Effects of Thymoquinone on Small-Molecule Metabolites in a Rat Model of Cerebral Ischemia Reperfusion Injury Assessed using MALDI-MSI
Abstract
1. Introduction
2. Results
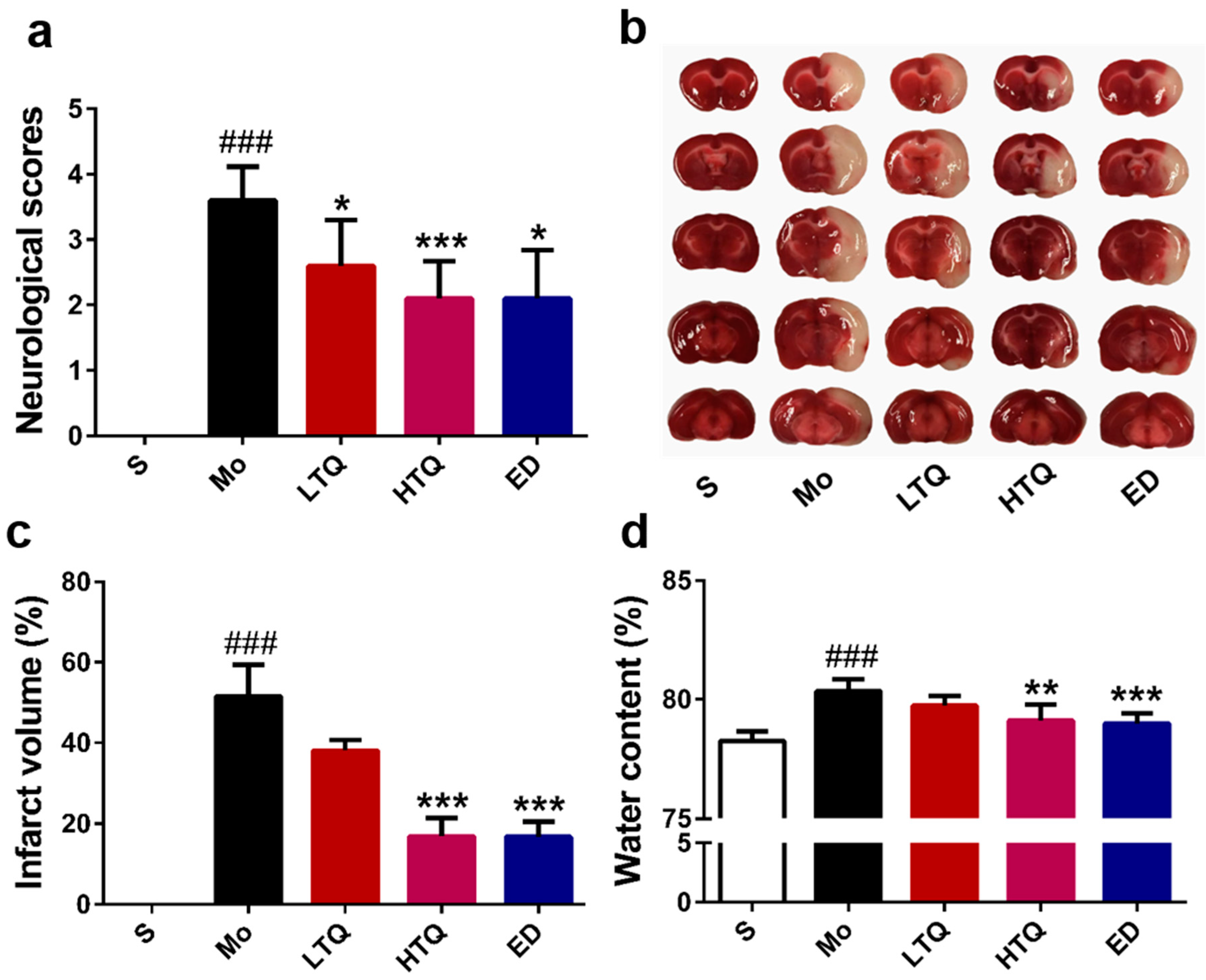
2.1. Neuroprotective Effects of Thymoquinone in the Cerebal Ischemia Reperfusion Injury Model
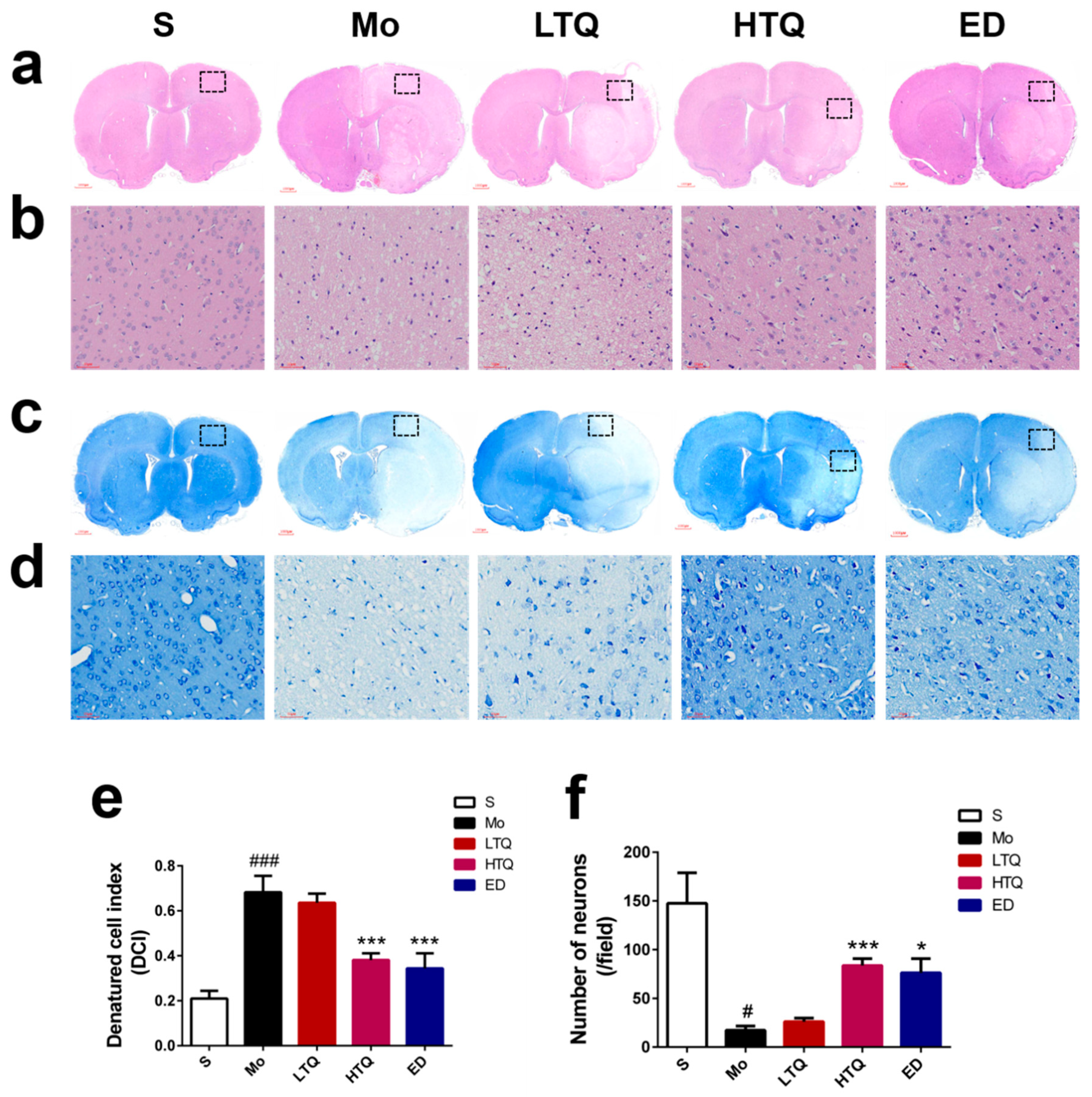
2.2. Histomorphological Changes Following Cerebral Ischemia–Reperfusion Injury
2.3. MALDI-MSI Analysis Following Cerebral Ischemia Reperfusion Injury
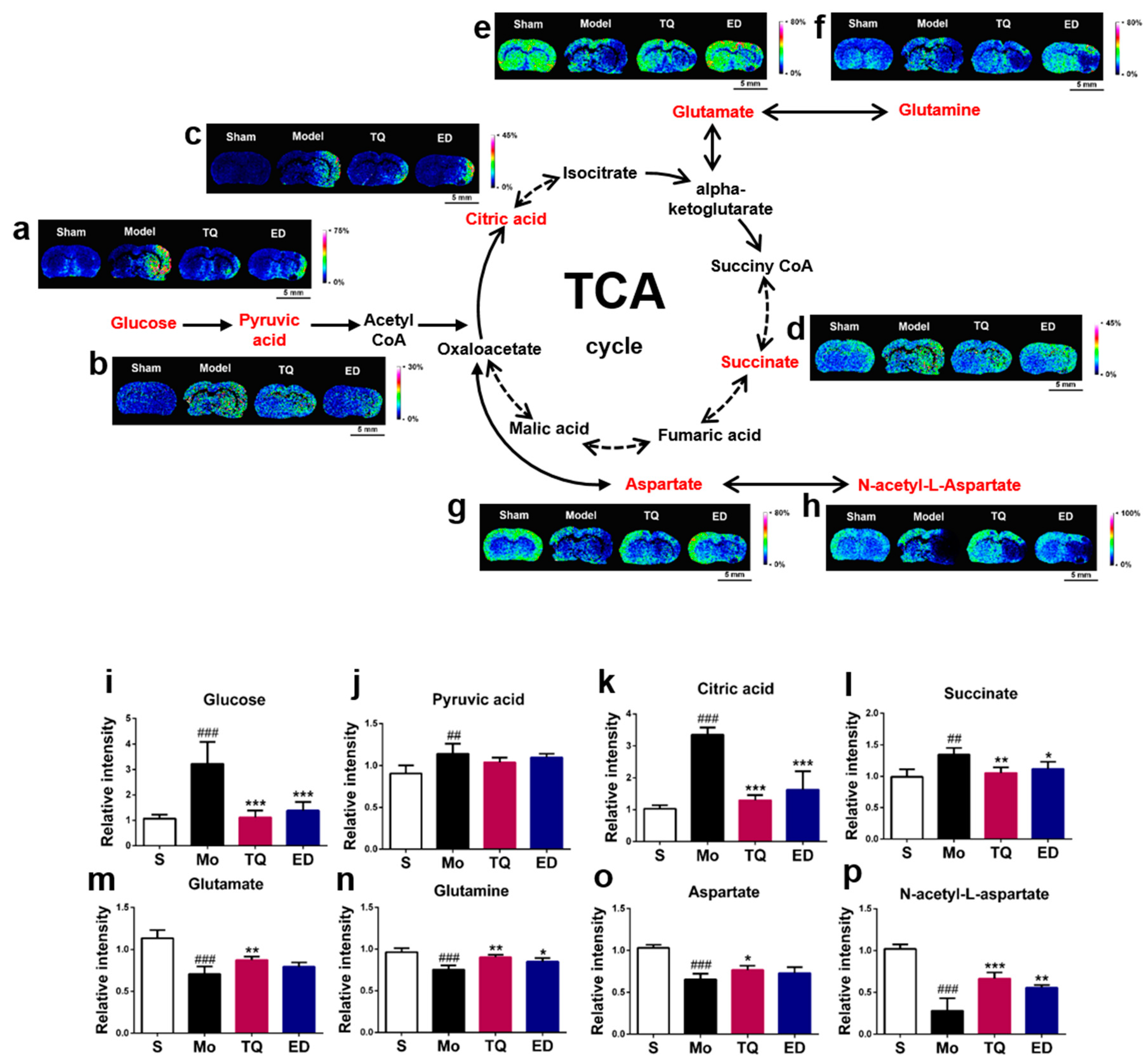
2.3.1. Effect of Thymoquinone Treatment on Aerobic Oxidation-Related Small Molecules in the Damaged Brain
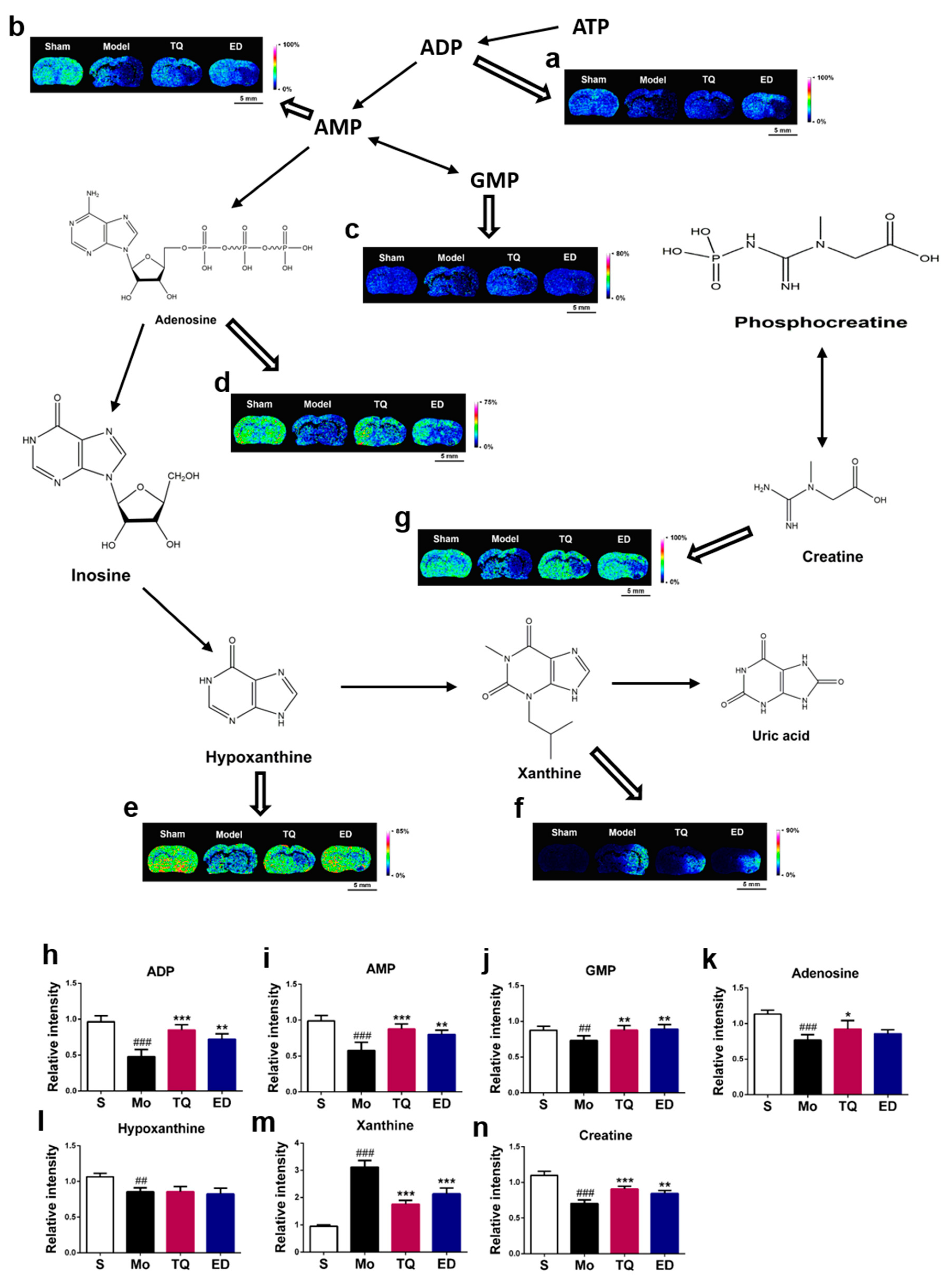
2.3.2. Effect of Thymoquinone Treatment on Small Molecules Involved in Energy Metabolism in the Damaged Brain
2.3.3. Effect of Thymoquinone Treatment on Lipid Molecules in the Damaged Brain
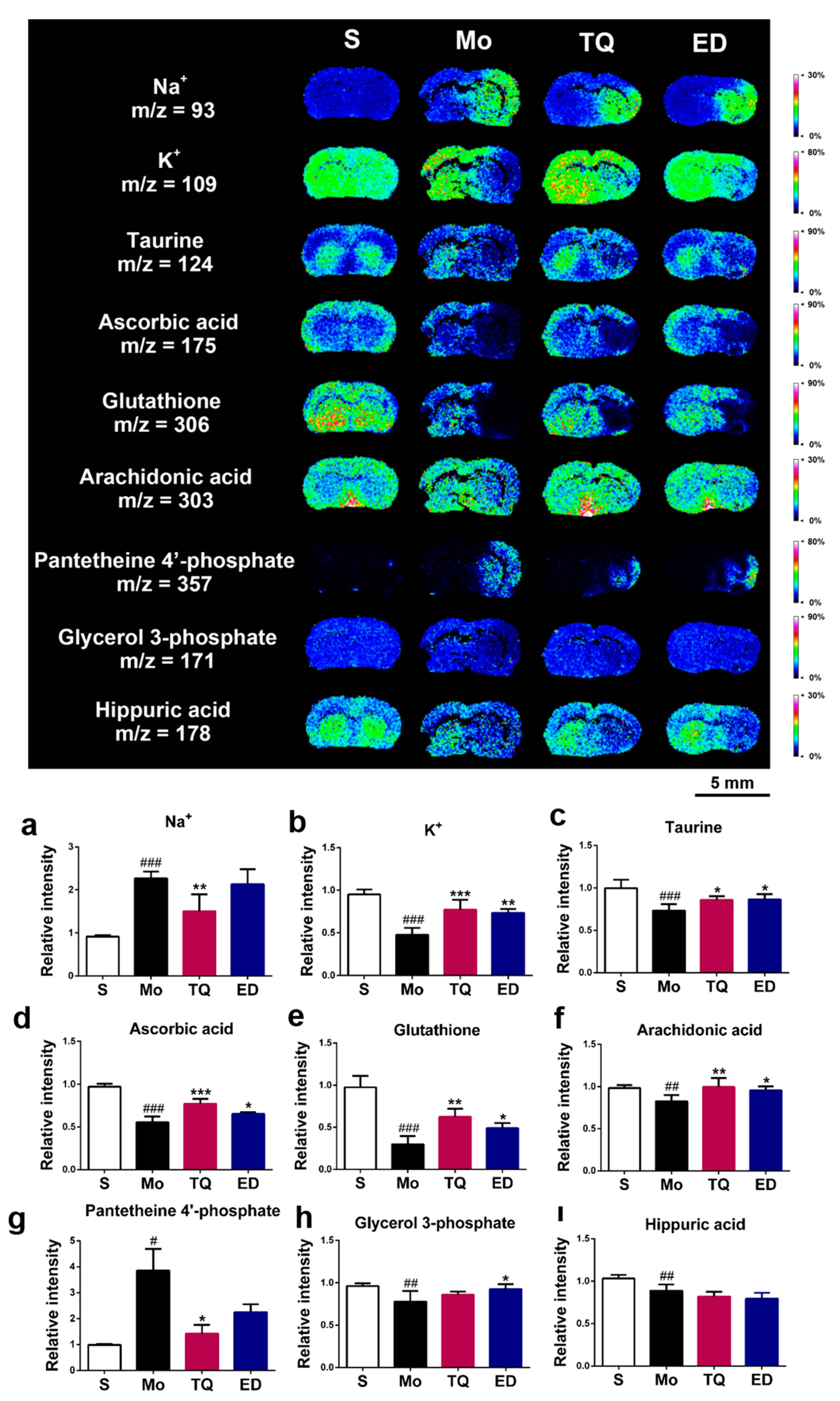
2.3.4. Effect of Thymoquinone Treatment on Metal Ions, Antioxidant Molecules, and Other Metabolic Small Molecules in the Damaged Brain
2.3.5. Segmentation Analysis
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Experimental Animals
4.3. Cerebral Ischemia–Reperfusion Model
4.4. Animal Behavioral Test
4.5. Infarct Volume and Edema Damage Assessment
4.6. Analysis of Histomorphological Features
4.7. Preparation of Samples for Mass Spectrometry Imaging
4.8. Mass Spectrometry Imaging
4.9. Segmentation Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, X.; Chen, L.; Dang, X.; Liu, J.; Ito, Y.; Sun, W. Neuroprotective effects of total steroid saponins on cerebral ischemia injuries in an animal model of focal ischemia/reperfusion. Planta Med. 2014, 80, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Husain, A.; Mujeeb, M.; Khan, S.A.; Najmi, A.K.; Siddique, N.A.; Damanhouri, Z.A.; Anwar, F. A review on therapeutic potential of Nigella sativa: A miracle herb. Asian Pac. J. Trop. Biomed. 2013, 3, 337–352. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Al-Omar, F.A.; Nagi, M.N. Neuroprotective effects of thymoquinone against transient forebrain ischemia in the rat hippocampus. Eur. J. Pharmacol. 2006, 543, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Ahmad, R.; Alam, M.A.; Samim, M.; Iqbal, Z.; Ahmad, F.J. Quantification and evaluation of thymoquinone loaded mucoadhesive nanoemulsion for treatment of cerebral ischemia. Int. J. Biol. Macromol. 2016, 88, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Ling, J.; Wang, X.; Li, T.; Liu, J.; Lai, Y.; Ji, H.; Peng, S.; Tian, J.; Zhang, Y. Discovery of a potential anti-ischemic stroke agent: 3-pentylbenzo[c]thiophen-1(3H)-one. J. Med. Chem. 2012, 55, 7173–7181. [Google Scholar] [CrossRef] [PubMed]
- Miura, D.; Fujimura, Y.; Yamato, M.; Hyodo, F.; Utsumi, H.; Tachibana, H.; Wariishi, H. Ultrahighly sensitive in situ metabolomic imaging for visualizing spatiotemporal metabolic behaviors. Anal. Chem. 2010, 82, 9789–9796. [Google Scholar] [CrossRef] [PubMed]
- Alhebshi, A.H.; Gotoh, M.; Suzuki, I. Thymoquinone protects cultured rat primary neurons against amyloid beta-induced neurotoxicity. Biochem. Biophys. Res. Commun. 2013, 433, 362–367. [Google Scholar] [CrossRef]
- Alhebshi, A.H.; Odawara, A.; Gotoh, M.; Suzuki, I. Thymoquinone protects cultured hippocampal and human induced pluripotent stem cells-derived neurons against alpha-synuclein-induced synapse damage. Neurosci. Lett. 2014, 570, 126–131. [Google Scholar] [CrossRef]
- Javidi, S.; Razavi, B.M.; Hosseinzadeh, H. A review of Neuropharmacology Effects of Nigella sativa and Its Main Component, Thymoquinone. Phytother. Res. 2016, 30, 1219–1229. [Google Scholar] [CrossRef]
- Xiao, X.Y.; Zhu, Y.X.; Bu, J.Y.; Li, G.W.; Zhou, J.H.; Zhou, S.P. Evaluation of Neuroprotective Effect of Thymoquinone Nanoformulation in the Rodent Cerebral Ischemia-Reperfusion Model. Biomed. Res. Int. 2016, 2016, 2571060. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Parvardeh, S.; Asl, M.N.; Sadeghnia, H.R.; Ziaee, T. Effect of thymoquinone and Nigella sativa seeds oil on lipid peroxidation level during global cerebral ischemia-reperfusion injury in rat hippocampus. Phytomedicine 2007, 14, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Katsura, K.; Rodriguez de Turco, E.B.; Folbergrova, J.; Bazan, N.G.; Siesjo, B.K. Coupling among energy failure, loss of ion homeostasis, and phospholipase A2 and C activation during ischemia. J. Neurochem. 1993, 61, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Li, C.; Xia, N.; Zhao, L.; Wang, D.; Yang, Y.; Gao, H. Neurochemical changes in unilateral cerebral hemisphere during the subacute stage of focal cerebral ischemia-reperfusion in rats: An ex vivo (1)H magnetic resonance spectroscopy study. Brain Res. 2018, 1684, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Karki, P.; Hong, P.; Johnson, J., Jr.; Pajarillo, E.; Son, D.S.; Aschner, M.; Lee, E.Y. Arundic Acid Increases Expression and Function of Astrocytic Glutamate Transporter EAAT1 Via the ERK, Akt, and NF-kappaB Pathways. Mol. Neurobiol. 2018, 55, 5031–5046. [Google Scholar] [CrossRef] [PubMed]
- Moffett, J.R.; Ross, B.; Arun, P.; Madhavarao, C.N.; Namboodiri, A.M. N-Acetylaspartate in the CNS: From neurodiagnostics to neurobiology. Prog. Neurobiol. 2007, 81, 89–131. [Google Scholar] [CrossRef]
- Duarte, J.M.; Lei, H.; Mlynarik, V.; Gruetter, R. The neurochemical profile quantified by in vivo 1H NMR spectroscopy. Neuroimage 2012, 61, 342–362. [Google Scholar] [CrossRef]
- Saunders, D.E. MR spectroscopy in stroke. Br. Med. Bull. 2000, 56, 334–345. [Google Scholar] [CrossRef]
- Fouad, I.A.; Sharaf, N.M.; Abdelghany, R.M.; El Sayed, N. Neuromodulatory Effect of Thymoquinone in Attenuating Glutamate-Mediated Neurotoxicity Targeting the Amyloidogenic and Apoptotic Pathways. Front. Neurol. 2018, 9, 236. [Google Scholar] [CrossRef]
- Panel, M.; Ruiz, I.; Brillet, R.; Lafdil, F.; Teixeira-Clerc, F.; Nguyen, C.T.; Calderaro, J.; Gelin, M.; Allemand, F.; Guichou, J.F.; et al. Small-Molecule Inhibitors of Cyclophilins Block Opening of the Mitochondrial Permeability Transition Pore and Protect Mice From Hepatic Ischemia/Reperfusion Injury. Gastroenterology 2019, 157, 1368–1382. [Google Scholar] [CrossRef]
- Sanderson, T.H.; Reynolds, C.A.; Kumar, R.; Przyklenk, K.; Huttemann, M. Molecular mechanisms of ischemia-reperfusion injury in brain: Pivotal role of the mitochondrial membrane potential in reactive oxygen species generation. Mol. Neurobiol. 2013, 47, 9–23. [Google Scholar] [CrossRef]
- Farooqui, Z.; Shahid, F.; Abidi, S.; Parwez, I.; Khan, F. Oral thymoquinone administration ameliorates: The effect of cisplatin on brush border membrane enzymes, energy metabolism, and redox status in rat kidney. Naunyn Schmiedebergs Arch. Pharmacol. 2017, 390, 1271–1284. [Google Scholar] [CrossRef] [PubMed]
- Shahid, F.; Farooqui, Z.; Abidi, S.; Parwez, I.; Khan, F. Oral administration of thymoquinone mitigates the effect of cisplatin on brush border membrane enzymes, energy metabolism and antioxidant system in rat intestine. Biomed. Pharmacother. 2017, 94, 1111–1120. [Google Scholar] [CrossRef]
- Shanta, S.R.; Choi, C.S.; Lee, J.H.; Shin, C.Y.; Kim, Y.J.; Kim, K.H.; Kim, K.P. Global changes in phospholipids identified by MALDI MS in rats with focal cerebral ischemia. J. Lipid Res. 2012, 53, 1823–1831. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.M.; Lambertsen, K.L.; Clausen, B.H.; Meyer, M.; Bhandari, D.R.; Larsen, S.T.; Poulsen, S.S.; Spengler, B.; Janfelt, C.; Hansen, H.S. Mass spectrometry imaging of biomarker lipids for phagocytosis and signalling during focal cerebral ischaemia. Sci. Rep. 2016, 6, 39571. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.Z.; Fan, C.X.; Zhang, Z.L.; Zhao, X.; Sun, Y.; Liu, H.H.; Nie, Z.X.; Pu, X.P. Effects of Dl-3 n-butylphthalide on Cerebral Ischemia Infarction in Rat Model by Mass Spectrometry Imaging. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef]
- Shimada, K.; Jong, C.J.; Takahashi, K.; Schaffer, S.W. Role of ROS Production and Turnover in the Antioxidant Activity of Taurine. Adv. Exp. Med. Biol. 2015, 803, 581–596. [Google Scholar] [CrossRef]
- Kakutani, S.; Egawa, K.; Saito, K.; Suzuki, T.; Horikawa, C.; Rogi, T.; Kawashima, H.; Shibata, H.; Sasaki, S. Arachidonic acid intake and asthma risk in children and adults: A systematic review of observational studies. J. Nutr. Sci. 2014, 3, e12. [Google Scholar] [CrossRef]
- Gong, Z.; Pan, J.; Shen, Q.; Li, M.; Peng, Y. Mitochondrial dysfunction induces NLRP3 inflammasome activation during cerebral ischemia/reperfusion injury. J. Neuroinflammation 2018, 15, 242. [Google Scholar] [CrossRef]
- Zhu, N.; Zhao, X.; Xiang, Y.; Ye, S.; Huang, J.; Hu, W.; Lv, L.; Zeng, C. Thymoquinone attenuates monocrotaline-induced pulmonary artery hypertension via inhibiting pulmonary arterial remodeling in rats. Int. J. Cardiol. 2016, 221, 587–596. [Google Scholar] [CrossRef]
- Ma, Y.; Li, L.; Niu, Z.; Song, J.; Lin, Y.; Zhang, H.; Du, G. Effect of recombinant plasminogen activator timing on thrombolysis in a novel rat embolic stroke model. Pharmacol. Res. 2016, 107, 291–299. [Google Scholar] [CrossRef]
- Li, W.; Liu, H.; Jiang, H.; Wang, C.; Guo, Y.; Sun, Y.; Zhao, X.; Xiong, X.; Zhang, X.; Zhang, K.; et al. (S)-Oxiracetam is the Active Ingredient in Oxiracetam that Alleviates the Cognitive Impairment Induced by Chronic Cerebral Hypoperfusion in Rats. Sci. Rep. 2017, 7, 10052. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, L.; Kong, L.; Zhu, Z.; Zhang, W.; Song, J.; Chang, J.; Du, G. Pinocembrin Protects Blood-Brain Barrier Function and Expands the Therapeutic Time Window for Tissue-Type Plasminogen Activator Treatment in a Rat Thromboembolic Stroke Model. Biomed. Res. Int. 2018, 2018, 8943210. [Google Scholar] [CrossRef] [PubMed]
- Tatlisumak, T.; Carano, R.A.; Takano, K.; Opgenorth, T.J.; Sotak, C.H.; Fisher, M. A novel endothelin antagonist, A-127722, attenuates ischemic lesion size in rats with temporary middle cerebral artery occlusion: A diffusion and perfusion MRI study. Stroke 1998, 29, 850–857, discussion 857–858. [Google Scholar] [CrossRef] [PubMed]
- Ya, B.L.; Liu, Q.; Li, H.F.; Cheng, H.J.; Yu, T.; Chen, L.; Wang, Y.; Yuan, L.L.; Li, W.J.; Liu, W.Y.; et al. Uric Acid Protects against Focal Cerebral Ischemia/Reperfusion-Induced Oxidative Stress via Activating Nrf2 and Regulating Neurotrophic Factor Expression. Oxid. Med. Cell. Longev. 2018, 2018, 6069150. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Cheng, J.; Pan, G.; Wu, S.; Hu, Q.; Jiang, H.; Wang, Y.; Xiong, J.; Pang, Q.; Chen, X. Treadmill exercise ameliorates focal cerebral ischemia/reperfusion-induced neurological deficit by promoting dendritic modification and synaptic plasticity via upregulating caveolin-1/VEGF signaling pathways. Exp. Neurol. 2019, 313, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Liu, C.; Ji, Y.; Teng, L.; Guo, Y. Neuregulin-1beta Plays a Neuroprotective Role by Inhibiting the Cdk5 Signaling Pathway after Cerebral Ischemia-Reperfusion Injury in Rats. J. Mol. Neurosci. 2018, 66, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.L.; Zhang, B.; Jing, L. MiR-125b blocks Bax/Cytochrome C/Caspase-3 apoptotic signaling pathway in rat models of cerebral ischemia-reperfusion injury by targeting p53. Neurol. Res. 2018, 40, 828–837. [Google Scholar] [CrossRef]
- Liu, H.; Chen, R.; Wang, J.; Chen, S.; Xiong, C.; Wang, J.; Hou, J.; He, Q.; Zhang, N.; Nie, Z.; et al. 1,5-Diaminonaphthalene hydrochloride assisted laser desorption/ionization mass spectrometry imaging of small molecules in tissues following focal cerebral ischemia. Anal. Chem. 2014, 86, 10114–10121. [Google Scholar] [CrossRef]
- Huang, X.; Zhan, L.; Sun, J.; Xue, J.; Liu, H.; Xiong, C.; Nie, Z. Utilizing a Mini-Humidifier To Deposit Matrix for MALDI Imaging. Anal. Chem. 2018, 90, 8309–8313. [Google Scholar] [CrossRef]
- Liu, H.; Li, W.; He, Q.; Xue, J.; Wang, J.; Xiong, C.; Pu, X.; Nie, Z. Mass Spectrometry Imaging of Kidney Tissue Sections of Rat Subjected to Unilateral Ureteral Obstruction. Sci. Rep. 2017, 7, 41954. [Google Scholar] [CrossRef]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, F.; Liu, R.; Fan, C.; Sun, Y.; Huang, X.; Nie, Z.; Zhao, X.; Pu, X. Effects of Thymoquinone on Small-Molecule Metabolites in a Rat Model of Cerebral Ischemia Reperfusion Injury Assessed using MALDI-MSI. Metabolites 2020, 10, 27. https://doi.org/10.3390/metabo10010027
Tian F, Liu R, Fan C, Sun Y, Huang X, Nie Z, Zhao X, Pu X. Effects of Thymoquinone on Small-Molecule Metabolites in a Rat Model of Cerebral Ischemia Reperfusion Injury Assessed using MALDI-MSI. Metabolites. 2020; 10(1):27. https://doi.org/10.3390/metabo10010027
Chicago/Turabian StyleTian, Fang, Runzhe Liu, Chaoxin Fan, Yi Sun, Xi Huang, Zongxiu Nie, Xin Zhao, and Xiaoping Pu. 2020. "Effects of Thymoquinone on Small-Molecule Metabolites in a Rat Model of Cerebral Ischemia Reperfusion Injury Assessed using MALDI-MSI" Metabolites 10, no. 1: 27. https://doi.org/10.3390/metabo10010027
APA StyleTian, F., Liu, R., Fan, C., Sun, Y., Huang, X., Nie, Z., Zhao, X., & Pu, X. (2020). Effects of Thymoquinone on Small-Molecule Metabolites in a Rat Model of Cerebral Ischemia Reperfusion Injury Assessed using MALDI-MSI. Metabolites, 10(1), 27. https://doi.org/10.3390/metabo10010027





