Environmental Modulation of Mini-Clonal Gardens for Cutting Production and Propagation of Hard- and Easy-to-Root Eucalyptus spp
Abstract
:1. Introduction
2. Results
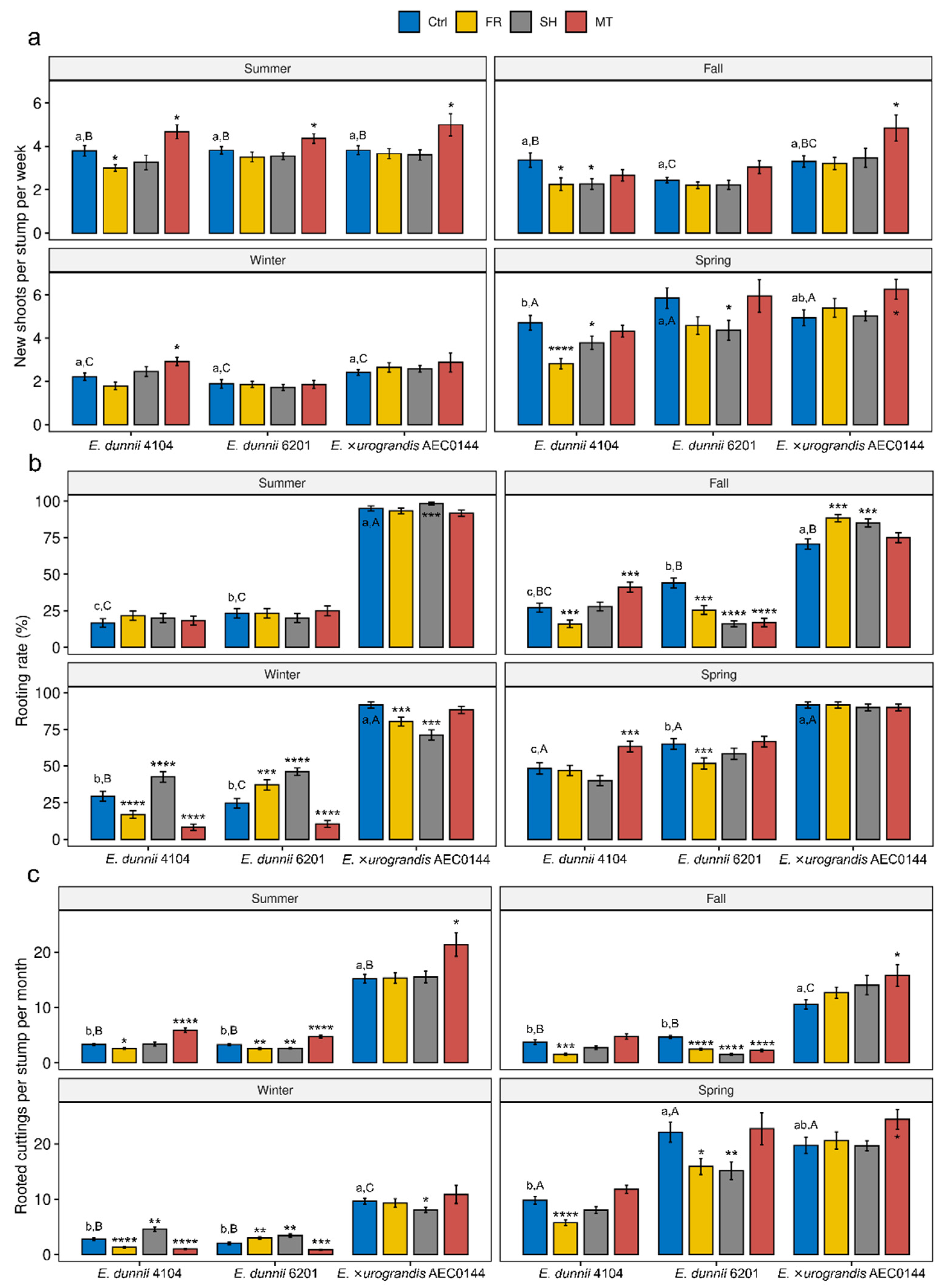
2.1. Branching, Rooting, and Overall Productivity
2.2. Developmental Outcomes of Cuttings and AR System Morphology
2.3. Carbohydrate Content
2.4. Gene Expression Patterns
2.5. Foliar Nutritional Profile
3. Discussion
3.1. Mini-Tunnels Modulate Overall Productivity in a Season-Dependent Manner by Increasing Branching Rates
3.2. Seasonality and Species Are Major Factors in Cutting Developmental Outcomes
3.3. Global Carbohydrate Content Is more Sensitive to Seasonality and Species Effects Than to Mini-Tunnel Treatment
3.4. Contrasting Seasons Are Linked to Consistently Different Cutting Expression Profiles of AR-Related Genes
3.5. Mini-Tunnels Significantly Alter Leaf Nutrient Profiles of Mini-Stumps and Cuttings in the Summer
4. Materials and Methods
4.1. Plant Material and Study Site
4.2. Experiment Design and Treatments
4.3. Cutting Excision and Rooting
4.4. Sample Collection, Morphological Assessments, and Chlorophyll Measurements
4.5. Carbohydrate Content Analysis
4.6. Gene Expression Analysis
4.7. Leaf Nutrient Profiling
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IBÁ-Brazilian Tree Industry Annual Report; 2021. Available online: https://iba.org/eng/datafiles/publicacoes/relatorios/relatorioiba2021.pdf (accessed on 2 March 2022).
- Comission on Genetic Resources for Food and Agriculture. The State of the World’s Forest Genetic Resources; FAO: Rome, Italy, 2014. [Google Scholar]
- Wendling, I.; Xavier, A.; Gomes, J.; Pires, I.; Andrade, H. Propagação Clonal de Híbridos de Eucalyptus spp. Por Miniestaquia. Rev. Árvore 2000, 24, 181–186. [Google Scholar]
- Assis, T.F.; Fett-Neto, A.G.; Alfenas, A.C. Current Techniques and Prospects for the Clonal Propagation of Hardwoods with Emphasis on Eucalyptus. In Plantation Forest Biotechnology for the 21st Century; Walter, C., Carson, M., Eds.; Research Signpost: Thiruvananthapuram, India, 2004; pp. 1109–1119. ISBN 81-7736-228-3. [Google Scholar]
- De Almeida, M.R.; Aumond, M.; Da Costa, C.T.; Schwambach, J.; Ruedell, C.M.; Correa, L.R.; Fett-Neto, A.G. Environmental Control of Adventitious Rooting in Eucalyptus and Populus Cuttings. Trees Struct. Funct. 2017, 31, 1377–1390. [Google Scholar] [CrossRef]
- Steffens, B.; Rasmussen, A. The Physiology of Adventitious Roots. Plant Physiol. 2016, 170, 603–617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- da Costa, C.T.; de Almeida, M.R.; Ruedell, C.M.; Schwambach, J.; Maraschin, F.S.; Fett-Neto, A.G. When Stress and Development go Hand in Hand: Main Hormonal Controls of Adventitious Rooting in Cuttings. Front. Plant Sci. 2013, 4, 133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bellini, C.; Pacurar, D.I.; Perrone, I. Adventitious Roots and Lateral Roots: Similarities and Differences. Annu. Rev. Plant Biol. 2014, 65, 639–666. [Google Scholar] [CrossRef] [PubMed]
- Pacurar, D.I.; Perrone, I.; Bellini, C. Auxin is a Central Player in the Hormone Cross-Talks that Control Adventitious Rooting. Physiol. Plant 2014, 151, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Druege, U.; Franken, P.; Hajirezaei, M.R. Plant Hormone Homeostasis, Signaling, and Function during Adventitious Root Formation in Cuttings. Front. Plant Sci. 2016, 7, 381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Druege, U.; Hilo, A.; Pérez-Pérez, J.M.; Klopotek, Y.; Acosta, M.; Shahinnia, F.; Zerche, S.; Franken, P.; Hajirezaei, M.R. Molecular and Physiological Control of Adventitious Rooting in Cuttings: Phytohormone Action Meets Resource Allocation. Ann. Bot. 2019, 123, 929–949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guan, L.; Murphy, A.S.; Peer, W.A.; Gan, L.; Li, Y.; Cheng, Z.M. (Max) Physiological and Molecular Regulation of Adventitious Root Formation. CRC Crit. Rev. Plant Sci. 2015, 34, 506–521. [Google Scholar] [CrossRef]
- Steffens, B.; Kovalev, A.; Gorb, S.N.; Sauter, M. Emerging Roots Alter Epidermal Cell Fate through Mechanical and Reactive Oxygen Species Signaling. Plant Cell 2012, 24, 3296–3306. [Google Scholar] [CrossRef] [Green Version]
- Hartmann, H.T.; Kester, D.E.; Davies, F.T., Jr.; Geneve, R.L. Principles of Propagation by Cuttings. In Hartmann and Kester’s Plant Propagation Principles and Practices; Pearson: London, UK, 2014; pp. 295–361. ISBN 1-292-02088-1. [Google Scholar]
- Gutierrez, L.; Mongelard, G.; Floková, K.; Pǎcurar, D.I.; Novák, O.; Staswick, P.; Kowalczyk, M.; Pǎcurar, M.; Demailly, H.; Geiss, G.; et al. Auxin Controls Arabidopsis Adventitious Root Initiation by Regulating Jasmonic Acid Homeostasis. Plant Cell 2012, 24, 2515–2527. [Google Scholar] [CrossRef] [PubMed]
- Lakehal, A.; Chaabouni, S.; Cavel, E.; le Hir, R.; Ranjan, A.; Raneshan, Z.; Novák, O.; Păcurar, D.I.; Perrone, I.; Jobert, F.; et al. A Molecular Framework for the Control of Adventitious Rooting by TIR1/AFB2-Aux/IAA-Dependent Auxin Signaling in Arabidopsis. Mol. Plant 2019, 12, 1499–1514. [Google Scholar] [CrossRef] [PubMed]
- da Costa, C.T.; Offringa, R.; Fett-Neto, A.G. The Role of Auxin Transporters and Receptors in Adventitious Rooting of Arabidopsis thaliana Pre-Etiolated Flooded Seedlings. Plant Sci. 2020, 290, 110294. [Google Scholar] [CrossRef] [PubMed]
- Ruedell, C.M.; de Almeida, M.R.; Fett-Neto, A.G. Concerted Transcription of Auxin and Carbohydrate Homeostasis-Related Genes Underlies Improved Adventitious Rooting of Microcuttings Derived from Far-Red Treated Eucalyptus globulus Labill Mother Plants. Plant Physiol. Biochem. 2015, 97, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Aumond, M.L.; de Araujo, A.T.; de Oliveira Junkes, C.F.; de Almeida, M.R.; Matsuura, H.N.; de Costa, F.; Fett-Neto, A.G. Events Associated with Early Age-Related Decline in Adventitious Rooting Competence of Eucalyptus globulus Labill. Front. Plant Sci. 2017, 8, 1734. [Google Scholar] [CrossRef] [Green Version]
- de Almeida, M.R.; de Bastiani, D.; Gaeta, M.L.; de Araújo Mariath, J.E.; de Costa, F.; Retallick, J.; Nolan, L.; Tai, H.H.; Strömvik, M.V.; Fett-Neto, A.G. Comparative Transcriptional Analysis Provides New Insights into the Molecular Basis of Adventitious Rooting Recalcitrance in Eucalyptus. Plant Sci. 2015, 239, 155–165. [Google Scholar] [CrossRef] [PubMed]
- da Cunha, A.C.M.C.M.; de Paiva, H.N.; Xavier, A.; Otoni, W.C. Papel Da Nutrição Mineral Na Formação de Raízes Adventícias Em Plantas Lenhosas. Pesqui. Florest. Bras. 2010, 58, 35. [Google Scholar] [CrossRef]
- Vilasboa, J.; Da Costa, C.T.; Fett-Neto, A.G. Rooting of Eucalypt Cuttings as a Problem-Solving Oriented Model in Plant Biology. Prog. Biophys. Mol. Biol. 2019, 146, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Christiaens, A.; Gobin, B.; Van Huylenbroeck, J.; Van Labeke, M.-C. Adventitious Rooting of Chrysanthemum is Stimulated by a Low Red:Far-Red Ratio. J. Plant Physiol. 2019, 236, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Ruedell, C.M.; de Almeida, M.R.; Schwambach, J.; Posenato, C.F.; Fett-Neto, A.G. Pre and Post-Severance Effects of Light Quality on Carbohydrate Dynamics and Microcutting Adventitious Rooting of Two Eucalyptus Species of Contrasting Recalcitrance. Plant Growth Regul. 2013, 69, 235–245. [Google Scholar] [CrossRef]
- Schwambach, J.; Fadanelli, C.; Fett-Neto, A.G. Mineral Nutrition and Adventitious Rooting in Microcuttings of Eucalyptus globulus. Tree Physiol. 2005, 25, 487–494. [Google Scholar] [CrossRef]
- da Cunha, A.C.M.C.M.; de Paiva, H.N.; Leite, H.G.; de Barros, N.F.; Leite, F.P. Relações Entre Variáveis Climáticas Com Produção e Enraizamento de Miniestacas de Eucalipto. Rev. Árvore 2009, 33, 195–203. [Google Scholar] [CrossRef] [Green Version]
- Assis, T. Hybrids and Mini-Cutting: A Powerful Combination that Has Revolutionized the Eucalyptus Clonal Forestry. BMC Proc. 2011, 5, I18. [Google Scholar] [CrossRef] [Green Version]
- Brondani, G.E.; de Oliveira, L.S.; Konzen, E.R.; da Silva, A.L.L.; Costa, J.L. Mini-Incubators Improve the Adventitious Rooting Performance of Corymbia and Eucalyptus Microcuttings According to the Environment in Which They Are Conditioned. An. Acad. Bras. Cienc. 2017, 90, 2409–2423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Lima, M.S.; Araujo, M.M.; Berghetti, Á.L.P.; Aimi, S.C.; Costella, C.; Griebeler, A.M.; Somavilla, L.M.; dos Santos, O.P.; dos Reis Teixeira Valente, B.M. Mini-cutting Technique Application in Corymbia and Eucalyptus: Effects of Mini-tunnel Use across Seasons of the Year. New For. 2022, 53, 161–179. [Google Scholar] [CrossRef]
- Batista, A.F.; dos Santos, G.A.; Silva, L.D.; Quevedo, F.F.; de Assis, T.F. The Use of Mini-Tunnels and the Effects of Seasonality in the Clonal Propagation of Eucalyptus in a Subtropical Environment. Aust. For. 2015, 78, 65–72. [Google Scholar] [CrossRef]
- Franklin, K.A. Shade Avoidance. New Phytol. 2008, 179, 930–944. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Li, L. Hormonal Regulation in Shade Avoidance. Front. Plant Sci. 2017, 8, 1527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konzen, E.R.; Saudade de Aguiar, N.; Navroski, M.C.; Mota, C.S.; Miranda, L.; Estopa, R.A.; Tonett, E.L.; de Pereira, M.O. Artificial Light Improves Productivity of Mini-Cuttings in a Clonal Minigarden of Eucalyptus benthamii and E. dunnii. South For. 2021, 83, 310–320. [Google Scholar] [CrossRef]
- Muñoz, P.; Cotado, A.; Munné-Bosch, S. Transient Photoinhibition and Photo-Oxidative Stress as an Integral Part of Stress Acclimation and Plant Development in a Dioecious Tree Adapted to Mediterranean Ecosystems. Tree Physiol. 2021, 41, 1212–1229. [Google Scholar] [CrossRef] [PubMed]
- Zerche, S.; Haensch, K.T.; Druege, U.; Hajirezaei, M.R. Nitrogen Remobilisation Facilitates Adventitious Root Formation on Reversible Dark-Induced Carbohydrate Depletion in Petunia hybrida. BMC Plant Biol. 2016, 16, 219. [Google Scholar] [CrossRef] [PubMed]
- Klopotek, Y.; Franken, P.; Klaering, H.P.; Fischer, K.; Hause, B.; Hajirezaei, M.R.; Druege, U. A Higher Sink Competitiveness of the Rooting Zone and Invertases Are Involved in Dark Stimulation of Adventitious Root Formation in Petunia hybrida Cuttings. Plant Sci. 2016, 243, 10–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villanova, J.; Cano, A.; Albacete, A.; López, A.; Cano, E.; Acosta, M.; Pérez-Pérez, J.M. Multiple Factors Influence Adventitious Rooting in Carnation (Dianthus caryophyllus L.) Stem Cuttings. Plant Growth Regul. 2017, 81, 511–521. [Google Scholar] [CrossRef]
- Brondani, G.E.; Baccarin, F.J.B.; de Wit Ondas, H.W.; Stape, J.L.; Gonçalves, A.N.; de Almeida, M. Low Temperature, IBA Concentrations and Optimal Time for Adventitious Rooting of Eucalyptus benthamii Mini-Cuttings. J. For. Res. 2012, 23, 583–592. [Google Scholar] [CrossRef]
- Eliyahu, A.; Duman, Z.; Sherf, S.; Genin, O.; Cinnamon, Y.; Abu-Abied, M.; Weinstain, R.; Dag, A.; Sadot, E. Vegetative Propagation of Elite Eucalyptus Clones as Food Source for Honeybees (Apis mellifera); Adventitious Roots versus Callus Formation. Isr. J. Plant Sci. 2020, 67, 83–97. [Google Scholar] [CrossRef] [Green Version]
- Abu-Abied, M.; Szwerdszarf, D.; Mordehaev, I.; Levy, A.; Rogovoy, O.; Belausov, E.; Yaniv, Y.; Uliel, S.; Katzenellenbogen, M.; Riov, J.; et al. Microarray Analysis Revealed Upregulation of Nitrate Reductase in Juvenile Cuttings of Eucalyptus grandis, Which Correlated with Increased Nitric Oxide Production and Adventitious Root Formation. Plant J. 2012, 71, 787–799. [Google Scholar] [CrossRef]
- Abu-Abied, M.; Szwerdszarf, D.; Mordehaev, I.; Yaniv, Y.; Levinkron, S.; Rubinstein, M.; Riov, J.; Ophir, R.; Sadot, E. Gene Expression Profiling in Juvenile and Mature Cuttings of Eucalyptus grandis Reveals the Importance of Microtubule Remodeling during Adventitious Root Formation. BMC Genom. 2014, 15, 826. [Google Scholar] [CrossRef] [Green Version]
- Pommerrenig, B.; Ludewig, F.; Cvetkovic, J.; Trentmann, O.; Klemens, P.A.W.; Neuhaus, H.E. In Concert: Orchestrated Changes in Carbohydrate Homeostasis Are Critical for Plant Abiotic Stress Tolerance. Plant Cell Physiol. 2018, 59, 1290–1299. [Google Scholar] [CrossRef]
- Oberschelp, G.P.J.; Guarnaschelli, A.B.; Teson, N.; Harrand, L.; Podestá, F.E.; Margarit, E.; Pedro Javier, O.G.; Beatriz, G.A.; Natalia, T.; Leonel, H.; et al. Cold Acclimation and Freezing Tolerance in Three Eucalyptus Species: A Metabolomic and Proteomic Approach. Plant Physiol. Biochem. 2020, 154, 316–327. [Google Scholar] [CrossRef]
- Trueman, S.J.; McMahon, T.V.; Bristow, M. Production of Cuttings in Response to Stock Plant Temperature in the Subtropical Eucalypts, Corymbia citriodora and Eucalyptus dunnii. New For. 2013, 44, 265–279. [Google Scholar] [CrossRef]
- Lakehal, A.; Bellini, C. Control of Adventitious Root Formation: Insights into Synergistic and Antagonistic Hormonal Interactions. Physiol. Plant 2019, 165, 90–100. [Google Scholar] [CrossRef]
- Druege, U.; Franken, P.; Lischewski, S.; Ahkami, A.H.; Zerche, S.; Hause, B.; Hajirezaei, M.R. Transcriptomic Analysis Reveals Ethylene as Stimulator and Auxin as Regulator of Adventitious Root Formation in Petunia Cuttings. Front. Plant Sci. 2014, 5, 494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramirez-Carvajal, G.A.; Morse, A.M.; Dervinis, C.; Davis, J.M. The Cytokinin Type-B Response Regulator PtRR13 is a Negative Regulator of Adventitious Root Development in Populus. Plant Physiol. 2009, 150, 759–771. [Google Scholar] [CrossRef] [Green Version]
- Smolka, A.; Welander, M.; Olsson, P.; Holefors, A.; Zhu, L.H. Involvement of the ARRO-1 Gene in Adventitious Root Formation in Apple. Plant Sci. 2009, 177, 710–715. [Google Scholar] [CrossRef]
- Lakehal, A.; Dob, A.; Novák, O.; Bellini, C. A DAO1-Mediated Circuit Controls Auxin and Jasmonate Crosstalk Robustness during Adventitious Root Initiation in Arabidopsis. Int. J. Mol. Sci. 2019, 20, 4428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lischweski, S.; Muchow, A.; Guthörl, D.; Hause, B. Jasmonates Act Positively in Adventitious Root Formation in Petunia Cuttings. BMC Plant Biol. 2015, 15, 229. [Google Scholar] [CrossRef] [Green Version]
- Reddy, S.K.; Finlayson, S.A. Phytochrome B Promotes Branching in Arabidopsis by Suppressing Auxin Signaling. Plant Physiol. 2014, 164, 1542–1550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, H.; Abernathy, S.D.; White, R.H.; Finlayson, S.A. Photosynthetic Photon Flux Density and Phytochrome B Interact to Regulate Branching in Arabidopsis. Plant Cell Environ. 2011, 34, 1986–1998. [Google Scholar] [CrossRef]
- Kumari, S.; Yadav, S.; Patra, D.; Singh, S.; Sarkar, A.K.; Panigrahi, K.C.S. Uncovering the Molecular Signature Underlying the Light Intensity-Dependent Root Development in Arabidopsis thaliana. BMC Genom. 2019, 20, 596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holalu, S.V.; Reddy, S.K.; Blackman, B.K.; Finlayson, S.A. Phytochrome Interacting Factors 4 and 5 Regulate Axillary Branching via Bud Abscisic Acid and Stem Auxin Signalling. Plant Cell Environ. 2020, 43, 2224–2238. [Google Scholar] [CrossRef]
- Pierik, R.; de Wit, M. Shade Avoidance: Phytochrome Signalling and Other Aboveground Neighbour Detection Cues. J. Exp. Bot. 2014, 65, 2815–2824. [Google Scholar] [CrossRef]
- Sun, J.; Qi, L.; Li, Y.; Chu, J.; Li, C. Pif4-Mediated Activation of Yucca8 Expression Integrates Temperature into the Auxin Pathway in Regulating Arabidopsis Hypocotyl Growth. PLoS Genet 2012, 8, e1002594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nomoto, Y.; Kubozono, S.; Miyachi, M.; Yamashino, T.; Nakamichi, N.; Mizuno, T. Circadian Clock and PIF4-Mediated External Coincidence Mechanism Coordinately Integrates Both of the Cues from Seasonal Changes in Photoperiod and Temperature to Regulate Plant Growth in Arabidopsis thaliana. Plant Signal. Behav. 2013, 8, e22863. [Google Scholar] [CrossRef] [Green Version]
- Xu, M.; Zhu, L.; Shou, H.; Wu, P. A PIN1 Family Gene, OsPIN1, Involved in Auxin-Dependent Adventitious Root Emergence and Tillering in Rice. Plant Cell Physiol. 2005, 46, 1674–1681. [Google Scholar] [CrossRef]
- Shinohara, N.; Taylor, C.; Leyser, O. Strigolactone Can Promote or Inhibit Shoot Branching by Triggering Rapid Depletion of the Auxin Efflux Protein PIN1 from the Plasma Membrane. PLoS Biol. 2013, 11, e1001474. [Google Scholar] [CrossRef] [Green Version]
- da Cunha, A.C.M.C.M.; de Paiva, H.N.; Leite, H.G.; de Barros, N.F.; Leite, F.P. Influência Do Estado Nutricional de Minicepas No Enraizamento de Miniestacas de Eucalipto. Rev. Árvore 2009, 33, 607–615. [Google Scholar] [CrossRef] [Green Version]
- Vasconcelos, E.; Ribeiro, H.; Ramos, A.; Coutinho, J. Influence of Nitrogen and Potassium on Eucalyptus globulus Labill. Mother Plants. Rev. Ciências Agrárias 2007, 30, 87–97. [Google Scholar]
- Oberschelp, G.P.J.; Gonçalves, A.N. Assessing the Effects of Basal Media on the in Vitro Propagation and Nutritional Status of Eucalyptus dunnii Maiden. Vitr. Cell. Dev. Biol.-Plant 2016, 52, 28–37. [Google Scholar] [CrossRef]
- Trueman, S.J.S.J.; McMahon, T.V.; Bristow, M. Production of Eucalyptus cloeziana Cuttings in Response to Stock Plant Temperature. J. Trop. For. Sci. 2013, 25, 60–69. [Google Scholar]
- Trueman, S.J.; McMahon, T.V.; Bristow, M. Nutrient Partitioning among the Roots, Hedge and Cuttings of Corymbia citriodora Stock Plants. J. Soil Sci. Plant Nutr. 2013, 13, 977–989. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- McCready, R.M.; Guggolz, J.; Silviera, V.; Owens, H.S. Determination of Starch and Amylose in Vegetables. Anal. Chem. 1950, 22, 1156–1158. [Google Scholar] [CrossRef]
- Chow, P.S.; Landhäusser, S.M. A Method for Routine Measurements of Total Sugar and Starch Content in Woody Plant Tissues. Tree Physiol. 2004, 24, 1129–1136. [Google Scholar] [CrossRef]
- White, E.J.; Venter, M.; Hiten, N.F.; Burger, J.T. Modified Cetyltrimethylammonium Bromide Method Improves Robustness and Versatility: The Benchmark for Plant RNA Extraction. Biotechnol. J. 2008, 3, 1424–1428. [Google Scholar] [CrossRef]
- Ye, J.; Coulouris, G.; Zaretskaya, I.; Cutcutache, I.; Rozen, S.; Madden, T.L. Primer-BLAST: A Tool to Design Target-Specific Primers for Polymerase Chain Reaction. BMC Bioinform. 2012, 13, 134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Almeida, M.R.; Ruedell, C.M.; Ricachenevsky, F.K.; Sperotto, R.A.; Pasquali, G.; Fett-Neto, A.G. Reference Gene Selection for Quantitative Reverse Transcription-Polymerase Chain Reaction Normalization during in Vitro Adventitious Rooting in Eucalyptus globulus Labill. BMC Mol. Biol. 2010, 11, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perkins, J.R.; Dawes, J.M.; McMahon, S.B.; Bennett, D.L.H.; Orengo, C.; Kohl, M. ReadqPCR and NormqPCR: R Packages for the Reading, Quality Checking and Normalisation of RT-QPCR Quantification Cycle (Cq) Data. BMC Genom. 2012, 13, 296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ritz, C.; Spiess, A.N. QpcR: An R Package for Sigmoidal Model Selection in Quantitative Real-Time Polymerase Chain Reaction Analysis. Bioinformatics 2008, 24, 1549–1551. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, M.; Kim, D.R. Pcr: An R Package for Quality Assessment, Analysis and Testing of QPCR Data. Peer J. 2018, 6, e4473. [Google Scholar] [CrossRef] [Green Version]
- Malavolta, E.; Vitti, G.C.; de Oliveira, S.A. Avaliação do Estado Nutricional das Plantas: Princípios e Aplicações, 2nd ed.; POTAFOS: Piracicaba, Brazil, 1997. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: New York, NY, USA, 2021. [Google Scholar]
- Kassambara, A. Rstatix: Pipe-Friendly Framework for Basic Statistical Tests; R Core Team: New York, NY, USA, 2021. [Google Scholar]
- Kassambara, A. Ggpubr: “ggplot2” Based Publication Ready Plots; R Core Team: New York, NY, USA, 2020. [Google Scholar]
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R Package for Multivariate Analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Kassambara, A.; Mundt, F. Factoextra: Extract and Visualize the Results of Multivariate Data Analyses; R Core Team: New York, NY, USA, 2020. [Google Scholar]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilasboa, J.; Da Costa, C.T.; Fett-Neto, A.G. Environmental Modulation of Mini-Clonal Gardens for Cutting Production and Propagation of Hard- and Easy-to-Root Eucalyptus spp. Plants 2022, 11, 3281. https://doi.org/10.3390/plants11233281
Vilasboa J, Da Costa CT, Fett-Neto AG. Environmental Modulation of Mini-Clonal Gardens for Cutting Production and Propagation of Hard- and Easy-to-Root Eucalyptus spp. Plants. 2022; 11(23):3281. https://doi.org/10.3390/plants11233281
Chicago/Turabian StyleVilasboa, Johnatan, Cibele T. Da Costa, and Arthur G. Fett-Neto. 2022. "Environmental Modulation of Mini-Clonal Gardens for Cutting Production and Propagation of Hard- and Easy-to-Root Eucalyptus spp" Plants 11, no. 23: 3281. https://doi.org/10.3390/plants11233281




