Niche Variation in Endemic Lilium pomponium on a Wide Altitudinal Gradient in the Maritime Alps
Abstract
:1. Introduction
2. Results
2.1. Local Topography
2.2. Micro-Niche Diversity
2.3. Marginality and Distribution Limits
2.4. Abundance and Phenotype
2.5. Vegetation Structure
3. Discussion
3.1. Climate Change: The Importance of Tracking the Fine-Scaled Niche
3.2. Ecological and Range Limits, Two Separate Issues
3.3. Perspectives for Conservation Management
4. Materials and Methods
4.1. Study Species and Area
4.2. Altitudinal Variation in Topographic Parameters
4.3. Population Sampling along Altitudinal and Bioclimatic Gradients
4.4. Micro-Niche Characterization
4.5. Ecological Gradients and Distribution Limits
4.6. Phenotypic Traits
4.7. Local Abundance
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kessler, M. Elevational Gradients in Species Richness and Endemism of Selected Plant Groups in the Central Bolivian Andes. Plant Ecol. 2000, 149, 181–193. [Google Scholar] [CrossRef]
- Thompson, J.D. Plant Evolution in the Mediterranean: Insights for Conservation, 2nd ed.; Oxford University Press: New York, NY, USA, 2020; ISBN 978-0-19-883514-1. [Google Scholar]
- Di Biase, L.; Pace, L.; Mantoni, C.; Fattorini, S. Variations in Plant Richness, Biogeographical Composition, and Life Forms along an Elevational Gradient in a Mediterranean Mountain. Plants 2021, 10, 2090. [Google Scholar] [CrossRef] [PubMed]
- Måsviken, J.; Dalerum, F.; Cousins, S.A.O. Contrasting Altitudinal Variation of Alpine Plant Communities along the Swedish Mountains. Ecol. Evol. 2020, 10, 4838–4853. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Hutchinson, I. Cohabitation of Species in an Artificial Grass-Legume Community on Ski-Slopes on Whistler Mountain, British Columbia, Canada. J. Appl. Ecol. 1986, 23, 239–250. [Google Scholar] [CrossRef]
- Gómez, J.M.; Zamora, R. Generalization vs. Specialization in the Pollination System of Hormathophylla Spinosa (Cruciferae). Ecology 1999, 80, 796–805. [Google Scholar] [CrossRef]
- Black, H.N.; Harrison, J.D.G.; Cron, G.V. Do Breeding System and Pollen Limitation Vary with Altitude in the Widespread Herb, Cineraria erodioides (Asteraceae)? S. Afr. J. Bot. 2019, 121, 377–385. [Google Scholar] [CrossRef]
- Totland, Ø. Environment-Dependent Pollen Limitation and Selection on Floral Traits in an Alpine Species. Ecology 2001, 82, 2233–2244. [Google Scholar] [CrossRef]
- Pailler, T.; Humeau, L.; Figier, J.; Thompson, J.D. Reproductive Trait Variation in the Functionally Dioecious and Morphologically Heterostylous Island Endemic Chassalia corallioides (Rubiaceae). Biol. J. Linn. Soc. 1998, 64, 297–313. [Google Scholar] [CrossRef]
- Alonso, C. Pollination Success across an Elevation and Sex Ratio Gradient in Gynodioecious Daphne laureola. Am. J. Bot. 2005, 92, 1264–1269. [Google Scholar] [CrossRef] [Green Version]
- Wagner, J.; Lechleitner, M.; Hosp, D. Pollen Limitation Is Not the Rule in Nival Plants: A Study from the European Central Alps. Am. J. Bot. 2016, 103, 375–387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vitousek, P.M.; Matson, P.A.; Turner, D.R. Elevational and Age Gradients in Hawaiian Montane Rainforest: Foliar and Soil Nutrients. Oecologia 1988, 77, 565–570. [Google Scholar] [CrossRef]
- Todorović, B.; Stevanović, B. Adaptive Characteristics of the Endemic Species Satureja horvatii Šilić (Lamiaceae) in Mountain-Mediterranean and Mediterranean Habitats. Bot. J. Linn. Soc. 1994, 114, 367–376. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, L.; Qi, D. Variation in Leaf Traits at Different Altitudes Reflects the Adaptive Strategy of Plants to Environmental Changes. Ecol. Evol. 2020, 10, 8166–8175. [Google Scholar] [CrossRef] [PubMed]
- Walther, G.-R.; Beißner, S.; Burga, C.A. Trends in the Upward Shift of Alpine Plants. J. Veg. Sci. 2005, 16, 541–548. [Google Scholar] [CrossRef]
- Walther, G.-R.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.C.; Fromentin, J.-M.; Hoegh-Guldberg, O.; Bairlein, F. Ecological Responses to Recent Climate Change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.E.; Goulden, M.L. Rapid Shifts in Plant Distribution with Recent Climate Change. Proc. Natl. Acad. Sci. USA 2008, 105, 11823–11826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, J.; Charpentier, A.; Bouguet, G.; Charmasson, F.; Roset, S.; Buatois, B.; Vernet, P.; Gouyon, P.-H. Evolution of a Genetic Polymorphism with Climate Change in a Mediterranean Landscape. Proc. Natl. Acad. Sci. USA 2013, 110, 2893–2897. [Google Scholar] [CrossRef] [Green Version]
- Emberger, L. La Végétation de La Région Méditerranéenne Essai d’une Classification Des Groupements Végétaux. Rev. Générale Bot. 1930, 42, 641–662. [Google Scholar]
- Ozenda, P. Les Limites de La Végétation Méditerranéenne Dans Les Alpes Maritimes. Bull. Société Bot. Fr. 1950, 97, 156–164. [Google Scholar] [CrossRef] [Green Version]
- Rivas-Martinez, S. Etages bioclimatiques, secteurs chorologiques et séries de végétation de l’Espagne méditerranéenne. Ecol. Mediterr. 1982, 8, 275–288. [Google Scholar] [CrossRef]
- Lavergne, S.; Thompson, J.D.; Garnier, E.; Debussche, M. The Biology and Ecology of Narrow Endemic and Widespread Plants: A Comparative Study of Trait Variation in 20 Congeneric Pairs. Oikos 2004, 107, 505–518. [Google Scholar] [CrossRef]
- Papuga, G.; Gauthier, P.; Pons, V.; Farris, E.; Thompson, J.D. Ecological Niche Differentiation in Peripheral Populations: A Comparative Analysis of Eleven Mediterranean Plant Species. Ecography 2018, 41, 1650–1664. [Google Scholar] [CrossRef] [Green Version]
- Gazaix, A.; Grillas, P.; Papuga, G.; Fontes, H.; Sabatier, F.; Pons, V.; Gauthier, P.; Thompson, J.D. Ecological Niche Differentiation among Six Annual Lythrum Species in Mediterranean Temporary Pools. Oecologia 2021, 197, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Murru, V.; Farris, E.; Santo, A.; Grillo, O.; Piazza, C.; Gaio, A.; Bacchetta, G.; Thompson, J.D. Niche Differentiation at Multiple Spatial Scales on Large and Small Mediterranean Islands for the Endemic Silene velutina Pourr. Ex Loisel. (Caryophyllaceae). Plants 2021, 10, 2298. [Google Scholar] [CrossRef] [PubMed]
- Noble, V.; Diadema, K. La Flore des Alpes-Maritimes et de la Principauté de Monaco: Originalité et Diversité; Conservatoires Botaniques Nationaux Alpin et Méditerranéen; Naturalia Publications Conservatoire Botanique National Méditerranéen: Rouen, France, 2011; ISBN 978-2-909717-75-3. [Google Scholar]
- Macrì, C.; Dagnino, D.; Guerrina, M.; Médail, F.; Minuto, L.; Thompson, J.D.; Casazza, G. Effects of Environmental Heterogeneity on Phenotypic Variation of the Endemic Plant Lilium pomponium in the Maritime and Ligurian Alps. Oecologia 2020, 195, 93–103. [Google Scholar] [CrossRef]
- Casazza, G.; Macrì, C.; Dagnino, D.; Guerrina, M.; Juin, M.; Minuto, L.; Thompson, J.D.; Baumel, A.; Médail, F. When Ecological Marginality Is Not Geographically Peripheral: Exploring Genetic Predictions of the Centre-Periphery Hypothesis in the Endemic Plant Lilium pomponium. PeerJ 2021, 9, e11039. [Google Scholar] [CrossRef]
- Pironon, S.; Papuga, G.; Villellas, J.; Angert, A.L.; García, M.B.; Thompson, J.D. Geographic Variation in Genetic and Demographic Performance: New Insights from an Old Biogeographical Paradigm. Biol. Rev. Camb. Philos. Soc. 2017, 92, 1877–1909. [Google Scholar] [CrossRef]
- Rameau, J.C.; Mansion, D.; Dumé, G.; Gauberville, C. Flore Forestière Française: Guide Écologique Illustré. Région Méditerranéenne Volume 3 de Flore Forestière Française. In Forêt Privée Française; IDF: Paris, France, 2008; ISBN 978-2-904740-16-9. [Google Scholar]
- Kirillova, I.A.; Kirillov, D.V. Impact of Weather Conditions on Seasonal Development, Population Structure and Reproductive Success on Dactylorhiza traunsteineri (Orchidaceae) in the Komi Republic (Russia). Nat. Conserv. Res. 2020, 5, 77–89. [Google Scholar] [CrossRef] [Green Version]
- Sletvold, N.; Ågren, J. Climate-Dependent Costs of Reproduction: Survival and Fecundity Costs Decline with Length of the Growing Season and Summer Temperature. Ecol. Lett. 2015, 18, 357–364. [Google Scholar] [CrossRef]
- Mursal, N.; Mehdiyeva, N.P.; Ibrahimova, A.G. Population Status and Ecology of Platanthera chlorantha (Orchidaceae) in the Greater Caucasus (Azerbaijan). Nat. Conserv. Res. 2020, 5, 114–124. [Google Scholar] [CrossRef]
- Barbero, M. Les remontées méditerranéennes sur le versant italien des Alpes. Ecol. Mediterr. 1978, 4, 109–132. [Google Scholar] [CrossRef]
- Barbero, M.; Bono, G.; Ozenda, P. Sur Les Groupements Végétaux En Limite d’aire Dans Les Alpes Maritimes et Ligures. Bull. Société Bot. Fr. 1970, 117, 593–607. [Google Scholar] [CrossRef] [Green Version]
- Casazza, G.; Zappa, E.; Mariotti, M.G.; Médail, F.; Minuto, L. Ecological and Historical Factors Affecting Distribution Pattern and Richness of Endemic Plant Species: The Case of the Maritime and Ligurian Alps Hotspot. Divers. Distrib. 2008, 14, 47–58. [Google Scholar] [CrossRef]
- Diadema, K.; Bretagnolle, F.; Affre, L.; Yuan, Y.-M.; Médail, F. Geographic Structure of Molecular Variation of Gentiana ligustica (Gentianaceae) in the Maritime and Ligurian Regional Hotspot, Inferred from ITS Sequences. Taxon 2005, 54, 887–894. [Google Scholar] [CrossRef]
- Médail, F.; Diadema, K. Glacial Refugia Influence Plant Diversity Patterns in the Mediterranean Basin. J. Biogeogr. 2009, 36, 1333–1345. [Google Scholar] [CrossRef]
- Jacquemyn, H.; Honnay, O.; Pailler, T. Range Size Variation, Nestedness and Species Turnover of Orchid Species along an Altitudinal Gradient on Réunion Island: Implications for Conservation. Biol. Conserv. 2007, 136, 388–397. [Google Scholar] [CrossRef]
- Rydin, H.; Snoeijs, P.; Diekmann, M.; Van der Maarel, E. (Eds.) Acta phytogeographica Suecica. In Swedish Plant Geography; Svenska Växtgeografiska Sällskapet: Uppsala, Sweden, 1999; ISBN 978-91-7210-484-6. [Google Scholar]
- Grytnes, J.; Kapfer, J.; Jurasinski, G.; Birks, H.H.; Henriksen, H.; Klanderud, K.; Odland, A.; Ohlson, M.; Wipf, S.; Birks, H.J.B. Identifying the Driving Factors behind Observed Elevational Range Shifts on European Mountains. Glob. Ecol. Biogeogr. 2014, 23, 876–884. [Google Scholar] [CrossRef] [Green Version]
- Körner, C. The Use of “altitude” in Ecological Research. Trends Ecol. Evol. 2007, 22, 569–574. [Google Scholar] [CrossRef]
- Casazza, G.; Giordani, P.; Benesperi, R.; Foggi, B.; Viciani, D.; Filigheddu, R.; Farris, E.; Bagella, S.; Pisanu, S.; Mariotti, M.G. Climate Change Hastens the Urgency of Conservation for Range-Restricted Plant Species in the Central-Northern Mediterranean Region. Biol. Conserv. 2014, 179, 129–138. [Google Scholar] [CrossRef]
- Gauthier, P.; Bernard, C.; Thompson, J.D. Exploring Vulnerability of Listed Mediterranean Plants in Relation to Risks of Population Loss. J. Nat. Conserv. 2019, 51, 125736. [Google Scholar] [CrossRef]
- SILENE. Available online: http://www.silene.eu/index.php?cont=accueil (accessed on 23 February 2021).
- IGN RGE ALTI®. Version 2.0. Descriptif de Contenu. 2013. Available online: https://geoservices.ign.fr/sites/default/files/2021-07/DC_RGEALTI_2-0.pdf (accessed on 15 February 2022).
- Hijmans, R.J.; van Etten, J.; Sumner, M.; Cheng, J.; Bevan, A.; Bivand, R.; Busetto, L.; Canty, M.; Forrest, D.; Ghosh, A.; et al. Raster: Geographic Data Analysis and Modeling. R Package Version 3.1-5. 2020. Available online: https://cran.r-project.org/web/packages/raster/index.html (accessed on 15 February 2022).
- R Development Core Team. R: A Language and Environment for Statistical Computing: Reference Index; R Foundation for Statistical Computing: Vienna, Austria, 2010; ISBN 978-3-900051-07-5. [Google Scholar]
- Hijmans, R.; Phillips, S.; Leathwick, J.; Elith, J. Dismo: Species Distribution Modeling. R Package Version 1.3-5. 2017. Available online: https://rspatial.org/raster/sdm/ (accessed on 15 February 2022).
- Husson, F.; Josse, J.; Le, S.; Mazet, J.; FactoMineR: Multivariate Exploratory Data Analysis and Data Mining. R Package Version 1.42. 2019. Available online: https://cran.r-project.org/web/packages/FactoMineR/index.html (accessed on 15 February 2022).
- Dray, S.; Dufour, A.; Thioulouse, J.; Jombart, T.; Pavoine, S.; Lobry, J.R.; Ollier, S.; Borcard, D.; Legendre, P.; Bougeard, S.; et al. Ade4: Analysis of Ecological Data: Exploratory and Euclidean Methods in Environmental Sciences. R Package Version 1.7-13. 2018. Available online: https://cran.r-project.org/web/packages/ade4/ade4.pdf (accessed on 15 February 2022).
- Martin, N.; Carrega, P.; Adnes, C. Downscaling à Fine Résolution Spatiale Des Températures Actuelles et Futures Par Modélisation Statistique Des Sorties ALADIN-Climat Sur Les Alpes-Maritimes (France). Climatologie 2013, 10, 51–72. [Google Scholar] [CrossRef] [Green Version]





| Description of the Micro-Niche Type * | Quadrats | Percentage of Quadrats | Position along Ecological Gradients ** | Geographic Limits ** | ||
|---|---|---|---|---|---|---|
| 1 | + Block cover, water retention capacity, first PCoA *** axis (characterised by E. spinosa and T. vulgaris) − Slope, Shannon diversity, litter cover, gravel and bare soil cover, number of species (+) Lichen cover, conductivity (−) pH, 4th PCoA axis (community characterised by Stipa juncea, Rhamnus alaternus, Smilax aspera, Brachypodium retusum, Ruta angustifolia, Pistacia lentiscus | 2 ABC 3 ABC 9 ABC | 13 C 1262 B | 15.9% | Close to the limits of Lilium distribution | |
| 2 | − Easting, northing (+) Bedrock cover, stone cover | 4 ABC 7 ABC 10 ABC 20 ABC 208 ABC 835 ABC 877 ABC | 13 AB 18 C 738 A 1262 AC | 39.1% | Intermediate distance to distribution limits | |
| 3 | + Litter cover, vegetation cover − 1st PCoA axis (characterised by high presence of Sesleria caerulea and Helictotrichon sempervirens), easting | 21 ABC 26 ABC 1148 ABC | 16 A 300 A 738 BC | 18.8% | High altitude | |
| 4 | + Northing, easting, pH − Water retention capacity, conductivity, soil carbon content, block cover (+) Gravel and bare soil cover, moss cover | 6 ABC 11 ABC 334 ABC 1208 ABC | 16_BC 18 AB 300_BC | 26.1% | Close to the distribution centre | |
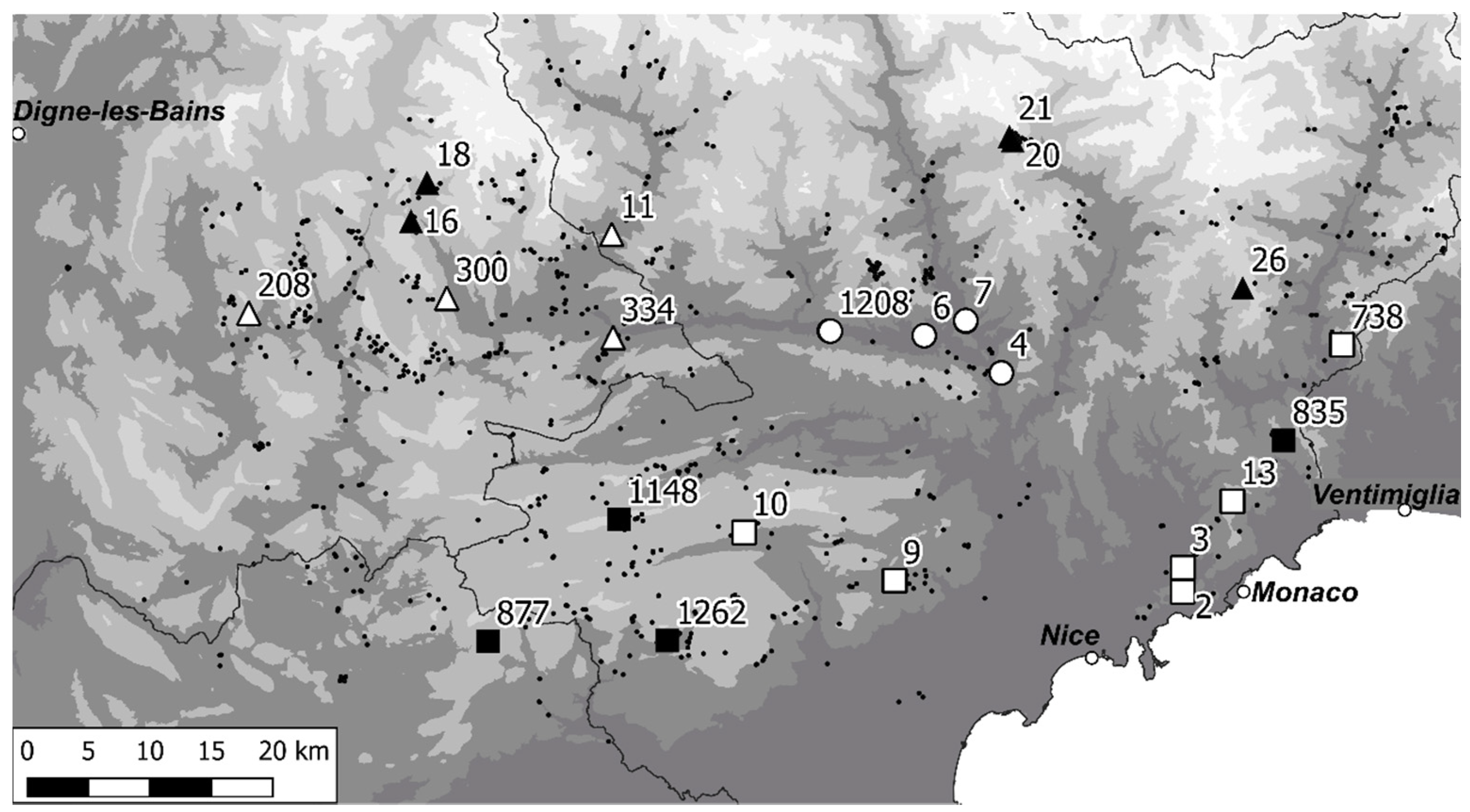
| Altitude (m) | ID | Sampling Group | Locality | Date | Habitat Type | Slope (°) | Aspect (°) |
|---|---|---|---|---|---|---|---|
| 191 | 4 | BA | Vallon d′Aïga Blanca 06450 Utelle | 19 May 2021 | Garrigue, scree slope | 35 | 250 |
| 244 | 7 | BA | M2205 06710 La Tour | 18 May 2021 | Garrigue, scree slope | 40 | 250 |
| 259 | 6 | BA | D126 06710 Massoins | 18 May 2021 | Mixed woodland | 35 | 80 |
| 336 | 1208 | BA | les Moulières 06710 Touët-sur-Var | 15 June 2021 | Shrubland | 45 | 10 |
| 544 | 3 | MA_med | Plateau Tercier 06340 La Trinité | 4 May 2021 | Garrigue on karst | 0 | - |
| 570 | 334 | MA_mont | D610 04320 Entrevaux | 15 June 2021 | Rocky grassland | 20 | 55 |
| 649 | 11 | MA_mont | path to l′Aiguille 06470 Daluis | 15 June 2021 | Shrubland | 25 | 70 |
| 665 | 2 | MA_med | Fort de la Revère 06360 Eze | 3 May 2021 | Garrigue, scree slope | 35 | 180 |
| 918 | 9 | MA_med | Col de Vence 06140 Vence | 19 June 2021 | Rocky grassland | 10 | 150 |
| 934 | 13 | MA_med | Col de la Madone de Gorbio 06440 Peille | 16 June 2021 | Rocky grassland | 35 | 90 |
| 1034 | 10 | MA_med | D2 06620 Gréolières | 19 June 2021 | Garrigue on karst | 45 | 170 |
| 1065 | 300 | MA_mont | Verdre 04240 Annot | 14 June 2021 | Forest edge | 20 | 90 |
| 1078 | 738 | MA_med | below Mont Tron 06540 Breil-sur-Roya | 17 June 2021 | Shrubland | 30 | 105 |
| 1099 | 208 | MA_mont | Chemin du Jas de Bernard 04170 Moriez | 14 June 2021 | Shrubland | 35 | 180 |
| 1118 | 1262 | HA_med | Forêt domaniale de Nans 06460 Saint-Vallier-de-Thiey | 20 June 2021 | Scree slope | 30 | 195 |
| 1142 | 835 | HA_med | below Mont Razet 06500 Castillon | 18 June 2021 | Shrubland | 30 | 230 |
| 1205 | 877 | HA_med | Combe de la Roque 83840 La Roque-Esclapon | 21 June 2021 | Shrubland | 40 | 235 |
| 1305 | 16 | HA_mont | D908, ravin des Baumettes 04170 Thorame-Haute | 28 June 2021 | Forest edge | 45 | 95 |
| 1447 | 1148 | HA_med | Col de Bleine 06750 Andon | 20 June 2021 | Garrigue on karst | 35 | 55 |
| 1505 | 18 | HA_mont | Peyresq, ravin de la Fontaine 04170 Thorame-Haute | 28 June 2021 | Shrubland | 40 | 200 |
| 1640 | 20 | HA_mont | l′Adrechas, la Colmiane 06420 Valdeblore | 29 June 2021 | Shrubland | 30 | 205 |
| 1791 | 21 | HA_mont | les Aiguillettes, la Colmiane 06420 Valdeblore | 29 June 2021 | Pine forest | 40 | 95 |
| 1816 | 26 | HA_mont | Pointe de Ventabren 06380 Moulinet | 30 June 2021 | Montane grassland | 45 | 130 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fontaine, N.; Gauthier, P.; Casazza, G.; Thompson, J.D. Niche Variation in Endemic Lilium pomponium on a Wide Altitudinal Gradient in the Maritime Alps. Plants 2022, 11, 833. https://doi.org/10.3390/plants11060833
Fontaine N, Gauthier P, Casazza G, Thompson JD. Niche Variation in Endemic Lilium pomponium on a Wide Altitudinal Gradient in the Maritime Alps. Plants. 2022; 11(6):833. https://doi.org/10.3390/plants11060833
Chicago/Turabian StyleFontaine, Ninon, Perrine Gauthier, Gabriele Casazza, and John D. Thompson. 2022. "Niche Variation in Endemic Lilium pomponium on a Wide Altitudinal Gradient in the Maritime Alps" Plants 11, no. 6: 833. https://doi.org/10.3390/plants11060833
APA StyleFontaine, N., Gauthier, P., Casazza, G., & Thompson, J. D. (2022). Niche Variation in Endemic Lilium pomponium on a Wide Altitudinal Gradient in the Maritime Alps. Plants, 11(6), 833. https://doi.org/10.3390/plants11060833






