Drought-Induced Responses in Maize under Different Vapor Pressure Deficit Conditions
Abstract
:1. Introduction
2. Results
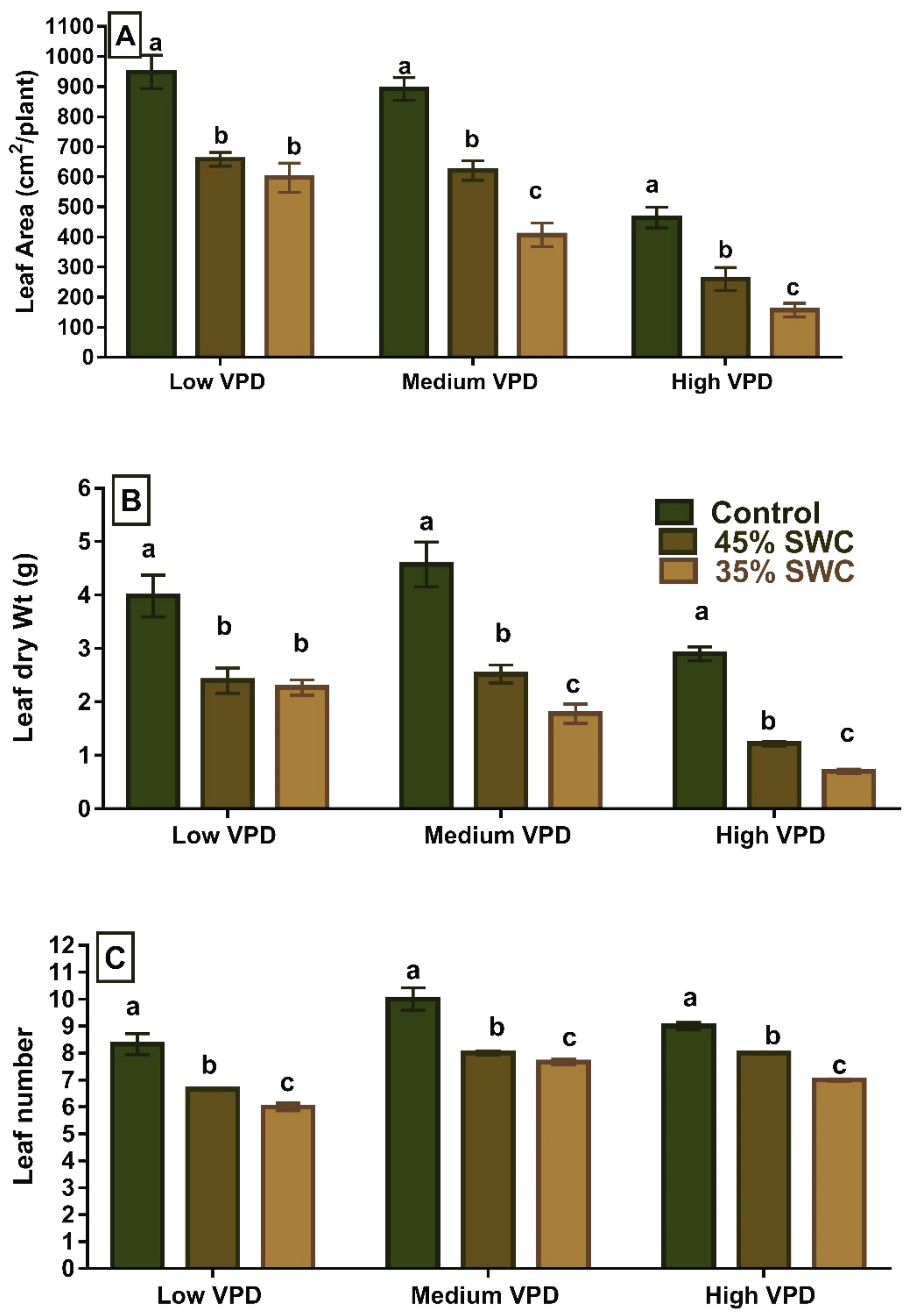
2.1. Leaf Traits
2.2. Gas Exchange
2.3. Enzyme Activities
2.4. Foliar ABA
2.5. Expression of Transcription Factors
3. Discussion
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Leaf Area, Weight, and Number
4.3. Gas Exchange
4.4. Analysis of Foliar ABA Content
4.5. Enzyme Extraction and Enzyme Activity Assays
4.6. RNA Extraction and Real Time Quantitative PCR
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. Water for Sustainable Food and Agriculture; Food and Agriculture Organization: Rome, Italy, 2017. [Google Scholar]
- Basso, B.; Ritchie, J.T. Evapotranspiration in High-Yielding Maize and under Increased Vapor Pressure Deficit in the US Midwest. Agric. Environ. Lett. 2018, 3, 170039. [Google Scholar] [CrossRef]
- Messina, C.D.; Sinclair, T.R.; Hammer, G.L.; Curan, D.; Thompson, J.; Oler, Z.; Gho, C.; Cooper, M. Limited-Transpiration Trait May Increase Maize Drought Tolerance in the US Corn Belt. Agronomy 2015, 107, 1978–1986. [Google Scholar] [CrossRef] [Green Version]
- Chavarria, G.; dos Santos, H.P. Plant water relations: Absorption, transport and control mechanisms. Advances in Selected Plant Physiology Aspects. Montanaro, G., Dichio, B., Eds.; Embrapa Uva e Vinho-Capítulo em livro científico (ALICE). 2012. Available online: https://books.google.com.sg/books?hl=en&lr=&id=o7qZDwAAQBAJ&oi=fnd&pg=PA105&dq=Plant+water+relations:+Absorption,+transport+and+control+mechanisms&ots=x5J_whObg2&sig=h-zqKOGerPTb6ngsUE7tFeNNes0&redir_esc=y#v=onepage&q=Plant%20water%20relations%3A%20Absorption%2C%20transport%20and%20control%20mechanisms&f=false (accessed on 18 October 2022).
- Devi, M.J.; Reddy, V.R. Transpiration Response of Cotton to Vapor Pressure Deficit and Its Relationship with Stomatal Traits. Front. Plant Sci. 2018, 9, 1572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Devi, M.J.; Reddy, V.R. Effect of temperature under different evaporative demand conditions on maize leaf expansion. Environ. Exp. Bot. 2018, 155, 509–517. [Google Scholar] [CrossRef]
- Lobell, D.B.; Roberts, M.J.; Schlenker, W.; Braun, N.; Little, B.B.; Rejesus, R.M.; Hammer, G.L. Greater Sensitivity to Drought Accompanies Maize Yield Increase in the U.S. Midwest. Science 2014, 344, 516–519. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.C. Agronomic options for improving rainfall-use efficiency of crops in dryland farming systems. J. Exp. Bot. 2004, 55, 2413–2425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kramer, P.J.; Boyer, J.S. Water Relations of Plants and Soils; Academic Press: Cambridge, MA, USA, 1995. [Google Scholar]
- Tardieu, F.; Reymond, M.; Hamard, P.; Granier, C.; Muller, B. Spatial distributions of expansion rate, cell division rate and cell size in maize leaves: A synthesis of the effects of soil water status, evaporative demand and temperature. J. Exp. Bot. 2000, 51, 1505–1514. [Google Scholar] [CrossRef] [Green Version]
- Devi, M.J.; Reddy, V.R. Stomatal closure response to soil drying at different vapor pressure deficit conditions in maize. Plant Physiol. Biochem. 2020, 154, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Devi, M.J.; Taliercio, E.W.; Sinclair, T.R. Leaf expansion of soybean subjected to high and low atmospheric vapour pressure deficits. J. Exp. Bot. 2015, 66, 1845–1850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avramova, V.; AbdElgawad, H.; Zhang, Z.; Fotschki, B.; Casadevall, R.; Vergauwen, L.; Knapen, D.; Taleisnik, E.; Guisez, Y.; Asard, H.; et al. Drought Induces Distinct Growth Response, Protection, and Recovery Mechanisms in the Maize Leaf Growth Zone. Plant Physiol. 2015, 169, 1382–1396. [Google Scholar] [CrossRef]
- Carmo-Silva, A.E.; Soares, A.S.; Marques da Silva, J.; Bernardes da Silva, A.; Keys, A.J.; Arrabaça, M.C. Photosynthetic responses of three C4 grasses of different metabolic subtypes to water deficit. Funct. Plant Biol. 2007, 34, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Gu, W.; Meng, Y.; Li, J.; Li, L.; Wang, Y.; Qu, D.; Wei, S. Exogenous DCPTA Ameliorates Simulated Drought Conditions by Improving the Growth and Photosynthetic Capacity of Maize Seedlings. Sci. Rep. 2017, 7, 12684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doubnerová, V.; Ryšlavá, H. What can enzymes of C4 photosynthesis do for C3 plants under stress? Plant Sci. Int. J. Exp. Plant Biol. 2011, 180, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Scheibe, R. Malate valves to balance cellular energy supply. Physiol. Plant. 2004, 120, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Saccardy, K.; Cornic, G.; Brulfert, J.; Reyss, A. Effect of drought stress on net CO2 uptake by Zea leaves. Planta 1996, 199, 589–595. [Google Scholar] [CrossRef]
- Buckley, T.N. How do stomata respond to water status? New Phytol. 2019, 224, 21–36. [Google Scholar] [CrossRef] [Green Version]
- Saradadevi, R.; Palta, J.A.; Siddique, K.H.M. ABA-Mediated Stomatal Response in Regulating Water Use during the Development of Terminal Drought in Wheat. Front. Plant Sci. 2017, 8, 1251. [Google Scholar] [CrossRef]
- Xie, X.; Wang, Y.; Williamson, L.; Holroyd, G.H.; Tagliavia, C.; Murchie, E.; Theobald, J.; Knight, M.R.; Davies, W.J.; Leyser, H.M.; et al. The identification of genes involved in the stomatal response to reduced atmospheric relative humidity. Curr. Biol. CB 2006, 16, 882–887. [Google Scholar] [CrossRef]
- Mittal, S.; Banduni, P.; Mallikarjuna, M.G.; Rao, A.R.; Jain, P.A.; Dash, P.K.; Thirunavukkarasu, N. Structural, Functional, and Evolutionary Characterization of Major Drought Transcription Factors Families in Maize. Front. Plant 2018, 6, 177. [Google Scholar] [CrossRef] [Green Version]
- Shikha, M.; Kanika, A.; Rao, A.R.; Mallikarjuna, M.G.; Gupta, H.S.; Nepolean, T. Genomic Selection for Drought Tolerance Using Genome-Wide SNPs in Maize. Front. Plant Sci. 2017, 8, 550. [Google Scholar] [CrossRef]
- Lawlor, D.W.; Cornic, G. Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ. 2002, 25, 275–294. [Google Scholar] [CrossRef] [Green Version]
- Perez-Martin, A.; Flexas, J.; Ribas-Carbó, M.; Bota, J.; Tomás, M.; Infante, J.M.; Diaz-Espejo, A. Interactive effects of soil water deficit and air vapour pressure deficit on mesophyll conductance to CO2 in Vitis vinifera and Olea europaea. J. Exp. Bot. 2009, 60, 2391–2405. [Google Scholar] [CrossRef] [PubMed]
- Aasamaa, K.; Sõber, A. Stomatal sensitivities to changes in leaf water potential, air humidity, CO2 concentration and light intensity, and the effect of abscisic acid on the sensitivities in six temperate deciduous tree species. Environ. Exp. Bot. 2011, 71, 72–78. [Google Scholar] [CrossRef]
- Shekoofa, A.; Sinclair, T.R.; Messina, C.D.; Cooper, M.J.C.S. Variation among maize hybrids in response to high vapor pressure deficit at high temperatures. Crop Sci. 2016, 56, 392–396. [Google Scholar] [CrossRef]
- Iglesias-Acosta, M.; Martínez-Ballesta, M.C.; Teruel, J.A.; Carvajal, M. The response of broccoli plants to high temperature and possible role of root aquaporins. Environ. Exp. Bot. 2010, 68, 83–90. [Google Scholar] [CrossRef]
- Ben-Asher, J.; Garcia, A.; Flitcroft, I.; Hoogenboom, G. Effect of atmospheric water vapor on photosynthesis, transpiration and canopy conductance: A case study in corn. Plant Soil Environ. 2013, 59, 549–555. [Google Scholar] [CrossRef] [Green Version]
- Bunce, J.A. Effects of water vapor pressure difference on leaf gas exchange in potato and sorghum at ambient and elevated carbon dioxide under field conditions. Field Crop Res. 2003, 82, 37–47. [Google Scholar] [CrossRef]
- Salah, H.; Tardieu, F. Control of Leaf Expansion Rate of Droughted Maize Plants under Fluctuating Evaporative Demand (A Superposition of Hydraulic and Chemical Messages?). Plant Physiol. 1997, 114, 893–900. [Google Scholar] [CrossRef] [Green Version]
- Fu, Z.; Ciais, P.; Prentice, I.C.; Gentine, P.; Makowski, D.; Bastos, A.; Luo, X.; Green, J.K.; Stoy, P.C.; Yang, H.; et al. Atmospheric dryness reduces photosynthesis along a large range of soil water deficits. Nat. Commun. 2022, 13, 989. [Google Scholar] [CrossRef]
- Zhang, P.; Yang, X.; Manevski, K.; Li, S.; Wei, Z.; Andersen, M.N.; Liu, F. Physiological and Growth Responses of Potato (Solanum Tuberosum L.) to Air Temperature and Relative Humidity under Soil Water Deficits. Plants 2022, 11, 1126. [Google Scholar] [CrossRef]
- Flore, J.; Moon, J.; Lakso, A. The effect of water stress and vapor pressure gradient on stomatal conductance, water use efficiency, and photosynthesis of fruit crops. In Proceedings of the International Symposium on Water Relations in Fruit Crops 171, Bologna-Rimini, Italy, 2–6 September 1984; pp. 207–218. [Google Scholar]
- Ward, D.A. The temperature acclimation of photosynthetic responses to CO2 in Zea mays and its relationship to the activities of photosynthetic enzymes and the CO2-concentrating mechanism of C4 photosynthesis. Plant Cell Environ. 1987, 10, 407–441. [Google Scholar] [CrossRef]
- Doubnerová Hýsková, V.; Miedzińska, L.; Dobrá, J.; Vankova, R.; Ryšlavá, H. Phosphoenolpyruvate carboxylase, NADP-malic enzyme, and pyruvate, phosphate dikinase are involved in the acclimation of Nicotiana tabacum L. to drought stress. J. Plant Physiol. 2014, 171, 19–25. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, B.; Park, J.; Plaxton, W.C. The remarkable diversity of plant PEPC (phosphoenolpyruvate carboxylase): Recent insights into the physiological functions and post-translational controls of non-photosynthetic PEPCs. Biochem. J. 2011, 436, 15–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taniguchi, M.; Miyake, H. Redox-shuttling between chloroplast and cytosol: Integration of intra-chloroplast and extra-chloroplast metabolism. Curr. Opin. Plant Biol. 2012, 15, 252–260. [Google Scholar] [CrossRef]
- Tatar, Ö.; Brück, H.; Asch, F. Atmospheric and soil water deficit induced changes in chemical and hydraulic signals in wheat (Triticum aestivum L.). J. Agron. Crop Sci. 2022, early view. [Google Scholar] [CrossRef]
- Daszkowska-Golec, A.; Szarejko, I. Open or close the gate—Stomata action under the control of phytohormones in drought stress conditions. Front. Plant Sci. 2013, 4, 138. [Google Scholar] [CrossRef] [Green Version]
- Leng, P.; Zhao, J. Transcription factors as molecular switches to regulate drought adaptation in maize. Theor. Appl. Genet. 2020, 133, 1455–1465. [Google Scholar] [CrossRef]
- Sakuma, Y.; Maruyama, K.; Qin, F.; Osakabe, Y.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Dual function of an Arabidopsis transcription factor DREB2A in water-stress-responsive and heat-stress-responsive gene expression. Proc. Natl. Acad. Sci. USA 2006, 103, 18822–18827. [Google Scholar] [CrossRef] [Green Version]
- Bhatnagar-Mathur, P.; Devi, M.J.; Reddy, D.S.; Lavanya, M.; Vadez, V.; Serraj, R.; Yamaguchi-Shinozaki, K.; Sharma, K.K. Stress-inducible expression of At DREB1A in transgenic peanut (Arachis hypogaea L.) increases transpiration efficiency under water-limiting conditions. Plant Cell Rep. 2007, 26, 2071–2082. [Google Scholar] [CrossRef] [Green Version]
- Ryu, S.B.; Li, P.H.; Brenner, M.L. An enzyme-immunoassay of abscisic acid in potato (Solanum commersonii) cultured cells. Plant Cell Rep. 1992, 12, 34–36. [Google Scholar] [CrossRef]
- Ryu, S.B.; Costa, A.; Xin, Z.; Li, P.H. Induction of Cold Hardiness by Salt Stress Involves Synthesis of Cold- and Abscisic Acid-Responsive Proteins in Potato (Solanum commersonii Dun). Plant Cell Physiol. 1995, 36, 1245–1251. [Google Scholar] [CrossRef]
- Maroco, J.P.; Edwards, G.E.; Ku, M.S.B. Photosynthetic acclimation of maize to growth under elevated levels of carbon dioxide. Planta 1999, 210, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-H.; Gitz, D.C.; Sicher, R.C.; Baker, J.T.; Timlin, D.J.; Reddy, V.R. Temperature dependence of growth, development, and photosynthesis in maize under elevated CO2. Environ. Exp. Bot. 2007, 61, 224–236. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]




| 45% Soil Water Content | |||
|---|---|---|---|
| % of Decrease | Low VPD | Medium VPD | HighVPD |
| Leaf Area | 30.62 | 30.40 | 43.97 |
| Leaf Wt. | 39.69 | 44.86 | 57.93 |
| Leaf Number | 20.00 | 23.33 | 11.12 |
| Stomatal Conductance | 15.63 | 31.61 | 14.53 |
| Photosynthesis | 9.07 | 12.14 | 14.50 |
| 35% Soil Water Content | |||
| Leaf Area | 37.07 | 54.40 | 66.38 |
| Leaf Wt. | 42.96 | 61.05 | 75.86 |
| Leaf Number | 28.01 | 20.03 | 11.12 |
| Stomatal Conductance | 21.41 | 33.78 | 23.84 |
| Photosynthesis | 15.38 | 19.02 | 26.16 |
| 45% Soil Water Content | |||
| % of Increase | Low VPD | Medium VPD | High VPD |
| Water Use Efficiency | 41.35 | 32.87 | 4.04 |
| ABA | 47.70 | 39.31 | 29.83 |
| PEPC | 0.00 | 21.05 | 52.00 |
| MDH | 15.38 | 18.52 | 31.82 |
| NADP-ME | 2.00 | 28.57 | 42.86 |
| PPDK | 38.89 | 47.80 | 51.65 |
| 35% Soil Water Content | |||
| Water Use Efficiency | 75.00 | 45.74 | 60.00 |
| ABA | 90.00 | 88.34 | 83.33 |
| PEPC | 14.55 | 39.47 | 140.00 |
| MDH | 92.31 | 48.15 | 72.73 |
| NADP-ME | 30.00 | 65.71 | 114.29 |
| PPDK | 108.33 | 105.77 | 133.36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devi, M.J.; Reddy, V.R.; Timlin, D. Drought-Induced Responses in Maize under Different Vapor Pressure Deficit Conditions. Plants 2022, 11, 2771. https://doi.org/10.3390/plants11202771
Devi MJ, Reddy VR, Timlin D. Drought-Induced Responses in Maize under Different Vapor Pressure Deficit Conditions. Plants. 2022; 11(20):2771. https://doi.org/10.3390/plants11202771
Chicago/Turabian StyleDevi, Mura Jyostna, Vangimalla R. Reddy, and Dennis Timlin. 2022. "Drought-Induced Responses in Maize under Different Vapor Pressure Deficit Conditions" Plants 11, no. 20: 2771. https://doi.org/10.3390/plants11202771





