Immunomodulatory Potential of Non-Classical HLA-G in Infections including COVID-19 and Parasitic Diseases
Abstract
:1. Introduction
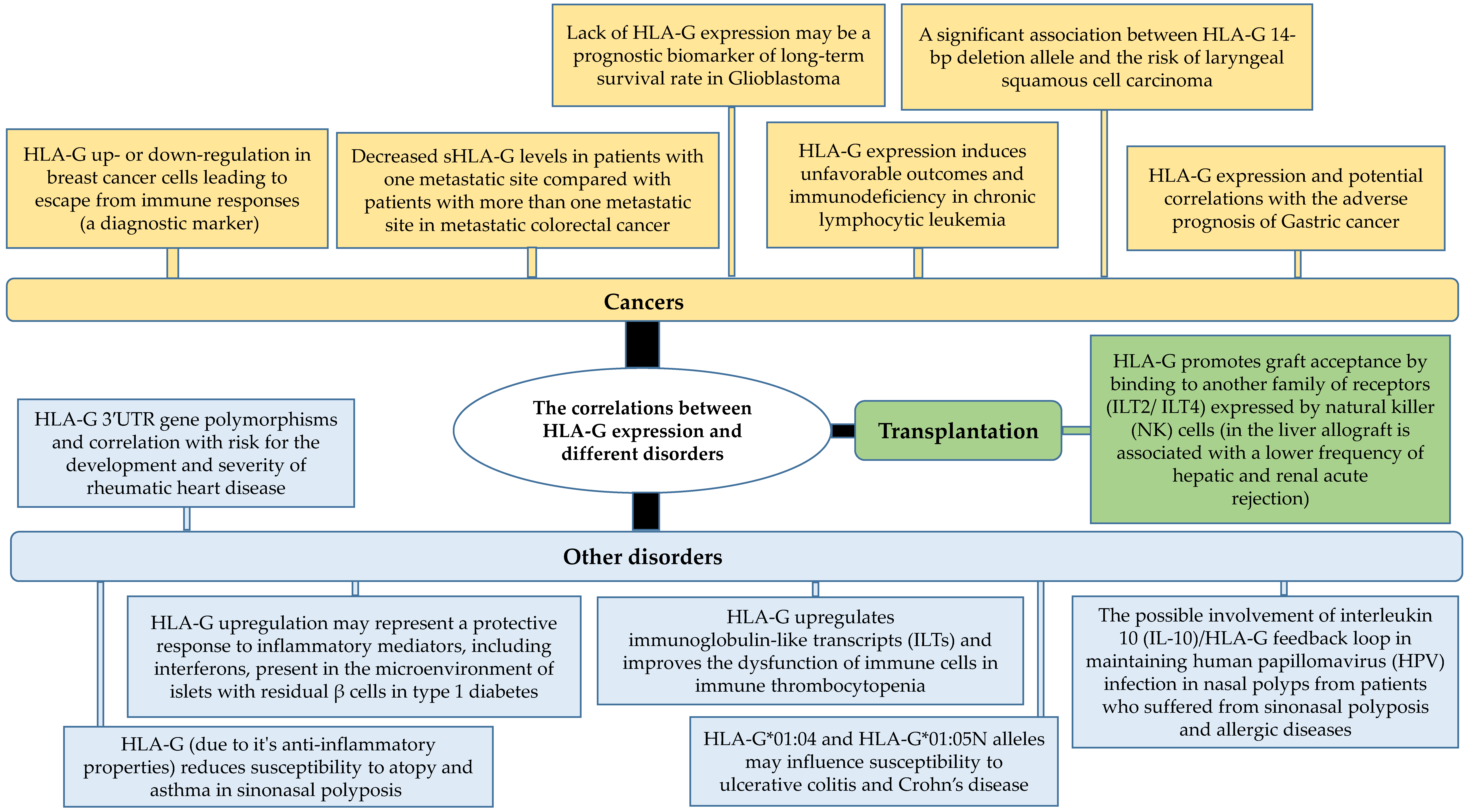
2. Functions of HLA-G in Diseases
| Pathogens or Diseases | HLA-G | Observations | References |
|---|---|---|---|
| Hepatitis B virus (HBV) | HLA-G (14 bp ins/ins genotype) | Positive correlation with worse clinical manifestations | [63] |
| sHLA-G | A significant correlation with the phase of HBV infection, clinical diagnosis, and disease persistence, and also progression toward hepatocellular carcinoma | [64] | |
| Hepatitis C virus (HCV) | Higher levels of sHLA-G and interleukin-10 (IL-10) | Negative correlation with response to treatment | [65] |
| HLA-G | A plausible function in the genesis of HCV liver fibrosis | [66] | |
| Human immunodeficiency virus 1 (HIV-1) | HLA-G polymorphisms | HLA-G polymorphisms independently and synergistically induce susceptibility to heterosexual acquisition of HIV-1 | [67] |
| Human Papillomavirus (HPV) | HLA-G polymorphism | Associations with the outcomes of oral HPV, affecting some characteristics of the women’s reproductive health, dual function (tumor progression and a good immunotherapeutic target) in cervical HPV | [68,69] |
| Herpesvirus (6A and 6B) | HLA-G | Inhibits in vitro angiogenesis through HLA-G | [70] |
| Cytomegalovirus (CMV) | HLA-G | Influences HLA-G expression in healthy individuals and probably contribute to viral immune evasion | [71] |
| sHLA-G | As a promising biomarker of diagnosis of maternal CMV in maternal blood and amniotic fluid | [72] | |
| Arbovirus | sHLA-G | A plausible biomarker to monitor neurological complications | [73] |
| SARS-CoV-2 | HLA- G | The possible induction of profound immune suppression leading to the escape of virus from immune attack | [10] |
| Helicobacter pylori | HLA-G | Correlated with milder colonization and milder inflammation | [74] |
| Pseudomonas aeruginosa | sHLA-G | Decreasing levels during antibiotic therapy in patients with cystic fibrosis (negative correlation with inflammation) | [75] |
| HLA-G | Inducing HLA-G expression in monocytes and T cells by P. aeruginosa (protecting from immune responses) | [76] | |
| Tropheryma whipplei | HLA-G | Increased HLA-G levels in patients’ sera (promoting bacteria colonization) | [23] |
| Human African trypanosomiasis (HAT) | HLA-G 3′ UTR-2 and UTR-5 haplotypes | Association with increased susceptibility to HAT | [77] |
| HLA-G 3′ UTR-4 haplotype | Association with a decreased risk of HAT | ||
| HLA-G 5′ URR-010102a/UTR-2 and 5′URR- 0103e/CR-G*01:03:01:02/UTR-5 haplotypes | Association with HAT disease progression | [78] | |
| HLA-G (rs1611139 T, rs17875389 A, rs9380142 G alleles) | Association with increased risk of infection | [79,80] | |
| HLA-G (rs1233330 A, rs1233330 G alleles) | Association with decreased risk of infection | [80,81] | |
| American trypanosomiasis (Chagas disease) | Cell-surface HLA-G | Reduced HLA-G expression on cardiac muscle and colonic cells in patients with cardiac or digestive forms of Chagas, respectively | [82] |
| HLA-G (+3003 T allele and +3003TT, +3187GG and +3196GC genotypes) | Association with an enhanced risk of symptomatic Chagas | ||
| HLA-G (+3003C allele and +3003CT and +3196CC genotypes) | Association with a decreased risk of symptomatic Chagas | ||
| HLA-G (+3027C and +3035C alleles and of +3027CC and +3035CC genotypes) | Association with the digestive form of Chagas | ||
| Malaria | sHLA-G | Increased sHLA-G levels in cord blood and correlation with low weight at birth and clinical outcome (positive correlation with high risk of infection in infancy) | [83] |
| HLA-G (+3187G allele and UTR1 haplotype) | Association with reduced level of parasite burden during Plasmodium falciparum asymptomatic infection | [84] | |
| HLA-G (UTR3 haplotype) | Association with enhanced level of parasite burden and increased severity of P. falciparum asymptomatic infection | ||
| HLA-G (+3010G and +3142C alleles) | Association with enhanced total IgG and IgG1 antibodies levels against P. falciparum glutamate-rich protein (GLURP) | [85] | |
| HLA-G (+3196G and UTR2 haplotype) | Association with a decreased IgG3 response against P. falciparum Merozoite Surface Protein 2 (MSP2) | ||
| sHLA-G | The correlation of increased sHLA-G levels in cord blood with low birth weight and an enhanced risk of malaria (P. falciparum) in the first year of life | [83,86] | |
| sHLA-G | The correlation of an increased mean sHLA-G levels during infancy with low birth weight and an enhanced risk of malaria (P. falciparum) during the two first years of life | [87] | |
| Visceral leishmaniasis (VL) | sHLA-G | Increased levels of blood sHLA-G in Leishmania-infected patients | [88] |
| Decreased levels of blood sHLA-G after anti-parasitic treatment of VL | [89] | ||
| Toxoplasma spp. | sHLA-G | Parasite increased the secretion of sHLA-G by trophoblast inducing apoptosis of decidual natural killer (dNK) cells (positive correlation with abnormal pregnancy) | [90] |
| Increased sHLA-G levels in the amniotic fluid of pregnant women infected by Toxoplasma and in congenitally infected fetuses | [91] | ||
| The increased secretion of sHLA-G by trophoblast due to Toxoplasma infection (inducing apoptosis of dNK cells) | [90] | ||
| Cell-surface HLA-G | Increased HLA-G expression in the Toxoplasma-infected cells (decreasing HLA-G after treatment of infected cell by IL-10) | [92] |
3. HLA-G Expression in Parasitic Diseases
4. HLA-G as Target for Potential Immunotherapeutic Strategy
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wyatt, R.C.; Lanzoni, G.; Russell, M.A.; Gerling, I.; Richardson, S.J. What the HLA-I!-Classical and Non-classical HLA Class I and Their Potential Roles in Type 1 Diabetes. Curr. Diabetes Rep. 2019, 19, 159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hauer, V.; Risti, M.; Miranda, B.L.; da Silva, J.S.; Cidral, A.L.; Pozzi, C.M.; de C Contieri, F.L.; Sadissou, I.A.; Donadi, E.A.; Augusto, D.G.; et al. The association of HLA-G polymorphisms and the synergistic effect of sMICA and sHLA-G with chronic kidney disease and allograft acceptance. PLoS ONE 2019, 14, e0212750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arns, T.; Antunes, D.A.; Abella, J.R.; Rigo, M.M.; Kavraki, L.E.; Giuliatti, S.; Donadi, E.A. Structural Modeling and Molecular Dynamics of the Immune Checkpoint Molecule HLA-G. Front. Immunol. 2020, 11, 575076. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.; Barker, D.J.; Georgiou, X.; Cooper, M.A.; Flicek, P.; Marsh, S.G. Ipd-imgt/hla database. Nucleic Acids. Res. 2020, 48, D948–D955. [Google Scholar] [PubMed]
- Curigliano, G.; Criscitiello, C.; Gelao, L.; Goldhirsch, A. Molecular pathways: Human leukocyte antigen G (HLA-G). Clin. Cancer. Res. 2013, 19, 5564–5571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- HoWangYin, K.-Y.; Loustau, M.; Wu, J.; Alegre, E.; Daouya, M.; Caumartin, J.; Sousa, S.; Horuzsko, A.; Carosella, E.D.; LeMaoult, J. Multimeric structures of HLA-G isoforms function through differential binding to LILRB receptors. Cell. Mol. Life Sci. 2012, 69, 4041–4049. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuroki, K.; Matsubara, H.; Kanda, R.; Miyashita, N.; Shiroishi, M.; Fukunaga, Y.; Kamishikiryo, J.; Fukunaga, A.; Fukuhara, H.; Hirose, K.; et al. Structural and functional basis for LILRB immune checkpoint receptor recognition of HLA-G isoforms. J. Immunol. 2019, 203, 3386–3394. [Google Scholar] [CrossRef]
- Bu, X.; Zhong, J.; Li, W.; Cai, S.; Gao, Y.; Ping, B. Immunomodulating functions of human leukocyte antigen-G and its role in graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Ann. Hematol. 2021, 100, 1391–1400. [Google Scholar] [CrossRef]
- Zaborek-Łyczba, M.; Łyczba, J.; Mertowska, P.; Mertowski, S.; Hymos, A.; Podgajna, M.; Niedźwiedzka-Rystwej, P.; Grywalska, E. The HLA-G Immune Checkpoint Plays a Pivotal Role in the Regulation of Immune Response in Autoimmune Diseases. Int. J. Mol. Sci. 2021, 22, 13348. [Google Scholar] [CrossRef]
- Lin, A.; Yan, W.-H. Perspective of HLA-G Induced Immunosuppression in SARS-CoV-2 Infection. Front. Immunol. 2021, 12, 788769. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, S.; Han, Y.; Wang, Y.; Sun, Y. Human leukocyte antigen-G expression and polymorphisms promote cancer development and guide cancer diagnosis/treatment. Oncol. Lett. 2018, 15, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Contini, P.; Murdaca, G.; Puppo, F.; Negrini, S. HLA-G Expressing Immune Cells in Immune Mediated Diseases. Front. Immunol. 2020, 11, 1613. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.K.; Brossay, L. The role of MHC class Ib-restricted T cells during infection. Immunogenetics 2016, 68, 677–691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Attia, J.V.; Dessens, C.E.; van de Water, R.; Houvast, R.D.; Kuppen, P.J.; Krijgsman, D. The Molecular and Functional Characteristics of HLA-G and the Interaction with Its Receptors: Where to Intervene for Cancer Immunotherapy? Int. J. Mol. Sci. 2020, 21, 8678. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Yan, W.-H. Human leukocyte antigen-G (HLA-G) expression in cancers: Roles in immune evasion, metastasis and target for therapy. Mol. Med. 2015, 21, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Morandi, F.; Rizzo, R.; Fainardi, E.; Rouas-Freiss, N.; Pistoia, V. Recent advances in our understanding of HLA-G biology: Lessons from a wide spectrum of human diseases. J. Immunol. Res. 2016, 2016, 4326495. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, I.L.; Montero-Montero, L.; Ferreira, E.; Quintanilla, M. New insights into the role of Qa-2 and HLA-G non-classical MHC-I complexes in malignancy. Front. Immunol. 2018, 9, 2894. [Google Scholar] [CrossRef]
- Rizzo, R.; Bortolotti, D.; Bolzani, S.; Fainardi, E. HLA-G molecules in autoimmune diseases and infections. Front. Immunol. 2014, 5, 592. [Google Scholar] [CrossRef] [Green Version]
- Créput, C.; Durrbach, A.; Menier, C.; Guettier, C.; Samuel, D.; Dausset, J.; Charpentier, B.; Carosella, E.D.; Rouas-Freiss, N. Human leukocyte antigen-G (HLA-G) expression in biliary epithelial cells is associated with allograft acceptance in liver-kidney transplantation. J. Hepatol. 2003, 39, 587–594. [Google Scholar] [CrossRef]
- Amodio, G.; Gregori, S. The discovery of HLA-G-bearing extracellular vesicles: New perspectives in HLA-G biology. Ann. Transl. Med. 2017, 5, 148. [Google Scholar] [CrossRef] [Green Version]
- Wan, R.; Wang, Z.-W.; Li, H.; Peng, X.-D.; Liu, G.-Y.; Ou, J.-M.; Cheng, A.-Q. Human leukocyte antigen-G inhibits the an-ti-tumor effect of natural killer cells via immunoglobulin-like transcript 2 in gastric cancer. Cell. Physiol. Biochem. 2017, 44, 1828–1841. [Google Scholar] [CrossRef] [PubMed]
- Hölzemer, A.; Garcia-Beltran, W.F.; Altfeld, M. Natural killer cell interactions with classical and non-classical human leukocyte antigen class I in HIV-1 infection. Front. Immunol. 2017, 8, 1496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azzouz, E.B.; Boumaza, A.; Mezouar, S.; Bardou, M.; Carlini, F.; Picard, C.; Raoult, D.; Mège, J.-L.; Desnues, B. Tropheryma whipplei increases expression of human Leukocyte Antigen-G on monocytes to reduce tumor necrosis factor and promote bacterial replication. Gastroenterology 2018, 155, 1553–1563. [Google Scholar] [CrossRef] [PubMed]
- Wastowski, I.J.; Simões, R.T.; Yaghi, L.; Donadi, E.A.; Pancoto, J.T.; Poras, I.; Lechapt-Zalcman, E.; Bernaudin, M.; Valable, S.; Carlotti, C.G., Jr.; et al. Human leukocyte antigen-G is frequently expressed in glioblastoma and may be induced in vitro by combined 5-aza-2′-deoxycytidine and interferon-γ treatments: Results from a multicentric study. Am. J. Pathol. 2013, 182, 540–552. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Toth, I.; Schulze zur Wiesch, J.; Pereyra, F.; Rychert, J.; Rosenberg, E.S.; van Lunzen, J.; Lichterfeld, M.; Yu, X.G. Func-tional characterization of HLA-G+ regulatory T cells in HIV-1 infection. PLoS Pathog. 2013, 9, e1003140. [Google Scholar] [CrossRef]
- Adolf, I.C.; Almars, A.; Dharsee, N.; Mselle, T.; Akan, G.; Nguma, I.J.; Nateri, A.S.; Atalar, F. HLA-G and single nucleotide polymorphism (SNP) associations with cancer in African populations: Implications in personal medicine. Genes Dis. 2021. [Google Scholar] [CrossRef]
- Arnaiz-Villena, A.; Juarez, I.; Suarez-Trujillo, F.; López-Nares, A.; Vaquero, C.; Palacio-Gruber, J.; Martin-Villa, J.M. HLA-G: Function, polymorphisms and pathology. Int. J. Immunogenet. 2021, 48, 172–192. [Google Scholar] [CrossRef]
- de Kruijf, E.M.; Sajet, A.; van Nes, J.G.; Natanov, R.; Putter, H.; Smit, V.T.; Liefers, G.J.; van den Elsen, P.J.; van de Velde, C.J.; Kuppen, P.J.K. HLA-E and HLA-G expression in classical HLA class I-negative tumors is of prognostic value for clinical outcome of early breast cancer patients. J. Immunol. 2010, 185, 7452–7459. [Google Scholar] [CrossRef] [Green Version]
- Nückel, H.; Rebmann, V.; Dürig, J.; Dührsen, U.; Grosse-Wilde, H. HLA-G expression is associated with an unfavorable out-come and immunodeficiency in chronic lymphocytic leukemia. Blood 2005, 105, 1694–1698. [Google Scholar]
- Li, X.; Sheng, Z.; Sun, Y.; Wang, Y.; Xu, M.; Zhang, Z.; Li, H.; Shao, L.; Zhang, Y.; Yu, J.; et al. Human leukocyte antigen-G upregulates immunoglobulin-like transcripts and corrects dysfunction of immune cells in immune thrombocytopenia. Haematologica 2021, 106, 770–781. [Google Scholar]
- Scarabel, L.; Garziera, M.; Fortuna, S.; Asaro, F.; Toffoli, G.; Geremia, S. Soluble HLA-G expression levels and HLA-G/irinotecan association in metastatic colorectal cancer treated with irinotecan-based strategy. Sci. Rep. 2020, 10, 8773. [Google Scholar] [CrossRef] [PubMed]
- Jucaud, V.; Ravindranath, M.; Terasaki, P. Immunobiology of HLA class-Ib molecules in transplantation. SOJ Immunol. 2015, 3, 1–15. [Google Scholar] [CrossRef]
- Avelino, M.A.G.; Wastowski, I.J.; Ferri, R.G.; Elias, T.G.A.; Lima, A.P.L.; Marinho, L.C.; Pignatari, S.S.N. The human leukocyte antigen G molecule (HLA-G) expression in patients with nasal polyposis. Braz. J. Otorhinolaryngol. 2014, 80, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Hussein, S.S.; Ali, E.N.; Zaki, N.H.; Ad’hiah, A.H. Genetic polymorphism of HLA-G gene (G*01:03, G*01:04, and G*01:05N) in Iraqi patients with inflammatory bowel disease (ulcerative colitis and Crohn’s disease). Eur. J. Hum. Genet. 2021, 22, 34. [Google Scholar] [CrossRef]
- Poomarimuthu, M.; Elango, S.; Soundrapandian, S.; Mariakuttikan, J. HLA-G 3′UTR gene polymorphisms and rheumatic heart disease: A familial study among South Indian population. Pediatr. Rheumatol. 2017, 15, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, R.; Malagutti, N.; Bortolotti, D.; Gentili, V.; Rotola, A.; Fainardi, E.; Pezzolo, T.; Aimoni, C.; Pelucchi, S.; Di Luca, D.; et al. Infection and HLA-G molecules in nasal polyposis. J. Immunol. Res. 2014, 2014, 407430. [Google Scholar] [CrossRef] [PubMed]
- Barakat, G.; Elsharkawy, A.; Nabiel, Y. Human leucocyte antigen-G gene polymorphism in laryngeal squamous cell carcinoma patients in Mansoura University Hospitals. Egypt. J. Basic Appl. Sci. 2021, 8, 214–221. [Google Scholar] [CrossRef]
- Amiot, L.; Vu, N.; Samson, M. Immunomodulatory properties of HLA-G in infectious diseases. J. Immunol. Res. 2014, 2014, 298569. [Google Scholar] [CrossRef]
- Sabbagh, A.; Sonon, P.; Sadissou, I.; Mendes-Junior, C.; Garcia, A.; Donadi, E.; Courtin, D. The role of HLA-G in parasitic diseases. HLA 2018, 91, 255–270. [Google Scholar] [CrossRef]
- Donadi, E.A.; Castelli, E.C.; Arnaiz-Villena, A.; Roger, M.; Rey, D.; Moreau, P. Implications of the polymorphism of HLA-G on its function, regulation, evolution and disease association. Cell. Mol. Life Sci. 2011, 68, 369–395. [Google Scholar] [CrossRef] [Green Version]
- Poláková, K.; Kuba, D.; Russ, G. The 4H84 monoclonal antibody detecting β2m free nonclassical HLA-G molecules also binds to free heavy chains of classical HLA class I antigens present on activated lymphocytes. Hum. Immunol. 2004, 65, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Swets, M.; König, M.H.; Zaalberg, A.; Dekker-Ensink, N.G.; Gelderblom, H.; van de Velde, C.J.; van den Elsen, P.J.; Kuppen, P.J. HLA-G and classical HLA class I expression in primary colorectal cancer and associated liver metastases. Hum. Immunol. 2016, 77, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Rebmann, V.; LeMaoult, J.; Rouas-Freiss, N.; Carosella, E.; Grosse-Wilde, H. Quantification and identification of soluble HLA-G isoforms. Tissue Antigens 2007, 1, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, R.; Trentini, A.; Bortolotti, D.; Manfrinato, M.C.; Rotola, A.; Castellazzi, M.; Melchiorri, L.; Di Luca, D.; Dallocchio, F.; Fainardi, E. Matrix metalloproteinase-2 (MMP-2) generates soluble HLA-G1 by cell surface proteolytic shedding. Mol. Cell. Biochem. 2013, 381, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.A. HLA-mediated tumor escape mechanisms that may impair immunotherapy clinical outcomes via T-cell acti-vation. Oncol. Lett. 2017, 14, 4415–4427. [Google Scholar] [CrossRef] [Green Version]
- Monneret, G.; Voirin, N.; Krawice-Radanne, I.; Bohé, J.; Lepape, A.; Rouas-Freiss, N.; Carosella, E.D. Soluble human leukocyte antigen-G5 in septic shock: Marked and persisting elevation as a predictor of survival. Crit. Care Med. 2007, 35, 1942–1947. [Google Scholar] [CrossRef]
- Schwich, E.; Hò, G.-G.T.; LeMaoult, J.; Bade-Döding, C.; Carosella, E.D.; Horn, P.A.; Rebmann, V. Soluble HLA-G and HLA-G bearing extracellular vesicles affect ILT-2 positive and ILT-2 negative CD8 T cells complementary. Front. Immunol. 2020, 11, 2046. [Google Scholar] [CrossRef]
- Bortolotti, D.; Gentili, V.; Rotola, A.; Potena, L.; Rizzo, R. Soluble HLA-G pre-transplant levels to identify the risk for devel-opment of infection in heart transplant recipients. Hum. Immunol. 2020, 81, 147–150. [Google Scholar] [CrossRef]
- Kluckova, K.; Durmanova, V.; Bucova, M. Soluble HLA-G, its diagnostic and prognostic value and potential target molecule for future therapy in cancer. Bratisl. Lek. Listy 2021, 122, 60–617. [Google Scholar] [CrossRef]
- Poras, I.; Yaghi, L.; Martelli-Palomino, G.; Mendes-Junior, C.T.; Muniz, Y.C.N.; Cagnin, N.F.; Sgorla de Almeida, B.; Castelli, E.C.; Carosella, E.D.; Donadi, E.A. Haplotypes of the HLA-G 3′ untranslated region respond to endogenous factors of HLA-G+ and HLA-G-cell lines differentially. PLoS ONE 2017, 12, e0169032. [Google Scholar]
- De Campos Fraga-Silva, T.F.; Maruyama, S.R.; Sorgi, C.A.; de Sousa Russo, E.M.; Fernandes, A.P.M.; de Barros Cardoso, C.R.; Faccioli, L.H.; Dias-Baruffi, M.; Bonato, V.L.D. COVID-19: Integrating the Complexity of Systemic and Pulmonary Immunopathology to Identify Biomarkers for Different Outcomes. Front. Immunol. 2021, 11, 599736. [Google Scholar] [CrossRef] [PubMed]
- Shahraki, P.K.; Alavian-Mehr, A.; Farjadian, S. HLA-G: Facts and Fictions. Asian Pac. J. Cancer Biol. 2018, 3, 37–45. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, A.; David, J.K.; Maden, S.K.; Wood, M.A.; Weeder, B.R.; Nellore, A.; Thompson, R.F. Human leukocyte antigen susceptibility map for severe acute respiratory syndrome coronavirus 2. J. Virol. 2020, 94, e00510–e00520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zidi, I. Puzzling out the COVID-19: Therapy targeting HLA-G and HLA-E. Hum. Immunol. 2020, 81, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Gan, J.; Chen, B.-G.; Zheng, D.; Zhang, J.-G.; Lin, R.-H.; Zhou, Y.-P.; Yang, W.-Y.; Lin, A.; Yan, W.-H. Dynamics of peripheral immune cells and their HLA-G and receptor expressions in a patient suffering from critical COVID-19 pneumonia to convalescence. Clin. Transl. Immunol. 2020, 9, e1128. [Google Scholar] [CrossRef]
- Al-Bayatee, N.T.; Ad’hiah, A.H. Soluble HLA-G is upregulated in serum of patients with severe COVID-19. Hum. Immunol. 2021, 82, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, D.; Gentili, V.; Rizzo, S.; Schiuma, G.; Beltrami, S.; Spadaro, S.; Strazzabosco, G.; Campo, G.; Carosella, E.D.; Papi, A. Increased sHLA-G Is Associated with Improved COVID-19 Outcome and Reduced Neutrophil Adhesion. Viruses 2021, 13, 1855. [Google Scholar] [CrossRef]
- Vivarelli, S.; Falzone, L.; Torino, F.; Scandurra, G.; Russo, G.; Bordonaro, R.; Pappalardo, F.; Spandidos, D.A.; Raciti, G.; Libra, M. Immune-checkpoint inhibitors from cancer to COVID-19: A promising avenue for the treatment of patients with COVID-19. Int. J. Oncol. 2021, 58, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.-H.; Gan, J.; Xu, D.-P.; Li, L.; Yan, W.-H. Comprehensive Transcriptomic Analysis Reveals the Role of the Immune Checkpoint HLA-G Molecule in Cancers. Front. Immunol. 2021, 12, 614773. [Google Scholar] [CrossRef] [PubMed]
- García, M.; Palma, M.B.; Verine, J.; Miriuka, S.; Inda, A.M.; Errecalde, A.L.; Desgrandchamps, F.; Carosella, E.D.; Tronik-Le Roux, D. The immune-checkpoint HLA-G/ILT4 is involved in the regulation of VEGF expression in clear cell renal cell carci-noma. BMC Cancer 2020, 20, 624. [Google Scholar] [CrossRef]
- Chen, Q.-Y.; Chen, Y.-X.; Han, Q.-Y.; Zhang, J.-G.; Zhou, W.-J.; Zhang, X.; Ye, Y.-H.; Yan, W.-H.; Lin, A. Prognostic Signifi-cance of Immune Checkpoints HLA-G/ILT-2/4 and PD-L1 in Colorectal Cancer. Front. Immunol. 2021, 12, 679090. [Google Scholar] [CrossRef] [PubMed]
- Manaster, I.; Goldman-Wohl, D.; Greenfield, C.; Nachmani, D.; Tsukerman, P.; Hamani, Y.; Yagel, S.; Mandelboim, O. MiRNA-mediated control of HLA-G expression and function. PLoS ONE 2012, 7, e33395. [Google Scholar] [CrossRef] [PubMed]
- Laaribi, A.; Zidi, I.; Hannachi, N.; Ben Yahia, H.; Chaouch, H.; Bortolotti, D.; Zidi, N.; Letaief, A.; Yacoub, S.; Boudabous, A. Association of an HLA-G 14-bp Insertion/Deletion polymorphism with high HBV replication in chronic hepatitis. J. Viral Hepat. 2015, 22, 835–841. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, S.; Farhadi, L.; Ghasemi, F.; Sheikhesmaeili, F.; Mohammadi, A. The potential role of HLA-G in the pathogenesis of HBV infection: Immunosuppressive or immunoprotective? Infect. Genet. Evol. 2020, 85, 104580. [Google Scholar] [CrossRef]
- Khorrami, S.; Mohammadpour, H.; Shahzamani, K.; Zarif, M.N.; Sharifi, A.H.; Merat, S.; Poustchi, H. The relationship between HLA-G and viral loads in non-responder HCV-infected patients after combined therapy with IFN-α2α and ribavirin. Hum. Immunol. 2015, 76, 181–186. [Google Scholar] [CrossRef]
- Amiot, L.; Vu, N.; Rauch, M.; L’Helgoualc’h, A.; Chalmel, F.; Gascan, H.; Turlin, B.; Guyader, D.; Samson, M. Expression of HLA-G by mast cells is associated with hepatitis C virus-induced liver fibrosis. J. Hepatol. 2014, 60, 245–252. [Google Scholar] [CrossRef] [Green Version]
- Lajoie, J.; Hargrove, J.; Zijenah, L.S.; Humphrey, J.H.; Ward, B.J.; Roger, M. Genetic variants in nonclassical major histo-compatibility complex class I human leukocyte antigen (HLA)-E and HLA-G molecules are associated with susceptibility to heterosexual acquisition of HIV-1. J. Infect. Dis. 2006, 193, 298–301. [Google Scholar] [CrossRef] [Green Version]
- Jaakola, A.; Roger, M.; Faucher, M.-C.; Syrjänen, K.; Grénman, S.; Syrjänen, S.; Louvanto, K. HLA-G polymorphism impacts the outcome of oral HPV infections in women. BMC Infect. Dis. 2021, 21, 419. [Google Scholar] [CrossRef]
- Xu, H.-H.; Yan, W.-H.; Lin, A. The Role of HLA-G in Human Papillomavirus Infections and Cervical Carcinogenesis. Front. Immunol. 2020, 11, 1349. [Google Scholar] [CrossRef]
- Rizzo, R.; D’Accolti, M.; Bortolotti, D.; Caccuri, F.; Caruso, A.; Di Luca, D.; Caselli, E. Human Herpesvirus 6A and 6B inhibit in vitro angiogenesis by induction of Human Leukocyte Antigen G. Sci. Rep. 2018, 8, 17683. [Google Scholar] [CrossRef]
- Albayati, Z.; Alyami, A.; Alomar, S.; Middleton, D.; Bonnett, L.; Aleem, S.; Flanagan, B.F.; Christmas, S.E. The Influence of Cytomegalovirus on Expression of HLA-G and its Ligand KIR 2 DL 4 by Human Peripheral Blood Leucocyte Subsets. Scand. J. Immunol. 2017, 86, 396–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, R.; Gabrielli, L.; Bortolotti, D.; Gentili, V.; Piccirilli, G.; Chiereghin, A.; Pavia, C.; Bolzani, S.; Guerra, B.; Simonazzi, G. Study of soluble HLA-G in congenital human cytomegalovirus infection. J. Immunol. Res. 2016, 2016, 3890306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almeida, R.S.; Ferreira, M.L.B.; Sonon, P.; Cordeiro, M.T.; Sadissou, I.; Diniz, G.T.N.; de Fatima Pessoa Militão-Albuquerque, M.; Franca, R.F.D.O.; Donadi, E.A.; Lucena-Silva, N. Cytokines and soluble HLA-G levels in the acute and recovery phases of arbo-virus-infected Brazilian patients exhibiting neurological complications. Front. Immunol. 2021, 12, 582935. [Google Scholar] [CrossRef] [PubMed]
- Oliveira Souza, D.M.B.; Genre, J.; Alves Silva, T.G.; Soares, C.P.; Borges Ferreira Rocha, K.; Nunes Oliveira, C.; Nunes Jatoba, C.A.; Marco de Leon Andrade, J.; Moreau, P.; da Cunha Medeiros, A.; et al. Upregulation of Soluble HLA-G5 and HLA-G6 Isoforms in the Milder Histopathological Stages of Helicobacter pylori Infection: A Role for Subverting Immune Responses? Scand. J. Immunol. 2016, 83, 38–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, R.; Bergamini, G.; Bortolotti, D.; Leal, T.; D’Orazio, C.; Pintani, E.; Melchiorri, L.; Zavatti, E.; Assael, B.M.; Sorio, C. HLA-G expression and regulation during Pseudomonas aeruginosa infection in cystic fibrosis patients. Future Microbiol. 2016, 11, 363–373. [Google Scholar] [CrossRef] [Green Version]
- Bortolotti, D.; LeMaoult, J.; Trapella, C.; Di Luca, D.; Carosella, E.D.; Rizzo, R. Pseudomonas aeruginosa quorum sensing molecule N-(3-oxododecanoyl)-L-homoserine-lactone induces HLA-G expression in human immune cells. Infect. Immun. 2015, 83, 3918–3925. [Google Scholar] [CrossRef] [Green Version]
- Courtin, D.; Milet, J.; Sabbagh, A.; Massaro, J.D.; Castelli, E.C.; Jamonneau, V.; Bucheton, B.; Sese, C.; Favier, B.; Rouas-Freiss, N. HLA-G 3′ UTR-2 haplotype is associated with Human African trypanosomiasis susceptibility. Infect. Genet. Evol. 2013, 17, 1–7. [Google Scholar] [CrossRef]
- Gineau, L.; Courtin, D.; Camara, M.; Ilboudo, H.; Jamonneau, V.; Dias, F.C.; Tokplonou, L.; Milet, J.; Mendonça, P.B.; Castelli, E.C.; et al. Human leukocyte antigen-G: A promising prognostic marker of disease progression to improve the control of human Af-rican trypanosomiasis. Clin. Infect. Dis. 2016, 63, 1189–1197. [Google Scholar] [CrossRef]
- Ahouty, B.; Koffi, M.; Ilboudo, H.; Simo, G.; Matovu, E.; Mulindwa, J.; Hertz-Fowler, C.; Bucheton, B.; Sidibé, I.; Jamonneau, V.; et al. Candidate genes-based investigation of susceptibility to Human African Trypanosomiasis in Côte d’Ivoire. PLoS Negl. Trop. Dis. 2017, 11, e0005992. [Google Scholar] [CrossRef] [Green Version]
- Ofon, E.; Noyes, H.; Mulindwa, J.; Ilboudo, H.; Simuunza, M.; Ebo’o, V.; Njiokou, F.; Koffi, M.; Bucheton, B.; Fogue, P.; et al. A polymorphism in the haptoglobin, haptoglobin related protein locus is associated with risk of human sleeping sickness within Cameroonian populations. PLoS Negl. Trop. Dis. 2017, 11, e0005979. [Google Scholar] [CrossRef] [Green Version]
- Kimuda, M.P.; Noyes, H.; Mulindwa, J.; Enyaru, J.; Alibu, V.P.; Sidibe, I.; Mumba Ngoyi, D.; Hertz-Fowler, C.; MacLeod, A.; Tastan Bishop, Ö.; et al. No evidence for association between APOL1 kidney disease risk alleles and Human African Trypanosomiasis in two Ugandan populations. PLoS Negl. Trop. Dis. 2018, 12, e0006300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dias, F.C.; Mendes-Junior, C.T.; Silva, M.C.; Tristão, F.S.; Dellalibera-Joviliano, R.; Moreau, P.; Soares, E.G.; Menezes, J.G.; Schmidt, A.; Dantas, R.O.; et al. Human leucocyte antigen-G (HLA-G) and its murine functional homolog Qa2 in the Trypanosoma cruzi Infection. Mediat. Inflamm. 2015, 2015, 595829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadissou, I.; d’Almeida, T.; Cottrell, G.; Luty, A.; Krawice-Radanne, I.; Massougbodji, A.; Moreau, P.; Moutairou, K.; Garcia, A.; Favier, B.; et al. High plasma levels of HLA-G are associated with low birth weight and with an increased risk of malaria in infancy. Malar. J. 2014, 13, 312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia, A.; Milet, J.; Courtin, D.; Sabbagh, A.; Massaro, J.D.; Castelli, E.C.; Migot-Nabias, F.; Favier, B.; Rouas-Freiss, N.; Donadi, E.A.; et al. Association of HLA-G 3′ UTR polymorphisms with response to malaria infection: A first insight. Infect. Genet. Evol. 2013, 16, 263–269. [Google Scholar] [CrossRef]
- Sabbagh, A.; Courtin, D.; Milet, J.; Massaro, J.D.; Castelli, E.C.; Migot-Nabias, F.; Favier, B.; Rouas-Freiss, N.; Moreau, P.; Garcia, A.; et al. Association of HLA-G 3′ untranslated region polymorphisms with antibody response against Plasmodium falciparum antigens: Preliminary results. Tissue Antigens 2013, 82, 53–58. [Google Scholar] [CrossRef]
- D’Almeida, T.C.; Sadissou, I.; Cottrell, G.; Tahar, R.; Moreau, P.; Favier, B.; Moutairou, K.; Donadi, E.A.; Massougbodji, A.; Rouass-Freiss, N. Evolution of the levels of human leukocyte antigen G (HLA-G) in Beninese infant during the first year of life in a malaria endemic area: Using latent class analysis. Malar. J. 2016, 15, 78. [Google Scholar] [CrossRef] [Green Version]
- d’Almeida, T.C.; Sadissou, I.; Milet, J.; Cottrell, G.; Mondière, A.; Avokpaho, E.; Gineau, L.; Sabbagh, A.; Massougbodji, A.; Moutairou, K.; et al. Soluble human leukocyte antigen-G during pregnancy and infancy in Benin: Mother/child resemblance and association with the risk of malaria infection and low birth weight. PLoS ONE 2017, 12, e0171117. [Google Scholar] [CrossRef]
- Donaghy, L.; Gros, F.; Amiot, L.; Mary, C.; Maillard, A.; Guiguen, C.; Gangneux, J.-P. Elevated levels of soluble non-classical major histocompatibility class I molecule human leucocyte antigen (HLA)-G in the blood of HIV-infected patients with or without visceral leishmaniasis. Clin. Exp. Immunol. 2007, 147, 236–240. [Google Scholar] [CrossRef]
- Gangneux, J.-P.; Poinsignon, Y.; Donaghy, L.; Amiot, L.; Tarte, K.; Mary, C.; Robert-Gangneux, F. Indoleamine 2,3-dioxygenase activity as a potential biomarker of immune suppression during visceral leishmaniasis. Innate Immun. 2013, 19, 564–568. [Google Scholar] [CrossRef]
- Han, M.; Jiang, Y.; Lao, K.; Xu, X.; Zhan, S.; Wang, Y.; Hu, X. sHLA-G involved in the apoptosis of decidual natural killer cells following Toxoplasma gondii infection. Inflammation 2014, 37, 1718–1727. [Google Scholar] [CrossRef]
- Robert-Gangneux, F.; Gangneux, J.-P.; Vu, N.; Jaillard, S.; Guiguen, C.; Amiot, L. High level of soluble HLA-G in amniotic fluid is correlated with congenital transmission of Toxoplasma gondii. Clin. Immunol. 2011, 138, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Zhang, R.; Xu, X.; Liu, Y.; Zhang, H.; Zhai, X.; Hu, X. IL-10 reduces levels of apoptosis in Toxoplasma gondii-infected trophoblasts. PLoS ONE 2013, 8, e56455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avokpaho, E.; d’Almeida, T.C.; Sadissou, I.; Tokplonou, L.; Adamou, R.; Sonon, P.; Milet, J.; Cottrell, G.; Mondière, A.; Mas-sougbodji, A. HLA-G expression during hookworm infection in pregnant women. Acta Trop. 2019, 196, 52–59. [Google Scholar] [CrossRef]
- Mariconti, M.; Meroni, V.; Badulli, C.; Brunetti, E.; Tinelli, C.; De Silvestri, A.; Tamarozzi, F.; Genco, F.; Casulli, A.; Martinetti, M. Correlation of serum sHLA-G levels with cyst stage in patients with cystic echinococcosis: Is it an immune evasion strategy? Parasite Immunol. 2016, 38, 414–418. [Google Scholar] [CrossRef] [PubMed]
- d’Almeida, T.C.; Sadissou, I.; Sagbohan, M.; Milet, J.; Avokpaho, E.; Gineau, L.; Sabbagh, A.; Moutairou, K.; Donadi, E.A.; Favier, B.; et al. High level of soluble human leukocyte antigen (HLA)-G at beginning of pregnancy as predictor of risk of malaria during infancy. Sci. Rep. 2019, 9, 9160. [Google Scholar] [CrossRef] [PubMed]
- Wilairatana, P.; Masangkay, F.R.; Kotepui, K.U.; Milanez, G.D.J.; Kotepui, M. Prevalence and characteristics of malaria among COVID-19 individuals: A systematic review, meta-analysis, and analysis of case reports. PLoS Negl. Trop. Dis. 2021, 15, e0009766. [Google Scholar] [CrossRef]
- Wilairatana, P.; Chanmol, W.; Rattaprasert, P.; Masangkay, F.R.; Milanez, G.D.J.; Kotepui, K.U.; Kotepui, M. Prevalence and characteristics of malaria co-infection among individuals with visceral leishmaniasis in Africa and Asia: A systematic review and meta-analysis. Parasites Vectors 2021, 14, 545. [Google Scholar] [CrossRef]
- Anchang-Kimbi, J.K.; Elad, D.M.; Sotoing, G.T.; Achidi, E.A. Coinfection with Schistosoma haematobium and Plasmodium falci-parum and anaemia severity among pregnant women in Munyenge, Mount Cameroon area: A cross-sectional study. J. Parasitol. Res. 2017, 2017, 6173465. [Google Scholar] [CrossRef] [Green Version]
- Lemaitre, M.; Watier, L.; Briand, V.; Garcia, A.; Le Hesran, J.-Y.; Cot, M. Coinfection with Plasmodium falciparum and Schistosoma haematobium: Additional evidence of the protective effect of Schistosomiasis on malaria in Senegalese children. Am. J. Trop. Med. Hyg. 2014, 90, 329–334. [Google Scholar] [CrossRef] [Green Version]
- Zawawi, A.; Alghanmi, M.; Alsaady, I.; Gattan, H.; Zakai, H.; Couper, K. The impact of COVID-19 pandemic on malaria elimination. Parasite Epidemiol. Control 2020, 11, e00187. [Google Scholar] [CrossRef]
- Tokplonou, L.; Nouatin, O.; Sonon, P.; M’po, G.; Glitho, S.; Agniwo, P.; Gonzalez-Ortiz, D.; Tchégninougbo, T.; Ayitchédji, A.; Favier, B.; et al. Schistosoma haematobium infection modulates Plasmodium falciparum parasite density and antimalarial antibody re-sponses. Parasite Immunol. 2020, 42, e12702. [Google Scholar] [CrossRef] [PubMed]
- Nouatin, O.; Ngoa, U.A.; Ibáñez, J.; Dejon-Agobe, J.C.; Mordmüller, B.; Edoa, J.R.; Mougeni, F.; Brückner, S.; Hounkpatin, A.B.; Esen, M.; et al. Effect of immune regulatory pathways after immunization with GMZ2 malaria vaccine candidate in healthy lifelong malaria-exposed adults. Vaccine 2020, 38, 4263–4272. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.-S.; Qu, X.-Y.; Zhang, W.-Z.; Li, J.; Lv, Z.-Y. Infection against infection: Parasite antagonism against parasites, viruses and bacteria. Infect. Dis. Poverty 2019, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Vasireddi, M.; Hilliard, J. Herpes B virus, macacine herpesvirus 1, breaks simplex virus tradition via major histocompatibility complex class I expression in cells from human and macaque hosts. J. Virol. 2012, 86, 12503–12511. [Google Scholar] [CrossRef] [Green Version]
- Iesa, M.A.; Osman, M.E.; Hassan, M.A.; Dirar, A.I.; Abuzeid, N.; Mancuso, J.J.; Pandey, R.; Mohammed, A.A.; Borad, M.J.; Babiker, H.M.; et al. SARS-CoV-2 and Plasmodium falciparum common immunodominant regions may explain low COVID-19 incidence in the malaria-endemic belt. New Microbes New Infect. 2020, 38, 100817. [Google Scholar] [CrossRef]
- Sartelet, H.; Schleiermacher, D.; Le-Hesran, J.-Y.; Graesslin, O.; Gaillard, D.; Fe, M.; Lechki, C.; Gaye, A.; Le Bouteiller, P.; Birembaut, P. Less HLA-G expression in Plasmodium falciparum-infected third trimester placentas is associated with more natural killer cells. Placenta 2005, 26, 505–511. [Google Scholar] [CrossRef]
- Sancho, L.; Martinez, C.; Nogales, A.; De La Hera, A. Reconstitution of natural-killer-cell activity in the newborn by interleu-kin-2. N. Engl. J. Med. 1986, 314, 57–58. [Google Scholar]
- Herrmann, M.; Schulte, S.; Wildner, N.H.; Wittner, M.; Brehm, T.T.; Ramharter, M.; Woost, R.; Lohse, A.W.; Jacobs, T.; Schulze zur Wiesch, J. Analysis of co-inhibitory receptor expression in COVID-19 infection compared to acute Plasmodium falciparum malaria: LAG-3 and TIM-3 correlate with t cell activation and course of disease. Front. Immunol. 2020, 11, 1870. [Google Scholar] [CrossRef]
- La Manna, M.P.; Orlando, V.; Tamburini, B.; Badami, G.D.; Dieli, F.; Caccamo, N. Harnessing unconventional T cells for immunotherapy of tuberculosis. Front. Immunol. 2020, 11, 2107. [Google Scholar] [CrossRef]
- Bai, X.; Yi, M.; Jiao, Y.; Chu, Q.; Wu, K. Blocking TGF-β signaling to enhance the efficacy of immune checkpoint inhibitor. OncoTargets Ther. 2019, 12, 9527. [Google Scholar] [CrossRef] [Green Version]
- Ullah, M.; Azazzen, D.; Kaci, R.; Benabbou, N.; Lauraine, E.P.; Pocard, M.; Mirshahi, M. High expression of HLA-G in ovarian carcinomatosis: The role of interleukin-1β. Neoplasia 2019, 21, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, H.; Faghihloo, E. Viruses as key modulators of the TGF-β pathway; a double-edged sword involved in cancer. Rev. Med. Virol. 2018, 28, e1967. [Google Scholar] [CrossRef] [PubMed]
- Kordelas, L.; Rebmann, V.; Ludwig, A.; Radtke, S.; Ruesing, J.; Doeppner, T.; Epple, M.; Horn, P.; Beelen, D.; Giebel, B. MSC-derived exosomes: A novel tool to treat therapy-refractory graft-versus-host disease. Leukemia 2014, 28, 970–973. [Google Scholar] [CrossRef]
- Uckun, F.M.; Saund, S.; Windlass, H.; Trieu, V. Repurposing Anti-Malaria Phytomedicine Artemisinin as a COVID-19 Drug. Front. Pharmacol. 2021, 12, 407. [Google Scholar] [CrossRef]
- Drewry, L.L.; Harty, J.T. Balancing in a black box: Potential immunomodulatory roles for TGF-β signaling during blood-stage malaria. Virulence 2020, 11, 159–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osman, M.; Faridi, R.M.; Sligl, W.; Shabani-Rad, M.-T.; Dharmani-Khan, P.; Parker, A.; Kalra, A.; Tripathi, M.B.; Storek, J.; Cohen Tervaert, J.W.; et al. Impaired natural killer cell counts and cytolytic activity in patients with severe COVID-19. Blood Adv. 2020, 4, 5035–5039. [Google Scholar] [CrossRef]
- Ahmed, F.; Jo, D.-H.; Lee, S.-H. Can Natural Killer Cells Be a Principal Player in Anti-SARS-CoV-2 Immunity? Front. Immunol. 2020, 11, 586765. [Google Scholar] [CrossRef]
- Ademe, M.; Girma, F. The Influence of Helminth Immune Regulation on COVID-19 Clinical Outcomes: Is it Beneficial or Det-rimental? Infect. Drug Resist. 2021, 14, 4421. [Google Scholar] [CrossRef]
- Wolday, D.; Gebrecherkos, T.; Arefaine, Z.G.; Kebede, Y.; Gebreegzabher, A.; Tasew, G.; Abdulkader, M.; Abraha, H.E.; Desta, A.A.; Atsbaha, A.H.; et al. Effect of Co-Infection with Intestinal Parasites on COVID-19 Severity: A Prospective Observational Cohort Study. eClinicalMedicine 2021, 39, 101054. [Google Scholar] [CrossRef]
- Hoque, M.N.; Akter, S.; Mishu, I.D.; Islam, M.R.; Rahman, M.S.; Akhter, M.; Islam, I.; Hasan, M.M.; Rahaman, M.M.; Sultana, M.; et al. Microbial co-infections in COVID-19: Associated microbiota and underlying mechanisms of pathogenesis. Microb. Pathog. 2021, 156, 104941. [Google Scholar] [CrossRef]
- Parker, W.; Sarafian, J.T.; Broverman, S.A.; Laman, J.D. Between a hygiene rock and a hygienic hard place: Avoiding SARS-CoV-2 while needing environmental exposures for immunity. Evol. Med. Public Health 2021, 9, 120–130. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rashidi, S.; Vieira, C.; Tuteja, R.; Mansouri, R.; Ali-Hassanzadeh, M.; Muro, A.; Nguewa, P.; Manzano-Román, R. Immunomodulatory Potential of Non-Classical HLA-G in Infections including COVID-19 and Parasitic Diseases. Biomolecules 2022, 12, 257. https://doi.org/10.3390/biom12020257
Rashidi S, Vieira C, Tuteja R, Mansouri R, Ali-Hassanzadeh M, Muro A, Nguewa P, Manzano-Román R. Immunomodulatory Potential of Non-Classical HLA-G in Infections including COVID-19 and Parasitic Diseases. Biomolecules. 2022; 12(2):257. https://doi.org/10.3390/biom12020257
Chicago/Turabian StyleRashidi, Sajad, Carmen Vieira, Renu Tuteja, Reza Mansouri, Mohammad Ali-Hassanzadeh, Antonio Muro, Paul Nguewa, and Raúl Manzano-Román. 2022. "Immunomodulatory Potential of Non-Classical HLA-G in Infections including COVID-19 and Parasitic Diseases" Biomolecules 12, no. 2: 257. https://doi.org/10.3390/biom12020257






