Induced Genetic Variations in Fruit Trees Using New Breeding Tools: Food Security and Climate Resilience
Abstract
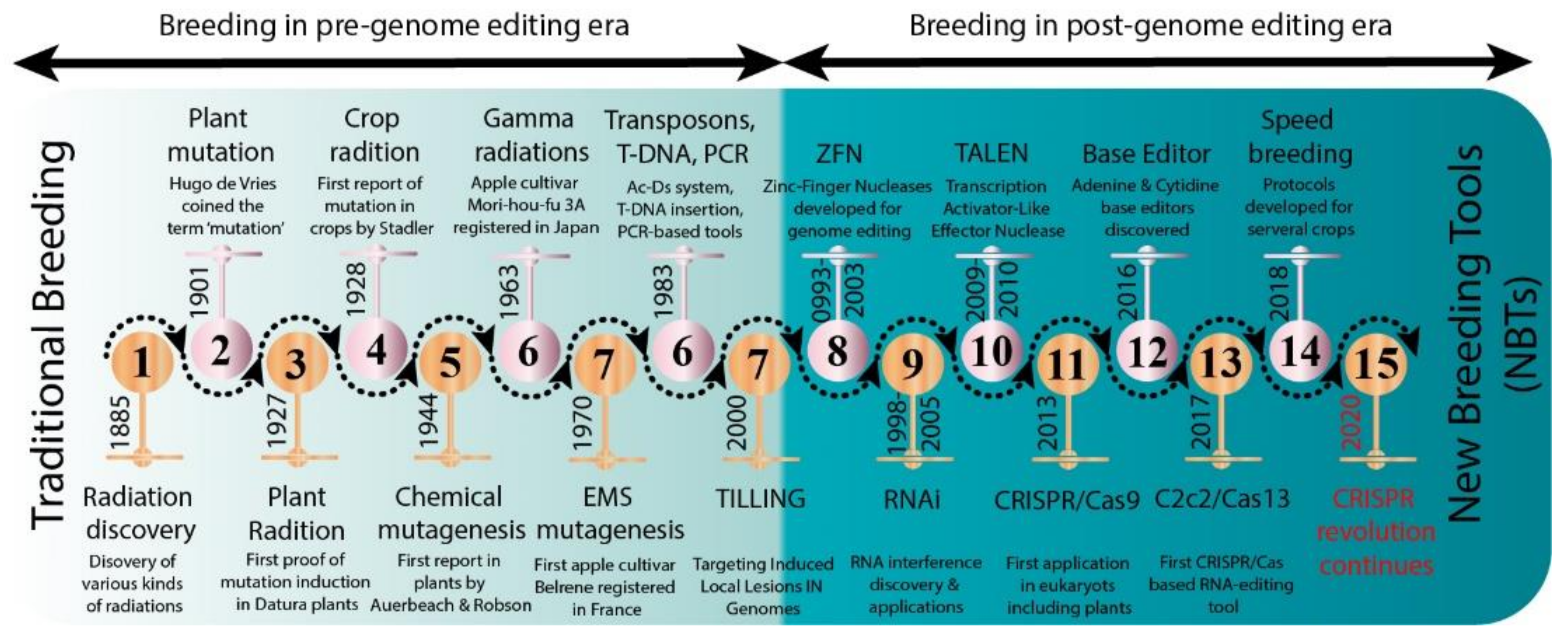
:1. Introduction
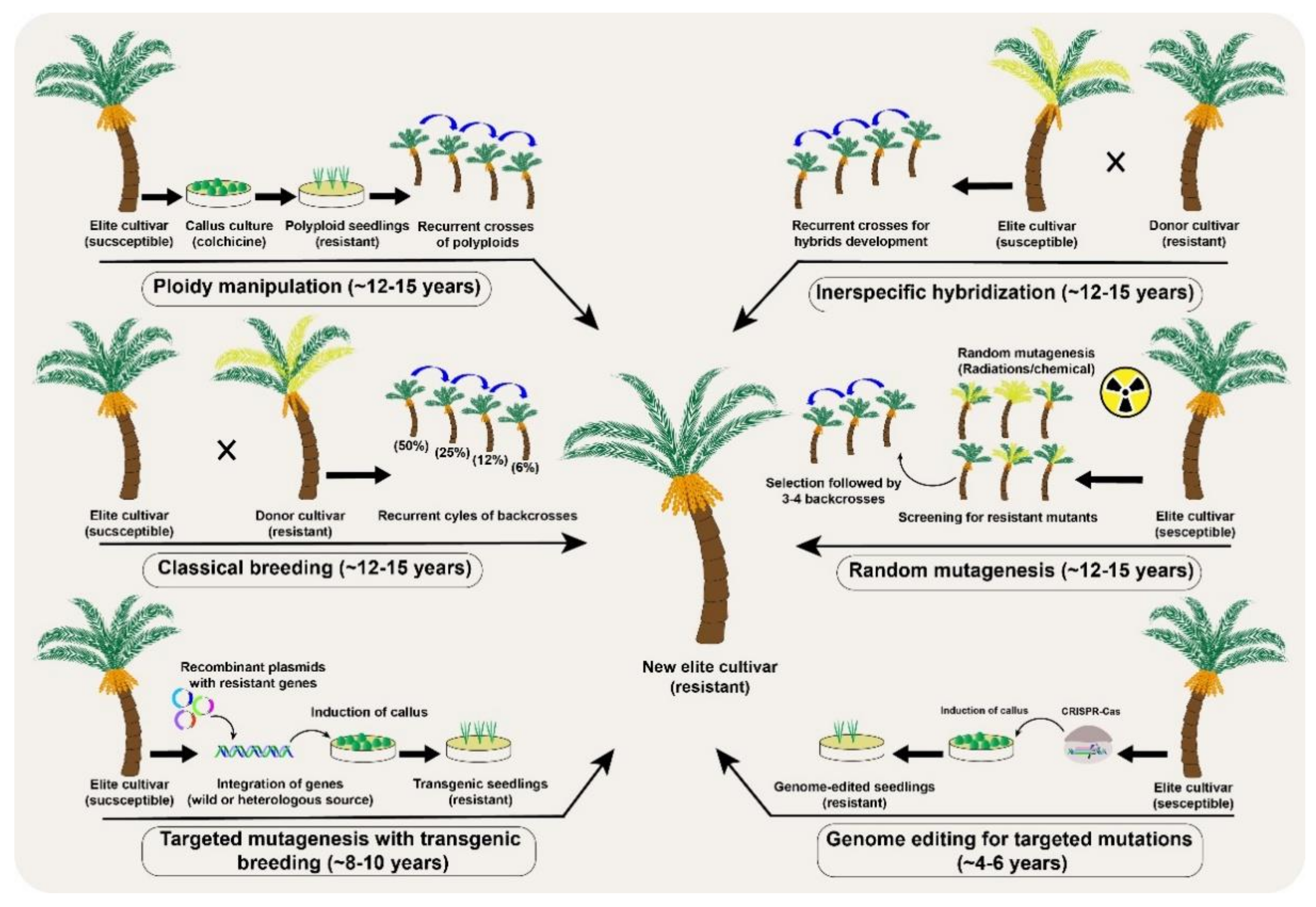
2. Tree Breeding under Climate Change
3. Mutagenesis as a Source of Genetic Variability in Tree Plants
TILLING as a Powerful Tool in Mutation Breeding
4. Genomics and Genetic Engineering Perspectives of Trees
5. Genome Editing in Precision Breeding
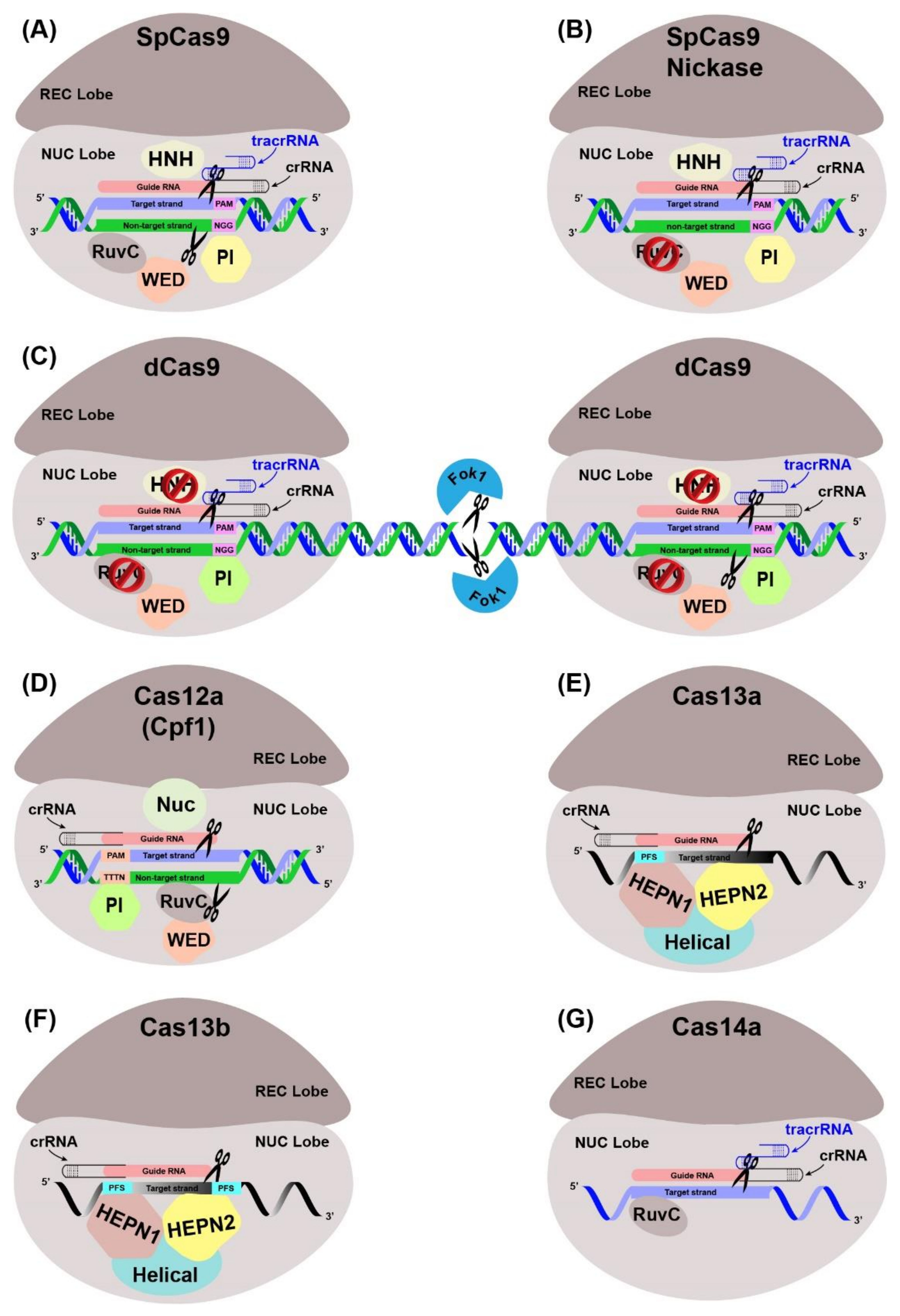
5.1. Principle and Types of CRISPR-Cas Systems
5.2. CRISPR/Cas Based GE Strategy in Trees
5.3. CRISPR-Mediated Genome Editing in Apples
5.4. CRISPR-Mediated Genome Editing in Banana
| Cas Type | Organism | Size (Amino Acids) | Class/ Type | PAM Site | Altered PAM | Types of End | Mutations | Plants | References |
|---|---|---|---|---|---|---|---|---|---|
| SpCas9 | Streptococcus pyogenes | 1368–1424 | 2/II | NGG | -- | Blunt/ds or ss | -- | Many plant species | [92,115,116,117] |
| SpCas9 VQR | S. pyogenes | 1372 | 2/II | NGA | Yes | Blunt/ds or ss | D1135V/R1335Q/T1337R | Rice | [118] |
| SpCas9 EQR | S. pyogenes | 1372 | 2/II | NGAG | Yes | Blunt/ds or ss | D1135E/R1335Q/T1337R | - | [118] |
| SpCas9 VRER | S. pyogenes | 1372 | 2/II | NGCG | Yes | Blunt/ds or ss | D1135V/G1218R/R1335E/T1337R | Rice | [118] |
| SpCas9 D1135E | S. pyogenes | 1372 | 2/II | NAG/NGA | Yes | Blunt/ds or ss | D1135E | - | [117] |
| SpCas9 QQR1 | S. pyogenes | 1372 | 2/II | NAAG | Yes | Blunt/ds or ss | G1218R/N1286Q/I1331F/D1332K/ R1333Q/R1335Q/T1337R | - | [119] |
| SpCas9-NG | S. pyogenes | 1372 | 2/II | NG | Yes | Blunt/ds or ss | R1335V/L1111R/D1135V/G1218R/ E1219F/A1322R/T1337R | Arabidopsis and rice | [120] |
| SpCas9-HF1 | S. pyogenes | 1368 | 2-II | NGG | Enhanced specificity | Blunt/ds or ss | N497A/R661A/Q695A/Q926A | Arabidopsis and rice | [121] |
| eHF1-Cas9 | S. pyogenes | 1368 | 2-II | NGG | Enhanced specificity | Blunt/ds or ss | N497A/R661A/Q695A/K848A/ Q926A/K1003A/R1060A | Rice | [122] |
| HiFi Cas9 | S. pyogenes | 1368 | 2-II | NGG | Enhanced specificity | Blunt/ds or ss | R691A | Rice | [123] |
| XCas9 | S. pyogenes | 1368 | 2-II | NG, GAA & GAT | Enhanced specificity | Blunt/ds or ss | A262T/R324L/S409I/E480K/ E543D/M694I/E1219V | Rice | [124] |
| dCas9 | S. pyogenes | 1368 | 2-II | NGG | No | Blunt/ds or ss | D10A/H840A | Arabidopsis and rice | [125,126] |
| nCas9 | S. pyogenes | 1368 | 2-II | NG, GAA & GAT | Enhanced specificity | Blunt/ds or ss | D10A | Rice, tobacco | [127,128] |
| SaCas9 | Staphylococcus aureus | 1053 | 2-II | NNGRRT | - | Blunt/ds or ss | - | Tobacco, rice, Arabidopsis and citrus | [129] |
| SaCas9-KKH | S. aureus | 1053 | 2-II | NNNRRT | Enhanced specificity | Blunt/ds or ss | E782K/N968K/R1015H | Tobacco, rice, Arabidopsis and citrus | [130] |
| BlatCas9 | Brevibacillus laterosporus | 1092 | 2-II | NNNNCND | Staggered/ds | - | Maize | [94] | |
| FnCas9 | Francisella novicida | 1629 | 2B-II | NGG | - | Staggered/ds | - | Arabidopsis | [131] |
| Cpf1 (Cas12a) | Prevotella & Franscisella | 1300 | 2-V | TTTN | - | Staggered/ds | - | Many plant species | [132,133] |
| AsCas12a RR | Acidaminococcus | 1307 | 2-V | TYCV & CCCC | Yes | Staggered/ds | S542R/K607R | - | [134,135] |
| AsCas12a RVR | Acidaminococcus | 1307 | 2-V | TATV | Yes | Staggered/ds | S542R/K548V/N552R | - | [135] |
| LbCas12a | Lachnospiraceae bacterium | 1228 | 2-V | TTTV | - | Staggered/ds | - | Many plant species | [134] |
| LbCas12a RR | Lachnospiraceae bacterium | 1228 | 2-V | TYCV & CCCC | Yes | Staggered/ds | G532R/K595R | Rice | [135] |
| LbCas12a RVR | Lachnospiraceae bacterium | 1228 | 2-V | TATV | Yes | Staggered/ds | G532R/K538V/Y542R | Rice | [135] |
| FnCas12a | F. novicida | 1300 | 2-V | TTV, TTTV & KYTV | - | Staggered/ds | - | Rice | [134] |
| FnCas12a RR | F. novicida | 1300 | 2-V | TYCV & TCTV | Yes | Staggered/ds | N607R/K671R | Rice | [136] |
| FnCas12a RVR | F. novicida | 1300 | 2-V | TWTV | Yes | Staggered/ds | N607R/K613V/N617R | Rice | [136] |
| Cas12b | Alicyclobacillus, Acidoterrestris, Bacillus Thermoamylovorans, A. acidiphilus | 1100–1300 | 2-VB | TTTN | - | Staggered/ds | - | Many plant species | [137,138] |
| Cas12X | Deltaproteobacteria | <1000 | 2-V | TTCN | - | Staggered/ds | - | - | [139,140] |
5.5. CRISPR-Mediated Genome Editing in Citrus
5.6. CRISPR-Mediated Genome Editing in Papaya
5.7. CRISPR-Mediated Genome Editing in Pear
5.8. Mitigation of Off-Target Mutations in GE Trees
5.8.1. GC Content of sgRNA
5.8.2. gRNA Length and Mismatches
5.8.3. Chemical Modification of gRNA
5.8.4. Concentration of Cas Protein/gRNA
5.8.5. Cas Protein Variants
| Tree Species | Gene Target | Trait Modified | CRISPR-Cas System | Reference |
|---|---|---|---|---|
| Apple | PDS | Albino phenotype | CRISPR-Cas9 | [102,104,106] |
| DIPM-1 DIPM-2 DIPM-4 | Fire blight | CRISPR-Cas9 | [103,107] | |
| TFL1 | Early flowering | CRISPR-Cas9 | [106] | |
| ALS | CRISPR-Cas9 | [39] | ||
| CNGC2 | B. dothidea resistance | CRISPR-Cas9 | [108] | |
| Detection of viruses and viroids | CRISPR-Cas12 | [109] | ||
| Banana | PDS | Albino phenotype | CRISPR-Cas9 | [113,114] |
| PDS | eBSV resistance | CRISPR-Cas9 | [141] | |
| PDS | Albino phenotype | CRISPR-Cas9 | [142] | |
| PDS | Albino phenotype | CRISPR-Cas12a | [142] | |
| ACO1 | Fruit ripening | CRISPR-Cas9 | [143] | |
| LCYε | β-carotene | CRISPR-Cas9 | [93] | |
| GA20ox2 | Gibberlic acid biosynthesis | CRISPR-Cas9 | [145] | |
| Cacao | TcNPR3 | Resistance to Phytophthora tropicalis | CRISPR-Cas9 | [177] |
| Citrus | PDS | Albino phenotype | CRISPR-Cas9 | [146,147] |
| LOB1 | Canker resistance | CRISPR-Cas9 | [149] | |
| PDS | Albino phenotype | CRISPR-Cas12 | [148] | |
| Papaya | Papain (PpalEPIC8) | Cysteine protease, P. palmivora resistance | CRISPR-Cas9 | [150] |
| Ppal15kDa | P. palmivora resistance | CRISPR-Cas9 | [151] | |
| Pear | TFL1 | Early flowering | CRISPR-Cas9 | [106] |
| PDS and ALS | Albino and chlorsulfuron | CRISPR-Cas9 C-to-T BE | [39] | |
| PbPAT14 | Dwarf and yellowing | CRISPR-Cas9 | [152] |
5.9. CRISPR Delivery Techniques and Vectors
5.10. Genome Editing Tools: Base, Prime and RNA Editors
5.10.1. Base Editors
5.10.2. Prime Editing
5.10.3. RNA Editing
6. Techniques to Estimate and Quantify the Mutation Rate
6.1. T7E1 Mismatch Cleavage Assay
6.2. High-Resolution Melting Assay
6.3. Sanger Sequencing
6.4. Next-Generation Sequencing
6.5. FLA-PCR (Fragment Analysis)
7. Critical Assessment of CRISPR-Cas Based GE in Fruit Trees
8. Biological and Regulatory Constraints to GE in Trees
9. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grattapaglia, D.; Silva-Junior, O.B.; Resende, R.T.; Cappa, E.P.; Muller, B.S.F.; Tan, B.Y.; Isik, F.; Ratcliffe, B.; El-Kassaby, Y.A. Quantitative genetics and genomics converge to accelerate forest tree breeding. Front. Plant Sci. 2018, 9, 1693. [Google Scholar] [CrossRef]
- Burdon, R.D.; Klapste, J. Alternative selection methods and explicit or implied economic-worth functions for different traits in tree breeding. Tree Genet. Genom. 2019, 15, 79. [Google Scholar] [CrossRef]
- Hough, L.F.; Shay, J.R.; Dayton, D.F. Apple Scab Resistance from Malus-Floribunda Sieb. Proc. Am. Soc. Hort. Sci. 1953, 62, 341–347. [Google Scholar]
- Schouten, H.J.; Krens, F.A.; Jacobsen, E. Cisgenic plants are similar to traditionally bred plants—International regulations for genetically modified organisms should be altered to exempt cisgenesis. EMBO Rep. 2006, 7, 750–753. [Google Scholar] [CrossRef] [Green Version]
- Semagn, K.; Bjornstad, A.; Ndjiondjop, M.N. Progress and prospects of marker assisted backcrossing as a tool in crop breeding programs. Afr. J. Biotechnol. 2006, 5, 2588–2603. [Google Scholar]
- Moose, S.P.; Mumm, R.H. Molecular plant breeding as the foundation for 21st century crop improvement. Plant Physiol. 2008, 147, 969–977. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Omura, M.; Shimada, T. Citrus breeding, genetics and genomics in Japan. Breed. Sci. 2016, 66, 3–17. [Google Scholar] [CrossRef] [Green Version]
- Feng, S.J.; Zhang, J.P.; Mu, Z.H.; Wang, Y.J.; Wen, C.L.; Wu, T.; Yu, C.; Li, Z.; Wang, H.S. Recent progress on the molecular breeding of Cucumis sativus L. in China. Theor. Appl. Genet. 2020, 133, 1777–1790. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Fujii, H.; Omura, M.; Shimada, T. Fast-track breeding system to introduce CTV resistance of trifoliate orange into citrus germplasm, by integrating early flowering transgenic plants with marker-assisted selection. BMC Plant Biol. 2020, 20, 224. [Google Scholar] [CrossRef]
- Elo, A.; Lemmetyinen, J.; Novak, A.; Keinonen, K.; Porali, I.; Hassinen, M.; Sopanen, T. BpMADS4 has a central role in inflorescence initiation in silver birch (Betula pendula). Physiol. Plant. 2007, 131, 149–158. [Google Scholar] [CrossRef]
- Schlathölter, I.; Jänsch, M.; Flachowsky, H.; Broggini, G.A.L.; Hanke, M.-V.; Patocchi, A. Generation of advanced fire blight-resistant apple (Malus × domestica) selections of the fifth generation within 7 years of applying the early flowering approach. Planta 2018, 247, 1475–1488. [Google Scholar] [CrossRef] [Green Version]
- Stadler, L.J. Genetic effects of x rays in maize. Proc. Natl. Acad. Sci. USA 1928, 14, 69–75. [Google Scholar] [CrossRef] [Green Version]
- Acquaah, G. Principles of Plant Genetics and Breeding; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Sink, K.; Jain, R.; Chowdhury, J. Somatic cell hybridization. In Distant Hybridization of Crop Plants; Springer: Berlin/Heidelberg, Germany, 1992; pp. 168–198. [Google Scholar]
- Kohli, A.; Christou, P. Stable transgenes bear fruit. Nat. Biotechnol. 2008, 26, 653–654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ammann, K. Genomic misconception: A fresh look at the biosafety of transgenic and conventional crops. A plea for a process agnostic regulation. New Biotechnol. 2014, 31, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Twyman, R.M.; Kohli, A.; Stoger, E.; Christou, P. Foreign DNA: Integration and expression in transgenic plants. In Genetic Engineering: Principles and Methods; Setlow, J.K., Ed.; Springer: Boston, MA, USA, 2002; pp. 107–136. [Google Scholar]
- Ghogare, R.; Williamson-Benavides, B.; Ramirez-Torres, F.; Dhingra, A. CRISPR-associated nucleases: The Dawn of a new age of efficient crop improvement. Transgenic Res. 2020, 29, 1–35. [Google Scholar] [CrossRef]
- Zhu, C.; Bortesi, L.; Baysal, C.; Twyman, R.M.; Fischer, R.; Capell, T.; Schillberg, S.; Christou, P. Characteristics of genome editing mutations in cereal crops. Trends Plant Sci. 2017, 22, 38–52. [Google Scholar] [CrossRef]
- Monsur, M.B.; Shao, G.; Lv, Y.; Ahmad, S.; Wei, X.; Hu, P.; Tang, S. Base editing: The ever expanding clustered regularly interspaced short palindromic repeats (CRISPR) tool kit for precise genome editing in plants. Genes 2020, 11, 466. [Google Scholar] [CrossRef]
- Quispe-Huamanquispe, D.G.; Gheysen, G.; Kreuze, J.F. Horizontal gene transfer contributes to plant evolution: The case of Agrobacterium T-DNAs. Front. Plant Sci. 2017, 8, 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isabel, N.; Holliday, J.A.; Aitken, S.N. Forest genomics: Advancing climate adaptation, forest health, productivity, and conservation. Evol. Appl. 2020, 13, 3–10. [Google Scholar] [CrossRef] [Green Version]
- Sullivan, M.J.P.; Lewis, S.L.; Affum-Baffoe, K.; Castilho, C.; Costa, F.; Sanchez, A.C.; Ewango, C.E.N.; Hubau, W.; Marimon, B.; Monteagudo-Mendoza, A.; et al. Long-term thermal sensitivity of Earth’s tropical forests. Science 2020, 368, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Cortes, A.J.; Restrepo-Montoya, M.; Bedoya-Canas, L.E. Modern strategies to assess and breed forest tree adaptation to changing climate. Front. Plant Sci. 2020, 11, 583323. [Google Scholar] [CrossRef] [PubMed]
- Neale, D.B.; Kremer, A. Forest tree genomics: Growing resources and applications. Nat. Rev. Genet. 2011, 12, 111–122. [Google Scholar] [CrossRef] [PubMed]
- White, T.L.; Adams, W.T.; Neale, D.B. Forest Genetics; Cabi: Wallingford, UK, 2007. [Google Scholar]
- Seehausen, O. Hybridization and adaptive radiation. Trends Ecol. Evol. 2004, 19, 198–207. [Google Scholar] [CrossRef]
- Burkhart, H.E.; Brunner, A.M.; Stanton, B.J.; Shuren, R.A.; Amateis, R.L.; Creighton, J.L. An assessment of potential of hybrid poplar for planting in the Virginia Piedmont. New For. 2017, 48, 479–490. [Google Scholar] [CrossRef]
- Cipollini, M.; Dingley, N.R.; Felch, P.; Maddox, C. Evaluation of phenotypic traits and blight-resistance in an American chestnut backcross orchard in Georgia. Glob. Ecol. Conserv. 2017, 10, 1–8. [Google Scholar] [CrossRef]
- Badenes, M.L.; Fernández, I.; Martí, A.; Ríos, G.; Rubio-Cabetas, M.J. Application of genomic technologies to the breeding of trees. Front. Genet. 2016, 7, 198. [Google Scholar] [CrossRef] [Green Version]
- Lebedev, V.G.; Lebedeva, T.N.; Chernodubov, A.I.; Shestibratov, K.A. Genomic selection for forest tree improvement: Methods, achievements and perspectives. Forests 2020, 11, 1190. [Google Scholar] [CrossRef]
- Thistlethwaite, F.R.; Ratcliffe, B.; Klápště, J.; Porth, I.; Chen, C.; Stoehr, M.U.; El-Kassaby, Y.A. Genomic selection of juvenile height across a single-generational gap in Douglas-fir. Heredity 2019, 122, 848–863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isik, F.; Bartholomé, J.; Farjat, A.; Chancerel, E.; Raffin, A.; Sanchez, L.; Plomion, C.; Bouffier, L. Genomic selection in maritime pine. Plant Sci. 2016, 242, 108–119. [Google Scholar] [CrossRef]
- Kaiser, N.; Douches, D.; Dhingra, A.; Glenn, K.C.; Herzig, P.R.; Stowe, E.C.; Swarup, S. The role of conventional plant breeding in ensuring safe levels of naturally occurring toxins in food crops. Trends Food Sci. Technol. 2020, 100, 51–66. [Google Scholar] [CrossRef]
- Iwata, H.; Minamikawa, M.F.; Kajiya-Kanegae, H.; Ishimori, M.; Hayashi, T. Genomics-assisted breeding in fruit trees. Breed. Sci. 2016, 66, 100–115. [Google Scholar] [CrossRef] [Green Version]
- Te Beest, M.; Le Roux, J.J.; Richardson, D.M.; Brysting, A.K.; Suda, J.; Kubesova, M.; Pysek, P. The more the better? The role of polyploidy in facilitating plant invasions. Ann. Bot. 2012, 109, 19–45. [Google Scholar] [CrossRef] [Green Version]
- Lamo, K.; Bhat, D.J.; Kour, K.; Solanki, S.P.S. Mutation studies in fruit crops: A review. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 3620–3633. [Google Scholar] [CrossRef]
- Zonneveld, B.J.M.; Pollock, W.I. Flow cytometric analysis of somaclonal variation in lineages of Hosta sports detects polyploidy and aneuploidy chimeras. Plant Biol. 2012, 14, 972–979. [Google Scholar] [CrossRef]
- Malabarba, J.; Chevreau, E.; Dousset, N.; Veillet, F.; Moizan, J.; Vergne, E. New strategies to overcome present CRISPR/Cas9 limitations in apple and pear: Efficient dechimerization and base editing. Int. J. Mol. Sci. 2020, 22, 319. [Google Scholar] [CrossRef]
- Gambino, G.; Moine, A.; Boccacci, P.; Perrone, I.; Pagliarani, C. Somatic embryogenesis is an effective strategy for dissecting chimerism phenomena in Vitis vinifera cv Nebbiolo. Plant Cell Rep. 2021, 40, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Kumawat, S.; Rana, N.; Bansal, R.; Vishwakarma, G.; Mehetre, S.T.; Das, B.K.; Kumar, M.; Yadav, S.K.; Sonah, H.; Sharma, T.R.; et al. Expanding avenue of fast neutron mediated mutagenesis for crop improvement. Plants 2019, 8, 164. [Google Scholar] [CrossRef] [Green Version]
- Predieri, S. Mutation induction and tissue culture in improving fruits. Plant Cell Tissue Organ Cult. 2001, 64, 185–210. [Google Scholar] [CrossRef]
- Lokko, Y.; Amoatey, H. Improvement of Pineapple Using in Vitro and Mutation Breeding Techniques; Biotechnology and Nuclear Agriculture Research Institute: Legon-Accra, Ghana, 2001; pp. 25–29. [Google Scholar]
- International Atomic Energy Agency. In Vitro Techniques for Selection of Radiation Induced Mutations Adapted to Adverse Environmental Conditions. In Proceedings of a Final Research Coordination Meeting; Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture: Vienna, Austria, 2001; pp. 1–96. [Google Scholar]
- Mba, C.; Afza, R.; Bado, S.; Mohan Jain, S. Induced mutagenesis in plants using physical and chemical agents. Plant Cell Cult. Essent. Methods 2010, 20, 111–130. [Google Scholar]
- Elhiti, M.; Wang, H.Y.; Austin, R.S.; Chen, B.; Brown, D.; Wang, A.M. Generation of chemically induced mutations using in vitro propagated shoot tip tissues for genetic improvement of fruit trees. Plant Cell Tissue Organ Cult. 2016, 124, 447–452. [Google Scholar] [CrossRef]
- Alvarez, D.; Cerda-Bennasser, P.; Stowe, E.; Ramirez-Torres, F.; Capell, T.; Dhingra, A.; Christou, P. Fruit crops in the era of genome editing: Closing the regulatory gap. Plant Cell Rep. 2021, 40, 915–930. [Google Scholar] [CrossRef] [PubMed]
- Sikora, P.; Chawade, A.; Larsson, M.; Olsson, J.; Olsson, O. Mutagenesis as a tool in plant genetics, functional genomics, and breeding. Int. J. Plant Genom. 2011, 2011, 314829. [Google Scholar] [CrossRef] [Green Version]
- Kumawat, S.; Rana, N.; Bansal, R.; Vishwakarma, G.; Mehetre, S.; Das, B.K.; Kumar, M.; Yadav, S.; Sonah, H.; Sharma, T.R.; et al. Fast neutron mutagenesis in plants: Advances, applicability and challenges. Plant Sci. 2019. [Google Scholar] [CrossRef]
- Spengler, R.N. Origins of the apple: The role of megafaunal mutualism in the domestication of Malus and rosaceous trees. Front. Plant Sci. 2019, 10, 617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, G.A.; Prochnik, S.; Jenkins, J.; Salse, J.; Hellsten, U.; Murat, F.; Perrier, X.; Ruiz, M.; Scalabrin, S.; Terol, T.; et al. Sequencing of diverse mandarin, pummelo and orange genomes reveals complex history of admixture during citrus domestication. Nat. Biotechnol. 2014, 32, 656–662. [Google Scholar] [CrossRef]
- Crosby, J.A.; Janick, J.; Pecknold, P.C.; Korban, S.S.; Oconnor, P.A.; Ries, S.M.; Goffreda, J.; Voordeckers, A. Breeding apples for scab resistance: 1945–1990. Fruit Var. J. 1992, 46, 145–166. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Ivancic, A.; Schmitzer, V.; Veberic, R.; Stampar, F. Comparison of major taste compounds and antioxidative properties of fruits and flowers of different Sambucus species and interspecific hybrids. Food Chem. 2016, 200, 134–140. [Google Scholar] [CrossRef]
- Rajan, R.P.; Singh, G. A review on application of somaclonal variation in important horticulture crops. Plant Cell Biotechnol. Mol. Biol. 2021, 22, 161–175. [Google Scholar]
- Ranghoo-Sanmukhiya, V.M. Somaclonal variation and methods used for its detection. In Propagation and Genetic Manipulation of Plants; Siddique, I., Ed.; Springer: Singapore, 2021; pp. 1–18. [Google Scholar]
- Orbović, V.; Ćalović, M.; Viloria, Z.; Nielsen, B.; Gmitter, F.G.; Castle, W.S.; Grosser, J.W. Analysis of genetic variability in various tissue culture-derived lemon plant populations using RAPD and flow cytometry. Euphytica 2008, 161, 329–335. [Google Scholar] [CrossRef]
- Cevallos-Cevallos, J.M.; Jines, C.; Maridueña-Zavala, M.G.; Molina-Miranda, M.J.; Ochoa, D.E.; Flores-Cedeno, J.A. GC-MS metabolite profiling for specific detection of dwarf somaclonal variation in banana plants. Appl. Plant Sci. 2018, 6, e01194. [Google Scholar] [CrossRef] [Green Version]
- Henao-Ramírez, A.M.; Salazar Duque, H.J.; Calle Tobón, A.F.; Urrea Trujillo, A.I. Determination of genetic stability in cacao plants (Theobroma Cacao L.) derived from somatic embryogenesis using microsatellite molecular markers (SSR). Int. J. Fruit Sci. 2021, 21, 284–298. [Google Scholar] [CrossRef]
- Mba, C. Induced mutations unleash the potentials of plant genetic resources for food and agriculture. Agronomy 2013, 3, 200–231. [Google Scholar] [CrossRef] [Green Version]
- McCallum, C.M.; Comai, L.; Greene, E.A.; Henikoff, S. Targeted screening for induced mutations. Nat. Biotechnol. 2000, 18, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Jankowicz-Cieslak, J.; Mba, C.; Till, B.J. Mutagenesis for crop breeding and functional genomics. In Biotechnologies for Plant Mutation Breeding; Springer: Cham, Switzerland, 2017; pp. 3–18. [Google Scholar]
- Sato, Y.; Sato, K.; Nishio, T. Interspecific pairs of class II S haplotypes having different recognition specificities between Brassica oleracea and Brassica rapa. Plant Cell Physiol. 2006, 47, 340–345. [Google Scholar] [CrossRef] [Green Version]
- Acevedo-Garcia, J.; Spencer, D.; Thieron, H.; Reinstädler, A.; Hammond-Kosack, K.; Phillips, A.L.; Panstruga, R. mlo-based powdery mildew resistance in hexaploid bread wheat generated by a non-transgenic TILLING approach. Plant Biotechnol. J. 2017, 15, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.; Howell, T.; Nitcher, R.; Missirian, V.; Watson, B.; Ngo, K.J.; Lieberman, M.; Fass, J.; Uauy, C.; Tran, R.K.; et al. Discovery of rare mutations in populations: TILLING by sequencing. Plant Physiol. 2011, 156, 1257–1268. [Google Scholar] [CrossRef] [Green Version]
- Taheri, S.; Abdullah, T.L.; Jain, S.M.; Sahebi, M.; Azizi, P. TILLING, high-resolution melting (HRM), and next-generation sequencing (NGS) techniques in plant mutation breeding. Mol. Breed. 2017, 37, 40. [Google Scholar] [CrossRef]
- Gilchrist, E.J.; Sidebottom, C.H.D.; Koh, C.S.; MacInnes, T.; Sharpe, A.G.; Haughn, G.W. A mutant Brassica napus (Canola) population for the identification of new genetic diversity via TILLING and next generation sequencing. PLoS ONE 2013, 8, e84303. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Hao, L.; Parry, M.A.J.; Phillips, A.L.; Hu, Y.-G. Progress in TILLING as a tool for functional genomics and improvement of crops. J. Integrat. Plant Biol. 2014, 56, 425–443. [Google Scholar] [CrossRef] [Green Version]
- Freeman, J.S.; Potts, B.M.; Downes, G.M.; Pilbeam, D.; Thavamanikumar, S.; Vaillancourt, R.E. Stability of quantitative trait loci for growth and wood properties across multiple pedigrees and environments in Eucalyptus globulus. New Phytol. 2013, 198, 1121–1134. [Google Scholar] [CrossRef] [Green Version]
- Osakabe, Y.; Sugano, S.S.; Osakabe, K. Genome engineering of woody plants: Past, present and future. J. Wood Sci. 2016, 62, 217–225. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Li, S.; Zhang, D.; Han, M.; Jin, X.; Zhao, C.; Wang, S.; Xing, L.; Ma, J.; Ji, J.; et al. Sequencing of a wild apple (Malus baccata) genome unravels the differences between cultivated and wild apple species regarding disease resistance and cold tolerance. G3-Genes Genom. Genet. 2019, 9, 2051–2060. [Google Scholar] [CrossRef] [Green Version]
- Torales, S.L.; El Mujtar, V.; Marcucci-Poltri, S.; Pomponio, F.; Soliani, C.; Villalba, P.; Estravis-Barcala, M.; Klein, L.; García, M.; Pentreath, V.; et al. Application of high-throughput sequencing technologies in native forest tree species in Argentina: Implications for breeding. In Low Intensity Breeding of Native Forest Trees in Argentina: Genetic Basis for their Domestication and Conservation; Pastorino, M.J., Marchelli, P., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 455–482. [Google Scholar]
- Ricroch, A.E.; Henard-Damave, M.C. Next biotech plants: New traits, crops, developers and technologies for addressing global challenges. Crit. Rev. Biotechnol. 2016, 36, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Yabor, L.; Perez, L.; Gomez, D.; Villalobos-Olivera, A.; Mendoza, J.R.; Martinez, J.; Escalante, D.; Garro, G.; Hajari, E.; Lorenzo, J.C. Histological evaluation of pineapple transgenic plants following 8 years of field growth. Euphytica 2020, 216, 23. [Google Scholar] [CrossRef]
- Ault, K.; Viswanath, V.; Jayawickrama, J.; Ma, C.; Eaton, J.; Meilan, R.; Beauchamp, G.; Hohenschuh, W.; Murthy, G.; Strauss, S.H. Improved growth and weed control of glyphosate-tolerant poplars. New For. 2016, 47, 653–667. [Google Scholar] [CrossRef] [Green Version]
- Gonsalves, D.; Tripathi, S.; Carr, J.B.; Suzuki, J.Y. Papaya ringspot virus. Plant Health Inst. 2010, 10, 1094. [Google Scholar] [CrossRef]
- Carter, N. Petition for Determination of Nonregulated Status: Arctic™ Apple (Malus × domestica) Events GD743 and GS784; United States Department of Agriculture—Animal and Plant Health Inspection Service: Summerland, BC, Canada, 2012.
- Stowe, E.; Dhingra, A. Development of the Arctic® Apple. Plant Breed. Rev. 2021, 44, 273–296. [Google Scholar]
- Scorza, R.; Callahan, A.; Ravelonandro, M.; Braverman, M. Development and regulation of the Plum pox virus resistant transgenic plum “HoneySweet”. In Regulation of Agricultural Biotechnology: The United States and Canada; Springer: Berlin/Heidelberg, Germany, 2012; pp. 269–280. [Google Scholar]
- Xu, K. An overview of Arctic apples: Basic facts and characteristics. NY Fruit Q. 2013, 21, 8–10. [Google Scholar]
- Food and Drug Administration (FDA). Biotechnology Consultations on Food from GE Plant Varieties; Food and Drug Administration (FDA): Silver Spring, MD, USA, 2017.
- Hu, L.; Lu, H.; Liu, Q.; Chen, X.; Jiang, X. Overexpression of mtlD gene in transgenic Populus tomentosa improves salt tolerance through accumulation of mannitol. Tree Physiol. 2005, 25, 1273–1281. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.L.; Su, X.H.; Zhang, B.Y.; Huang, Q.J.; Zhang, X.H.; Huang, R.F. Expression of jasmonic ethylene responsive factor gene in transgenic poplar tree leads to increased salt tolerance. Tree Physiol. 2009, 29, 273–279. [Google Scholar] [CrossRef] [Green Version]
- Randle, M.; Tennant, P. Transgenic Papaya. In Genetically Modified Crops: Current Status, Prospects and Challenges; Kavi Kishor, P.B., Rajam, M.V., Pullaiah, T., Eds.; Springer: Singapore, 2021; Volume 2, pp. 129–160. [Google Scholar]
- Vadlamudi, T.; Patil, B.L.; Kaldis, A.; Sai Gopal, D.V.R.; Mishra, R.; Berbati, M.; Voloudakis, A. DsRNA-mediated protection against two isolates of Papaya ringspot virus through topical application of dsRNA in papaya. J. Virol. Meth. 2020, 275, 113750. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, E.; Nanto, K.; Oishi, M.; Ebinuma, H.; Morishita, Y.; Sakurai, N.; Suzuki, H.; Shibata, D.; Shimada, T. Agrobacterium-mediated transformation of Eucalyptus globulus using explants with shoot apex with introduction of bacterial choline oxidase gene to enhance salt tolerance. Plant Cell Rep. 2012, 31, 225–235. [Google Scholar] [CrossRef]
- Kweon, J.; Kim, D.-E.; Jang, A.-H.; Kim, Y. CRISPR/Cas-based customization of pooled CRISPR libraries. PLoS ONE 2018, 13, e0199473. [Google Scholar] [CrossRef]
- Veillet, F.; Durand, M.; Kroj, T.; Cesari, S.; Gallois, J.-L. Precision Breeding made real with CRISPR: Illustration through genetic resistance to pathogens. Plant Commun. 2020, 1, 100102. [Google Scholar] [CrossRef]
- Horvath, P.; Barrangou, R. CRISPR/Cas, the immune system of bacteria and archaea. Science 2010, 327, 167–170. [Google Scholar] [CrossRef] [Green Version]
- Makarova, K.S.; Wolf, Y.I.; Iranzo, J.; Shmakov, S.A.; Alkhnbashi, O.S.; Brouns, S.J.J.; Charpentier, E.; Cheng, D.; Haft, D.H.; Horvath, P.; et al. Evolutionary classification of CRISPR–Cas systems: A burst of class 2 and derived variants. Nat. Rev. Microbiol. 2020, 18, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Makarova, K.S.; Koonin, E.V. Annotation and classification of CRISPR-Cas systems. In Methods in Molecular Biology; Lundgren, M., Emmanuelle, C., Peter, F., Eds.; Springer Science+Business Media: New York, NY, USA, 2015; Volume 1311, pp. 47–75. [Google Scholar]
- Hille, F.; Richter, H.; Wong, S.P.; Bratovič, M.; Ressel, S.; Charpentier, E. The biology of CRISPR-Cas: Backward and forward. Cell 2018, 172, 1239–1259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A programmable dual-RNA–guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Deveau, H.; Barrangou, R.; Garneau, J.E.; Labonté, J.; Fremaux, C.; Boyaval, P.; Romero, D.A.; Horvath, P.; Moineau, S. Phage response to CRISPR-encoded resistance in Streptococcus thermophilus. J. Bacteriol. 2008, 190, 1390–1400. [Google Scholar] [CrossRef] [Green Version]
- Karvelis, T.; Gasiunas, G.; Young, J.; Bigelyte, G.; Silanskas, A.; Cigan, M.; Siksnys, V. Rapid characterization of CRISPR-Cas9 protospacer adjacent motif sequence elements. Genome Biol. 2015, 16, 253. [Google Scholar] [CrossRef] [Green Version]
- Symington, L.S.; Gautier, J. Double-strand break end resection and repair pathway choice. Annu. Rev. Genet. 2011, 45, 247–271. [Google Scholar] [CrossRef] [PubMed]
- Budman, J.; Chu, G. Processing of DNA for nonhomologous end-joining by cell-free extract. EMBO J. 2005, 24, 849–860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gong, C.; Bongiorno, P.; Martins, A.; Stephanou, N.C.; Zhu, H.; Shuman, S.; Glickman, M.S. Mechanism of nonhomologous end-joining in mycobacteria: A low-fidelity repair system driven by Ku, ligase D and ligase C. Nat. Struct. Mol. Biol. 2005, 12, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Burstein, D.; Harrington, L.B.; Strutt, S.C.; Probst, A.J.; Anantharaman, K.; Thomas, B.C.; Doudna, J.A.; Banfield, J.F. New CRISPR-Cas systems from uncultivated microbes. Nature 2017, 542, 237–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shmakov, S.; Smargon, A.; Scott, D.; Cox, D.; Pyzocha, N.; Yan, W.; Abudayyeh, O.O.; Gootenberg, J.S.; Makarova, K.S.; Wolf, Y.I.; et al. Diversity and evolution of class 2 CRISPR–Cas systems. Nat. Rev. Microbiol. 2017, 15, 169–182. [Google Scholar] [CrossRef] [Green Version]
- Koonin, E.V.; Makarova, K.S.; Zhang, F. Diversity, classification and evolution of CRISPR-Cas systems. Curr. Opin. Microbiol. 2017, 37, 67–78. [Google Scholar] [CrossRef]
- FAOSTAT. Food and Agriculture Organization of the United Nations Statistics Division. 2021. Available online: http://faostat3.fao.org/home/E (accessed on 28 January 2021).
- Nishitani, C.; Hirai, N.; Komori, S.; Wada, M.; Okada, K.; Osakabe, K.; Yamamoto, T.; Osakabe, Y. Efficient genome editing in apple using a CRISPR/Cas9 system. Sci. Rep. 2016, 6, 1–8. [Google Scholar]
- Malnoy, M.; Viola, R.; Jung, M.H.; Koo, O.J.; Kim, S.; Kim, J.S.; Velasco, R.; Nagamangala Kanchiswamy, C. DNA-free genetically edited grapevine and apple protoplast using CRISPR/Cas9 ribonucleoproteins. Front. Plant Sci. 2016, 7, 1904. [Google Scholar] [CrossRef]
- Osakabe, Y.; Liang, Z.; Ren, C.; Nishitani, C.; Osakabe, K.; Wada, M.; Komori, S.; Malnoy, M.; Velasco, R.; Poli, M.; et al. CRISPR–Cas9-mediated genome editing in apple and grapevine. Nat. Protoc. 2018, 13, 2844–2863. [Google Scholar] [CrossRef] [PubMed]
- Chevreau, E.; Dousset, N.; Joffrion, C.; Richer, A.; Charrier, A.; Vergne, E. Agroinfiltration is a key factor to improve the efficiency of apple and pear transformation. Sci. Hortic. 2019, 251, 150–154. [Google Scholar] [CrossRef]
- Charrier, A.; Vergne, E.; Dousset, N.; Richer, A.; Petiteau, A.; Chevreau, E. Efficient targeted mutagenesis in apple and first time edition of pear using the CRISPR-Cas9 system. Front. Plant Sci. 2019, 10, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pompili, V.; Dalla Costa, L.; Piazza, S.; Pindo, M.; Malnoy, M. Reduced fire blight susceptibility in apple cultivars using a high-efficiency CRISPR/Cas9-FLP/FRT-based gene editing system. Plant Biotechnol. J. 2020, 18, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Bai, S.; Wang, N.; Sun, X.; Zhang, Y.; Zhu, J.; Dong, C. CRISPR/Cas9-mediated mutagenesis of MdCNGC2 in apple callus and VIGS-mediated silencing of MdCNGC2 in fruits improve resistance to Botryosphaeria dothidea. Front. Plant Sci. 2020, 11, 575477. [Google Scholar] [CrossRef]
- Jiao, J.; Kong, K.; Han, J.; Song, S.; Bai, T.; Song, C.; Wang, M.; Yan, Z.; Zhang, H.; Zhang, R.; et al. Field detection of multiple RNA viruses/viroids in apple using a CRISPR/Cas12a-based visual assay. Plant Biotechnol. J. 2021, 19, 394–405. [Google Scholar] [CrossRef] [PubMed]
- Picq, C. Bananas; International Network for the Improvement of Banana and Plantain (INIBAP): Montpellier, France, 2000; Volume 14, p. 14. [Google Scholar]
- Perrier, X.; De Langhe, E.; Donohue, M.; Lentfer, C.; Vrydaghs, L.; Bakry, F.; Carreel, F.; Hippolyte, I.; Horry, J.-P.; Jenny, C.; et al. Multidisciplinary perspectives on banana (Musa spp.) domestication. Proc. Natl. Acad. Sci. USA 2011, 108, 11311–11318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dash, P.K.; Rai, R. Translating the “banana genome” to delineate stress resistance, dwarfing, parthenocarpy and mechanisms of fruit ripening. Front. Plant Sci. 2016, 7, 1543. [Google Scholar] [CrossRef] [Green Version]
- Kaur, N.; Alok, A.; Shivani, M.; Pandey, P.; Awasthi, P.; Tiwari, S. CRISPR/Cas9-mediated efficient editing in phytoene desaturase (PDS) demonstrates precise manipulation in banana cv. Rasthali genome. Funct. Integr. Genom. 2018, 18, 89–99. [Google Scholar] [CrossRef]
- Naim, F.; Dugdale, B.; Kleidon, J.; Brinin, A.; Shand, K.; Waterhouse, P.; Dale, J. Gene editing the phytoene desaturase alleles of Cavendish banana using CRISPR/Cas9. Transgenic Res. 2018, 27, 451–460. [Google Scholar] [CrossRef] [Green Version]
- Makarova, K.S.; Wolf, Y.I.; Alkhnbashi, O.S.; Costa, F.; Shah, S.A.; Saunders, S.J.; Barrangou, R.; Brouns, S.J.J.; Charpentier, E.; Haft, D.H.; et al. An updated evolutionary classification of CRISPR–Cas systems. Nat. Rev. Microbiol. 2015, 13, 722–736. [Google Scholar] [CrossRef] [Green Version]
- Barrangou, R.; Fremaux, C.; Deveau, H.; Richards, M.; Boyaval, P.; Moineau, S.; Romero, D.A.; Horvath, P. CRISPR provides acquired resistance against viruses in prokaryotes. Science 2007, 315, 1709–1713. [Google Scholar] [CrossRef]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A. Multiplex genome engineering using CRISPR/Cas systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef] [Green Version]
- Kleinstiver, B.P.; Prew, M.S.; Tsai, S.Q.; Topkar, V.V.; Nguyen, N.T.; Zheng, Z.; Gonzales, A.P.W.; Li, Z.; Peterson, R.T.; Yeh, J.-R.J.; et al. Engineered CRISPR-Cas9 nucleases with altered PAM specificities. Nature 2015, 523, 481–485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anders, C.; Bargsten, K.; Jinek, M. Structural Plasticity of PAM Recognition by Engineered Variants of the RNA-Guided Endonuclease Cas9. Mol. Cell 2016, 61, 895–902. [Google Scholar] [CrossRef] [Green Version]
- Nishimasu, H.; Shi, X.; Ishiguro, S.; Gao, L.; Hirano, S.; Okazaki, S.; Noda, T.; Abudayyeh, O.O.; Gootenberg, J.S.; Mori, H.; et al. Engineered CRISPR-Cas9 nuclease with expanded targeting space. Science 2018, 361, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Kleinstiver, B.P.; Pattanayak, V.; Prew, M.S.; Tsai, S.Q.; Nguyen, N.T.; Zheng, Z.; Joung, J.K. High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 2016, 529, 490–495. [Google Scholar] [CrossRef] [Green Version]
- Liang, Z.; Chen, K.; Yan, Y.; Zhang, Y.; Gao, C. Genotyping genome-edited mutations in plants using CRISPR ribonucleoprotein complexes. Plant Biotechnol. J. 2018, 16, 2053–2062. [Google Scholar] [CrossRef] [PubMed]
- Vakulskas, C.A.; Dever, D.P.; Rettig, G.R.; Turk, R.; Jacobi, A.M.; Collingwood, M.A.; Bode, N.M.; McNeill, M.S.; Yan, S.; Camarena, J.; et al. A high-fidelity Cas9 mutant delivered as a ribonucleoprotein complex enables efficient gene editing in human hematopoietic stem and progenitor cells. Nat. Med. 2018, 24, 1216–1224. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.H.; Miller, S.M.; Geurts, M.H.; Tang, W.; Chen, L.; Sun, N.; Zeina, C.M.; Gao, X.; Rees, H.A.; Lin, Z.; et al. Evolved Cas9 variants with broad PAM compatibility and high DNA specificity. Nature 2018, 556, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Piatek, A.; Ali, Z.; Baazim, H.; Li, L.; Abulfaraj, A.; Al-Shareef, S.; Aouida, M.; Mahfouz, M.M. RNA-guided transcriptional regulation in planta via synthetic dCas9-based transcription factors. Plant Biotechnol. J. 2015, 13, 578–589. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, D.; Xiong, X.; Yan, B.; Xie, W.; Sheen, J.; Li, J.-F. A potent Cas9-derived gene activator for plant and mammalian cells. Nat. Plants 2017, 3, 930–936. [Google Scholar] [CrossRef] [Green Version]
- Mikami, M.; Toki, S.; Endo, M. Precision targeted mutagenesis via Cas9 paired nickases in rice. Plant Cell Physiol. 2016, 57, 1058–1068. [Google Scholar] [CrossRef] [Green Version]
- Hsu, C.-T.; Cheng, Y.-J.; Yuan, Y.-H.; Hung, W.-F.; Cheng, Q.-W.; Wu, F.-H.; Lee, L.-Y.; Gelvin, S.B.; Lin, C.-S. Application of Cas12a and nCas9-activation-induced cytidine deaminase for genome editing and as a non-sexual strategy to generate homozygous/multiplex edited plants in the allotetraploid genome of tobacco. Plant Mol. Biol. 2019, 101, 355–371. [Google Scholar] [CrossRef] [PubMed]
- Endo, M.; Mikami, M.; Endo, A.; Kaya, H.; Itoh, T.; Nishimasu, H.; Nureki, O.; Toki, S. Genome editing in plants by engineered CRISPR–Cas9 recognizing NG PAM. Nat. Plants 2019, 5, 14–17. [Google Scholar] [CrossRef]
- Kleinstiver, B.P.; Prew, M.S.; Tsai, S.Q.; Nguyen, N.T.; Topkar, V.V.; Zheng, Z.; Joung, J.K. Broadening the targeting range of Staphylococcus aureus CRISPR-Cas9 by modifying PAM recognition. Nat. Biotechnol. 2015, 33, 1293–1298. [Google Scholar] [CrossRef] [PubMed]
- Hirano, H.; Gootenberg, J.S.; Horii, T.; Abudayyeh, O.O.; Kimura, M.; Hsu, P.D.; Nakane, T.; Ishitani, R.; Hatada, I.; Zhang, F.; et al. Structure and engineering of Francisella novicida Cas9. Cell 2016, 164, 950–961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Endo, A.; Toki, S. FnCpf1-Mediated targeted mutagenesis in plants. Meth. Mol. Biol. 2018, 1795, 223–239. [Google Scholar]
- Begemann, M.B.; Gray, B.N.; January, E.; Gordon, G.C.; He, Y.; Liu, H.; Wu, X.; Brutnell, T.P.; Mockler, T.C.; Oufattole, M. Precise insertion and guided editing of higher plant genomes using Cpf1 CRISPR nucleases. Sci. Rep. 2017, 7, 11606. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zetsche, B.; Gootenberg Jonathan, S.; Abudayyeh Omar, O.; Slaymaker Ian, M.; Makarova Kira, S.; Essletzbichler, P.; Volz Sara, E.; Joung, J.; van der Oost, J.; Regev, A.; et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 2015, 163, 759–771. [Google Scholar] [CrossRef] [Green Version]
- Gao, L.; Cox, D.B.T.; Yan, W.X.; Manteiga, J.C.; Schneider, M.W.; Yamano, T.; Nishimasu, H.; Nureki, O.; Crosetto, N.; Zhang, F. Engineered Cpf1 variants with altered PAM specificities. Nat. Biotechnol. 2017, 35, 789–792. [Google Scholar] [CrossRef] [Green Version]
- Tóth, E.; Czene, B.C.; Kulcsár, P.I.; Krausz, S.L.; Tálas, A.; Nyeste, A.; Varga, É.; Huszár, K.; Weinhardt, N.; Ligeti, Z.; et al. Mb- and FnCpf1 nucleases are active in mammalian cells: Activities and PAM preferences of four wild-type Cpf1 nucleases and of their altered PAM specificity variants. Nucleic Acids Res. 2018, 46, 10272–10285. [Google Scholar] [CrossRef]
- Shmakov, S.; Abudayyeh, O.O.; Makarova, K.S.; Wolf, Y.I.; Gootenberg, J.S.; Semenova, E.; Minakhin, L.; Joung, J.; Konermann, S.; Severinov, K.; et al. Discovery and functional characterization of diverse class 2 CRISPR-Cas systems. Mol. Cell 2015, 60, 385–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, H.; Gao, P.; Rajashankar, K.R.; Patel, D.J. PAM-Dependent Target DNA Recognition and cleavage by C2c1 CRISPR-Cas endonuclease. Cell 2016, 167, 1814–1828.e12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, H.; Patel, D.J. CasX: A new and small CRISPR gene-editing protein. Cell Res. 2019, 29, 345–346. [Google Scholar] [CrossRef]
- Liu, J.-J.; Orlova, N.; Oakes, B.L.; Ma, E.; Spinner, H.B.; Baney, K.L.M.; Chuck, J.; Tan, D.; Knott, G.J.; Harrington, L.B.; et al. CasX enzymes comprise a distinct family of RNA-guided genome editors. Nature 2019, 566, 218–223. [Google Scholar] [CrossRef]
- Tripathi, J.N.; Ntui, V.O.; Ron, M.; Muiruri, S.K.; Britt, A.; Tripathi, L. CRISPR/Cas9 editing of endogenous banana streak virus in the B genome of Musa spp. overcomes a major challenge in banana breeding. Commun. Biol. 2019, 2, 46. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.; Zhu, H.; Liu, J.; Yang, Q.; Shao, X.; Bi, F.; Hu, C.; Huo, H.; Chen, K.; Yi, G. Establishment of a PEG-mediated protoplast transformation system based on DNA and CRISPR/Cas9 ribonucleoprotein complexes for banana. BMC Plant Biol. 2020, 20, 425. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Sheng, O.; Deng, G.; He, W.; Dong, T.; Yang, Q.; Dou, T.; Li, C.; Gao, H.; Liu, S.; et al. CRISPR/Cas9-mediated genome editing of MaACO1 (aminocyclopropane-1-carboxylate oxidase 1) promotes the shelf life of banana fruit. Plant Biotechnol. J. 2020. [Google Scholar] [CrossRef]
- Kaur, N.; Alok, A.; Shivani, M.; Kumar, P.; Awasthi, P.; Chaturvedi, S.; Pandey, P.; Pandey, A.; Pandey, A.K.; Tiwari, S. CRISPR/Cas9 directed editing of lycopene epsilon-cyclase modulates metabolic flux for β-carotene biosynthesis in banana fruit. Metab. Eng. 2020, 59, 76–86. [Google Scholar] [CrossRef]
- Shao, X.; Wu, S.; Dou, T.; Zhu, H.; Hu, C.; Huo, H.; He, W.; Deng, G.; Sheng, O.; Bi, F.; et al. Using CRISPR/Cas9 genome editing system to create MaGA20ox2 gene-modified semi-dwarf banana. Plant Biotechnol. J. 2020, 18, 17–19. [Google Scholar] [CrossRef] [Green Version]
- Jia, H.; Wang, N. Xcc-facilitated agroinfiltration of citrus leaves: A tool for rapid functional analysis of transgenes in citrus leaves. Plant Cell Rep. 2014, 33, 1993–2001. [Google Scholar] [CrossRef]
- Jia, H.; Xu, J.; Orbović, V.; Zhang, Y.; Wang, N. Editing citrus genome via SaCas9/sgRNA system. Front. Plant Sci. 2017, 8, 2135. [Google Scholar] [CrossRef]
- Jia, H.; Orbović, V.; Wang, N. CRISPR-LbCas12a-mediated modification of citrus. Plant Biotechnol. J. 2019, 17, 1928–1937. [Google Scholar] [CrossRef] [Green Version]
- Jia, H.; Zhang, Y.; Orbović, V.; Xu, J.; White, F.F.; Jones, J.B.; Wang, N. Genome editing of the disease susceptibility gene CsLOB1 in citrus confers resistance to citrus canker. Plant Biotechnol. J. 2017, 15, 817–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gumtow, R.; Wu, D.; Uchida, J.; Tian, M. A Phytophthora palmivora extracellular cystatin-like protease inhibitor targets papain to contribute to virulence on papaya. Mol. Plant Microbe Interact. 2018, 31, 363–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pettongkhao, S.; Navet, N.; Schornack, S.; Tian, M.; Churngchow, N. A secreted protein of 15 kDa plays an important role in Phytophthora palmivora development and pathogenicity. Sci. Rep. 2020, 10, 2319. [Google Scholar] [CrossRef] [PubMed]
- Pang, H.; Yan, Q.; Zhao, S.; He, F.; Xu, J.; Qi, B.; Zhang, Y. Knockout of the S-acyltransferase gene, PbPAT14, confers the dwarf yellowing phenotype in first generation pear by ABA accumulation. Int. J. Mol. Sci. 2019, 20, 6347. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Cheng, X.; Shan, Q.; Zhang, Y.; Liu, J.; Gao, C.; Qiu, J.L. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat. Biotechnol. 2014, 32, 947–951. [Google Scholar] [CrossRef]
- Doench, J.G.; Hartenian, E.; Graham, D.B.; Tothova, Z.; Hegde, M.; Smith, I.; Sullender, M.; Ebert, B.L.; Xavier, R.J.; Root, D.E. Rational design of highly active sgRNAs for CRISPR-Cas9–mediated gene inactivation. Nat. Biotechnol. 2014, 32, 1262–1267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, N.; Liu, W.; Wang, X. WU-CRISPR: Characteristics of functional guide RNAs for the CRISPR/Cas9 system. Genome Biol. 2015, 16, 218. [Google Scholar] [CrossRef] [Green Version]
- Sugano, S.S.; Nishihama, R.; Shirakawa, M.; Takagi, J.; Matsuda, Y.; Ishida, S.; Shimada, T.; Hara-Nishimura, I.; Osakabe, K.; Kohchi, T. Efficient CRISPR/Cas9-based genome editing and its application to conditional genetic analysis in Marchantia polymorpha. PLoS ONE 2018, 13, e0205117. [Google Scholar] [CrossRef] [Green Version]
- Fu, Y.; Sander, J.D.; Reyon, D.; Cascio, V.M.; Joung, J.K. Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat. Biotechnol. 2014, 32, 279–284. [Google Scholar] [CrossRef] [Green Version]
- Rose, J.C.; Popp, N.A.; Richardson, C.D.; Stephany, J.J.; Mathieu, J.; Wei, C.T.; Corn, J.E.; Maly, D.J.; Fowler, D.M. Suppression of unwanted CRISPR-Cas9 editing by co-administration of catalytically inactivating truncated guide RNAs. Nat. Commun. 2020, 11, 2697. [Google Scholar] [CrossRef]
- Zhang, F.; LeBlanc, C.; Irish, V.F.; Jacob, Y. Rapid and efficient CRISPR/Cas9 gene editing in Citrus using the YAO promoter. Plant Cell Rep. 2017, 36, 1883–1887. [Google Scholar] [CrossRef]
- Lee, C.M.; Cradick, T.J.; Fine, E.J.; Bao, G. Nuclease target site selection for maximizing on-target activity and minimizing off-target effects in genome editing. Mol. Ther. 2016, 24, 475–487. [Google Scholar] [CrossRef] [Green Version]
- Ryan, D.E.; Taussig, D.; Steinfeld, I.; Phadnis, S.M.; Lunstad, B.D.; Singh, M.; Vuong, X.; Okochi, K.D.; McCaffrey, R.; Olesiak, M.; et al. Improving CRISPR–Cas specificity with chemical modifications in single-guide RNAs. Nucleic Acids Res. 2017, 46, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Kocak, D.D.; Josephs, E.A.; Bhandarkar, V.; Adkar, S.S.; Kwon, J.B.; Gersbach, C.A. Increasing the specificity of CRISPR systems with engineered RNA secondary structures. Nat. Biotechnol. 2019, 37, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Shan, Q.; Wang, Y.; Li, J.; Zhang, Y.; Chen, K.; Liang, Z.; Zhang, K.; Liu, J.; Xi, J.J.; Qiu, J.L.; et al. Targeted genome modification of crop plants using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 686–688. [Google Scholar] [CrossRef]
- Xu, R.; Li, H.; Qin, R.; Wang, L.; Li, L.; Wei, P.; Yang, J. Gene targeting using the Agrobacterium tumefaciens-mediated CRISPR-Cas system in rice. Rice 2014, 7, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miao, J.; Guo, D.; Zhang, J.; Huang, Q.; Qin, G.; Zhang, X.; Wan, J.; Gu, H.; Qu, L.J. Targeted mutagenesis in rice using CRISPR-Cas system. Cell Res. 2013, 23, 1233–1236. [Google Scholar] [CrossRef] [Green Version]
- Liang, Z.; Zhang, K.; Chen, K.; Gao, C. Targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas system. J. Genet. Gen. 2014, 41, 63–68. [Google Scholar] [CrossRef]
- Hyun, Y.; Kim, J.; Cho, S.W.; Choi, Y.; Kim, J.-S.; Coupland, G. Site-directed mutagenesis in Arabidopsis thaliana using dividing tissue-targeted RGEN of the CRISPR/Cas system to generate heritable null alleles. Planta 2015, 241, 271–284. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.-P.; Xing, H.-L.; Dong, L.; Zhang, H.-Y.; Han, C.-Y.; Wang, X.-C.; Chen, Q.-J. Egg cell-specific promoter-controlled CRISPR/Cas9 efficiently generates homozygous mutants for multiple target genes in Arabidopsis in a single generation. Genome Biol. 2015, 16, 144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.-F.; Norville, J.E.; Aach, J.; McCormack, M.; Zhang, D.; Bush, J.; Church, G.M.; Sheen, J. Multiplex and homologous recombination–mediated genome editing in Arabidopsis and Nicotiana benthamiana using guide RNA and Cas9. Nat. Biotechnol. 2013, 31, 688–691. [Google Scholar] [CrossRef]
- Sun, X.; Hu, Z.; Chen, R.; Jiang, Q.; Song, G.; Zhang, H.; Xi, Y. Targeted mutagenesis in soybean using the CRISPR-Cas9 system. Sci. Rep. 2015, 5, 10342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.; Zhang, Y.; Kleinstiver, B.P.; Guo, J.A.; Aryee, M.J.; Miller, J.; Malzahn, A.; Zarecor, S.; Lawrence-Dill, C.J.; Joung, J.K.; et al. Activities and specificities of CRISPR/Cas9 and Cas12a nucleases for targeted mutagenesis in maize. Plant Biotechnol. J. 2019, 17, 362–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolter, F.; Klemm, J.; Puchta, H. Efficient in planta gene targeting in Arabidopsis using egg cell-specific expression of the Cas9 nuclease of Staphylococcus aureus. Plant J. 2018, 94, 735–746. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, A.; Ishida, T.; Yoshimura, M.; Kimura, Y.; Sawa, S. Developing heritable mutations in Arabidopsis thaliana using a modified CRISPR/Cas9 toolkit comprising PAM-altered Cas9 variants and gRNAs. Plant Cell Physiol. 2019, 60, 2255–2262. [Google Scholar] [CrossRef] [PubMed]
- Brocken, D.J.; Tark-Dame, M.; Dame, R.T. dCas9: A versatile tool for epigenome editing. Curr. Issues Mol. Biol. 2017, 26, 15–32. [Google Scholar]
- Bikard, D.; Jiang, W.; Samai, P.; Hochschild, A.; Zhang, F.; Marraffini, L.A. Programmable repression and activation of bacterial gene expression using an engineered CRISPR-Cas system. Nucleic Acids Res. 2013, 41, 7429–7437. [Google Scholar] [CrossRef] [Green Version]
- Lowder, L.G.; Zhang, D.; Baltes, N.J.; Paul, J.W.; Tang, X.; Zheng, X.; Voytas, D.F.; Hsieh, T.-F.; Zhang, Y.; Qi, Y. A CRISPR/Cas9 toolbox for multiplexed plant genome editing and transcriptional regulation. Plant Physiol. 2015, 169, 971–985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fister, A.S.; Landherr, L.; Maximova, S.N.; Guiltinan, M.J. Transient expression of CRISPR/Cas9 machinery targeting TcNPR3 enhances defense response in Theobroma cacao. Front. Plant Sci. 2018, 9, 268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandhya, D.; Jogam, P.; Allini, V.R.; Abbagani, S.; Alok, A. The present and potential future methods for delivering CRISPR/Cas9 components in plants. J. Genet. Eng. Biotechnol. 2020, 18, 25. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.-l.; Liang, H.-z.; Wang, S.-f.; Lian, Y.; Wei, Y.-l.; Wang, T.-f. Research progress and commercialization on transgenic soybean in china. Soybean Sci. 2010, 29, 143–150. [Google Scholar]
- Li, S.; Cong, Y.; Liu, Y.; Wang, T.; Shuai, Q.; Chen, N.; Gai, J.; Li, Y. Optimization of Agrobacterium-mediated transformation in soybean. Front. Plant Sci. 2017, 8, 246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castel, B.; Tomlinson, L.; Locci, F.; Yang, Y.; Jones, J.D.G. Optimization of T-DNA architecture for Cas9-mediated mutagenesis in Arabidopsis. PLoS ONE 2019, 14, e0204778. [Google Scholar] [CrossRef] [Green Version]
- Zale, J.M.; Agarwal, S.; Loar, S.; Steber, C.M. Evidence for stable transformation of wheat by floral dip in Agrobacterium tumefaciens. Plant Cell Rep. 2009, 28, 903–913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bastaki, N.K.; Cullis, C.A. Floral-dip transformation of flax (Linum usitatissimum) to generate transgenic progenies with a high transformation rate. J. Visual. Exp. 2014, 19, 52189. [Google Scholar] [CrossRef] [Green Version]
- Curtis, I.S.; Nam, H.G. Transgenic radish (Raphanus sativus L. longipinnatus Bailey) by floral-dip method-plant development and surfactant are important in optimizing transformation efficiency. Transgenic Res. 2001, 10, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Sharada, M.S.; Kumari, A.; Pandey, A.K.; Sharma, S.; Sharma, P.; Sreelakshmi, Y.; Sharma, R. Generation of genetically stable transformants by Agrobacterium using tomato floral buds. Plant Cell Tissue Org. Cult. 2017, 129, 299–312. [Google Scholar] [CrossRef]
- Murovec, J.; Guček, K.; Bohanec, B.; Avbelj, M.; Jerala, R. DNA-free genome editing of Brassica oleracea and B. rapa protoplasts using CRISPR-Cas9 ribonucleoprotein complexes. Front. Plant Sci. 2018, 9, 1594. [Google Scholar] [CrossRef]
- Svitashev, S.; Young, J.K.; Schwartz, C.; Gao, H.; Falco, S.C.; Cigan, A.M. Targeted mutagenesis, precise gene editing, and site-specific gene insertion in maize using Cas9 and guide RNA. Plant Physiol. 2015, 169, 931–945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andersson, M.; Turesson, H.; Olsson, N.; Fält, A.-S.; Ohlsson, P.; Gonzalez, M.N.; Samuelsson, M.; Hofvander, P. Genome editing in potato via CRISPR-Cas9 ribonucleoprotein delivery. Physiol. Plant. 2018, 164, 378–384. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Liu, Z.-B.; Xing, A.; Moon, B.P.; Koellhoffer, J.P.; Huang, L.; Ward, R.T.; Clifton, E.; Falco, S.C.; Cigan, A. Cas9-guide RNA directed genome editing in soybean. Plant Physiol. 2015, 169, 960–970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.; Choi, J.; Won, K.-H. A stable DNA-free screening system for CRISPR/RNPs-mediated gene editing in hot and sweet cultivars of Capsicum annuum. BMC Plant Biol. 2020, 20, 449. [Google Scholar] [CrossRef]
- Foster, A.J.; Martin-Urdiroz, M.; Yan, X.; Wright, H.S.; Soanes, D.M.; Talbot, N.J. CRISPR-Cas9 ribonucleoprotein-mediated co-editing and counterselection in the rice blast fungus. Sci. Rep. 2018, 8, 14355. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Choi, S.; Park, S.; Yoon, J.; Park, A.Y.; Choe, S. DNA-free genome editing via ribonucleoprotein (RNP) delivery of CRISPR/Cas in lettuce. In Plant Genome Editing with CRISPR Systems: Methods and Protocols; Qi, Y., Ed.; Springer: New York, NY, USA, 2019; pp. 337–354. [Google Scholar]
- Waltz, E. Gene-edited CRISPR mushroom escapes US regulation. Nature 2016, 532, 293. [Google Scholar] [CrossRef] [Green Version]
- Baltes, N.J.; Gil-Humanes, J.; Cermak, T.; Atkins, P.A.; Voytas, D.F. DNA replicons for plant genome engineering. Plant Cell 2014, 26, 151–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cermak, T.; Baltes, N.; Cegan, R.; Zhang, Y.; Voytas, D. High-frequency, precise modification of the tomato genome. Genome Biol. 2015, 16, 232–246. [Google Scholar] [CrossRef] [Green Version]
- Gil-Humanes, J.; Wang, Y.; Liang, Z.; Shan, Q.; Ozuna, C.V.; Sánchez-León, S.; Baltes, N.J.; Starker, C.; Barro, F.; Gao, C.; et al. High-efficiency gene targeting in hexaploid wheat using DNA replicons and CRISPR/Cas9. Plant J. 2017, 89, 1251–1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, K.; Han, T.; Liu, G.; Chen, T.; Wang, Y.; Yu, A.Y.L.; Liu, Y. A geminivirus-based guide RNA delivery system for CRISPR/Cas9 mediated plant genome editing. Sci. Rep. 2015, 5, 14926. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.; Abul-faraj, A.; Piatek, M.; Mahfouz, M.M. Activity and specificity of TRV-mediated gene editing in plants. Plant Signal. Behav. 2015, 10, e1044191. [Google Scholar] [CrossRef]
- Ramakrishna, S.; Kwaku Dad, A.-B.; Beloor, J.; Gopalappa, R.; Lee, S.-K.; Kim, H. Gene disruption by cell-penetrating peptide-mediated delivery of Cas9 protein and guide RNA. Genome Res. 2014, 24, 1020–1027. [Google Scholar] [CrossRef] [Green Version]
- Sun, W.; Ji, W.; Hall, J.M.; Hu, Q.; Wang, C.; Beisel, C.L.; Gu, Z. Self-assembled DNA nanoclews for the efficient delivery of CRISPR–Cas9 for genome editing. Angew. Chem. Int. Ed. 2015, 54, 12029–12033. [Google Scholar] [CrossRef] [PubMed]
- Mout, R.; Ray, M.; Yesilbag Tonga, G.; Lee, Y.-W.; Tay, T.; Sasaki, K.; Rotello, V.M. Direct cytosolic delivery of CRISPR/Cas9-ribonucleoprotein for efficient gene editing. ACS Nano 2017, 11, 2452–2458. [Google Scholar] [CrossRef] [Green Version]
- Gori, J.L.; Hsu, P.D.; Maeder, M.L.; Shen, S.; Welstead, G.G.; Bumcrot, D. Delivery and specificity of CRISPR/Cas9 genome editing technologies for human gene therapy. Hum. Gene Ther. 2015, 26, 443–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molla, K.A.; Yang, Y. CRISPR/Cas-mediated base editing: Technical considerations and practical applications. Trends Biotechnol. 2019, 37, 1121–1142. [Google Scholar] [CrossRef]
- Rees, H.A.; Komor, A.C.; Yeh, W.-H.; Caetano-Lopes, J.; Warman, M.; Edge, A.S.B.; Liu, D.R. Improving the DNA specificity and applicability of base editing through protein engineering and protein delivery. Nat. Commun. 2017, 8, 15790. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Zong, Y.; Wang, Y.; Jin, S.; Zhang, D.; Song, Q.; Zhang, R.; Gao, C. Expanded base editing in rice and wheat using a Cas9-adenosine deaminase fusion. Genome Biol. 2018, 19, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, B.-C.; Yun, J.-Y.; Kim, S.-T.; Shin, Y.; Ryu, J.; Choi, M.; Woo, J.W.; Kim, J.-S. Precision genome engineering through adenine base editing in plants. Nat. Plants 2018, 4, 427–431. [Google Scholar] [CrossRef]
- Hua, K.; Tao, X.; Yuan, F.; Wang, D.; Zhu, J.-K. Precise A·T to G·C base editing in the rice genome. Mol. Plant 2018, 11, 627–630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hua, K.; Tao, X.; Zhu, J.-K. Expanding the base editing scope in rice by using Cas9 variants. Plant Biotechnol. J. 2019, 17, 499–504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, L.; Li, J.; Wang, Q.; Xu, Z.; Sun, L.; Alariqi, M.; Manghwar, H.; Wang, G.; Li, B.; Ding, X.; et al. High-efficient and precise base editing of C•G to T•A in the allotetraploid cotton (Gossypium hirsutum) genome using a modified CRISPR/Cas9 system. Plant Biotechnol. J. 2020, 18, 45–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, F.; Kuang, Y.; Ren, B.; Wang, J.; Zhang, D.; Lin, H.; Yang, B.; Zhou, X.; Zhou, H. Highly efficient A.T to G.C base editing by Cas9n-guided tRNA adenosine deaminase in rice. Mol. Plant 2018, 11, 631–634. [Google Scholar] [CrossRef] [Green Version]
- Anzalone, A.V.; Randolph, P.B.; Davis, J.R.; Sousa, A.A.; Koblan, L.W.; Levy, J.M.; Chen, P.J.; Wilson, C.; Newby, G.A.; Raguram, A.; et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 2019, 576, 149–157. [Google Scholar] [CrossRef]
- Yan, J.; Cirincione, A.; Adamson, B. Prime editing: Precision genome editing by reverse transcription. Mol. Cell 2020, 77, 210–212. [Google Scholar] [CrossRef]
- Xue, C.; Zhang, H.; Lin, Q.; Fan, R.; Gao, C. Manipulating mRNA splicing by base editing in plants. Sci. China Life Sci. 2018, 61, 1293–1300. [Google Scholar] [CrossRef]
- Li, Z.; Xiong, X.; Wang, F.; Liang, J.; Li, J.-F. Gene disruption through base editing-induced messenger RNA missplicing in plants. New Phytol. 2019, 222, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Butt, H.; Rao, G.S.; Sedeek, K.; Aman, R.; Kamel, R.; Mahfouz, M. Engineering herbicide resistance via prime editing in rice. Plant Biotechnol. J. 2020, 18, 2370–2372. [Google Scholar] [CrossRef]
- Tang, X.; Sretenovic, S.; Ren, Q.; Jia, X.; Li, M.; Fan, T.; Yin, D.; Xiang, S.; Guo, Y.; Liu, L.; et al. Plant prime editors enable precise gene editing in rice cells. Mol. Plant 2020, 13, 667–670. [Google Scholar] [CrossRef]
- Xu, R.; Li, J.; Liu, X.; Shan, T.; Qin, R.; Wei, P. Development of plant prime-editing systems for precise genome editing. Plant Commun. 2020, 1, 100043. [Google Scholar] [CrossRef]
- Li, H.; Li, J.; Chen, J.; Yan, L.; Xia, L. Precise modifications of both exogenous and endogenous genes in rice by prime editing. Mol. Plant 2020, 13, 671–674. [Google Scholar] [CrossRef] [PubMed]
- Vouillot, L.; Thélie, A.; Pollet, N. Comparison of T7E1 and surveyor mismatch cleavage assays to detect mutations triggered by engineered nucleases. G3 Genes Genomes Genet. 2015, 5, 407–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, K.; Yang, Y. RNA-guided genome editing in plants using A CRISPR-Cas system. Mol. Plant 2013, 6, 1975–1983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sentmanat, M.F.; Peters, S.T.; Florian, C.P.; Connelly, J.P.; Pruett-Miller, S.M. A survey of validation strategies for CRISPR-Cas9 editing. Sci. Rep. 2018, 8, 888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richter, J.; Watson, J.M.; Stasnik, P.; Borowska, M.; Neuhold, J.; Berger, M.; Stolt-Bergner, P.; Schoft, V.; Hauser, M.-T. Multiplex mutagenesis of four clustered CrRLK1L with CRISPR/Cas9 exposes their growth regulatory roles in response to metal ions. Sci. Rep. 2018, 8, 12182. [Google Scholar] [CrossRef]
- Pan, C.; Ye, L.; Qin, L.; Liu, X.; He, Y.; Wang, J.; Chen, L.; Lu, G. CRISPR/Cas9-mediated efficient and heritable targeted mutagenesis in tomato plants in the first and later generations. Sci. Rep. 2016, 6, 24765. [Google Scholar] [CrossRef]
- Denbow, C.J.; Lapins, S.; Dietz, N.; Scherer, R.; Nimchuk, Z.L.; Okumoto, S. Gateway-compatible CRISPR-Cas9 vectors and a rapid detection by high-resolution melting curve analysis. Front. Plant Sci. 2017, 8, 1171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samarut, É.; Lissouba, A.; Drapeau, P. A simplified method for identifying early CRISPR-induced indels in zebrafish embryos using High Resolution Melting analysis. BMC Genom. 2016, 17, 547. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.; Li, K.; Jin, L.; Xu, R.; Miao, K.; Yang, F.; Qi, C.; Zhang, L.; Botella, J.R.; Wang, R.; et al. A simple and cost-effective method for screening of CRISPR/Cas9-induced homozygous/biallelic mutants. Plant Meth. 2018, 14, 40. [Google Scholar] [CrossRef]
- Chen, L.; Li, W.; Katin-Grazzini, L.; Ding, J.; Gu, X.; Li, Y.; Gu, T.; Wang, R.; Lin, X.; Deng, Z.; et al. A method for the production and expedient screening of CRISPR/Cas9-mediated non-transgenic mutant plants. Hort. Res. 2018, 5, 13. [Google Scholar] [CrossRef]
- Li, S.; Liu, S.; Liu, Y.; Lu, H.; Tan, Y.; Huang, J.; Wei, P.; Shu, Q.Y. HRM-facilitated rapid identification and genotyping of mutations induced by CRISPR/Cas9 mutagenesis in rice. Crop Breed. Appl. Biotechnol. 2018, 18, 184–191. [Google Scholar] [CrossRef] [Green Version]
- Rocafort, M.; Arshed, S.; Hudson, D.; Singh, J.; Bowen, J.K.; Plummer, K.M.; Bradshaw, R.E.; Johnson, R.D.; Johnson, L.J.; Mesarich, C.H. CRISPR-Cas9 gene editing and rapid detection of gene-edited mutants using high-resolution melting in the apple scab fungus, Venturia inaequalis. bioRxiv 2021. [Google Scholar] [CrossRef]
- Bewg, W.P.; Ci, D.; Tsai, C.-J. Genome editing in trees: From multiple repair pathways to long-term stability. Front. Plant Sci. 2018, 9, 1732. [Google Scholar] [CrossRef]
- Ren, Q.; Zhong, Z.; Wang, Y.; You, Q.; Li, Q.; Yuan, M.; He, Y.; Qi, C.; Tang, X.; Zheng, X.; et al. Bidirectional promoter-based CRISPR-Cas9 systems for plant genome editing. Front. Plant Sci. 2019, 10, 1173. [Google Scholar] [CrossRef] [PubMed]
- Bruegmann, T.; Deecke, K.; Fladung, M. Evaluating the efficiency of gRNAs in CRISPR/Cas9 mediated genome editing in poplars. Int. J. Mol. Sci. 2019, 20, 3623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalla Costa, L.; Piazza, S.; Pompili, V.; Salvagnin, U.; Cestaro, A.; Moffa, L.; Vittani, L.; Moser, C.; Malnoy, M. Strategies to produce T-DNA free CRISPRed fruit trees via Agrobacterium tumefaciens stable gene transfer. Sci. Rep. 2020, 10, 20155. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Chen, Y.; Ma, Y.; Wang, H.; Wei, J. Effective reduction in chimeric mutants of poplar trees produced by CRISPR/Cas9 through a second round of shoot regeneration. Plant Biotechnol. Rep. 2020, 14, 549–558. [Google Scholar] [CrossRef]
- Hendel, A.; Fine, E.J.; Bao, G.; Porteus, M.H. Quantifying on- and off-target genome editing. Trends Biotechnol. 2015, 33, 132–140. [Google Scholar] [CrossRef] [Green Version]
- Macovei, A.; Sevilla, N.R.; Cantos, C.; Jonson, G.B.; Slamet-Loedin, I.; Čermák, T.; Voytas, D.F.; Choi, I.-R.; Chadha-Mohanty, P. Novel alleles of rice eIF4G generated by CRISPR/Cas9-targeted mutagenesis confer resistance to Rice tungro spherical virus. Plant Biotechnol. J. 2018, 16, 1918–1927. [Google Scholar] [CrossRef] [Green Version]
- de Pater, S.; Klemann, B.; Hooykaas, P.J.J. True gene-targeting events by CRISPR/Cas-induced DSB repair of the PPO locus with an ectopically integrated repair template. Sci. Rep. 2018, 8, 3338. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Wu, J.-J.; Tang, T.; Liu, K.-D.; Dai, C. CRISPR/Cas9-mediated genome editing efficiently creates specific mutations at multiple loci using one sgRNA in Brassica napus. Sci. Rep. 2017, 7, 7489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minkenberg, B.; Xie, K.; Yang, Y. Discovery of rice essential genes by characterizing a CRISPR-edited mutation of closely related rice MAP kinase genes. Plant J. 2017, 89, 636–648. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Zhang, J.; Wei, P.; Zhang, B.; Gou, F.; Feng, Z.; Mao, Y.; Yang, L.; Zhang, H.; Xu, N.; et al. The CRISPR/Cas9 system produces specific and homozygous targeted gene editing in rice in one generation. Plant Biotechnol. J. 2014, 12, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Fernandez i Marti, A.; Dodd, R.S. Using CRISPR as a gene editing tool for validating adaptive gene function in tree landscape genomics. Front. Ecol. Evol. 2018, 6. [Google Scholar] [CrossRef] [Green Version]
- Lonowski, L.A.; Narimatsu, Y.; Riaz, A.; Delay, C.E.; Yang, Z.; Niola, F.; Duda, K.; Ober, E.A.; Clausen, H.; Wandall, H.H.; et al. Genome editing using FACS enrichment of nuclease-expressing cells and indel detection by amplicon analysis. Nat. Protoc. 2017, 12, 581–603. [Google Scholar] [CrossRef]
- Tsai, C.J.; Xue, L.J. CRISPRing into the woods. GM Crops Food 2015, 6, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wu, H.; Chen, Y.; Yin, T. Efficient CRISPR/Cas9-mediated gene editing in an interspecific hybrid poplar with a highly heterozygous genome. Front. Plant Sci. 2020, 11, 996. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.S.; Sattar, M.N.; Iqbal, Z.; Raza, A.; Al-Sadi, A.M. Next-generation sequencing and the CRISPR-Cas nexus: A molecular plant virology perspective. Front. Microbiol. 2021, 11, 3456. [Google Scholar] [CrossRef] [PubMed]
- Montecillo, J.A.V.; Chu, L.L.; Bae, H. CRISPR-Cas9 system for plant genome editing: Current approaches and emerging developments. Agronomy 2020, 10, 1033. [Google Scholar] [CrossRef]
- Chang, S.; Mahon, E.L.; MacKay, H.A.; Rottmann, W.H.; Strauss, S.H.; Pijut, P.M.; Powell, W.A.; Coffey, V.; Lu, H.; Mansfield, S.D.; et al. Genetic engineering of trees: Progress and new horizons. In Vitro Cell. Develop. Biol. Plant 2018, 54, 341–376. [Google Scholar] [CrossRef]
- Vats, S.; Kumawat, S.; Kumar, V.; Patil, G.B.; Joshi, T.; Sonah, H.; Sharma, T.R.; Deshmukh, R. Genome editing in plants: Exploration of technological advancements and challenges. Cells 2019, 8, 1386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, Y.; Xin, S.; Dai, X.; Yang, X.; Huang, H.; Hua, Y. Efficient genome editing of rubber tree (Hevea brasiliensis) protoplasts using CRISPR/Cas9 ribonucleoproteins. Ind. Crops Prod. 2020, 146, 112146. [Google Scholar] [CrossRef]
- Tsai, C.J.; Xu, P.; Xue, L.J.; Hu, H.; Nyamdari, B.; Naran, R.; Zhou, X.; Goeminne, G.; Gao, R.; Gjersing, E.; et al. Compensatory guaiacyl lignin biosynthesis at the expense of syringyl lignin in 4CL1-knockout poplar. Plant Physiol. 2020, 183, 123–136. [Google Scholar] [CrossRef] [Green Version]
- Lowe, K.; Wu, E.; Wang, N.; Hoerster, G.; Hastings, C.; Cho, M.-J.; Scelonge, C.; Lenderts, B.; Chamberlin, M.; Cushatt, J.; et al. Morphogenic regulators Baby boom and Wuschel improve monocot transformation. Plant Cell 2016, 28, 1998–2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lotan, T.; Ohto, M.-A.; Yee, K.M.; West, M.A.L.; Lo, R.; Kwong, R.W.; Yamagishi, K.; Fischer, R.L.; Goldberg, R.B.; Harada, J.J. Arabidopsis LEAFY COTYLEDON1 is sufficient to induce embryo development in vegetative cells. Cell 1998, 93, 1195–1205. [Google Scholar] [CrossRef] [Green Version]
- Stone, S.L.; Braybrook, S.A.; Paula, S.L.; Kwong, L.W.; Meuser, J.; Pelletier, J.; Hsieh, T.-F.; Fischer, R.L.; Goldberg, R.B.; Harada, J.J. Arabidopsis LEAFY COTYLEDON2 induces maturation traits and auxin activity: Implications for somatic embryogenesis. Proc. Natl. Acad. Sci. USA 2008, 105, 3151–3156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srinivasan, C.; Liu, Z.; Heidmann, I.; Supena, E.D.J.; Fukuoka, H.; Joosen, R.; Lambalk, J.; Angenent, G.; Scorza, R.; Custers, J.B.M.; et al. Heterologous expression of the BABY BOOM AP2/ERF transcription factor enhances the regeneration capacity of tobacco (Nicotiana tabacum L.). Planta 2006, 225, 341. [Google Scholar] [CrossRef]
- Deng, W.; Luo, K.; Li, Z.; Yang, Y. A novel method for induction of plant regeneration via somatic embryogenesis. Plant Sci. 2009, 177, 43–48. [Google Scholar] [CrossRef]
- Bisi, R.B.; Pio, R.; da Hora Farias, D.; Locatelli, G.; de Alcântara Barbosa, C.M.; Pereira, W.A. Molecular characterization of the S-alleles and compatibility among hybrid pear tree cultivars for subtropical regions. Hort. Sci. 2019, 54, 2104–2110. [Google Scholar] [CrossRef] [Green Version]
- Peng, C.; Wang, H.; Xu, X.; Wang, X.; Chen, X.; Wei, W.; Lai, Y.; Liu, G.; Godwin, I.D.; Li, J.; et al. High-throughput detection and screening of plants modified by gene editing using quantitative real-time polymerase chain reaction. Plant J. 2018, 95, 557–567. [Google Scholar] [CrossRef]
- Abu-Qaoud, H.; Skirvin, R.M.; Chevreau, E. In vitro separation of chimeral pears into their component genotypes. Euphytica 1990, 48, 189–196. [Google Scholar] [CrossRef]
- Strauss, S.H.; Costanza, A.; Séguin, A. Genetically engineered trees: Paralysis from good intentions. Science 2015, 349, 794–795. [Google Scholar] [CrossRef] [PubMed]
- Sattar, M.N.; Iqbal, Z.; Tahir, M.N.; Shahid, M.S.; Khurshid, M.; Al-Khateeb, A.A.; Al-Khateeb, S.A. CRISPR/Cas9: A practical approach in date palm genome editing. Front. Plant Sci. 2017, 8, 1469. [Google Scholar] [CrossRef] [Green Version]
- Nadakuduti, S.S.; Buell, C.R.; Voytas, D.F.; Starker, C.G.; Douches, D.S. Genome editing for crop improvement—Applications in clonally propagated polyploids with a focus on potato (Solanum tuberosum L.). Front. Plant Sci. 2018, 9, 1607. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Liang, Z.; Zong, Y.; Wang, Y.; Liu, J.; Chen, K.; Qiu, J.L.; Gao, C. Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nat. Commun. 2016, 7, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Liang, Z.; Chen, K.; Li, T.; Zhang, Y.; Wang, Y.; Zhao, Q.; Liu, J.; Zhang, H.; Liu, C.; Ran, Y.; et al. Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat. Commun. 2017, 8, 14261. [Google Scholar] [CrossRef]
- Hu, N.; Xian, Z.; Li, N.; Liu, Y.; Huang, W.; Yan, F.; Su, D.; Chen, J.; Li, Z. Rapid and user-friendly open-source CRISPR/Cas9 system for single- or multi-site editing of tomato genome. Hort. Res. 2019, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Araki, M. A future scenario of the global regulatory landscape regarding genome-edited crops. GM Crops Food 2017, 8, 44–56. [Google Scholar] [CrossRef] [Green Version]
- Ellens, K.W.; Levac, D.; Pearson, C.; Savoie, A.; Strand, N.; Louter, J.; Tibelius, C. Canadian Regulatory Aspects of Gene Editing Technologies. Transgenic Res. 2019, 28, 165–168. [Google Scholar] [CrossRef]
- Mallapaty, S. Australian gene-editing rules adopt“middle ground”. Nature 2019. [Google Scholar] [CrossRef]
- Callaway, E. CRISPR plants now subject to tough GM laws in European Union. Nature 2018, 560, 16–17. [Google Scholar] [CrossRef] [PubMed]
- Wight, A.J. Strict EU ruling on gene-edited crops squeezes science. Nature 2018, 563, 15–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
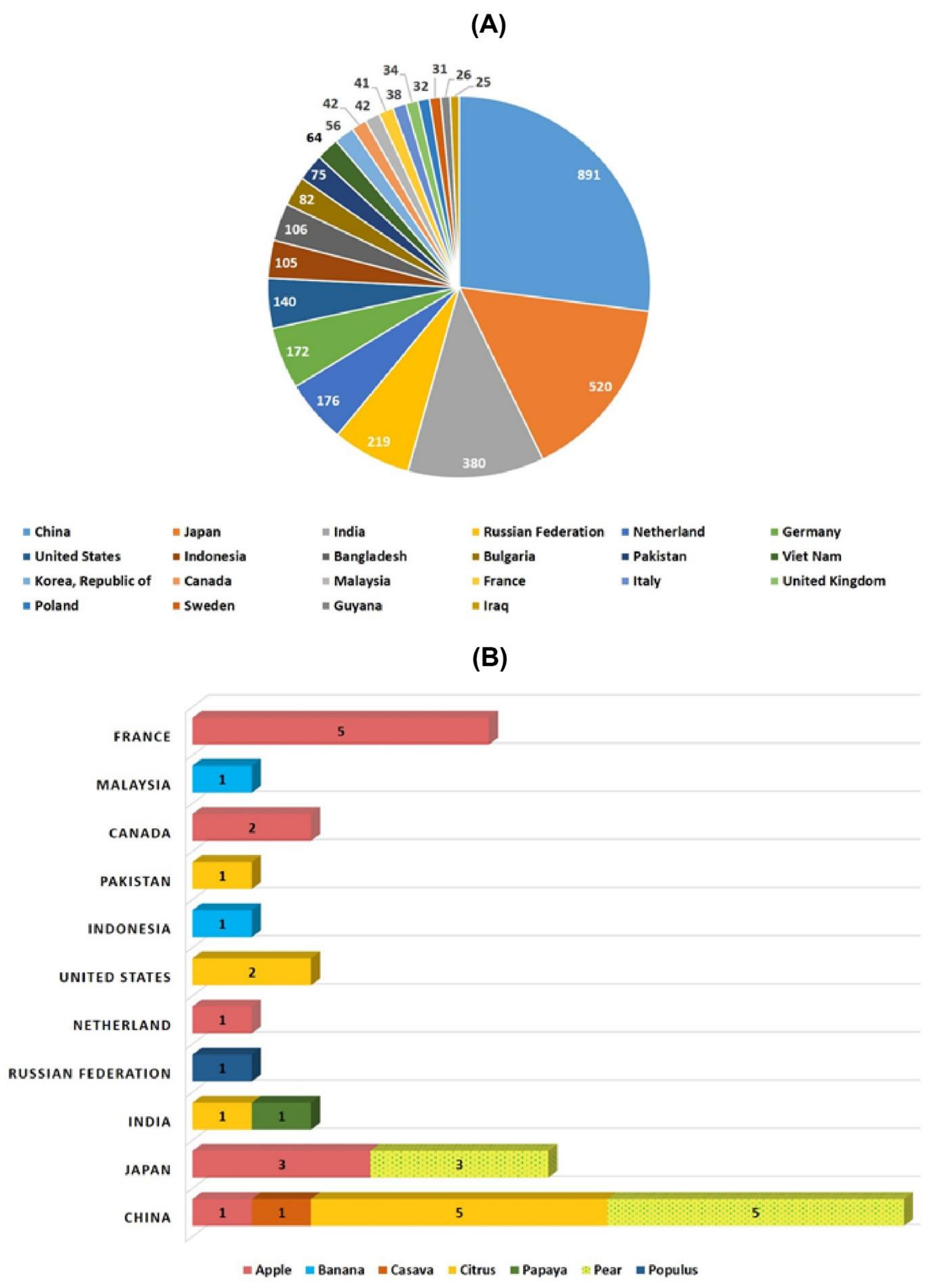

| Common Name | Botanical Name | Variety | Mutagen Source | Country | Year of Registration |
|---|---|---|---|---|---|
| Almond | Prunus dulcis Mill. | Supernova | Gamma rays (30 Gy) | Italy | 1987 |
| Apple | Malus pumila Mill. | Mori-hou-fu 3A | Gamma rays (30 Gy) | Japan | 1963 |
| Senbatsu-Fuji-2-Kei | Gamma rays (60 Gy) | 1985 | |||
| Belrene | EMS | France | 1970 | ||
| Blackjoin BA 2 520 | Gamma rays (50 Gy) | 1970 | |||
| Courtagold | 1972 | ||||
| Courtavel | 1972 | ||||
| Lysgolden | 1972 | ||||
| Donghenghongpingguo | Gamma rays (250 Gy) | China | 1987 | ||
| Dovar | X-rays (30–35 Gy) | Netherlands | 1978 | ||
| Golden Haidegg | Gamma rays (50 Gy) | Austria | 1986 | ||
| James Grieve Double Red | Gamma rays (62 Gy) | Czech Republic | 1995 | ||
| McIntosh 8F-2-32 | Gamma rays | Canada | 1970 | ||
| Shamrock | Gamma rays | Canada | 1986 | ||
| Apricot | Prunus armeniaca L. | Early Blenheim | thermal neutrons (thN) | Canada | 1970 |
| Banana | Musa paradisiaca L. | Klue Hom Thong KU1 | Gamma rays (25 Gy) | Thailand | 1985 |
| Novaria | Gamma rays (60 Gy) | Malaysia | 1995 | ||
| AL-BEELY | Gamma rays | Sudan | 2007 | ||
| Pirama 1 | Gamma rays (30 Gy) | Indonesia | 2019 | ||
| Fuxuan 01 | Gamma rays | China | 2005 | ||
| Clementina | Citrus celementina L. | Nero | Fast neutron (6 Gy) | Spain | 2006 |
| Neufina | 2010 | ||||
| CLEMENVERD | Fast neutron (5 Gy) | Spain | |||
| Ficus | Ficus benjamina L. | Golden King | X-rays (25 Gy) AND Gamma rays (20 Gy) | Belgium | 1980 |
| Golden Princess | |||||
| Fig | Ficus carica L. | Bol (Abundant) | Gamma rays (50–70 Gy) | Russian Federation | 1979 |
| Grapefruit | Citrus paradisi Macf. | Rio Red | Thermal neutrons (thN) | United States | 1970 |
| Star Ruby | 1984 | ||||
| Indian Jujube | Ziziphus mauritiana Lamk. | Dao tien | MNH (0.02–0.04%) | Viet Nam | 1986 |
| Ma hong | |||||
| Japanese pear | Pyrus pyriforia Nak. | Gold Nijisseiki | Gamma rays (0.12–0.15 Gy) | Japan | 1991 |
| Kotobuki Shinsui | Gamma rays (80 Gy) | 1997 | |||
| Osa Gold | 1997 | ||||
| Lemon | Citrus limon L. | Eureka 22 INTA | X-rays (10 Gy) | Argentina | 1987 |
| Loquat | Eriobotrya japonica L. | Shiro-mogi | Gamma rays (200 Gy) | Japan | 1982 |
| Mandarin | Citrus reticulata L. | Zhongyu 7 | Gamma rays (100 Gy) | China | 1985 |
| Zhongyu 8 | 1986 | ||||
| Hongju 420 | |||||
| NIAB Kinnow | Gamma rays (20 Gy) | Pakistan | 2017 | ||
| PAU Kinnow-1 | Gamma rays (30 Gy) | India | 2017 | ||
| Mulberry | Morus alba L. | Sangfu 1 | Gamma rays (75 Gy) | China | 1974 |
| Fuzaofeng | Gamma rays (5 Gy) | 1992 | |||
| Ji 7681 | N2 laser | 1988 | |||
| Fusang 10 | Gamma rays | 1980 | |||
| Shansang 871 | Gamma rays (60 Gy) | 1994 | |||
| Shigu 11-6 | Gamma rays (100 Gy) | 1995 | |||
| Lala Berry | Colchicine | Japan | 2003 | ||
| Pop Berry | Colchicine | 2004 | |||
| S54 | EMS | India | 1974 | ||
| Orange | Citrus sinensis L. | Hongju 418 | Gamma rays (100 Gy) | China | 1983 |
| Xuegan 9-12-1 | |||||
| Valencia 2 INTA | X-rays (20 Gy) | Argentina | 1987 | ||
| IAC 2014 | Gamma rays (40 Gy) | Brazil | 2016 | ||
| Papaya | Carica papaya L. | Pusa nanha | Gamma rays (150 Gy) | India | 1987 |
| Peach | Prunus persica L. | Magnif 135 | Gamma rays | Argentina | 1968 |
| Shaji 1 | CO2 laser | China | 1985 | ||
| Shaji 2 | |||||
| Fuku-ekubo | Gamma rays (30 Gy) | Japan | 1996 | ||
| Shimizu Hakutou RS | 2004 | ||||
| Plovdiv 6 | Gamma rays (10 Gy) | Bulgaria | 1981 | ||
| Pear | Pyrus communis L. | Fuxiangyanghongdli | Gamma rays (2.5 Gy) | China | 1983 |
| Chaofu 1 | 1989 | ||||
| Chaofu 10 | |||||
| Chaofu 10 | |||||
| Chaofu 2 | |||||
| Plum | Prunus domestica L. | Spurdente-Ferco | Gamma rays | France | 1988 |
| Pomegranate | Punica granatum L. | Karabakh | Gamma rays (50–70 Gy) | Russian Federation | 1979 |
| Khyrda | |||||
| Sour cherry | Prunus cerasus L. | Plodorodnaya Michurina | X-rays | Russian Federation | 1977 |
| Karlik Samorodka | Gamma rays | 1979 | |||
| Polukarlik Orlovskoi Rannei | |||||
| Polukarlik Turgenevki | |||||
| Nishina Zao (DT2008) | Ion beams | Japan | 2009 | ||
| Sweet cherry | Prunus avium L. | Compact Lambert | X-rays (40 Gy) | Canada | 1964 |
| Compact Stella 35B-11 | 1974 | ||||
| Van 2D-14-11 | 1972 | ||||
| Lapins | X-rays | 1983 | |||
| Lambert 2B-17-18-EC | X-rays (50 Gy) | 1972 | |||
| Stella | 1968 | ||||
| Stella 16A-7 | 1972 | ||||
| Sunburst | 1983 | ||||
| Sumste Samba | Gamma rays | 2000 | |||
| ALDAMLA | Gamma rays (25 Gy) | Turkey | 2014 | ||
| BURAK | Gamma rays (50 Gy) | ||||
| Burlat C1 | Gamma rays | Italy | 1983 | ||
| Nero II C1 | |||||
| Ferrovia spur | X-rays (4 Gy) | 1992 | |||
| Super 6 | Colchicine | Japan | 1997 | ||
| Roman Nishiki | 2002 |
| Country | Cartagena Protocol of Biosafety (CPB) Status | Regulations for Different GE Categories | ||
|---|---|---|---|---|
| SDN-1 | SDN-2 | SDN-3 | ||
| Argentina | Non-ratified | Non-GMO | Cas-by-case decision | Non-GMO if no transgene |
| Australia | Non-ratified | Non-GMO | GMO | Not clear |
| Brazil | Ratified | Non-GMO (with previous consultation) | Cas-by-case decision | Non-GMO if no transgene |
| Canada | Non-ratified | Case-by-case (based upon novelty) | ||
| Chile | Non-ratified | Non-GMO (with previous consultation) | Non-GMO | Non-GMO if no transgene |
| China | Ratified | Under review with new legislations | ||
| Colombia | Ratified | Non-GMO (with previous consultation) | Non-GMO (Case-by-case decision) | Non-GMO if no transgene |
| European Union | Ratified | GMO | ||
| Guatemala El Salvador | Ratified | No clear regulations | ||
| Honduras | Ratified | Case-by-case non-GMO | Non-GMO if no transgene | |
| India | Ratified | Currently unclear (under discussion) | ||
| Israel | Non-ratified | Non-GMO | Non-GMO if no transgene | |
| Japan | Ratified | Non-GMO if no extracellular footprints were integrated into the organism genome | GMO (if extracellular footprints remain) | |
| New Zealand | Ratified | GMO | ||
| Norway | Ratified | Currently unclear (proposal under review) | ||
| Paraguay | Ratified | Unclear (may vary Case-by-case) | ||
| Russian Federation | Non-ratified | Unclear due to new expected policies | ||
| South Africa | Non-ratified | Currently unclear (under discussion) | ||
| Switzerland | Ratified | Currently unclear (under discussion) | ||
| United States | Non-ratified | Case-by-case (USDA); clarification under discussion (FDA) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sattar, M.N.; Iqbal, Z.; Al-Khayri, J.M.; Jain, S.M. Induced Genetic Variations in Fruit Trees Using New Breeding Tools: Food Security and Climate Resilience. Plants 2021, 10, 1347. https://doi.org/10.3390/plants10071347
Sattar MN, Iqbal Z, Al-Khayri JM, Jain SM. Induced Genetic Variations in Fruit Trees Using New Breeding Tools: Food Security and Climate Resilience. Plants. 2021; 10(7):1347. https://doi.org/10.3390/plants10071347
Chicago/Turabian StyleSattar, Muhammad Naeem, Zafar Iqbal, Jameel M. Al-Khayri, and S. Mohan Jain. 2021. "Induced Genetic Variations in Fruit Trees Using New Breeding Tools: Food Security and Climate Resilience" Plants 10, no. 7: 1347. https://doi.org/10.3390/plants10071347








