Anticancer Potential of Natural Bark Products—A Review
Abstract
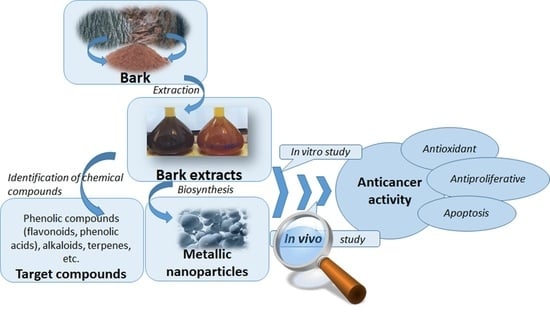
:1. Introduction
2. Research Methodology
3. Anticancer Potential of Bioactive Compounds from Bark Extracts (BE) in Different Type of Suppressed Cancer Cells Lines
4. Biosynthesized Metallic Nanoparticles (BMN) Mediated by Bark Extracts as Anticancer Agents
5. BE and BMNs Mechanism of AntiCancer Action
6. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ediriweera, M.K.; Tennekoon, K.H.; Samarakoon, S.R.; Thabrew, I.; De Silva, E.D. A study of the potential anticancer activity of Mangifera zeylanica bark: Evaluation of cytotoxic and apoptotic effects of the hexane extract and bioassay-guided fractionation to identify phytochemical constituents. Oncol. Lett. 2016, 11, 1335–1344. [Google Scholar] [CrossRef] [Green Version]
- Chanda, S.; Nagani, K. In Vitro and in Vivo Methods for Anticancer Activity Evaluation and Some Indian Medicinal Plants Possessing Anticancer Properties: An Overview. J. Pharm. Phytochem. 2013, 2, 140–152. [Google Scholar]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse Effects of Cancer Chemotherapy: Anything New to Improve Tolerance and Reduce Sequelae? Front. Pharmacol. 2018, 9, 245. [Google Scholar] [CrossRef]
- Fock, K.M. Review article: The epidemiology and prevention of gastric cancer. Aliment. Pharmacol. Ther. 2014, 40, 250–260. [Google Scholar] [CrossRef]
- Alawode, T. An Overview of the Anticancer Properties of Some Plants Used in Traditional Medicine in Nigeria. Int. Res. J. Biochem. Bioinform. 2013, 3, 7–14. [Google Scholar]
- Mishra, T.; Arya, R.K.; Meena, S.; Joshi, P.; Pal, M.; Meena, B.; Upreti, D.K.; Rana, T.S.; Datta, D. Isolation, Characterization and Anticancer Potential of Cytotoxic Triterpenes from Betula utilis Bark. PLoS ONE 2016, 11, e0159430. [Google Scholar] [CrossRef]
- De Wilt, L.H.; Jansen, G.; Assaraf, Y.G.; van Meerloo, J.; Cloos, J.; Schimmer, A.; Chan, E.T.; Kirk, C.J.; Peters, G.J.; Kruyt, F.A. Proteasome-based mechanisms of intrinsic and acquired bortezomib resistance in non-small cell lung cancer. Biochem. Pharmacol. 2012, 83, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Thamizhiniyan, V.; Young-Woong, C.; Young-Kyoon, K. The cytotoxic nature of Acanthopanax sessiliflorus stem bark extracts in human breast cancer cells. Saudi J. Biol. Sci. 2015, 22, 752–759. [Google Scholar] [CrossRef] [Green Version]
- Santos-Sánchez, N.F.; Salas-Coronado, R.; Villanueva-Cañongo, C.; Hernández-Carlos, B. Antioxidant Compounds and Their Antioxidant Mechanism; IntechOpen: London, UK, 2019. [Google Scholar]
- Greenwell, M.; Rahman, P. Medicinal Plants: Their Use in Anticancer Treatment. Int. J. Pharm. Sci. Res. 2015, 6, 4103–4112. [Google Scholar] [CrossRef]
- Jerez, M.; Selga, A.; Sineiro, J.; Torres, J.L.; Núñez, M.J. A comparison between bark extracts from Pinus pinaster and Pinus radiata: Antioxidant activity and procyanidin composition. Food Chem. 2007, 100, 439–444. [Google Scholar] [CrossRef]
- Tariq, A.; Sadia, S.; Pan, K.; Ullah, I.; Mussarat, S.; Sun, F.; Abiodun, O.O.; Batbaatar, A.; Li, Z.; Song, D.; et al. A systematic review on ethnomedicines of anti-cancer plants. Phytother. Res. 2017, 31, 202–264. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.; Kim, G.-D. Eco-friendly approach for nanoparticles synthesis and mechanism behind antibacterial activity of silver and anticancer activity of gold nanoparticles. Appl. Microbiol. Biotechnol. 2017, 101, 79–92. [Google Scholar] [CrossRef] [PubMed]
- El-Shemy, H.A.; Aboul-Enein, A.M.; Aboul-Enein, K.M.; Fujita, K. Willow Leaves’ Extracts Contain Anti-Tumor Agents Effective against Three Cell Types. PLoS ONE 2007, 2, e178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhandari, J.; Muhammad, B.; Thapa, P.; Shrestha, B.G. Study of phytochemical, anti-microbial, anti-oxidant, and anti-cancer properties of Allium wallichii. BMC Complement. Altern. Med. 2017, 17, 102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prema, R.; Sekar, S.D.; Chandra Sekhar, K.B. Review On: Herbs as Anticancer Agents. Int. J. Pharma Ind. Res. 2011, 1, 105. [Google Scholar]
- BioRender. Available online: https://App.Biorender.Com/ (accessed on 31 August 2021).
- Iqbal, J.; Abbasi, B.H.; Mahmood, T.; Kanwal, S.; Ali, B.; Shah, S.A.; Khalil, A.T. Plant-derived anticancer agents: A green anticancer approach. Asian Pac. J. Trop. Biomed. 2017, 7, 1129–1150. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, Y.; Padilla-Zakour, O.; Yang, G. Polyphenols, antioxidant and antimicrobial activities of leaf and bark extracts of Solidago canadensis L. Ind. Crop. Prod. 2015, 75, 803–809. [Google Scholar] [CrossRef]
- Kasote, D.; Katyare, S.S.; Hegde, M.V.; Bae, H. Significance of Antioxidant Potential of Plants and its Relevance to Therapeutic Applications. Int. J. Biol. Sci. 2015, 11, 982–991. [Google Scholar] [CrossRef] [Green Version]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [Green Version]
- Scalbert, A.; Manach, C.; Morand, C.; Rémésy, C.; Jiménez, L. Dietary Polyphenols and the Prevention of Diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Niedzwiecki, A.; Roomi, M.W.; Kalinovsky, T.; Rath, M. Anticancer Efficacy of Polyphenols and Their Combinations. Nutrients 2016, 8, 552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsieh, T.-C.; Wu, J.M. Targeting CWR22Rv1 prostate cancer cell proliferation and gene expression by combinations of the phytochemicals EGCG, genistein and quercetin. Anticancer. Res. 2009, 29, 4025–4032. [Google Scholar] [PubMed]
- Tang, S.-N.; Singh, C.; Nall, D.; Meeker, D.; Shankar, S.; Srivastava, R.K. The dietary bioflavonoid quercetin synergizes with epigallocathechin gallate (EGCG) to inhibit prostate cancer stem cell characteristics, invasion, migration and epithelial-mesenchymal transition. J. Mol. Signal. 2010, 5, 14. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, K.A.; Harris, N.H.; Johnson, A.D.; Lindvall, H.C.; Wang, G.; Ahmed, K. Protein kinase CK2 modulates apoptosis induced by resveratrol and epigallocatechin-3-gallate in prostate cancer cells. Mol. Cancer Ther. 2007, 6, 1006–1012. [Google Scholar] [CrossRef] [Green Version]
- Elansary, H.O.; Szopa, A.; Kubica, P.; El-Ansary, D.O.; Ekiert, H.; Al-Mana, F.A. Malus baccata var. gracilis and Malus toringoides Bark Polyphenol Studies and Antioxidant, Antimicrobial and Anticancer Activities. Processes 2020, 8, 283. [Google Scholar] [CrossRef] [Green Version]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Mattar, M.A.; Al-Yafrasi, M.A.; El-Ansary, D.O.; El-Abedin, T.K.Z.; Yessoufou, K. Polyphenol Profile and Pharmaceutical Potential of Quercus spp. Bark Extracts. Plants 2019, 8, 486. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.-J.; Bao, J.-L.; Chen, X.-P.; Huang, M.; Wang, Y.-T. Alkaloids Isolated from Natural Herbs as the Anticancer Agents. Evid. Based Complement. Altern. Med. 2012, 2012, 485042. [Google Scholar] [CrossRef] [Green Version]
- Boyer, N.C.; Morrison, K.C.; Kim, J.; Hergenrother, P.J.; Movassaghi, M. Synthesis and anticancer activity of epipolythiodiketopiperazine alkaloids. Chem. Sci. 2013, 4, 1646–1657. [Google Scholar] [CrossRef] [Green Version]
- Kingston, D.G.I.; Gerhart, B.B.; Ionescu, F.; Mangino, M.M.; Sami, S.M. Plant Anticancer Agents V: New Bisindole Alkaloids from Tabernaemontana johnstonii Stem Bark. J. Pharm. Sci. 1978, 67, 249–251. [Google Scholar] [CrossRef] [PubMed]
- Isah, T. Anticancer alkaloids from trees: Development into drugs. Pharmacogn. Rev. 2016, 10, 90–99. [Google Scholar] [CrossRef] [Green Version]
- Jaafari, A.; Tilaoui, M.; Mouse, H.A.; M’Bark, L.A.; Aboufatima, R.; Chait, A.; Lepoivre, M.; Zyad, A. Comparative study of the antitumor effect of natural monoterpenes: Relationship to cell cycle analysis. Rev. Bras. Farm. 2012, 22, 534–540. [Google Scholar] [CrossRef] [Green Version]
- Lombrea, A.; Scurtu, A.; Avram, S.; Pavel, I.; Turks, M.; Lugiņina, J.; Peipiņš, U.; Dehelean, C.; Soica, C.; Danciu, C. Anticancer Potential of Betulonic Acid Derivatives. Int. J. Mol. Sci. 2021, 22, 3676. [Google Scholar] [CrossRef]
- Wang, X.-H.; Zhou, S.-Y.; Qian, Z.-Z.; Zhang, H.-L.; Qiu, L.-H.; Song, Z.; Zhao, J.; Wang, P.; Hao, X.-S.; Wang, H.-Q. Evaluation of toxicity and single-dose pharmacokinetics of intravenous ursolic acid liposomes in healthy adult volunteers and patients with advanced solid tumors. Expert Opin. Drug Metab. Toxicol. 2012, 9, 117–125. [Google Scholar] [CrossRef]
- Dehelean, C.A.; Şoica, C.; Ledeţi, I.; Aluaş, M.; Zupko, I.; Gǎluşcan, A.; Cinta-Pinzaru, S.; Munteanu, M. Study of the betulin enriched birch bark extracts effects on human carcinoma cells and ear inflammation. Chem. Cent. J. 2012, 6, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González-Sarrías, A.; Li, L.; Seeram, N.P. Effects of Maple (Acer) Plant Part Extracts on Proliferation, Apoptosis and Cell Cycle Arrest of Human Tumorigenic and Non-tumorigenic Colon Cells. Phytother. Res. 2011, 26, 995–1002. [Google Scholar] [CrossRef]
- Bagheri, G.; Mirzaei, M.; Mehrabi, R.; Sharifi-Rad, J. Cytotoxic and Antioxidant Activities of Alstonia scholaris, Alstonia venenata and Moringa oleifera Plants from India. Jundishapur J. Nat. Pharm. Prod. 2016, 11, 31129. [Google Scholar] [CrossRef] [Green Version]
- Dolai, N.; Karmakar, I.; Kumar, R.S.; Kar, B.; Bala, A.; Haldar, P.K. Evaluation of antitumor activity and in vivo antioxidant status of Anthocephalus cadamba on Ehrlich ascites carcinoma treated mice. J. Ethnopharmacol. 2012, 142, 865–870. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.K.; Ezzat, M.O.; Majid, A.S.A.; Majid, A.M.S.A. The Anticancer, Antioxidant and Antimicrobial Properties of the Sesquiterpene β-Caryophyllene from the Essential Oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef] [PubMed]
- Hassan, L.E.A.; Dahham, S.S.; Saghir, S.; Mohammed, A.M.A.; Eltayeb, N.M.; Majid, A.M.S.A.; Majid, A.S.A. Chemotherapeutic potentials of the stem bark of Balanite aegyptiaca (L.) Delile: An antiangiogenic, antitumor and antioxidant agent. BMC Complement. Altern. Med. 2016, 16, 396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basri, D.F.; Alamin, Z.A.Z.; Chan, K.M. Assessment of cytotoxicity and genotoxicity of stem bark extracts from Canarium odontophyllum Miq. (dabai) against HCT 116 human colorectal cancer cell line. BMC Complement. Altern. Med. 2015, 16, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Al-Mana, F.A.; Mahmoud, E.A.; El-Abedin, T.K.A.Z.; Mattar, M.A.; Ekiert, H. Phenolic Compounds of Catalpa speciosa, Taxus cuspidate, and Magnolia acuminata have Antioxidant and Anticancer Activity. Molecules 2019, 24, 412. [Google Scholar] [CrossRef] [Green Version]
- Badrinarayanan, V.; Anand, V.; Karpagam, T.; Bai, J.S.; Manikandan, R. In Vitro Antimicrobial and Anticancer Activity of Cinnamomum Zeylanicum Linn Bark Extracts. Int. J. Pharm. Pharmaceut. Sci. 2014, 6, 12–18. [Google Scholar]
- Sathuvan, M.; Vignesh, A.; Thangam, R.; Palani, P.; Rengasamy, R.; Murugesan, K. In Vitro Antioxidant and Anticancer potential of Bark of Costus pictus D.DON. Asian Pac. J. Trop. Biomed. 2012, 2, S741–S749. [Google Scholar] [CrossRef]
- Baloglu, M.C.; Llorent-Martínez, E.J.; Aumeeruddy, M.Z.; Mahomoodally, M.F.; Altunoglu, Y.C.; Ustaoglu, B.; Ocal, M.; Gürel, S.; Bene, K.; Sinan, K.I.; et al. Multidirectional insights on Chrysophyllum perpulchrum leaves and stem bark extracts: HPLC-ESI-MSn profiles, antioxidant, enzyme inhibitory, antimicrobial and cytotoxic properties. Ind. Crop. Prod. 2019, 134, 33–42. [Google Scholar] [CrossRef]
- Kanunfre, C.C.; Leffers, T.; Cruz, L.S.; Luz, L.E.; Crisma, A.R.; Wang, M.; Avula, B.; Khan, I.A.; Beltrame, F.L. Euphorbia umbellata bark extracts—an in vitro cytotoxic study. Rev. Bras. Farm. 2016, 27, 206–213. [Google Scholar] [CrossRef]
- Coșarcă, S.-L.; Moacă, E.-A.; Tanase, C.; Muntean, D.L.; Pavel, I.Z.; Dehelean, C.A. Spruce and beech bark aqueous extracts: Source of polyphenols, tannins and antioxidants correlated to in vitro antitumor potential on two different cell lines. Wood Sci. Technol. 2019, 53, 313–333. [Google Scholar] [CrossRef]
- Yessoufou, K.; Elansary, H.O.; Mahmoud, E.A.; Skalicka-Woźniak, K. Antifungal, antibacterial and anticancer activities of Ficus drupacea L. stem bark extract and biologically active isolated compounds. Ind. Crop. Prod. 2015, 74, 752–758. [Google Scholar] [CrossRef]
- Prasanthi, D.; Adikay, S. Pharmacognostic Studies and Nephroprotective Potential of Hydroalcoholic Extract of Trichosanthes cucumerina in Acute Renal Failure. Pharmacogn. J. 2017, 9, 176–184. [Google Scholar] [CrossRef] [Green Version]
- Al-Asmari, A.K.; AlBalawi, S.M.; Athar, T.; Khan, A.Q.; Al-Shahrani, H.; Islam, M. Moringa oleifera as an Anti-Cancer Agent against Breast and Colorectal Cancer Cell Lines. PLoS ONE 2015, 10, e0135814. [Google Scholar] [CrossRef]
- Johnson-Ajinwo, O.R.; Richardson, A.; Li, W.-W. Cytotoxic effects of stem bark extracts and pure compounds from Margaritaria discoidea on human ovarian cancer cell lines. Phytomedicine 2015, 22, 1–4. [Google Scholar] [CrossRef]
- Moirangthem, D.S.; Talukdar, N.C.; Bora, U.; Kasoju, N.; Das, R.K. Differential effects of Oroxylum indicum bark extracts: Antioxidant, antimicrobial, cytotoxic and apoptotic study. Cytotechnology 2012, 65, 83–95. [Google Scholar] [CrossRef] [Green Version]
- Wu, D.-C.; Li, S.; Yang, D.-Q.; Cui, Y.-Y. Effects of Pinus massoniana bark extract on the adhesion and migration capabilities of HeLa cells. Fitoterapia 2011, 82, 1202–1205. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.K.; Saini, K.S.; Hossain, Z.; Omer, A.; Sharma, C.; Gayen, J.R.; Singh, P.; Arya, K.R.; Singh, R.K. Saraca indicaBark Extract ShowsIn VitroAntioxidant, Antibreast Cancer Activity and Does Not Exhibit Toxicological Effects. Oxidative Med. Cell. Longev. 2015, 2015, 205360. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Jain, S.K. Screening of in vitro cytotoxic activity of some medicinal plants used traditionally to treat cancer in Chhattisgarh state, India. Asian Pac. J. Trop. Biomed. 2011, 1, S147–S150. [Google Scholar] [CrossRef]
- Ghate, N.B.; Hazra, B.; Sarkar, R.; Mandal, N. In vitro anticancer activity of Spondias pinnata bark on human lung and breast carcinoma. Cytotechnology 2013, 66, 209–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baldivia, D.D.S.; Leite, D.F.; De Castro, D.T.H.; Campos, J.F.; Dos Santos, U.P.; Paredes-Gamero, E.J.; Carollo, C.A.; Silva, D.B.; Souza, K.D.P.; Dos Santos, E.L. Evaluation of In Vitro Antioxidant and Anticancer Properties of the Aqueous Extract from the Stem Bark of Stryphnodendron adstringens. Int. J. Mol. Sci. 2018, 19, 2432. [Google Scholar] [CrossRef] [Green Version]
- Ravi, A.; Mallika, A.; Sama, V.; Begum, A.S.; Khan, R.S.; Reddy, B.M. Antiproliferative activity and standardization of Tecomella undulata bark extract on K562 cells. J. Ethnopharmacol. 2011, 137, 1353–1359. [Google Scholar] [CrossRef]
- Baharum, Z.; Akim, A.M.; Taufiq-Yap, Y.H.; Hamid, R.A.; Kasran, R. In Vitro Antioxidant and Antiproliferative Activities of Methanolic Plant Part Extracts of Theobroma cacao. Molecules 2014, 19, 18317–18331. [Google Scholar] [CrossRef] [Green Version]
- Nagati, V.; Koyyati, R.; Donda, M.; Alwala, J.; Kudle, K. Green Synthesis and Characterization of Silver Nanoparticles from Cajanus Cajanleaf Extract and Its Antibacterial Activity. Int. J. Nanomater. Biostruct. 2012, 2, 39–43. [Google Scholar]
- Mousavi, B.; Tafvizi, F.; Bostanabad, S.Z. Green synthesis of silver nanoparticles using Artemisia turcomanica leaf extract and the study of anti-cancer effect and apoptosis induction on gastric cancer cell line (AGS). Artif. Cells Nanomed. Biotechnol. 2018, 46, 499–510. [Google Scholar] [CrossRef] [Green Version]
- Rajan, R.; Chandran, K.; Harper, S.L.; Yun, S.-I.; Kalaichelvan, P.T. Plant extract synthesized silver nanoparticles: An ongoing source of novel biocompatible materials. Ind. Crop. Prod. 2015, 70, 356–373. [Google Scholar] [CrossRef]
- Nayak, D.; Ashe, S.; Rauta, P.R.; Kumari, M.; Nayak, B. Bark extract mediated green synthesis of silver nanoparticles: Evaluation of antimicrobial activity and antiproliferative response against osteosarcoma. Mater. Sci. Eng. C 2016, 58, 44–52. [Google Scholar] [CrossRef]
- Odeyemi, S.W.; De La Mare, J.; Edkins, A.L.; Afolayan, A.J. In vitro and in vivo toxicity assessment of biologically synthesized silver nanoparticles from Elaeodendron croceum. J. Complement. Integr. Med. 2019, 16, 20180184. [Google Scholar] [CrossRef]
- Saravanakumar, K.; Chelliah, R.; MubarakAli, D.; Oh, D.-H.; Kathiresan, K.; Wang, M.-H. Unveiling the potentials of biocompatible silver nanoparticles on human lung carcinoma A549 cells and Helicobacter pylori. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Khan, S.A.; Bello, B.A.; Khan, J.A.; Anwar, Y.; Mirza, M.B.; Qadri, F.; Farooq, A.; Adam, I.K.; Asiri, A.; Khan, S.B. Albizia chevalier based Ag nanoparticles: Anti-proliferation, bactericidal and pollutants degradation performance. J. Photochem. Photobiol. B Biol. 2018, 182, 62–70. [Google Scholar] [CrossRef]
- Umar, H.; Kavaz, D.; Rizaner, N. Biosynthesis of zinc oxide nanoparticles using Albizia lebbeck stem bark, and evaluation of its antimicrobial, antioxidant, and cytotoxic activities on human breast cancer cell lines. Int. J. Nanomed. 2018, 14, 87–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanjikar, A.P.; Hugar, A.L.; Londonkar, R.L. Characterization of phyto-nanoparticles from Ficus krishnae for their antibacterial and anticancer activities. Drug Dev. Ind. Pharm. 2018, 44, 377–384. [Google Scholar] [CrossRef]
- Zhang, X.; Xiao, C. Biofabrication of silver nanoparticles and their combined effect with low intensity ultrasound for treatment of lung cancer. J. Photochem. Photobiol. B Biol. 2018, 181, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Vasanth, K.; Ilango, K.; MohanKumar, R.; Agrawal, A.; Dubey, G.P. Anticancer activity of Moringa oleifera mediated silver nanoparticles on human cervical carcinoma cells by apoptosis induction. Colloids Surfaces B Biointerfaces 2014, 117, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Barai, A.C.; Paul, K.; Dey, A.; Manna, S.; Roy, S.; Bag, B.G.; Mukhopadhyay, C. Green synthesis of Nerium oleander-conjugated gold nanoparticles and study of its in vitro anticancer activity on MCF-7 cell lines and catalytic activity. Nano Converg. 2018, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Arya, G.; Kumari, R.M.; Gupta, N.; Kumar, A.; Chandra, R.; Nimesh, S. Green synthesis of silver nanoparticles using Prosopis juliflora bark extract: Reaction optimization, antimicrobial and catalytic activities. Artif. Cells Nanomed. Biotechnol. 2018, 46, 985–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Francis, S.; Koshy, E.P.; Mathew, B. Green synthesis of Stereospermum suaveolens capped silver and gold nanoparticles and assessment of their innate antioxidant, antimicrobial and antiproliferative activities. Bioprocess Biosyst. Eng. 2018, 41, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Yugandhar, P.; Vasavi, T.; Devi, P.U.M.; Savithramma, N. Bioinspired green synthesis of copper oxide nanoparticles from Syzygium alternifolium (Wt.) Walp: Characterization and evaluation of its synergistic antimicrobial and anticancer activity. Appl. Nanosci. 2017, 7, 417–427. [Google Scholar] [CrossRef] [Green Version]
- Yallappa, S.; Manjanna, J.; Dhananjaya, B.L.; Vishwanatha, U.; Ravishankar, B.; Gururaj, H.; Niranjana, P.; Hungund, B.S. Phytochemically Functionalized Cu and Ag Nanoparticles Embedded in MWCNTs for Enhanced Antimicrobial and Anticancer Properties. Nano-Micro Lett. 2015, 8, 120–130. [Google Scholar] [CrossRef] [Green Version]
- Majoumouo, M.S.; Sharma, J.R.; Sibuyi, N.R.S.; Tincho, M.B.; Boyom, F.F.; Meyer, M. Synthesis of Biogenic Gold Nanoparticles from Terminalia mantaly Extracts and the Evaluation of Their In Vitro Cytotoxic Effects in Cancer Cells. Molecules 2020, 25, 4469. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, K.; Deore, S.; Dhamecha, D.; Jagwani, S.; Jalalpure, S.; Bohara, R. Phytosynthesis of Silver Nanoparticles: Characterization, Biocompatibility Studies, and Anticancer Activity. ACS Biomater. Sci. Eng. 2018, 4, 892–899. [Google Scholar] [CrossRef]
- Bhattacharya, S. Anticarcinogenic Property of Medicinal Plants: Involvement of Antioxidant Role; Research Signpost: Kerala, India, 2012; pp. 83–96. [Google Scholar]
- Bold, R.J.; Termuhlen, P.M.; McConkey, D.J. Apoptosis, cancer and cancer therapy. Surg. Oncol. 1997, 6, 133–142. [Google Scholar] [CrossRef]
- Bellucci, M.; Agostini, F.; Masin, M.; Tartaglia, G.G. Predicting protein associations with long noncoding RNAs. Nat. Methods 2011, 8, 444–445. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, X.-L.; Li, Y.-Y.; Cui, Y.-Y.; Chen, Y.-H. Pinus massoniana Bark Extract: Structure–Activity Relationship and Biomedical Potentials. Am. J. Chin. Med. 2016, 44, 1559–1577. [Google Scholar] [CrossRef]
- Burlacu, E.; Tanase, C.; Coman, N.-A.; Berta, L. A Review of Bark-Extract-Mediated Green Synthesis of Metallic Nanoparticles and Their Applications. Molecules 2019, 24, 4354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, K.-R.; Nam, D.; Yun, H.-M.; Lee, S.-G.; Jang, H.-J.; Sethi, G.; Cho, S.K.; Ahn, K.S. β-Caryophyllene oxide inhibits growth and induces apoptosis through the suppression of PI3K/AKT/mTOR/S6K1 pathways and ROS-mediated MAPKs activation. Cancer Lett. 2011, 312, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxid. Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, J.D.; Subramanian, T.; Liu, K.; King, M.R. Rafting Down the Metastatic Cascade: The Role of Lipid Rafts in Cancer Metastasis, Cell Death, and Clinical Outcomes. Cancer Res. 2021, 81, 5–17. [Google Scholar] [CrossRef]
- Hocman, G. Chemoprevention of cancer: Phenolic antioxidants (BHT, BHA). Int. J. Biochem. 1988, 20, 639–651. [Google Scholar] [CrossRef]
- Carlson, C.; Hussain, S.M.; Schrand, A.M.; Braydich-Stolle, L.K.; Hess, K.L.; Jones, R.L.; Schlager, J.J. Unique Cellular Interaction of Silver Nanoparticles: Size-Dependent Generation of Reactive Oxygen Species. J. Phys. Chem. B 2008, 112, 13608–13619. [Google Scholar] [CrossRef]


| Source of Bark: Scientific Name (Family)—Common Name | Extracts Type | Main Bioactive Compounds | Experiment Type | Type of Suppressed Cancer Cells Lines | References |
|---|---|---|---|---|---|
| Acanthopanax sessiliflorus (Decne & Planch) Miq. (Araliaceae)—thorny ginseng | n-hexane fraction of stem bark methanolic extract | - | In vitro | MDA-MB-231 and MCF-7 human breast cancer | [8] |
| Acer rubrum L. (Sapindaceae)—red maple | Methanolic extract | Polyphenols—ginnalin-A | In vitro | HCT-116 Caco-2 HT-29 colon cancer cell lines | [38] |
| Acer saccharum March. (Sapindaceae)—sugar maple | Methanolic extract | Polyphenols—ginnalin-A | In vitro | HCT-116 Caco-2 HT-29 colon cancer cell lines | [38] |
| Alstonia scholaris (L.) R.Br. (Apocynaceae)—devil`s tree | Hexane extract | In vitro | DLA: dalton`s lymphoma ascitic cell line | [39] | |
| Alstonia venenata R. Br. (Apocynaceae) | Hexane extract | In vitro | DLA: dalton`s lymphoma ascitic cell line | [39] | |
| Antocephalus cadamba (Roxb.) Miq. (Rubiaceae)—kadam | Petroleum ether Methanolic extract | - | In vivo on Swiss albino mice | EAC: Ehrlich ascites carcinoma | [40] |
| Aquilaria crassna Pierre (Thymelaeaceae)—Agar wood | essential oil from stem bark extract | β-caryophyllene | In vitro | PANC-1 pancreatic cancer, HCT-116 and HT29 colorectal cancer | [41] |
| Balanite aegyptiaca (L.) Delile (Balamitaceae)—desert date | Choloform Methanolic water Hexane extract | Alkaloids | In vivo antitumor activity in nude mice induced with HCT-116 tumor | HCT-116 colon cancer K562 myelogenous leukemia U937 histyocitic lymphoma MCF-7 breast cancer | [42] |
| Betula pendula Roth (Betulaceae)—common silver birch | 2-propanol and ethanolic extract | Betulin and betulinic acid | In vitro | HeLa human cervical cancer; A431 human squamous carcinoma; A2780 ovarian carcinoma; MCF-7 breast cancer | [37] |
| Betula utilis D.Don (Betulaceae)—Himalayan silver birch | Ethyl acetate extract | Triterpenes | In vitro | MCF-7 | [6] |
| Canarium odontophyllum Miq. (Burseraceae)—dabai | Acetone extract | Flavonoids Saponins Tannins Terpenoids and phenolic compounds | In vitro | HCT 116 colorectal cancer | [43] |
| Catalpa speciosa Warder (Bignoniaceae)—northern catalpa | Methanolic bark extract | Phenolic acids catechins | In vitro | HeLa cervical cancer; MCF-7 breast cancer; Jurkat leukemic T cells; T24 urinary bladder carcinoma; HT-29 colorectal adenocarcinoma | [44] |
| Cinnamomum zeylanicum Blume. (Lauraceae)—true cinnamon tree | Methanolic extract | - | In vitro | HepG2 hepato carcinoma | [45] |
| Costus pictus D.Don (Costaceae)—painted spiral ginger | Methanolic extract | Polyphenols Flavonoids | In vitro | HT-29 colon cancer; A549 lung carcinoma | [46] |
| Crysophillum perpulchrum L. (Sapotaceae) | Methanolic bark extract | Polyphenols | In vitro | HeLa cervical cancer | [47] |
| Euphorbia umbellata (Pax) Bruyns (Euphorbiaceae)—African milk bush | Water and methanolic extract | Triterpenes Steroids | In vitro | Jurkat cells T-cell leukemia | [48] |
| Fagus sylvatica L. (Fagaceae)—beech | Water bath extraction and ultrasound-assisted extraction | Polyphenols | In vitro | A375 melanoma; A549 lung carcinoma | [49] |
| Ficus drupacea Thunb. (Moraceae) —brown-woolly fig | Methanolic extract, n-hexane fraction | Oleanolic acid, friedelin, and epilupeol acetate | In vitro | HeLa cervical cancer; MCF-7 breast cancer; Jurkat leukemic T cells; HT-29 colorectal cancer; and T24 urinary bladder carcinoma | [50] |
| Khaya senegalensis (Desr.) A Juss (Meliaceae)—African mahogany | Methanolic, water, and SFE (subcritical fluid extraction) | Monoterpenes Sesquiterpene | In vitro | Caco 2 colorectal cancer; HeLa cervical cancer | [51] |
| Magnolia acuminata L. (Magnoliaceae)—cucumber tree | - | Phenolic acids—protocatechuic acid Catechin Epicatechin | In vitro | HeLa cervical cancer; MCF-7 breast cancer; Jurkat leukemic T cells; T24 urinary bladder carcinoma; HT-29 colorectal adenocarcinoma | [44] |
| Malus baccata var.gracilis (Rehder) T.C.Ku. (Rosaceae)—Siberian crab apple | Methanolic extract | Polyphenols protocatechuic acid, gallic acid, and catechin | In vitro | MCF-7 breast cancer cells, HeLa cervical cancer Jurkat cells T cell leukemia | [28] |
| Malus toringoides Hughes (Rosaceae)—cut-leaf crabapple | Methanolic extract | Polyphenols protocatechuic acid, gallic acid, and catechin | In vitro | MCF-7 breast cancer cells, HeLa cervical cancer Jurkat cells T cell leukemia | [28] |
| Mangifera zeylanica (Blume) Hook. f (Anacardiaceae)— Sri Lanka wild mango | Hexane, chloroform, ethyl acetate and methanol extract | Steroids, flavonoids, phenolic compounds, and tannins | In vitro | MCF-7 and MDA-MB-231 breast cancer SKOV-3 ovarian cancer | [1] |
| Moringa oleifera Lam. (Moringaceae)—drumstick tree | Hexane and benzene extract | - | In vitro | DLA: dalton`s lymphoma ascitic cell line | [39] |
| Moringa oleifera Lam. (Moringaceae)—drumstick tree | Ethanolic extract | Eugenol, isopropyl isothiocynate, D-allose, and hexadeconoic acid ethyl ester | In vitro | MDA-MB-231 breast cancer HCT–8 ileocecal adenocarcinoma | [52] |
| Margaritaria discoidea (Baill.) G. L. Webster (Euphorbiaceae)—pheasant-berry | Dichloroethane and methanol extract | Gallic acid Securinine | In vitro | OVCAR-8; A2780 ovarian cancer cell lines A2780cis cisplatin resistant ovarian cancer | [53] |
| Oroxylum indicum (L.)Benth. Ex Kurtz (Bignoniaceae)—Indian trumpet flower | Petroleum ether Dichloromethane Methanol extract | Polyphenols Flavonoids | In vitro | HeLa cervical adenocarcinoma | [54] |
| Picea abies L. (Pinaceae)—spruce | Water bath extraction and ultrasound-assisted extraction | Polyphenols, tannins, flavonoids, and flavonols | In vitro | A375 human melanoma; A549 lung carcinoma | [49] |
| Pinus massoniana Lamb. (Pinaceae)—chinese red pine | Flavonoids (Tannins)—proanthocyanidins | In vitro | HeLa cells cervical cancer | [55] | |
| Quercus acutissima Carruth. (Fagaceae)—Sawtooth oak | Methanolic extract | Phenolic acid—scaffeic acid, ellagic acid, gallic acid, and protocatechuic acid | In vitro | MCF-7 breast cancer; HeLa Cervical cancer Jurkat T-cell leukemia cell line | [29] |
| Quercus macrocarpa Michx. (Fagaceae)—bur oak | Methanolic extract | Phenolic acids—caffeic acid and catechins | In vitro | MCF-7 breast cancer; HeLa Cervical cancer Jurkat T-cell leukemia cell line | [29] |
| Quercus robur L. (Fagaceae)—common oak | Methanolic extract | Phenolic acids—ellagic acid, gallic acid, protocatechuic acid, and vanillic acid | In vitro | MCF-7 breast cancer; HeLa Cervical cancer Jurkat T-cell leukemia cell line T24 bladder transitional cell carcinoma | [29] |
| Saraca indica L. (Fabaceae)—asoka-tree | Alcoholic extract | Polyphenols | In vitro | MCF-7, MDA-MB-231 breast cancer | [56] |
| Sesbania grandiflora (L.) Poiret (Fabaceae)—vegetable hummingbird | Ethyl alcoholic extract | Phenolics terpenoids and phenolics flavonoids | In vitro | MCF-7 human breast cancer and HL-60 human leukemia | [57] |
| Spondias pinnata L.f.Kurz (Anacardiaceae)—wild mango | Water methanolic extract | - | In vitro | A549 lung carcinoma; MCF-7 breast carcinoma | [58] |
| Stryphnodendron adstringens (Mart.) Coville (Fabaceae)—barbatimao | Water extract | Proanthocyanidins (Tannins) Triterpenoids Gallic acid Gallocatechin epigallocathechin | In vitro | B16F10Nex-2 melanoma cells | [59] |
| Taxus cuspidata Siebold &Zucc (Taxaceae)—Japanese yew | Phenolic acids | In vitro | HeLa cervical cancer; MCF-7 breast cancer; Jurkat leukemic T cells; T24 urinary bladder carcinoma; HT-29 colorectal adenocarcinoma | [44] | |
| Tecomella undulata (Sm.) Seem. (Bignoniaceae)—desert teak | Chloroform extract | Steroids and Triterpenes Flavonoids | In vitro | K562 erythroleukemic cell line | [60] |
| Theobroma cacao L. (Malvaceae)—cocoa tree | Methanolic extract | Flavonoids Saponins Triterpenes Condensed tannins Steroids | In vitro | MCF-7 breast cancer | [61] |
| Wrghtia tinctoria (Roxb.)R. Br. (Apocynaceae)—pala indigo tree | Ethyl alcoholic extract | Alkaloids terpenoids Phenolics | In vitro | MCF-7 human breast cancer | [57] |
| Source of Bark: Scientific Name (Family)—Common Name | BMNs Type | Experiment Type | Type of Suppressed Cancer Cell Lines | References |
|---|---|---|---|---|
| Albizia chevaalieri Harms (Fabaceae) | Ag | In vitro | MDA-MB231, MCF-7 breast cancer and HepG2 liver cancer | [68] |
| Albizia lebbeck L. Benth. (Fabaceae)—lebbeck tree | ZnO | In vitro | MDA-MB-231; MCF-7 breast cancer | [69] |
| Azadirachta indica A.Juss. (Meliaceae)—Nimtree or Indian lillac | Ag | In vitro | MG-63 osteosarcoma | [65] |
| Elaeodendrum croceum (Thunb.) DC. (Celastraceae) —saffron | Ag | In vitro | MDA-MB-231 breast cancer | [66] |
| Ficus benghalensis L. (Moraceae)—banyan fig | Ag | In vitro | MG-63 osteosarcoma | [65] |
| Ficus benghalensis var. krishnae L. (Moraceae)—Krishna`s butter cup | Ag | In vitro | SKOV-3 ovarian cancer | [70] |
| Garcinia mangostana L. (Clusiaceae)—Mangosteen | Ag | In vitro | A549 lung cancer | [71] |
| Moringa oleifera L. (Moringaceae)—drumstick tree | Ag | In vitro | HeLa cervical cancer | [72] |
| Nerium oleander L. (Apocynaceae)—Karabi | Au | In vitro | MCF-7 breast cancer | [73] |
| Prosopis juliflora Sw. DC. (Fabaceae)—Mesquite | Ag | In vitro | A549 lung cancer | [74] |
| Stereospermum suaveolens Roxb. DC.—(Bignoniaceae) | Ag Au | In vitro | A549 lung cancer | [75] |
| Syzygium alternifolium (Wt.) Walp (Myrtiaceae)—North Arcot | Cu | In vitro | MDA-MB-231 breast cancer | [76] |
| Terminalia arjuna Wigh and Arn (Combretaceae)—Arjuna tree | Cu–Ag | In vitro | MDA-MB-231 breast cancer; HeLA cervical cancer; SiHa squamous cell carcinoma; HepG2 liver carcinoma | [77] |
| Terminalia mantaly H. Perrier (Combretaceae)—Madagascar almond, umbrella tree | Au | In vitro | Caco-2 colon cancer; MCF-7 breast cancer; HepG2 liver cancer | [78] |
| Toxicodendron vernicifluum (Stokes) F.A. Barkley (Anacardiaceae)—Chinese lacquer tree | Ag | In vitro | A549 Adenocarcinomic human alveolar basal epithelial cells | [67] |
| Salacia chinensis L. (Celastraceae)—Chinese salacia | Ag | In vitro | HepG2 liver cancer; L-132 lung cancer; MIA-Pa-Ca-2 pancreas cancer; MDA-MB-231 breast cancer; KB cells oral cancer; PC-3 prostate cancer; HeLa cervical cancer cells | [79] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burlacu, E.; Tanase, C. Anticancer Potential of Natural Bark Products—A Review. Plants 2021, 10, 1895. https://doi.org/10.3390/plants10091895
Burlacu E, Tanase C. Anticancer Potential of Natural Bark Products—A Review. Plants. 2021; 10(9):1895. https://doi.org/10.3390/plants10091895
Chicago/Turabian StyleBurlacu, Ema, and Corneliu Tanase. 2021. "Anticancer Potential of Natural Bark Products—A Review" Plants 10, no. 9: 1895. https://doi.org/10.3390/plants10091895