Motilin, a Novel Orexigenic Factor, Involved in Feeding Regulation in Yangtze Sturgeon (Acipenser dabryanus)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Fish and Procedures
2.3. Molecular Cloning and Sequence Analysis of Motilin and Motilin Receptor
2.4. Reagents
2.5. Quantitative Real-Time PCR
2.6. Statistical Analysis
3. Results
3.1. Molecular Cloning of AdMotilin and AdMotilinR
3.2. Tissue Distribution
3.3. Effects of Weaning on the Expression of AdMotilin and AdMotilinR
3.4. Effects of Fasting on AdMotilin and AdMotilinR Expression
3.5. Effects of Motilin-NH2 Injection on Food Intake of Yangtze Sturgeon
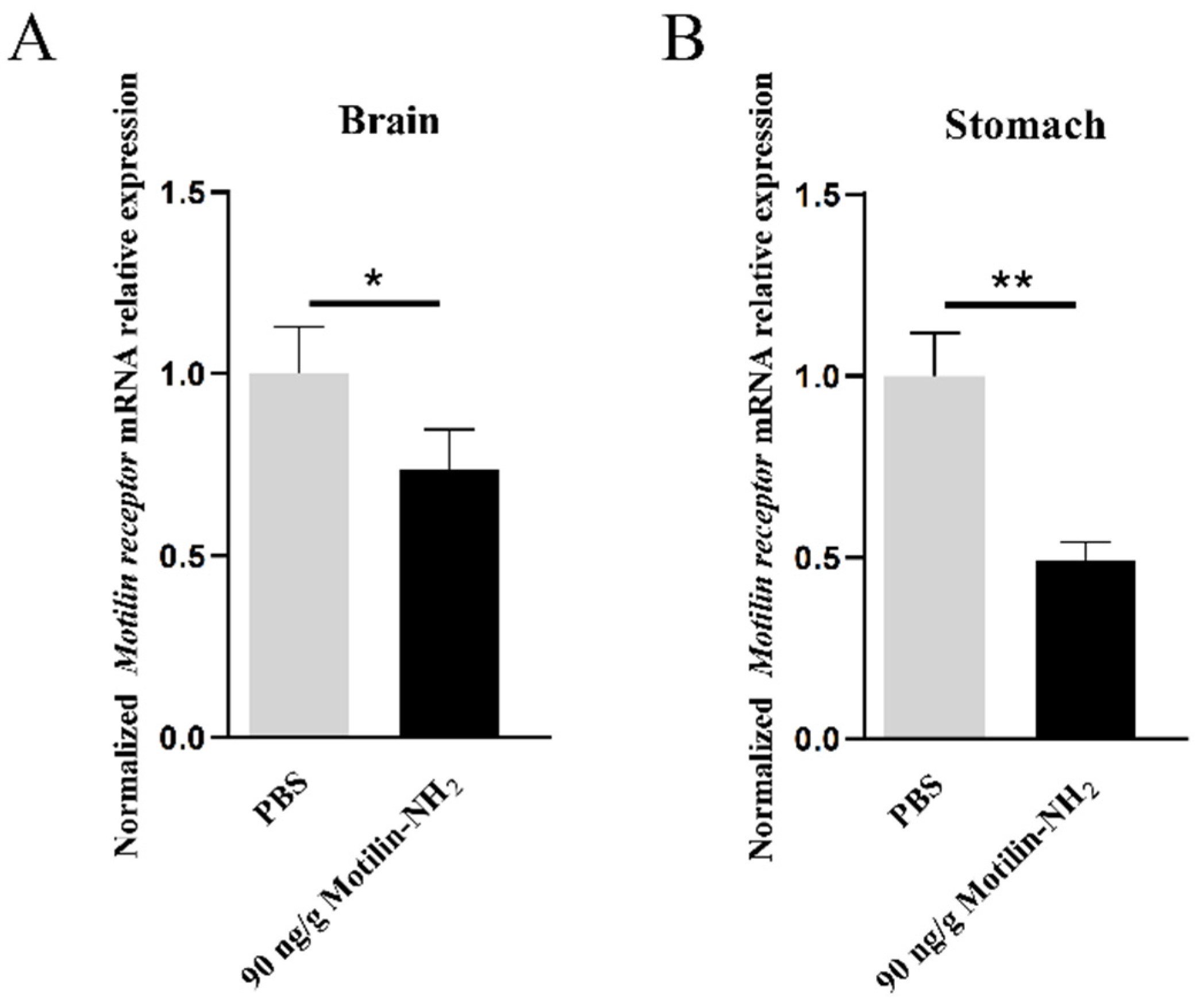
3.6. Effects of Motilin-NH2 Injection on AdMotilinR Expression
3.7. Effects of Motilin-NH2 Injection on Appetite Factors Expression
3.8. In Vitro Effects of Motilin-NH2 Incubation on Appetite Factors Expression in the Brain and Stomach Fragments
4. Discussion
4.1. The Analysis of AdMotilin and AdMotilinR Sequences in Yangtze Sturgeon
4.2. The Tissue Expression of AdMotilin and AdMotilinR
4.3. Weaning and Fasting Induced AdMotilin and AdMotilinR Expression Changes
4.4. Motilin Polypeptide Promoted Food Intake of Yangtze Sturgeon
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- St-Pierre, B.; Perez Palencia, J.Y.; Samuel, R.S. Impact of Early Weaning on Development of the Swine Gut Microbiome. Microorganisms 2023, 11, 1753. [Google Scholar] [CrossRef] [PubMed]
- Morison, B.J.; Heath, A.M.; Haszard, J.J.; Hein, K.; Fleming, E.A.; Daniels, L.; Erickson, E.W.; Fangupo, L.J.; Wheeler, B.J.; Taylor, B.J.; et al. Impact of a Modified Version of Baby-Led Weaning on Dietary Variety and Food Preferences in Infants. Nutrients 2018, 10, 1092. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; He, K.; Luo, J.; Sun, J.; Liao, L.; Tang, X.; Liu, Q.; Yang, S. Co-modulation of Liver Genes and Intestinal Microbiome of Largemouth Bass Larvae (Micropterus salmoides) During Weaning. Front. Microbiol. 2020, 11, 1332. [Google Scholar] [CrossRef] [PubMed]
- Bertucci, J.I.; Blanco, A.M.; Sundarrajan, L.; Rajeswari, J.J.; Velasco, C.; Unniappan, S. Nutrient Regulation of Endocrine Factors Influencing Feeding and Growth in Fish. Front. Endocrinol. 2019, 10, 83. [Google Scholar] [CrossRef] [PubMed]
- Volkoff, H. The Neuroendocrine Regulation of Food Intake in Fish: A Review of Current Knowledge. Front. Neurosci. 2016, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Pham, L.P.; Jordal, A.O.; Nguyen, M.V.; Rønnestad, I. Food intake, growth, and expression of neuropeptides regulating appetite in clown anemonefish (Amphiprion ocellaris) exposed to predicted climate changes. Gen. Comp. Endocrinol. 2021, 304, 113719. [Google Scholar] [CrossRef]
- Yuan, D.; Gao, Y.; Zhang, X.; Wang, B.; Chen, H.; Wu, Y.; Chen, D.; Wang, Z.; Li, Z. NPY and NPY receptors in the central control of feeding and interactions with CART and MC4R in Siberian sturgeon. Gen. Comp. Endocrinol. 2019, 284, 113239. [Google Scholar] [CrossRef] [PubMed]
- Fabian Canosa, L.; Ignacio Bertucci, J. Nutrient regulation of somatic growth in teleost fish. The interaction between somatic growth, feeding and metabolism. Mol. Cell. Endocrinol. 2020, 518, 111029. [Google Scholar] [CrossRef] [PubMed]
- Lau, J.; Farzi, A.; Qi, Y.; Heilbronn, R.; Mietzsch, M.; Shi, Y.C.; Herzog, H. CART neurons in the arcuate nucleus and lateral hypothalamic area exert differential controls on energy homeostasis. Mol. Metab. 2018, 7, 102–118. [Google Scholar] [CrossRef]
- Chen, H.; Wang, B.; Zhou, B.; Qi, J.W.; Tang, N.; Wang, S.Y.; Tian, Z.Z.; Wang, M.; Xu, S.Q.; Yu, N.Y.; et al. Characterization, phylogeny, and responses of leptin to different nutritional states in critically endangered Yangtze sturgeon (Acipenser dabryanus). Aquaculture 2020, 525, 735296. [Google Scholar] [CrossRef]
- Abdalla, M.M. Central and peripheral control of food intake. Endocr. Regul. 2017, 51, 52–70. [Google Scholar] [CrossRef]
- Cawthon, C.R.; de La Serre, C.B. The critical role of CCK in the regulation of food intake and diet-induced obesity. Peptides 2021, 138, 170492. [Google Scholar] [CrossRef] [PubMed]
- Hyland, L.; Park, S.B.; Abdelaziz, Y.; Abizaid, A. Ghrelin infused into the dorsomedial hypothalamus of male mice increases food intake and adiposity. Physiol. Behav. 2020, 220, 7. [Google Scholar] [CrossRef]
- Kitazawa, T.; Kaiya, H. Motilin Comparative Study: Structure, Distribution, Receptors, and Gastrointestinal Motility. Front. Endocrinol. 2021, 12, 700884. [Google Scholar] [CrossRef]
- Cooper, T.A.; Jenkins, S.J.; Wojakiewicz, L.; Kattesh, H.G.; Kojima, C.J. Effects of weaning and syndyphalin-33 on expression of melanocortinergic appetite-regulating genes in swine. Domest. Anim. Endocrinol. 2011, 40, 165–172. [Google Scholar] [CrossRef]
- Depoortere, I.; De Clercq, P.; Svoboda, M.; Bare, L.; Peeters, T.L. Identification of motilin mRNA in the brain of man and rabbit. Conservation of polymorphism of the motilin gene across species. Peptides 1997, 18, 1497–1503. [Google Scholar] [CrossRef]
- Huang, Z.; De Clercq, P.; Depoortere, I.; Peeters, T.L. Isolation and sequence of cDNA encoding the motilin precursor from monkey intestine. Demonstration of the motilin precursor in the monkey brain. FEBS Lett. 1998, 435, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, C.; Kajihara, K.; Yanaka, T.; Sakata, I.; Itoh, Z.; Oda, S.I.; Sakai, T. House musk shrew (Suncus murinus, order: Insectivora) as a new model animal for motilin study. Peptides 2009, 30, 318–329. [Google Scholar] [CrossRef] [PubMed]
- De Clercq, P.; Depoortere, I.; Macielag, M.; Vandermeers, A.; Vandermeers-Piret, M.C.; Peeters, T.L. Isolation, sequence, and bioactivity of chicken motilin. Peptides 1996, 17, 203–208. [Google Scholar] [CrossRef]
- Apu, A.S.; Mondal, A.; Kitazawa, T.; Takemi, S.; Sakai, T.; Sakata, I. Molecular cloning of motilin and mechanism of motilin-induced gastrointestinal motility in Japanese quail. Gen. Comp. Endocrinol. 2016, 233, 53–62. [Google Scholar] [CrossRef]
- Zhang, S.; Kaiya, H.; Kitazawa, T. Motilin is a regulator of gastric contraction in Japanese fire belly newts (Cynops pyrrhogaster), in vitro studies using isolated gastrointestinal strips of newts, rabbits, and chickens. Gen. Comp. Endocrinol. 2023, 330, 114140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Okuhara, Y.; Iijima, M.; Takemi, S.; Sakata, I.; Kaiya, H.; Teraoka, H.; Kitazawa, T. Identification of pheasant ghrelin and motilin and their actions on contractility of the isolated gastrointestinal tract. Gen. Comp. Endocrinol. 2020, 285, 11. [Google Scholar] [CrossRef] [PubMed]
- Goswami, C.; Shimada, Y.; Yoshimura, M.; Mondal, A.; Oda, S.; Tanaka, T.; Sakai, T.; Sakata, I. Motilin Stimulates Gastric Acid Secretion in Coordination with Ghrelin in Suncus murinus. PLoS ONE 2015, 462, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Li, H.; Jin, Y.; He, Y.; Mei, L.; Jin, C. Differential expression of motilin receptors on the endothelium of dog gastrointestinal arteries and motilin-induced motilin receptor dependent relaxation of corresponding arteries. Peptides 2021, 143, 170574. [Google Scholar] [CrossRef] [PubMed]
- Konturek, P.C.; Brzozowski, T.; Konturek, S.J. Gut clock: Implication of circadian rhythms in the gastrointestinal tract. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2011, 62, 139–150. [Google Scholar]
- Miegueu, P.; Cianflone, K.; Richard, D.; St-Pierre, D.H. Motilin stimulates preadipocyte proliferation and differentiation and adipocyte lipid storage. Am. J. Physiol.-Endocrinol. Metab. 2011, 301, E758–E766. [Google Scholar] [CrossRef] [PubMed]
- Tack, J.; Deloose, E.; Ang, D.; Scarpellini, E.; Vanuytsel, T.; Van Oudenhove, L.; Depoortere, I. Motilin-induced gastric contractions signal hunger in man. Gut 2016, 65, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Garthwaite, T.L. Peripheral motilin administration stimulates feeding in fasted rats. Peptides 1985, 6, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Bungo, T.; Ueda, H.; Kitazawa, T.; Taneike, T.; Furuse, M. Intracerebroventricular administration of chicken motilin does not induce hyperphagia in meat-type chicks. Physiol. Behav. 2004, 82, 199–203. [Google Scholar] [CrossRef]
- Ando, R.; Bungo, T.; Kanwakami, S.; Shimojo, M.; Masuda, Y.; Furuse, M. Intracerebroventricular injection of mammalian motilin, melanin-concentrating hormone or galanin does not stimulate food intake in neonatal chicks. Br. Poult. Sci. 2000, 41, 508–511. [Google Scholar] [CrossRef]
- Liu, Y.; Li, S.S.; Huang, X.G.; Lu, D.Q.; Liu, X.C.; Ko, W.H.; Zhang, Y.; Cheng, C.H.K.; Lin, H.R. Identification and characterization of a motilin-like peptide and its receptor in teleost. Gen. Comp. Endocrinol. 2013, 186, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Olsson, C.; Holbrook, J.D.; Bompadre, G.; Jönsson, E.; Hoyle, C.H.; Sanger, G.J.; Holmgren, S.; Andrews, P.L. Identification of genes for the ghrelin and motilin receptors and a novel related gene in fish, and stimulation of intestinal motility in zebrafish (Danio rerio) by ghrelin and motilin. Gen. Comp. Endocrinol. 2008, 155, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Lie, K.K.; Tørresen, O.K.; Solbakken, M.H.; Rønnestad, I.; Tooming-Klunderud, A.; Nederbragt, A.J.; Jentoft, S.; Sæle, Ø. Loss of stomach, loss of appetite? Sequencing of the ballan wrasse (Labrus bergylta) genome and intestinal transcriptomic profiling illuminate the evolution of loss of stomach function in fish. BMC Genom. 2018, 19, 186. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.Y.; Qi, X.; Wen, H.S.; Zhang, K.Q.; Zhang, X.Y.; Li, J.; Li, Y.; Fan, H.Y. Identification, expression analysis, and functional characterization of motilin and its receptor in spotted sea bass (Lateolabrax maculatus). Gen. Comp. Endocrinol. 2019, 277, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Ke, F.E.; Wei, Q.; He, X.; Cen, Y.J.E.B.o.F. Biology and life history of Dabry’s sturgeon, Acipenser dabryanus, in the Yangtze River. Environ. Biol. Fish 1997, 48, 257–264. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, G.M.; Wang, Q.S. China Red Data Book of Endangered Animals: Pisces; Science Press: Beijing, China, 1998. [Google Scholar]
- Chandra, G.; Fopp-Bayat, D. Trends in aquaculture and conservation of sturgeons: A review of molecular and cytogenetic tools: Genetic Tools. Rev. Aquacult. 2020, 3, 119–137. [Google Scholar]
- Wei, Q.J.F. Foundation and Prospects of Wild Population Reconstruction of Acipenser dabryanus. Fish 2021, 6, 55. [Google Scholar]
- Rosenfeld, D.J.; Garthwaite, T.L. Central administration of motilin stimulates feeding in rats. Physiol. Behav. 1987, 39, 753–756. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Mei, W.A.; Yl, A.; Yl, A.; Bo, Z.B.; Ni, T.A.; Sz, A.; Sx, A.; Ny, A.; Ql, A. Using the transcriptome to evaluate the best reference genes for studying nutrition of the critically endangered Yangtze sturgeon (Acipenser dabryanus). Aquaculture 2021, 543, 736894. [Google Scholar]
- Xu, L.; Depoortere, I.; Tang, M.; Peeters, T.L. Identification and expression of the motilin precursor in the guinea pig. FEBS Lett. 2001, 490, 7–10. [Google Scholar] [CrossRef]
- Bemis, W.E.; Findeis, E.K.; Grande, L. An overview of Acipenseriforms. Environ. Biol. Fish 2002, 48, 25–71. [Google Scholar] [CrossRef]
- Kitazawa, T.; Harada, R.; Sakata, I.; Sakai, T.; Kaiya, H. A verification study of gastrointestinal motility-stimulating action of guinea-pig motilin using isolated gastrointestinal strips from rabbits and guinea-pigs. Gen. Comp. Endocrinol. 2019, 274, 106–112. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Irwin, D.M.; Chen, R.; Zhang, Y.-P. Stepwise loss of motilin and its specific receptor genes in rodents. J. Mol. Endocrinol. 2010, 44, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Aerssens, J.; Depoortere, I.; Thielemans, L.; Mitselos, A.; Coulie, B.; Peeters, T. The rat lacks functional genes for motilin and the motilin receptor. Neurogastroenterol. Motil. 2004, 16, 841. [Google Scholar]
- Cui, H.Y.; Luo, X.U.; Zhou, H.F. The Impact study of motilin digestive function in hypothalamus paraventricular nucleus. Prog. Mod. Biomed. 2011, 11, 460–464. [Google Scholar]
- Dudani, A.; Aizawa, S.; Zhi, G.; Tanaka, T.; Jogahara, T.; Sakata, I.; Sakai, T. The proximal gastric corpus is the most responsive site of motilin-induced contractions in the stomach of the Asian house shrew. J. Comp. Physiol. B Biochem. Syst. Environ. Physiol. 2016, 186, 665–675. [Google Scholar] [CrossRef]
- Du, K.; Stöck, M.; Kneitz, S.; Klopp, C.; Woltering, J.M.; Adolfi, M.C.; Feron, R.; Prokopov, D.; Makunin, A.; Kichigin, I.; et al. The sterlet sturgeon genome sequence and the mechanisms of segmental rediploidization. Nat. Ecol. Evol. 2020, 4, 841–852. [Google Scholar] [CrossRef] [PubMed]
- Ohshiro, H.; Nonaka, M.; Ichikawa, K. Molecular identification and characterization of the dog motilin receptor. Regul. Pept. 2008, 146, 80–87. [Google Scholar] [CrossRef]
- Suzuki, A.; Ishida, Y.; Aizawa, S.; Sakata, I.; Tsutsui, C.; Mondal, A.; Kanako, K.; Sakai, T. Molecular identification of GHS-R and GPR38 in Suncus murinus. Peptides 2012, 36, 29–38. [Google Scholar] [CrossRef]
- Feighner, S.D.; Tan, C.P.; McKee, K.K.; Palyha, O.C.; Hreniuk, D.L.; Pong, S.S.; Austin, C.P.; Figueroa, D.; MacNeil, D.; Cascieri, M.A.; et al. Receptor for motilin identified in the human gastrointestinal system. Science 1999, 284, 2184–2188. [Google Scholar] [CrossRef]
- Yamamoto, I.; Kaiya, H.; Tsutsui, C.; Sakai, T.; Tsukada, A.; Miyazato, M.; Tanakae, M. Primary structure, tissue distribution, and biological activity of chicken motilin receptor. Gen. Comp. Endocrinol. 2008, 156, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, T.; Matsuura, B.; Miyake, T.; Abe, M.; Ikeda, Y.; Hiasa, Y. Effects of Motilin Receptor Agonists and Ghrelin in Human motilin receptor Transgenic Mice. Int. J. Mol. Sci. 2019, 20, 1521. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Depoortere, I.; Tomasetto, C.; Zandecki, M.; Tang, M.; Timmermans, J.-P.; Peeters, T.L. Evidence for the presence of motilin, ghrelin, and the motilin and ghrelin receptor in neurons of the myenteric plexus. Regul. Pept. 2005, 124, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Palmegiano, G.B.; Gai, F.; Daprà, F.; Gasco, L.; Pazzaglia, M.; Peiretti, P.G.J.A.R. Effects of Spirulina and plant oil on the growth and lipid traits of white sturgeon (Acipenser transmontanus) fingerlings. Aquac. Res. 2010, 39, 587–595. [Google Scholar] [CrossRef]
- Laczynska, B.; Siddique, M.; Ziomek, E.; Shelton, W.L.; Aquaculture, D.F.J.N.A.J.o. Early Weaning Effects on Survival, Growth, and Histopathology of Larval Sterlet Acipenser ruthenus. North Am. J. Aquac. 2020, 82, 181–189. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Chen, S.; Wang, B.; Wu, H.; Tang, N.; Zhao, L.; Yang, S.; Liu, Q.; Zhou, B.; et al. Transcriptome Reveals the Effects of Early Weaning on Lipid Metabolism and Liver Health of Yangtze Sturgeon (Acipenser dabryanus). Int. J. Mol. Sci. 2022, 23, 10866. [Google Scholar] [CrossRef] [PubMed]
- Dendy, R.; Stinson, E.J.; Guerithault, N.; Gluck, M.E. Brain Stimulation to Modulate Food Intake and Eating Behavior. Curr. Diabetes Rep. 2019, 19, 152. [Google Scholar] [CrossRef] [PubMed]
- Manning, A.J.; Murray, H.M.; Gallant, J.W.; Matsuoka, M.P.; Radford, E.; Douglas, S.E. Ontogenetic and tissue-specific expression of preproghrelin in the Atlantic halibut, Hippoglossus hippoglossus L. J. Endocrinol. 2008, 196, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Yu, X.; Han, J.; Yu, H.; Xue, M. Effects of dietary protein sources on growth performance and feed intake regulation of grass carp (Ctenopharyngodon idellus). Aquaculture 2019, 510, 216–224. [Google Scholar] [CrossRef]
- Deloose, E.; Biesiekierski, J.R.; Vanheel, H.; Depoortere, I.; Tack, J. Effect of motilin receptor activation on food intake and food timing. Am. J. Clin. Nutr. 2018, 107, 537–543. [Google Scholar] [CrossRef]
- Tack, J.; Verbeure, W.; Mori, H.; Schol, J.; Van den Houte, K.; Huang, I.H.; Balsiger, L.; Broeders, B.; Colomier, E.; Scarpellini, E.; et al. The gastrointestinal tract in hunger and satiety signalling. United Eur. Gastroenterol. J. 2021, 9, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Moura-Assis, A.; Friedman, J.M.; Velloso, L.A. Gut-to-brain signals in feeding control. American journal of physiology. Endocrinol. Metab. 2021, 320, E326–E332. [Google Scholar]
- Deloose, E.; Vos, R.; Janssen, P.; Van den Bergh, O.; Van Oudenhove, L.; Depoortere, I.; Tack, J. The motilin receptor agonist erythromycin stimulates hunger and food intake through a cholinergic pathway. Am. J. Clin. Nutr. 2016, 103, 730–737. [Google Scholar] [CrossRef]
- Asakawa, A.; Inui, A.; Momose, K.; Ueno, N.; Fujino, M.A.; Kasuga, M. Motilin increases food intake in mice. Peptides 1998, 19, 987–990. [Google Scholar] [CrossRef]
- Delgado, M.J.; Cerdá-Reverter, J.M.; Soengas, J.L. Hypothalamic Integration of Metabolic, Endocrine, and Circadian Signals in Fish: Involvement in the Control of Food Intake. Front. Neurosci. 2017, 11, 354. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.S.; Begg, D.P. The regulation of food intake by insulin in the central nervous system. J. Neuroendocrinol. 2021, 33, E12952. [Google Scholar] [CrossRef]
- Bunner, W.; Landry, T.; Laing, B.T.; Li, P.; Rao, Z.; Yuan, Y.; Huang, H. ARC(AgRP/NPY) Neuron Activity Is Required for Acute Exercise-Induced Food Intake in Un-Trained Mice. Front. Physiol. 2020, 11, 411. [Google Scholar] [CrossRef] [PubMed]
- Kihara, M.; Kaiya, H.; Hirai, Y.; Katayama, H.; Terao, A.; Nishikawa, M. Salmon acyl-ghrelin increases food intake and reduces doxorubicin-induced myocardial apoptosis in rats, likely by anti-oxidative activity. Peptides 2021, 137, 170471. [Google Scholar] [CrossRef] [PubMed]
- Shainer, I.; Buchshtab, A.; Hawkins, T.A.; Wilson, S.W.; Cone, R.D.; Gothilf, Y. Novel hypophysiotropic AgRP2 neurons and pineal cells revealed by BAC transgenesis in zebrafish. Sci. Rep. 2017, 7, 44777. [Google Scholar] [CrossRef]
- Kwon, E.; Jo, Y.H. Activation of the ARC(POMC)→MeA Projection Reduces Food Intake. Front. Neural Circuits 2020, 14, 595783. [Google Scholar] [CrossRef]
- Soengas, J.L. Integration of Nutrient Sensing in Fish Hypothalamus. Front. Neurosci. 2021, 15, 653928. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, Y.; Tang, N.; Qi, J.; Wu, Y.; Hao, J.; Wang, S.; Chen, D.; Li, Z. One evidence of cocaine- and amphetamine-regulated transcript (CART) has the bidirectional effects on appetite in Siberian sturgeon (Acipenser baerii). Fish Physiol. Biochem. 2018, 44, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Liu, Q.; Zhang, X.; Wu, Y.; Zhu, J.; Qi, J.; Tang, N.; Wang, S.; Wang, H.; Chen, D.; et al. The evidence of apelin has the bidirectional effects on feeding regulation in Siberian sturgeon (Acipenser baerii). Peptides 2017, 94, 78–85. [Google Scholar] [CrossRef]
- Kalananthan, T.; Murashita, K.; Rønnestad, I.; Ishigaki, M.; Takahashi, K.; Silva, M.S.; Wakabayashi, Y.; Lai, F.; Shimizu, M.; Nilsen, T.O.; et al. Hypothalamic agrp and pomc mRNA Responses to Gastrointestinal Fullness and Fasting in Atlantic Salmon (Salmo salar, L.). Front. Physiol. 2020, 11, 61. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, X.; Lai, J.; Liu, Y.; Song, M.; Li, F.; Gong, Q. Molecular characterization and tissue distribution of cholecystokinin and its receptor in Yangtze sturgeon (Acipenser dabryanus) and their response to different feeding conditions. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2022, 265, 111129. [Google Scholar] [CrossRef] [PubMed]
- Larsson, T.A.; Larson, E.T.; Fredriksson, R.; Conlon, J.M.; Larhammar, D. Characterization of NPY receptor subtypes Y2 and Y7 in rainbow trout Oncorhynchus mykiss. Peptides 2006, 27, 1320–1327. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.; Jia, J.; Yang, G.; Wang, D.; Sun, C.; Li, W. Duplication of neuropeptide Y and peptide YY in Nile tilapia Oreochromis niloticus and their roles in food intake regulation. Peptides 2017, 88, 97–105. [Google Scholar] [CrossRef]
- Tolås, I.; Kalananthan, T.; Gomes, A.S.; Lai, F.; Norland, S.; Murashita, K.; Rønnestad, I. Regional Expression of npy mRNA Paralogs in the Brain of Atlantic Salmon (Salmo salar, L.) and Response to Fasting. Front. Physiol. 2021, 12, 720639. [Google Scholar] [CrossRef]











| Primer Name | Sequences (5′–3′) | TM (°C) | Useage |
|---|---|---|---|
| Motilin-F | AGCAAACAGCACAAGGAAAC | 56 | Clone |
| Motilin-R | ATGTATCTCTCTGGAGCGGC | ||
| Motilin-F1 | GACACGATGGTCCACGGCAAGGT | 62 | 3′ RACE |
| Motilin-F2 | GATGGAGAAGGAGAAGAGCAAGACGG | ||
| Motilin receptor-F | ATGCACCCGACCAGATCTCAGACC | 68 | Clone |
| Motilin receptor-R | TTAAACGCCTGTGCTGGTCTCGGT | ||
| Motilin-qF | GCCTCTTGGTGGTGTGTTTGATG | 63.5 | RT-qPCR |
| Motilin-qR | ACTTCTTCCCCGTCTTGCTCTTCT | ||
| Motilin receptor-qF | GTTTATCGTTGGCGTCACTGGGA | 63.5 | RT-qPCR |
| Motilin receptor-qR | GGTCAAAGGGCAGGCAAAGGAAT | ||
| POMC-qF | AGCACCACCCTTAGCGTTCT | 59 | RT-qPCR |
| POMC-qR | ACCTCTTGTCATCCCGCCT | ||
| NPY-qF | GCTGGCTACCGTGGCTTTC | 59 | RT-qPCR |
| NPY-qR | GACTGGACCTCTTCCCATACCT | ||
| CART-qF | CGACTGTGGTTGAGAGCCG | 55.7 | RT-qPCR |
| CART-qR | GACAGTCACACAACTTGCCGAT | ||
| AgRP-qF | AGGCTGTGCGTCTCAGTGTC | 55.7 | RT-qPCR |
| AgRP-qR | GAATCGGAAGTCCTGTATCGG | ||
| Apelin-qF | CAGACACGCTGTTTTACACCAC | 59 | RT-qPCR |
| Apelin-qR | GCACAGCATGGACACCAAGAT | ||
| NUCB2-qF | TGGAGACAGACCAGCATTTCAG | 55.7 | RT-qPCR |
| NUCB2-qR | GGCTCCGTAACCTGTTCACTTC | ||
| NPFF-qF | GCGGATGAGCGGGTAATGACT | 63.5 | RT-qPCR |
| NPFF-qR | GACTCTACCTGCTCCTCGCCG | ||
| CCK-qF | GAGGGTAGTCCTGTAGCATCTGA | 62.3 | RT-qPCR |
| CCK-qR | TTCTACCAGACGAGCCTTTCC | ||
| Ghrelin-qF | CCAAGGTGACACGTCGAGATTC | 63.5 | RT-qPCR |
| Ghrelin-qR | TCCTGATACTGAGATTCTGACATTGAG | ||
| Gastrin-qF | GAGTTTCGTCAAGGTATGCGTGT | 63.5 | RT-qPCR |
| Gastrin-qR | GCAGCCAGTGTCTTCTCCCG | ||
| EF-1α-qF | ATGTTCACAATGGCAGCGTC | 60 | RT-qPCR |
| EF-1α-qR | AAGATTGACCGTCGTTCCG | ||
| β-actin-qF | GCCCCACCTGAGCGTAAAT | 60 | RT-qPCR |
| β-actin-qR | TCCTGCTTGCTGATCCACAT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, N.; Li, Y.; Li, Y.; Xu, S.; Wang, M.; Wang, B.; Liu, Y.; Zhang, S.; Wu, H.; Zhang, X.; et al. Motilin, a Novel Orexigenic Factor, Involved in Feeding Regulation in Yangtze Sturgeon (Acipenser dabryanus). Biomolecules 2024, 14, 433. https://doi.org/10.3390/biom14040433
Tang N, Li Y, Li Y, Xu S, Wang M, Wang B, Liu Y, Zhang S, Wu H, Zhang X, et al. Motilin, a Novel Orexigenic Factor, Involved in Feeding Regulation in Yangtze Sturgeon (Acipenser dabryanus). Biomolecules. 2024; 14(4):433. https://doi.org/10.3390/biom14040433
Chicago/Turabian StyleTang, Ni, Ya Li, Yingzi Li, Shaoqi Xu, Mei Wang, Bin Wang, Yanling Liu, Shupeng Zhang, Hongwei Wu, Xin Zhang, and et al. 2024. "Motilin, a Novel Orexigenic Factor, Involved in Feeding Regulation in Yangtze Sturgeon (Acipenser dabryanus)" Biomolecules 14, no. 4: 433. https://doi.org/10.3390/biom14040433
APA StyleTang, N., Li, Y., Li, Y., Xu, S., Wang, M., Wang, B., Liu, Y., Zhang, S., Wu, H., Zhang, X., Zhou, B., & Li, Z. (2024). Motilin, a Novel Orexigenic Factor, Involved in Feeding Regulation in Yangtze Sturgeon (Acipenser dabryanus). Biomolecules, 14(4), 433. https://doi.org/10.3390/biom14040433





