A Landauer Formula for Bioelectronic Applications
Abstract
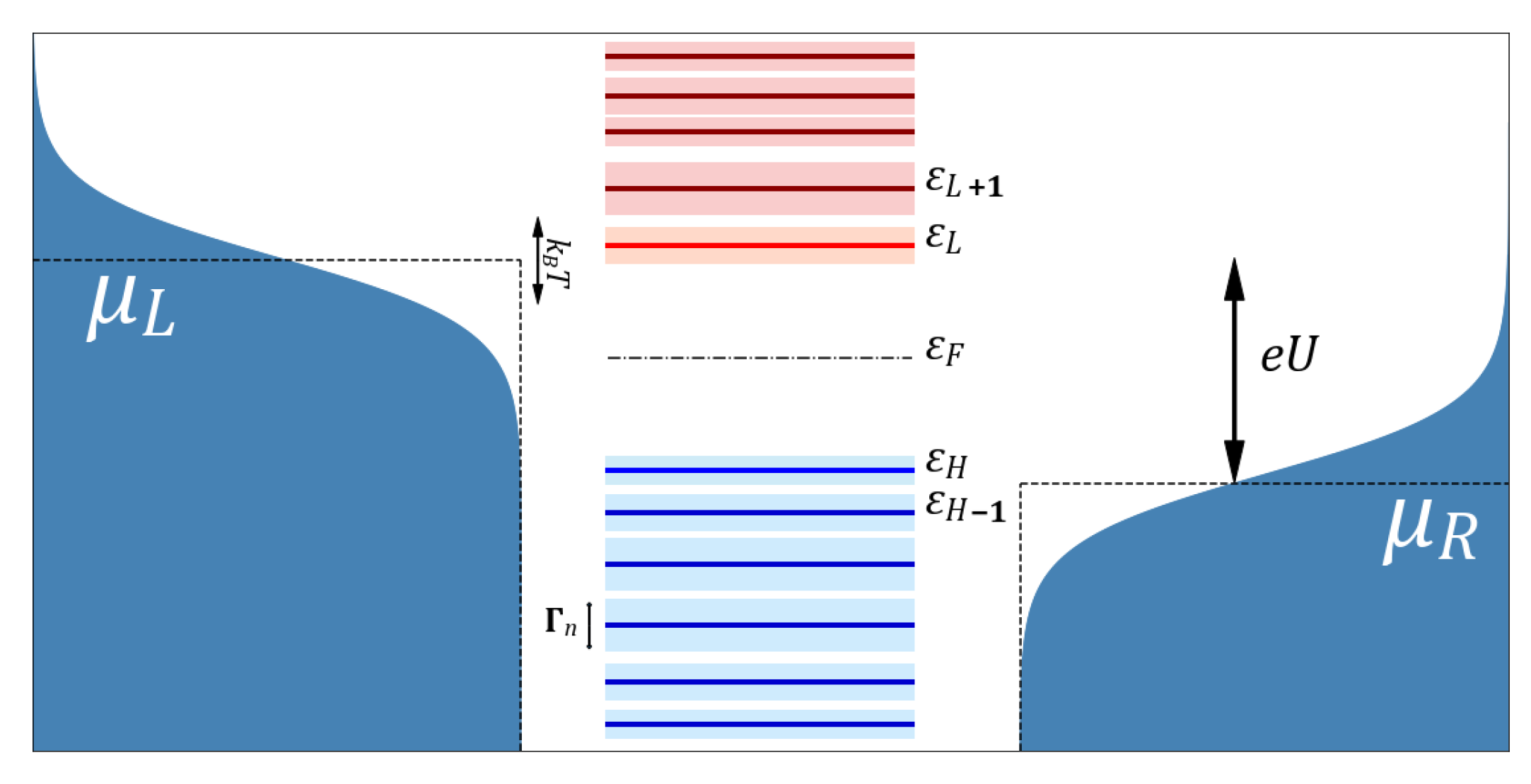
:1. Introduction
- The conductance remains nearly constant when temperature is changed from tens of Kelvins to ambient temperatures [10].
2. Derivation of a Landauer Formula for Bioelectronics
3. Electron Transfer
4. Weak Contacts
5. Strong Contacts
5.1. Strong and Weak Contact Mixed
5.2. Strong Decoherence
5.3. Temperature Dependence
5.4. Distance Dependence
5.5. Order of Magnitude
5.6. Electron Transfer at Strong Decoherence
6. Experiments
6.1. Temperature Dependence
6.2. Distance Dependence of Electron Transfer and Conductance
6.3. Distribution of Conductance
7. Discussion
8. Materials and Methods
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
Appendix C
Appendix D
References
- Bostick, C.D.; Mukhopadhyay, S.; Pecht, I.; Sheves, M.; Cahen, D.; Lederman, D. Protein bioelectronics: A review of what we do and do not know. Rep. Prog. Phys. 2018, 81, 026601. [Google Scholar] [CrossRef] [PubMed]
- Amdursky, N.; Marchak, D.; Sepunaru, L.; Pecht, I.; Sheves, M.; Cahen, D. Electronic transport via proteins. Adv. Mater. 2014, 26, 7142–7161. [Google Scholar] [CrossRef] [PubMed]
- Kaldor, N. Capital accumulation and economic growth. In The Theory of Capital; Springer: Berlin/Heidelberg, Germany, 1961; pp. 177–222. [Google Scholar]
- Artés, J.M.; Díez-Pérez, I.; Gorostiza, P. Transistor-like behavior of single metalloprotein junctions. Nano Lett. 2011, 12, 2679–2684. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Song, W.; Pang, P.; Zhao, Y.; Zhang, P.; Csabai, I.; Vattay, G.; Lindsay, S. Observation of giant conductance fluctuations in a protein. Nano Futur. 2017, 1, 035002. [Google Scholar] [CrossRef]
- Zhang, B.; Song, W.; Pang, P.; Lai, H.; Chen, Q.; Zhang, P.; Lindsay, S. Role of contacts in long-range protein conductance. Proc. Natl. Acad. Sci. USA 2019, 116, 5886–5891. [Google Scholar] [CrossRef] [Green Version]
- Yoo, S.Y.; Lee, T.; Chung, Y.H.; Min, J.; Choi, J.W. Fabrication of biofilm in nanoscale consisting of cytochrome f/2-MAA bilayer on Au surface for bioelectronic devices by self-assembly technique. J. Nanosci. Nanotechnol. 2011, 11, 7069–7072. [Google Scholar] [CrossRef] [PubMed]
- Korpany, K.V.; Langat, P.; Kim, D.M.; Edelman, N.; Cooper, D.R.; Nadeau, J.; Blum, A.S. Conductance switching in the photoswitchable protein dronpa. J. Am. Chem. Soc. 2012, 134, 16119–16122. [Google Scholar] [CrossRef]
- Zhang, B.; Lindsay, S. Electronic Decay Length in a Protein Molecule. Nano Lett. 2019, 6, 4017–4022. [Google Scholar] [CrossRef]
- Sepunaru, L.; Pecht, I.; Sheves, M.; Cahen, D. Solid-state electron transport across azurin: From a temperature-independent to a temperature-activated mechanism. J. Am. Chem. Soc. 2011, 133, 2421–2423. [Google Scholar] [CrossRef]
- Lambert, C. Basic concepts of quantum interference and electron transport in single-molecule electronics. Chem. Soc. Rev. 2015, 44, 875–888. [Google Scholar] [CrossRef]
- Marcus, R.A. On the theory of oxidation-reduction reactions involving electron transfer. I. J. Chem. Phys. 1956, 24, 966–978. [Google Scholar] [CrossRef]
- Segal, D.; Nitzan, A.; Davis, W.B.; Wasielewski, M.R.; Ratner, M.A. Electron transfer rates in bridged molecular systems 2. A steady-state analysis of coherent tunneling and thermal transitions. J. Phys. Chem. B 2000, 104, 3817–3829. [Google Scholar] [CrossRef]
- Nitzan, A. Electron transmission through molecules and molecular interfaces. Annu. Rev. Phys. Chem. 2001, 52, 681–750. [Google Scholar] [CrossRef] [PubMed]
- Nitzan, A. A relationship between electron-transfer rates and molecular conduction. J. Phys. Chem. A 2001, 105, 2677–2679. [Google Scholar] [CrossRef]
- Sowa, J.K.; Mol, J.A.; Briggs, G.A.D.; Gauger, E.M. Beyond Marcus theory and the Landauer-Büttiker approach in molecular junctions: A unified framework. J. Chem. Phys. 2018, 149, 154112. [Google Scholar] [CrossRef] [PubMed]
- Sarovar, M.; Ishizaki, A.; Fleming, G.R.; Whaley, K.B. Quantum entanglement in photosynthetic light-harvesting complexes. Nat. Phys. 2010, 6, 462. [Google Scholar] [CrossRef]
- Engel, G.S.; Calhoun, T.R.; Read, E.L.; Ahn, T.K.; Mančal, T.; Cheng, Y.C.; Blankenship, R.E.; Fleming, G.R. Evidence for wavelike energy transfer through quantum coherence in photosynthetic systems. Nature 2007, 446, 782. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, M.; Rebentrost, P.; Lloyd, S.; Aspuru-Guzik, A. Environment-assisted quantum walks in photosynthetic energy transfer. J. Chem. Phys. 2008, 129, 11B603. [Google Scholar] [CrossRef]
- Zahid, F.; Paulsson, M.; Datta, S. Electrical conduction through molecules. In Advanced Semiconductor and Organic Nano-Techniques; Elsevier: Amsterdam, The Netherlands, 2003; pp. 1–41. [Google Scholar]
- Datta, S. Electronic Transport in Mesoscopic Systems; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Breuer, H.P.; Petruccione, F. The Theory of Open Quantum Systems; Oxford University Press on Demand: New York, NY, USA, 2002. [Google Scholar]
- Lever, G.; Cole, D.J.; Hine, N.D.; Haynes, P.D.; Payne, M.C. Electrostatic considerations affecting the calculated HOMO–LUMO gap in protein molecules. J. Phys. Condens. Matter 2013, 25, 152101. [Google Scholar] [CrossRef]
- Nitzan, A. The relationship between electron transfer rate and molecular conduction 2. The sequential hopping case. Isr. J. Chem. 2002, 42, 163–166. [Google Scholar] [CrossRef]
- Rebentrost, P.; Mohseni, M.; Kassal, I.; Lloyd, S.; Aspuru-Guzik, A. Environment-assisted quantum transport. New J. Phys. 2009, 11, 033003. [Google Scholar] [CrossRef]
- Raichlin, S.; Pecht, I.; Sheves, M.; Cahen, D. Protein electronic conductors: Hemin–substrate bonding dictates transport mechanism and efficiency across myoglobin. Angew. Chem. 2015, 127, 12556–12560. [Google Scholar] [CrossRef]
- Amdursky, N.; Ferber, D.; Bortolotti, C.A.; Dolgikh, D.A.; Chertkova, R.V.; Pecht, I.; Sheves, M.; Cahen, D. Solid-state electron transport via cytochrome c depends on electronic coupling to electrodes and across the protein. Proc. Natl. Acad. Sci. USA 2014, 111, 5556–5561. [Google Scholar] [CrossRef] [PubMed]
- Wierzbinski, E.; Venkatramani, R.; Davis, K.L.; Bezer, S.; Kong, J.; Xing, Y.; Borguet, E.; Achim, C.; Beratan, D.N.; Waldeck, D.H. The single-molecule conductance and electrochemical electron-transfer rate are related by a power law. ACS Nano 2013, 7, 5391–5401. [Google Scholar] [CrossRef]
- Wolak, M.A.; Balaeff, A.; Gutmann, S.; Helmrich, H.J.; Vosloo, R.; Beerbom, M.M.; Wierzbinski, E.; Waldeck, D.H.; Bezer, S.; Achim, C.; et al. Electronic structure of self-assembled peptide nucleic acid thin films. J. Phys. Chem. C 2011, 115, 17123–17135. [Google Scholar] [CrossRef]
- Szent-Gyorgyi, A. The study of energy—Levels in biochemistry. Nature 1941, 148, 157. [Google Scholar] [CrossRef]



| in | in | |
|---|---|---|
| Native m-Mb | ||
| apo Mb | ||
| Reconstituted m-Mb |
| Energy in eV | |
|---|---|
| LUMO+1 | −8.9052 |
| LUMO | −9.5185 |
| HOMO | −9.9637 |
| HOMO-1 | −10.0282 |
| in | in | in eV | |
|---|---|---|---|
| Covalent binding (E104C) | |||
| Electrostatic binding (WT) |
| Energy in eV | |
|---|---|
| LUMO+1 | −9.9457 |
| LUMO | −9.9252 |
| HOMO | −9.6021 |
| HOMO-1 | −9.0685 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papp, E.; Jelenfi, D.P.; Veszeli, M.T.; Vattay, G. A Landauer Formula for Bioelectronic Applications. Biomolecules 2019, 9, 599. https://doi.org/10.3390/biom9100599
Papp E, Jelenfi DP, Veszeli MT, Vattay G. A Landauer Formula for Bioelectronic Applications. Biomolecules. 2019; 9(10):599. https://doi.org/10.3390/biom9100599
Chicago/Turabian StylePapp, Eszter, Dávid P. Jelenfi, Máté T. Veszeli, and Gábor Vattay. 2019. "A Landauer Formula for Bioelectronic Applications" Biomolecules 9, no. 10: 599. https://doi.org/10.3390/biom9100599






