The Role of the GH/IGF1 Axis on the Development of MAFLD in Pediatric Patients with Obesity
Abstract
:1. Introduction
2. Materials and Methods
3. Statistical Analysis
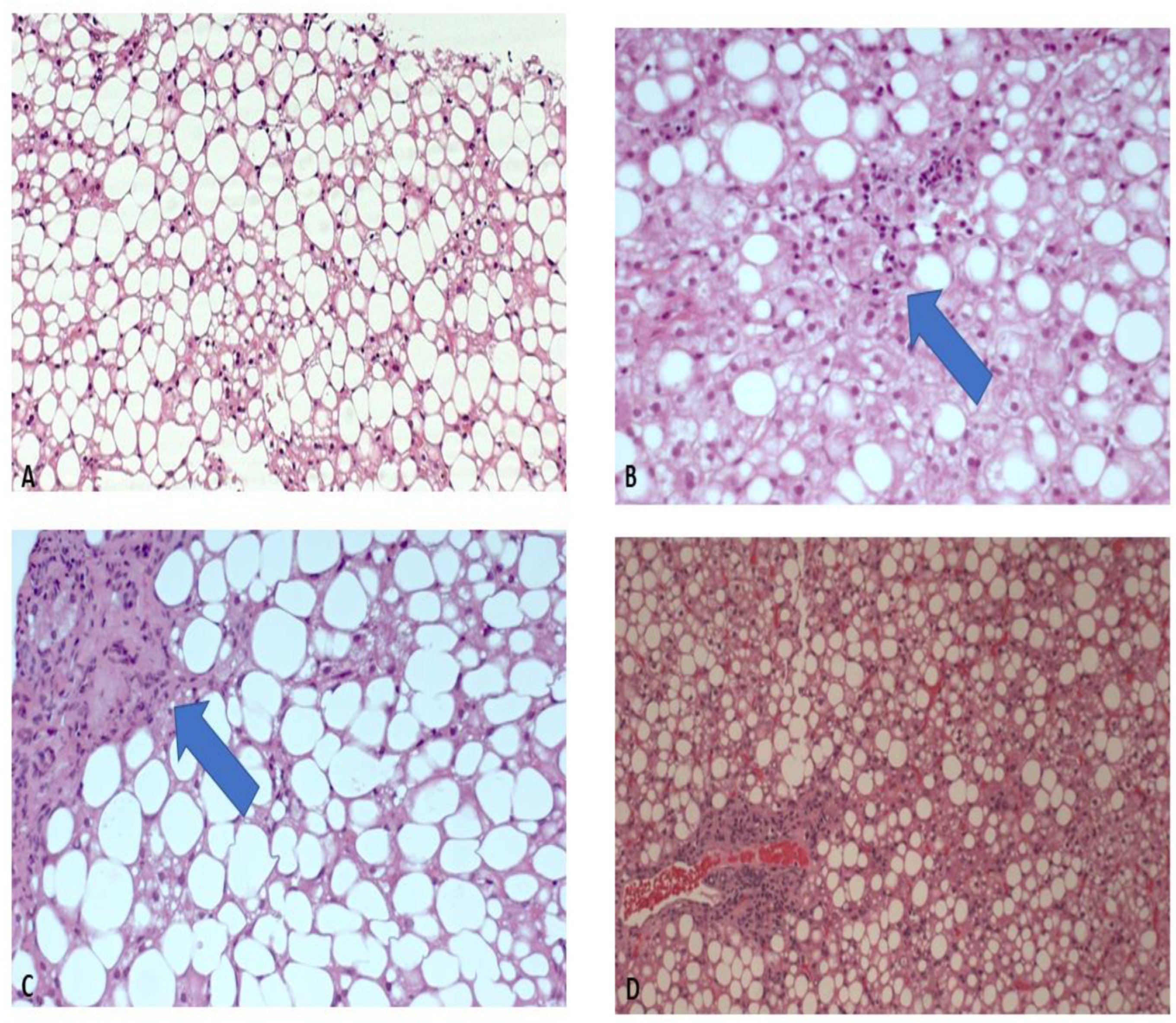
4. Results
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Liang, S.; Yu, Z.; Song, X.; Wang, Y.; Li, M.; Xue, J. Reduced Growth Hormone Secretion is Associated with Nonalcoholic Fatty Liver Disease in Obese Children. Horm. Metab. Res. 2018, 50, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, D.; Cabello-Verrugio, C.; Solís, N.; San Martín, D.; Cofré, C.; Pizarro, M.; Arab, J.P.; Abrigo, J.; Campos, F.; Irigoyen, B.; et al. Somatotropic Axis Dysfunction in Non-Alcoholic Fatty Liver Disease: Beneficial Hepatic and Systemic Effects of Hormone Supplementation. Int. J. Mol. Sci. 2018, 19, 1339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stanley, T.L.; Fourman, L.T.; Zheng, I.; McClure, C.M.; Feldpausch, M.N.; Torriani, M.; Corey, K.E.; Chung, R.T.; Lee, H.; Kleiner, D.E.; et al. Relationship of IGF-1 and IGF-Binding Proteins to Disease Severity and Glycemia in Nonalcoholic Fatty Liver Disease. J. Clin. Endocrinol. Metab. 2021, 106, e520–e533. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, T.; Nakao, K.; Hamasaki, K.; Furukawa, R.; Tsuruta, S.; Ueda, Y.; Taura, N.; Shibata, H.; Fujimoto, M.; Toriyama, K.; et al. Role of growth hormone, insulin-like growth factor 1 and insulin-like growth factor-binding protein 3 in development of non-alcoholic fatty liver disease. Hepatol. Int. 2007, 1, 287–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clemmons, D.R. The relative roles of growth hormone and IGF-1 in controlling insulin sensitivity. J. Clin. Investig. 2004, 113, 25–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, Y.; Menon, R.K.; Cohen, P.; Hwang, D.; Clemens, T.; DiGirolamo, D.J.; Kopchick, J.J.; Le Roith, D.; Trucco, M.; Sperling, M.A. Liver-specific deletion of the growth hormone receptor reveals essential role of growth hormone signaling in hepatic lipid metabolism. J. Biol. Chem. 2009, 284, 19937–19944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fusco, A.; Miele, L.; D’Uonnolo, A.; Forgione, A.; Riccardi, L.; Cefalo, C.; Barini, A.; Bianchi, A.; Giampietro, A.; Cimino, V.; et al. Nonalcoholic fatty liver disease is associated with increased GHBP and reduced GH/IGF-I levels. Clin. Endocrinol. 2012, 77, 531–536. [Google Scholar] [CrossRef]
- Bredella, M.A.; Torriani, M.; Thomas, B.J.; Ghomi, R.H.; Brick, D.J.; Gerweck, A.V.; Miller, K.K. Peak growth hormone-releasing hormone-arginine-stimulated growth hormone is inversely associated with intramyocellular and intrahepatic lipid content in premenopausal women with obesity. J. Clin. Endocrinol. Metab. 2009, 94, 3995–4002. [Google Scholar] [CrossRef] [Green Version]
- Clemmons, D.R. Role of IGF-binding proteins in regulating IGF responses to changes in metabolism. J. Mol. Endocrinol. 2018, 61, T139–T169. [Google Scholar] [CrossRef] [Green Version]
- Martinez-Castillo, M.; Rosique-Oramas, D.; Medina-Avila, Z.; Perez-Hernandez, J.L.; Higuera-De la Tijera, F.; Santana-Vargas, D.; Montalvo-Jave, E.E.; Sanchez-Avila, F.; Torre, A.; Kershenobich, D.; et al. Differential production of insulin-like growth factor-binding proteins in liver fibrosis progression. Mol. Cell. Biochem. 2020, 469, 65–75. [Google Scholar] [CrossRef]
- Petaja, E.M.; Zhou, Y.; Havana, M.; Hakkarainen, A.; Lundbom, N.; Ihalainen, J.; Yki-Jarvinen, H. Phosphorylated IGFBP 1 as a non-invasive predictor of liver fat in NAFLD. Sci. Rep. 2016, 6, 24740. [Google Scholar] [CrossRef] [Green Version]
- Kotronen, A.; Lewitt, M.; Hall, K.; Brismar, K.; Yki-Jarvinen, H. Insulin-like growth factor binding protein 1 as a novel specific marker of hepatic insulin sensitivity. J. Clin. Endocrinol. Metab. 2008, 93, 4867–4872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fahlbusch, P.; Knebel, B.; Horbelt, T.; Barbosa, D.M.; Nikolic, A.; Jacob, S.; Al-Hasani, H.; Van de Velde, F.; Van Nieuwenhove, Y.; Muller-Wieland, D.; et al. Physiological Disturbance in Fatty Liver Energy Metabolism Converges on IGFBP2 Abundance and Regulation in Mice and Men. Int. J. Mol. Sci. 2020, 21, 4144. [Google Scholar] [CrossRef] [PubMed]
- Dali-Youcef, N.; Vix, M.; Costantino, F.; El-Saghire, H.; Lhermitte, B.; Callari, C.; D’Agostino, J.; Perretta, S.; Paveliu, S.; Gualtierotti, M.; et al. Interleukin-32 Contributes to Human Nonalcoholic Fatty Liver Disease and Insulin Resistance. Hepatol. Commun. 2019, 3, 1205–1220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Min, H.K.; Maruyama, H.; Jang, B.K.; Shimada, M.; Mirshahi, F.; Ren, S.; Oh, Y.; Puri, P.; Sanyal, A.J. Suppression of IGF binding protein-3 by palmitate promotes hepatic inflammatory responses. FASEB J. 2016, 30, 4071–4082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopez-Bermejo, A.; Khosravi, J.; Fernandez-Real, J.M.; Hwa, V.; Pratt, K.L.; Casamitjana, R.; Garcia-Gil, M.M.; Rosenfeld, R.G.; Ricart, W. Insulin resistance is associated with increased serum concentration of IGF-binding protein-related protein 1 (IGFBP-rP1/MAC25). Diabetes 2006, 55, 2333–2339. [Google Scholar] [CrossRef]
- Yan, H.; Li, T.; Wang, Y.; Li, H.; Xu, J.; Lu, X. Insulin-like growth factor binding protein 7 accelerates hepatic steatosis and insulin resistance in non-alcoholic fatty liver disease. Clin. Exp. Pharmacol. Physiol. 2019, 46, 1101–1110. [Google Scholar] [CrossRef]
- Takahashi, Y.; Iida, K.; Takahashi, K.; Yoshioka, S.; Fukuoka, H.; Takeno, R.; Imanaka, M.; Nishizawa, H.; Takahashi, M.; Seo, Y.; et al. Growth hormone reverses nonalcoholic steatohepatitis in a patient with adult growth hormone deficiency. Gastroenterology 2007, 132, 938–943. [Google Scholar] [CrossRef]
- Sobrevals, L.; Rodriguez, C.; Romero-Trevejo, J.L.; Gondi, G.; Monreal, I.; Paneda, A.; Juanarena, N.; Arcelus, S.; Razquin, N.; Guembe, L.; et al. Insulin-like growth factor I gene transfer to cirrhotic liver induces fibrolysis and reduces fibrogenesis leading to cirrhosis reversion in rats. Hepatology 2010, 51, 912–921. [Google Scholar]
- Vajro, P.; Lenta, S.; Socha, P.; Dhawan, A.; McKiernan, P.; Baumann, U.; Durmaz, O.; Lacaille, F.; McLin, V.; Nobili, V. Diagnosis of nonalcoholic fatty liver disease in children and adolescents: Position paper of the ESPGHAN Hepatology Committee. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 700–713. [Google Scholar] [CrossRef]
- Feldstein, A.E.; Charatcharoenwitthaya, P.; Treeprasertsuk, S.; Benson, J.T.; Enders, F.B.; Angulo, P. The natural history of non-alcoholic fatty liver disease in children: A follow-up study for up to 20 years. Gut 2009, 58, 1538–1544. [Google Scholar] [CrossRef] [Green Version]
- Rosenfeld, R.G.; Albertsson-Wikland, K.; Cassorla, F.; Frasier, S.D.; Hasegawa, Y.; Hintz, R.L.; LaFranchi, S.; Lippe, B.; Loriaux, L.; Melmed, S. Diagnostic controversy: The diagnosis of childhood growth hormone deficiency revisited. J. Clin. Endocrinol. Metab. 1995, 80, 1532–1540. [Google Scholar] [PubMed]
- Linea Guida Clinica Sulla Diagnosi del Deficit di GH. Available online: https://www.ospedalebambinogesu.it/ (accessed on 10 September 2022).
- Conwell, L.S.; Trost, S.G.; Brown, W.J.; Batch, J.A. Indexes of insulin resistance and secretion in obese children and adolescents: A validation study. Diabetes Care 2004, 27, 314–319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunt, E.M.; Kleiner, D.E.; Wilson, L.A.; Belt, P.; Neuschwander-Tetri, B.A.; NASH Clinical Research Network (CRN). Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: Distinct clinicopathologic meanings. Hepatology 2011, 53, 810–820. [Google Scholar] [CrossRef] [Green Version]
- Santiago-Rolón, A.; Purcell, D.; Rosado, K.; Toro, D.H. A comparison of brunt’s criteria, the non-alcoholic fatty liver disease activity score (NAS), and a proposed NAS scoring that includes fibrosis in non-alcoholic fatty liver disease staging. Puerto Rico Health Sci. J. 2015, 34, 189–194. [Google Scholar]
- Rufinatscha, K.; Ress, C.; Folie, S.; Haas, S.; Salzmann, K.; Moser, P.; Dobner, J.; Weiss, G.; Iruzubieta, P.; Arias-Loste, M.T.; et al. Metabolic effects of reduced growth hormone action in fatty liver disease. Hepatol. Int. 2018, 12, 474–481. [Google Scholar] [CrossRef] [Green Version]
- Dichtel, L.E.; Corey, K.E.; Misdraji, J.; Bredella, M.A.; Schorr, M.; Osganian, S.A.; Young, B.J.; Sung, J.C.; Miller, K.K. The Association Between IGF-1 Levels and the Histologic Severity of Nonalcoholic Fatty Liver Disease. Clin. Transl. Gastroenterol. 2017, 8, e217. [Google Scholar] [CrossRef] [PubMed]
- Dichtel, L.E.; Cordoba-Chacon, J.; Kineman, R.D. Growth Hormone and Insulin-Like Growth Factor 1 Regulation of Nonalcoholic Fatty Liver Disease. J. Clin. Endocrinol. Metab. 2022, 107, 1812–1824. [Google Scholar] [CrossRef]
- Osganian, S.A.; Subudhi, S.; Masia, R.; Drescher, H.K.; Bartsch, L.M.; Chicote, M.L.; Chung, R.T.; Gee, D.W.; Witkowski, E.R.; Bredella, M.A.; et al. Expression of IGF-1 receptor and GH receptor in hepatic tissue of patients with nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Growth Horm. IGF Res. 2022, 65, 101482. [Google Scholar] [CrossRef]
- Mauras, N.; O’Brien, K.O.; Welch, S.; Rini, A.; Helgeson, K.; Vieira, N.E.; Yergey, A.L. Insulin-like growth factor I and growth hormone (GH) treatment in GH-deficient humans: Differential effects on protein, glucose, lipid, and calcium metabolism. J. Clin. Endocrinol. Metab. 2000, 85, 1686–1694. [Google Scholar] [CrossRef] [Green Version]
- Lambert, J.E.; Ramos-Roman, M.A.; Browning, J.D.; Parks, E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology 2014, 146, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Cordoba-Chacon, J.; Sarmento-Cabral, A.; Del Rio-Moreno, M.; Diaz-Ruiz, A.; Subbaiah, P.V.; Kineman, R.D. Adult-Onset Hepatocyte GH Resistance Promotes NASH in Male Mice, Without Severe Systemic Metabolic Dysfunction. Endocrinology 2018, 159, 3761–3774. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.S.; Weiss, J.J.; Fourman, L.T.; Buckless, C.; Branch, K.L.; Lee, H.; Torriani, M.; Misra, M.; Stanley, T.L. Effect of recombinant human growth hormone on liver fat content in young adults with nonalcoholic fatty liver disease. Clin. Endocrinol. 2021, 94, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Hjelholt, A.; Høgild, M.; Bak, A.M.; Arlien-Søborg, M.C.; Bæk, A.; Jessen, N.; Richelsen, B.; Pedersen, S.B.; Møller, N.; Jørgensen, J.O.L. Growth Hormone and Obesity. Endocrinol. Metab. Clin. N. Am. 2020, 49, 239–250. [Google Scholar] [CrossRef]
- Hribal, M.L.; Procopio, T.; Petta, S.; Sciacqua, A.; Grimaudo, S.; Pipitone, R.M.; Perticone, F.; Sesti, G. Insulin-like growth factor-I, inflammatory proteins, and fibrosis in subjects with nonalcoholic fatty liver disease. J. Clin. Endocrinol. Metab. 2013, 98, E304–E308. [Google Scholar] [CrossRef] [Green Version]
- Attallah, H.; Friedlander, A.L.; Hoffman, A.R. Visceral obesity, impaired glucose tolerance, metabolic syndrome, and growth hormone therapy. Growth Horm. IGF Res. 2006, 16, S62–S67. [Google Scholar] [CrossRef]
- Bredella, M.A.; Gerweck, A.V.; Lin, E.; Landa, M.G.; Torriani, M.; Schoenfeld, D.A.; Hemphill, L.C.; Miller, K.K. Effects of GH on body composition and cardiovascular risk markers in young men with abdominal obesity. J. Clin. Endocrinol. Metab. 2013, 98, 3864–3872. [Google Scholar] [CrossRef]

| Variables | Mean (SD) or Median (25th–75th Centile) |
|---|---|
| Age, years | 12.9 (2.1) |
| Sex (M/F)% | 88/103 (46.2/53.8) |
| BMI, kg/sqm | 28.5 (4.7) |
| WC, cm (IQR) | 88.5 (81–99) |
| Uric acid, mg/dL | 5.2 (1.6) |
| ALT, UI/L (IQR) | 51 (31–78) |
| AST, UI/L (IQR) | 39 (27–54) |
| GGT, UI/L (IQR) | 21 (15–29) |
| Total-cholesterol, mg/dL (IQR) | 158 (132–179) |
| HDL cholesterol, mg/dL(IQR) | 43 (36–48) |
| Triglycerides, mg/dL (IQR) | 138 (73–151) |
| Glucose, mg/dL (IQR) | 82.9 (76–91) |
| Insulin, μUI/ML (IQR) | 17.6 (10–28.3) |
| HOMA-IR | 3.7 (3.4) |
| DBP, mmHg (IQR) | 68 (60–74) |
| SBP, mmHg (IQR) | 110 (101–118) |
| GH, ng/mL (IQR) | 7.8 (5.8–9) |
| IGF1, ng/mL (IQR) | 266 (198–345) |
| IGFBP-3, µg/mL (IQR) | 2.34 (1.5–3.2) |
| TNF-a (IQR), pg/ml | 51 (7.5–77.3) |
| IL-6 (IQR), pg/mL | 29 (19–33.5) |
| Tanner’s Stage I–II–III (Approximatively 12–15 Years) | Tanner’s Stage IV–V (Approximatively > 15 Years) | Tanner’s Stage: I–II–III vs. IV–V p Values | |||||
|---|---|---|---|---|---|---|---|
| All (151) | F (85) | M (66) | All (40) | F (18) | M (22) | ||
| Age, years | 13.5 (1.33) | 12.9 (1.3) | 12.6(1.3) | 15.9 (1.1) | 15.9 (1.3) | 16 (0.9) | 0.0001 |
| BMI, kg/sqm | 27.1 (4.2) | 27 (24–29) | 27.1 (24–29.5) | 29.8 (5.6) | 30 (25–33) | 28.8 (25–31) | 0.01 |
| WC, cm (IQR) | 84 (78–92) | 83 (78–92) | 83 (77–90.5) | 88 (78–97) | 87 (76–96) | 88 (81–97) | 0.04 |
| Uric acid, mg/dL | 5.2 (1.6) | 5.4 (1.6) | 5 (1.5) | 5.7 (1.6) | 5.3 (1.9) | 6 (1.2) | 0.14 |
| ALT, UI/L (IQR) | 59.7 (32–75.5) | 63 (33–78) | 55 (31–73) | 73 (28–96) | 85 (32–130) | 65 (28–78) | 0.08 |
| AST, UI/L (IQR) | 43 (27–52.5) | 44 (30–56) | 41.5 (26–49) | 45 (25–58) | 57 (24–67) | 39.5 (25–44.7) | 0.65 |
| GGT, UI/L (IQR) | 23.6 (15–27) | 25 (15–30) | 22 (14–25) | 32 (14–39) | 39 (31–18) | 26 (13–28) | 0.01 |
| Total-cholesterol, mg/dL (IQR) | 158 (132–178) | 165 (145–189) | 149 (123–169) | 161 (131–180) | 173 (131–216) | 151 (132–165) | 0.66 |
| HDL, mg/dL (IQR) | 46 (38–48.5) | 44.6 (39–49) | 47 (36–48) | 45 (36–48) | 46 (36–53) | 45 (35–44) | 0.82 |
| Triglycerides, mg/dL (IQR) | 104 (69–117) | 109 (97–122) | 99 (59–110) | 126 (89–168) | 125 (90–170) | 127 (89–163) | 0.05 |
| Glucose, mg/dL (IQR) | 83.6 (76–90) | 83 (76–89) | 84 (77–93) | 80 (76–86) | 85 (77–92) | 78 (71–83) | 0.13 |
| Insulin, μUI/ML (IQR) | 17 (10–24) | 19 (10–25.5) | 16 (10–21) | 18(10–24) | 22 (11–29) | 15 (10–19) | 0.71 |
| HOMA-IR (IQR) | 3.7 (2–4.9) | 3.9 (2.2–4.9) | 3.4 (2–5) | 3.8 (2–4.5) | 4.9 (2.8–6.9) | 3.1 (1.8–4) | 0.81 |
| DBP, mmHg (IQR) | 67 (60–74) | 67 (59–79) | 67.5 (61–73) | 66 (60–73) | 70 (68–75) | 63 (58–68) | 0.49 |
| SBP, mmHg (IQR) | 110 (101–118) | 110.5 (102–118) | 111 (102–115) | 111.5 (103–120) | 110 (105–118) | 112 (102–120) | 0.62 |
| TNF-a | 40 (7–47) | 41 (7–22.5) | 36.5(12–59) | 55.2 (43–89) | 44.5 (30–49) | 41 (33–78) | 0.06 |
| IL-6 | 26 (18–31.2) | 24 (19–29.5) | 27 (18.5–32) | 36.5 (25–50) | 25 (21–35) | 39 (26–49) | 0.13 |
| GH, ng/mL (IQR) | 10.2 (8–11) | 10.5(3.2) | 10 (8–11) | 10 (8–11) | 8.1 (7.5–11) | 10.4 (2–11.3) | 0.67 |
| IGF1, ng/mL(IQR) | 272 (198–345) | 267 (198–305) | 279 (208–347) | 268 (198–345) | 233 (164–305) | 395 (125–205) | 0.81 |
| IGFBP-3, µg/mL (IQR) | 2.4 (1.5–3.2) | 2.4 (1.5–3.2) | 2.4 (1.5–3.2) | 2.2 (1.5–2.8) | 2.2 (1.4–2.6) | 2.3 (1.6–2.9) | 0.5 |
| Histologic Characteristic | Number of Patients | Percentage |
|---|---|---|
| Steatosis | ||
| 0 | 16 | 8.4% |
| 1 | 64 | 33.7% |
| 2 | 72 | 37.9% |
| 3 | 38 | 20% |
| Portal inflammation | ||
| 0 | 19 | 10% |
| 1 | 139 | 73.2% |
| 2 | 32 | 16.8% |
| Lobular inflammation | ||
| 0 | 73 | 38.6% |
| 1 | 100 | 52.9% |
| 2 | 16 | 8.5% |
| Ballooning | ||
| 0 | 99 | 52.1% |
| 1 | 62 | 32.6% |
| 2 | 29 | 15.3% |
| Fibrosis | ||
| 0 | 57 | 30% |
| 1 | 108 | 56.8% |
| 2 | 18 | 9.5% |
| 3 | 7 | 3.7% |
| NAS | ||
| 0 | 13 | 6.8% |
| 1 | 11 | 5.8% |
| 2 | 43 | 22.6% |
| 3 | 34 | 17.9% |
| 4 | 39 | 20.5% |
| 5 | 23 | 12.6% |
| 6 | 24 | 12.6% |
| 7 | 3 | 1.6% |
| Non-Steatohepatitis (N 102) | Steatohepatitis (N 89) | p | |
|---|---|---|---|
| Age, years | 12.9 (1.6) | 13.1 (1.8) | 0.40 |
| Sex, (F/M) | 57/45 | 46/43 | 0.79 |
| BMI, kg/sqm | 26.8 (4.2) | 28.3 (4.4) | 0.01 |
| WC, cm (IQR) | 85.9 (72–95) | 90 (80–99) | 0.02 |
| Uric acid, mg/dL | 5.4 (1.6) | 6 (1.4) | 0.01 |
| ALT, UI/L (IQR) | 49 (24–66) | 75 (39–90) | 0.001 |
| AST, UI/L (IQR) | 37 (25–45) | 50 (33–62) | 0.001 |
| GGT, UI/L (IQR) | 21 (13–24) | 30 (19–38) | 0.04 |
| Total-cholesterol, mg/dL (IQR) | 158 (93–175) | 161 (78–183) | 0.22 |
| HDL, mg/dL (IQR) | 44 (37–49) | 47 (35–48) | 0.70 |
| Triglycerides, mg/dL (IQR) | 100 (73–138) | 127 (74–146) | 0.04 |
| Glucose, mg/dL (IQR) | 80 (75–86) | 89 (76–96) | 0.03 |
| Insulin, μUI/ML (IQR) | 17 (10–24) | 24 (18–32) | 0.02 |
| HOMA-IR (IQR) | 3.5 (2.2–4.5) | 4.6 (3.2–6.2) | 0.001 |
| DBP, mmHg (IQR) | 65 (58–72) | 68 (61–75) | 0.08 |
| SBP, mmHg (IQR) | 110 (101–118) | 114 (103–121) | 0.31 |
| TNF-a, pg/mL | 44 (7.5–44) | 72 (12–72) | 0.02 |
| IL-6, pg/mL | 25 (22–35.5) | 35 (15–39) | 0.048 |
| GH, ng/mL (IQR) | 7.5 (6–8.2) | 5.4 (4–6.8) | 0.001 |
| IGF1, ng/mL(IQR) | 284 (208–345) | 245 (150–310) | 0.01 |
| IGFBP-3, µg/mL (IQR) | 2.6 (1.6–3.4) | 2 (1.4–2.8) | 0.02 |
| Logistic Regression Analysis * | Standardized Beta Coefficients | SE | p Value |
|---|---|---|---|
| GH, ng/mL | |||
| Total Cholesterol | 0.13 | 0.04 | 0.003 |
| HDL-Cholesterol | −0.17 | 0.06 | 0.008 |
| Triglycerides | −0.01 | 0.02 | 0.37 |
| Glucose | 1.7 | 0.02 | 0.002 |
| Insulin | 0.03 | 0.04 | 0.45 |
| HOMA-IR | −0.33 | 0.2 | 0.09 |
| Fibrosis | −2.3 | 0.31 | 0.001 |
| Portal Inflammation | 0.56 | 0.51 | 0.41 |
| Lobular Inflammation | −0.36 | 0.34 | 0.28 |
| TNF-a | −1.2 | 0.12 | 0.04 |
| IGF1, ng/mL | |||
| Total Cholesterol | 0.41 | 1.3 | 0.75 |
| HDL-Cholesterol | 0.68 | 1.97 | 0.72 |
| Triglycerides | −0.07 | 0.35 | 0.83 |
| Glucose | −0.69 | 0.71 | 0.32 |
| Insulin | 3.8 | 1.5 | 0.01 |
| HOMA-IR | −2.7 | 1.7 | 0.001 |
| Fibrosis | −2.8 | 1.1 | 0.001 |
| Portal Inflammation | −1.3 | 1.7 | 0.09 |
| Lobular Inflammation | −1.0 | 1.1 | 0.42 |
| TNF-a | −0.78 | 0.34 | 0.25 |
| IGFBP-3, µg/mL | |||
| Total Cholesterol | 0.01 | 0.01 | 0.46 |
| HDL-Cholesterol | −0.02 | 0.03 | 0.21 |
| Triglycerides | 0.01 | 0.04 | 0.69 |
| Glucose | −0.01 | 0.07 | 0.16 |
| Insulin | −0.02 | 0.016 | 0.94 |
| HOMA-IR | −0.09 | 0.07 | 0.20 |
| Fibrosis | −0.02 | 0.04 | 0.45 |
| Portal Inflammation | 0.41 | 0.54 | 0.35 |
| Lobular Inflammation | −0.01 | 0.9 | 0.74 |
| TNF-a | −0.11 | 0.02 | 0.85 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mosca, A.; Della Volpe, L.; Alisi, A.; Panera, N.; Maggiore, G.; Vania, A. The Role of the GH/IGF1 Axis on the Development of MAFLD in Pediatric Patients with Obesity. Metabolites 2022, 12, 1221. https://doi.org/10.3390/metabo12121221
Mosca A, Della Volpe L, Alisi A, Panera N, Maggiore G, Vania A. The Role of the GH/IGF1 Axis on the Development of MAFLD in Pediatric Patients with Obesity. Metabolites. 2022; 12(12):1221. https://doi.org/10.3390/metabo12121221
Chicago/Turabian StyleMosca, Antonella, Luca Della Volpe, Anna Alisi, Nadia Panera, Giuseppe Maggiore, and Andrea Vania. 2022. "The Role of the GH/IGF1 Axis on the Development of MAFLD in Pediatric Patients with Obesity" Metabolites 12, no. 12: 1221. https://doi.org/10.3390/metabo12121221






