Modeling Epithelial Homeostasis and Perturbation in Three-Dimensional Human Esophageal Organoids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture and 3D Organoid Culture
2.2. IL-13 and UAB30 Treatments
2.3. Histology, Immunohistochemistry and Immunofluorescence
2.4. Immunoblotting
2.5. Quantitative Reverse-Transcription Polymerase Chain Reaction (qRT-PCR)
2.6. Statistical Analyses
3. Results
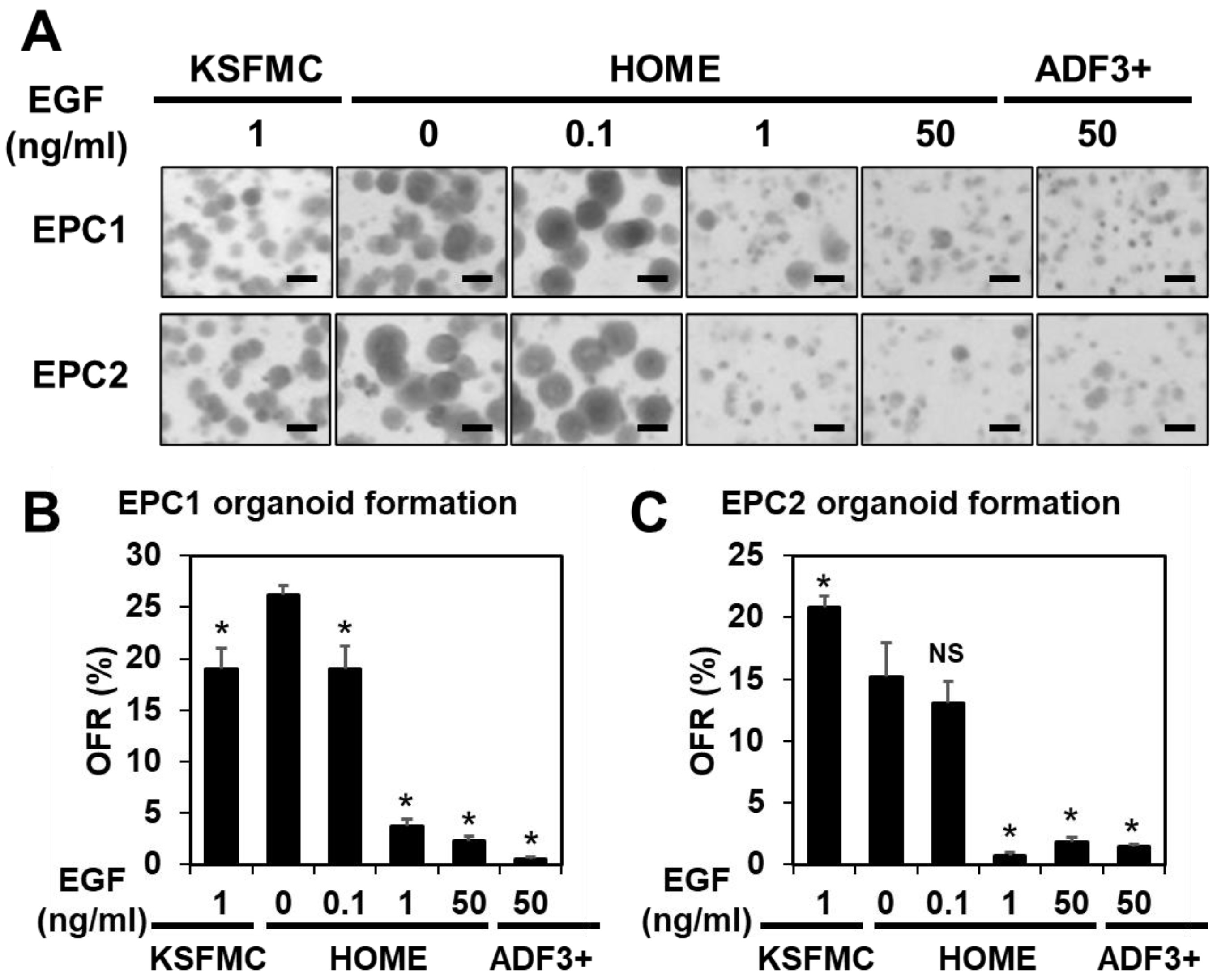
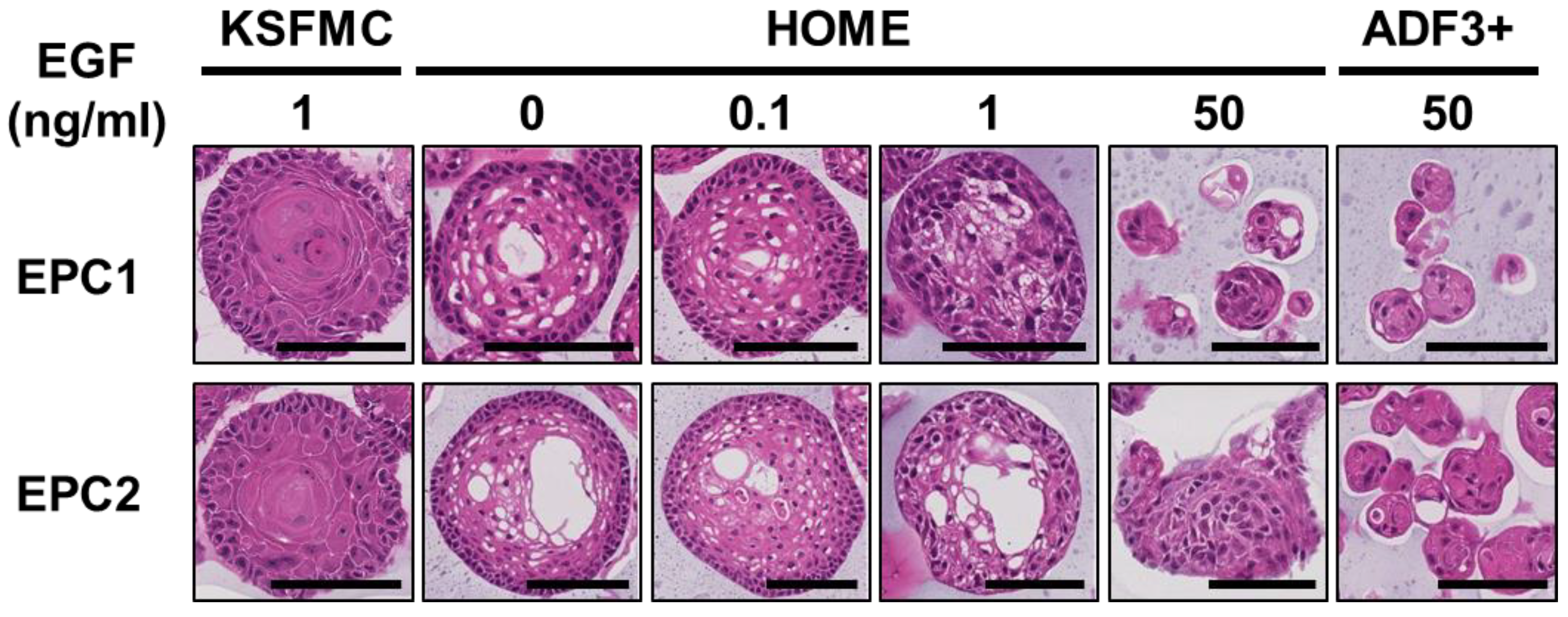
3.1. Exogenous EGF Is Dispensable for Normal Human Esophageal Organoid Growth in ADF-Based Medium
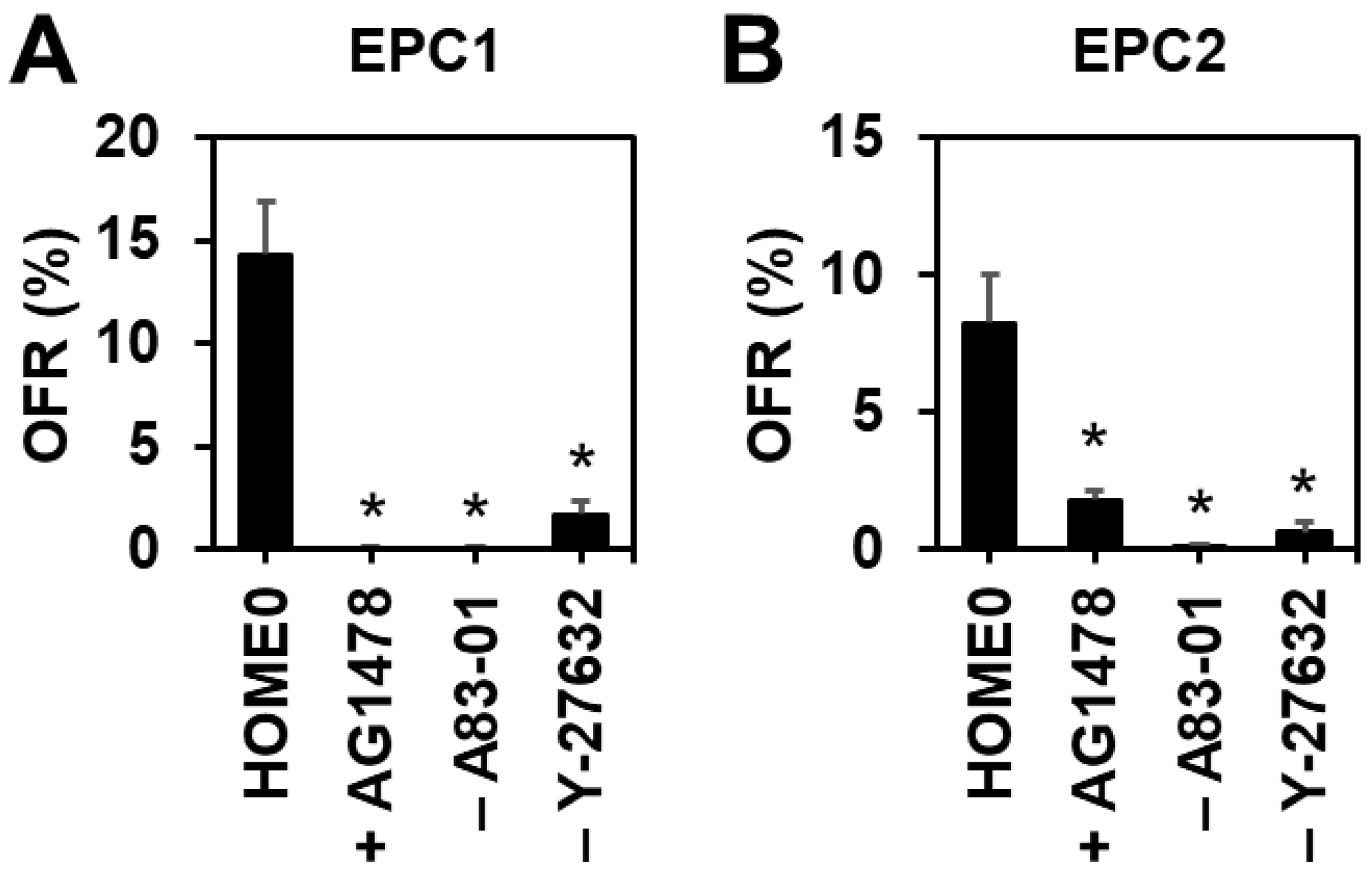
3.2. Inhibition of TGFβ Receptor-Mediated Signaling or Rho-Associated Kinase (ROCK) Increases Organoid Formation Efficiency
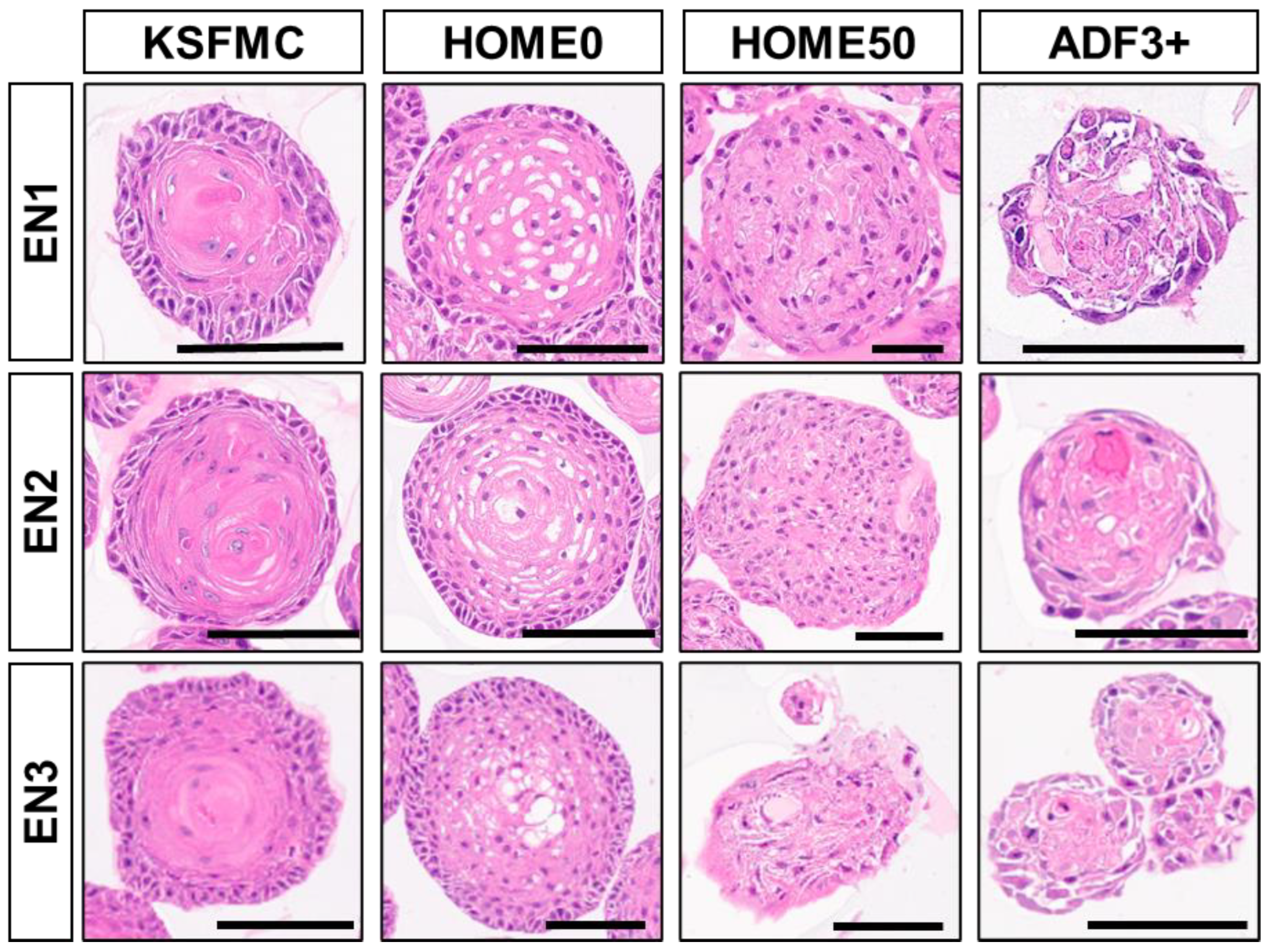
3.3. HOME0 Permits Normal Esophageal Organoid Formation from Patient Biopsies
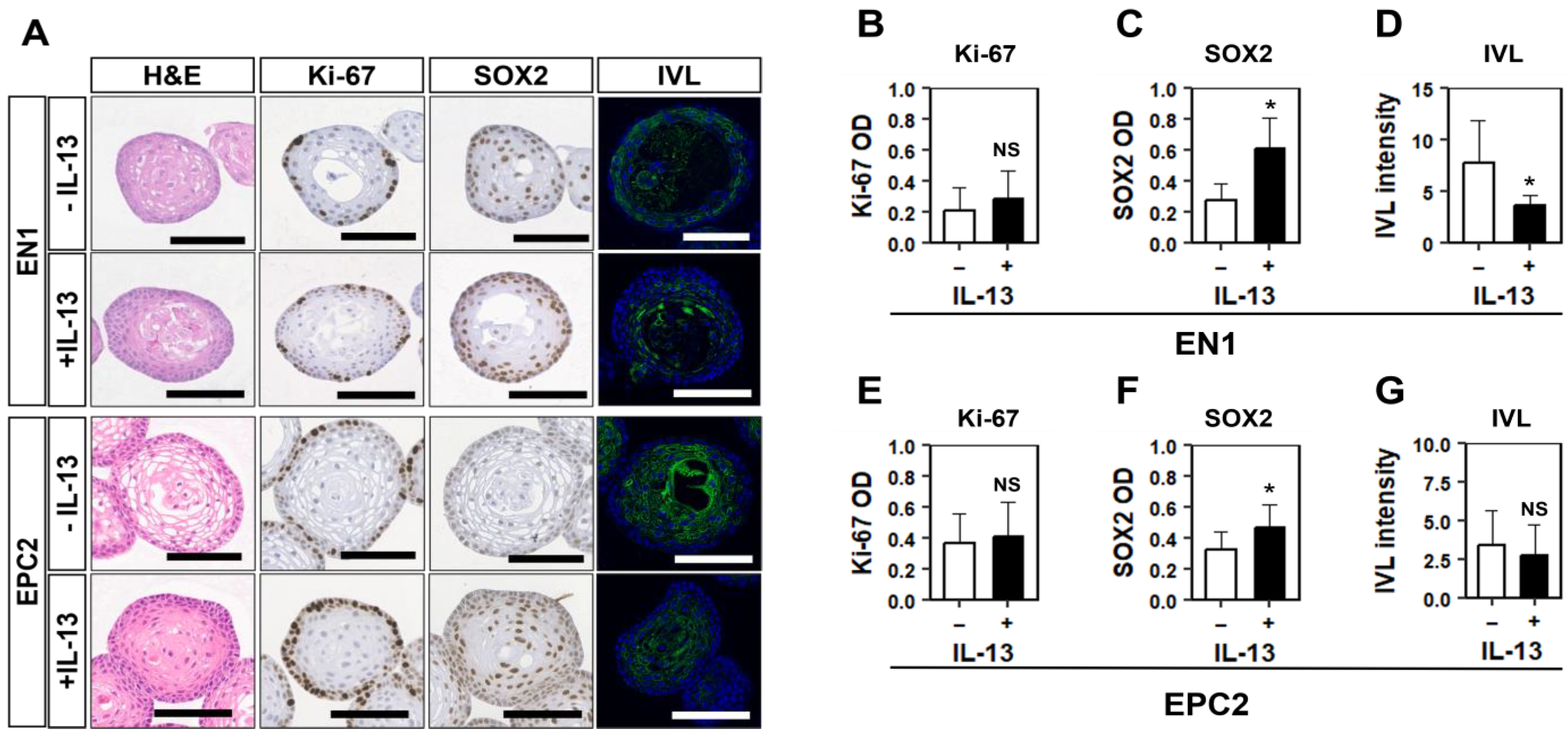
3.4. IL-13 Induces BCH-like Changes in Normal Esophageal Organoids Formed in HOME0
3.5. RXR Stimulation Induces BCH-like Changes in Normal Esophageal Organoids Formed in HOME0
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pan, Q.; Nicholson, A.M.; Barr, H.; Harrison, L.A.; Wilson, G.D.; Burkert, J.; Jeffery, R.; Alison, M.R.; Looijenga, L.; Lin, W.R.; et al. Identification of lineage-uncommitted, long-lived, label-retaining cells in healthy human esophagus and stomach, and in metaplastic esophagus. Gastroenterology 2013, 144, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Barbera, M.; di Pietro, M.; Walker, E.; Brierley, C.; MacRae, S.; Simons, B.D.; Jones, P.H.; Stingl, J.; Fitzgerald, R.C. The human squamous oesophagus has widespread capacity for clonal expansion from cells at diverse stages of differentiation. Gut 2015, 64, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Shipley, G.D.; Pittelkow, M.R. Control of growth and differentiation in vitro of human keratinocytes cultured in serum-free medium. Arch. Dermatol. 1987, 123, 1541a–1544a. [Google Scholar] [CrossRef] [PubMed]
- Rodeck, U.; Jost, M.; Kari, C.; Shih, D.T.; Lavker, R.M.; Ewert, D.L.; Jensen, P.J. EGF-R dependent regulation of keratinocyte survival. J. Cell Sci. 1997, 110 Pt 2, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Marchese, C.; Rubin, J.; Ron, D.; Faggioni, A.; Torrisi, M.R.; Messina, A.; Frati, L.; Aaronson, S.A. Human keratinocyte growth factor activity on proliferation and differentiation of human keratinocytes: Differentiation response distinguishes KGF from EGF family. J. Cell Physiol. 1990, 144, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Pittelkow, M.R.; Cook, P.W.; Shipley, G.D.; Derynck, R.; Coffey, R.J., Jr. Autonomous growth of human keratinocytes requires epidermal growth factor receptor occupancy. Cell Growth Differ. 1993, 4, 513–521. [Google Scholar]
- Cook, P.W.; Mattox, P.A.; Keeble, W.W.; Shipley, G.D. Inhibition of autonomous human keratinocyte proliferation and amphiregulin mitogenic activity by sulfated polysaccharides. In Vitro Cell Dev. Biol. 1992, 28, 218–222. [Google Scholar] [CrossRef]
- Garach-Jehoshua, O.; Ravid, A.; Liberman, U.A.; Koren, R. 1,25-Dihydroxyvitamin D3 increases the growth-promoting activity of autocrine epidermal growth factor receptor ligands in keratinocytes. Endocrinology 1999, 140, 713–721. [Google Scholar] [CrossRef]
- Piepkorn, M.; Lo, C.; Plowman, G. Amphiregulin-dependent proliferation of cultured human keratinocytes: Autocrine growth, the effects of exogenous recombinant cytokine, and apparent requirement for heparin-like glycosaminoglycans. J. Cell Physiol. 1994, 159, 114–120. [Google Scholar] [CrossRef]
- Coffey, R.J., Jr.; Derynck, R.; Wilcox, J.N.; Bringman, T.S.; Goustin, A.S.; Moses, H.L.; Pittelkow, M.R. Production and auto-induction of transforming growth factor-alpha in human keratinocytes. Nature 1987, 328, 817–820. [Google Scholar] [CrossRef]
- DeWard, A.D.; Cramer, J.; Lagasse, E. Cellular heterogeneity in the mouse esophagus implicates the presence of a nonquiescent epithelial stem cell population. Cell Rep. 2014, 9, 701–711. [Google Scholar] [CrossRef]
- Gibbs, S.; Backendorf, C.; Ponec, M. Regulation of keratinocyte proliferation and differentiation by all-trans-retinoic acid, 9-cis-retinoic acid and 1,25-dihydroxy vitamin D3. Arch. Dermatol. Res. 1996, 288, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.H.; Vivero, M.; Gudas, L.J. Overexpression of CRABPI in suprabasal keratinocytes enhances the proliferation of epidermal basal keratinocytes in mouse skin topically treated with all-trans retinoic acid. Exp. Cell Res. 2008, 314, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Kolev, V.; Mandinova, A.; Guinea-Viniegra, J.; Hu, B.; Lefort, K.; Lambertini, C.; Neel, V.; Dummer, R.; Wagner, E.F.; Dotto, G.P. EGFR signalling as a negative regulator of Notch1 gene transcription and function in proliferating keratinocytes and cancer. Nat. Cell Biol. 2008, 10, 902–911. [Google Scholar] [CrossRef]
- Rangarajan, A.; Talora, C.; Okuyama, R.; Nicolas, M.; Mammucari, C.; Oh, H.; Aster, J.C.; Krishna, S.; Metzger, D.; Chambon, P.; et al. Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J. 2001, 20, 3427–3436. [Google Scholar] [CrossRef]
- Jiang, M.; Ku, W.Y.; Zhou, Z.; Dellon, E.S.; Falk, G.W.; Nakagawa, H.; Wang, M.L.; Liu, K.; Wang, J.; Katzka, D.A.; et al. BMP-driven NRF2 activation in esophageal basal cell differentiation and eosinophilic esophagitis. J. Clin. Investig. 2015, 125, 1557–1568. [Google Scholar] [CrossRef] [PubMed]
- Rochman, M.; Travers, J.; Miracle, C.E.; Bedard, M.C.; Wen, T.; Azouz, N.P.; Caldwell, J.M.; Kc, K.; Sherrill, J.D.; Davis, B.P.; et al. Profound loss of esophageal tissue differentiation in patients with eosinophilic esophagitis. J. Allergy Clin. Immunol. 2017, 140, 738–749.e733. [Google Scholar] [CrossRef]
- Schoepfer, A.M.; Simko, A.; Bussmann, C.; Safroneeva, E.; Zwahlen, M.; Greuter, T.; Biedermann, L.; Vavricka, S.; Godat, S.; Reinhard, A.; et al. Eosinophilic Esophagitis: Relationship of Subepithelial Eosinophilic Inflammation with Epithelial Histology, Endoscopy, Blood Eosinophils, and Symptoms. Am. J. Gastroenterol. 2018, 113, 348–357. [Google Scholar] [CrossRef]
- Cheng, E.; Souza, R.F.; Spechler, S.J. Tissue remodeling in eosinophilic esophagitis. Am. J. Physiol.-Gastrointest. Liver Physiol. 2012, 303, G1175–G1187. [Google Scholar] [CrossRef]
- Flashner, S.; Martin, C.; Matsuura, N.; Shimonosono, M.; Tomita, Y.; Morimoto, M.; Okolo, O.; Yu, V.X.; Parikh, A.S.; Klein-Szanto, A.J.P.; et al. Modeling Oral-Esophageal Squamous Cell Carcinoma in 3D Organoids. J. Vis. Exp. 2022, 190, e64676. [Google Scholar] [CrossRef]
- Karakasheva, T.A.; Kijima, T.; Shimonosono, M.; Maekawa, H.; Sahu, V.; Gabre, J.T.; Cruz-Acuna, R.; Giroux, V.; Sangwan, V.; Whelan, K.A.; et al. Generation and Characterization of Patient-Derived Head and Neck, Oral, and Esophageal Cancer Organoids. Curr. Protoc. Stem Cell Biol. 2020, 53, e109. [Google Scholar] [CrossRef] [PubMed]
- Kasagi, Y.; Chandramouleeswaran, P.M.; Whelan, K.A.; Tanaka, K.; Giroux, V.; Sharma, M.; Wang, J.; Benitez, A.J.; DeMarshall, M.; Tobias, J.W.; et al. The Esophageal Organoid System Reveals Functional Interplay Between Notch and Cytokines in Reactive Epithelial Changes. Cell Mol. Gastroenterol. Hepatol. 2018, 5, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Driehuis, E.; Kretzschmar, K.; Clevers, H. Establishment of patient-derived cancer organoids for drug-screening applications. Nat. Protoc. 2020, 15, 3380–3409. [Google Scholar] [CrossRef] [PubMed]
- Tiriac, H.; Belleau, P.; Engle, D.D.; Plenker, D.; Deschenes, A.; Somerville, T.D.D.; Froeling, F.E.M.; Burkhart, R.A.; Denroche, R.E.; Jang, G.H.; et al. Organoid Profiling Identifies Common Responders to Chemotherapy in Pancreatic Cancer. Cancer Discov. 2018, 8, 1112–1129. [Google Scholar] [CrossRef]
- Seppala, T.T.; Zimmerman, J.W.; Suri, R.; Zlomke, H.; Ivey, G.D.; Szabolcs, A.; Shubert, C.R.; Cameron, J.L.; Burns, W.R.; Lafaro, K.J.; et al. Precision Medicine in Pancreatic Cancer: Patient-Derived Organoid Pharmacotyping Is a Predictive Biomarker of Clinical Treatment Response. Clin. Cancer Res. 2022, 28, 3296–3307. [Google Scholar] [CrossRef]
- Compadre, A.J.; van Biljon, L.N.; Valentine, M.C.; Llop-Guevara, A.; Graham, E.; Fashemi, B.; Herencia-Ropero, A.; Kotnik, E.N.; Cooper, I.; Harrington, S.P.; et al. RAD51 Foci as a Biomarker Predictive of Platinum Chemotherapy Response in Ovarian Cancer. Clin. Cancer Res. 2023, 29, 2466–2479. [Google Scholar] [CrossRef]
- Ooft, S.N.; Weeber, F.; Dijkstra, K.K.; McLean, C.M.; Kaing, S.; van Werkhoven, E.; Schipper, L.; Hoes, L.; Vis, D.J.; van de Haar, J.; et al. Patient-derived organoids can predict response to chemotherapy in metastatic colorectal cancer patients. Sci. Transl. Med. 2019, 11, eaay2574. [Google Scholar] [CrossRef]
- Millen, R.; De Kort, W.W.B.; Koomen, M.; van Son, G.J.F.; Gobits, R.; Penning de Vries, B.; Begthel, H.; Zandvliet, M.; Doornaert, P.; Raaijmakers, C.P.J.; et al. Patient-derived head and neck cancer organoids allow treatment stratification and serve as a tool for biomarker validation and identification. Med 2023, 4, 290–310.e212. [Google Scholar] [CrossRef]
- Ji, S.; Feng, L.; Fu, Z.; Wu, G.; Wu, Y.; Lin, Y.; Lu, D.; Song, Y.; Cui, P.; Yang, Z.; et al. Pharmaco-proteogenomic characterization of liver cancer organoids for precision oncology. Sci. Transl. Med. 2023, 15, eadg3358. [Google Scholar] [CrossRef]
- Sato, T.; Stange, D.E.; Ferrante, M.; Vries, R.G.; Van Es, J.H.; Van den Brink, S.; Van Houdt, W.J.; Pronk, A.; Van Gorp, J.; Siersema, P.D.; et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 2011, 141, 1762–1772. [Google Scholar] [CrossRef]
- Kagawa, S.; Natsuizaka, M.; Whelan, K.A.; Facompre, N.; Naganuma, S.; Ohashi, S.; Kinugasa, H.; Egloff, A.M.; Basu, D.; Gimotty, P.A.; et al. Cellular senescence checkpoint function determines differential Notch1-dependent oncogenic and tumor-suppressor activities. Oncogene 2015, 34, 2347–2359. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, H.; Kasagi, Y.; Karakasheva, T.A.; Hara, T.; Aaron, B.; Shimonosono, M.; Kijima, T.; Giroux, V.; Bailey, D.; Wilkins, B.; et al. Modeling Epithelial Homeostasis and Reactive Epithelial Changes in Human and Murine Three-Dimensional Esophageal Organoids. Curr. Protoc. Stem Cell Biol. 2020, 52, e106. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Nakagawa, H.; Oyama, K.; Takaoka, M.; Andl, C.D.; Jacobmeier, B.; von Werder, A.; Enders, G.H.; Opitz, O.G.; Rustgi, A.K. Telomerase induces immortalization of human esophageal keratinocytes without p16INK4a inactivation. Mol. Cancer Res. 2003, 1, 729–738. [Google Scholar] [PubMed]
- Whelan, K.A.; Merves, J.F.; Giroux, V.; Tanaka, K.; Guo, A.; Chandramouleeswaran, P.M.; Benitez, A.J.; Dods, K.; Que, J.; Masterson, J.C.; et al. Autophagy mediates epithelial cytoprotection in eosinophilic oesophagitis. Gut 2017, 66, 1197–1207. [Google Scholar] [CrossRef]
- Harada, K.; Sakamoto, N. Cancer organoid applications to investigate chemotherapy resistance. Front. Mol. Biosci. 2022, 9, 1067207. [Google Scholar] [CrossRef]
- Ma, H.C.; Zhu, Y.J.; Zhou, R.; Yu, Y.Y.; Xiao, Z.Z.; Zhang, H.B. Lung cancer organoids, a promising model still with long way to go. Crit. Rev. Oncol. Hematol. 2022, 171, 103610. [Google Scholar] [CrossRef]
- Xu, H.; Jiao, Y.; Qin, S.; Zhao, W.; Chu, Q.; Wu, K. Organoid technology in disease modelling, drug development, personalized treatment and regeneration medicine. Exp. Hematol. Oncol. 2018, 7, 30. [Google Scholar] [CrossRef]
- Birk, D.; Gansauge, F.; Gansauge, S.; Formentini, A.; Lucht, A.; Beger, H.G. Serum and correspondent tissue measurements of epidermal growth factor (EGF) and epidermal growth factor receptor (EGF-R). Clinical relevance in pancreatic cancer and chronic pancreatitis. Int. J. Pancreatol. 1999, 25, 89–96. [Google Scholar] [CrossRef]
- Ohashi, S.; Natsuizaka, M.; Wong, G.S.; Michaylira, C.Z.; Grugan, K.D.; Stairs, D.B.; Kalabis, J.; Vega, M.E.; Kalman, R.A.; Nakagawa, M.; et al. Epidermal growth factor receptor and mutant p53 expand an esophageal cellular subpopulation capable of epithelial-to-mesenchymal transition through ZEB transcription factors. Cancer Res. 2010, 70, 4174–4184. [Google Scholar] [CrossRef]
- Driehuis, E.; Kolders, S.; Spelier, S.; Lohmussaar, K.; Willems, S.M.; Devriese, L.A.; de Bree, R.; de Ruiter, E.J.; Korving, J.; Begthel, H.; et al. Oral Mucosal Organoids as a Potential Platform for Personalized Cancer Therapy. Cancer Discov. 2019, 9, 852–871. [Google Scholar] [CrossRef]
- Chen, S.; Crawford, M.; Day, R.M.; Briones, V.R.; Leader, J.E.; Jose, P.A.; Lechleider, R.J. RhoA modulates Smad signaling during transforming growth factor-beta-induced smooth muscle differentiation. J. Biol. Chem. 2006, 281, 1765–1770. [Google Scholar] [CrossRef]
- Ji, H.; Tang, H.; Lin, H.; Mao, J.; Gao, L.; Liu, J.; Wu, T. Rho/Rock cross-talks with transforming growth factor-beta/Smad pathway participates in lung fibroblast-myofibroblast differentiation. Biomed. Rep. 2014, 2, 787–792. [Google Scholar] [CrossRef]
- Jacobse, J.; Brown, R.; Revetta, F.; Vaezi, M.; Buendia, M.A.; Williams, C.S.; Higginbotham, T.; Washington, M.K.; Goettel, J.; Hiremath, G.; et al. A synthesis and subgroup analysis of the eosinophilic esophagitis tissue transcriptome. J. Allergy Clin. Immunol. 2023, 153, 759–771. [Google Scholar] [CrossRef]
- Shoda, T.; Morita, H.; Nomura, I.; Ishimura, N.; Ishihara, S.; Matsuda, A.; Matsumoto, K.; Kinoshita, Y. Comparison of gene expression profiles in eosinophilic esophagitis (EoE) between Japan and Western countries. Allergol. Int. 2015, 64, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Voorhees, J.J. Molecular mechanisms of retinoid actions in skin. FASEB J. 1996, 10, 1002–1013. [Google Scholar] [CrossRef] [PubMed]
- Melo, N.; Belyaeva, O.V.; Berger, W.K.; Halasz, L.; Yu, J.; Pilli, N.; Yang, Z.; Klyuyeva, A.V.; Elmets, C.A.; Atigadda, V.; et al. Next-generation retinoid X receptor agonists increase ATRA signaling in organotypic epithelium cultures and have distinct effects on receptor dynamics. J. Biol. Chem. 2023, 299, 102746. [Google Scholar] [CrossRef] [PubMed]
- Chapellier, B.; Mark, M.; Messaddeq, N.; Calleja, C.; Warot, X.; Brocard, J.; Gerard, C.; Li, M.; Metzger, D.; Ghyselinck, N.B.; et al. Physiological and retinoid-induced proliferations of epidermis basal keratinocytes are differently controlled. EMBO J. 2002, 21, 3402–3413. [Google Scholar] [CrossRef]
- Xiao, J.H.; Feng, X.; Di, W.; Peng, Z.H.; Li, L.A.; Chambon, P.; Voorhees, J.J. Identification of heparin-binding EGF-like growth factor as a target in intercellular regulation of epidermal basal cell growth by suprabasal retinoic acid receptors. EMBO J. 1999, 18, 1539–1548. [Google Scholar] [CrossRef]
- Rittie, L.; Varani, J.; Kang, S.; Voorhees, J.J.; Fisher, G.J. Retinoid-induced epidermal hyperplasia is mediated by epidermal growth factor receptor activation via specific induction of its ligands heparin-binding EGF and amphiregulin in human skin in vivo. J. Investig. Dermatol. 2006, 126, 732–739. [Google Scholar] [CrossRef]
- Kimura, R.; Iwamoto, R.; Mekada, E. Soluble form of heparin-binding EGF-like growth factor contributes to retinoic acid-induced epidermal hyperplasia. Cell Struct. Funct. 2005, 30, 35–42. [Google Scholar] [CrossRef]
- Bottenstein, J.E.; Sato, G.H. Growth of a rat neuroblastoma cell line in serum-free supplemented medium. Proc. Natl. Acad. Sci. USA 1979, 76, 514–517. [Google Scholar] [CrossRef]
- Chen, Y.; Stevens, B.; Chang, J.; Milbrandt, J.; Barres, B.A.; Hell, J.W. NS21: Re-defined and modified supplement B27 for neuronal cultures. J. Neurosci. Methods 2008, 171, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, M.; Smith, C.E.; Mashiba, M.K.; Okawa, T.; Andl, C.D.; El-Deiry, W.S.; Nakagawa, H. EGF-mediated regulation of IGFBP-3 determines esophageal epithelial cellular response to IGF-I. Am. J. Physiol.-Gastrointest. Liver Physiol. 2006, 290, G404–G416. [Google Scholar] [CrossRef] [PubMed]
- Chapman, S.; Liu, X.; Meyers, C.; Schlegel, R.; McBride, A.A. Human keratinocytes are efficiently immortalized by a Rho kinase inhibitor. J. Clin. Investig. 2010, 120, 2619–2626. [Google Scholar] [CrossRef]
- Hennings, H.; Michael, D.; Cheng, C.; Steinert, P.; Holbrook, K.; Yuspa, S.H. Calcium regulation of growth and differentiation of mouse epidermal cells in culture. Cell 1980, 19, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Boyce, S.T.; Ham, R.G. Calcium-regulated differentiation of normal human epidermal keratinocytes in chemically defined clonal culture and serum-free serial culture. J. Investig. Dermatol. 1983, 81, 33s–40s. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Kawahira, N.; Nakashima, Y.; Nakano, H.; Iwase, A.; Uchijima, Y.; Wang, M.; Wu, S.M.; Minamisawa, S.; Kurihara, H.; et al. Notch and retinoic acid signals regulate macrophage formation from endocardium downstream of Nkx2-5. Nat. Commun. 2023, 14, 5398. [Google Scholar] [CrossRef]

| Abbreviation | Name |
|---|---|
| AI | Artificial Intelligence |
| BCH | basal cell hyperplasia |
| BMP | bone morphogenetic proteins |
| DAPI | 4′,6-diamidino-2-phenylindole |
| DMSO | Dimethyl sulfoxide |
| EoE | eosinophilic esophagitis |
| H&E | Hematoxylin and Eosin |
| IF | immunofluorescence |
| IHC | immunohistochemistry |
| IL | interleukin |
| IVL | Involucrin |
| OFR | Organoid formation rate |
| PBS | phosphate-buffered saline |
| PDL | population doubling level |
| PDO | Patient-derived organoids |
| qRT-PCR | quantitative reverse-transcription polymerase chain reaction |
| RAR | retinoic acid receptor |
| RXR | retinoic-X receptor |
| 3D | three-dimensional |
| TGF-β | transforming growth factor-beta |
| Abbreviation | Name |
|---|---|
| ADF | advanced Dulbecco’s Modified Eagle Medium/Ham’s F-12 |
| ADF3+ | advanced DMEM +/+/+ |
| CM | conditioned medium |
| CM-NR | CM containing Noggin and R-Spondin1 |
| EGF | epidermal growth factor |
| FGF2 | fibroblast growth factor 2 |
| FGF10 | fibroblast growth factor 10 |
| HB-EGF | heparin-binding EGF-like growth factor |
| HOME | human organoid medium containing EGF at indicated concentrations |
| HOME0 | human organoid medium containing 0 ng/mL EGF |
| HOME0.1 | human organoid medium containing 0.1 ng/mL EGF |
| HOME1 | human organoid medium containing 1 ng/mL EGF |
| HOME10 | human organoid medium containing 10 ng/mL EGF |
| HOME0 ΔA | HOME0 devoid of A83-01 |
| KSFM | keratinocyte serum-free medium |
| KSFMC | KSFM medium containing 0.6 mM Ca2+ |
| MOM | murine organoid medium |
| Name | ADF3+ | HOME | HOME | MOM | KSFMC |
|---|---|---|---|---|---|
| Base medium | ADF | ADF | ADF | ADF | KSFM |
| B27 | (+) | (+) | (+) | (+) | U |
| N2 | (−) | (+) | (+) | (+) | U |
| NAC | (+) | (+) | (+) | (+) | U |
| CM-NR | (+) | (+) | (+) | (+) | U |
| EGF (ng/mL) | 50 † | 0 or 50 † | Various † (0–50) | 50 †† | 1 † |
| A83-01 (µM) | 0.5 | 0 | 5 | 0 | U |
| Nicotinamide | (+) | (−) | (−) | (−) | U |
| FGF110 | (+) | (−) | (−) | (−) | U |
| FGF2 | (+) | (−) | (−) | (−) | U |
| CHI99021 | (+) | (−) | (−) | (−) | U |
| Forskolin | (+) | (−) | (−) | (−) | U |
| Prostaglandin E2 | (+) | (−) | (−) | (−) | U |
| CaCl2 (mM) | 1 | 1 | 1 | 1 | 0.6 |
| Y-27632 | (+) | (+) | (+) | (+) | (+) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shimonosono, M.; Morimoto, M.; Hirose, W.; Tomita, Y.; Matsuura, N.; Flashner, S.; Ebadi, M.S.; Okayasu, E.H.; Lee, C.Y.; Britton, W.R.; et al. Modeling Epithelial Homeostasis and Perturbation in Three-Dimensional Human Esophageal Organoids. Biomolecules 2024, 14, 1126. https://doi.org/10.3390/biom14091126
Shimonosono M, Morimoto M, Hirose W, Tomita Y, Matsuura N, Flashner S, Ebadi MS, Okayasu EH, Lee CY, Britton WR, et al. Modeling Epithelial Homeostasis and Perturbation in Three-Dimensional Human Esophageal Organoids. Biomolecules. 2024; 14(9):1126. https://doi.org/10.3390/biom14091126
Chicago/Turabian StyleShimonosono, Masataka, Masaki Morimoto, Wataru Hirose, Yasuto Tomita, Norihiro Matsuura, Samuel Flashner, Mesra S. Ebadi, Emilea H. Okayasu, Christian Y. Lee, William R. Britton, and et al. 2024. "Modeling Epithelial Homeostasis and Perturbation in Three-Dimensional Human Esophageal Organoids" Biomolecules 14, no. 9: 1126. https://doi.org/10.3390/biom14091126






