1. Introduction
Membrane lipid composition (MLC), especially anionic lipids, can affect lipid bilayer and membrane protein dynamics, structure and function [
1,
2,
3,
4]. The MLC has been shown to affect small, drug-like molecules partitioning and translocation through a membrane [
5]. Moreover, MLC differences have also been suggested to affect receptor entry processes of drugs and neurotransmitters [
6,
7,
8]. Accordingly, it is vital to determine the biologically relevant MLCs and then utilize these lipidomics data to reveal the exact roles of the specific lipid components and contribution to properties of a membrane. Such studies have been performed for, e.g., exosomes [
9,
10], yeast endoplasmic reticulum, plasma membrane, and trans-Golgi network [
11], and adipose tissue [
12].
Prior experimental studies have indicated that specific lipid composition changes of the mitochondrial outer membrane (OMM) can affect the function of membrane proteins such as monoamine oxidase B (MAO-B) [
13,
14]. MAO-B is a monotopic membrane protein that degrades, e.g., benzylamine and phenylamine, but, most notably, monoamine neurotransmitters such as dopamine [
15]. Thus, the OMM composition directly or indirectly affects the regulation of the synaptic dopamine level and, thus, is crucial for treating neurological disorders such as Parkinson’s disease and depression [
16]. Despite the apparent importance of OMM for a multitude of cellular functions such as apoptosis [
17], inflammation [
18,
19], and monoamine degradation [
20], the tendency has been to oversimplify the OMM composition in prior in silico studies, e.g., [
21,
22,
23]. It is prudent to represent or inspect the OMM with the most accurate and current lipid compositions thus updating the situation regarding the roles of different lipid species and ensuring that future studies on membrane proteins involving the OMM are performed with the most accurate MLC possible. Notably, a major anionic component, phosphatidylinositol (PI; 13% of OMM mass; [
24,
25]), has been excluded from prior simulation studies concentrating on the OMM [
21,
22,
23]. Because the necessary parameters are now available for the CHARMM36 force field [
26], a representative of these anionic lipids can now be included in the studied model systems.
This in silico study aimed to explore the effect of a complex MLC for the OMM using atomistic molecular dynamics (MD) simulations (Models #1–5;
Table 1). Firstly, we wanted to examine how the simple two-component model (Model #5;
Table 1) performs in comparison to a model housing all the major lipids of OMM in correct ratios (Model #2;
Table 1). Secondly, we also wanted to see how the varying of PS concentration affected the membrane structure and dynamics (Models #1–4). The increased levels of this anionic lipid species have been linked to schizophrenia [
14] and, more specifically, to the inactivation of the MAO-B enzyme [
13]. Thirdly, the idea was to set the foundation for pursuing more simulation work using these membrane models in the future with small-molecules (e.g., dopamine) and membrane proteins residing at the OMM (e.g., MOA-B). The membrane environment affects neurotransmitter dynamics throughout their life cycle, which also includes their removal from the synaptic cleft, intracellular synthesis and eventual degradation (e.g., dopamine + MAO-B) [
7].
The results indicate that the four-component OMM models produce notably different results than the previously applied two-component models containing phosphatidylcholine (PC) and phosphatidylethanolamine (PE) [
27]. There are changes in the bilayer thickness, area per lipid values, lateral diffusion rates, membrane surface charge, and the amount of membrane-ion contacts directly attributed to the lipid composition. The results suggest that the overall effect of the accurate OMM lipid composition on the water-membrane interface should not be ignored in future studies, when for example studying the membrane protein conformation and dynamics.
In short, based on the simulations, we present an OMM model (Model #2;
Table 1) with realistic lipid composition, which is ready for various membrane protein simulations. These equilibrated membrane model coordinates are made freely available (see the
Supplementary File simulated_systems.zip).
4. Discussion
Prior in silico studies focusing on properties of the outer mitochondrial membrane (OMM) have been conducted using relatively simple single- or two-component membrane models [
28,
43,
44]. However, due to ongoing advancements in lipid force field development [
43,
45,
46], it is becoming increasingly possible to study more realistic and complex membrane lipid compositions (MLCs) than ever before by using atomistic molecular dynamics (MD) simulations. Here, the MD simulations were performed using models with 2–4-lipid components (
Table 1;
Figure 1) and, thus, the interplay and combined effect of all major OMM lipid types [
24,
25] could be established (
Figure 2,
Figure 3,
Figure 4 and
Figure 5;
Table 2,
Table 3,
Table 4 and
Table 5).
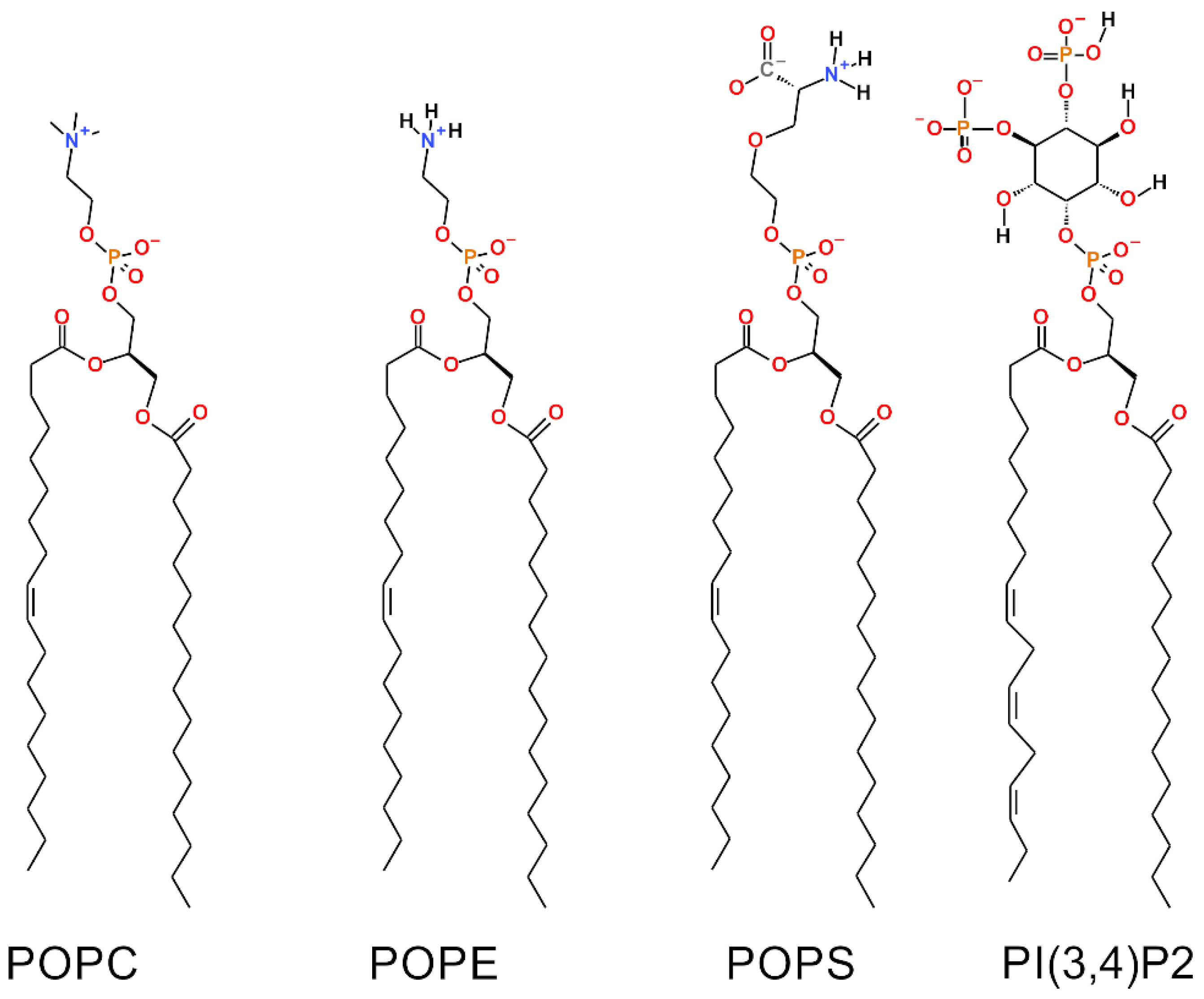
Notably, the effects of two charged lipid types, PI(3,4)P2 (1-palmitoyl-2-linolenoyl-inositol-(4,5)-bisphosphate (PIP2) with protonation on P4;
Figure 1) and POPS (phosphatidylserine;
Figure 1), were probed by varying their concentration among the five OMM models (Models #1–#5;
Table 1;
Figure 5). Overall, the results show that the structural properties such as membrane thickness, area per lipid, and S
CD order parameter were not primarily affected by the MLC changes within the studied membrane models. These basic metrics were close to the equivalent numbers found for neutral POPC-POPE membrane in the simulations (Model #5 in
Table 1 and
Figure 5) and experiments [
38,
39]. This behavior was to be expected based on prior simulation studies, which similarly have shown that charged lipids at moderate physiological concentrations induce only minor effects on the structural properties of membranes [
23,
44,
47] or even phase behavior [
48]. However, the effect of charged lipids may be modulated by other factors, e.g., phosphatidic acid significantly affects the ordering of the bilayer composed of saturated lipids while its effect on unsaturated lipids is tiny [
49].
The detailed analysis of the trajectories from the MD simulations indicated that the changes in the charged lipid levels of the OMM models cause substantial effects on the dynamical parameters. The results suggest that increasing the POPS levels in the OMM models slows down the lateral diffusion of all lipids forming the bilayer. The second observation is that the headgroup rotation of POPE was slowed down in the OMM models containing charged lipid species without POPS or PI(3,4)P2-specific interactions. The rotation of the POPS headgroup was also slowed down, but only in response to high POPS levels.
The water-membrane interface, where the OMM simulations are likely to be more accurate than on the above-mentioned diffusion metrics, is expected to be affected by any changes to the lipid headgroup [
50,
51,
52,
53]; thus it is not surprising that there are indeed notable differences between studied models. In general, the POPE hydration was reduced when anionic lipids were included (
Table 4). The surface charge density or POPS concentration increase generated a hydration difference of 5% for POPS between Models #2 and #4. A PI(3,4)P2 hydration difference of ~7% was seen between Models #2 and #4. In addition, Na
+ ions neutralize the membrane surface, when PI(3,4)P2 was present in a high concentration; however, a similar effect was not seen for POPS. This effect on surface charge or adherence of cations due to anionic lipid composition changes could also affect the dynamics of other solutes such as proteins or small molecules.
The results described above highlight a persistent problem: an over-simplification of the membrane environment in both in silico and in vitro studies of membrane proteins. This is commonplace even though the importance of a specific lipid environment for the membrane protein is recognized. For example, cholesterol, a lipid significantly modulating the structure of the lipid bilayer, is also known to affect a protein’s structure, dynamics, and function [
54,
55,
56]. Likewise, polyunsaturated lipids regulate the behavior of G-protein coupled receptors [
57,
58]. As a single structural parameter, the membrane thickness is recognized as an essential factor affecting membrane proteins in multiple ways via the so-called hydrophobic mismatch mechanism [
59,
60,
61]. Nevertheless, these three examples concern transmembrane proteins and structural properties of lipid bilayers, while the relation between properties of the water-membrane interface and peripheral, monotopic or bitopic membrane proteins remains elusive. A few studies have showed that changes in the behavior of the lipid headgroup, due to the membrane composition or the presence of divalent cations, might prevent the association of peripheral membrane proteins with the membrane [
62,
63].
In the context of OMM, the increased POPS levels have been linked to monoamine oxidase B (MOA-B) inactivation [
13] and schizophrenia [
14] decades ago. Likewise, the MAO-B activity has been linked to progressive brain diseases such as Alzheimer’s disease and Parkinson’s disease [
16,
64]. Accordingly, the increase of the POPS concentration at the OMM is potentially causing indirect or direct effects on the membrane protein’s structure and function. Firstly, the POPS-rich membrane could restrict the diffusion of monoamines (e.g., dopamine) by binding them more firmly onto the membrane surface. The anionic lipids are known to increase membrane adherence of amphipathic non-peptidic neurotransmitters in particular monoamines [
6], e.g., dopamine, which likely accesses or enters its post-synaptic receptor via membrane-mediated pathway [
65]. Secondly, it is also possible that increased POPS levels affect the MOA-B structure and dynamics adversely at the OMM, i.e., the monoamine entry via the membrane surface is hindered, or the subsequent catalysis is prevented due to a conformational shift. The opening of channels leading towards the catalytic center of MOA-A and -B occurs at the membrane surface [
27,
66,
67], and loops controlling entrance to the channel are affected by the interactions with the nearby lipids [
27]. Moreover, Cytochrome P, a protein of similar topology to MOA (single transmembrane helix and globular extra membrane catalytic domain), has been shown to be affected by lipid composition changes such as the addition of cholesterol [
68] or charged lipids [
69].