1. Introduction
Maize (
Zea mays, L.) is the third-most important crop following wheat and rice and therefore considered the primary staple cereal for food and livestock feed, in addition to uses in the industry for oils and biofuel production as alcohol [
1,
2]. Maize originated from Americans and was then cultivated in different agroecological zones with a wide range of environmental conditions around the world [
3].
According to the environmental conditions and climatic factors, diverse pathogenic fungi were reported on maize grains, causing malformation, growth reduction and decreasing of photosynthetic capacity, and deficiency in nutritional elements due to the depletion of nitrogen, carbon, and other inorganic salts from the host [
4]. These changes reduce the quality, quantity, and nutritional values of grain. Moreover, pathogenic fungi produce mycotoxins (aflatoxins and ochratoxins) that are, in addition, harmful to humans and animals [
5]. Moreover, since grains are a good substrate for microbial growth, many species of toxicogenic fungi, e.g.,
Aspergillus, Penicillium, and
Curvularia, and the
Fusarium genera infect maize and were stated to cause serious diseases such as seed, root, stem, ear, and kernel rots [
6,
7].
It was found that fungi can distribute and survive in different wide types of ecosystems [
8]. The biodiversity and populations of the fungal pathogens correlate with biotic factors like the plant and/or microbial community and abiotic elements such as temperature, humidity, moisture, salinity, and pH of the soil. Both biotic and abiotic factors usually affect also the pathogenicity of the phytopathogenic fungi [
9,
10].
Little of the environmental literature has taken care of seed-borne mycobiomes. For instance,
Fusarium ear rot, caused mainly by
Fusarium verticillioides and
F. proliferatum, is an important seed-borne pathogenic fungi of maize. Both fungi can survive in infected maize grains and have a wide distribution in temperate regions [
11,
12]. The moisture content and temperature stress during plant field growth and postharvest are the most related factors that affect fungal growth, spreading, and mycotoxins production by both pathogens on maize grains [
5,
11,
13]. In this connection, maize seed-borne
Fusarium graminearum and
Stenocarpella maydis pathogens were reported at a higher incidence in the wetter seasons of the studied sites. It was concluded that the inoculum levels of each of the fungal species can be affected by local factors like the microenvironment in field conditions, as well as land preparation, previous crops, the volume of debris from the preseason, and fertilizer application, which have a bigger impact compared to the drought condition on the fungal community structure of ear rot pathogens [
14].
However, the lytic activity of enzymes (e.g., cellulases, amylases, pectinases, and proteases) of the phytopathogenic fungi is found to be linked to the pathogenicity features and therefore play a vital role in disease incidence and development. These enzymes could be used as a differentiating tool to study the biodiversity of phytopathogens [
15].
The fungal cell wall supports and protects internal structures and organelles. Protein constitutes 20–30% of the cell wall and is firmly intertwined within the glucan and chitin-based matrix of the filamentous fungi [
16]. Among the various components, the protein content mediates molecule absorption, aids in adhesion for cell migration and fusion, maintains the cell shape, protects the cell against irrelevant substances, mediates the molecule’s absorption, transmits intracellular signals from exterior stimuli, and synthesizes and remodels the cell wall components [
17]. These features represent a protective barrier against various adverse ecosystems and could therefore be used as an indicator for the survivability of the fungal pathogens under certain ecosystem conditions.
During the pathogenesis, to survive and cause the disease, the fungus produces specialized proteins to suppress the plant’s immune response [
18]. In general, amino acids are considered as fuel for the defense genes in a plant, and fuel for the fungus during infection as well; a battle may arise between the fungus and the plant, provided that either of them acquires a large number of amino acids. If the plant contains many amino acids, its resistance to infection is higher [
19,
20]. Moreover, if the fungus possesses a large number of amino acids, its ability to infect is high; contrarily, a lack of proteins in the plant facilitates the infection process, and their deficiency in the fungus reduces the ability of the fungus to cause the disease [
21,
22]. That is why the current study investigated both amino acids and proteins of the pathogenic fungi.
Molecular characterization is another tool that uses DNA sequencing for the identification of fungal groups and, in comparison, for the description of fungal biodiversity. Genetic markers as genes associated with internal transcribed spacer (ITS) sections and the highly conserved 5.8S gene between ITS1 and ITS2 are used for taxonomic identification and phylogenetic relationships among fungal communities [
23,
24].
To the authors’ knowledge, the current pioneer study is the first report on the biodiversity of maize grain-borne pathogenic fungi in Saudi Arabia, representing a case study to draw the attention of the biodiversity of phytopathogenic fungal communities associated with Zea mays grain in relation to the pathology, enzymatic activity, cell wall protein content, and molecular identifications. This may enable understanding the ecobiological relationships of these pathogens to facilitate the management process and get rid of them in the future.
4. Discussion
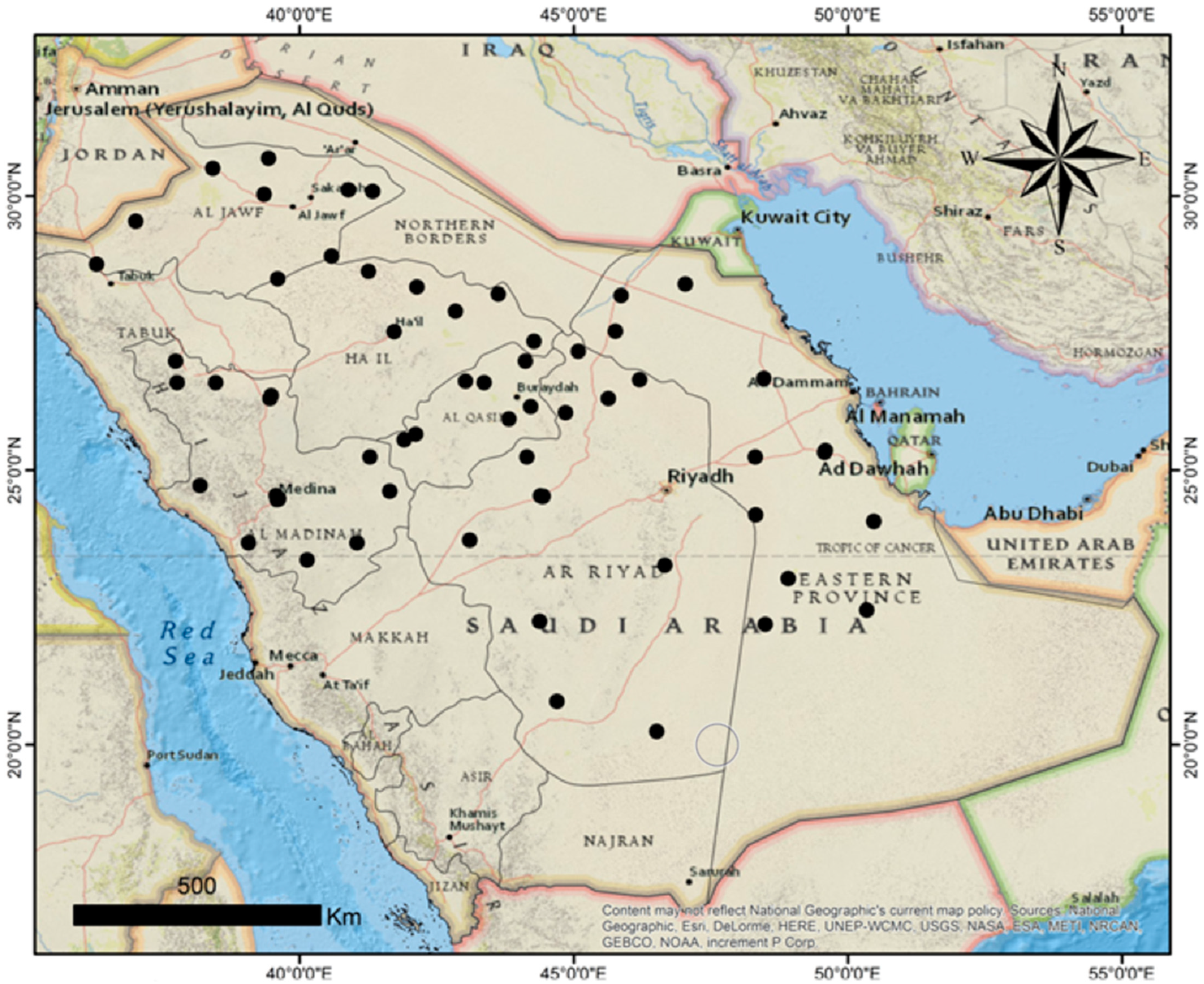
The distribution of maize seed-borne mycobiota was explored by several seed health testing methods. Sixty maize seed samples collected from different locations in 12 provinces of Saudi Arabia were examined for the incidence and frequency of seed-borne fungi using the SMB, DFB, and AP techniques. Forty-one fungal species comprising saprophytic and pathogenic ones were detected, in which their percent infestation was variable according to the locations. Our results revealed that maize seeds were infected by varying degrees with several pathogenic fungi, like
F. verticillioides,
F. proliferatum,
Alternaria alternata,
Drechslera maydis, and
S. strictum, which are the primary causal agents of pink ear rot,
Alternaria leaf blight, Southern corn leaf blight, and wilt diseases in most world maize-growing areas [
39,
40,
41,
42].
Regarding the detection methods, SMB, AP, and DFB showed substantial variances in the frequencies of the recovered fungi. The SMB technique was superior in detecting the maximum number (36 species) of seed-borne fungi compared to the DFB (21 species) and AP (26 species) techniques. The obtained results are consistent with the findings of many studies of research that the SMB technique provided ideal conditions for the development of mycelial growth and spore formation of many Hyphomycetes [
43].
Fusarium verticillioides, often in association with
F. subglutinans and
F. proliferatum, are the most destructive prevalent causal pathogens of
Fusarium ear rot or pink ear rot disease transmitted by maize grains worldwide and are dominant in drier and warmer climates worldwide [
44], causing a reduction in maize output by 10% typically and 30–50% in severely affected areas [
12]. Mycotoxin’s accumulation in preharvest-infected plants or stored grains like deoxynivalenol, zearalenol, fusaric acid, and fumonisin yielded by such pathogens impair human health and animals, as well as support the fungal virulence in severely infecting seedlings of various maize genotypes [
13,
41]. Moreover,
A. niger and
A. flavus were the most dominant in all used seed health techniques, recording <80% frequencies for each.
Aspergillus species, e.g.,
A. flavus and
A. niger are well-known agricultural pathogens that cause high economic losses by reducing the quality of infected maize grain, especially when ear rot symptoms appear, which are commonly associated with the secretion of the fungal aflatoxins and ochratoxins, as well as less-prominent toxins like patulin [
45].
Our findings revealed that maize seeds were infected by the C. lunata, C. australiensis, F. incarnatum, F. chlamydosporum, S. zeae, and S. strictum pathogens at lower percentages. The detection of the above-mentioned fungal pathogens of maize grains constitutes the first comprehensive report of seed-borne of maize in Saudi Arabia. The presence of so many fungal pathogens on corn grains in high proportions calls for the need for field studies for these and other pathogens. Public awareness of the need to study seed health and develop practical resistance methods appropriate to improve the quality of these grains should also be increased.
The isolated maize grain-borne fungi were subjected to a pathogenicity test. The growing-on test showed symptom similarities in the infestation treatments of
Fusarium species, especially
F. verticillioides,
F. proliferatum, and
F. chlamydosporum, which were rotted seeds and stunted and yellow seedlings. In this respect,
F. verticillioides KSU1M1-3 and
F. proliferatum KSU1M2-2 recorded the highest significant decreases in seedling survival (56.67 and 62.83%, respectively) as compared with the uninoculated control (96.0%). Similar findings were reported earlier on
F. verticillioides and
F. proliferatum as the most destructive abundant pathogens associated with maize grains worldwide [
35,
39]. Both pathogens can survive in infected maize seeds without causing apparent symptoms on seed tissues and subsequently transmitted systemically to growing seedlings through the stalk up to the ear, causing blights and rot of the root, stem, and ear in the next season [
12].
Although
Acremonium zeae Gams & Sumner (
Sarocladium zeae) was previously described as the seed-borne causal agent of the black bundle and
Acremonium stalk rot diseases on maize [
46], the current study reported the susceptibility of maize infection by
S. zeae,
C. lunata, and
C. australiensis, recorded for the first time in the studied area of Saudi Arabia. Our data declare the ability of
S. zeae (KSU2M2-1 and KSU2M2-2),
C. lunata KSU3M1-1, and
C.
australiensis KSU3M2-1 pathogens to develop symptoms on maize seedlings in the form of a significant decrease in seedling survival as compared to the uninfected control.
Sarocladium zeae was previously described as producing extracellular hydrolases, e.g., cellulases, amylases, and proteases [
47], which are reported to play a role in the host-pathogen interaction; these enzymes were also reported in our isolates. On the other hand, a recent study identified
S. zeae as the beneficial endophyte in maize seeds [
42]. This contradiction may be due to the presence of different strains having, or not, various pathogenic activities on plants. However, information about the role of endophytic
Sarocladium species, including
S. zeae, in maize is still unclear.
Curvularia lunata was previously reported as a causal pathogen of
Curvularia leaf spot disease of corn in the United States and China under hot–humid and tropical areas, causing significant yield loss [
48].
C. lunata can produce a non-host-specific toxin, furanoid (methyl 5-(hydroxymethyl) furan-2-carboxylate), as the virulence factor that induces the development of symptoms on maize plants comparable to those of natural infection [
49].
The results obtained revealed that
Alternaria alternata KSU4M1-1 caused dramatic losses of seedling survival (67%) as compared with the control treatment (96%). Recently,
A. alternata was reported to be among four causal agents of
Alternaria leaf spot disease on maize seedlings [
50]. Other studies have stated that the risk is not only due to the yield loss they cause but also due to reduction in the grain quality (discoloration) and nutritive value, in addition to an array of
Alternaria mycotoxins that belong to three derivatives: tetramic acid, dibenzopyrone, and perylene, which play an important role in the pathogenesis and can inhibit the germination of maize as well [
51,
52].
The pathogenicity test concluded C. australiensis KSU3M2-1, C. lunata KSU3M1-1, and S. zeae (KSU2M2-1 and KSU2M2-2) as the first recorded in KSA. However, based on the pathogenicity test, the most severe pathogenic fungi were selected for the rest of the study.
The biodiversity of the fungal species in every maize crop governorate was investigated. The Shannon diversity index is usually applied as a popular metric used in ecology for estimating species diversity. The index considers the number of species living in a habitat (richness) and their relative abundance (evenness). Therefore, the distribution level of the fungi among the species and their abundance in the governorates could be observed. The current biodiversity metrics exhibited that the Najran governorate recorded the greatest species richness and diversity. The biodiversity and species abundance among the studied region may be due to the variability of the climatic conditions that were characterized by semi-arid to arid climates with moderate humidity and wind speed levels and relatively high temperatures, which aid in the growth and dispersal of a wide range of fungi.
The 12 isolated pathogenic fungi exhibited lytic activity during growth on the fermented medium. The tissue structure of maize plants, which was utilized during the fermentation, is hard to degrade, composed mainly of lignin, cellulose, pectin, and starch, as well as protein in lower amounts. Such a complicated construction restricts the penetration of pathogenic fungi into plant tissues. Therefore, the cooperation of various lytic enzymes may be required for the pathogenesis process. That is why the various hydrolytic enzymes, i.e., cellulases (FPase, CMCase, and β-glucosidase); amylase; and proteinase, were assayed. The previous enzymes are reported to be involved in the pathogenicity process since these enzymes are known as cell wall-degrading enzymes (CWDE) that facilitate the penetration of the fungal hyphae to enter plant tissue [
15,
30]. The great disparity in the enzymatic profile among fungal isolates may be due to the type of strain, the ecological conditions, and the genetic variation.
Cellulose is the main natural polymer composed of glucose units. The overall lytic activity of cellulase enzymes leads to the catalyzation of cellulose to single glucose units. The intermediate step of hydrolysis is performed by endoglucanase that randomly cleaves the β-1,4-glucosidic bonds in the inner cellulose, which, in combination with the catalytic action of two other exoglycanase enzymes (cellodextrinase and cellobiohydrolase) and β-glucosidase, lead to the complete degradation of the cellulose skeleton into glucose units [
30,
53,
54].
Virtually, pectin is present in all plants and contributes to the cell structure. It is composed of galacturonic acid monomers. Pectin-degrading enzymes have two major classes: esterases and depolymerases. PGase is the main heterogeneous pectinase catalyzing the hydrolysis of the pectin polymer into galacturonic acid by cleaving the α-1,4-glycosidic bond [
32,
55].
Starch is the main component of maize grains, which is decomposed by amylase, a starch-degrading enzyme. The lytic behavior of this enzyme is mediated by β-amylase and γ-amylase, leading to the hydrolysis of starch into glucose [
56].
The nitrogenous part of the plant is mainly hydrolyzed by proteases that cleave the protein into peptides and amino acids [
15]. Exopeptidases catalyze the terminal peptide bond, and endopeptidases cleave the nonterminal bonds between amino acids [
57].
The combined action of various lytic enzymes leads to the maceration of plant tissue, which facilitates the infection route by the pathogen. Phytopathogenic fungi produce CWDE to penetrate the host tissues through the degradation of the components of the plant cell wall (physical barrier against pathogenic invasion) [
15] and, therefore, may be essential for pathogenicity.
Chitinase activity was detected in the majority of the tested pathogens. Chitinases catalyze the hydrolysis of the 1–4 β-glycoside bond of N-acetyl-D-glucosamine in chitin that is present in the cell wall of fungi. Therefore, chitinase is used as a fungicide and insecticide agent [
35,
58]. The pathogenic fungus may use chitinase as an additional aid in the pathogenicity process by antagonizing the other beneficial fungi that are present on plants, which guarantee its sole presence on plant tissue and the success of the pathogenicity process. It is worth mentioning that the fermentation medium was free from the chitinase substrate, consequently indicating that chitinase is secreted constitutively by the tested fungi. Three mechanisms are supposed to manage the biosynthesis of the microbial enzymes, which may be induced, constitutive, or both. The induced (inducible) enzyme is one whose secretion is stimulated by a specific substrate (chitin), whereas the constitutive enzyme does not require the presence of the specific substrate [
15]. This leads to the conclusion that the current pathogens may have more than one mechanism of chitinase secretion, representing an additional threat for plant infection. Collectively, the current pathogens have a complementary profile of hydrolytic enzymes, capable of causing a serious infection of maize plants.
Regarding the amino acids and protein content, this is the first investigation that differentiated the essential and nonessential amino acids in the studied seed-borne fungi of maize, which is very important considering their pathogenicity degree and biological properties. As stated earlier, amino acids are an integral part of the hydrophilic–hydrophobic properties of fungal cell walls, and they are included in the structure of microbial organic substances [
59].
In this study, the amount of essential and nonessential amino acids of the 12 fungi was diverse and related to the degree of infection capacity of the fungi and the resistance ability of the host. For example,
F. chlamydosporum (KSU1M3-1) has the highest amount of most of the essential amino acids (tryptophan, leucine, isoleucine, methionine phenylalanine, and lysine) that were related to its lower pathogenicity, whereas the highest content of nonessential amino acids (alanine, glutamic acid, and aspartic acid) was observed in
F. verticillioides (KSU1M1-3) with the highest percentage of pathogenicity and the highest content of threonine. Pathogenic fungi contain different amino acids supporting and facilitating the invasion of fungi and characteristics as an important impact on fungal growth and fungal pathogenicity, as well as induce and activate virulence traits such as hyphal morphogenesis, the formation of a biofilm, and formation of the capsule [
60,
61,
62].
Exploring protein polymorphism has been a central approach in the studying of the classification, identification, and genetic variability of specific and subspecific taxa of fungi, and further, the pathogenic fungi are classified based on the total protein into (i) fungi with the highest protein, (ii) fungi with moderate protein, and (iii) fungi with the lowest protein [
63,
64]. In this study,
F. verticillioides (KSU1M1-3),
F. verticillioides (KSU1M1-2), and
S. zeae (KSU2M2-1) have the highest pathogenicity related to the highest induction of total protein. To establish successful colonization, plant pathogens, including vascular wilt fungi, secrete effector proteins during the pathogen-host interaction [
65,
66], which are key elements for the pathogen virulence against plants and are particularly important during the biotrophic phase of infection [
67]. The resistant plants are recognized by having a special resistance protein that stimulates the activation of induced defense response against effector proteins [
66,
67].
The current data on amino acids in relation to pathogenicity encourage performing additional studies to discover this point. However, the differences among the 12 tested fungi in this study are expected to be caused by the diversity of the species and strains, as well as the environmental conditions, the different geographical origins, and the growth conditions of the analyzed species.
In this study,
F. verticillioides (KSU1M1-3),
F. verticillioides (KSU1M1-2), and
S. zeae (KSU2M2-1) had the highest pathogenicity related to the highest total protein. The analysis of the protein polymorphism has been an important approach in the classification and identification, as well as the genetic variability, of specific and subspecific taxa of fungi. Several authors have reported that the total proteins can be used as a criterion method for identifying species of the same genus, such as
Fusarium species [
63,
64].
Plant pathogens, including vascular wilt fungi, secrete proteins during colonization to establish a successful pathogen-host interaction [
65]. Microbial pathogens secrete effector proteins, which are recognized by a special group of resistance proteins that stimulates the activation of the induced defense response [
66]. These effector proteins are key elements produced by fungal pathogens for their virulence against plants and are particularly important during the biotrophic phase of infection [
67].
In this study, the total amount of amino acids in the mycelium of the individual fungal strains tested was diverse. The highest total content of bounded amino acids was related to the degree of infection and the resistance ability of the host.
A pathogen requires amino acids (AA) to support its physiological functions, and alterations in the AA availability have remarkable effects on the growth of a pathogen and its expression of virulence factors [
68]. Amino acid metabolism is crucial to pathogenicity. Metabolic adaptation to the microenvironment of the host is associated with fungal morphogenesis, cell wall remodeling, biofilm formation, stress responses, and commensalism, all of which influence the progression of infections. Metabolic adaptation is regulated by complex transcriptional networks, such as the general control of amino acid metabolism [
69]. The utilization of arginine by both host and pathogen represents a metabolic bottleneck that is critical in determining the outcome of a pathogenic infection [
70]. The highest contents of amino acids facilitate the invasion of fungi and characteristics as an important impact on the fungal pathogenicity and growth, as well as induce and activate virulence traits such as hyphal morphogenesis, formation of the biofilm, and formation of the capsule [
60,
61,
62].
The heatmap was used to study the similarities and dissimilarities among fungal taxa. A hierarchical cluster heatmap is one visual method that can be used to clarify the associations between different parameters of samples, such as weather variables and the different pathogenic fungi, whereas the PCA is an unsupervised learning method that reduces the dimensionality of a dataset and extracts only the most important information for analysis [
71]. Both heatmap and the multivariate PCA were performed for visualization of the relationships, similarities, and dissimilarities based on the distances between various parameters against the genotypes. In this respect, the result of the PCA separated fungi according to their biochemical and physiological parameters. Fungal isolates from the Al-Qaseem and Middle Regions were separated from the rest of the fungi.
The ordination diagram of CCA shows that the environmental conditions are critical limiting factors for the biology of fungi, affecting their growth, reproduction, and pathogenicity in many ways. The cumulative percentage for the correlations between species and environmental factors for the first and second axes illuminated 98.88% and 1.12% of the total variance. These results indicated that the environmental factors had a great impact on the distribution patterns of the studied pathogenic fungi. The current data indicate that temperature was the most influential environmental variable, followed by humidity, pressure, and wind speed.
Environmental conditions such as humidity, wind, pH, pressure, and temperature affect the survival, growth, pathogenicity, and infestation of mycoflora [
72]. Saudi Arabia is located in a tropical area, which has a desert climate characterized by high temperatures with high humidity and low rainfall. Accordingly, the correlation between the environmental conditions and colonization of phytopathogenic fungi in maize grain in Saudi Arabia was studied in this investigation by using CCA. The CCA is a multivariate method used for the arrangement of different species along with environmental variables. Moreover, the CCA demonstrates the linear combination of weather variables with the distribution of fungal species, as well as investigates the response of different species to the environmental factors [
73].
It was stated that climatic factors such as temperature, pH, and humidity not only influence the fungal distribution but also affect the growth, survival, and infestation of
Fusarium species in maize [
72]. Furthermore, many studies have indicated that
Fusarium species differed in their optimal temperature requirement for growth that was observed between 25 and 29 °C [
74,
75].
On the other hand, the high temperature may kill the pathogens directly or weaken them by sublethal heat, which makes them unable to damage crops [
76]. We can conclude that temperature can affect fungal growth by affecting the kinetics of the cellular enzymatic reactions and modulating the secretum of the cell [
77]. We assume that the lethal temperature is what stops these reactions and may affect the growth of the fungus and its ability to cause infection. Not only that, but the temperature difference may lead to the emergence of mutagenic isolates that may resist the high temperatures and can cause disease after adapting to the environmental changes.
Additionally, the humidity level was found to be a critical limiting factor for fungus vitality, affecting their distribution, reproduction, and pathogenicity in many cases. In general, the high water content may be necessary for some types of fungi for spore germination, as it is required for the initiation of germination; for instance, temperature and relative humidity are important metrological variables affecting ear and stalk rot infection of maize by
Fusarium spp. [
78].
CCA showed a low correlation of the incidence of
A. alternata, as well as the current newly detected pathogenic fungi in KSA (
C. australiensis, C. lunata, and
S. zeae) with air temperature, relative humidity, pressure, and wind speed compared to the other pathogens. Our results showed that dry conditions and moderate temperature were the critical factors enhancing the prevalence and distribution of the above-mentioned pathogens, which may be explained by their sensitivity to low oxygen conditions in wet soils. High humidity conditions can be appropriate for the presence of antagonistic saprophytic microorganisms, including fungi, yeast, and bacteria, that may reduce the pathogen’s survival potential [
79], whereas the
Magnaporthiopsis maydis DNA levels increased 10 times under drought-stressed corn buds compared to non-stressed plants [
80].
Curvularia lunata is a foliar destructive causal pathogen of
Curvularia leaf spot disease of maize related to subtropical and tropical, as well as sometimes temperate areas, of the world [
81]. Previously,
C. lunata and
C. inaequalis were first reported as the causal agent of
Curvularia leaf spot disease of maize in North China [
82]. It has been reported that the frequency of seed-borne
A. alternata and
S. strictum pathogens in maize grains varies significantly in drought and heat climates in different growing areas of South Africa [
14].
The optimum range of
C. lunta was from 28 to 32 °C and the pH values from 4 to 12. Similarly, the optimum in vitro growth of the
C. lunata pathogen is around 30 °C [
83]. Generally, high temperature and humidity conditions were found to be favorites for the development of
Curvularia leaf spot disease during the flowering to grain filling stages [
84]. Under such conditions, the accumulation of
C. lunata mycotoxin (e.g., furanoid-type toxin) may occur both in vitro and in the plant, which can lead to leaf lesions [
49]. On the contrary, a strong relationship between the dry weather conditions in the field and the
C. lunata success overwintering in infected leaves and becoming primary infection sources in the following season was reported [
82].
Regarding the molecular biology relationships, the phylogenetic framework was explored to detect the fungal evolution and taxonomic studies. In this study, studying the biodiversity and molecular identification of 12 pathogenic fungi in maize in KSA using the ITS region revealed 12 isolates from different species of fungi like
Fusarium,
Curvularia,
Alternaria, and
Sarocladium species. The studied isolates were classified with their nearby relative isolates, which showed highly significant relations among the members of each species. The obtained DNA nucleotide sequences of the 12 fungi displayed 98–100% similarity with their nearby relative isolates in GenBank. The phylogenetic tree classified the fungal strains into clusters, one containing only
Alternaria alternaria (MW553174, outer group). The second cluster included all the other fungal strains, including the Fusarium strains obtained in this study. Intraspecies variability was noted in the ITS regions, which may be due to variances between the phenotypic and molecular taxonomic structures, as reported for the
Fusarium genera [
85].
In the current study, the discriminatory power of the RAPD markers was determined using various parameters. RAPD3 demonstrated the highest efficiency, which was characterized by the highest values of MI (23.81), EMR (26.00), and the percentage of polymorphic loci (100%). The value of PIC ranges from 0 to 1, with values closer to 1 indicating higher polymorphisms, i.e., PIC > 0.5 is classified as highly informative, values of 0.25 < PIC < 0.5 are moderately informative, and PIC < 0. 25 are lowly informative [
86]. The present PIC values ranged from 0.82 in RAPD 2 to 0.95 in RAPD 1, which was higher than the average PIC value (0.261) revealed from 17 RAPD primers used to study the genetic diversity of
F. fujikuroi from maize [
87]. Such results indicate that the reliability and significance of RAPD in the characterization of the populations’ diversity is high. Some authors even indicated a higher RAPD sensitivity in the diversity determination when compared to other molecular techniques, such as RFLP [
88] or SSR [
89]. What is more, RAPD has been widely used in the judgment of genetic similarity [
90] or in the analysis of genetic diversity within a species [
91] or between closely related species [
92]. Therefore, RAPD markers are dominant and efficient in the detection of DNA-sequence polymorphisms of many loci throughout the genome. For example, within the
Fusarium genus, RAPD has been applicable in the distinction between nonpathogenic and pathogenic isolates of
F. oxysporum [
93] or
F. verticillioides strain differentiation, depending on the host plant [
94].
It has been observed that the results obtained by RAPD-PCR when compared with those obtained from the ITS DNA sequences confirm each other. The similarity among the examined individuals within the same species reached more than 95%, which indicates the importance of these methods in the differentiation between individuals within the same species.