Metabolic Profiling and Antioxidant Analysis for the Juvenile Red Fading Leaves of Sweetpotato
Abstract
:1. Introduction
2. Results
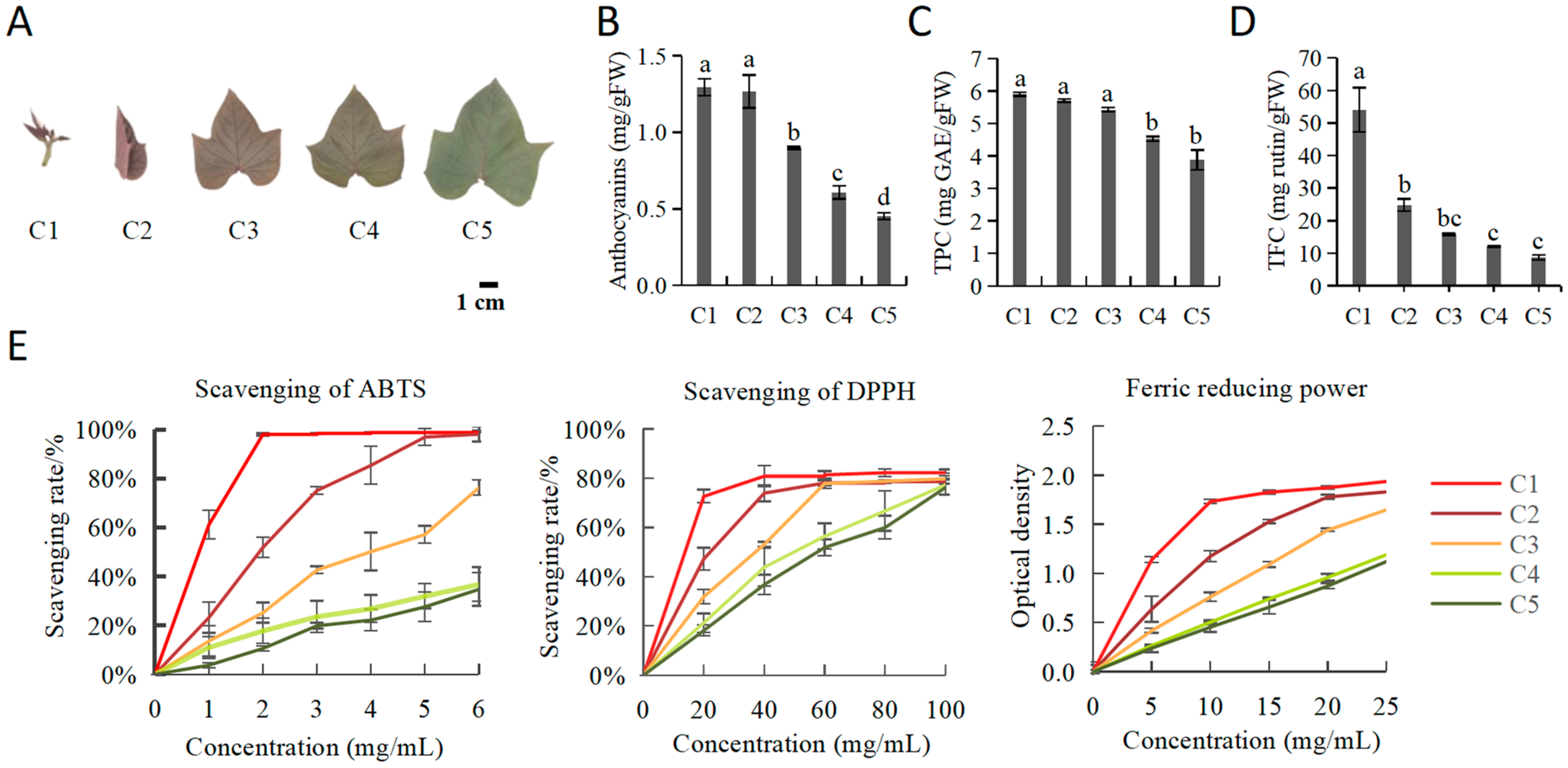
2.1. The Antioxidant Capacities for Juvenile Red Fading Leaves of “Chuanshan Zi”
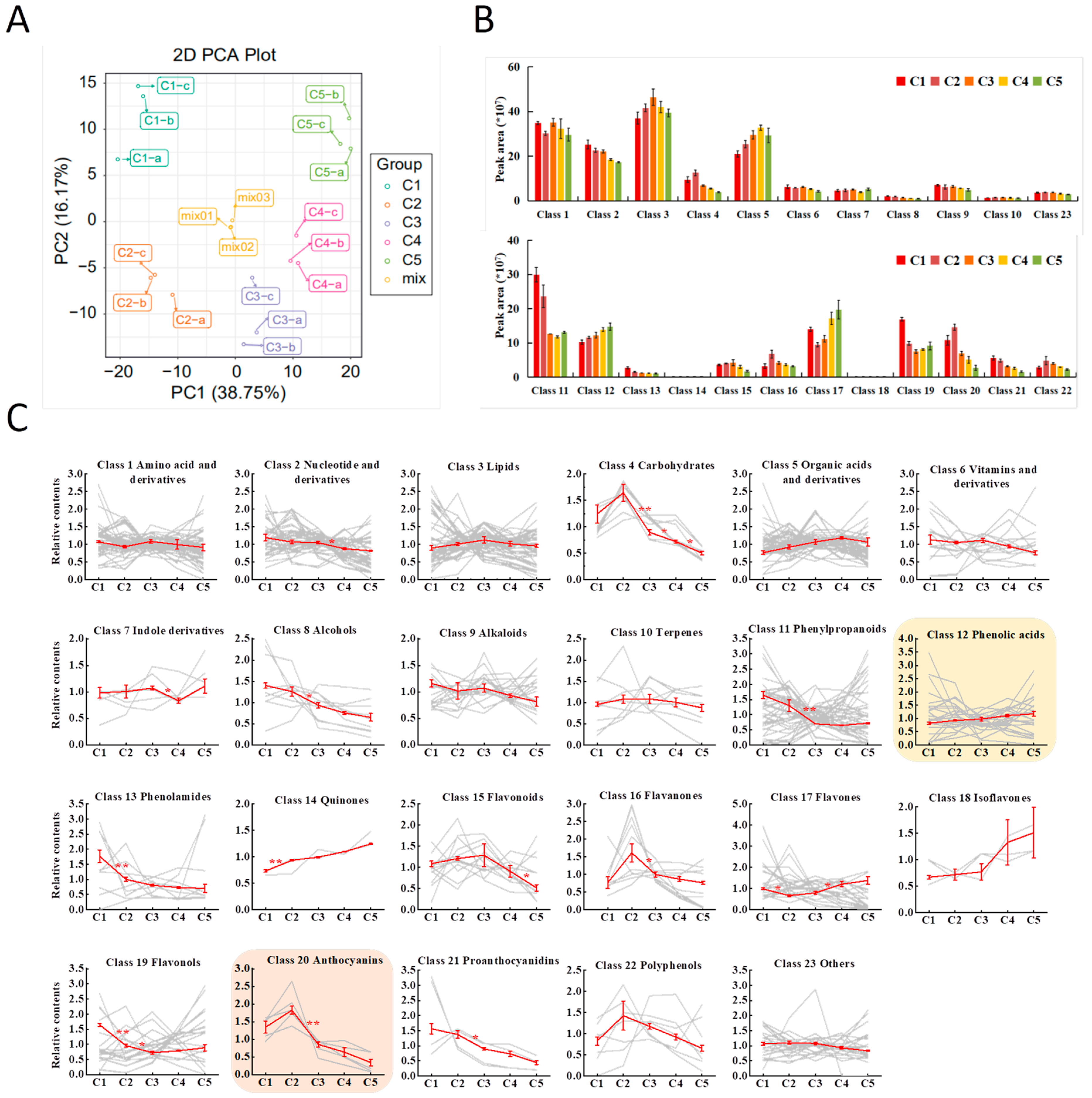
2.2. Metabolic Profiling for Juvenile Red Fading Leaves of “Chuanshan Zi”
2.3. Accumulation Trends for the 23 Metabolite Classes
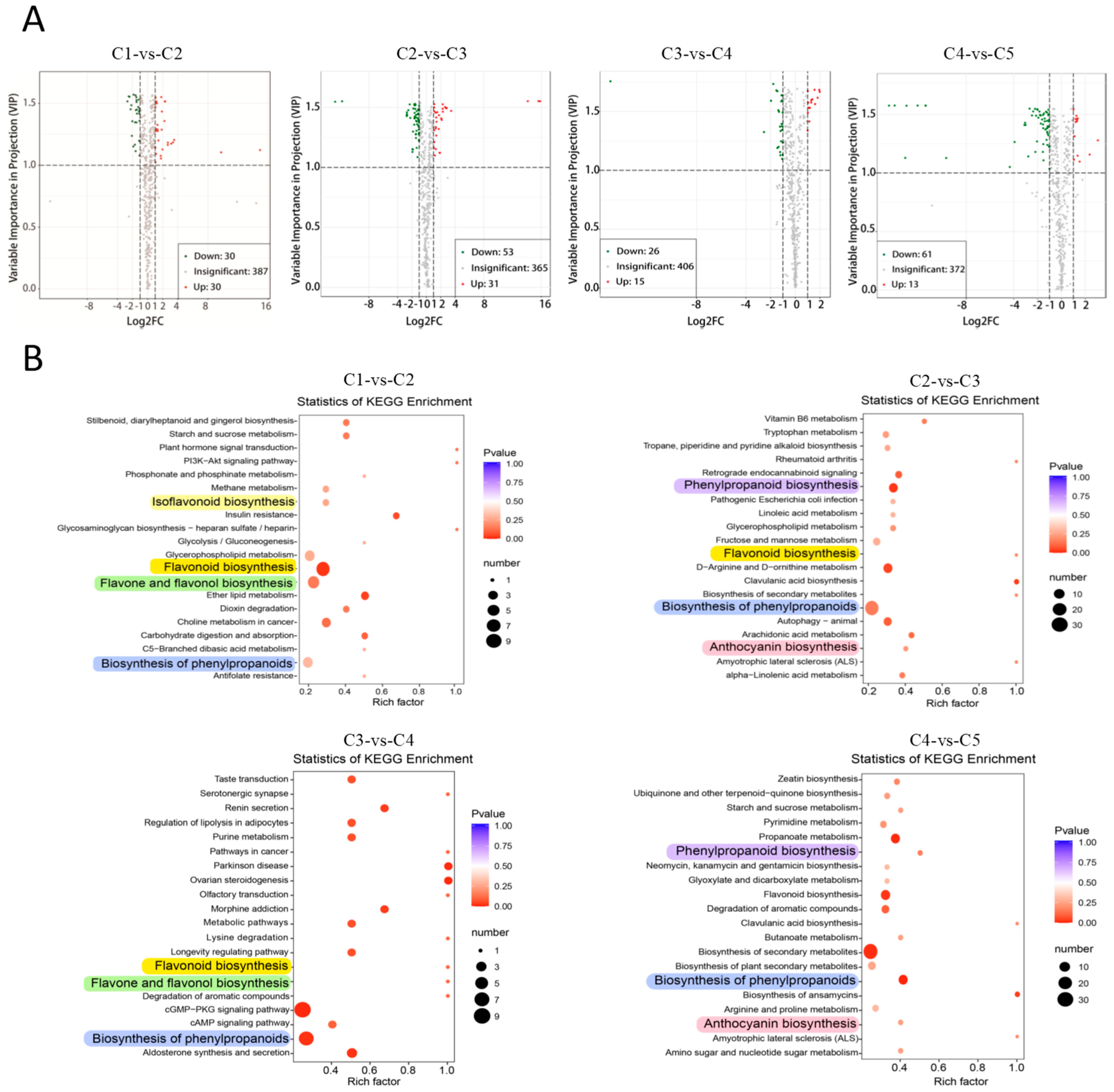
2.4. Differential Metabolites between Adjacent Leaf Stages
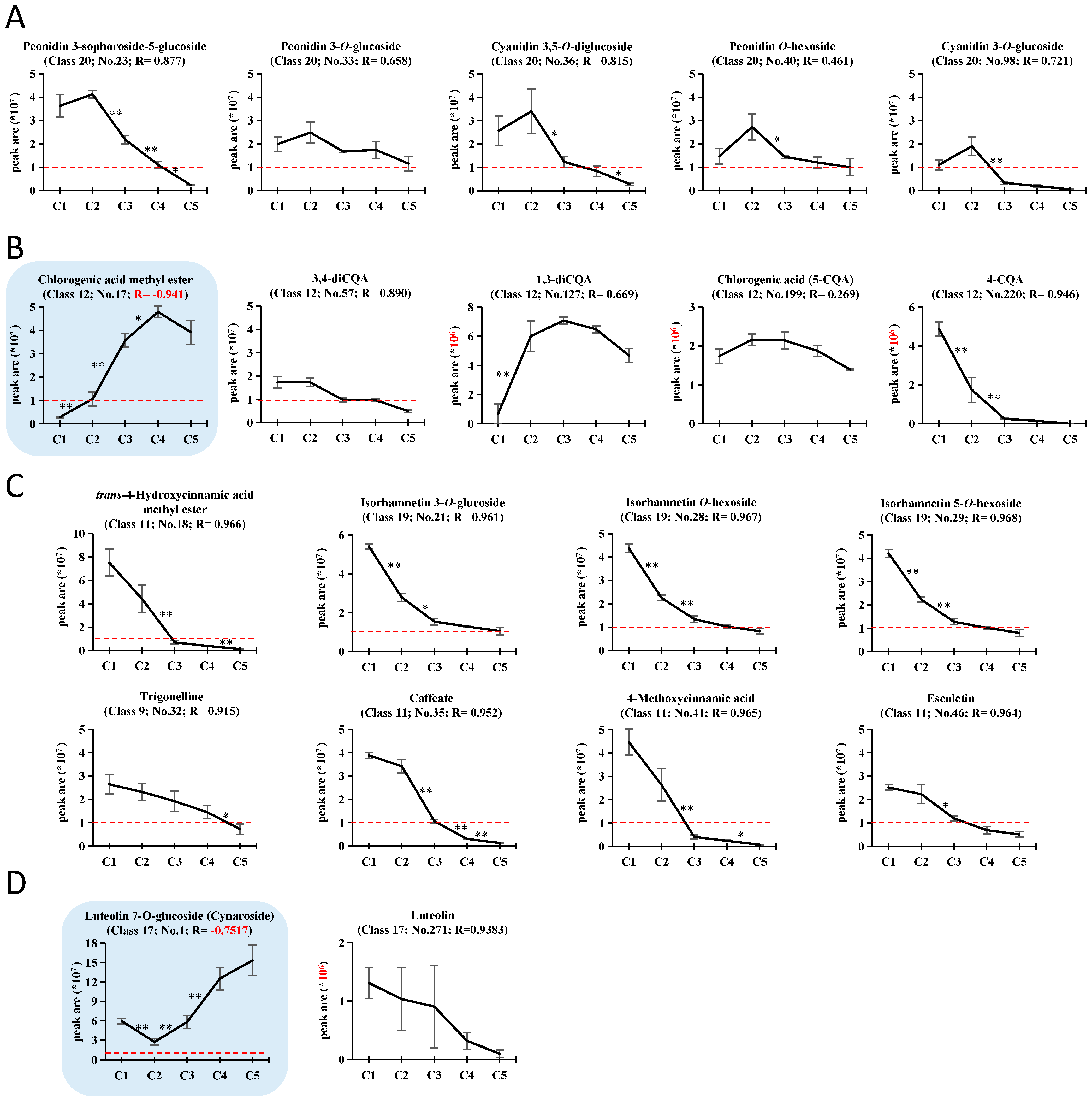
2.5. Analysis of the Top 50 Abundantly Accumulated Compounds
2.6. Correlation between Antioxidant Capacities and Metabolites
3. Discussion
3.1. The Metabolic Profile for Leaves of Sweetpotato “Chuanshan Zi”
3.2. Key Compounds Responsible for Declining Antioxidant Capacities in Leaves of “Chuanshan Zi”
3.3. Anthocyanins in Juvenile Leaves May Have Multiple Functions Relating to Both the Red Color and Their Antioxidant Capacities
4. Materials and Methods
4.1. Plant Materials
4.2. Measurement of Anthocyanins, TPC, TFC, and Antioxidant Activities
4.3. Sample Preparation for Metabolomic Analysis
4.4. LC-ESI-MS/MS Analysis
4.5. Qualitative and Quantitative Analysis of Metabolites
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Luo, J.; Duan, J.; Huo, D.; Shi, Q.; Niu, L.; Zhang, Y. Transcriptomic analysis reveals transcription factors related to leaf anthocyanin biosynthesis in Paeonia qiui. Molecules 2017, 22, 2186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, J.; Wu, D.; Shi, J.; Balfour, K.; Wang, H.; Zhu, G.; Liu, Y.; Wang, J.; Zhu, Z. Multiple MYB activators and repressors collaboratively regulate the juvenile red fading in leaves of sweetpotato. Front. Plant Sci. 2020, 11, 941. [Google Scholar] [CrossRef] [PubMed]
- Chao, P.Y.; Lin, S.Y.; Lin, K.H.; Liu, Y.F.; Hsu, J.I.; Yang, C.M.; Lai, J.Y. Antioxidant activity in extracts of 27 indigenous Taiwanese vegetables. Nutrients 2014, 6, 2115–2130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gould, K.S. Nature’s swiss army knife: The diverse protective roles of anthocyanins in leaves. J. Biomed. Biotechnol. 2004, 5, 314–320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gong, W.C.; Liu, Y.H.; Wang, C.M.; Chen, Y.Q.; Martin, K.; Meng, L.Z. Why are there so many plant species that transiently flush young leaves red in the tropics? Front. Plant Sci. 2020, 11, 83. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, H.C.; Chen, C.C.; Lin, K.H.; Chao, P.Y.; Lin, H.H.; Huang, M.Y. Bioactive compounds, antioxidants, and health benefits of sweet potato leaves. Molecules 2021, 26, 1820. [Google Scholar] [CrossRef]
- Islam, M.S.; Yoshimoto, M.; Yahara, S.; Okuno, S.; Ishiguro, K.; Yamakawa, O. Identification and characterization of foliar polyphenolic composition in sweetpotato (Ipomoea batatas L.) genotypes. J. Agric. Food Chem. 2002, 50, 3718–3722. [Google Scholar] [CrossRef]
- Islam, S. Sweetpotato (Ipomea batatas L.) leaf: Its potential effect on human health and nutrition. J. Food Sci. 2006, 71, R13–R18. [Google Scholar] [CrossRef]
- Sun, H.; Mu, T.; Xi, L.; Zhang, M.; Chen, J. Sweet potato (Ipomoea batatas L.) leaves as nutritional and functional foods. Food Chem. 2014, 156, 380–389. [Google Scholar] [CrossRef]
- Zhang, L.; Tu, Z.C.; Yuan, T.; Wang, H.; Xie, X.; Fu, Z.F. Antioxidants and alpha-glucosidase inhibitors from Ipomoea batatas leaves identified by bioassay-guided approach and structure-activity relationships. Food Chem. 2016, 208, 61–67. [Google Scholar] [CrossRef]
- Krochmal-Marczak, B.; Cebulak, T.; Kapusta, I.; Oszmianski, J.; Kaszuba, J.; Zurek, N. The content of phenolic acids and flavonols in the leaves of nine varieties of sweet potatoes (Ipomoea batatas L.) depending on their development, grown in Central Europe. Molecules 2020, 25, 3473. [Google Scholar] [CrossRef] [PubMed]
- Thu, N.N.; Sakurai, C.; Uto, H.; Van Chuyen, N.; Lien, D.T.; Yamamoto, S.; Ohmori, R.; Kondo, K. The polyphenol content and antioxidant activities of the main edible vegetables in northern Vietnam. J. Nutr. Sci. Vitaminol. 2004, 50, 203–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiu, C.H.; Lin, K.H.; Lin, H.H.; Chu, W.X.; Lai, Y.C.; Chao, P.Y. Analysis of chlorogenic acid in sweet potato leaf extracts. Plants 2022, 11, 2063. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Gong, L.; Guo, Z.; Wang, W.; Zhang, H.; Liu, X.; Yu, S.; Xiong, L.; Luo, J. A novel integrated method for large-scale detection, identification, and quantification of widely targeted metabolites: Application in the study of rice metabolomics. Mol. Plant 2013, 6, 1769–1780. [Google Scholar] [CrossRef] [Green Version]
- Clifford, M.N.; Jaganath, I.B.; Ludwig, I.A.; Crozier, A. Chlorogenic acids and the acyl-quinic acids: Discovery, biosynthesis, bioavailability and bioactivity. Nat. Prod. Rep. 2017, 34, 1391–1421. [Google Scholar] [CrossRef] [Green Version]
- Lin, L.C.; Pai, Y.F.; Tsai, T.H. Isolation of luteolin and luteolin-7-O-glucoside from Dendranthema morifolium Ramat Tzvel and their pharmacokinetics in rats. J. Agric. Food Chem. 2015, 63, 7700–7706. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, F.; Lian, Y.; Xiao, H.; Zheng, J. Biosynthesis of citrus flavonoids and their health effects. Crit. Rev. Food Sci. 2020, 60, 566–583. [Google Scholar] [CrossRef]
- Hyun, S.K.; Jung, Y.J.; Chung, H.Y.; Jung, H.A.; Choi, J.S. Isorhamnetin glycosides with free radical and ONOO-scavenging activities from the stamens of Nelumbo nucifera. Arch. Pharm. Res. 2006, 29, 287–292. [Google Scholar] [CrossRef]
- Cho, J.Y.; Moon, J.H.; Seong, K.Y.; Park, K.H. Antimicrobial activity of 4-hydroxybenzoic acid and trans 4-hydroxycinnamic acid isolated and identified from rice hull. Biosci. Biotechnol. Biochem. 1998, 62, 2273–2276. [Google Scholar] [CrossRef] [Green Version]
- Sova, M. Antioxidant and antimicrobial activities of cinnamic acid derivatives. Mini-Rev. Med. Chem. 2012, 12, 749–767. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Hossain, H.; Hossain, M.; Reza, H.M.; Rahman, M.M.; Ullah, M.O. Hydroxycinnamic acid derivatives: A potential class of natural compounds for the management of lipid metabolism and obesity. Nutr. Metab. 2016, 13, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Hsia, C.W.; Lin, K.C.; Lee, T.Y.; Hsia, C.H.; Chou, D.S.; Jayakumar, T.; Velusamy, M.; Chang, C.C.; Sheu, J.R. Esculetin, a coumarin derivative, prevents thrombosis: Inhibitory signaling on PLCgamma2-PKC-AKT activation in human platelets. Int. J Mol. Sci. 2019, 20, 2731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, B.C.; Lee, S.Y.; Lee, H.J.; Sim, G.S.; Kim, J.H.; Kim, J.H.; Cho, Y.H.; Lee, D.H.; Pyo, H.B.; Choe, T.B.; et al. Anti-oxidative and photo-protective effects of coumarins isolated from Fraxinus chinensis. Arch. Pharm. Res. 2007, 30, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Berglund, T.; Kalbin, G.; Strid, A.; Rydstrom, J.; Ohlsson, A.B. UV-B- and oxidative stress-induced increase in nicotinamide and trigonelline and inhibition of defensive metabolism induction by poly(ADP-ribose)polymerase inhibitor in plant tissue. FEBS Lett. 1996, 380, 188–193. [Google Scholar] [CrossRef] [Green Version]
- Ashihara, H.; Watanabe, S. Accumulation and function of trigonelline in non-leguminous plants. Nat. Prod. Commun. 2014, 9, 795–798. [Google Scholar] [CrossRef] [Green Version]
- Mondolot, L.; La Fisca, P.; Buatois, B.; Talansier, E.; de Kochko, A.; Campa, C. Evolution in caffeoylquinic acid content and histolocalization during Coffea canephora leaf development. Ann. Bot. 2006, 98, 33–40. [Google Scholar] [CrossRef]
- Meyer, S.; Louis, J.; Moise, N.; Piolot, T.; Baudin, X.; Cerovic, Z.G. Developmental changes in spatial distribution of in vivo fluorescence and epidermal UV absorbance over Quercus petraea leaves. Ann. Bot. 2009, 104, 621–633. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.-Z.; Huang, S.-Q. Red young leaves have less mechanical defence than green young leaves. Oikos 2013, 122, 1035–1041. [Google Scholar] [CrossRef]
- Whitney, H.M.; Reed, A.; Rands, S.A.; Chittka, L.; Glover, B.J. Flower iridescence increases object detection in the insect visual system without compromising object identity. Curr. Biol. 2016, 26, 802–808. [Google Scholar] [CrossRef]
- Sun, Y.; Li, M.; Mitra, S.; Hafiz Muhammad, R.; Debnath, B.; Lu, X.; Jian, H.; Qiu, D. Comparative phytochemical profiles and antioxidant enzyme activity analyses of the southern highbush blueberry (Vaccinium corymbosum) at different developmental stages. Molecules 2018, 23, 2209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Chen, D.; Mai, Y.; Wen, B.; Wang, X. Concordance between antioxidant activities in vitro and chemical components of Radix Astragali (Huangqi). Nat. Prod. Res. 2012, 26, 1050–1053. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, G.; Long, C.; Xu, J.; Cen, J.; Yang, X. The antioxidant activity and genotoxicity of isogarcinol. Food Chem. 2018, 253, 5–12. [Google Scholar] [CrossRef] [PubMed]

| Category (Compound Number) | Class No. | Class | Compound Number | Correlation with Antioxidant Abilities | |
|---|---|---|---|---|---|
| Primary metabolites (233) | Class 1 | Amino acid and derivatives | 55 | 0.477 | |
| Class 2 | Nucleotide and derivatives | 41 | 0.955 ** | ||
| Class 3 | Lipids | 52 | −0.418 | ||
| Class 4 | Carbohydrates | 9 | 0.759 | ||
| Class 5 | Organic acids and derivatives | 47 | −0.938 * | ||
| Class 6 | Vitamins and derivatives | 14 | 0.769 | ||
| Class 7 | Indole derivatives | 6 | 0.003 | ||
| Class 8 | Alcohols | 9 | 0.971 ** | ||
| Secondary metabolites (193) | |||||
| Alkaloids (18) | Class 9 | Alkaloids | 18 | 0.877 | |
| Terpenes (8) | Class 10 | Terpenes | 8 | 0.205 | |
| Phenolics (167) | |||||
| Simple Phenols (82) | Class 11 | Phenylpropanoids | 46 | 0.944 * | |
| Class 12 | Phenolic acids | 25 | −0.966 ** | ||
| Class 13 | Phenolamides | 9 | 0.937 * | ||
| Class 14 | Quinones | 2 | −0.963 ** | ||
| Flavonoids (85) | Class 15 | Flavonoids | 12 | 0.578 | |
| Class 16 | Flavanones | 10 | 0.192 | ||
| Class 17 | Flavones | 23 | −0.578 | ||
| Class 18 | Isoflavones | 3 | −0.852 | ||
| Class 19 | Flavonols | 21 | 0.846 | ||
| Class 20 | Anthocyanins | 5 | 0.776 | ||
| Class 21 | Proanthocyanidins | 4 | 0.956 ** | ||
| Class 22 | Polyphenols | 7 | 0.251 | ||
| Others (23) | Class 23 | Others | 23 | 0.734 | |
| Rank | Compounds | Class | Average Peak Area (×107) | Rank | Compounds | Class | Average Peak Area (×107) |
|---|---|---|---|---|---|---|---|
| No.1 # | Cynaroside (Luteolin 7-O-glucoside) | Class 17 | 8.47 ± 2.34 | No.26 | MAG (18:3) isomer2 | Class 3 | 2.07 ± 0.14 |
| No.2 | L(-)-Malic acid | Class 5 | 6.82 ± 0.54 | No.27 | 6,7-Dihydroxycoumarin 7-O-quinic acid | Class 11 | 2.03 ± 0.22 |
| No.3 | L-Leucine | Class 1 | 5.49 ± 0.15 | No.28 * | Isorhamnetin O-hexoside | Class 19 | 1.97 ± 0.65 |
| No.4 | MAG (18:3) isomer3 | Class 3 | 5.20 ± 0.35 | No.29 * | Isorhamnetin 5-O-hexoside | Class 19 | 1.91 ± 0.62 |
| No.5 | alpha-Aminocaproic acid | Class 1 | 5.09 ± 0.17 | No.30 | Procyanidin B3 | Class 21 | 1.83 ± 0.33 |
| No.6 # | Kynurenic acid | Class 5 | 4.38 ± 1.67 | No.31 | Glutamic acid | Class 1 | 1.82 ± 0.20 |
| No.7 | Citric acid | Class 5 | 3.71 ± 0.38 | No.32 #,* | Trigonelline | Class 9 | 1.81 ± 0.34 |
| No.8 | LysoPC 20:4 | Class 3 | 3.64 ± 0.40 | No.33 | Peonidin 3-O-glucoside | Class 20 | 1.81 ± 0.22 |
| No.9 | Niacinamide | Class 6 | 3.57 ± 0.32 | No.34 | 2-Isopropylmalate | Class 5 | 1.80 ± 0.15 |
| No.10 | 2-Aminoisobutyric acid | Class 1 | 3.55 ± 0.47 | No.35 #,* | Caffeate | Class 11 | 1.76 ± 0.79 |
| No.11 # | Galactinol | Class 4 | 3.37 ± 0.75 | No.36 # | Cyanidin 3,5-O-diglucoside | Class 20 | 1.67 ± 0.57 |
| No.12 | Indole | Class 7 | 3.30 ± 0.16 | No.37 # | LysoPC 20:1 | Class 3 | 1.67 ± 0.38 |
| No.13 | L-Phenylalanine | Class 1 | 3.21 ± 0.19 | No.38 # | D-( + )-Sucrose | Class 4 | 1.63 ± 0.33 |
| No.14 | MAG (18:3) isomer5 | Class 3 | 3.10 ± 0.36 | No.39 # | MAG (18:4) isomer1 | Class 3 | 1.62 ± 0.38 |
| No.15 | Guanosine | Class 2 | 3.02 ± 0.50 | No.40 | Peonidin 3-O-hexoside | Class 20 | 1.57 ± 0.30 |
| No.16 | Adenosine | Class 2 | 2.73 ± 0.31 | No.41 #,* | 4-Methoxycinnamic acid | Class 11 | 1.56 ± 0.86 |
| No.17 #,* | Chlorogenic acid methyl ester | Class 12 | 2.73 ± 0.87 | No.42 | Hesperidin (Hesperetin 7-rutinoside) | Class 16 | 1.51 ± 0.10 |
| No.18 #,* | trans-4-Hydroxycinnamic acid methyl ester | Class 11 | 2.63 ± 1.46 | No.43 | Citric acid monohydrate | Class 5 | 1.48 ± 0.15 |
| No.19 * | D-(-)-Valine | Class 1 | 2.55 ± 0.23 | No.44 | LysoPC 15:0 | Class 3 | 1.46 ± 0.18 |
| No.20 | 14,15-Dehydrocrepenynic acid | Class 3 | 2.53 ± 0.39 | No.45 # | 2-Methylsuccinic acid | Class 5 | 1.46 ± 0.27 |
| No.21 * | Isorhamnetin 3-O-glucoside | Class 19 | 2.42 ± 0.80 | No.46 * | Esculetin (6,7-Dihydroxycoumarin) | Class 11 | 1.42 ± 0.40 |
| No.22 | 6-MethylCoumarin | Class 11 | 2.34 ± 0.25 | No.47 | LysoPC 16:2 (2n isomer) | Class 3 | 1.42 ± 0.15 |
| No.23 # | Peonidin 3-O-sophoroside-5-O-glucoside | Class 20 | 2.26 ± 0.73 | No.48 # | LysoPC 18:3 | Class 3 | 1.42 ± 0.54 |
| No.24 | 2,5-Dihydroxy benzoic acid O-hexside | Class 12 | 2.25 ± 0.41 | No.49 # | LysoPC 20:1 (2n isomer) | Class 3 | 1.42 ± 0.32 |
| No.25 | Succinyladenosine | Class 2 | 2.17 ± 0.22 | No.50 | Hesperetin 7-O-neohesperidoside | Class 16 | 1.36 ± 0.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Wu, Q.; Deng, J.; Balfour, K.; Chen, Z.; Liu, Y.; Kumar, S.; Chen, Y.; Zhu, Z.; Zhu, G. Metabolic Profiling and Antioxidant Analysis for the Juvenile Red Fading Leaves of Sweetpotato. Plants 2022, 11, 3014. https://doi.org/10.3390/plants11223014
Shi J, Wu Q, Deng J, Balfour K, Chen Z, Liu Y, Kumar S, Chen Y, Zhu Z, Zhu G. Metabolic Profiling and Antioxidant Analysis for the Juvenile Red Fading Leaves of Sweetpotato. Plants. 2022; 11(22):3014. https://doi.org/10.3390/plants11223014
Chicago/Turabian StyleShi, Jie, Qiang Wu, Jiliang Deng, Kelly Balfour, Zhuo Chen, Yonghua Liu, Sunjeet Kumar, Yanli Chen, Zhixin Zhu, and Guopeng Zhu. 2022. "Metabolic Profiling and Antioxidant Analysis for the Juvenile Red Fading Leaves of Sweetpotato" Plants 11, no. 22: 3014. https://doi.org/10.3390/plants11223014






