Exploring the Biological Functions and Anti-Melanogenesis of Phallus indusiatus for Mushroom-Based Cosmetic Applications
Abstract
:1. Introduction
2. Materials and Methods
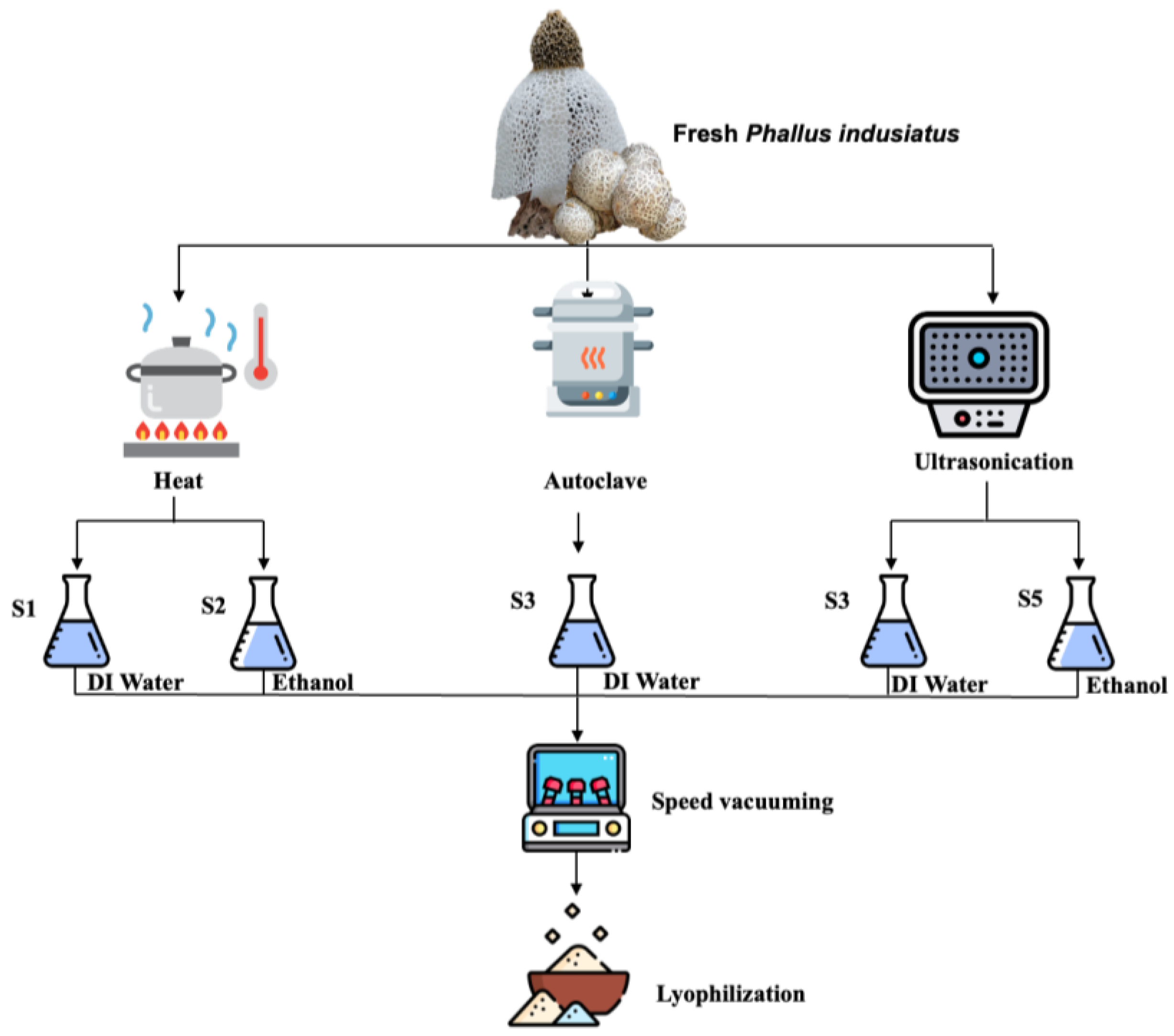
2.1. Phallus indusiatus Extraction
2.2. Phytochemical Analysis of Crude Extracts
2.2.1. Total Flavonoids Content Determination
2.2.2. Total Phenolics and Tannin Content
2.2.3. Total Sugar Analysis
2.3. Antioxidant Analysis
2.3.1. DPPH Assay
2.3.2. ABTS Assay
2.3.3. ORAC Assay
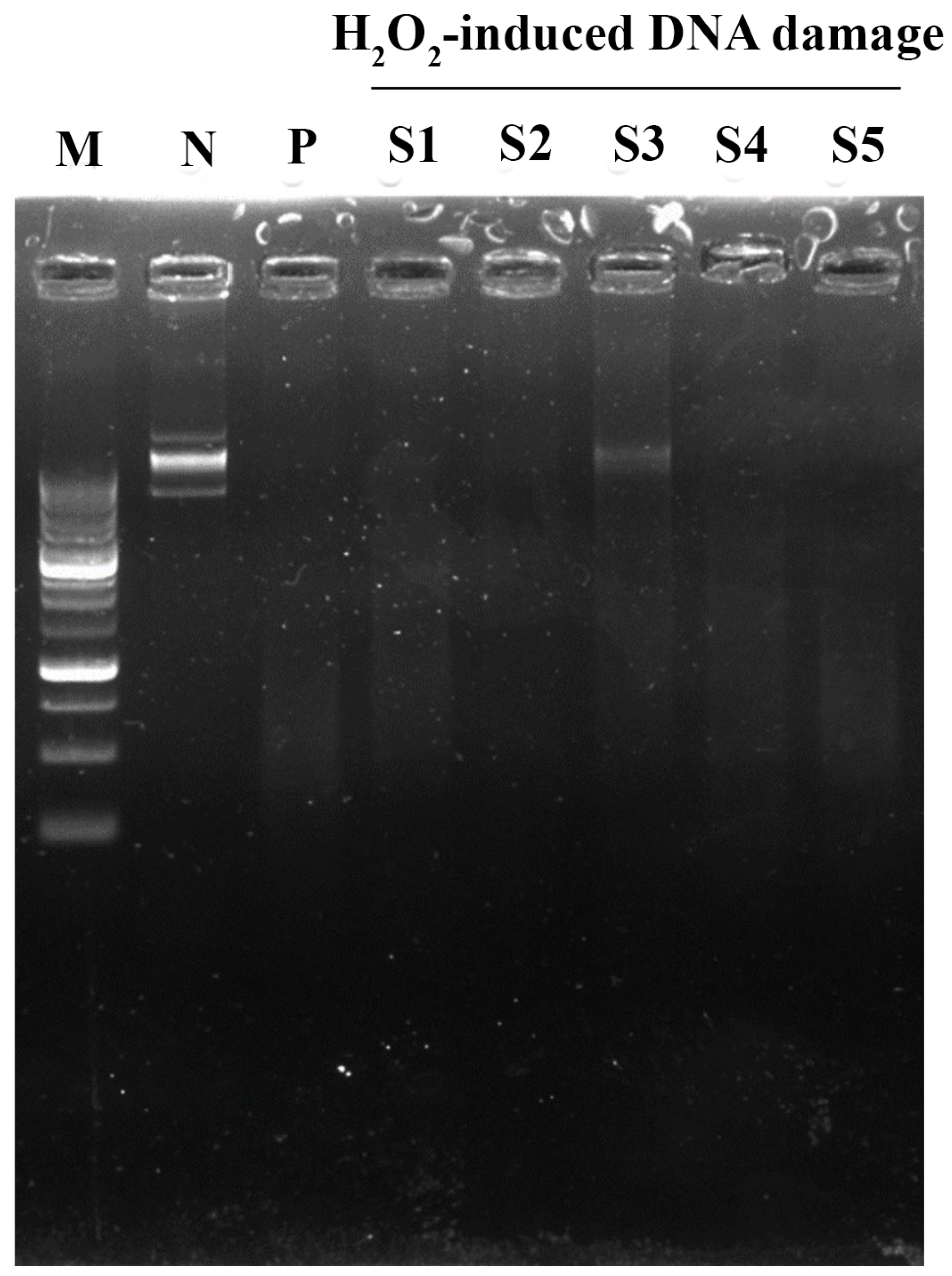
2.3.4. DNA Damage Assay
2.4. Biological Function Analysis
2.4.1. Collagenase Inhibition Assay
2.4.2. Elastase Inhibition Assay
2.4.3. Tyrosinase Inhibition Assay
2.4.4. Cellular Tyrosinase Inhibition Assay
2.4.5. Melanin Quantitation
2.4.6. Anti-Inflammatory Assay
2.5. Statistical Analysis
3. Results
3.1. Study of Phytochemical Contents of Fresh P. indusiatus Extraction by Different Methods
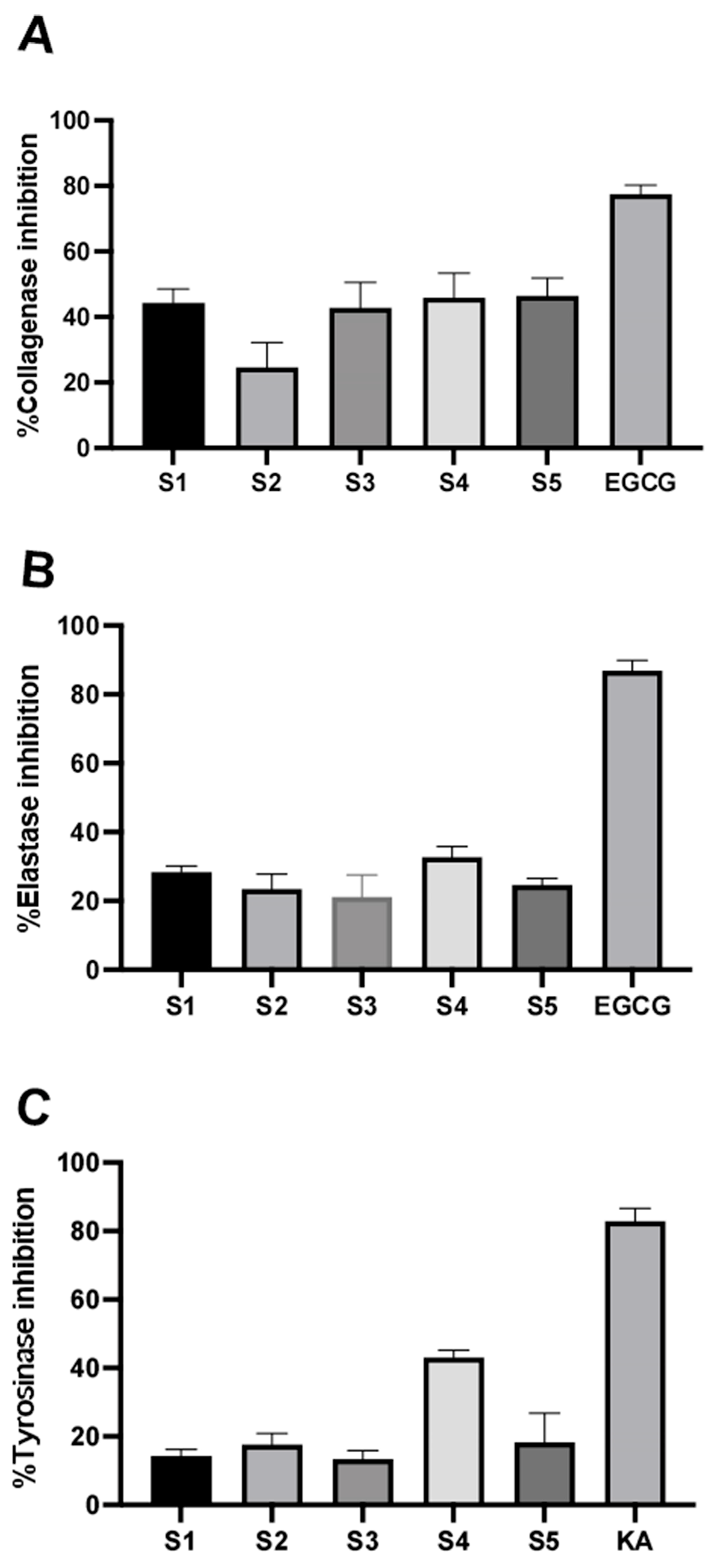
3.2. Inhibitory Effect on ECM-Degrading Enzymes of the P. indusiatus Extracts
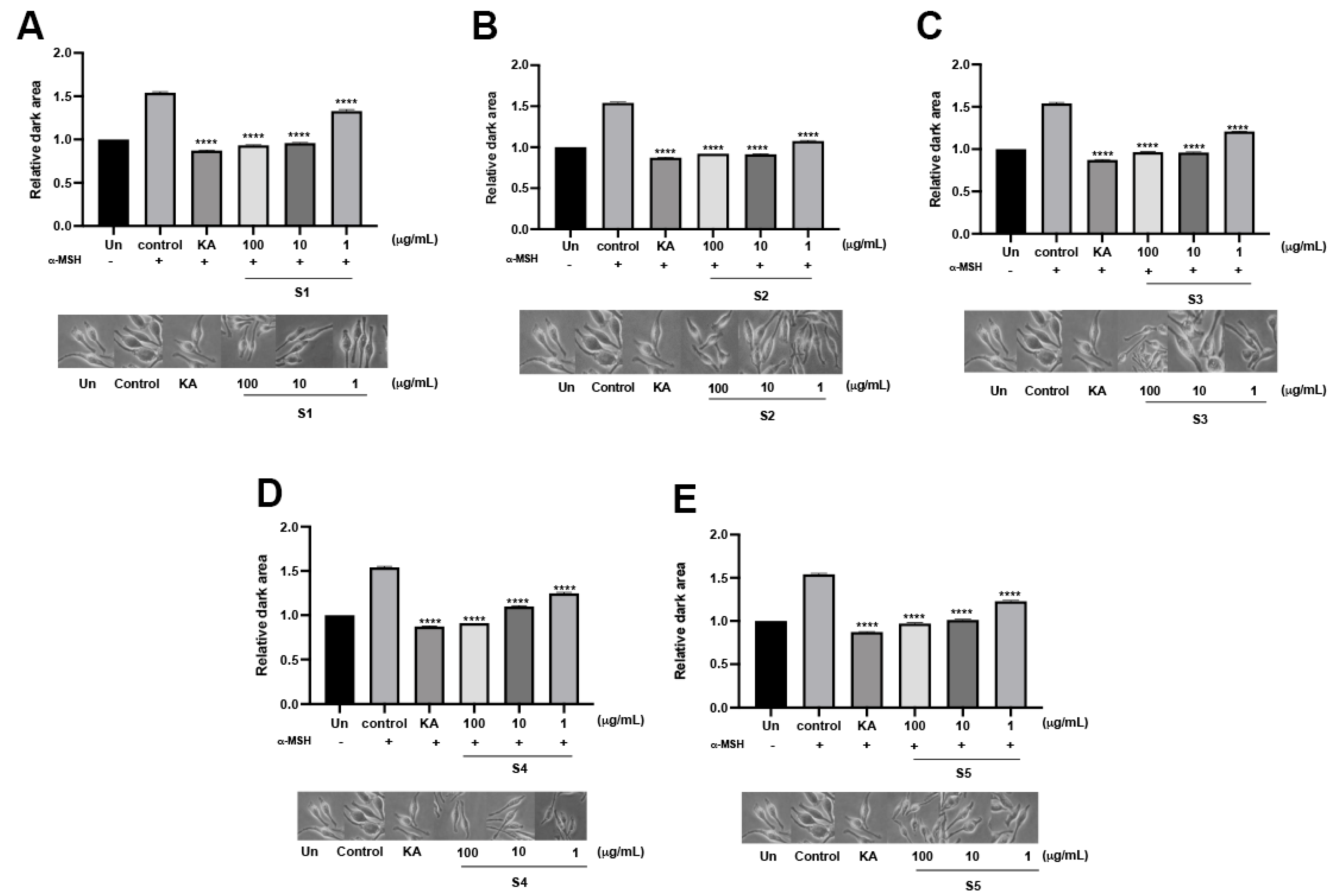
3.3. Assessment of Anti-Melanogenesis of the P. indusiatus Extracts in Both Enzymatic and Cellular Functions
3.4. Anti-Inflammatory Activity of the P. indusiatus Extracts by Decreasing NO Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, Y.; Choi, M.-H.; Li, J.; Yang, H.; Shin, H.-J. Mushroom Cosmetics: The Present and Future. Cosmetics 2016, 3, 22. [Google Scholar] [CrossRef]
- Taofiq, O.; Heleno, S.A.; Calhelha, R.C.; Alves, M.J.; Barros, L.; Barreiro, M.F.; Gonzalez-Paramas, A.M.; Ferreira, I.C. Development of Mushroom-Based Cosmeceutical Formulations with Anti-Inflammatory, Anti-Tyrosinase, Antioxidant, and Antibacterial Properties. Molecules 2016, 21, 1372. [Google Scholar] [CrossRef]
- Visvanathan, S.; Krishnamoorthy, R.; Sabesan, G.S. Fungal Cosmetics: Mushrooms in Beauty Care and the New Age of Natural Cosmetics. In Fungal Biology; Springer: Berlin/Heidelberg, Germany, 2022; pp. 1–37. [Google Scholar]
- Wasmeier, C.; Hume, A.N.; Bolasco, G.; Seabra, M.C. Melanosomes at a glance. J. Cell Sci. 2008, 121 Pt 24, 3995–3999. [Google Scholar] [CrossRef]
- Maranduca, M.A.; Branisteanu, D.; Serban, D.N.; Branisteanu, D.C.; Stoleriu, G.; Manolache, N.; Serban, I.L. Synthesis and physiological implications of melanic pigments. Oncol. Lett. 2019, 17, 4183–4187. [Google Scholar] [CrossRef]
- Kameyama, K.; Takemura, T.; Hamada, Y.; Sakai, C.; Kondoh, S.; Nishiyama, S.; Urabe, K.; Hearing, V.J. Pigment production in murine melanoma cells is regulated by tyrosinase, tyrosinase-related protein 1 (TRP1), DOPAchrome tautomerase (TRP2), and a melanogenic inhibitor. J. Investig. Dermatol. 1993, 100, 126–131. [Google Scholar] [CrossRef] [PubMed]
- McCabe, M.C.; Hill, R.C.; Calderone, K.; Cui, Y.; Yan, Y.; Quan, T.; Fisher, G.J.; Hansen, K.C. Alterations in extracellular matrix composition during aging and photoaging of the skin. Matrix Biol. Plus 2020, 8, 100041. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Qin, Z.; Xia, W.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Matrix-degrading metalloproteinases in photoaging. J. Investig. Dermatol. Symp. Proc. 2009, 14, 20–24. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- de Jager, T.L.; Cockrell, A.E.; Du Plessis, S.S. Ultraviolet Light Induced Generation of Reactive Oxygen Species. Adv. Exp. Med. Biol. 2017, 996, 15–23. [Google Scholar] [CrossRef]
- Fussell, J.C.; Kelly, F.J. Oxidative contribution of air pollution to extrinsic skin ageing. Free Radic. Biol. Med. 2020, 151, 111–122. [Google Scholar] [CrossRef]
- Garcia-Gavin, J.; Gonzalez-Vilas, D.; Fernandez-Redondo, V.; Toribio, J. Pigmented contact dermatitis due to kojic acid. A paradoxical side effect of a skin lightener. Contact Dermat. 2010, 62, 63–64. [Google Scholar] [CrossRef]
- Xu, H.; Li, X.; Xin, X.; Mo, L.; Zou, Y.; Zhao, G.; Yu, Y.; Chen, K. Antityrosinase Mechanism and Antimelanogenic Effect of Arbutin Esters Synthesis Catalyzed by Whole-Cell Biocatalyst. J. Agric. Food Chem. 2021, 69, 4243–4252. [Google Scholar] [CrossRef] [PubMed]
- Arndt, K.A.; Fitzpatrick, T.B. Topical use of hydroquinone as a depigmenting agent. JAMA 1965, 194, 965–967. [Google Scholar] [CrossRef] [PubMed]
- Breathnach, A.C.; Nazzaro-Porro, M.; Passi, S.; Zina, G. Azelaic acid therapy in disorders of pigmentation. Clin. Dermatol. 1989, 7, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Neering, H. Treatment of melasma (chloasma) by local application of a steroid cream. Dermatologica 1975, 151, 349–353. [Google Scholar] [CrossRef]
- Zasada, M.; Budzisz, E. Retinoids: Active molecules influencing skin structure formation in cosmetic and dermatological treatments. Adv. Dermatol. Allergol. 2019, 36, 392–397. [Google Scholar] [CrossRef]
- Sangthong, S.; Pintathong, P.; Pongsua, P.; Jirarat, A.; Chaiwut, P. Polysaccharides from Volvariella volvacea Mushroom: Extraction, Biological Activities and Cosmetic Efficacy. J. Fungi 2022, 8, 572. [Google Scholar] [CrossRef]
- Ha, H.T.; Tran-Van, H.; Tran-Van, H.; Tran, T.V.; Ha, H.T.; Tran-Van, H.; Tran, T.V.; Nguyen, H.T.N.; Phan, D.T.A. Study on chemical compositions, antioxidants and intracellular anti-melanogenic activities of varieties of Ganoderma lucidum in Vietnam. Int. J. Food Sci. Technol. 2023, 58, 4127–4135. [Google Scholar] [CrossRef]
- Habtemariam, S. The Chemistry, Pharmacology and Therapeutic Potential of the Edible Mushroom Dictyophora indusiata (Vent ex. Pers.) Fischer (Synn. Phallus indusiatus). Biomedicines 2019, 7, 98. [Google Scholar] [CrossRef]
- Oyetayo, V.; Dong, C.; Yao, Y. Antioxidant and antimicrobial properties of aqueous extract from Dictyophora indusiata. Open Mycol. J. 2009, 3, 20–26. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Wu, L.; Wu, X.; Huang, Y.; Liu, B. Optimization of polysaccharides extraction from Dictyophora indusiata and determination of its antioxidant activity. Int. J. Biol. Macromol. 2017, 103, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Choi, J.; Sharma, N.; Choi, M.; Seo, S.Y. In vitro anti-tyrosinase activity of 5-(hydroxymethyl)-2-furfural isolated from Dictyophora indusiata. Phytother. Res. 2004, 18, 841–844. [Google Scholar] [CrossRef] [PubMed]
- Nazir, Y.; Linsaenkart, P.; Khantham, C.; Chaitep, T.; Jantrawut, P.; Chittasupho, C.; Rachtanapun, P.; Jantanasakulwong, K.; Phimolsiripol, Y.; Sommano, S.R.; et al. High Efficiency In Vitro Wound Healing of Dictyophora indusiata Extracts via Anti-Inflammatory and Collagen Stimulating (MMP-2 Inhibition) Mechanisms. J. Fungi 2021, 7, 1100. [Google Scholar] [CrossRef]
- Giordano, A.; Morales-Tapia, P.; Moncada-Basualto, M.; Pozo-Martinez, J.; Olea-Azar, C.; Nesic, A.; Cabrera-Barjas, G. Polyphenolic Composition and Antioxidant Activity (ORAC, EPR and Cellular) of Different Extracts of Argylia radiata Vitroplants and Natural Roots. Molecules 2022, 27, 610. [Google Scholar] [CrossRef] [PubMed]
- Phonphoem, W.; Sinthuvanich, C.; Aramrak, A.; Sirichiewsakul, S.; Arikit, S.; Yokthongwattana, C. Nutritional Profiles, Phytochemical Analysis, Antioxidant Activity and DNA Damage Protection of Makapuno Derived from Thai Aromatic Coconut. Foods 2022, 11, 3912. [Google Scholar] [CrossRef]
- Taofiq, O.; González-Paramás, A.M.; Martins, A.; Barreiro, M.F.; Ferreira, I.C.F.R. Mushrooms extracts and compounds in cosmetics, cosmeceuticals and nutricosmetics—A review. Ind. Crops Prod. 2016, 90, 38–48. [Google Scholar] [CrossRef]
- Mago, P.; Sharma, R.; Hafeez, I.; Nawaz, I.; Joshi, M.; Mehrotra, R. Mushroom based Cosmeceuticals: An Upcoming Biotechnology Sector. Biosci. Biotechnol. Res. Asia 2023, 20, 2. [Google Scholar] [CrossRef]
- Bristy, A.T.; Islam, T.; Ahmed, R.; Hossain, J.; Reza, H.M.; Jain, P. Evaluation of Total Phenolic Content, HPLC Analysis, and Antioxidant Potential of Three Local Varieties of Mushroom: A Comparative Study. Int. J. Food Sci. 2022, 2022, 3834936. [Google Scholar] [CrossRef]
- Rensing, S.A. Great moments in evolution: The conquest of land by plants. Curr. Opin. Plant Biol. 2018, 42, 49–54. [Google Scholar] [CrossRef]
- Gil-Ramírez, A.; Pavo-Caballero, C.; Baeza, E.; Baenas, N.; Garcia-Viguera, C.; Marín, F.R.; Soler-Rivas, C. Mushrooms do not contain flavonoids. J. Funct. Foods 2016, 25, 1–13. [Google Scholar] [CrossRef]
- Wang, S.; Liu, Z.; Wang, X.; Liu, R.; Zou, L. Mushrooms Do Produce Flavonoids: Metabolite Profiling and Transcriptome Analysis of Flavonoid Synthesis in the Medicinal Mushroom Sanghuangporus baumii. J. Fungi 2022, 8, 582. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Hu, Z.; Fu, H.; Hu, M.; Xu, X.; Chen, J. Chemical analysis and antioxidant activity in vitro of a beta-D-glucan isolated from Dictyophora indusiata. Int. J. Biol. Macromol. 2012, 51, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Floegel, A.; Kim, D.-O.; Chung, S.-J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compos. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Sies, H.; Belousov, V.V.; Chandel, N.S.; Davies, M.J.; Jones, D.P.; Mann, G.E.; Murphy, M.P.; Yamamoto, M.; Winterbourn, C. Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nat. Rev. Mol. Cell Biol. 2022, 23, 499–515. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, G.P. Mechanisms of UV-induced mutations and skin cancer. Genome Instab. Dis. 2020, 1, 99–113. [Google Scholar] [CrossRef]
- Hasnat, M.A.; Pervin, M.; Debnath, T.; Lim, B.O. DNA Protection, Total Phenolics and Antioxidant Potential of the Mushroom Russula Virescens. J. Food Biochem. 2014, 38, 6–17. [Google Scholar] [CrossRef]
- Widgerow, A.D.; Fabi, S.G.; Palestine, R.F.; Rivkin, A.; Ortiz, A.; Bucay, V.W.; Chiu, A.; Naga, L.; Emer, J.; Chasan, P.E. Extracellular Matrix Modulation: Optimizing Skin Care and Rejuvenation Procedures. J. Drugs Dermatol. 2016, 15, s63–s71. [Google Scholar]
- Chowdhury, A.; Nosoudi, N.; Karamched, S.; Parasaram, V.; Vyavahare, N. Polyphenol treatments increase elastin and collagen deposition by human dermal fibroblasts; Implications to improve skin health. J. Dermatol. Sci. 2021, 102, 94–100. [Google Scholar] [CrossRef]
- Serre, C.; Busuttil, V.; Botto, J.M. Intrinsic and extrinsic regulation of human skin melanogenesis and pigmentation. Int. J. Cosmet. Sci. 2018, 40, 328–347. [Google Scholar] [CrossRef]
- Varghese, P.K.; Abu-Asab, M.; Dimitriadis, E.K.; Dolinska, M.B.; Morcos, G.P.; Sergeev, Y.V. Tyrosinase Nanoparticles: Understanding the Melanogenesis Pathway by Isolating the Products of Tyrosinase Enzymatic Reaction. Int. J. Mol. Sci. 2021, 22, 734. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- El-Nashar, H.A.S.; El-Din, M.I.G.; Hritcu, L.; Eldahshan, O.A. Insights on the Inhibitory Power of Flavonoids on Tyrosinase Activity: A Survey from 2016 to 2021. Molecules 2021, 26, 7546. [Google Scholar] [CrossRef] [PubMed]
- Pillaiyar, T.; Manickam, M.; Namasivayam, V. Skin whitening agents: Medicinal chemistry perspective of tyrosinase inhibitors. J. Enzyme Inhib. Med. Chem. 2017, 32, 403–425. [Google Scholar] [CrossRef] [PubMed]
- Wulf, H.C.; Sandby-Moller, J.; Kobayasi, T.; Gniadecki, R. Skin aging and natural photoprotection. Micron 2004, 35, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Lucy, T.T.; Mamun-Or-Rashid, A.N.M.; Yagi, M.; Yonei, Y. Serial Passaging of RAW 264.7 Cells Modulates Intracellular AGE Formation and Downregulates RANKL-Induced In Vitro Osteoclastogenesis. Int. J. Mol. Sci. 2022, 23, 2371. [Google Scholar] [CrossRef] [PubMed]


| Samples | Total Phenolics µg QE/g FW | Total Flavonoids mg GAE/g FW | Total Tannin mg TAE/g FW | Total Sugar mg/g FW | DPPH IC50 µg/mL | ABTS IC50 µg/mL | ORAC (NetAUC) mM Ascorbic/g FW |
|---|---|---|---|---|---|---|---|
| S1 | 11.4 ± 4.68 | 173.19 ± 6.22 | 8.86 ± 5.83 | 61.09 ± 26.99 b | >100 | 28.29 ± 2.63 bc | 1135.64 ± 12.81 b |
| S2 | 12.27 ± 6.58 | 237.49 ± 34.72 | 8.31 ± 3.325 | 62.95 ± 24.84 b | 88.33 ± 11.10 | 28.77 ± 1.14 bc | 203.08 ± 17.73 c |
| S3 | 17.59 ± 12.44 | 192 ± 41.23 | 13.3 ± 2.87 | 104.98 ± 47.33 ab | 80.16 ± 6.53 | 32.59 ± 1.25 ab | N.D. |
| S4 | 9.92 ± 3.23 | 174.80 ± 32.7 | 7.71 ± 1.91 | N.D. | 84.88 ± 11.59 | 41.25 ± 7.91 a | 1302.79 ± 59.70 a |
| S5 | 11.95 ± 3.75 | 188.12 ± 25 | 8.86 ± 1.91 | 265.26 ± 123.42 a | >100 | 42.44 ± 3.76 a | 187.33 ± 39.07 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theeraraksakul, K.; Jaengwang, K.; Choowongkomon, K.; Tabtimmai, L. Exploring the Biological Functions and Anti-Melanogenesis of Phallus indusiatus for Mushroom-Based Cosmetic Applications. Cosmetics 2023, 10, 121. https://doi.org/10.3390/cosmetics10050121
Theeraraksakul K, Jaengwang K, Choowongkomon K, Tabtimmai L. Exploring the Biological Functions and Anti-Melanogenesis of Phallus indusiatus for Mushroom-Based Cosmetic Applications. Cosmetics. 2023; 10(5):121. https://doi.org/10.3390/cosmetics10050121
Chicago/Turabian StyleTheeraraksakul, Kanyanat, Kittitat Jaengwang, Kiattawee Choowongkomon, and Lueacha Tabtimmai. 2023. "Exploring the Biological Functions and Anti-Melanogenesis of Phallus indusiatus for Mushroom-Based Cosmetic Applications" Cosmetics 10, no. 5: 121. https://doi.org/10.3390/cosmetics10050121
APA StyleTheeraraksakul, K., Jaengwang, K., Choowongkomon, K., & Tabtimmai, L. (2023). Exploring the Biological Functions and Anti-Melanogenesis of Phallus indusiatus for Mushroom-Based Cosmetic Applications. Cosmetics, 10(5), 121. https://doi.org/10.3390/cosmetics10050121





