populations, and the presence of the population increases the intra-population genetic variation, inter-population genetic differentiation, or overall population allelic richness. Negative values indicate a negative effect, which means that the genetic contribution of the population is lower than the average genetic contribution of the populations, and the presence of the taxon decreases the intra-population genetic variation, inter-population genetic differentiation, or overall population allelic richness [
13].
Thus, the GE, GPO, and GPR populations have a positive impact on the entire population of
Gymnocypris species in terms of genetic contribution, indicating that they have a positive role in maintaining population diversity; therefore, these populations should be prioritized for conservation. In contrast, other populations play a negative role in maintaining the genetic diversity of the entire population (
Figure 2b). On the basis of the different clades of the phylogenetic tree, the GN, GW, GD, and GE populations contribute positively to the genetic diversity of all populations on clade A; therefore, they should be prioritized for conservation. The GE population is unique in clade B, so it should also be protected on a priority basis. The GPR population plays a positive role in the diversity of the other three populations in clade C, so it should be prioritized for protection. Therefore, without considering the systematic variability of alleles, we suggest that the GE and GPR populations be grouped into the same conservation unit because they are in the same geographic pattern and political region or distribution, and that the GPOs be grouped into a separate conservation unit [
25].