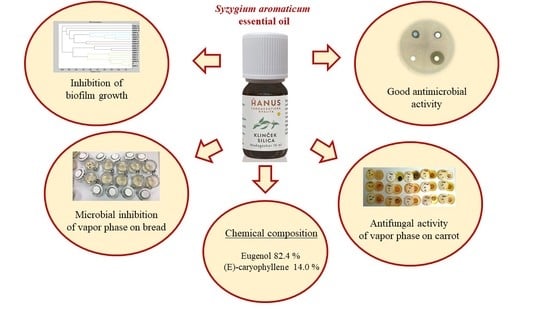
Chemical Composition, In Vitro and In Situ Antimicrobial and Antibiofilm Activities of Syzygium aromaticum (Clove) Essential Oil
Abstract
:1. Introduction
2. Results
2.1. Chemical Composition of S. aromaticum EO
2.2. Antimicrobial Activity of S. aromaticum EO
2.3. Antimicrobial Analysis In Situ Using a Food Model
2.4. Analysis of Biofilm Developmental Phases and Evaluation of Molecular Differences on Different Surfaces Using MALDI-TOF MS Biotyper
3. Discussion
4. Materials and Methods
4.1. Essential Oil
4.2. Chemical Characterization of Essential Oil Samples by Gas Chromatography/Mass Spectrometry (GC/MS) and Gas Chromatography (GC-FID)
4.3. Tested Microorganisms
4.4. Antimicrobial Activity: Disc Diffusion Method
4.5. Minimum Inhibitory Concentrations (MIC)
4.6. Antimicrobial Analysis In Situ on a Food Model
4.7. Analysis of Differences in Biofilm Development with MALDI-TOF MS Biotyper
4.8. Statistical Data Evaluation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, J.; Baghotia, A.; Goel, S.P. Eugenia caryophyllata Thunberg (Family Myrtaceae): A review. Int. J. Res. Pharm. Biomed. Sci. 2012, 3, 1469–1475. [Google Scholar]
- Lee, S.; Najiah, M.; Wendy, W.; Nadirah, M. Chemical composition and antimicrobial activity of the essential oil of Syzygium aromaticum flower bud (Clove) against fish systemic bacteria isolated from aquaculture sites. Front. Agric. China 2009, 3, 332–336. [Google Scholar] [CrossRef]
- Chaieb, K.; Zmantar, T.; Ksouri, R.; Hajlaoui, H.; Mahdouani, K.; Abdelly, C.; Bakhrouf, A. Antioxidant properties of the essential oil of Eugenia caryophyllata and its antifungal activity against a large number of clinical Candida species. Mycoses 2007, 50, 403–406. [Google Scholar] [CrossRef]
- Shobana, S.; Naidu, K.A. Antioxidant activity of selected Indian spices. Prostaglandins Leukot. Essent. Fat. Acids (PLEFA) 2000, 62, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Soliman, K.M.; Badeaa, R.I. Effect of oil extracted from some medicinal plants on different mycotoxigenic fungi. Food Chem. Toxicol. 2002, 40, 1669–1675. [Google Scholar] [CrossRef]
- Budri, P.E.; Silva, N.C.C.; Bonsaglia, E.C.R.; Júnior, A.F.; Júnior, J.P.A.; Doyama, J.T.; Gonçalves, J.L.; Santos, M.V. Effect of essential oils of Syzygium aromaticum and Cinnamomum zeylanicum and their major components on biofilm production in Staphylococcus aureus strains isolated from milk of cows with mastitis. J. Dairy Sci. 2015, 98, 5899–5904. [Google Scholar] [CrossRef] [PubMed]
- Rajkowska, K.; Nowicka-Krawczyk, P.; Kunicka-Styczyńska, A. Effect of clove and thyme essential oils on Candida biofilm formation and the oil distribution in yeast cells. Molecules 2019, 24, 1954. [Google Scholar] [CrossRef] [Green Version]
- Adil, M.; Singh, K.; Verma, P.K.; Khan, A.U. Eugenol-induced suppression of biofilm-forming genes in Streptococcus mutans: An approach to inhibit biofilms. J. Glob. Antimicrob. Resist. 2014, 2, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, J.D.F.; Paroul, N.; Czyewski, E.; Lerin, L.; Rotava, I.; Cansian, R.L.; Mossi, A.; Toniazzo, G.; de Oliveira, D.; Treichel, H. Chemical composition and antioxidant and antibacterial activities of clove essential oil (Eugenia caryophyllata Thunb). Rev. Ceres 2018, 57, 589–594. [Google Scholar] [CrossRef]
- Radünz, M.; da Trindade, M.L.M.; Camargo, T.M.; Radünz, A.L.; Dellinghausen Borges, C.; Gandra, E.A.; Helbig, E. Antimicrobial and antioxidant activity of unencapsulated and encapsulated clove (Syzygium aromaticum, L.) essential oil. Food Chem. 2019, 276, 180–186. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Zhu, X.; Cao, P.; Wei, S.; Lu, Y. Antibacterial and antibiofilm activities of eugenol from essential oil of Syzygium aromaticum (L.) merr. & L. M. Perry (clove) leaf against periodontal pathogen Porphyromonas gingivalis. Microb. Pathog. 2017, 113, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Ebani, V.V.; Najar, B.; Bertelloni, F.; Pistelli, L.; Mancianti, F.; Nardoni, S. Chemical composition and in vitro antimicrobial efficacy of sixteen essential oils against Escherichia coli and Aspergillus fumigatus isolated from poultry. J. Vet. Sci. 2018, 5, 62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade, B.F.M.T.; Nunes Barbosa, L.; Probst, I.P.; Fernandes Júnior, A. Antimicrobial activity of essential oils. J. Essent. Oil Res. 2017, 26, 34–40. [Google Scholar] [CrossRef]
- Besten, M.A.; Jasinski, V.C.; Costa, Â.D.G.; Nunes, D.S.; Sens, S.L.; Wisniewski, A., Jr.; Simionatto, E.L.; Riva, D.; Dalmarco, J.B.; Granato, D. Chemical composition similarity between the essential oils isolated from male and female specimens of each five Baccharis species. J. Braz. Chem. Soc. 2020, 23, 1041–1047. [Google Scholar] [CrossRef] [Green Version]
- Hu, Q.; Zhou, M.; Wei, S. Progress on the Antimicrobial Activity Research of Clove Oil and Eugenol in the Food Antisepsis Field. J. Food Sci. 2018, 83, 1476–1483. [Google Scholar] [CrossRef] [Green Version]
- Teles, A.M.; Silva-Silva, J.V.; Fernandes, J.M.P.; Abreu-Silva, A.L.; Calabrese, K.D.S.; Mendes Filho, N.E.; Mouchrek, A.N.; Almeida-Souza, F. GC-MS Characterization of Antibacterial, Antioxidant, and Antitrypanosomal Activity of Syzygium aromaticum Essential Oil and Eugenol. Evid. Based Complement Alternat. Med. 2021, 2021, 6663255. [Google Scholar] [CrossRef]
- Lalami, A.E.; Moukhafi, K.; Bouslamti, R.; Lairini, S. Evaluation of antibacterial and antioxidant effects of cinnamon and clove essential oils from Madagascar. Mater. Today Proc. 2019, 13, 762–770. [Google Scholar] [CrossRef]
- Cimanga, K.; Kambu, K.; Tona, L.; Apers, S.; De Bruyne, T.; Hermans, N.; Totté, J.; Pieters, L.; Vlietinck, A.J. Correlation between chemical composition and antibacterial activity of essential oils of some aromatic medicinal plants growing in the democratic republic of Congo. J. Ethnopharmacol. 2020, 79, 213–220. [Google Scholar] [CrossRef]
- Gislene, G.F.; Paulo, C.; Giuliana, L. Antibacterial activity of plant extracts and phytochemicals on antibiotic resistant bacteria. Braz. J. Microbiol. 2000, 31, 314–325. [Google Scholar] [CrossRef]
- Hili, P.; Evans, C.S.; Veness, R.G. Antimicrobial action of essential oils: The effect of dimethylsulphoxide on the activity of cinnamon oil. Lett. Appl. Microbiol. 1997, 24, 269–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nzeako, B.C.; Al-Kharousi, Z.S.; Al-Mahrooqui, Z. Antimicrobial activities of clove and thyme extracts. Sultan Qaboos Univ. Med. J. 2006, 6, 33–39. [Google Scholar] [PubMed]
- Thosar, N.; Basak, S.; Bahadure, R.N.; Rajurkar, M. Antimicrobial efficacy of five essential oils against oral pathogens: An in vitro study. Eur. J. Dent. 2013, 07, S071–S077. [Google Scholar] [CrossRef] [Green Version]
- Fu, Y.; Zu, Y.; Chen, L.; Shi, X.; Wang, Z.; Sun, Z.; Effert, T. Antimicrobial activity of clove and rosemary essential oils alone and in combination. Phytother. Res. 2007, 21, 989–994. [Google Scholar] [CrossRef]
- Nuñez, L.; D’Aquino, M. Microbicide activity of clove essential oil (Eugenia caryophyllata). Braz. J. Microbiol. 2012, 43, 1255–1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.G.; Liu, T.; Hu, Q.P.; Cao, X.M. Chemical composition, antibacterial properties and mechanism of action of essential oil from clove buds against Staphylococcus aureus. Molecules 2016, 21, 1194. [Google Scholar] [CrossRef]
- Panizzi, L.; Falmini, G.; Cioni, P.L.; Morelli, I. Composition and antimicrobial properties of essential oil of four Mediterranean Lamiaceae. J. Ethnopharm. 1993, 39, 167–170. [Google Scholar] [CrossRef]
- Rana, I.S.; Rana, A.S.; Rajak, R.C. Evaluation of antifungal activity in essential oil of the Syzygium aromaticum (L.) by extraction, purification and analysis of its main component eugenol. Braz. J. Microbiol. 2011, 42, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Nunez, L.; D’aquino, M.; Chirife, J. Antifungal properties of clove oil (Eugenia caryophylata) in sugar solution. Braz. J. Microbiol. 2001, 32, 123–126. [Google Scholar] [CrossRef]
- Ahmad, N.; Alam, M.K.; Shehbaz, A.; Khan, A.; Mannan, A.; Rashid, H.S.; Bisht, B.; Owais, M. Antimicrobial activity of clove oil and its potential in the treatment of vaginal candidiasis. J. Drug Target. 2005, 13, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Vale-Silva, L.; Cavaleiro, C.; Salgueiro, L. Antifungal activity of the clove essential oil from Syzygium aromaticum on Candida, Aspergillus and dermatophyte species. J. Med. Microbiol. 2009, 58, 1454–1462. [Google Scholar] [CrossRef]
- Chami, N.; Chami, F.; Bennis, S.; Trouillas, J.; Remmal, A. Antifungal treatment with carvacrol and eugenol of oral candidiasis in immunosuppressed rats. Braz. J. Infect. Dis. 2004, 8, 217–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chee, H.Y.; Lee, M.H. Antifungal activity of clove essential oil and its volatile vapour against dermatophytic fungi. Mycobiology 2007, 35, 241–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, S.K.; Agrawal, S.C. Fungistatic activity of some perfumes against otomycotic pathogens. Mycoses 2002, 45, 88–90. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, P. Antimicrobial activity of plant essential oils using food model media, efficacy, synergistic potential and interactions with food components. Food Microbiol. 2009, 26, 142–150. [Google Scholar] [CrossRef]
- Moreira, M.R.; Ponce, A.G.; Del Valle, C.E.; Roura, S.I. Effects of clove and tea tree oils on Escherichia coli O157:H7 in blanched spinach and minced cooked beef. J. Food Process. Preserv. 2007, 31, 379–391. [Google Scholar] [CrossRef]
- Aguilar-Gonzalez, A.E.; Palou, E.; Lopez-Malo, A. Antifungal activity of essential oils of clove (Syzygium aromaticum) and/or mustard (Brassica nigra) in vapor phase against gray mold (Botrytis cinerea) in strawberries. Innov. Food Sci. Emerg. Technol. 2015, 32, 181–185. [Google Scholar] [CrossRef]
- Lindsay, D.; Brözel, V.S.; von Holy, A. Spore formation in Bacillus subtilis biofilms. J. Food Protect. 2005, 68, 860–865. [Google Scholar] [CrossRef]
- Kalemba, D.; Kunicka, A. Antibacterial and antifungal properties of essential oils. Curr. Med. Chem. 2003, 10, 813–829. [Google Scholar] [CrossRef]
- Kunicka-Styczyńska, A.; Tyfa, A.; Laskowski, D.; Plucińska, A.; Rajkowska, K.; Kowal, K. Clove Oil (Syzygium aromaticum L.) Activity Against Alicyclobacillus acidoterrestris Biofilm on Technical Surfaces. Molecules 2020, 25, 3334. [Google Scholar] [CrossRef]
- Li, T.; Wang, D.; Liu, N.; Ma, Y.; Ding, T.; Mei, Y.; Li, J. Inhibition of Quorum Sensing-Controlled Virulence Factors and Biofilm Formation in Pseudomonas fluorescens by Cinnamaldehyde. Int. J. Food Microbiol. 2018, 269, 98–106. [Google Scholar] [CrossRef]
- Kubesová, A.; Šalplachta, J.; Horká, M.; Růžička, F.; Šlais, K. Candida “Psilosis”—Electromigration Techniques and MALDI-TOF Mass Spectrometry for Phenotypical Discrimination. Analyst 2012, 137, 1937–1943. [Google Scholar] [CrossRef] [PubMed]
- Rams, T.E.; Sautter, J.D.; Getreu, A.; van Winkelhoff, A.J. Phenotypic Identification of Porphyromonas Gingivalis Validated with Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Microb. Pathog. 2016, 94, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Kačániová, M.; Terentjeva, M.; Galovičová, L.; Ivanišová, E.; Štefániková, J.; Valková, V.; Borotová, P.; Kowalczewski, P.Ł.; Kunová, S.; Felšöciová, S.; et al. Biological Activity and Antibiofilm Molecular Profile of Citrus aurantium Essential Oil and Its Application in a Food Model. Molecules 2020, 25, 3956. [Google Scholar] [CrossRef]
- Kačániová, M.; Galovičová, L.; Ivanišová, E.; Vukovic, N.L.; Štefániková, J.; Valková, V.; Borotová, P.; Žiarovská, J.; Terentjeva, M.; Felšöciová, S.; et al. Antioxidant, Antimicrobial and Antibiofilm Activity of Coriander (Coriandrum Sativum L.) Essential Oil for Its Application in Foods. Foods 2020, 9, 282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry; Allured Publishing Corporation: Carol Stream, IL, USA, 2007; ISBN 978-1-93233-11-4. [Google Scholar]
- Van Den Dool, H.; Kratz, P.D. A Generalization of the Retention Index System Including Linear Temperature Programmed Gas—Liquid Partition Chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Dao, T.; Bensoussan, M.; Gervais, P.; Dantigny, P. Inactivation of Conidia of Penicillium chrysogenum, P. digitatum and P. italicum by Ethanol Solutions and Vapours. Int. J. Food Microbiol. 2008, 122, 68–73. [Google Scholar] [CrossRef]
- Talibi, I.; Askarne, L.; Boubaker, H.; Boudyach, E.H.; Msanda, F.; Saadi, B.; Aoumar, A.A.B. Antifungal activity of some Moroccan plants against Geotrichum candidum, the causal agent of postharvest citrus sour rot. Crop Prot. 2013, 35, 41–46. [Google Scholar] [CrossRef]
- Aman, M.; Rai, V.R. Antifungal activity of fungicides and plant extracts against yellow sigatoka disease causing Mycosphaerella musicola. Curr. Res. Environ. Appl. Mycol. 2015, 5, 277–284. [Google Scholar] [CrossRef]







| No | RI a | Compound b | % c |
|---|---|---|---|
| 1 | 830 | furfural | Tr |
| 2 | 983 | 6-methyl-5-hepten-2-one | Tr |
| 3 | 1190 | methyl salicylate | Tr |
| 4 | 1193 | 2-allyl-phenol | Tr |
| 5 | 1360 | eugenol | 82.4 |
| 6 | 1379 | a-copaene | Tr |
| 7 | 1422 | (E)-caryophyllene | 14.0 |
| 8 | 1456 | a-humulene | 1.8 |
| 9 | 1519 | eugenol acetate | 0.9 |
| 10 | 1583 | caryophyllene oxide | 0.7 |
| 11 | 1755 | benzyl benzoate | Tr |
| total | 99.7 |
| Microorganism | Zone Inhibition (mm) | Activity of EO | MIC 50 (µL/mL) | MIC 90 (µL/mL) | ATB |
|---|---|---|---|---|---|
| S. enteritidis | 9.44 ± 1.01 | * | 216.23 | 265.41 | 29.00 ± 0.10 |
| P. aeruginosa | 5.22 ± 1.20 | * | 223.38 | 284.56 | 28.00 ± 0.06 |
| Y. enterocolitica | 7.67 ± 1.41 | * | 224.46 | 278.92 | 28.00 ± 0.05 |
| S. marcescens | 10.44 ± 1.01 | ** | 112.13 | 145.25 | 27.00 ± 0.02 |
| S. aureus | 15.78 ± 0.67 | *** | 85.46 | 93.35 | 29.00 ± 0.03 |
| B. subtilis | 11.22 ± 0.83 | ** | 121.13 | 138.91 | 26.00 ± 0.05 |
| E. faecalis | 4.67 ± 1.32 | * | 218.36 | 264.55 | 29.00 ± 0.03 |
| C. albicans | 8.22 ± 1.53 | * | 221.43 | 226.32 | 28.00 ± 0.06 |
| C. krusei | 9.89 ± 1.76 | * | 211.82 | 265.33 | 26.00 ± 0.12 |
| C. tropicalis | 9.33 ± 0.58 | * | 214.54 | 271.36 | 29.00 ± 0.02 |
| C. glabrata | 9.33 ± 1.00 | * | 217.22 | 264.36 | 26.00 ± 0.03 |
| Biofilm S. maltophilia | 8.89 ± 1.36 | * | 223.18 | 284.32 | 27.00 ± 0.05 |
| Biofilm B. subtilis | 10.44 ± 0.88 | ** | 103.64 | 128.64 | 26.00 ± 0.03 |
| P. commune | 16.52 ± 0.15 | *** | 74.32 | 81.26 | 25.00 ± 0.05 |
| P. expansum | 18.56 ± 0.22 | *** | 64.25 | 75.12 | 26.00 ±0.11 |
| P. glabrum | 18.32 ± 0.21 | *** | 68.41 | 78.12 | 27.00 ± 0.06 |
| P. chrysogenum | 17.36 ± 0.08 | *** | 75.11 | 86.92 | 26.00 ± 0.09 |
| Mycelial Growth Inhibition [%] | ||||
|---|---|---|---|---|
| Fungi | Concentration of EO | |||
| 62.5 µL/L | 125 µL/L | 250 µL/L | 500 µL/L | |
| P. commune | 16.41 ± 6.84 a | 45.72 ± 5.49 b | 96.22 ± 1.20 c | 98.78 ± 1.72 cd |
| P. expansum | 16.73 ± 6.38 a | 90.49 ± 12.26 b | 100.00 ± 0.00 bc | 100.00 ± 0.00 bd |
| P. glabrum | 42.91 ± 5.87 a | 54.50 ± 9.01 a | 93.42 ± 9.30 b | 100.00 ± 0.00 bc |
| P. chrysogenum | 30.31 ± 9.70 a | 12.21 ± 7.87 a | 97.90 ± 1.49 b | 99.45 ± 0.78 bc |
| Bacterial Growth Inhibition [%] | ||||
|---|---|---|---|---|
| Microorganisms | Concentration of EO | |||
| 62.5 µL/L | 125 µL/L | 250 µL/L | 500 µL/L | |
| P. chrysogenum | 16.93 ± 0.37 a | 51.04 ± 4.43 ab | 61.22 ± 15.73 bc | 93.53 ± 5.11 c |
| P. expansum | 19.43 ± 0.89 a | 59.81 ± 4.33 b | 64.98 ± 2.81 b | 75.94 ± 0.91 c |
| S. marcescens | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 49.11 ± 13.73 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kačániová, M.; Galovičová, L.; Borotová, P.; Valková, V.; Ďúranová, H.; Kowalczewski, P.Ł.; Said-Al Ahl, H.A.H.; Hikal, W.M.; Vukic, M.; Savitskaya, T.; et al. Chemical Composition, In Vitro and In Situ Antimicrobial and Antibiofilm Activities of Syzygium aromaticum (Clove) Essential Oil. Plants 2021, 10, 2185. https://doi.org/10.3390/plants10102185
Kačániová M, Galovičová L, Borotová P, Valková V, Ďúranová H, Kowalczewski PŁ, Said-Al Ahl HAH, Hikal WM, Vukic M, Savitskaya T, et al. Chemical Composition, In Vitro and In Situ Antimicrobial and Antibiofilm Activities of Syzygium aromaticum (Clove) Essential Oil. Plants. 2021; 10(10):2185. https://doi.org/10.3390/plants10102185
Chicago/Turabian StyleKačániová, Miroslava, Lucia Galovičová, Petra Borotová, Veronika Valková, Hana Ďúranová, Przemysław Łukasz Kowalczewski, Hussein A. H. Said-Al Ahl, Wafaa M. Hikal, Milena Vukic, Tatsiana Savitskaya, and et al. 2021. "Chemical Composition, In Vitro and In Situ Antimicrobial and Antibiofilm Activities of Syzygium aromaticum (Clove) Essential Oil" Plants 10, no. 10: 2185. https://doi.org/10.3390/plants10102185