PRAT Proteins Operate in Organellar Protein Import and Export in Arabidopsis thaliana
Abstract
:1. Introduction
2. Results
2.1. HP20 and HP30 Interact during the Import of Transit Sequence-Less Precursors into Chloroplasts
2.2. Isolation and Characterization of HP20 and HP30 Knock-Out Mutants of A. thaliana
2.3. Etiolated Athp30-2 Seedlings Show Reduced Viability during Greening
2.4. HP22 Is Part of a Unique Protein Export Pathway from Chloroplasts that Operates during Leaf Senescence
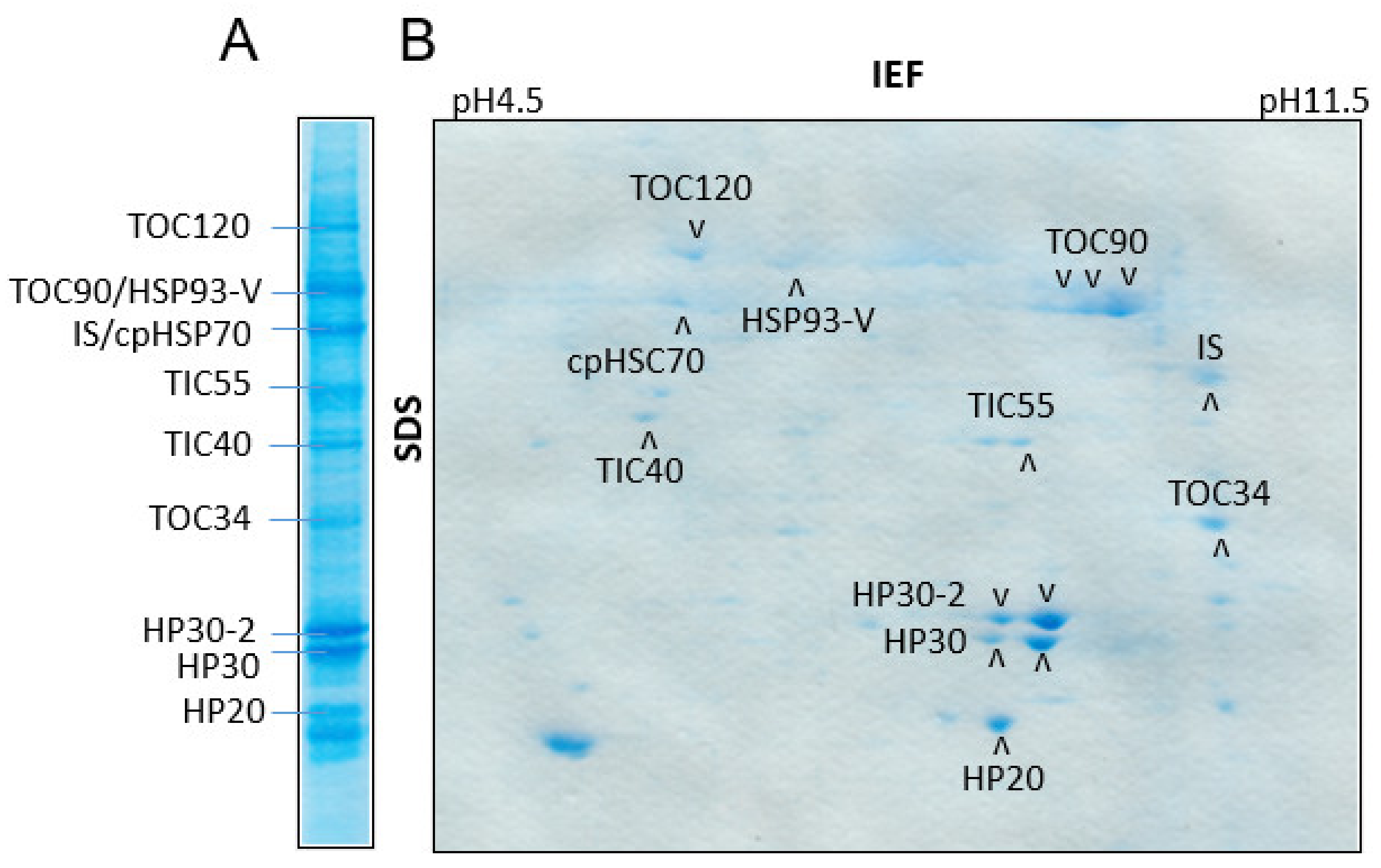
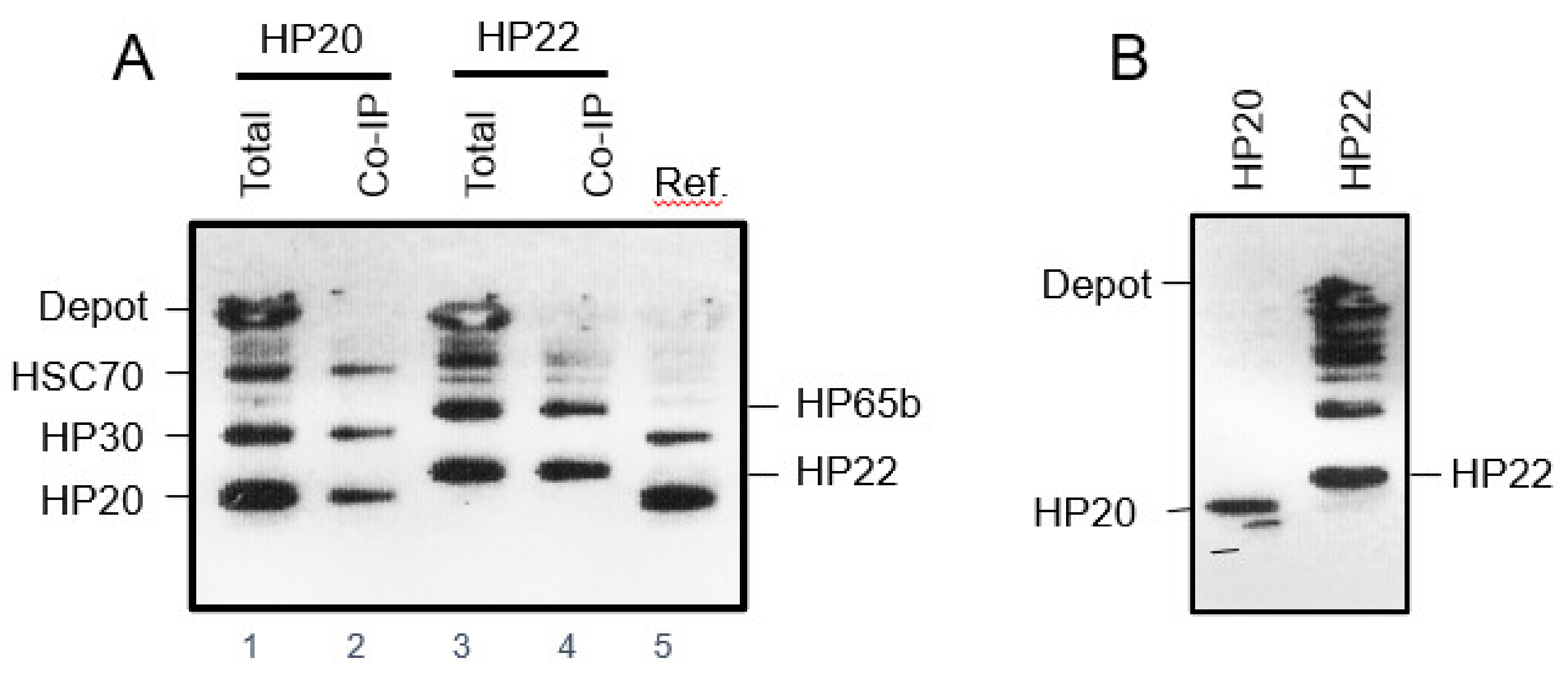
2.5. Isolation of Proteins Interacting with HP22 in Chloroplasts
2.6. Genetic Interaction of HP22 with HP65b
3. Discussion
3.1. Function of HP20, HP30 and HP30-2 in Chloroplasts
3.2. Role of Mitochondrial HP30-2 in planta
3.3. Role of Chloroplast HP22 during Leaf Senescence
4. Materials and Methods
4.1. Arabidopsis Mutants and RNAi Lines
4.2. Plant Growth Conditions
4.3. Seedling Viability Tests
4.4. Pulse-Labeling of Total Leaf Proteins with 35S-Methionine
4.5. Chloroplast Isolation, Amino Acid Uptake and In Organello-Protein Synthesis
4.6. Isolation of Plastid Envelope Protein Complexes
4.7. Plastid Leakage Assay
4.8. Analysis of Mitochondrial Membrane Proteins
4.9. Other Techniques
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GFP | green fluorescent protein |
| HP | hypothetical protein |
| PRAT | preprotein and amino acid transporter |
| TIC | translocase of the inner envelope of chloroplasts |
| TOC | translocase of the outer envelope of chloroplasts |
References
- Kirk, J.T.O.; Tilney-Basset, R.A.E. The Plastids: Their Chemistry, Structure, Growth and Inheritance; Elsevier: Amsterdam, The Netherlands, 1978. [Google Scholar]
- Von Wettstein, D.; Gough, S.; Kannangara, C.G. Chlorophyll biosynthesis. Plant Cell 1995, 7, 1039–1057. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, P.; López-Juez, E. Biogenesis and homeostasis of chloroplasts and other plastids. Nat. Rev. Mol. Cell Biol. 2013, 14, 787–7802. [Google Scholar] [CrossRef]
- Zimorski, V.; Ku, C.; Martin, W.F.; Gould, S.B. Endosymbiotic theory for organelle origins. Curr. Opin. Microbiol. 2014, 22, 38–48. [Google Scholar] [CrossRef]
- Lyska, D.; Meierhoff, K.; Westhoff, P. How to build functional thylakoid membranes: From plastid transcription to protein complex assembly. Planta 2013, 237, 413–428. [Google Scholar] [CrossRef] [Green Version]
- Dall’Osto, L.; Bressan, M.; Bassi, R. Biogenesis of light harvesting proteins. Biochim. Biophys. Acta 2015, 1847, 861–871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, L.X.; Theg, S.M. The chloroplast protein import system: From algae to trees. Biochim. Biophys. Acta 2013, 1833, 314–331. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Kim, D.H.; Hwang, I. Specific targeting of proteins to outer envelope membranes of endosymbiotic organelles, chloroplasts, and mitochondria. Front. Plant Sci. 2014, 29, 173. [Google Scholar] [CrossRef] [Green Version]
- Demarsy, E.; Lakshmanan, A.M.; Kessler, F. Border control: Selectivity of chloroplast protein import and regulation at the TOC-complex. Front. Plant Sci. 2014, 17, 483. [Google Scholar] [CrossRef] [Green Version]
- Richardson, L.G.; Paila, Y.D.; Siman, S.R.; Chen, Y.; Smith, M.D.; Schnell, D.J. Targeting and assembly of components of the TOC protein import complex at the chloroplast outer envelope membrane. Front. Plant Sci. 2014, 11, 269. [Google Scholar] [CrossRef] [Green Version]
- Paila, Y.D.; Richardson, L.G.; Schnell, D.J. New insights into the mechanism of chloroplast protein import and its integration with protein quality control, organelle biogenesis and development. J. Mol. Biol. 2015, 427, 1038–1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakai, M. The TIC complex uncovered: The alternative view on the molecular mechanism of protein translocation across the inner envelope membrane of chloroplasts. Biochim. Biophys. Acta 2015, 1847, 957–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolland, N.; Curien, G.; Finazzi, G.; Kuntz, M.; Maréchal, E.; Matringe, M.; Ravanel, S.; Seigneurin-Berny, D. The biosynthetic capacities of the plastids and integration between cytoplasmic and chloroplast processes. Ann. Rev. Genet. 2012, 46, 233–264. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K. Emerging roles of the chloroplast outer envelope membrane. Trends Plant Sci. 2011, 16, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Facchinelli, F.; Weber, A.P. The metabolite transporters of the plastid envelope: An update. Front. Plant Sci. 2011, 12, 50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Breuers, F.K.; Bräutigam, A.; Weber, A.P. The plastid outer envelope–A highly dynamic interface between plastid and cytoplasm. Front. Plant Sci. 2011, 14, 97. [Google Scholar] [CrossRef] [Green Version]
- Rassow, J.; Dekker, P.J.; van Wilpe, S.; Meijer, M.; Soll, J. The preprotein translocase of the mitochondrial inner membrane: Function and evolution. J. Mol. Biol. 1999, 286, 105–120. [Google Scholar] [CrossRef]
- Drea, S.C.; Lao, N.T.; Wolfe, K.H.; Kavanagh, T.A. Gene duplication, exon gain and neofunctionalization of OEP16-related genes in land plants. Plant J. 2006, 46, 723–735. [Google Scholar] [CrossRef]
- Murcha, M.W.; Elhafez, D.; Lister, R.; Tonti-Filippini, J.; Baumgartner, M.; Philippar, K.; Carrie, C.; Mokranjac, D.; Soll, J.; Whelan, J. Characterization of the preprotein and amino acid transporter gene family in Arabidopsis. Plant Physiol. 2007, 143, 199–212. [Google Scholar] [CrossRef] [Green Version]
- Anderson, J.J.; Oxender, D.L. Escherichia coli transport mutants lacking binding protein and other components of the branched chain amino acid transport systems. J. Bacteriol. 1977, 130, 384–392. [Google Scholar] [CrossRef] [Green Version]
- Neupert, W.; Herrmann, J.M. Translocation of proteins into mitochondria. Annu. Rev. Biochem. 2007, 76, 723–7249. [Google Scholar] [CrossRef] [Green Version]
- Wiedemann, N.; Pfanner, N. Mitochondrial machineries for protein import and assembly. Annu. Rev. Biochem. 2017, 20, 685–714. [Google Scholar] [CrossRef] [Green Version]
- Meier, S.; Neupert, W.; Herrmann, J.M. Conserved N-terminal negative charges in the Tim17 subunit of the TIM23 translocase play a critical role in the import of preproteins into mitochondria. J. Biol. Chem. 2005, 280, 7777–7785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez, D.E.; Costa, M.L.; Guiamet, J.J. Senescence-associated degradation of chloroplast proteins inside and outside the organelle. Plant Biol. 2008, 10, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Carrie, C.; Murcha, M.W.; Kuehn, K.; Duncan, O.; Barthet, M.; Smith, P.M.; Eubel, H.; Meyer, E.; Day, D.A.; Millar, A.H.; et al. Type II NAD(P) dehydrogenases are targeted to mitochondria and chloroplasts or peroxisomes in Arabidopsis thaliana. FEBS Lett. 2008, 582, 3073–3079. [Google Scholar] [CrossRef] [Green Version]
- Duncan, O.; Murcha, M.W.; Whelan, J. Unique components of the plant mitochondrial protein import apparatus. Biochim. Biophys. Acta 2013, 1833, 304–313. [Google Scholar] [CrossRef] [Green Version]
- Murcha, M.W.; Kubiszewski-Jakubiak, S.; Teixeira, P.F.; Gugel, I.L.; Kmiec, B.; Narsai, R.; Ivanova, A.; Megel, C.; Shock, A.; Kraus, S.; et al. Plant-specific preprotein and amino acid transporter proteins are required for tRNA import into mitochondria. Plant Physiol. 2016, 172, 2471–2490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossig, C.; Gray, J.; Valdes, O.; Rustgi, S.; von Wettstein, D.; Reinbothe, C.; Reinbothe, S. HP30–2, a mitochondrial PRAT protein for import of signal sequence-less precursor proteins in Arabidopsis Thaliana. J. Integr. Plant Biol. 2017, 59, 535–551. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Gan, S. Leaf senescence: Signals, execution, and regulation. Curr. Top. Dev. Biol. 2005, 71, 83–112. [Google Scholar]
- Zentgraf, U.; Laun, T.; Miao, Y. The complex regulation of WRKY53 during leaf senescence of Arabidopsis thaliana. Eur. J. Cell Biol. 2010, 89, 1333–1337. [Google Scholar] [CrossRef]
- Christiansen, M.W.; Gregersen, P.L. Members of the barley NAC transcription factor gene family show differential co-regulation with senescence-associated genes during senescence of flag leaves. J. Exp. Bot. 2014, 65, 4009–4022. [Google Scholar] [CrossRef] [Green Version]
- Penfold, C.A.; Buchanan-Wollaston, V. Modelling transcriptional networks in leaf senescence. J. Exp. Bot. 2014, 65, 3859–3873. [Google Scholar] [CrossRef] [Green Version]
- Springer, A.; Kang, C.; Rustgi, S.; von Wettstein, D.; Reinbothe, C.; Pollmann, S.; Reinbothe, S. Programmed chloroplast destruction during leaf senescence involves 13-lipoxygenase (13-LOX). Proc. Natl. Acad. Sci. USA 2016, 113, 3383–3388. [Google Scholar] [CrossRef] [Green Version]
- Buchanan-Wollaston, V.; Earl, S.; Harrison, E.; Mathas, E.; Navabpour, S.; Page, T.; Pink, D. The molecular analysis of leaf senescence–A genomic approach. Plant Biotechnol. J. 2003, 1, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Reinbothe, C.; Reinbothe, S. Regulation of photosynthetic gene expression by the environment: From seedling de-etiolation to leaf senescence. In Photoprotection, Photoinhibition, Gene Regulation, and Environment; Demmig-Adams, B., Adams, W., III, Mattoo, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 333–365. [Google Scholar]
- Sarwat, M.; Tuteja, N. Senescence Signalling and Control in Plants; Elsevier Academic Press: Cambridge, MA, USA, 2018; pp. 1–322. ISBN 9780128131879. [Google Scholar]
- Rossig, C.; Reinbothe, C.; Gray, J.; Valdes, O.; von Wettstein, D.; Reinbothe, S. Three proteins mediate import of transit sequence-less precursors into the inner envelope of chloroplasts in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2013, 110, 19962–19967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossig, C.; Reinbothe, C.; Gray, J.; Valdes, O.; von Wettstein, D.; Reinbothe, S. New functions of the chloroplast Preprotein and Amino acid Transporter (PRAT) family members in protein import. Plant Signal. Behav. 2014, 9, e27693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinbothe, S.; Rossig, C.; Gray, J.; Valdes, O.; Reinbothe, C. Plastid import of a transit sequence-less precursor protein in Arabidopsis thaliana. Cell Mol. Biol. 2014, 60. [Google Scholar] [CrossRef]
- Miras, S.; Salvi, D.; Piette, L.; Seigneurin-Berny, D.; Grunwald, D.; Reinbothe, C.; Joyard, J.; Reinbothe, S.; Rolland, N. Toc159- and Toc75-independent import of a transit sequence-less precursor into the inner envelope of chloroplasts. J. Biol. Chem. 2007, 282, 29482–29492. [Google Scholar] [CrossRef] [Green Version]
- Schnell, D.J.; Kessler, F.; Blobel, G. Isolation of components of the chloroplast protein import machinery. Science 1994, 266, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Chen, K.; Hiltbunner, A.; Wehrli, E.; Eugster, M.; Schnell, D.; Kessler, F. The major protein import receptor of plastids is essential for chloroplast biogenesis. Nature 2000, 403, 203–207. [Google Scholar] [CrossRef] [Green Version]
- Kubis, S.; Patel, R.; Combe, J.; Bedard, J.; Kovacheva, S.; Lilley, K.; Biehl, A.; Leister, D.; Rios, G.; Koncz, C.; et al. Functional specialization amongst the Arabidopsis Toc159 family of chloroplast protein import receptors. Plant Cell 2004, 16, 2059–2077. [Google Scholar] [CrossRef] [Green Version]
- Inoue, H.; Rounds, C.; Schnell, D.J. The molecular basis for distinct pathways for protein import into Arabidopsis chloroplasts. Plant Cell 2010, 22, 1947–1960. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.; Zhong, D.; Monteiro, A. Comparative genomics and evolution of the HSP90 family of genes across all kingdoms of organisms. BMC Genom. 2006, 17, 156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flores-Pérez, Ú.; Jarvis, P. Molecular chaperone involvement in chloroplast protein import. Biochim. Biophys. Acta 2013, 1833, 332–340. [Google Scholar] [CrossRef] [Green Version]
- Alonso, J.M.; Stepanova, A.N.; Leisse, T.J.; Kim, C.J.; Chen, H.; Shinn, P.; Stevenson, D.K.; Zimmerman, J.; Barajas, P.; Cheuk, R.; et al. Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 2003, 301, 653–657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Innis, M.A.; Gelfand, D.H.; Sninsky, J.J.; White, T.J. PCR Protocols; Academic Press: San Diego, CA, USA, 1990. [Google Scholar]
- Pollmann, S.; Springer, A.L.; Buhr, F.; Lahroussi, A.; Samol, I.; Bonneville, J.-M.; Tichtinsky, G.; von Wettstein, D.; Reinbothe, C.; Reinbothe, S. A plant porphyria related to defects in plastid import of protochlorophyllide oxidoreductase A. Proc. Natl. Acad. Sci. USA 2007, 104, 2019–2023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosso, M.G.; Li, Y.; Strizhov, N.; Reiss, B.; Dekker, K.; Weisshaar, B. An Arabidopsis thaliana T-DNA mutagenized population (GABI-Kat) for flanking sequence tag-based reverse genetics. Plant Mol. Biol. 2003, 53, 247–259. [Google Scholar] [CrossRef]
- Caroca, R.; Howell, K.A.; Hasse, C.; Ruf, S.; Bock, R. Design of chimeric expression elements that confer high-level gene activity in chromoplasts. Plant J. 2013, 73, 368–379. [Google Scholar] [CrossRef]
- Ferro, M.; Salvi, D.; Brugière, S.; Miras, S.; Kowalski, S.; Louwagie, M.; Garin, J.; Joyard, J.; Rolland, N. Proteomics of the chloroplast envelope membranes from Arabidopsis thaliana. Mol. Cell Proteom. 2003, 25, 325–345. [Google Scholar] [CrossRef] [Green Version]
- Bhandari, V.; Houry, W.A. Substrate interaction networks of the Escherichia coli chaperones: Trigger factor, DnaK and GroEL. Adv. Exp. Med. Biol. 2015, 883, 271–294. [Google Scholar] [PubMed]
- Krapp, A.; Hofmann, B.; Schäfer, C.; Stitt, M. Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates: A mechanism for the ‘sink regulation’ of photosynthesis? Plant J. 1993, 3, 817–828. [Google Scholar] [CrossRef]
- Van Dingenen, J.; de Milde, L.; Vermeersch, M.; Maleux, K.; de Rycke, R.; de Bruyne, M.; Storme, V.; Gonzalez, N.; Dhondt, S.; Inzé, D. Chloroplasts are central players in sugar-induced leaf growth. Plant Physiol. 2016, 171, 590–605. [Google Scholar] [CrossRef] [Green Version]
- Carrie, C.; Murcha, M.W.; Whelan, J. An in silico analysis of the mitochondrial import apparatus of plants. BMC Plant Biol. 2010, 16, 249. [Google Scholar] [CrossRef] [Green Version]
- Piller, L.E.; Besagni, C.; Ksas, B.L.; Rumeau, D.; Bréhélin, C.; Glauser, G.; Kessler, F.; Havaux, M. Chloroplast lipid droplet type II NAD(P)H quinine oxidoreductase is essential for prenylquinone metabolism and vitamin K1 accumulation. Proc. Natl. Acad. Sci. USA 2011, 108, 14354–14359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elhafez, D.; Murcha, M.W.; Clifton, R.; Soole, K.L.; Day, D.A.; Whelan, J. Characterization of mitochondrial alternative NAD(P)H dehydrogenases in Arabidopsis: Intraorganelle location and expression. Plant Cell Physiol. 2006, 47, 43–54. [Google Scholar] [CrossRef] [Green Version]
- Singhal, K.; Vreede, J.; Mashaghi, A.; Tans, S.J.; Bolhuis, P.G. The trigger factor chaperone encapsulates and stabilizes partial folds of substrate proteins. PLoS Comput. Biol. 2015, 11, e1004444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saio, T.; Guan, X.; Rossi, P.; Economou, A.; Kalodimos, C.G. Structural basis for protein antiaggregation activity of the trigger factor chaperone. Science 2014, 344, 1250494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samol, I.; Buhr, F.; Springer, A.; Pollmann, S.; Lahroussi, A.; Rossig, C.; von Wettstein, D.; Reinbothe, C.; Reinbothe, S. Implication of the oep16–1 mutation in a flu-independent, singlet oxygen-regulated cell death pathway in Arabidopsis thaliana. Plant Cell Physiol. 2011, 52, 84–95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samol, I.; Rossig, C.; Buhr, F.; Springer, A.; Pollmann, S.; Lahroussi, A.; von Wettstein, D.; Reinbothe, C.; Reinbothe, S. The outer chloroplast envelope protein OEP16–1 for plastid import of NADPH:protochlorophyllide oxidoreductase A in Arabidopsis thaliana. Plant Cell Physiol. 2011, 52, 96–111. [Google Scholar] [CrossRef] [Green Version]
- Reinbothe, S.; Reinbothe, C.; Parthier, B. Methyl jasmonate-regulated translation of nuclear-encoded chloroplast proteins in barley (Hordeum vulgare L. cv. Salome). J. Biol. Chem. 1993, 268, 10606–10611. [Google Scholar] [CrossRef]
- Nortin, J.D. Testing of plum pollen viability with tetrazolium salts. Proc. Amer. Soc. Hort. Sci. 1966, 89, 132–134. [Google Scholar]
- Mullet, J.E.; Klein, R.R.; Grossman, A.R. Optimization of protein synthesis in isolated higher plant chloroplasts. Identification of paused translation intermediates. Eur. J. Biochem. 1986, 155, 331–338. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Scharf, K.-D.; Nover, L. Synthesis, modification and structural binding of heat-shock proteins in tomato cell cultures. Eur. J. Biochem. 1984, 139, 303–313. [Google Scholar]
- Towbin, M.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets; procedure and some applications. Proc. Natl. Acad. Sci USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef] [Green Version]
- Reinbothe, C.; Mache, R.; Reinbothe, S. A second, substrate-dependent site of protein import into chloroplasts. Proc. Natl. Acad. Sci. USA 2000, 97, 9795–9800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theg, S.M.; Bauerle, C.; Olsen, L.J.; Selman, B.R.; Keegstra, K. Internal ATP is the only energy requirement for the translocation of precursor proteins across chloroplastic membranes. J. Biol. Chem. 1989, 264, 6730–6736. [Google Scholar] [CrossRef]
- Mans, R.J.; Novelli, G.D. A convenient, rapid and sensitive method for measuring the incorporation of radioactive amino acids into protein. Biochem. Biophys. Res. Commun. 1960, 3, 540–543. [Google Scholar] [CrossRef]
- O’Farrell, P.H. High resolution two-dimensional electrophoresis of proteins. J. Biol. Chem. 1975, 250, 4007–4021. [Google Scholar] [CrossRef]
- Chang, J.Y. Manual micro-sequence analysis of polypeptides using dimethyl-aminobenzene isothiocyanate. Methods Enzymol. 1983, 91, 455–466. [Google Scholar]
- Van Leyen, K.; Duvolsini, R.M.; Engelhardt, H.; Wiedmann, M. A function for lipoxygenase in programmed organelle degradation. Nature 1998, 395, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Day, D.A.; Neuburger, M.; Douce, R. Biochemical characterisation of chlorophyll-free mitochondria from pea leaves. J. Plant Physiol. 1985, 12, 219–228. [Google Scholar]
- Berthold, J.; Bauer, M.F.; Schneider, H.C.; Klaus, C.; Dietmeier, K.; Neupert, W.; Brunner, M. The MIM complex mediates preprotein translocation across the mitochondrial inner membrane and couples it to the mt-Hsp70/ATP driving system. Cell 1995, 81, 1085–1093. [Google Scholar] [CrossRef] [Green Version]
- Porra, R.J.; Thompson, W.A.; Kriedemann, P.E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: Verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 1989, 975, 384–394. [Google Scholar] [CrossRef]
- Martinez-Caballero, S.; Grigoriev, S.M.; Herrmann, J.M.; Campo, M.L.; Kinnally, K.W. Tim17p regulates the twin pore structure and voltage gating of the mitochondrial protein import complex TIM. J. Biol. Chem. 2007, 282, 3584–3593. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bassham, D.C. Autophagy: Pathways for self-eating in plant cells. Annu. Rev. Plant Biol. 2012, 63, 215–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Blumwald, E. Stress-induced chloroplast degradation in Arabidopsis is regulated via a process independent of autophagy and senescence-associated vacuoles. Plant Cell 2014, 26, 4875–4888. [Google Scholar] [CrossRef] [Green Version]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossig, C.; Gray, J.; Valdes, O.; Springer, A.; Rustgi, S.; von Wettstein, D.; Reinbothe, C.; Rassow, J.; Reinbothe, S. PRAT Proteins Operate in Organellar Protein Import and Export in Arabidopsis thaliana. Plants 2021, 10, 958. https://doi.org/10.3390/plants10050958
Rossig C, Gray J, Valdes O, Springer A, Rustgi S, von Wettstein D, Reinbothe C, Rassow J, Reinbothe S. PRAT Proteins Operate in Organellar Protein Import and Export in Arabidopsis thaliana. Plants. 2021; 10(5):958. https://doi.org/10.3390/plants10050958
Chicago/Turabian StyleRossig, Claudia, John Gray, Oscar Valdes, Armin Springer, Sachin Rustgi, Diter von Wettstein, Christiane Reinbothe, Joachim Rassow, and Steffen Reinbothe. 2021. "PRAT Proteins Operate in Organellar Protein Import and Export in Arabidopsis thaliana" Plants 10, no. 5: 958. https://doi.org/10.3390/plants10050958





