Anti-Inflammatory Activities of Constituents from Cinnamomum insularimontanum Hayata Leaves and Their Mechanisms
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Composition Analysis and Its Anti-Inflammatory Activity of C. insularimontanum Leaves Essential Oil (CILE)
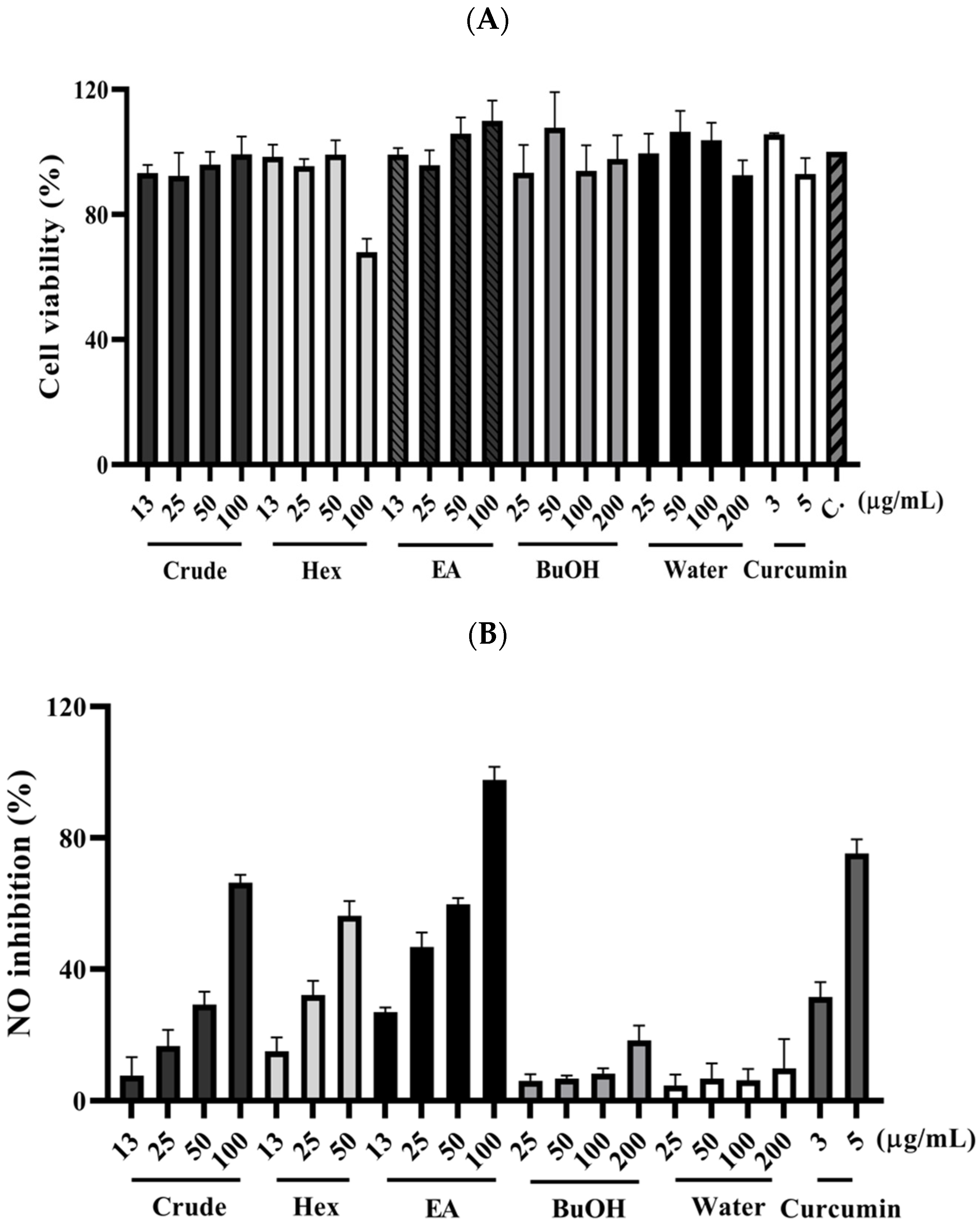
2.2. Anti-Inflammatory Activity of Ethanolic Extract and Its Bioactive Compounds Isolation
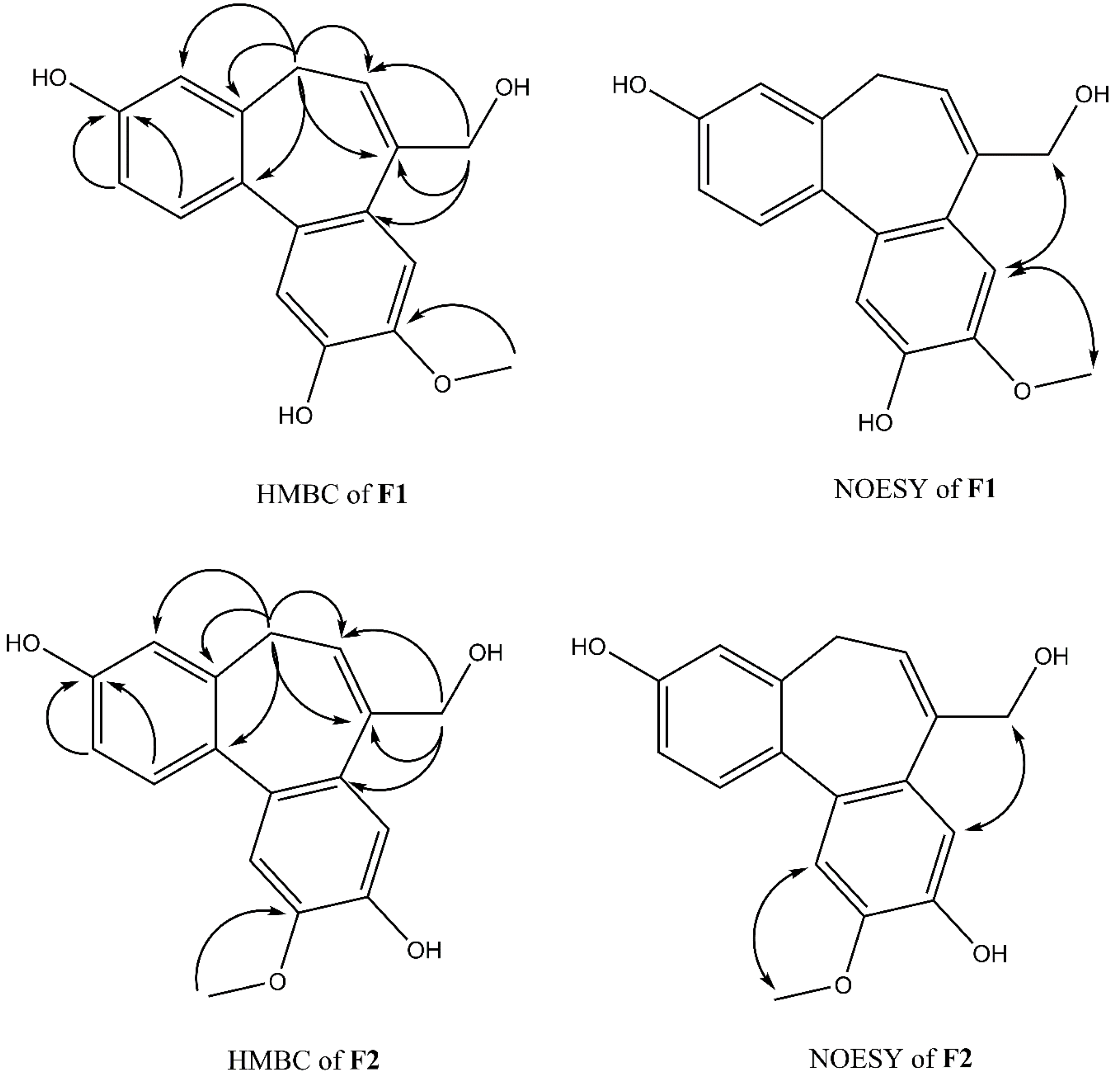
2.3. Anti-Inflammatory Activity of Isoburmanol (F1) and Burmanol (F2)
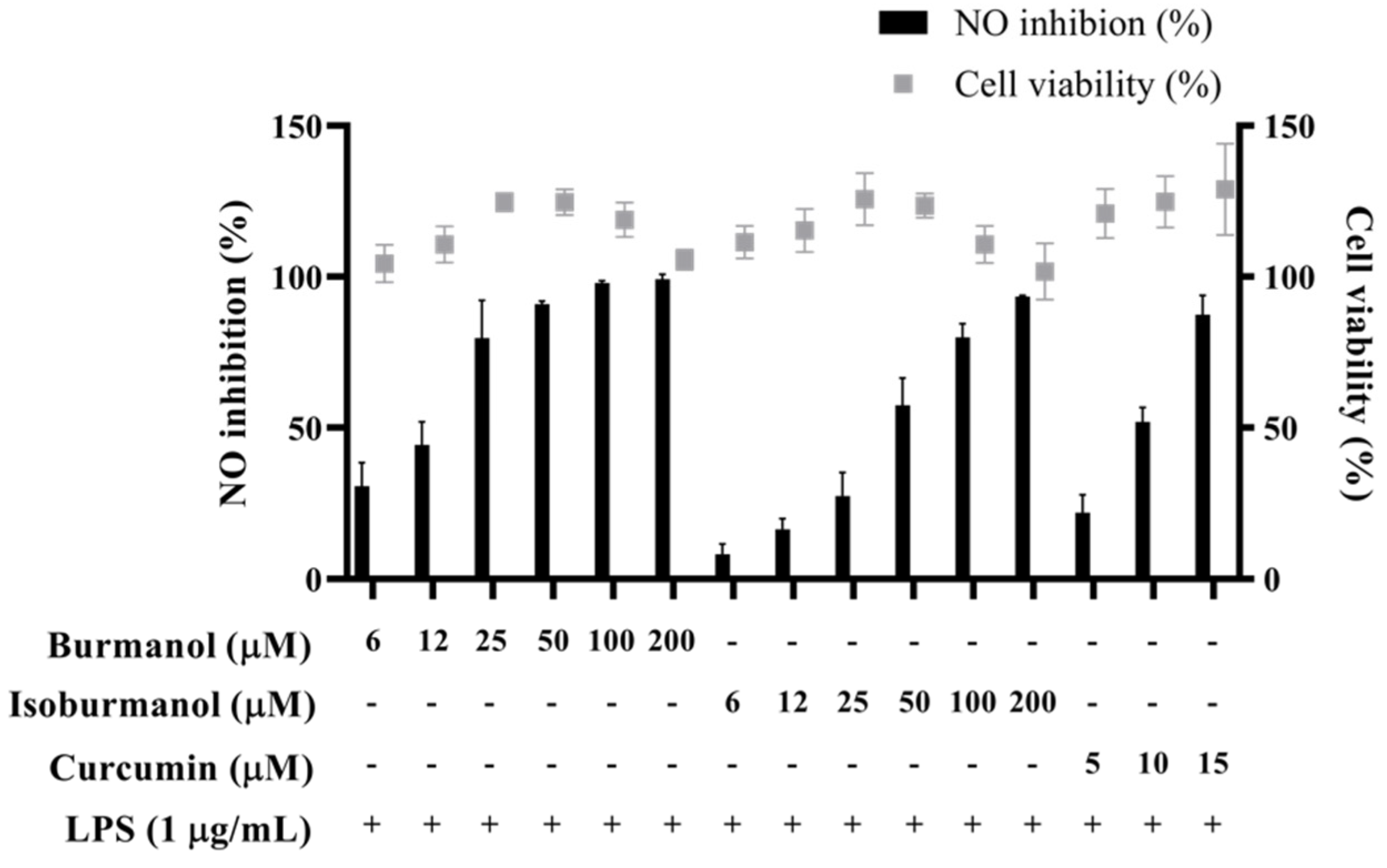
2.3.1. NO Production and PGE2 Synthesis Inhibitory Activity
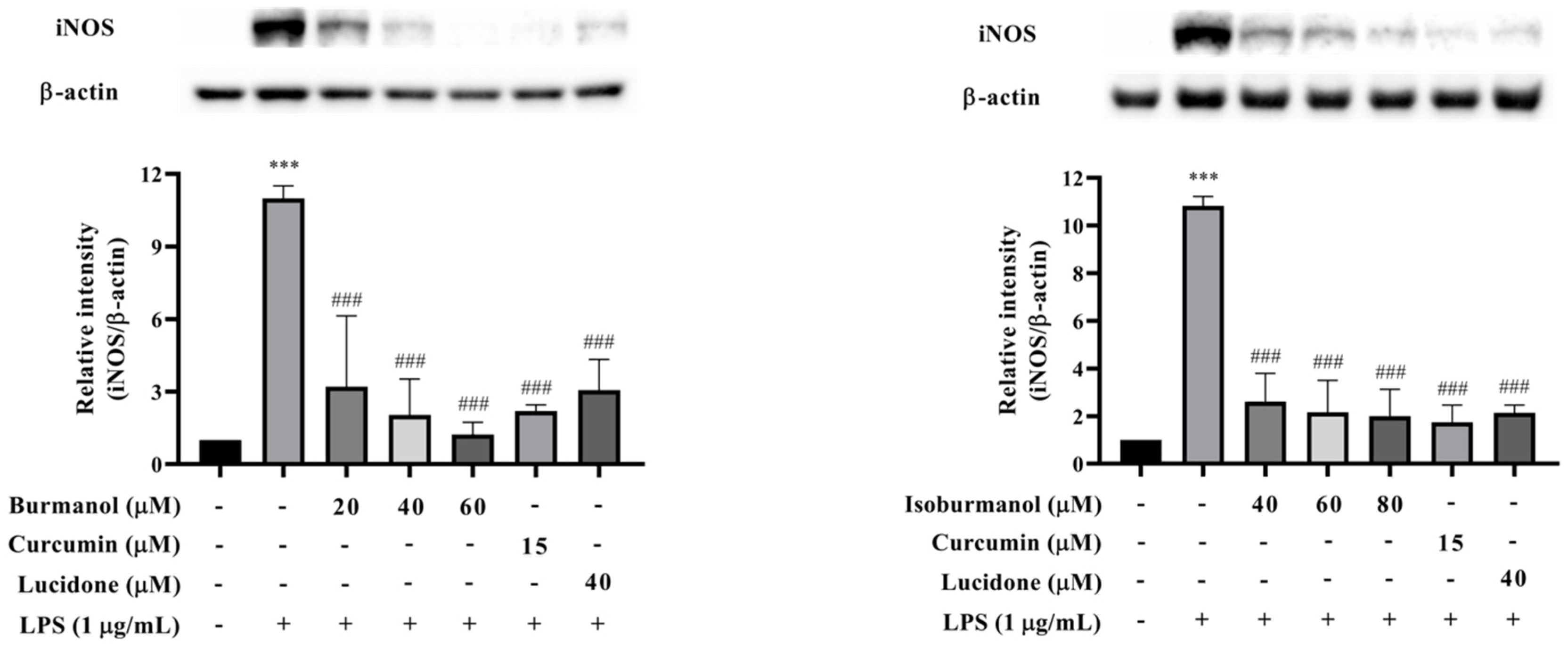
2.3.2. Effects of F1 and F2 on iNOS Expression in LPS-Induced Macrophages
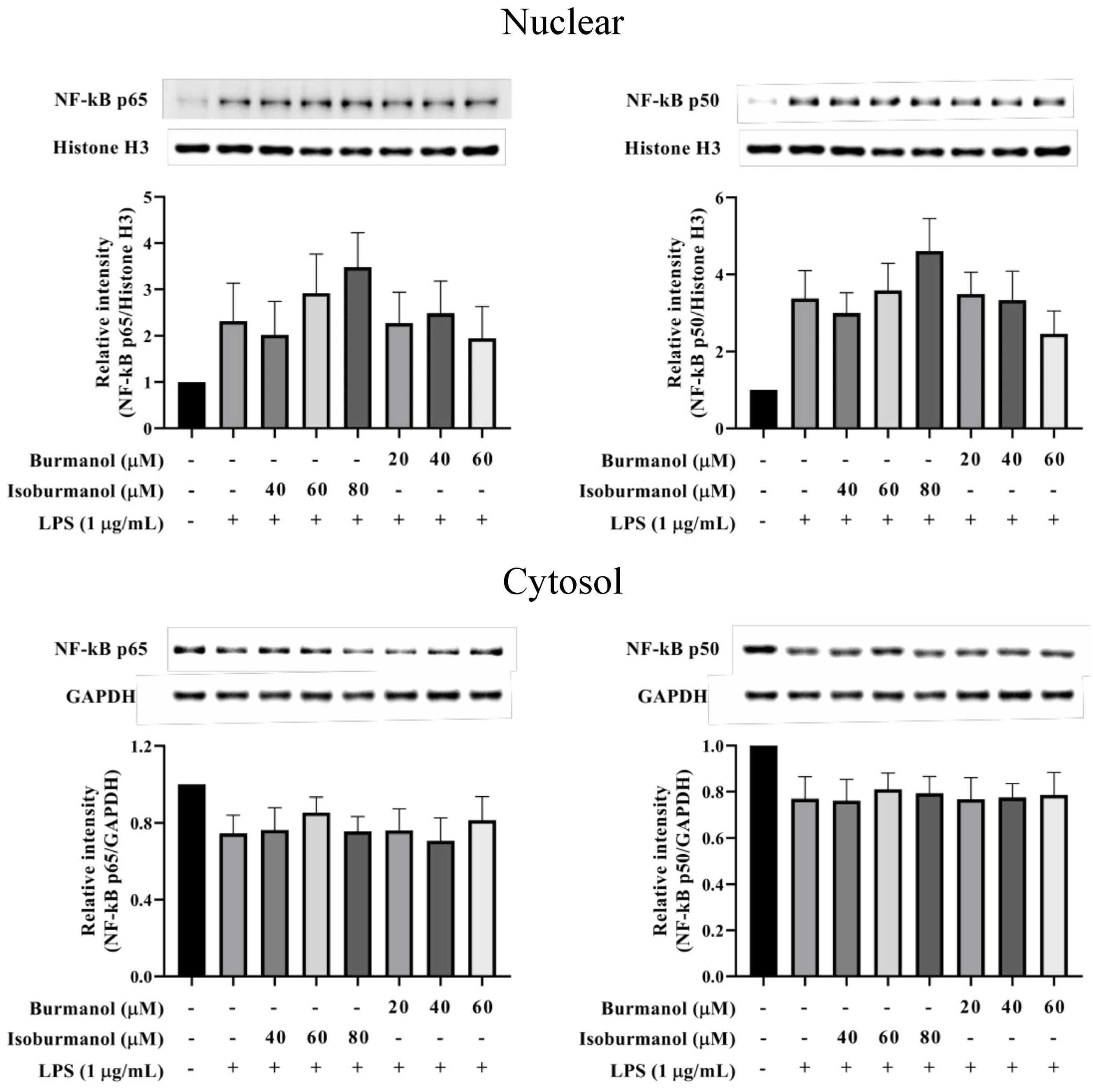
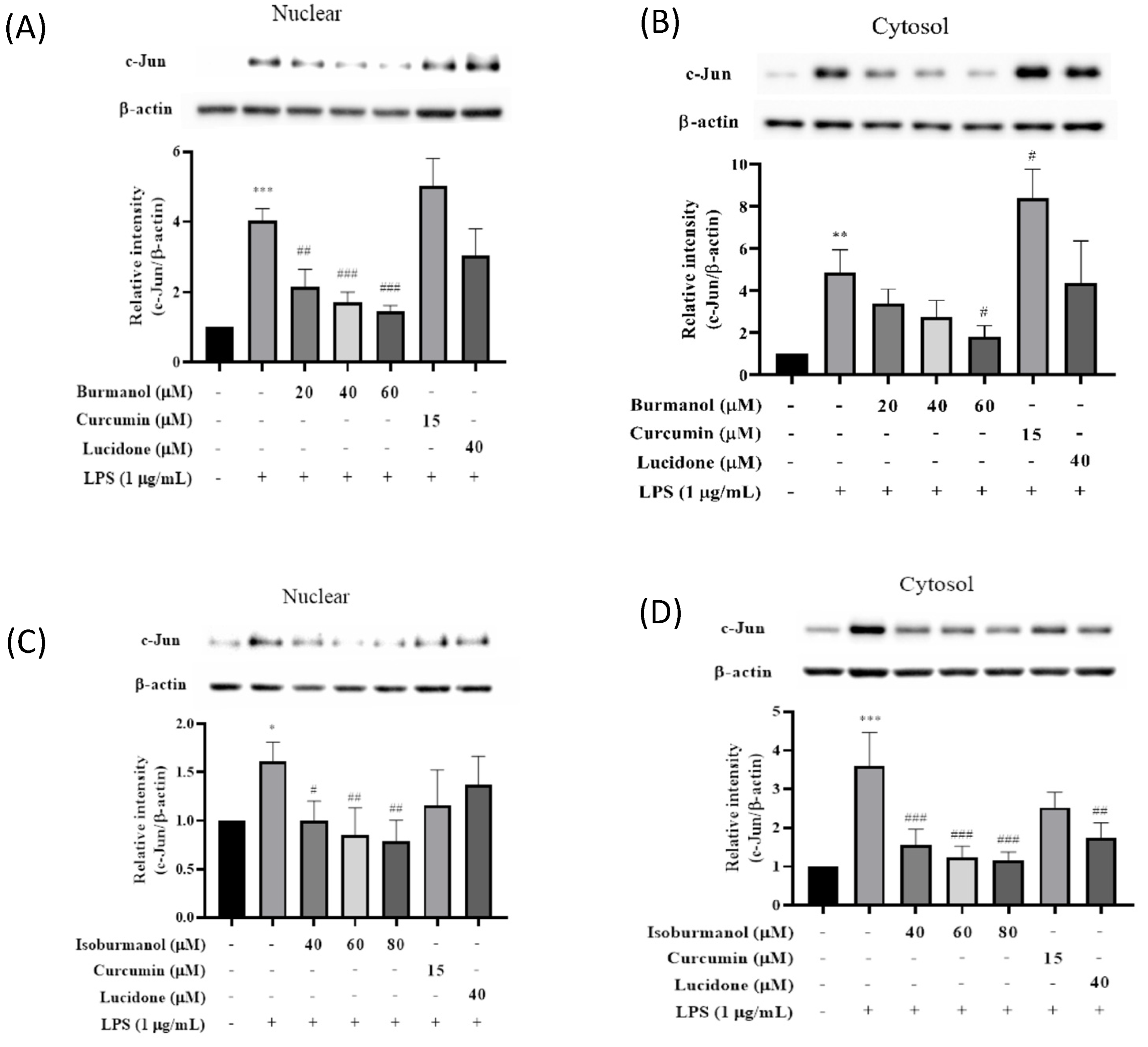
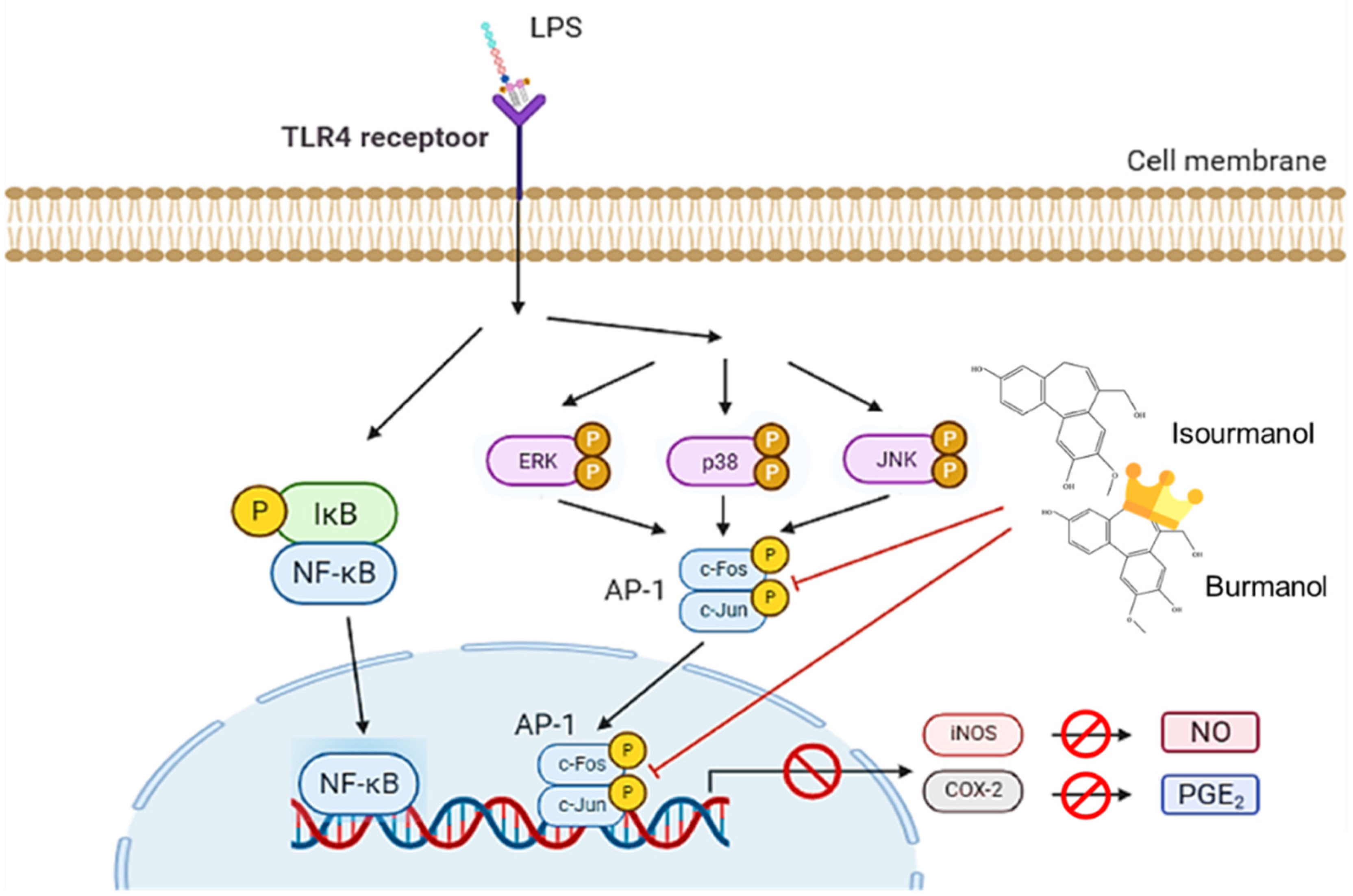
2.3.3. Effects of F1 and F2 on Upstream Transcription Factors NF-κB and AP-1 Expression in LPS-Induced Macrophages
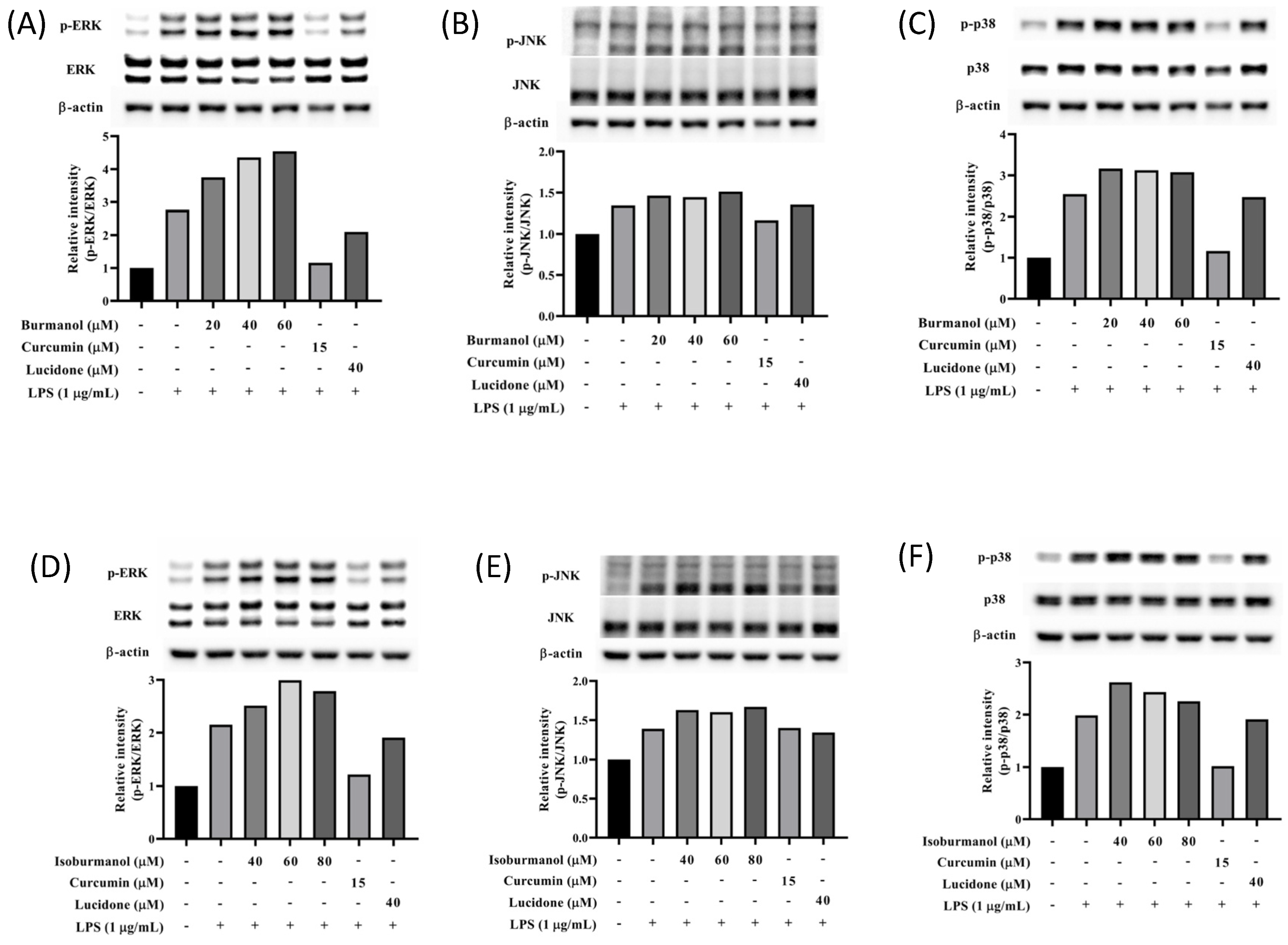
2.3.4. Effects of Isoburmanol and Burmanol on MAPK
2.4. Molecular Docking Simulation Analysis
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Preparation of C. insularimontanum Leaves Essential Oil (CILE) and GC/MS Analysis
3.4. Extraction and Isolation
3.5. Cell Culture and Cell Survival Assay
3.6. Anti-Inflammatory Assay
3.7. Protein Extraction and Immunoblotting
3.8. Molecular Docking
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhao, M.; Wu, L.; Yan, G.; Chen, Y.; Zhou, M.; Wu, Y.; Li, Y. Inflammation and tumor progression: Signaling pathways and targeted intervention. Signal Transduct. Target. Ther. 2021, 6, 263. [Google Scholar] [CrossRef]
- Aghasafari, P.; George, U.; Pidaparti, R. A review of inflammatory mechanism in airway diseases. Inflamm. Res. 2019, 68, 59–74. [Google Scholar] [CrossRef]
- Wawrzyniak-Gramacka, E.; Hertmanowska, N.; Tylutka, A.; Morawin, B.; Wacka, E.; Gutowicz, M.; Zembron-Lacny, A. The association of anti-inflammatory diet ingredients and lifestyle exercise with inflammaging. Nutrients 2021, 13, 3696. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.d.R.; Barreto Arantes, M.; Menezes de Faria Pereira, S.; Leandro da Cruz, L.; de Souza Passos, M.; Pereira de Moraes, L.; Vieira, I.J.C.; Barros de Oliveira, D. Plants as Sources of Anti-Inflammatory Agents. Molecules 2020, 25, 3726. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; The International Natural Product Sciences Taskforce; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Desborough, M.J.R.; Keeling, D.M. The aspirin story—From willow to wonder drug. Br. J. Haematol. 2017, 177, 674–683. [Google Scholar] [CrossRef] [Green Version]
- Mansukh, C.; Wani, M.; Horwitz, S.B. Nature as a Remarkable Chemist: A Personal Story of the Discovery and Development of Taxol®. Anticancer Drugs 2014, 25, 482–487. [Google Scholar]
- Lin, C.T.; Chen, C.J.; Lin, T.Y.; Tung, J.C.; Wang, S.Y. Anti-inflammation activity of fruit essential oil from Cinnamomum insularimontanum Hayata. Bioresour. Technol. 2008, 99, 8783–8787. [Google Scholar] [CrossRef]
- Hsieh, T.; Hsieh, S.; Chen, C. Chemical constituents from the stems of Cinnamomum insulari-montanum. Chem. Nat. Compd. 2010, 46, 99–100. [Google Scholar] [CrossRef]
- Chen, C.; Lin, C.; Kao, C.; Chen, C.; Li, H. Chemical constituents of the leaves of Cinnamomum insulari-montanum. Chem. Nat. Compd. 2019, 55, 922–923. [Google Scholar] [CrossRef]
- Hsieh, T.J.; Liu, T.Z.; Lu, F.J.; Hsieh, P.Y.; Chen, C.H. Actinodaphnine induces apoptosis through increased nitric oxide, reactive oxygen species and down-regulation of NF-κB signaling in human hepatoma Mahlavu cells. Food Chem. Toxicol. 2006, 44, 344–354. [Google Scholar] [CrossRef]
- Lin, C.C.; Cheng, H.Y.; Fang, B.J. Anti-herpes virus type 2 activity of herbal medicines from Taiwan. Pharm. Biol. 2003, 41, 259–262. [Google Scholar] [CrossRef]
- Kaewchoothong, A.; Tewtrakul, S.; Panichayupakaranant, P. Inhibitory effect of phenylbutanoid-rich Zingiber cassumunar extracts on nitric oxide production by murine macrophage-like RAW264.7 cells. Phytother. Res. 2012, 26, 1789–1792. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Hong, Z.L.; Yang, W.L.; Wu, M.H.; Huang, J.C.; Lee, J.Y. A novel homosesquiterpenoid from the stems of Cinnamomum burmanii. Nat. Prod. Res. 2012, 26, 1218–1223. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.L.; Yang, W.L.; Li, Y.P.; Kang, Y.F.; Wu, H.M.; Chen, C.L.; Chen, C.T.; Chen, W.Y.; Chen, S.J.; Chen, C.Y. A novel homosesquiterpene from Cinnamomum macrostemon Hayata. J. Med. Med. Sci. 2012, 3, 90–092. [Google Scholar]
- Yang, C.L.; Wu, H.C.; Hwang, T.L.; Lin, C.H.; Cheng, Y.H.; Wang, C.C.; Kan, H.L.; Kuo, Y.H.; Chen, I.S.; Chang, H.S. Anti-inflammatory and antibacterial activity constituents from the stem of Cinnamomum validinerve. Molecules 2020, 25, 3382. [Google Scholar] [CrossRef]
- Pflästerer, D.; Rudolph, M.; Yates, B.F.; Ariafard, A.; Hashmi, A.S.K. Total synthesis of (±)-dihydroisosubamol. Adv. Synth. Catal. 2017, 359, 866–874. [Google Scholar] [CrossRef]
- Kumar, K.S.; Hsieh, H.W.; Wang, S.Y. Anti-inflammatory effect of lucidone in mice via inhibition of NF-κB/MAP kinase pathway. Int. Immunopharmacol. 2010, 10, 385–392. [Google Scholar] [CrossRef]
- Li, J.-k.; Nie, L.; Zhao, Y.-P.; Zhang, Y.-Q.; Wang, X.; Wang, S.-S.; Liu, Y.; Zhao, H.; Cheng, L. IL-17 mediates inflammatory reactions via p38/c-Fos and JNK/c-Jun activation in an AP-1-dependent manner in human nucleus pulposus cells. J. Transl. Med. 2016, 14, 77. [Google Scholar] [CrossRef] [Green Version]
- Hannemann, N.; Jordan, J.; Paul, S.; Reid, S.; Baenkler, H.W.; Sonnewald, S.; Bäuerle, T.; Vera, J.; Schett, G.; Bozec, A. The AP-1 transcription factor c-Jun promotes arthritis by regulating cyclooxygenase-2 and arginase-1 expression in macrophages. J. Immunol. 2017, 198, 3605–3614. [Google Scholar] [CrossRef] [Green Version]
- Kang, G.; Kong, P.J.; Yuh, Y.J.; Lim, S.Y.; Yim, S.V.; Chun, W.; Kim, S.S. Curcumin suppresses lipopolysaccharide-induced cyclooxygenase-2 expression by inhibiting activator protein 1 and nuclear factor κB bindings in BV2 microglial cells. J. Pharmacol. Sci. 2004, 94, 325–328. [Google Scholar] [CrossRef] [Green Version]
- Leclercq, I.A.; Farrell, G.C.; Sempoux, C.; dela Peña, A.; Horsmans, Y. Curcumin inhibits NF-κB activation and reduces the severity of experimental steatohepatitis in mice. J. Hepatol. 2004, 41, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Xia, Y. c-Jun, at the crossroad of the signaling network. Protein Cell 2011, 2, 889–898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, Z.; Machius, M.; Nestler, E.J.; Rudenko, G. Activator Protein-1: Redox switch controlling structure and DNA-binding. Nucleic Acids Res. 2017, 5, 11425–11436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juergens, U.R. Anti-inflammatory properties of the monoterpene 1.8-cineole: Current evidence for co-medication in inflammatory airway diseases. Drug Res. 2014, 64, 638–646. [Google Scholar] [CrossRef] [PubMed]




| RT (min) | Name | Concentration% | KI a | Identification b |
|---|---|---|---|---|
| 15.97 | α-Pinene | 7.55 | 934 | KI/MS/ST |
| 17.12 | Camphene | 2.44 | 950 | KI/MS/ST |
| 18.97 | Sabinene | 5.06 | 974 | KI/MS/ST |
| 19.12 | β-Pinene | 3.14 | 976 | KI/MS/ST |
| 21.93 | α-Terpinene | 0.53 | 1014 | KI/MS/ST |
| 22.44 | m-Cymene | 0.74 | 1023 | KI/MS |
| 22.69 | Limonene | 4.14 | 1028 | KI/MS/ST |
| 22.85 | 1,8-Cineole | 35.94 | 1030 | KI/MS/ST |
| 24.51 | γ-Terpinene | 2.46 | 1059 | KI/MS/ST |
| 31.21 | 4-Terpeneol | 1.54 | 1180 | KI/MS/ST |
| 32.02 | α-Terpineol | 3.61 | 1194 | KI/MS/ST |
| 36.29 | Isobornyl acetate | 4.81 | 1283 | KI/MS/ST |
| 40.27 | α-Cubebene | 0.46 | 1371 | KI/MS |
| 42.13 | Caryophyllene | 0.93 | 1413 | KI/MS/ST |
| 45.06 | (+)-Valencene | 0.85 | 1483 | KI/MS |
| 45.36 | β-Guaiene | 0.37 | 1490 | KI/MS |
| 46.32 | Cubebol | 0.61 | 1514 | KI/MS/ST |
| 47.21 | α-Calacorene | 0.55 | 1537 | KI/MS |
| 47.57 | β-Elemol | 0.93 | 1546 | KI/MS/ST |
| 48.77 | Nerolidol | 0.87 | 1576 | KI/MS/ST |
| 50.79 | Guaiol | 0.83 | 1628 | KI/MS |
| 51.61 | α-Eudesmol | 6.17 | 1650 | KI/MS/ST |
| 64.95 | Kaurene | 1.47 | 2034 | KI/MS/ST |
| Fractions | A | B | C | D | E | F | G | H | I | J | CUR a |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Weight (g) | 0.1 | 0.3 | 0.7 | 1.5 | 5.0 | 4.2 | 2.4 | 3.2 | 1.8 | 3.4 | - |
| IC50 b (μg/mL) | >40 | >20 | 19.0 | 25.5 | 37.2 | 15.8 | 23.5 | 25.1 | 33.9 | 38.0 | 2.6 |
| Positions | 13C | 1H | HMBC |
|---|---|---|---|
| 1 | 131.5 | 7.26 (d, J = 8.3 Hz, 1H) | H-1/C-2, H-1/C-3, H-1/C-11a, H-1/C-11b |
| 2 | 114.1 | 6.70 (dd, J = 8.3, 2.5 Hz, 1H) | H2/C-1, H-2/C-3, H-2/C-4 |
| 3 | 158.1 | ||
| 4 | 114.0 | 6.65 (d, J = 2.5 Hz, 1H) | H-4/C-2, H-4/C-3, H-4/C-4a, H-4/C-5 |
| 4a | 131.8 | ||
| 5 | 34.1 | Ha: 2.72 (dd, J = 12.6, 6.3 Hz, 1H) | H-5/C-4, H-5/C-4a, H-5/C-6, H-5/C-7, H-5/C-11b |
| Hb: 3.03 (dd, J = 12.6, 8.2 Hz, 1H) | |||
| 6 | 128.0 | 6.13 (t, J = 7.4 Hz, 1H) | H-6/C-5, H-6/C-7a, H-6/C-12 |
| 7 | 139.1 | ||
| 7a | 129.6 | ||
| 8 | 110.8 | 7.18 (s, 1H) | H-8/C-7, H-8/C-9, H-8/C-10 |
| 9 | 147.8 | ||
| 10 | 146.5 | ||
| 11 | 117.2 | 7.03 (s, 1H) | H-11/C-7a, H-11/C-9, H-11/C-11a |
| 11a | 135.6 | ||
| 11b | 145.1 | ||
| 12 | 66.4 | Ha: 4.33 (d, J = 13.0 Hz, 1H) | H-12/C-6, H-12/C-7, H-12/C-7a |
| Hb: 4.45 (d, J = 13.0 Hz, 1H) | |||
| OMe | 56.6 | 3.92 (s, 3H) | OMe/C-9 |
| Positions | 13C | 1H | HMBC |
|---|---|---|---|
| 1 | 131.4 | 7.32 (d, J = 8.4 Hz, 1H) | H-1/C-3, H-1/C-11a, H-1/C-11b |
| 2 | 114.3 | 6.71 (dd, J = 8.4, 2.6 Hz, 1H) | H-2/C-3, H-2/C-4 |
| 3 | 158.3 | ||
| 4 | 114.1 | 6.64 (d, J = 2.6 Hz, 1H) | H-4/C-2, H-4/C-3, H-4/C-4a, H-4/C-5 |
| 4a | 132.0 | ||
| 5 | 34.1 | Ha: 2.72 (dd, J = 12.6, 6.3 Hz, 1H) | H-5/C-4, H-5/C-4a, H-5/C-6, H-5/C-7, H-5/C-11b |
| Hb: 3.03 (dd, J = 12.6, 8.2 Hz, 1H) | |||
| 6 | 127.9 | 6.13 (t, J = 7.6 Hz, 1H) | H-6/C-5, H-6/C-7, H-6/C-12 |
| 7 | 138.9 | ||
| 7a | 130.8 | ||
| 8 | 114.1 | 7.06 (s, 1H) | H-8/C-7, H-8/C-9, H-8/C-10 |
| 9 | 146.5 | ||
| 10 | 148.1 | ||
| 11 | 113.7 | 7.08 (s, 1H) | H-11/C-7a, H-11/C-9, H-11/C-10, H-11/C-11a |
| 11a | 134.2 | ||
| 11b | 145.2 | ||
| 12 | 66.1 | Ha: 4.30 (d, J = 13.0 Hz, 1H) | H-12/C-6, H-12/C-7, H-12/C-7a |
| Hb: 4.39 (d, J = 13.0 Hz, 1H) | |||
| OMe | 56.5 | 3.93 (s, 3H) | OMe/C-10 |
| Compound | Protein | PDB Code | IC50 (μM) | Minimum Binding Energy |
|---|---|---|---|---|
| Isoburmanol | AP-1 | 5VPA | 14.0 | −103.53 |
| AP-1 (oxidized form) | 5VPB | −95.50 | ||
| AP-1 (bound to DNA) | 1FOS | −85.33 | ||
| JNK1 (MYU binding sites) | 3V3V | −96.61 | ||
| IKKβ (KSA binding sites) | 4KIK | −94.71 | ||
| NF-κB | 1BFT | −86.78 | ||
| Burmanol | AP-1 | 5VPA | 43.8 | −98.88 |
| AP-1 (oxidized form) | 5VPB | −87.82 | ||
| AP-1 (bound to DNA) | 1FOS | −88.51 | ||
| JNK1 (MYU binding sites) | 3V3V | −91.95 | ||
| IKKβ (KSA binding sites) | 4KIK | −90.82 | ||
| NF-κB | 1BFT | −89.70 | ||
| Curcumin | AP-1 | 5VPA | 9.4 | −113.54 |
| AP-1 (oxidized form) | 5VPB | −113.95 | ||
| AP-1 (bound to DNA) | 1FOS | −103.62 | ||
| JNK1 (MYU binding sites) | 3V3V | −114.44 | ||
| IKKβ (KSA binding sites) | 4KIK | −122.01 | ||
| NF-κB | 1BFT | −120.81 | ||
| Ethyl p-coumarate | AP-1 | 5VPA | 58.0 | −89.43 |
| AP-1 (oxidized form) | 5VPB | −82.96 | ||
| AP-1 (bound to DNA | 1FOS | −72.42 | ||
| JNK1 (MYU binding sites) | 3V3V | −77.13 | ||
| IKKβ (KSA binding sites) | 4KIK | −73.09 | ||
| NF-κB | 1BFT | −74.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-Y.; Wu, P.-C.; Tsao, N.-W.; Tseng, Y.-H.; Chu, F.-H.; Wang, S.-Y. Anti-Inflammatory Activities of Constituents from Cinnamomum insularimontanum Hayata Leaves and Their Mechanisms. Plants 2022, 11, 3252. https://doi.org/10.3390/plants11233252
Chen C-Y, Wu P-C, Tsao N-W, Tseng Y-H, Chu F-H, Wang S-Y. Anti-Inflammatory Activities of Constituents from Cinnamomum insularimontanum Hayata Leaves and Their Mechanisms. Plants. 2022; 11(23):3252. https://doi.org/10.3390/plants11233252
Chicago/Turabian StyleChen, Chieh-Yin, Pei-Chen Wu, Nai-Wen Tsao, Yen-Hsueh Tseng, Fang-Hua Chu, and Sheng-Yang Wang. 2022. "Anti-Inflammatory Activities of Constituents from Cinnamomum insularimontanum Hayata Leaves and Their Mechanisms" Plants 11, no. 23: 3252. https://doi.org/10.3390/plants11233252








