Differences in Germination of ACCase-Resistant Biotypes Containing Isoleucine-1781-Leucine Mutation and Susceptible Biotypes of Wild Oat (Avena sterilis ssp. ludoviciana)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Seed Source
2.2. Whole Plant Dose Response Assay
2.3. Investigating Molecular Basis of Resistance
2.4. Germination and Seedling Growth
3. Results and Discussion
3.1. Whole Plant Dose Response Assay
3.2. Molecular Basis for Resistance
3.3. Germination and Seedling Growth
Author Contributions
Funding
Conflicts of Interest
References
- Kaundun, S.S.; Windass, J.D. Derived cleaved amplified polymorphic, a simple method to detect a key point mutation conferring acethyl CoA carboxylase inhibitor herbicide resistance in grass weeds. Weed Res. 2006, 46, 34–39. [Google Scholar] [CrossRef]
- Heap, I. The International Survey of Herbicide Resistant Weeds. Available online: http://www.weedscience.com (accessed on 22 August 2021).
- Lonhienne, T.; Garcia, M.D.; Pierens, G.; Mobli, M.; Nouwens, A.; Guddat, L.W. Structural insights into the mechanism of inhibition of AHAS by herbicides. Proc. Natl. Acad. Sci. USA 2018, 115, 1945–1954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, Q.; Collavo, A.; Zheng, M.Q.; Owen, M.; Sattin, M.; Powles, S.B. Diversity of acetyl-coenzyme A carboxylase mutations in resistant Lolium populations: Evaluation using clethodim. Plant Physiol. 2007, 145, 547–558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delye, C.; Michel, S.; Berard, A.; Chauvel, B.; Brunel, D.; Guillemin, J.P.; Le Corre, V. Geographical variation in resistance to acetyl-coenzyme A carboxylase-inhibiting herbicides across the range of the arable weed Alopecurus myosuroides (blackgrass). New Phytol. 2010, 186, 1005–1017. [Google Scholar] [CrossRef] [PubMed]
- Powles, S.B.; Yu, Q. Evolution in action: Plants resistant to herbicides. Ann. Rev. Plant. Biol. 2010, 61, 317–347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coustau, C.; Chevillon, C.; French-Constant, R. Resistance to xenobiotics and parasites: Can we count the cost? Trends Ecol. Evol. 2000, 15, 378–383. [Google Scholar] [CrossRef]
- Pavlicev, M.; Wagner, G.P. A model of developmental evolution: Selection, pleiotropy and compensation. Trends Ecol. Evolution. 2012, 27, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Marjanovic, J.; Gornicki, P. Resistance to herbicides caused by single amino acid mutations in acetyl-C o A carboxylase in resistant populations of grassy weeds. New Phytol. 2013, 197, 1110–1116. [Google Scholar] [CrossRef] [PubMed]
- Benakashani, F.; Zand, E.; Naghavi, M.R.; Sasanfar, H. Mutations in Acetyl-CoA Carboxylase enzyme, mechanism of cross-resistance in wild oat (Avena ludoviciana Deuri.) biotypes to ACCase inhibitor herbicides. Iran J. Weed Sci. 2015, 10, 179–190. [Google Scholar]
- Sasaki, Y.; Nagano, Y. Plant acetyl-CoA carboxylase: Structure, biosynthesis, regulation, and gene manipulation for plant breeding. Biosci. Biotechnol. Biochem. 2004, 68, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Kaundun, S.S. Resistance to acetyl-CoA carboxylase-inhibiting herbicides. Pest Manag. Sci. 2014, 70, 1405–1417. [Google Scholar] [CrossRef]
- Park, K.W.; Mallory-Smith, C.A. Multiple herbicide resistant in downy brome (Bromus tectorum) and its impact on fitness. Weed Sci. 2005, 53, 780–786. [Google Scholar] [CrossRef]
- Weaver, E.S.; Thomas, G. Germination responses to temperature of atrazine-resistant and -S biotype of two pigweed (Amaranthus) species. Weed Sci. 1986, 34, 865–870. [Google Scholar] [CrossRef]
- Alcocer-Ruthling, M.; Thill, D.C.; Shafii, B. Seed biology of sulfonylurea- resistant and -S biotype of prickly lettuce (Lactuca serriola). Weed Technol. 1992, 6, 858–864. [Google Scholar] [CrossRef]
- Dyer, W.E.; Chee, P.W.; Fay, P.K. Rapid germination of sulfonylurea-resistant Kochia scoparia L. accessions is associated with elevated seed levels of branched chain amino acids. Weed Sci. 1993, 41, 18–22. [Google Scholar] [CrossRef]
- Torres-Garcia, J.S.; Uscanga-Mortera, E.; Trejo, C.; Conde-Martinez, V.; Kohashi-Shibata, J.; Núñez-Farfán, J.; Martínez-Moreno, D. Effect of herbicide resistance on seed physiology of Phalaris minor (Little seed canarygrass). Bot. Sci. 2015, 93, 661–667. [Google Scholar] [CrossRef] [Green Version]
- Osipitan, O.A.; Dille, J.A. Fitness outcomes related to glyphosate resistance in kochia (Kochia scoparia): What life history stage to examine? Front. Plant Sci. 2017, 8, 1090. [Google Scholar] [CrossRef]
- Batlla, D.; Bench-Arnold, R.L. A predictive model for dormancy loss in Polygonum aviculare L. seeds based on changes in population hydrotime parameters. Seed Sci. Res. 2004, 14, 277–286. [Google Scholar] [CrossRef]
- Soltani, E.; Gruber, S.; Oveisi, M.; Salehi, N.; Alahdadi, I.; Javid, M.G. Water stress, temperature regimes, and light control secondary dormancy induction and loss in Brassica napus L. seeds. Seed Sci. Res. 2017, 27, 217–230. [Google Scholar] [CrossRef]
- Gummerson, R.J. The effect of constant temperatures and osmotic potentials on the germination of sugar beet. J. Exp. Bot. 1986, 37, 729–741. [Google Scholar] [CrossRef]
- Bradford, K.J. Applications of hydrothermal time to quantifying and modeling seed germination and dormancy. Weed Sci. 2002, 50, 248–260. [Google Scholar] [CrossRef]
- Soltani, E.; Farzaneh, S. Hydrotime analysis for determination of seed vigor in cotton. Seed Sci. Technol. 2014, 42, 260–273. [Google Scholar] [CrossRef]
- Soltani, A.; Gholipoor, M.; Zeinali, E. Seed reserve utilization and seedling growth of wheat as affected by drought and salinity. Environ. Exp. Bot. 2006, 55, 195–200. [Google Scholar] [CrossRef]
- Cheng, X.; Cheng, J.; Huang, X.; Lai, Y.; Wang, L.; Du, W.; Zhang, H. Dynamic quantitative trait loci analysis of seed reserve utilization during three germination stages in rice. PLoS ONE 2013, 8, e80002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seyyedi, S.A.; Khajeh-Hosseini, M.; Moghaddam, P.R.; Shahandeh, H. Effects of phosphorus and seed priming on seed vigor, fatty acids composition and heterotrophic seedling growth of black seed (Nigella sativa L.) grown in a calcareous soil. Ind. Crop. Prod. 2015, 74, 939–949. [Google Scholar] [CrossRef]
- Zheng, Y.L.; Ma, H.C. Effects of seed aging on seed germination and seed reserve utilization in mumian. HortTechnology 2014, 24, 471–474. [Google Scholar] [CrossRef] [Green Version]
- Castellanos-Frías, E.; García de León, D.; Pujadas-Salva, A.; Dorado, J.; Gonzalez-Andujar, J.L. Potential distribution of Avena sterilis L. in Europe under climate change. Ann. Appl. Biol. 2014, 165, 53–61. [Google Scholar] [CrossRef]
- Beckie, H.J.; Warwick, S.I.; Sauder, C.A. Basis for herbicide resistance in Canadian populations of wild oat (Avena fatua). Weed Sci. 2012, 60, 10–18. [Google Scholar] [CrossRef]
- Torner, C.; Gonzalez-Andujar, J.L.; Fernandez-Quintanilla, C. Wild oat (Avena sterilis L.) competition with winter barley: Plant density effects. Weed Res. 1991, 31, 301–307. [Google Scholar] [CrossRef]
- Vidal, R.A.; Fleck, N.G. The risk of finding herbicide resistant weed biotypes. Planta Daninha 1997, 31, 887–892. [Google Scholar]
- Zand, E.; Benakashani, F.; Alizadeh, H.M.; Soufizadeh, S.; Ramezani, K.; Maknali, S.; Fereydounpoor, M. Resistance to aryloxyphenoxypropionate herbicides in wild oat (Avena ludoviciana). Iran J. Weed Sci. 2006, 2, 17–32. [Google Scholar]
- Gherekhloo, J.; Oveisi, M.; Zand, E.; Deprado, R. A review of herbicide resistance in Iran. Weed Sci. 2016, 64, 551–561. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; ISBN 3-900051-07-0. Available online: https://www.r-project.org/ (accessed on 10 September 2021).
- Ritz, C.; Streibig, J.C. Bioassay analysis using R. J. Stat. Softw. 2005, 12, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Keshtkar, E.; Kudsk, P.; Mesgaran, M.B. Perspective: Common errors in dose–response analysis and how to avoid them. Pest Manag. Sci. 2021, 77, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Christoffers, M.J.; Berg, M.L.; Messersmith, C.G. An isoleucine to leucine mutation in acetyl-CoA carboxylase confers herbicide resistance in wild oat. Genome 2002, 45, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Michel, B.E.; Kaufmann, M.R. The osmotic potential of polyethylene glycol 6000. Plant Physiol. 1973, 51, 914–916. [Google Scholar] [CrossRef]
- Dahal, P.; Bradford, K.J. Effects of priming and endosperm integrity on seed germination rates of tomato genotypes: II Germination at reduced water potential. J. Exp. Bot. 1990, 41, 1441–1453. [Google Scholar] [CrossRef]
- Beckie, H.J.; Tardif, F.J. Herbicide cross resistance in weeds. Crop. Prot. 2012, 35, 15–28. [Google Scholar] [CrossRef]
- Torun, H.; Uygur, F.N. Determination and mapping of resistant wild oat (Avena sterilis L.) populations to most commonly used herbicides in wheat fields for Osmaniye, Turkey. Int. J. Agric. Sci. 2018, 11, 8–14. [Google Scholar]
- Beckie, H.J.; Hall, L.M.; Meers, S.; Laslo, J.J.; Stevenson, F.C. Management practices influencing herbicide resistance in wild oat. Weed Technol. 2004, 18, 853–859. [Google Scholar] [CrossRef]
- Deihimfard, R.; Zand, E. Evaluating environmental impacts of herbicides on wheat agroecosystems in the provinces of Iran using EIQ model. Environ. Sci. 2005, 6, 1–9. [Google Scholar]
- Benakashani, F.; Zand, E.; Alizadeh, H.M. Resistance of wild oat (Avena ludoviciana) biotypes to clodinafop-propargil herbicide. Appl. Entomol. Phytopathol. 2007, 74, 127–151. [Google Scholar]
- Benakashani, F.; Rahimian Mashhadi, H.; Zand, E.; Alizadeh, H.; Naghavi, M.R. Investigation of the cross resistance to ACCase inhibitor herbicides in wild oat (Avena ludoviciana Durieu.) populations from Khuzestan province and chemical control of resistant populations. Iran J. Weed Sci. 2010, 6, 95–112. [Google Scholar]
- Rastgou, M.; Rashed, M.H.; Zand, E.; Nasiri, M. Resistance of winter wild oat (Avena ludoviciana Durieu.) to aryloxyphenoxy propionate herbicides in wheat fields of Khuzestan province: First screening test. Iran J. Weed Sci. 2006, 2, 96–104. [Google Scholar]
- Zand, E.; Benakashani, F.; Baghestani, M.A.; Maknali, A.; Minbashi, M.; Soufizadeh, S. Investigating the distribution of resistant wild oat (Avena ludoviciana) populations to clodinafop-propargil herbicide in South Western. Iran. Environ. Sci. 2007, 3, 85–92. [Google Scholar]
- Delye, C. Weed resistance to acetyl coenzyme A carboxylase inhibitors: An update. Weed Sci. 2005, 53, 728–746. [Google Scholar] [CrossRef]
- Kukorelli, G.; Reisinger, P.; Pinke, G. ACCase inhibitor herbicides—selectivity, weed resistance and fitness cost: A review. Int. J. Pest Manag. 2013, 59, 165–173. [Google Scholar] [CrossRef]
- Gaines, T.A.; Duke, S.O.; Morran, S.; Rigon, C.A.G.; Tranel, P.J.; Küpper, A.; Dayan, F.E. Mechanisms of evolved herbicide resistance. J. Biol. Chem. 2020, 295, 10307–10330. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Q.; Powles, S.B. The molecular bases for resistance to acethyl co-enzyme A carboxylase (ACCase) inhibiting herbicides in two target based resistant biotypes of annual ryegrass (Lolium rigidum). Planta 2006, 223, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Delye, C.; Pernin, F.; Michel, S. ‘Universal’ PCR assays detecting mutations in acetyl-coenzyme A carboxylase or acetolactate synthase that endow herbicide resistance in grass weeds. Weed Res. 2011, 51, 353–362. [Google Scholar] [CrossRef]
- Ahmad-Hamdani, M.S.; Yu, Q.; Han, H.; Cawthray, G.R.; Wang, S.F.; Powles, S.B. Herbicide resistance endowed by enhanced rates of herbicide metabolism in wild oat (Avena spp.). Weed Sci. 2013, 61, 55–62. [Google Scholar] [CrossRef]
- Mahajan, G.; Loura, D.; Raymont, K.; Chauhan, B.S. Influence of soil moisture levels on the growth and reproductive behavior of Avena fatua and Avena ludoviciana. PLoS ONE 2020, 15, e0234648. [Google Scholar]
- Matzrafi, M.; Gerson, O.; Rubin, B.; Peleg, Z. Different Mutations Endowing Resistance to Acetyl-CoA Carboxylase Inhibitors Results in Changes in Ecological Fitness of Lolium rigidum Populations. Front. Plant Sci. 2017, 8, 1078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabet Zangeneh, H.; Mohammaddust Chamanabad, H.R.; Zand, E.; Asghari, A.; Alamisaeid, K.; Travlos, I.S.; Alebrahim, M.T. Study of fitness cost in three rigid ryegrass populations susceptible and resistant to acetyl-CoA carboxylase inhibiting herbicides. Front. Ecol. Evol. 2016, 4, 142. [Google Scholar] [CrossRef] [Green Version]



| Table. Cont. | City | Location | The History of Grass Herbicide Application (Last 5 Years before Seed Sampling) |
|---|---|---|---|
| S | Andimeshk | 32.45° N, 48.35° E | None |
| WR1 | Andimeshk | 32.45° N, 48.35° E | Clodinafop-propargyl |
| WR2 | Shush | 32.20° N, 48.25° E | Clodinafop-propargyl |
| WR3 | Ahvaz | 31.32° N, 48.67° E | Clodinafop-propargyl |
| WR4 | Andimeshk | 32.45° N, 48.35° E | Clodinafop-propargyl |
| Primer | Sequence 5′-3′ | Usage | Reference |
|---|---|---|---|
| ACCF1 | CACAGACCATGATGCAGCTC | CAPS for 2041and 2088 | [4] |
| ACCR1 | CTCCCTGGAGTTGTGCTTTC | - | |
| NsiI1781f | CTGTCTGAAGAAGACTATGGCCG | dCAPS for 1781 | [1] |
| NsiI1781r | AGAATACGCACTGGCAATAGCAGCACTTCCATGCA | - |
| Enzyme | Commercial Isoschizomers | Restriction Site | Technique | Reference |
|---|---|---|---|---|
| NsiI | AvaIII, EcoT22I, Mph11031, Zsp2I | 5′-ATGCA^T-3′ 3′-T^ACGTA-5′ | dCAPS (1781) | [1] |
| EcoRI | FunII | 5′-G^AATTC-3′ 3′-CTTAA^G-5′ | CAPS (2041) | [4] |
| EcoRV | Eco32I | 5′-GAT^ATC-3′ 3′-CTA^TAG-5′ | dCAPS (2078) | [4] |
| Eco47III | AfeI, Aor51HI, FunI | 5′-AGC^GCT-3′ | CAPS (2088) | [4] |
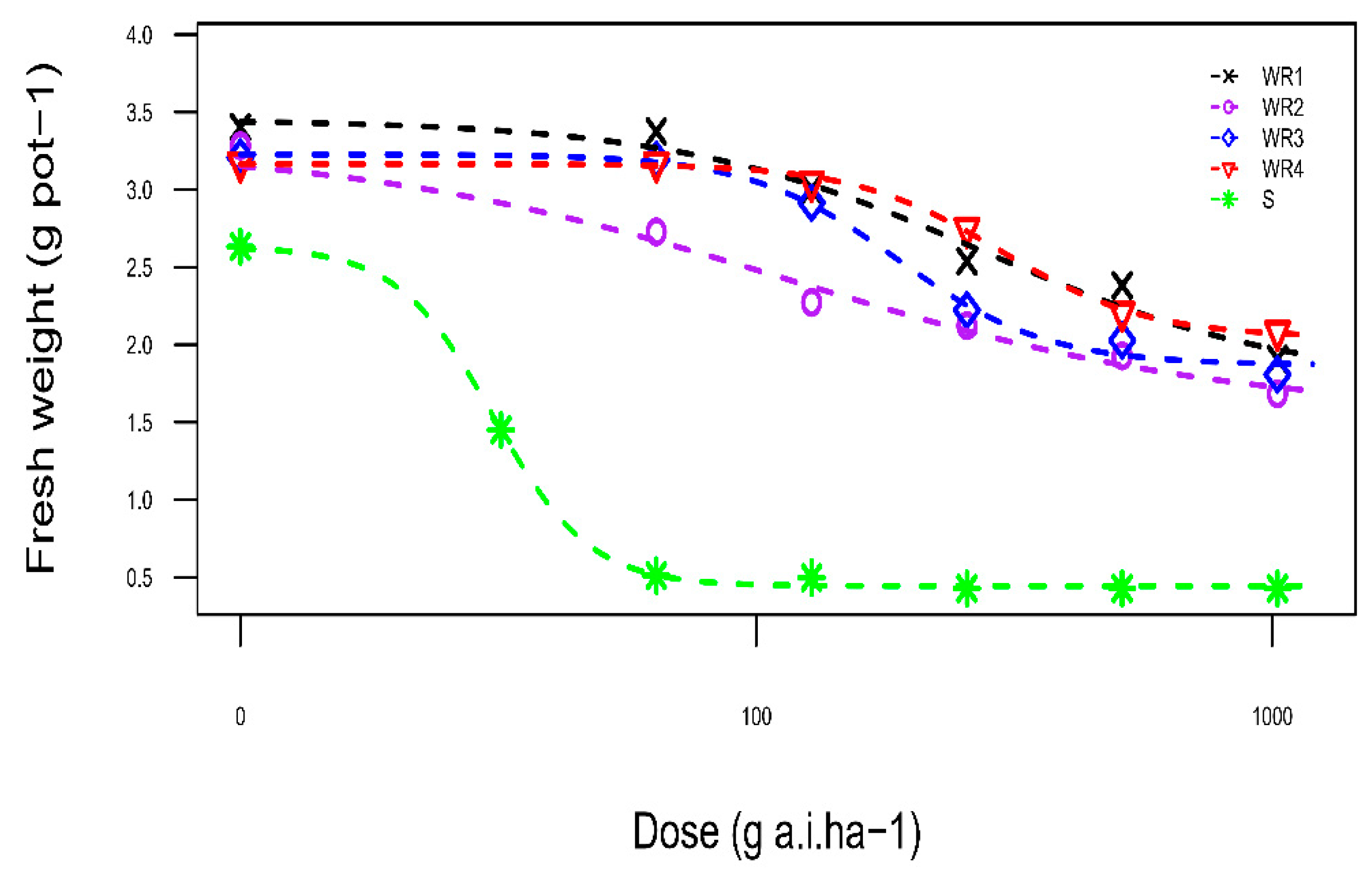
| Biotype | B | C (g.pot−1) | D (g.pot−1) | Absolute ED50 (g a.i. ha−1) | RI |
|---|---|---|---|---|---|
| WR1 | 1.45 (0.58) | 1.74 (0.42) | 3.45 (0.11) | >1024 NA | >214.22 |
| WR2 | 1.00 (0.58) | 1.55 (0.40) | 3.28 (0.13) | >1024 NA | >214.22 |
| WR3 | 3.01 (1.25) | 1.87 (0.12) | 3.23 (0.11) | >1024 NA | >214.22 |
| WR4 | 4.67 (3.13) | 2.04 (0.16) | 3.16 (0.09) | >1024 NA | >214.22 |
| S | 3.26 (2.93) | 0.44 (0.06) | 2.63 (0.13) | 1.90 | - |
| SOV | Df | ISDW | SLDW | FSDW | MSR | CEM | FSR |
|---|---|---|---|---|---|---|---|
| Experiment | 1 | 0.0198 | 0.0035 | 0.220 | 0.373 | 0.0013 | 0.0019 |
| Replication (Experiment) | 2 | 0.0228 | 0.0909 | 0.335 | 0.387 | 0.0011 | 0.0024 |
| Biotype | 4 | 64.429 ** | 31.641 ** | 51.454 ** | 15.812 ** | 1.8749 ** | 0.0911 ** |
| Water potential | 4 | 0.0004 | 70.958 ** | 114.168 ** | 114.305 ** | 0.1843 ** | 0.8008 ** |
| Bio × Exp | 4 | 0.0004 | 0.0817 | 1.032 | 1.042 | 0.0155 | 0.0068 |
| Bio × WP | 16 | 0.0008 | 3.423 ** | 6.404 ** | 6.449 ** | 0.1286 ** | 0.0416 ** |
| Exp × WP | 4 | 0.0002 | 0.0551 | 0.898 | 0.893 | 0.0027 | 0.0052 |
| Bio × Exp × WP | 16 | 0.0005 | 0.0695 | 0.544 | 0.540 | 0.0071 | 0.0032 |
| Residual | 98 | 0.0074 | 0.2371 | 0.502 | 0.514 | 0.0114 | 0.0035 |
| Biotype | θH (MPa-Hours) | Ψb(50) (MPa) | σΨb | RMSE | R2 |
|---|---|---|---|---|---|
| First experiment | |||||
| S | 47.80 ± 6.34 | −0.793 ± 0.012 | 0.268 ± 0.112 | 8.73 | 0.90 |
| WR1 | 87.42 ± 5.87 | −0.609 ± 0.024 | 0.529 ± 0.164 | 9.28 | 0.84 |
| WR2 | 72.34 ± 4.45 | −0.832 ± 0.008 | 0.431 ± 0.067 | 6.04 | 0.90 |
| WR3 | 48.01 ± 5.12 | −0.778 ± 0.009 | 0.239 ± 0.082 | 7.43 | 0.80 |
| WR4 | 61.09 ± 6.78 | −0.530 ± 0.010 | 0.502 ± 0.105 | 5.65 | 0.92 |
| Second experiment | |||||
| S | 48.99 ± 4.31 | −0.908 ± 0.014 | 0.369 ± 0.089 | 10.56 | 0.81 |
| WR1 | 51.41 ± 3.44 | −0.280 ± 0.018 | 0.393 ± 0.077 | 5.63 | 0.82 |
| WR2 | 34.41 ± 2.13 | −0.701 ± 0.004 | 0.220 ± 0.084 | 4.71 | 0.95 |
| WR3 | 31.15 ± 2.87 | −0.750 ± 0.008 | 0.222 ± 0.094 | 5.27 | 0.95 |
| WR4 | 36.31 ± 3.83 | −0.687 ± 0.009 | 0.321 ± 0.075 | 6.05 | 0.93 |
| Water Potential (MPa) | Biotype | SLDW (mg) | FSDW (mg) | MSR (mg) | CEM (mg mg−1) | FSR (mg mg−1) |
|---|---|---|---|---|---|---|
| −0.6 | S | 2.12 a | 10.26 b | 3.22 a | 0.66 b | 0.24 a |
| WR1 | 0.00 c | 11.58 a | 1.98 bc | 0.00 c | 0.15 b | |
| WR2 | 2.00 a | 8.82 c | 3.27 a | 0.53 b | 0.27 a | |
| WR3 | 1.22 b | 8.75 c | 1.40 c | 0.89 a | 0.14 b | |
| WR4 | 1.53 b | 8.76 c | 2.47 b | 0.63 b | 0.22 a | |
| −0.45 | S | 3.16 a | 9.45 b | 4.03 a | 0.79 a | 0.30 b |
| WR1 | 0.00 c | 10.80 a | 2.76 b | 0.00 c | 0.20 c | |
| WR2 | 2.66 a | 7.31 d | 4.80 a | 0.59 b | 0.39 a | |
| WR3 | 1.67 b | 8.26 c | 1.88 c | 0.92 a | 0.19 c | |
| WR4 | 1.87 b | 8.14 c | 3.10 b | 0.61 b | 0.28 b | |
| −0.3 | S | 3.41 b | 8.83 b | 4.65 b | 0.74 ab | 0.35 bc |
| WR1 | 0.00 d | 11.10 a | 2.44 d | 0.00 c | 0.18 d | |
| WR2 | 4.66 a | 6.63 d | 5.49 a | 0.86 a | 0.45 a | |
| WR3 | 2.90 b | 6.17 d | 3.96 bc | 0.74 ab | 0.39 ab | |
| WR4 | 2.04 c | 7.95 c | 3.29 c | 0.64 b | 0.29 c | |
| −0.15 | S | 3.97 b | 8.51 a | 4.98 b | 0.80 a | 0.37 c |
| WR1 | 3.27 c | 7.31 b | 6.26 a | 0.52 c | 0.46 b | |
| WR2 | 5.37 a | 5.68 c | 6.42 a | 0.84 a | 0.53 a | |
| WR3 | 3.93 b | 5.50 c | 4.67 b | 0.84 a | 0.46 b | |
| WR4 | 2.87 c | 6.85 b | 4.37 b | 0.67 b | 0.39 c | |
| 0 | S | 4.80 b | 8.11 a | 5.36 d | 0.89 a | 0.40 c |
| WR1 | 5.03 b | 3.84 bc | 9.72 a | 0.53 c | 0.72 a | |
| WR2 | 7.10 a | 3.78 bc | 8.33 b | 0.85 a | 0.69 a | |
| WR3 | 5.04 b | 3.03 c | 7.12 c | 0.71 b | 0.70 a | |
| WR4 | 3.79 c | 4.50 b | 6.74 c | 0.56 c | 0.59 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benakashani, F.; Gonzalez-Andujar, J.L.; Soltani, E. Differences in Germination of ACCase-Resistant Biotypes Containing Isoleucine-1781-Leucine Mutation and Susceptible Biotypes of Wild Oat (Avena sterilis ssp. ludoviciana). Plants 2021, 10, 2350. https://doi.org/10.3390/plants10112350
Benakashani F, Gonzalez-Andujar JL, Soltani E. Differences in Germination of ACCase-Resistant Biotypes Containing Isoleucine-1781-Leucine Mutation and Susceptible Biotypes of Wild Oat (Avena sterilis ssp. ludoviciana). Plants. 2021; 10(11):2350. https://doi.org/10.3390/plants10112350
Chicago/Turabian StyleBenakashani, Fatemeh, Jose L. Gonzalez-Andujar, and Elias Soltani. 2021. "Differences in Germination of ACCase-Resistant Biotypes Containing Isoleucine-1781-Leucine Mutation and Susceptible Biotypes of Wild Oat (Avena sterilis ssp. ludoviciana)" Plants 10, no. 11: 2350. https://doi.org/10.3390/plants10112350








