Antifungal Activity of the Sesquiterpene Lactones from Psephellus bellus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Identification of Compounds from P. bellus for Antifungal Studies
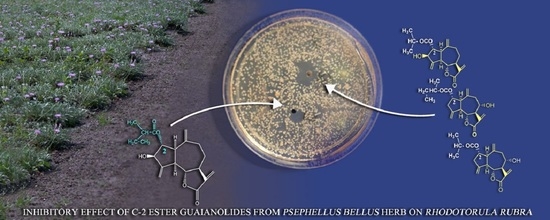
2.2. Antifungal Activity of the Guaianolides from P. bellus
3. Materials and Methods
3.1. Plant Material
3.2. Antifungal Susceptibility Study
3.3. Cytotoxicity Experiments
3.4. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Perfect, J.R. The antifungal pipeline: A reality check. Nature reviews. Drug Discov. 2017, 16, 603–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badiee, P.; Hashenmizadeh, Z. Opportunistic invasive fungal infections: Diagnosis & clinical management. Indian J. Med. Res. 2014, 139, 195–204. [Google Scholar] [PubMed]
- Bongomin, F.; Gago, S.; Oladele, R.O.; Denning, D.W. Global and Multi-National Prevalence of Fungal Diseases-Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef] [PubMed]
- Barrero, A.F.; Oltra, J.E.; Alvarez, M.; Raslan, D.S.; Saude, D.A.; Aksira, M. New sources and antifungal activity of sesquiterpene lactone. Fitoterapia 2000, 71, 60–64. [Google Scholar] [CrossRef]
- Bruno, M.; Bancheva, S.; Roselli, S.; Maggio, A. Sesquiterpenoids in subtribe Centaureinae (Cass) Dumort (tribe Cardueae, Asteraceae): Distribution, 13C NMR spectra data and biological properties. Phytochemistry 2013, 95, 19–93. [Google Scholar] [CrossRef] [Green Version]
- Nawrot, J.; Gornowicz-Porowska, J.; Nowak, G. Phytotherapy Perspectives for Treating Fungal Infections, Migraine, Sebohrreic Dermatitis and Hyperpigmentations with the Plants of the Centaureinae Subtribe (Asteraceae). Molecules 2020, 25, 5329. [Google Scholar] [CrossRef]
- Stefanović, O.D. Synergistic Activity of Antibiotics and Bioactive Plant Extracts: A Study Against Gram-Positive and GramNegative Bacteria. Bact. Pathog. Antibact. Control 2017, 23. [Google Scholar] [CrossRef] [Green Version]
- Heptinstall, S.; Groenwegen, W.A.; Spangenberg, P.; Loesche, W. Extracts feverfew may inhibit behavior via naturalization of sulphydryl groups. J. Pharm. Pharmacol. 1987, 39, 459–465. [Google Scholar] [CrossRef]
- Nowak, G.; Drozdz, B.; Holub, M. Sesquiterpene lactones of the Cardueae, subtribe Centaureinae. In Compositae: Systematics, Proceedings of the International Compositae Conference, Kew, Richmond, UK, 1994; Hind, D.J.N., Beentje, Eds.; Royal Botanic Gardens, Kew: Richmond, UK, 1996; Volume 1, pp. 219–227. [Google Scholar]
- Wyganowska-Swiatkowska, M.; Nohawica, M.; Grocholewicz, K.; Nowak, G. Influence of herbal medicines, on HMCBG1 release SARS-CoV-2 viral attachment, acute respiratory failure and sepsis. A literature review. Int. J. Mol. Sci. 2020, 21, 4639. [Google Scholar] [CrossRef]
- Hadas, E.; Derda, M.; Nawrot, J.; Nowak, G.; Thiem, B. Evaluation of the amoebicidal activities of Centaurea bella, Centaurea daghestanica, Rhaponticum pulchrum and Tanacetum vulgarae against pathogenic Acanthamoeba spp. Acta Pol. Pharm. 2017, 74, 1827–1832. [Google Scholar]
- Nawrot, J.; Napierała, M.; Kaczerowska-Pietrzak, K.; Florek, E.; Gornowicz-Porowska, J.; Pelant, E.; Nowak, G. The antiserotonin effect of parthenolide derivatives and standardised extract from the leaves of Stizolophus balsamita. Molecules 2019, 24, 4131. [Google Scholar] [CrossRef] [Green Version]
- Budesinsky, M.; Nowak, G.; Rychlewska, U.; Hodgson, D.J.; Saman, D.; Daniewski, W.M.; Holub, M. Structure of sesquiterpenic lactones of some species of subtribe Centaureinae. Collect. Czech. Chem. Commun. 1994, 59, 1175–1201. [Google Scholar] [CrossRef]
- Daniewski, W.M.; Nowak, G. Further sesquiterpene lactones of Centaurea bella. Phytochemistry 1993, 32, 204–205. [Google Scholar] [CrossRef]
- Nowak, G. A chemotaxonomy study of sesquiterpene lactones from subtribe Centaureinae of the Compositae. Phytochemistry 1992, 31, 2363–2368. [Google Scholar] [CrossRef]
- Abad, M.J.; Ansuategui, M.; Bermejo, P. Active antifungal substances from natural sources. Arkivoc 2007, 7, 116–145. [Google Scholar]
- Arif, T.; Bhosale, J.B.; Kumar, N.; Mandal, T.K.; Bendre, R.S.; Lavekar, G.S.; Dabur, R. Natural products-antifungal agents derived from plants. J. Asian Nat. Prod. Res. 2009, 11, 621–638. [Google Scholar] [CrossRef] [PubMed]
- Ivanescu, B.; Miron, A.; Corciova, A. Sesquiterpene Lactones from Artemisia Genus: Biological Activities and Methods of Analysis. J. Anal. Methods Chem. 2015, 247685. [Google Scholar] [CrossRef] [Green Version]
- Wedge, D.E.; Galindo, J.C.; Macías, F.A. Fungicidal activity of natural and synthetic sesquiterpene lactone analogs. Phytochemistry 2000, 53, 747–757. [Google Scholar] [CrossRef]
- Nowak, G. Chromatography of twenty six sesquiterpene lactones from Centaurea bella. Chromatographia 1993, 35, 325–328. [Google Scholar] [CrossRef]
- Lockhart, S.R.; Guarner, J. Emerging and reemerging fungal infections. Semin. Diagn. Pathol. 2019, 36, 177–181. [Google Scholar] [CrossRef]
- Mehra, T.; Köberle, M.; Braunsdorf, C.; Mailänder-Sanchez, D.; Borelli, C.; Schaller, M. Alternative approaches to antifungal therapies. Exp. Dermatol. 2012, 21, 778–782. [Google Scholar] [CrossRef] [Green Version]
- Magaldi, S.; Mata-Essayag, S.; Hartung de Capriles, C.; Perez, C.; Colella, M.T.; Olaizola, C.; Ontiveros, Y. Well diffusion for antifungal susceptibility testing. Int. J. Infect. Dis. 2004, 8, 39–45. [Google Scholar] [CrossRef] [Green Version]
- Rex, J.H.; Pfaller, M.A.; Walsh, T.J.; Chaturvedi, V.; Espinel-Ingroff, A.; Ghannoum, M.A.; Gosey, L.L.; Odds, F.C.; Rinaldi, M.G.; Sheehan, D.J.; et al. Antifungal susceptibility testing: Practical aspects and current challenges. Clin. Microbiol. Rev. 2001, 14, 43–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shirani, M.; Samimi, A.; Kalantari, H.; Madani, M.; Kord-Zanganeh, A. Chemical composition and antifungal effect of hydroalcoholic extract of Allium tripedale Trautv. against Candida species. Curr. Med. Mycol. 2017, 3, 6–12. [Google Scholar] [PubMed]
- Binder, U.; Aigner, M.; Risslegger, B.; Hörtnagl, C.; Lass-Flörl, C.; Lackner, M. Minimal Inhibitory Concentration (MIC)-Phenomena in Candida albicans and Their Impact on the Diagnosis of Antifungal Resistance. J. Fungi 2019, 5, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Pos. | 1 a | 11 a | 12 a | 13 b |
|---|---|---|---|---|
| 1 | 2.93 dd (6.5; 9.8) | 2.98 m | 2.98 m | 3.38 m |
| 2α | - | - | - | 2.51 m |
| 2β | 4.94 dd (6.5; 5.8) | 4.98 dd (9.3; 7.3) | 4.96 dd (9.0; 7.2) | 1.81 m |
| 3 | 4.41 m | 4.43 m | 4.43 m | 3.99 m |
| 5 | 3.01 t | 2.98 m | 2.85 m | 2.02 d (11.3) |
| 6 | 4.16 dd (9.0; 9.8) | 4.16 dd (10.6; 9.0) | 4.25 dd (10.1; 9.0) | 4.08 d 9.3) |
| 7 | 2.88 m | 2.81 m | 3.19 m | 3.08 t (9.4; 3.5; 3.2) |
| 8α | 2.25 m | 4.01 m | 4.01 dd (3.0; 5.3) | 5.24 m |
| 8β | 2.24 m | - | - | - |
| 9α | 2.25 m | 2.32 dd (5.4; 14.6) | 2.37 dd (5.3; 14.6) | 2.49 dd (15.2; 2.7) |
| 9β | 2.47 m | 2.70 dd (14.6; 5.4) | 2.70 dd (14.6; 5.3) | 2.71 dd (15.2; 5.2) |
| 13 | 6.25 d (3.6) | 6.29 dd (3.5; 0.8) | 6.21 d (0.8) | 6.24 d (1.6) |
| 13′ | 5.52 d (3.2) | 6.16 dd (3.2; 0.8) | 6.16 d (0.8) | 5.57 d (1.6) |
| 14 | 4.98 d (2.5) | 5.11 d (1.6) | 5.14 d (1.7) | 5.21 d (1.8) |
| 14′ | 4.93 d (2.1) | 5.03 d (1.6) | 4.92 d (1.7) | 5.11 d (1.8) |
| 15 | 5.52 d (2.1) | 5.66 dd (1.5; 0.8) | 5.66 dd (0) | 3.34 d (4.2) |
| 15′ | 5.44 d (1.8) | 5.47 dd (1.7; 0.8) | 5.47 m | 3.07 d (4.2) |
| 8-OH | 1.85 brs | 1.85 brs | ||
| 2′ | 2.40 m | 2.42 m | 2.48 m | - |
| 3′ | 1.67 m; 1.48 m | 1.63 m; 1.51 m | 1.17 d | 3.88 d |
| 4′ | 0.90 m | 1.15 d | 1.16 d | 1.55 s |
| 5′ | 1.16 q | 0.69 m | - | - |
| Name of Fungal Strains | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Candida albicans | Candida famata | Candida glabrata | Candida parapsilosis | Rhodotorula rubra | Trichophyton rubrum | Trichophyton mentagrophytes var. interdigitale | Trichophyton mentagrophytes var. interdigitale | Microsporum canis | Scopulariopsis brevicaulis | ||||||||||
| G | I | G | I | G | I | G | I | G | I | G | I | G | I | G | I | G | I | G | I | |
| Acroptilin (13) | yes | no | yes | 6.25 ± 0.35 | yes | no | yes | no | yes | no | yes | 21.5 ± 0.71 | yes | 6.5 ± 0.71 | yes | 6.25 ± 0.35 | yes | no | yes | no |
| Cebellin A (11) | yes | no | X | X | yes | 18.5 ± 0.71 | X | X | yes | 11.75 ± 0.35 | no | no | yes | 15.5 ± 0.71 | no | no | yes | no | yes | 10.5 ± 0.71 |
| Cebellin B (12) | yes | no | 10.5 ± 0.71 | yes | 12.5 ± 0.71 | yes | 8.25 ± 0.35 | yes | no | yes | 12.25 ± 0.35 | yes | 10.25 ± 0.35 | yes | 12.25 ± 0.35 | yes | no | yes | 8.5 ± 0.71 | |
| Cebellin L (1) | yes | no | yes | 15.5 ± 0.71 | yes | 14.25 ± 0.35 | yes | 26.5 ± 0.71 | yes | no | yes | 12.5 ± 0.71 | yes | 20.5 ± 0.71 | yes | 18.5 ± 0.71 | yes | no | yes | 10.5 ± 0.71 |
| Cebellin K + N + O (2-4) | yes | 12.25 ± 0.35 | X | X | yes | 19.75 ± 0.35 | X | X | yes | 21.5 ± 0.71 | no | no | yes | 24.25 ± 0.35 | no | no | yes | 20.25 ± 0.35 | yes | 9.25 ± 0.35 |
| Full extract of P. bellus | yes | no | yes | 12 | yes | 12.5 ± 0.71 | yes | 4 ± 1.41 | no | yes | 24.5 ± 0.71 | yes | 18.5 ± 0.71 | yes | 33.5 ± 0.71 | yes | no | yes | no | |
| Compound | Candida famata | Candida glabrata | Candida parapsilosis | Rhodotorula rubra | Trichophyton rubrum | Trichophyton mentagrophytes var interdigitale | Scopulariopsis brevicaulis | |
|---|---|---|---|---|---|---|---|---|
| p-Value | ||||||||
| Acroptilin (13) | Cebellin A (11) | - | - | - | - | - | 0.0061 * | - |
| Cebellin B (12) | 0.0169 * | - | - | - | 0.0036 * | 0.0216 * | - | |
| Cebellin L (1) | 0.0036 * | - | - | - | 0.0062 * | 0.0026 * | - | |
| Cebellin K + N + O (2–4) | - | - | - | - | - | 0.001 * | - | |
| Full extract of P. bellus | - | - | - | - | 0.0517 | 0.0035 * | - | |
| Cebellin A (11) | Cebellin B (12) | - | 0.0137 * | - | - | - | 0.0112 * | 0.1063 |
| Cebellin L (1) | - | 0.0169 * | - | - | - | 0.0196 * | 1 | |
| Cebellin K + N + O (2–4) | - | 0.1552 | - | 0.0033 * | - | 0.0041 * | 0.1552 | |
| Full extract of P. bellus | - | 0.0137 * | - | - | - | 0.0517 | - | |
| Cebellin B (12) | Cebellin L (1) | 0.0196 * | 0.0889 | 0.0009 * | - | 0.6988 | 0.003 * | 0.1063 |
| Cebellin K + N + O (2–4) | - | 0.0059 * | - | - | - | 0.0016 * | 0.3122 | |
| Full extract of P. bellus | - | 1 | 0.0531 | - | 0.0021 * | 0.0046 * | - | |
| Cebellin L (1) | Cebellin K + N + O (2–4) | - | 0.004 * | - | - | - | 0.0216 * | 0.1552 |
| Full extract of P. bellus | - | 0.0889 | 0.002 * | - | 0.0035 * | 0.1063 | - | |
| Cebellin K + N + O (2–4) | Full extract of P. bellus | - | 0.0059 * | - | - | - | 0.0093 * | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nawrot, J.; Adamski, Z.; Kamińska-Kolat, B.; Kubisiak-Rzepczyk, H.; Kroma, A.; Nowak, G.; Gornowicz-Porowska, J. Antifungal Activity of the Sesquiterpene Lactones from Psephellus bellus. Plants 2021, 10, 1180. https://doi.org/10.3390/plants10061180
Nawrot J, Adamski Z, Kamińska-Kolat B, Kubisiak-Rzepczyk H, Kroma A, Nowak G, Gornowicz-Porowska J. Antifungal Activity of the Sesquiterpene Lactones from Psephellus bellus. Plants. 2021; 10(6):1180. https://doi.org/10.3390/plants10061180
Chicago/Turabian StyleNawrot, Joanna, Zygmunt Adamski, Beata Kamińska-Kolat, Honorata Kubisiak-Rzepczyk, Anna Kroma, Gerard Nowak, and Justyna Gornowicz-Porowska. 2021. "Antifungal Activity of the Sesquiterpene Lactones from Psephellus bellus" Plants 10, no. 6: 1180. https://doi.org/10.3390/plants10061180