Fruit Growth Stage Transitions in Two Mango Cultivars Grown in a Mediterranean Environment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Orchard Characteristics and Plant Material
2.2. Environmental Conditions
2.3. Fruit Measurements and Experimental Design
2.4. Fruit Development Parameters
2.5. Statistical Analysis
3. Results and Discussion
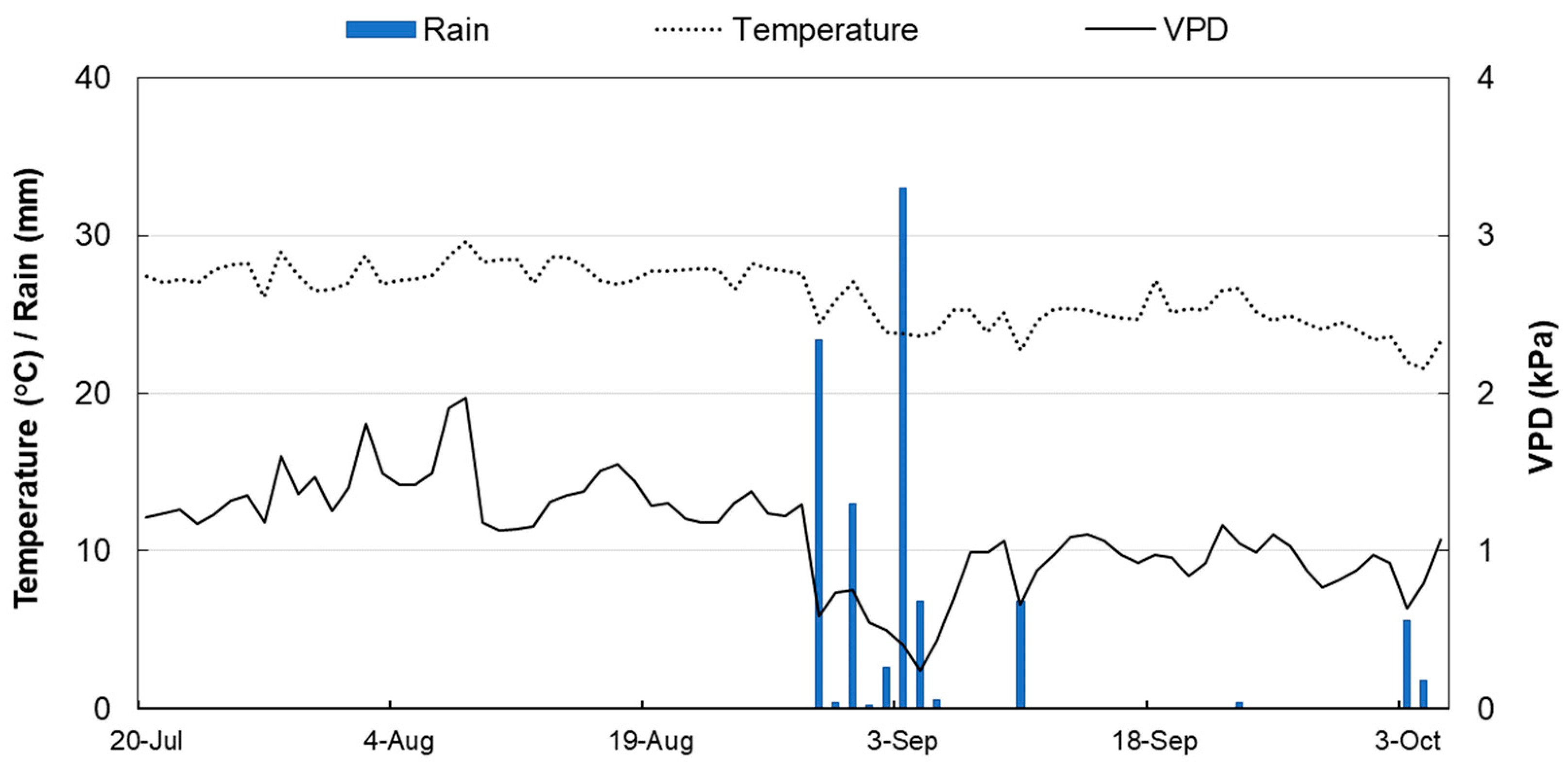
3.1. Climate Data
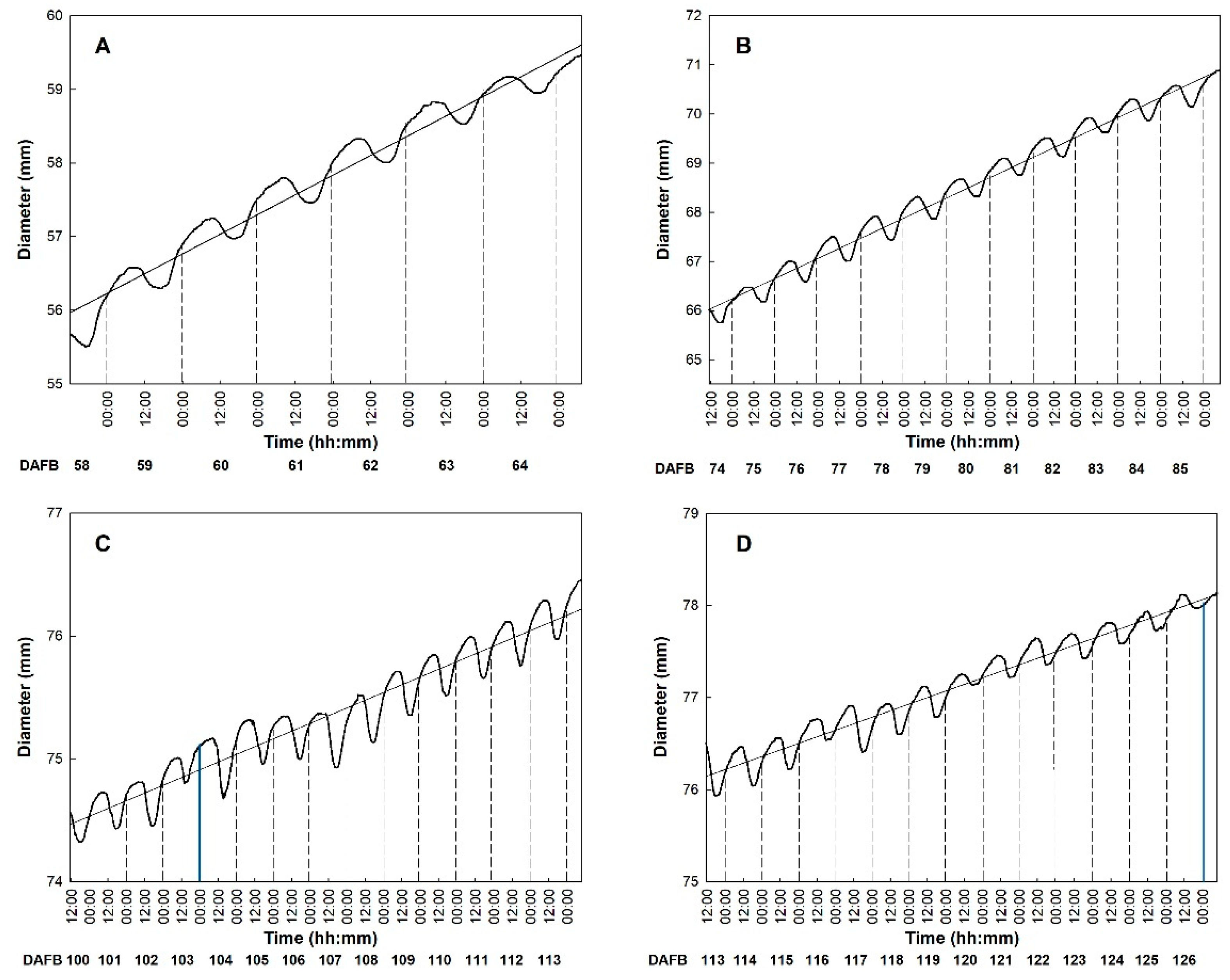
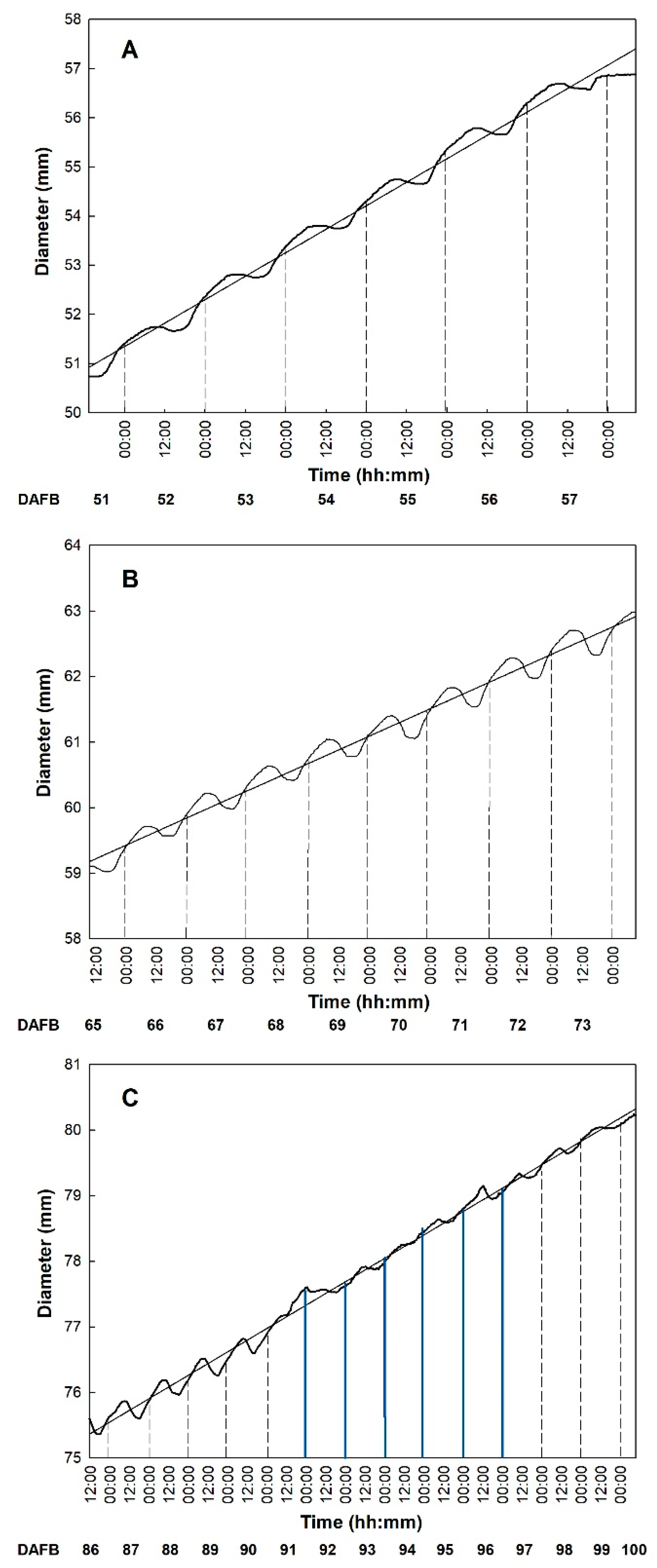
3.2. Fruit Growth
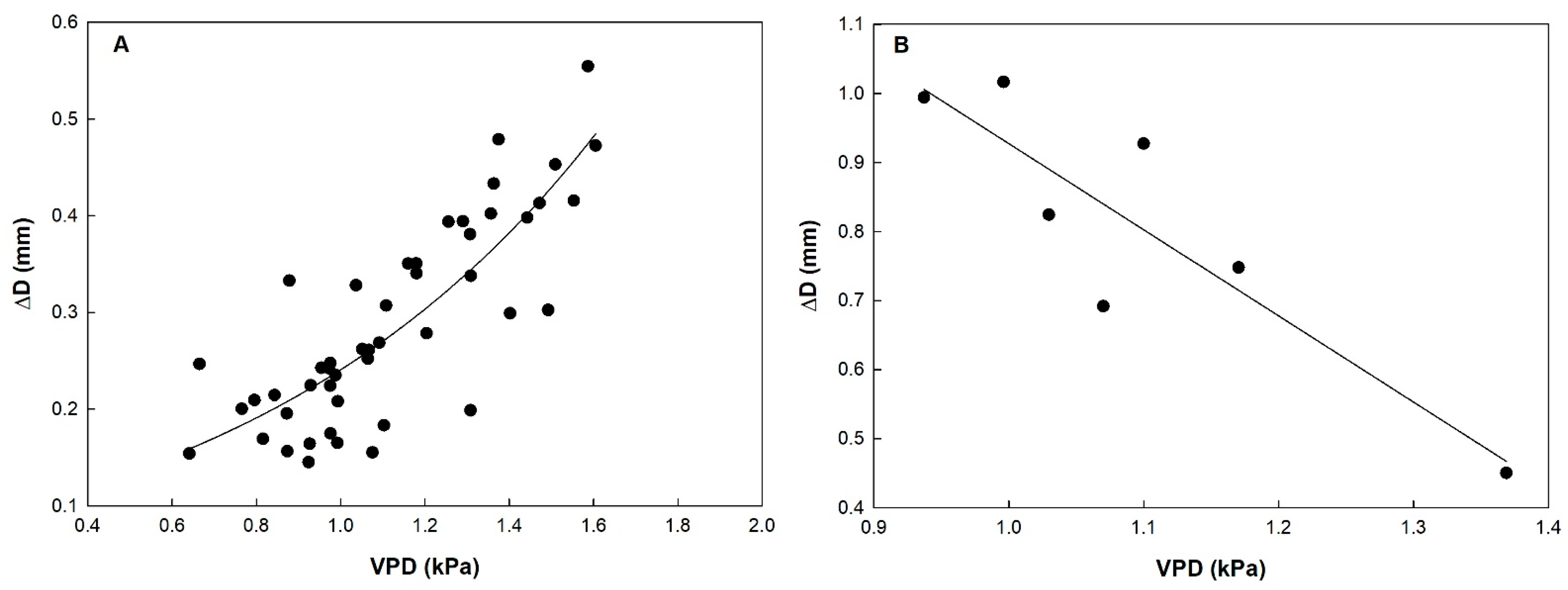
3.3. Fruit Development
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lionello, P.; Malanotte-Rizzoli, P.; Boscolo, R.; Alpert, P.; Artale, V.; Li, L.; Luterbacher, J.; May, W.; Trigo, R.; Tsimplis, M. The Mediterranean Climate: An Overview of the Main Characteristics and Issues. Dev. Earth Environ. Sci. 2006, 4, 1–26. [Google Scholar]
- Giorgi, F. Climate Change Hot-Spots. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Giorgi, F.; Lionello, P. Climate Change Projections for the Mediterranean Region. Glob. Planet. Chang. 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Mariani, L.; Parisi, S.; Cola, G.; Failla, O. Climate Change in Europe and Effects on Thermal Resources for Crops. Int. J. Biometeorol. 2012, 56, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, R. The Impact of Climate Change on Agriculture in Developing Countries. J. Nat. Resour. Policy Res. 2008, 1, 5–19. [Google Scholar] [CrossRef] [Green Version]
- Nath, V.; Kumar, G.; Pandey, S.; Pandey, S. Impact of climate change on tropical fruit production systems and its mitigation strategies. In Climate Change and Agriculture in India: Impact and Adaptation; Springer: Berlin/Heidelberg, Germany, 2019; pp. 129–146. [Google Scholar]
- Farina, V.; Barone, F.; Mazzaglia, A.; Lanza, C.M. Evaluation of Fruit Quality in Loquat Using Both Chemical and Sensory Analyses. In Proceedings of the Acta Horticulturae, International Society for Horticultural Science (ISHS), Leuven, Belgium, 31 January 2011; pp. 345–349. [Google Scholar]
- Mossad, A.; El Helew, W.K.M.; Elsheshetawy, H.E.; Farina, V. Mass Modelling by Dimension Attributes for Mango (Mangifera Indica Cv. Zebdia) Relevant to Post-Harvest and Food Plants Engineering. Agric. Eng. Int. CIGR J. 2016, 18, 219–229. [Google Scholar]
- Farina, V.; Tripodo, L.; Gianguzzi, G.; Sortino, G.; Giuffre, D.; Cicero, U.L.; Candia, R.; Collura, A. Innovative Techniques to Reduce Chilling Injuries in Mango (Mangifera Indica L.) Trees under Mediterranean Climate. Chem. Eng. Trans. 2017, 58, 823–828. [Google Scholar]
- Ochse, J.; Dijkman, M.; Wehlburg, M. Cultivo y Mejoramiento de Plantas Tropicales y Subtropicales; Limusa-Wiley: Ciudad de México, Mexico, 1972. [Google Scholar]
- Purseglove, J.W. Tropical Crops. Dicotyledons 2; Longmans: London, UK, 1968; pp. 333–719. [Google Scholar]
- Chacko, E.K. Physiology of Vegetative and Reproductive Growth in Mango (Mangifera Indica L.) Trees; CSIRO: Melbourne, Australia, 1986; Volume 1, pp. 54–70.
- Gianguzzi, L.; Papini, F.; Cusimano, D. Phytosociological Survey Vegetation Map of Sicily (Mediterranean Region). J. Maps 2016, 12, 845–851. [Google Scholar] [CrossRef] [Green Version]
- Boini, A.; Bresilla, K.; Perulli, G.; Manfrini, L.; Corelli Grappadelli, L.; Morandi, B. Photoselective Nets Impact Apple Sap Flow and Fruit Growth. Agric. Water Manag. 2019, 226, 105738. [Google Scholar] [CrossRef]
- Lizada, C. Mango. In Biochemistry of Fruit Ripening; Springer: Berlin/Heidelberg, Germany, 1993; pp. 255–271. [Google Scholar]
- da Silva, A.C.; de Souza, A.P.; Leonel, S.; de Souza, M.E.; Ramos, D.P.; Tanaka, A.A. Growth and Flowering of Five Mango Cultivar under Subtropics Conditions of Brazil. Am. J. Plant Sci. 2014, 2014, 42759. [Google Scholar] [CrossRef] [Green Version]
- Sergent, E. El Cultivo Del Mango (Mangifera Indica L.): Botánica, Manejo y Comercialización; CDCH UCV: Caracas, Venezuela, 1999; Volume 72, ISBN 980-00-1389-X. [Google Scholar]
- Galán Saúco, V. El Cultivo Del Mango; Mundi-Prensa Libros: Madrid, Spain, 2009; ISBN 84-8476-367-6. [Google Scholar]
- Li, M.; Feng, F.; Cheng, L. Expression Patterns of Genes Involved in Sugar Metabolism and Accumulation during Apple Fruit Development. PLoS ONE 2012, 7, e33055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agustí, M. Fruticultura; Mundi-Prensa: Madrid, Spain, 2010; pp. 140–144. ISBN 978-84-8476-398-7. [Google Scholar]
- Génard, M.; Dauzat, J.; Franck, N.; Lescourret, F.; Moitrier, N.; Vaast, P.; Vercambre, G. Carbon Allocation in Fruit Trees: From Theory to Modelling. Trees 2008, 22, 269–282. [Google Scholar] [CrossRef]
- Lescourret, F.; Génard, M.; Habib, R.; Fishman, S. Variation in Surface Conductance to Water Vapor Diffusion in Peach Fruit and Its Effects on Fruit Growth Assessed by a Simulation Model. Tree Physiol. 2001, 21, 735–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morandi, B.; Rieger, M.; Grappadelli, L.C. Vascular Flows and Transpiration Affect Peach (Prunus Persica Batsch.) Fruit Daily Growth. J. Exp. Bot. 2007, 58, 3941–3947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Léchaudel, M.; Lopez-Lauri, F.; Vidal, V.; Sallanon, H.; Joas, J. Response of the Physiological Parameters of Mango Fruit (Transpiration, Water Relations and Antioxidant System) to Its Light and Temperature Environment. J. Plant Physiol. 2013, 170, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, H.; Sakuratani, T. Water Dynamics in Mango (Mangifera Indica L.) Fruit during the Young and Mature Fruit Seasons as Measured by the Stem Heat Balance Method. J. Jpn. Soc. Hortic. Sci. 2006, 75, 11–19. [Google Scholar] [CrossRef]
- Dražeta, L.; Lang, A.; Hall, A.J.; Volz, R.K.; Jameson, P.E. Causes and Effects of Changes in Xylem Functionality in Apple Fruit. Ann. Bot. 2004, 93, 275–282. [Google Scholar] [CrossRef] [Green Version]
- Morandi, B.; Manfrini, L.; Zibordi, M.; Losciale, P.; Corelli Grappadelli, L. Effects of Drought Stress on the Growth, Water Relations and Vascular Flows of Young ’Summerkiwi’ Fruit. In Proceedings of the XXVIII International Horticultural Congress on Science and Horticulture for People (IHC2010): International Symposium on CLIMWATER 2010: Horticultural Use of Water in a Changing Climate, Lisbon, Portugal, 22–27 August 2010; pp. 355–359. [Google Scholar]
- Mazzeo, M.; Dichio, B.; Clearwater, M.J.; Montanaro, G.; Xiloyannis, C. Hydraulic Resistance of Developing Actinidia Fruit. Ann. Bot. 2013, 112, 197–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morandi, B.; Manfrini, L.; Zibordi, M.; Noferini, M.; Fiori, G.; Grappadelli, L.C. A Low-Cost Device for Accurate and Continuous Measurements of Fruit Diameter. HortScience 2007, 42, 1380–1382. [Google Scholar] [CrossRef] [Green Version]
- Grierson, W. Fruit Development, Maturation and Ripening. In Handbook of Plant and Crop Physiology; CRC Press: Boca Raton, FL, USA, 2002; pp. 143–160. [Google Scholar]
- Lakshminarayana, S. Respiration and Ripening Patterns in the Life Cycle of the Mango Fruit. J. Hortic. Sci. 1973, 48, 227–233. [Google Scholar] [CrossRef]
- Giovannoni, J. Molecular Biology of Fruit Maturation and Ripening. Annu. Rev. Plant Biol. 2001, 52, 725–749. [Google Scholar] [CrossRef]
- Morandi, B.; Losciale, B.P.; Manfrini, L.; Zibordi, M.; Studhalter, M.; Grappadelli, L.C. The Growth of the Kiwifruit in Its Final Stages. In VI International Symposium on Kiwifruit; ISHS: Leuven, Belgium, 2006; pp. 369–374. [Google Scholar]
- Morandi, B.; Manfrini, L.; Losciale, P.; Zibordi, M.; Corelli Grappadelli, L. Changes in Vascular and Transpiration Flows Affect the Seasonal and Daily Growth of Kiwifruit (Actinidia Deliciosa) Berry. Ann. Bot. 2010, 105, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Morandi, B.; Anconelli, S.; Losciale, P.; Manfrini, L.; Pierpaoli, E.; Zibordi, M.; Grappadelli, L. How Irrigation Level Affects the Daily Growth Pattern of Young Pear Fruit of the Cv.’Abbé Fetel’. Acta Hortic. 2014, 163–169. [Google Scholar] [CrossRef]
- Scalisi, A.; O’Connell, M.G.; Stefanelli, D.; Lo Bianco, R. Fruit and Leaf Sensing for Continuous Detection of Nectarine Water Status. Front. Plant Sci. 2019, 10, 805. [Google Scholar] [CrossRef] [Green Version]
- Grilo, F.; Scalisi, A.; Pernice, F.; Morandi, B.; Lo Bianco, R. Recurrent Deficit Irrigation and Fruit Harvest Affect Tree Water Relations and Fruitlet Growth in ‘Valencia’Orange. Eur. J. Hortic. Sci 2019, 84, 177–187. [Google Scholar] [CrossRef] [Green Version]
- Scalisi, A.; Marino, G.; Marra, F.P.; Caruso, T.; Bianco, R.L. A Cultivar-Sensitive Approach for the Continuous Monitoring of Olive (Olea Europaea L.) Tree Water Status by Fruit and Leaf Sensing. Front. Plant Sci. 2020, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Marino, G.; Scalisi, A.; Guzmán-Delgado, P.; Caruso, T.; Marra, F.P.; Lo Bianco, R. Detecting Mild Water Stress in Olive with Multiple Plant-Based Continuous Sensors. Plants 2021, 10, 131. [Google Scholar] [CrossRef]
- KOPPEN, W. das Das Geographische System Der Klimat. In Handbuch der klimatologie; Gebrüder Borntraeger: Berlin, Germany, 1936; pp. 1–44. [Google Scholar]
- Duro, A.; Piccione, V.; Scalia, C.; Zampino, D. Precipitazioni e Temperature Medie Mensili in Sicilia Relative al Sessantennio 1926–1985. In 5 Workshop Progetto Strategico Clima, Ambiente e Territorio nel Mezzogiorno CNR; ISHS: Leuven, Belgium, 1996; pp. 17–103. [Google Scholar]
- Drago, A.; Cartabellotta, D.; Lo Bianco, B.; Monterosso, I. Atlante Climatologico Della Sicilia. Seconda Edizione. Riv. Ital. Di Agrometeorol. 2005, 2, 67–83. [Google Scholar]
- Farina, V.; Gianguzzi, G.; D’Asaro, A.; Mazzaglia, A.; Palazzolo, E. Fruit Production and Quality Evaluation of Four Litchi Cultivars (Litchi Chinensis Sonn.) Grown in Mediterranean Climate. Fruits 2017, 72, 203–211. [Google Scholar] [CrossRef]
- Torres, A.C.; Galván, D.F.; Saúco, V.G. Guía Descriptiva de Cultivares de Mango; Consejería de Agricultura, Pesca y Alimentación: Seville, Spain, 1997; ISBN 84-86840-11-2. [Google Scholar]
- Yahia, E.M. Mango (Mangifera Indica L.). In Postharvest Biology and Technology of Tropical and Subtropical Fruits; Elsevier: Amsterdam, The Netherlands, 2011; pp. 492–567e. [Google Scholar]
- Léchaudel, M.; Joas, J. An Overview of Preharvest Factors Influencing Mango Fruit Growth, Quality and Postharvest Behaviour. Braz. J. Plant Physiol. 2007, 19, 287–298. [Google Scholar] [CrossRef] [Green Version]
- Faust, M. Physiology of Temperate Zone Fruit Trees; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1989; ISBN 0-471-81781-3. [Google Scholar]
- Torres-Ruiz, J.M.; Perulli, G.D.; Manfrini, L.; Zibordi, M.; Velasco, G.L.; Anconelli, S.; Pierpaoli, E.; Corelli-Grappadelli, L.; Morandi, B. Time of Irrigation Affects Vine Water Relations and the Daily Patterns of Leaf Gas Exchanges and Vascular Flows to Kiwifruit (Actinidia Deliciosa Chev.). Agric. Water Manag. 2016, 166, 101–110. [Google Scholar] [CrossRef]
- Léchaudel, M.; Génard, M.; Lescourret, F.; Urban, L.; Jannoyer, M. Modeling Effects of Weather and Source–Sink Relationships on Mango Fruit Growth. Tree Physiol. 2005, 25, 583–597. [Google Scholar] [CrossRef] [Green Version]
- Dambreville, A.; Normand, F.; Lauri, P.-E.; Guedon, Y. Modeling Developmental Growth Stages in Mango on the Basis of Absolute vs. Relative Growth Rates. In Proceedings of the International Symposium on Modelling in Fruit Research and Orchard Management, Montpellier, France, 2–5 June 2015; pp. 69–74. [Google Scholar]
- Camacho-Vázquez, C.; Ruiz-May, E.; Guerrero-Analco, J.A.; Elizalde-Contreras, J.M.; Enciso-Ortiz, E.J.; Rosas-Saito, G.; López-Sánchez, L.; Kiel-Martínez, A.L.; Bonilla-Landa, I.; Monribot-Villanueva, J.L. Filling Gaps in Our Knowledge on the Cuticle of Mangoes (Mangifera Indica) by Analyzing Six Fruit Cultivars: Architecture/Structure, Postharvest Physiology and Possible Resistance to Fruit Fly (Tephritidae) Attack. Postharvest Biol. Technol. 2019, 148, 83–96. [Google Scholar] [CrossRef]
- Nordey, T.; Léchaudel, M.; Génard, M. The Decline in Xylem Flow to Mango Fruit at the End of Its Development Is Related to the Appearance of Embolism in the Fruit Pedicel. Funct. Plant Biol. 2015, 42, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Montanaro, G.; Dichio, B.; Xiloyannis, C. Significance of Fruit Transpiration on Calcium Nutrition in Developing Apricot Fruit. J. Plant Nutr. Soil Sci. 2010, 173, 618–622. [Google Scholar] [CrossRef]
- Montanaro, G.; Dichio, B.; Xiloyannis, C.; Lang, A. Fruit Transpiration in Kiwifruit: Environmental Drivers and Predictive Model. AoB Plants 2012, 2012, pls036. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maguire, K.M.; Banks, N.H.; Opara, L.U. Factors Affecting Weight Loss of Apples. Hortic. Rev. 2001, 25, 197–234. [Google Scholar]
- Lechaudel, M.; Génard, M.; Lescourret, F.; Urban, L.; Jannoyer, M. Leaf-to-Fruit Ratio Affects Water and Dry-Matter Content of Mango Fruit. J. Hortic. Sci. Biotechnol. 2002, 77, 773–777. [Google Scholar] [CrossRef]
- Mukherjee, S.; Litz, R.E. Introduction: Botany and Importance. In The Mango: Botany, Production and Uses, 2nd ed.; CABI Publishing: Wallingford, UK, 2009; pp. 1–18. [Google Scholar]
- Guillén, L.; Adaime, R.; Birke, A.; Velázquez, O.; Angeles, G.; Ortega, F.; Ruíz, E.; Aluja, M. Effect of Resin Ducts and Sap Content on Infestation and Development of Immature Stages of Anastrepha Obliqua and Anastrepha Ludens (Diptera: Tephritidae) in Four Mango (Sapindales: Anacardiaceae) Cultivars. J. Econ. Entomol. 2017, 110, 719–730. [Google Scholar]
- Brüggenwirth, M.; Winkler, A.; Knoche, M. Xylem, Phloem, and Transpiration Flows in Developing Sweet Cherry Fruit. Trees 2016, 30, 1821–1830. [Google Scholar] [CrossRef]
- Choat, B.; Gambetta, G.A.; Shackel, K.A.; Matthews, M.A. Vascular Function in Grape Berries across Development and Its Relevance to Apparent Hydraulic Isolation. Plant Physiol. 2009, 151, 1677–1687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González-Aguilar, G.; Sotelo-Mundo, R.; Báez-Sañudo, R.; Petit-Jiménez, D.; González-León, A. Permeability of Cuticular Membrane during the Ontogeny of Mangifera Indica L. In Proceedings of the VIII International Mango Symposium Conference, Sun City, South Africa, 5–10 February 2006; pp. 213–220. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carella, A.; Gianguzzi, G.; Scalisi, A.; Farina, V.; Inglese, P.; Bianco, R.L. Fruit Growth Stage Transitions in Two Mango Cultivars Grown in a Mediterranean Environment. Plants 2021, 10, 1332. https://doi.org/10.3390/plants10071332
Carella A, Gianguzzi G, Scalisi A, Farina V, Inglese P, Bianco RL. Fruit Growth Stage Transitions in Two Mango Cultivars Grown in a Mediterranean Environment. Plants. 2021; 10(7):1332. https://doi.org/10.3390/plants10071332
Chicago/Turabian StyleCarella, Alessandro, Giuseppe Gianguzzi, Alessio Scalisi, Vittorio Farina, Paolo Inglese, and Riccardo Lo Bianco. 2021. "Fruit Growth Stage Transitions in Two Mango Cultivars Grown in a Mediterranean Environment" Plants 10, no. 7: 1332. https://doi.org/10.3390/plants10071332
APA StyleCarella, A., Gianguzzi, G., Scalisi, A., Farina, V., Inglese, P., & Bianco, R. L. (2021). Fruit Growth Stage Transitions in Two Mango Cultivars Grown in a Mediterranean Environment. Plants, 10(7), 1332. https://doi.org/10.3390/plants10071332








