In Silico Characterization of Calcineurin from Pathogenic Obligate Intracellular Trypanosomatids: Potential New Biological Roles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Database Inspection
2.2. Obtaining Consensus Sequences and Multiple Alignment of CaN
2.3. Evaluation of Conserved Domains, Determination of Physicochemical Parameters, and Hydrophobicity Profiles
2.4. In Silico Identification of Protein–Protein Interactions of CaN Regulatory Subunits
2.5. Prediction of the Subcellular Localization of CaN in Pathogenic Intracellular Trypanosomatids
2.6. Prediction of Entry to the Non-Classical Secretory Pathway of CaN in Pathogenic Intracellular Trypanosomatids
2.7. Prediction of Cleavage by Calpains in CaN Catalytic Subunits of Pathogenic Intracellular Trypanosomatids
2.8. Prediction of Phosphorylation Sites in the CaM-Binding Domain (CaM-BD) of CaN Catalytic Subunits from Leishmania Spp.
3. Results
3.1. The CaN Subunits of Obligate Intracellular Trypanosomatids Possess Diverse, Partially Conserved Domain Architecture and Potential Calpain Cleavage Sites
3.2. The CaM-Binding Domain in Leishmania Spp. Has Greater Potential to Be Regulated by Phosphorylation Than Its Human Ortholog
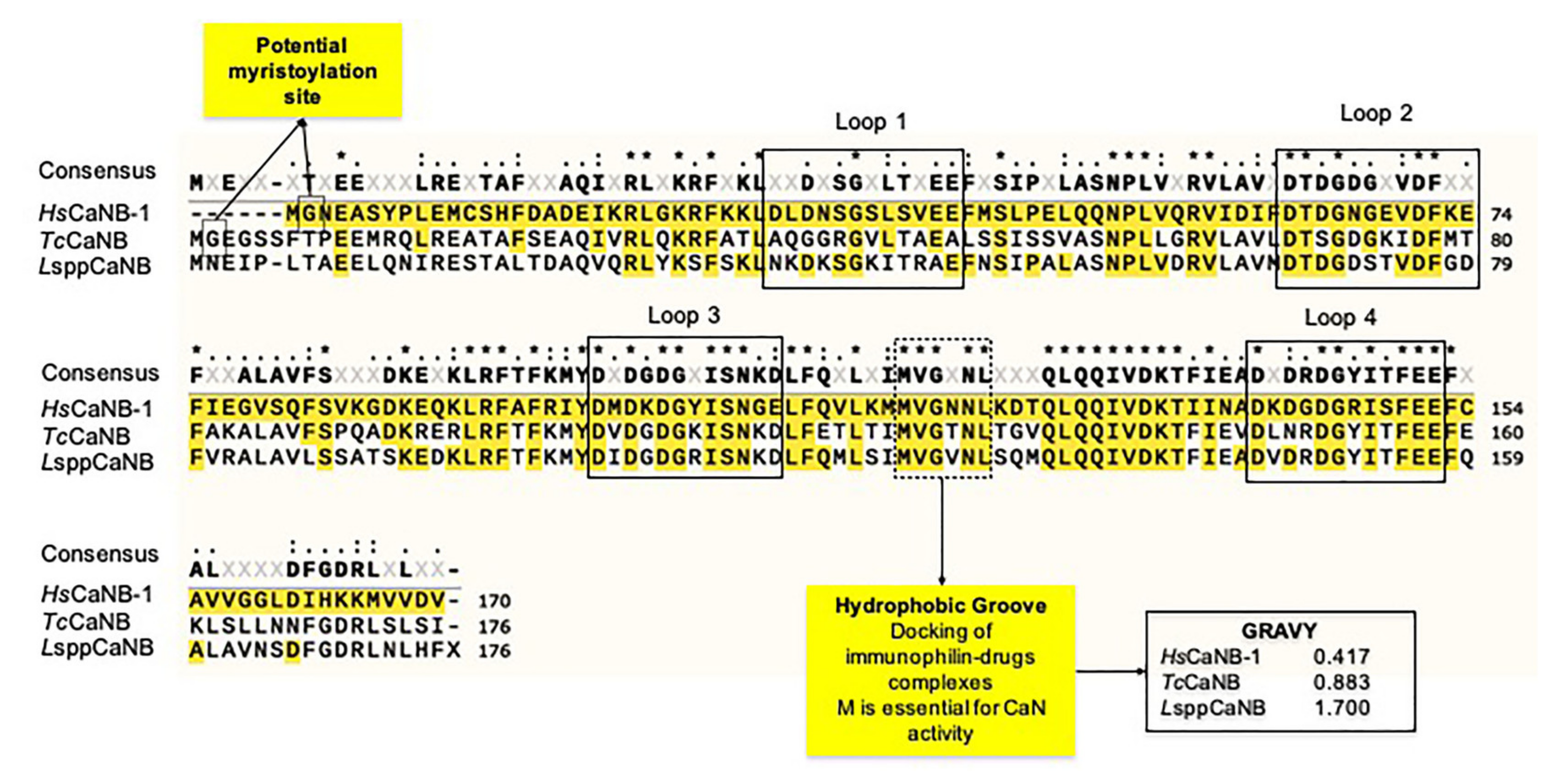
3.3. CaN Regulatory Subunits of Obligate Intracellular Trypanosomatids Differ in Their Calcium-Binding Domains from Their Human Counterpart and Myristoylation Potential, but Preserve Some Canonical EF-Loops and the Docking Site for Immunophilin–Immunosuppressive Drug Complexes
3.4. CaN Regulatory and Catalytic Subunits of Obligate Intracellular and Human Trypanosomatids Would Share the Subcellular Distribution Pattern, with Some Exceptions
3.5. CaN Regulatory Subunits of Obligate Intracellular Trypanosomatids Interact Only with Their Catalytic Monomer and Related Immunophilins
3.6. CaN Regulatory and Catalytic Subunits of Obligate Intracellular Trypanosomatids Have a Differential Potential to Be Secreted by the Non-Classical Pathway
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alvar, J.; Vélez, I.D.; Bern, C.; Herrero, M.; Desjeux, P.; Cano, J.; Jannin, J.; de Boer, M. Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE 2012, 7, e35671. [Google Scholar] [CrossRef]
- Norman, F.F.; López-Vélez, R. Chagas disease: Comments on the 2018 PAHO Guidelines for diagnosis and management. J. Travel Med. 2019, 26, taz060. [Google Scholar] [CrossRef]
- WHO. World Health Organization: Leishmaniasis and Chagas Disease. 2020. Available online: https://www.who.int/news/item/30-01-2009-leishmaniasis-the-global-trend; https://www.who.int/news-room/fact-sheets/detail/chagas-disease-(american-trypanosomiasis) (accessed on 1 May 2021).
- Sangenito, L.S.; da Silva Santos, V.; d’Avila-Levy, C.M.; Branquinha, M.H.; Souza dos Santos, A.L.; de Oliveira, S.S.C. Leishmaniasis and Chagas Disease—Neglected Tropical Diseases: Treatment Updates. Curr. Top. Med. Chem. 2019, 19, 174–177. [Google Scholar] [CrossRef]
- Aragão Horoiwa, T.; Cortez, M.; Sauter, I.P.; Migotto, A.; Bandeira, C.L.; Cerize, N.N.P.; de Oliveira, A.M. Sugar-based colloidal nanocarriers for topical meglumine antimoniate application to cutaneous leishmaniasis treatment: Ex vivo cutaneous retention and in vivo evaluation. Eur. J. Pharm. Sci. 2020, 147, 105295. [Google Scholar] [CrossRef]
- Ferreira, T.C.S.; Sauter, I.P.; Borda-Samper, L.; Bentivoglio, E.; DaMata, J.P.; Taniwaki, N.N.; Orrego, P.R.; Araya, J.E.; Lincopan, N.; Cortez, M. Effect of DODAB Nano-Sized Cationic Bilayer Fragments against Leishmania amazonensis. Molecules 2020, 25, 5741. [Google Scholar] [CrossRef] [PubMed]
- Luczywo, A.; Sauter, I.P.; da Silva Ferreira, T.C.; Cortez, M.; Romanelli, G.P.; Sathicq, G.; Asís, S.E. Microwave-assisted synthesis of 2-styrylquinoline-4-carboxylic acid derivatives to improve the toxic effect against Leishmania (Leishmania) amazonensis. J. Heterocycl. Chem. 2020, 58, 822–832. [Google Scholar] [CrossRef]
- Geiger, A.; Bossard, G.; Sereno, D.; Pissarra, J.; Lemesre, J.L.; Vincendeau, P.; Holzmuller, P. Escaping deleterious immune response in their hosts: Lessons from trypanosomatids. Front. Immunol. 2016, 7, 212. [Google Scholar] [CrossRef] [Green Version]
- De Morais, C.; Castro Lima, A.; Terra, R.; dos Santos, R.; Da-Silva, S.; Dutra, P. The dialogue of the host-parasite relationship: Leishmania spp. and Trypanosoma cruzi infection. Biomed. Res. Int. 2015, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, N.; Cortez, M. Trypanosoma cruzi: Parasite and host cell signaling during the invasion process. Subcell. Biochem. 2008, 47, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, S.; Docampo, R. Membrane proteins in trypanosomatids involved in Ca2+ homeostasis and signaling. Genes 2018, 9, 304. [Google Scholar] [CrossRef] [Green Version]
- Park, H.S.; Lee, S.C.; Cardenas, M.E.; Heitman, J. Calcium-Calmodulin-Calcineurin Signaling: A Globally Conserved Virulence Cascade in Eukaryotic Microbial Pathogens. Cell Host Microbe 2019, 26, 453–462. [Google Scholar] [CrossRef]
- Klee, C.B.; Crouch, T.H.; Krinks, M.H. Calcineurin: A calcium- and calmodulin-binding protein of the nervous system. Proc. Natl. Acad. Sci. USA 1979, 76, 6270–6273. [Google Scholar] [CrossRef] [Green Version]
- Klee, C.B.; Draetta, G.F.; Hubbard, M.J. Calcineurin. Adv. Enzymol. Relat. Areas Mol. Biol. 1988, 61, 149–200. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, R. Calmodulin-dependent protein phosphatases from microorganisms to man. A study in structural conservatism and biological diversity. Adv. Second. Messenger Phosphoprot. Res. 1993, 27, 1–23. [Google Scholar]
- Creamer, T.P. Calcineurin. Cell Commun. Signal. 2020, 18, 1–12. [Google Scholar] [CrossRef]
- Rusnak, F.; Mertz, P. Calcineurin: Form and function. Physiol. Rev. 2000, 80, 1483–1521. [Google Scholar] [CrossRef] [PubMed]
- Crabtree, G.R. Calcium, calcineurin, and the control of transcription. J. Biol. Chem. 2001, 276, 2313–2316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sikkink, R.; Haddy, A.; MacKelvie, S.; Mertz, P.; Litwiller, R.; Rusnak, F. Calcineurin subunit interactions: Mapping the calcineurin B binding domain on calcineurin A. Biochemistry 1995, 34, 8348–8356. [Google Scholar] [CrossRef]
- Watanabe, Y.; Perrino, B.A.; Chang, B.H.; Soderling, T.R. Identification in the calcineurin A subunit of the domain that binds the regulatory B subunit. J. Biol. Chem. 1995, 270, 456–460. [Google Scholar] [CrossRef] [Green Version]
- Kincaid, R.L.; Nightingale, M.S.; Martin, B.M. Characterization of a cDNA clone encoding the calmodulin-binding domain of mouse brain calcineurin. Proc. Natl. Acad. Sci. USA 1988, 85, 8983–8987. [Google Scholar] [CrossRef] [Green Version]
- Guerini, D.; Montell, C.; Klee, C.B. Molecular cloning and characterization of the genes encoding the two subunits of Drosophila melanogaster calcineurin. J. Biol. Chem. 1992, 267, 22542–22549. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Perrino, B.A.; Soderling, T.R. Identification of an autoinhibitory domain in calcineurin. J. Biol. Chem. 1990, 265, 1924–1927. [Google Scholar] [CrossRef]
- Kissinger, C.R.; Parge, H.E.; Knighton, D.R.; Lewis, C.T.; Pelletier, L.A.; Tempczyk, A.; Kalish, V.J.; Tucker, K.D.; Showalter, R.E.; Moomaw, E.W.; et al. Crystal structures of human calcineurin and the human FKBP12–FK506–calcineurin complex. Nature 1995, 378, 641–644. [Google Scholar] [CrossRef] [PubMed]
- Aramburu, J.; Heitman, J.; Crabtree, G.R. Calcineurin: A central controller of signalling in eukaryotes. EMBO Rep. 2004, 5, 343–348. [Google Scholar] [CrossRef] [Green Version]
- Schreiber, S.L.; Crabtree, G.R. The mechanism of action of cyclosporin A and FK506. Immunol. Today 1992, 13, 136–142. [Google Scholar] [CrossRef]
- Ho, S.; Clipstone, N.; Timmermann, L.; Northrop, J.; Graef, I.; Fiorentino, D.; Nourse, J.; Crabtree, G.R. The mechanism of action of cyclosporin A and FK506. Clin. Immunol. Immunopathol. 1996, 80, S40–S45. [Google Scholar] [CrossRef]
- Juvvadi, P.R.; Lee, S.C.; Heitman, J.; Steinbach, W.J. Calcineurin in fungal virulence and drug resistance: Prospects for harnessing targeted inhibition of calcineurin for an antifungal therapeutic approach. Virulence 2017, 8, 186–197. [Google Scholar] [CrossRef] [Green Version]
- Fraga, D.; Sehring, I.M.; Kissmehl, R.; Reiss, M.; Gaines, R.; Hinrichsen, R.; Plattner, H. Protein phosphatase 2B (PP2B, calcineurin) in Paramecium: Partial characterization reveals that two members of the unusually large catalytic subunit family have distinct roles in calcium-dependent processes. Eukaryot. Cell 2010, 9, 1049–1063. [Google Scholar] [CrossRef] [Green Version]
- Stathopoulos-Gerontides, A.; Guo, J.J.; Cyert, M.S. Yeast calcineurin regulates nuclear localization of the Crz1p transcription factor through dephosphorylation. Genes Dev. 1999, 13, 798–803. [Google Scholar] [CrossRef] [Green Version]
- Balaji, S.; Madan Babu, M.; Iyer, L.M.; Aravind, L. Discovery of the principal specific transcription factors of Apicomplexa and their implication for the evolution of the AP2-integrase DNA binding domains. Nucleic Acids Res. 2005, 33, 3994–4006. [Google Scholar] [CrossRef] [Green Version]
- Araya, J.E.; Cornejo, A.; Orrego, P.R.; Cordero, E.M.; Cortéz, M.; Olivares, H.; Neira, I.; Sagua, H.; Franco da Silveira, J.; Yoshida, N.; et al. Calcineurin B of the human protozoan parasite Trypanosoma cruzi is involved in cell invasion. Microbes Infect. 2008, 10, 892–900. [Google Scholar] [CrossRef]
- Orrego, P.R.; Olivares, H.; Cordero, E.M.; Bressan, A.; Cortez, M.; Sagua, H.; Neira, I.; González, J.; da Silveira, J.F.; Yoshida, N.; et al. A cytoplasmic new catalytic subunit of calcineurin in Trypanosoma cruzi and its molecular and functional characterization. PLoS Negl. Trop. Dis. 2014, 8, e2676. [Google Scholar] [CrossRef] [Green Version]
- Naderer, T.; Dandash, O.; Mcconville, M.J. Calcineurin is required for Leishmania major stress response pathways and for virulence in the mammalian host. Mol. Microbiol. 2011, 80, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, A.G.; Dey, C.S. Role of calmodulin and calcineurin in regulating flagellar motility and wave polarity in Leishmania. Parasitol. Res. 2017, 116, 3221–3228. [Google Scholar] [CrossRef]
- Aslett, M.; Aurrecoechea, C.; Berriman, M.; Brestelli, J.; Brunk, B.P.; Carrington, M.; Depledge, D.P.; Fischer, S.; Gajria, B.; Gao, X.; et al. TriTrypDB: A functional genomic resource for the Trypanosomatidae. Nucleic Acids Res. 2010, 38, D457–D462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno, V.R.; Agüero, F.; Tekiel, V.; Sánchez, D.O. The Calcineurin A homologue from Trypanosoma cruzi lacks two important regulatory domains. Acta Trop. 2007, 101, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, R.; Tariq, H.; McElhinney, H.; Szöor, B.; Huxley-Jones, J.; Stevens, R.; Matthews, K.; Tabernero, L. The TriTryp phosphatome: Analysis of the protein phosphatase catalytic domains. BMC Genomics 2007, 8, 434. [Google Scholar] [CrossRef] [Green Version]
- Leifso, K.; Cohen-Freue, G.; Dogra, N.; Murray, A.; McMaster, W.R. Genomic and proteomic expression analysis of Leishmania promastigote and amastigote life stages: The Leishmania genome is constitutively expressed. Mol. Biochem. Parasitol. 2007, 152, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Rice, P.; Longden, L.; Bleasby, A. EMBOSS: The european molecular biology open software suite. Trends Genet. 2000, 16, 276–277. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- Marchler-Bauer, A.; Lu, S.; Anderson, J.B.; Chitsaz, F.; Derbyshire, M.K.; DeWeese-Scott, C.; Fong, J.H.; Geer, L.Y.; Geer, R.C.; Gonzales, N.R.; et al. CDD: A Conserved Domain Database for the functional annotation of proteins. Nucleic Acids Res. 2011, 39, D225–D229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marchler-Bauer, A.; Derbyshire, M.K.; Gonzales, N.R.; Lu, S.; Chitsaz, F.; Geer, L.Y.; Geer, R.C.; He, J.; Gwadz, M.; Hurwitz, D.I.; et al. CDD: NCBI’s conserved domain database. Nucleic Acids Res. 2015, 43, D222–D226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, S.; Wang, J.; Chitsaz, F.; Derbyshire, M.K.; Geer, R.C.; Gonzales, N.R.; Gwadz, M.; Hurwitz, D.I.; Marchler, G.H.; Song, J.S.; et al. CDD/SPARCLE: The conserved domain database in 2020. Nucleic Acids Res. 2020, 48, D265–D268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Castro, E.; Sigrist, C.J.A.; Gattiker, A.; Bulliard, V.; Langendijk-Genevaux, P.S.; Gasteiger, E.; Bairoch, A.; Hulo, N. ScanProsite: Detection of PROSITE signature matches and ProRule-associated functional and structural residues in proteins. Nucleic Acids Res. 2006, 34, W362–W365. [Google Scholar] [CrossRef] [PubMed]
- Hulo, N.; Bairoch, A.; Bulliard, V.; Cerutti, L.; Cuche, B.A.; De castro, E.; Lachaize, C.; Langendijk-Genevaux, P.S.; Sigrist, C.J.A. The 20 years of PROSITE. Nucleic Acids Res. 2008, 36, D245–D249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasteiger, E.; Hoogland, C.; Gattiker, A.; Duvaud, S.; Wilkins, M.R.; Appel, R.D.; Bairoch, A. Protein Identification and Analysis Tools on the ExPASy Server. In The Proteomics Protocols Handbook; Walker, J.M., Ed.; Humana Press: Totowa, NJ, USA, 2005. [Google Scholar]
- Kyte, J.; Doolittle, R.F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 1982, 157, 105–132. [Google Scholar] [CrossRef] [Green Version]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef] [Green Version]
- Gazestani, V.H.; Yip, C.W.; Nikpour, N.; Berghuis, N.; Salavati, R. TrypsNetDB: An integrated framework for the functional characterization of trypanosomatid proteins. PLoS Negl. Trop. Dis. 2017, 11, e0005368. [Google Scholar] [CrossRef]
- Horton, P.; Park, K.J.; Obayashi, T.; Fujita, N.; Harada, H.; Adams-Collier, C.J.; Nakai, K. WoLF PSORT: Protein localization predictor. Nucleic Acids Res. 2007, 35, W585–W587. [Google Scholar] [CrossRef] [Green Version]
- Yu, C.S.; Cheng, C.W.; Su, W.C.; Chang, K.C.; Huang, S.W.; Hwang, J.K.; Lu, C.H. CELLO2GO: A web server for protein subCELlular lOcalization prediction with functional gene ontology annotation. PLoS ONE 2014, 9, e99368. [Google Scholar] [CrossRef] [Green Version]
- Bendtsen, J.D.; Jensen, L.J.; Blom, N.; Von Heijne, G.; Brunak, S. Feature-based prediction of non-classical and leaderless protein secretion. Protein Eng. Des. Sel. 2004, 17, 349–356. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Cao, J.; Gao, X.; Ma, Q.; Ren, J.; Xue, Y. GPS-CCD: A novel computational program for the prediction of calpain cleavage sites. PLoS ONE 2011, 6, e19001. [Google Scholar] [CrossRef]
- Blom, N.; Gammeltoft, S.; Brunak, S. Sequence and structure-based prediction of eukaryotic protein phosphorylation sites. J. Mol. Biol. 1999, 294, 1351–1362. [Google Scholar] [CrossRef] [PubMed]
- Blom, N.; Sicheritz-Pontén, T.; Gupta, R.; Gammeltoft, S.; Brunak, S. Prediction of post-translational glycosylation and phosphorylation of proteins from the amino acid sequence. Proteomics 2004, 4, 1633–1649. [Google Scholar] [CrossRef] [PubMed]
- Kakalis, L.T.; Kennedy, M.; Sikkink, R.; Rusnak, F.; Armitage, I.M. Characterization of the calcium-binding sites of calcineurin B. FEBS Lett. 1995, 362, 55–58. [Google Scholar] [CrossRef] [Green Version]
- Szöör, B. Trypanosomatid protein phosphatases. Mol. Biochem. Parasitol. 2010, 173, 53–63. [Google Scholar] [CrossRef]
- Ferreira, T.C.S.; Siess-Portugal, C.; Orrego, P.R.; Araya, J.E.; Cortez, M. Papel de calcineurinas na interação Leishmania-macrófago. In Atualidades em Medicina Tropical no Brasil: Protozoários; de Oliveira Meneguetti, D.U., de Oliveira, J., Aranha Camargo, L.M., Eds.; Stricto Sensu: Rio Branco, Brazil, 2020; pp. 319–339. ISBN 978-65-86283-13-6. [Google Scholar]
- Baksh, S.; Burakoff, S.J. The role of calcineurin in lymphocyte activation. Semin. Immunol. 2000, 12, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Kurotani, A.; Tokmakov, A.A.; Sato, K.I.; Stefanov, V.E.; Yamada, Y.; Sakurai, T. Localization-specific distributions of protein pI in human proteome are governed by local pH and membrane charge. BMC Mol. Cell Biol. 2019, 20, 36. [Google Scholar] [CrossRef]
- Kiraga, J.; Mackiewicz, P.; Mackiewicz, D.; Kowalczuk, M.; Biecek, P.; Polak, N.; Smolarczyk, K.; Dudek, M.R.; Cebrat, S. The relationships between the isoelectric point and: Length of proteins, taxonomy and ecology of organisms. BMC Genomics 2007, 8, 163. [Google Scholar] [CrossRef] [Green Version]
- Norris, C.M. Calpain interactions with the protein phosphatase calcineurin in neurodegeneration. In Role of Proteases in Cellular Dysfunction; Dhalla, N., Chakraborti, S., Eds.; Advances in Biochemistry in Health and Disease; Springer: New York, NY, USA, 2014; Volume 8, ISBN 9781461490999. [Google Scholar] [CrossRef]
- Wu, H.Y.; Tomizawa, K.; Matsui, H. Calpain-calcineurin signaling in the pathogenesis of calcium-dependent disorder. Acta Med. Okayama 2007, 61, 123–137. [Google Scholar] [CrossRef]
- Wu, H.Y.; Tomizawa, K.; Oda, Y.; Wei, F.Y.; Lu, Y.F.; Matsushita, M.; Li, S.T.; Moriwaki, A.; Matsui, H. Critical role of calpain-mediated cleavage of calcineurin in excitotoxic neurodegeneration. J. Biol. Chem. 2004, 279, 4929–4940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, P.; Huang, C.; Jia, Z.; Yi, F.; Yu, D.-Y.; Wei, Q. Non-catalytic domains of subunit A negatively regulate the activity of calcineurin. Biochimie 2005, 87, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; Li, X.; Li, B.; Guo, J.; Zhang, Y.; Ding, J. Calpain-mediated cleavage of calcineurin in puromycin aminonucleoside-induced podocyte injury. PLoS ONE 2016, 11, e0155504. [Google Scholar] [CrossRef]
- Sangenito, L.S.; Ennes-Vidal, V.; Marinho, F.A.; Da Mota, F.F.; Santos, A.L.; D′Avila-Levy, C.M.; Branquinha, M.H. Arrested growth of Trypanosoma cruzi by the calpain inhibitor MDL28170 and detection of calpain homologues in epimastigote forms. Parasitology 2009, 136, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Ennes-Vidal, V.; Menna-Barreto, R.F.S.; Santos, A.L.S.; Branquinha, M.H.; d’Avila-Levy, C.M. Effects of the calpain inhibitor MDL28170 on the clinically relevant forms of Trypanosoma cruzi in vitro. J. Antimicrob. Chemother. 2010, 65, 1395–1398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ennes-Vidal, V.; Menna-Barreto, R.F.S.; Santos, A.L.S.; Branquinha, M.H.; d’Avila-Levy, C.M. MDL28170, a calpain inhibitor, affects Trypanosoma cruzi metacyclogenesis, ultrastructure and attachment to Rhodnius prolixus midgut. PLoS ONE 2011, 6, e18371. [Google Scholar] [CrossRef] [Green Version]
- Marinho, F.A.; Gonçalves, K.C.S.; Oliveira, S.S.C.; Gonçalves, D.S.; Matteoli, F.P.; Seabra, S.H.; Oliveira, A.C.S.; Bellio, M.; Oliveira, S.S.; Souto-Padrón, T.; et al. The calpain inhibitor MDL28170 induces the expression of apoptotic markers in Leishmania amazonensis promastigotes. PLoS ONE 2014, 9, e87659. [Google Scholar] [CrossRef] [Green Version]
- Branquinha, M.; Marinho, F.; Sangenito, L.; Oliveira, S.; Goncalves, K.; Ennes-Vidal, V.; d’Avila-Levy, C.; Santos, A. Calpains: Potential targets for alternative chemotherapeutic intervention against human pathogenic trypanosomatids. Curr. Med. Chem. 2013, 20, 3174–3185. [Google Scholar] [CrossRef] [Green Version]
- Ono, Y.; Saido, T.C.; Sorimachi, H. Calpain research for drug discovery: Challenges and potential. Nat. Rev. Drug Discov. 2016, 15, 854–876. [Google Scholar] [CrossRef]
- Calalb, M.B.; Kincaid, R.L.; Soderling, T.R. Phosphorylation of calcineurin: Effect on calmodulin binding. Biochem. Biophys. Res. Commun. 1990, 172, 551–556. [Google Scholar] [CrossRef]
- Colbran, R.J.; Soderling, T.R. Calcium/calmodulin-independent autophosphorylation sites of calcium/calmodulin-dependent protein kinase II. Studies on the effect of phosphorylation of threonine 305/306 and serine 314 on calmodulin binding using synthetic peptides. J. Biol. Chem. 1990, 265, 11213–11219. [Google Scholar] [CrossRef]
- Jaureguia, E.; Du, L.; Gleasonc, C.; Poovaiah, B.W. Autophosphorylation of calcium/calmodulin-dependent protein kinase (CCaMK) at S343 or S344 generates an intramolecular interaction blocking the CaM-binding. Plant Signal. Behav. 2017, 12, e1343779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrino, B.A. Regulation of calcineurin phosphatase activity by its autoinhibitory domain. Arch. Biochem. Biophys. 1999, 372, 159–165. [Google Scholar] [CrossRef]
- Li, S.J.; Wang, J.; Ma, L.; Lu, C.; Wang, J.; Wu, J.W.; Wang, Z.X. Cooperative autoinhibition and multi-level activation mechanisms of calcineurin. Cell Res. 2016, 26, 336–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montenegro, M.; Cardenas, C.; Cuervo, C.; Bernal, C.; Grisard, E.C.; Thomas, M.C.; Lopez, M.C.; Puerta, C.J. Molecular characterization of calcineurin B from the non-virulent Trypanosoma rangeli kinetoplastid indicates high gene conservation. Mol. Biol. Rep. 2013, 40, 4901–4912. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Stemmer, P.M. Interactions of calcineurin A, calcineurin B, and Ca2+. Biochemistry 1999, 38, 12481–12489. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, W.; Kirberger, M.; Lee, H.W.; Ayalasomayajula, G.; Yang, J.J. Prediction of EF-hand calcium-binding proteins and analysis of bacterial EF-hand proteins. Proteins Struct. Funct. Genet. 2006, 65, 643–655. [Google Scholar] [CrossRef]
- Yang, S.A.; Klee, C.B. Low affinity Ca2+—Binding sites of calcineurin B mediate conformational changes in calcineurin A. Biochemistry 2000, 39, 16147–16154. [Google Scholar] [CrossRef]
- Natarajan, K.; Ness, J.; Wooge, C.H.; Janovick, J.A.; Conn, P.M. Specific identification and subcellular localization of three calmodulin-binding proteins in the rat gonadotrope: Spectrin, caldesmon, and calcineurin. Biol. Reprod. 1991, 44, 43–52. [Google Scholar] [CrossRef] [Green Version]
- Politino, M.; King, M.M. Calcium- and calmodulin-sensitive interactions of calcineurin with phospholipids. J. Biol. Chem. 1987, 262, 10109–10113. [Google Scholar] [CrossRef]
- Shibasaki, F.; Price, E.R.; Milan, D.; McKeon, F. Role of kinases and the phosphatase calcineurin in the nuclear shuttling of transcription factor NF-AT4. Nature 1996, 382, 370–373. [Google Scholar] [CrossRef]
- Usuda, N.; Arai, H.; Sasaki, H.; Hanai, T.; Nagata, T.; Muramatsu, T.; Kincaid, R.L.; Higuchi, S. Differential subcellular localization of neural isoforms of the catalytic subunit of calmodulin-dependent protein phosphatase (calcineurin) in central nervous system neurons: Immunohistochemistry on formalin-fixed paraffin sections employing antigen retri. J. Histochem. Cytochem. 1996, 44, 13–18. [Google Scholar] [CrossRef]
- Bueno, O.F.; Brandt, E.B.; Rothenberg, M.E.; Molkentin, J.D. Defective T cell development and function in calcineurin Aβ-deficient mice. Proc. Natl. Acad. Sci. USA 2002, 99, 9398–9403. [Google Scholar] [CrossRef] [Green Version]
- Gooch, J.L.; Toro, J.J.; Guler, R.L.; Barnes, J.L. Calcineurin A-α but not A-β is required for normal kidney development and function. Am. J. Pathol. 2004, 165, 1755–1765. [Google Scholar] [CrossRef]
- Singh, S.; More, K.R.; Chitnis, C.E. Role of calcineurin and actin dynamics in regulated secretion of microneme proteins in Plasmodium falciparum merozoites during erythrocyte invasion. Cell. Microbiol. 2014, 16, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, C.; Sarkar, D.; Bhaduri, A. Ca2+ and calmodulin-dependent protein phosphatase from Leishmania donovani. Parasitology 1999, 118, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Leitherer, S.; Clos, J.; Liebler-Tenorio, E.M.; Schleicher, U.; Bogdan, C.; Soulat, D. Characterization of the protein tyrosine phosphatase LmPRL-1 secreted by Leishmania major via the exosome pathway. Infect. Immun. 2017, 85, e00084-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, A.C.S.; Soares, D.C.; Saraiva, E.M.; Meyer-Fernandes, J.R.; Souto-Padrón, T. Different secreted phosphatase activities in Leishmania amazonensis. FEMS Microbiol. Lett. 2013, 340, 117–128. [Google Scholar] [CrossRef] [Green Version]
- Soulat, D.; Bogdan, C. Function of macrophage and parasite phosphatases in leishmaniasis. Front. Immunol. 2017, 8, 1838. [Google Scholar] [CrossRef]
- Kolukisaoglu, Ü.; Weinl, S.; Blazevic, D.; Batistic, O.; Kudla, J. Calcium sensors and their interacting protein kinases: Genomics of the Arabidopsis and rice CBL-CIPK signaling networks. Plant Physiol. 2004, 134, 43–58. [Google Scholar] [CrossRef] [Green Version]
- Silverman, J.M.; Clos, J.; De’Oliveira, C.C.; Shirvani, O.; Fang, Y.; Wang, C.; Foster, L.J.; Reiner, N.E. An exosome-based secretion pathway is responsible for protein export from Leishmania and communication with macrophages. J. Cell Sci. 2010, 123, 842–852. [Google Scholar] [CrossRef] [Green Version]
- Atayde, V.; Aslan, H.; Townsend, S.; Hassani, K.; Kamhawi, S.; Olivier, M. Exosome secretion by the parasitic protozoan Leishmania within the sand fly midgut. Cell Rep. 2015, 13, 957–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bayer-Santos, E.; Aguilar-Bonavides, C.; Rodrigues, S.P.; Cordero, E.M.; Marques, A.F.; Varela-Ramirez, A.; Choi, H.; Yoshida, N.; Da Silveira, J.F.; Almeida, I.C. Proteomic analysis of Trypanosoma cruzi secretome: Characterization of two populations of extracellular vesicles and soluble proteins. J. Proteome Res. 2013, 12, 883–897. [Google Scholar] [CrossRef] [PubMed]
- Sauter, I.P.; Madrid, K.G.; de Assis, J.B.; Sá-Nunes, A.; Torrecilhas, A.C.; Staquicini, D.I.; Pasqualini, R.; Arap, W.; Cortez, M. TLR9/MyD88/TRIF signaling activates host immune inhibitory CD200 in Leishmania infection. JCI Insight 2019, 4, e126207. [Google Scholar] [CrossRef]
- Rossi, I.V.; Ferreira Nunes, M.A.; Vargas-Otalora, S.; da Silva Ferreira, T.C.; Cortez, M.; Ramirez, M.I. Extracellular vesicles during TriTryps infection: Complexity and future challenges. Mol. Immunol. 2021, 132, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Neves, R.F.C.; Fernandes, A.C.S.; Meyer-Fernandes, J.R.; Souto-Padrón, T. Trypanosoma cruzi-secreted vesicles have acid and alkaline phosphatase activities capable of increasing parasite adhesion and infection. Parasitol. Res. 2014, 113, 2961–2972. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Toloza, G.; Aguilar-Guzmán, L.; Valck, C.; Abello, P.; Ferreira, A. Is it all that bad when living with an intracellular protozoan? the role of Trypanosoma cruzi calreticulin in angiogenesis and tumor growth. Front. Oncol. 2015, 4, 382. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Bunn, P.T.; Singh, S.S.; Ng, S.S.; Montes de Oca, M.; De Labastida Rivera, F.; Chauhan, S.B.; Singh, N.; Faleiro, R.J.; Edwards, C.L.; et al. Type I Interferons Suppress Anti-parasitic Immunity and Can Be Targeted to Improve Treatment of Visceral Leishmaniasis. Cell Rep. 2020, 30, 2512–2525. [Google Scholar] [CrossRef] [Green Version]
- Rashidi, S.; Fernández-Rubio, C.; Manzano-Román, R.; Mansouri, R.; Shafiei, R.; Ali-Hassanzadeh, M.; Barazesh, A.; Karimazar, M.; Hatam, G.; Nguewa, P. Potential therapeutic targets shared between leishmaniasis and cancer. Parasitology 2021, 148, 655–671. [Google Scholar] [CrossRef] [PubMed]
- Cortez, M.; Huynh, C.; Fernandes, M.C.; Kennedy, K.A.; Aderem, A.; Andrews, N.W. Leishmania promotes its own virulence by inducing expression of the host immune inhibitory ligand CD200. Cell Host Microbe 2011, 9, 463–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyenne, V.; Ghoroghi, S.; Collot, M.; Bons, J.; Follain, G.; Harlepp, S.; Mary, B.; Bauer, J.; Mercier, L.; Busnelli, I.; et al. Studying the Fate of Tumor Extracellular Vesicles at High Spatiotemporal Resolution Using the Zebrafish Embryo. Dev. Cell 2019, 48, 554–572. [Google Scholar] [CrossRef] [Green Version]
- Caner, A.; Sadıqova, A.; Erdoğan, A.; Namlıses, D.; Nalbantsoy, A.; Oltulu, F.; Toz, S.; Yiğittürk, G.; Ozkök, E.; Gunduz, C.; et al. Targeting of antitumor ımmune responses with live-attenuated Leishmania strains in breast cancer model. Breast Cancer 2020, 27, 1082–1095. [Google Scholar] [CrossRef]
- Zhao, H.; Achreja, A.; Iessi, E.; Logozzi, M.; Mizzoni, D.; Di Raimo, R.; Nagrath, D.; Fais, S. The key role of extracellular vesicles in the metastatic process. Biochim. Biophys. Biochim. Biophys. Acta (BBA) Rev. Cancer 2018, 1869, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; DeWitt, J.P. The multiple mechanism of anti-tumor function of calcineurin B subunit. Jacobs J. Mol. Transl. Med. 2015, 1, 1–3. [Google Scholar]
- Liu, L.; Su, Z.; Xin, S.; Cheng, J.; Li, J.; Xu, L.; Wei, Q. The calcineurin B subunit (CnB) is a new ligand of integrin αM that mediates CnB-induced Apo2L/TRAIL expression in macrophages. J. Immunol. 2012, 188, 238–247. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Qin, N.; Zhang, H.; Yang, R.; Xiang, B.; Wei, Q. Cellular uptake of exogenous calcineurin B is dependent on TLR4/MD2/CD14 complexes, and CnB is an endogenous ligand of TLR4. Sci. Rep. 2016, 6, 24346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, Y.; Huang, Y.; Tian, S.; Xie, X.; Xing, G.; Fu, J. Genetically engineered drug rhCNB induces apoptosis and cell cycle arrest in both gastric cancer cells and hepatoma cells. Drug Des. Dev. Ther. 2018, 12, 2567–2575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, J.; Gao, Y.; Zhu, Z.; Qin, N.; Wei, Q. Identification of a targeting-delivery peptide based on rhCNB. J. Pept. Sci. 2019, 25, e3159. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.J.B.; Vega, M.C.; González-Rey, E.; Fernández-Carazo, R.; Macedo-Ribeiro, S.; Gomis-Rüth, F.X.; González, A.; Coll, M. Trypanosoma cruzi macrophage infectivity potentiator has a rotamase core and a highly exposed α-helix. EMBO Rep. 2002, 3, 88–94. [Google Scholar] [CrossRef] [Green Version]
- Moro, A.; Ruiz-Cabello, F.; Fernández-Cano, A.; Stock, R.P.; Gonzalez, A. Secretion by Trypanosoma cruzi of a peptidyl-prolyl cis-trans isomerase involved in cell infection. EMBO J. 1995, 14, 2483–2490. [Google Scholar] [CrossRef]
- Kulkarni, M.M.; Karafova, A.; Kamysz, W.; Schenkman, S.; Pelle, R.; McGwire, B.S. Secreted trypanosome cyclophilin inactivates lytic insect defense peptides and induces parasite calcineurin activation and infectivity. J. Biol. Chem. 2013, 288, 8772–8784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Field, M.C.; Carrington, M. The trypanosome flagellar pocket. Nat. Rev. Microbiol. 2009, 7, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Murta, A.C.; Persechini, P.M.; Padron, T.d.S.; de Souza, W.; Guimarães, J.A.; Scharfstein, J. Structural and functional identification of GP57/51 antigen of Trypanosoma cruzi as a cysteine proteinase. Mol. Biochem. Parasitol. 1990, 43, 27–38. [Google Scholar] [CrossRef]
- Baba, Y.; Hirukawa, N.; Tanohira, N.; Sodeoka, M. Structure-based design of a highly selective catalytic site-directed inhibitor of Ser/Thr protein phosphatase 2B (calcineurin). J. Am. Chem. Soc. 2003, 125, 9740–9749. [Google Scholar] [CrossRef]
- Baba, Y.; Hirukawa, N.; Sodeoka, M. Optically active cantharidin analogues possessing selective inhibitory activity on Ser/Thr protein phosphatase 2B (calcineurin): Implications for the binding mode. Bioorg. Med. Chem. 2005, 13, 5164–5170. [Google Scholar] [CrossRef] [PubMed]








| Protein | #aa | MW (Da) | pI |
|---|---|---|---|
| HsCaNAα | 521 | 58,687.85 | 5.58 |
| TcCaNA1 | 382 | 43,244.64 | 4.83 |
| TcCaNA2 | 392 | 44,619.50 | 8.13 |
| LsppCaNA1 | 545 | 59,747.65 | 5.00 |
| LsppCaNA1_var | 503 | 55,559.36 | 5.61 |
| LsppCaNA2 | 407 | 45,138.00 | 6.37 |
| TcCaNB | 176 | 19,458.18 | 4.98 |
| LsppCaNB | 176 | 19,785.54 | 4.60 |
| HsCaNAα | #x | Context | Score | Kinase | Answer |
|---|---|---|---|---|---|
| # Sequence | 21 S | ARVFSVLRE | 0.974 | unsp | YES |
| # Sequence | 27 S | LREES---- | 0.556 | PKA | YES |
| LsppCaNA1 and LsppCaNA1var | #x | Context | Score | Kinase | Answer |
| # Sequence | 17 S | MGRLSRMFH | 0.956 | unsp | YES |
| # Sequence | 17 S | MGRLSRMFH | 0.725 | PKA | YES |
| # Sequence | 17 S | MGRLSRMFH | 0.556 | PKC | YES |
| # Sequence | 22 T | RMFHTLCEG | 0.766 | Unsp | YES |
| # Sequence | 22 T | RMFHTLCEG | 0.517 | CKII | YES |
| A. WoLF PSORT of Catalytic Subunits of CaN | ||||||
|---|---|---|---|---|---|---|
| Site | HsCaNAα | TcCaNA1 | TcCaNA2 | LsppCaNA1 | LsppCaNA1_var | LsppCaNA2 |
| extr | 3 | |||||
| plas | 1 | |||||
| cyto | 21.5 | 12 | 8.5 | 13.5 | 14.5 | 9.5 |
| cysk | 6 | |||||
| E.R. | 3 | 1 | ||||
| golg | 1 | |||||
| mito | 6 | 3 | 1 | 2 | 9 | |
| pero | 3 | 2 | 5 | 3 | 1 | 2 |
| vacu | 1 | |||||
| nucl | 4 | 4 | 9.5 | 3 | 9.5 | 3.5 |
| cyto_nucl | 13.5 | 9 | 10.5 | 10 | 13.5 | 7 |
| B. WoLF PSORT of Regulatory Subunits of CaN | ||||||
| Site | HsCaNB-1 | TcCaNB | LsppCaNB | |||
| extr | 1 | 1 | ||||
| plas | 2 | |||||
| cyto | 25 | 5.5 | 9 | |||
| cysk | 1 | 6 | ||||
| mito | 1 | |||||
| pero | 3 | 3 | 2 | |||
| nucl | 2 | 9.5 | 15 | |||
| Cyto_nucl | 14.5 | 8 | 14 | |||
| Name | NN-Score 1 | Odds | Weighted by Prior | Warning |
|---|---|---|---|---|
| HsCaNAα | 0.441 | 0.861 | 0.002 | - |
| HsCaNB-1 | 0.548 | 1.252 | 0.003 | - |
| TcCaNA1 | 0.682 | 1.863 | 0.004 | - |
| TcCaNA2 | 0.442 | 0.858 | 0.002 | - |
| TcCaNB | 0.637 | 1.758 | 0.004 | - |
| LsppCaNA1 | 0.528 | 1.094 | 0.002 | - |
| LsppCaNA1_var | 0.477 | 0.926 | 0.002 | - |
| LsppCaNA2 | 0.718 | 2.165 | 0.004 | - |
| LsppCaNB | 0.382 | 0.709 | 0.001 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orrego, P.R.; Serrano-Rodríguez, M.; Cortez, M.; Araya, J.E. In Silico Characterization of Calcineurin from Pathogenic Obligate Intracellular Trypanosomatids: Potential New Biological Roles. Biomolecules 2021, 11, 1322. https://doi.org/10.3390/biom11091322
Orrego PR, Serrano-Rodríguez M, Cortez M, Araya JE. In Silico Characterization of Calcineurin from Pathogenic Obligate Intracellular Trypanosomatids: Potential New Biological Roles. Biomolecules. 2021; 11(9):1322. https://doi.org/10.3390/biom11091322
Chicago/Turabian StyleOrrego, Patricio R., Mayela Serrano-Rodríguez, Mauro Cortez, and Jorge E. Araya. 2021. "In Silico Characterization of Calcineurin from Pathogenic Obligate Intracellular Trypanosomatids: Potential New Biological Roles" Biomolecules 11, no. 9: 1322. https://doi.org/10.3390/biom11091322






