1. Introduction
The world population is expected to grow to almost 9.73 billion by 2050 [
1] at an annual rate of 77 million people per year [
2], which will drive the need for increased food production. To meet the projected global food and fiber demands for a growing population by 2050, current crop production will need to double [
3] requiring a ~2.4% growth rate per year [
4]. However, the biggest challenge for modern agriculture is to increase plant productivity in an environmentally sustainable manner [
5]. Soil infertility is a major crop yield constraint in developing nations [
6]. Chemical fertilizers are the principal input used to increase soil fertility and crop yield. However, excessive use of chemical fertilizers leads to environmental pollution and soil structure degradation [
4]. In this context, there is a need for technologies to sustainably improve nutrient absorption by plants and reduce the use of chemical fertilizers [
7].
Soybean and other legumes are important components of crop rotations due to the ability to promote nitrogen fertility via biological nitrogen fixation (BNF) via diazotrophic symbionts such as
Bradyrhizobium japonicum [
8] that provide an abundant source of biologically available nitrogen for plants [
9]. In addition, biofertilizers, substances that contain viable microorganisms capable of enhancing nutrient uptake and transportation by plants when applied to the seeds or soil [
10], are an environmentally friendly and cost-effective source of plant nutrients that can promote sustainable crop production [
6]. Plant Growth Promotion Rhizobacteria (PGPR) have been used as biofertilizers either by helping to provide nutrients to the plants or by influencing plant growth [
11].
PGPRs are microorganisms that evolved plant associations [
12] and consume root exudates released by the plant host and in return secrete metabolites (e.g., root hormones such as indole-3-acetic acid) that can promote plant growth [
13]. For example, bacterial strains from the genera
Pseudomonas,
Azospirillum,
Azotobacter,
Bacillus,
Klebsiella,
Enterobacter,
Xanthomonas,
Arthrobacter,
Burkhoolderia,
Paenibacillus and
Serratia have been shown to have PGPR activity [
14,
15]. The genus
Bacillus in particular have been studied as PGPR due to their advantageous physiological traits, such as the ability to form hardy spores, which contributes to their survival in soils and a long shelf life that is conducive to field applications [
16]. Many
Bacillus spp. strains have been identified as PGPR with commercial potential use as biofertilizers and biocontrol agents [
17]. Within this genus, strains of
B. velezensis (Bv; previously known as
Bacillus amyloliquefaciens subsp.
plantarum) such as Bv BAC03 [
18] and Bv FZB42 exert plant growth-promoting activities through efficient colonization of plant roots [
19].
Most of the experiments with PGPRs have been performed under controlled environmental conditions, such as greenhouses. In addition, these studies are usually performed in small pots where the soil can be easily sterilized to ensure that the introduced PGPR gets established into the rhizosphere. However, when PGPRs are used in agricultural soils under field conditions their efficacy is more variable and even inexistent [
20]. The lack of consistent success suggests that the introduced microbial population declines rapidly after inoculation [
21,
22]. The decline in viability, numbers and/or metabolic activity of the PGPR population is thought to be caused by the inability of the inoculated PGPR to compete with the native microbiota of that specific soil [
23] and/or by the decrease in the PGPR population and activity due to environmental factors [
24]. To observe significant plant growth promotion, it has been estimated that it requires a PGPR population of 10
3–10
4 cells per gram of root tissue [
15]. PGPRs that are adapted to agricultural soils typically are observed to have a faster growth rate and an ability to metabolize various natural and xenobiotic compounds [
25]. Furthermore, there are plant cultivar-specific differences that can select for or against rhizosphere microbial populations [
5,
26].
Previous research determined that PGPR strains of Bv commonly can use pectin as a sole carbon source, and that soybean seed co-inoculation with Bv and purified pectin or pectin-rich orange peel (a “synbiotic” that consists of a prebiotic and probiotic) can promote soybean growth and nodulation [
27]. Pectin is a complex polysaccharide first isolated and described by Henri Braconnot in 1825 [
28] which consists primarily of galacturonic acids and is present in the primary cell walls and middle lamella in plants [
20,
29]. While purified pectin has been demonstrated to increase the survival of some Bv strains and promote soybean growth, it is not a cost-effective source of pectin for use as a seed amendment. Therefore, other less expensive, pectin-rich amendments were investigated for compatibility with PGPRs to increase soybean growth. Pectin is present in most of the plant tissues, but apple pomace and orange peel are the two most important sources of industrial pectin [
30]. Citrus crops are among the most widely cultivated fruits and more than 80% of citrus is manufactured to obtain juice, jams, jellies, etc. [
31]. After the industrial extraction of orange juice, large amounts of orange peel remain as a by-product [
32]. Orange peel (OP) is a good source of pectin as it contains up to 52.9% pectin content [
33] and has been identified to be used by PGPR strains such as pectinolytic Bv AP193 [
27]. Therefore, to increase the sustainability of agricultural systems by using PGPRs as biofertilizers, it is important to research new strategies to improve PGPR efficacy in agricultural soils in combination with a cost-effective pectin-rich amendment such as orange peel. The primary objective of this study was to test the responsiveness of different soybean cultivars to seed treatment consisting of PGPR and orange peel.
4. Discussion
The effect of soybean seed inoculation with Bv AP193 plus OP was previously tested with positive results in greenhouse and field experiments but only for a one-year experiment and with one soybean cultivar [
27,
42]. For that reason, this study explored the response of 20 soybean cultivars to inoculation with Bv AP193 supplemented with OP as a seed treatment to assess the consistency of the synbiotic inoculant in promoting plant growth. Our results demonstrate that there was a statistically significant positive effect of the inoculation with PGPR plus OP on plant growth promotion of 13.2% for aboveground biomass (
Table 2), 40% for nodule dry weight (
Table 3) and 12.5% for root dry weight (
Table 4) when the response was averaged across all cultivars. In addition, this significant inoculation effect occurred in the absence of inoculation by cultivar interaction. If these positive results are translated to an improvement in seedling vigor and later yield in the field, this PGPR plus OP inoculation treatment could have a very significant impact on soybean crop production and contribute to yield improvement.
However, when the effect of inoculation is analyzed by percentage of change for each cultivar, we observed that the response to the synbiotic inoculation was highly cultivar specific. Cultivars S49XT39, S52XT08, G4190RX and AG69XT0 were found to have a positive response to the inoculation with several plant growth parameters observed to increase; in contrast, cultivars AG53X0, REV4940X, LS5588X and S54XT17 showed a negative response to the inoculation (
Table 2,
Table 3 and
Table 4). For example, the inoculation with AP193 plus OP increased root dry weight by 73 and 28% in S49XT39 and S52XT08 cultivars, respectively, while decreasing 12% in cultivar AG53X0 (
Table 4). This phenomenon could be caused by different compatibility between cultivars and the PGPR strain that is derived from the capability of the PGPR strain to metabolize and use specific root exudates that can vary between the cultivar within each crop species [
43], as well as the presence of plant pathogens that could metabolize pectin and the relative susceptibility of the soybean cultivars to those pathogens. It has been found that rhizosphere populations change depending on the soybean cultivar planted [
44]; therefore, it is possible that PGPR compatibility and effectiveness in promoting growth can change with the cultivar of soybean tested. Similarly to our study, Kuzmicheva et al. [
45] found that inoculation with
Pseudomonas oryzihabitans (strain Ep4) stimulated root growth of the soybean cultivars Nice-Mecha and Svapa, which produced more organic acids; meanwhile, the cultivar Bara that secreted less organic acids did not show root growth promotion. While our preliminary results showed a positive trend of inoculation when all the cultivars response was averaged, we have found that some cultivars did not respond to inoculations as demonstrated in other studies [
43,
45,
46,
47,
48]. These cultivar by PGPR differential responses could be a constraint for inoculant manufacturers since there is a need for consistently effective biologic inoculants that can broadly be used in agriculture. For that reason, the factors that can affect the genotype by PGPR strain responses need to be further studied.
The different response of the cultivars to the inoculation could also be explained by the response of each cultivar to the OP amendment used in this preliminary study. While carrier materials can increase plant growth in combination with PGPR, the inoculation method can also cause stress to the introduced bacteria [
49]. Furthermore, OP has a complex chemical composition, including phenolic fractions that could affect the PGPR and/or each cultivar performance [
27,
42]. For that reason, the greenhouse experiment investigated the combined and separate effects of PGPR and OP on a set of cultivars selected for their high responsiveness (S49XT39 and S52XT08) or lack of response (AG53X0) to inoculation.
As in the preliminary greenhouse experiment, the cultivar S49XT39 showed positive plant growth promotion after inoculation with AP193 plus OP with a 9%, 60% and 32% increase on aboveground biomass, nodule dry weight and nitrogen fixation, respectively, in comparison with the non-inoculated treatment (
Table 5 and
Table 6). In contrast, the cultivar AG53X0 that did not show an increase in aboveground biomass with the inoculation in the preliminary experiment showed a 31.8% increase in biomass in this experiment. Additionally, one of the other responsive cultivars during the preliminary experiment, S52XT08, showed a negative response to the inoculation. This lack of consistency between experiments could be due to a strong influence of the environment and/or soil microbiota on the plant response to PGPR and OP inoculation. According to Nadeem et al. [
50], the effectiveness of inoculation with PGPRs on plant growth promotion might vary depending on microbial populations and their interactions with environmental factors such as soil nutrition, moisture and temperature. Since our preliminary experiment was conducted during the winter of 2020 and the second experiment during spring 2020 in a greenhouse, the environmental conditions such as light intensity, quality and temperature may have affected plant responses to synbiotic inoculation. Light intensity and quality can affect photosynthesis [
51], which ultimately affects the amount of root exudates produced by the plant and therefore might interfere in the plant–PGPR crosstalk [
43].
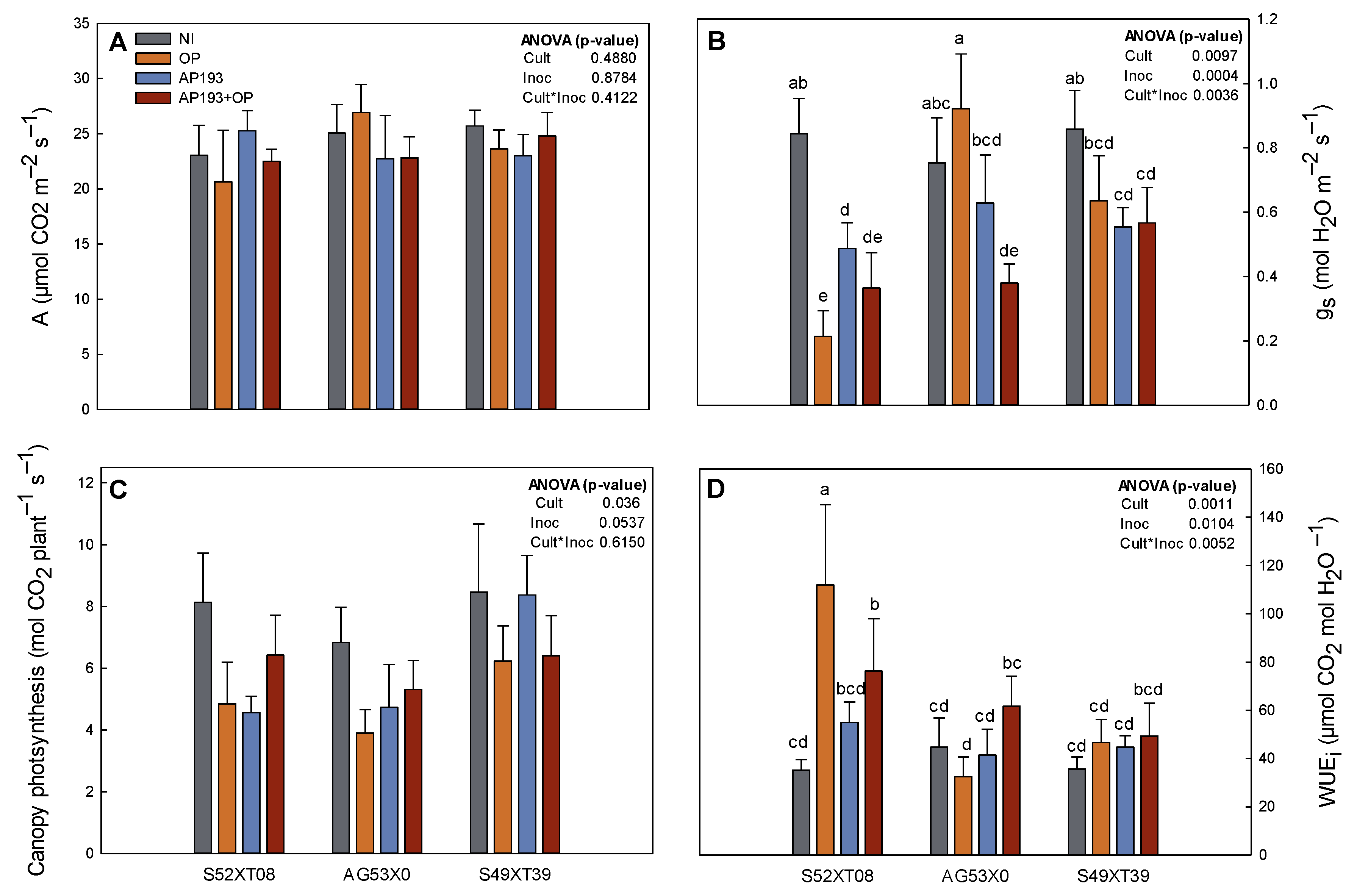
The greenhouse experiment also showed that inoculated plants with PGPR plus OP had lower stomatal conductance (g
s) without any negative effect on plant photosynthesis, which leads to superior intrinsic water-use efficiency (WUE
i). WUE
i is an instantaneous measurement of the efficiency of carbon gain per water loss. The WUE
i tended to be higher in all the cultivars inoculated with PGPR plus OP but was significantly increased in S52XT08 (
Figure 1). Changes in WUE
i are the result of decreases in transpiration rate or increases in photosynthesis activity [
52]. In maize, soil inoculation with
Burkholderia sp. LD-
11 also improved WUE
i through reduction of stomatal aperture provoked by small increases in abscisic acid (ABA) concentration in the leaves, which also promoted biomass accumulation [
53]. While in our current experiment we did not measure ABA concentrations, we hypothesize that inoculation with Bv AP193 may produce an increase in WUE
i due to ABA production. These results are supported by peanut experiments performed also at Auburn University where the inoculation with Bv AP203 with OP amendment resulted in increased WUE
i under well-watered and drought stress conditions [
42]. These results indicate that inoculation with some PGPR strains plus OP may be an important tool to alleviate water stress and benefit plant survival under water shortage environments.
In the field trials, there was no significant effect of inoculation and the interaction between variables for plant growth, nitrogen fixation and yield (
Figure 2 and
Figure 3). While non-significant, cultivar AG69X0 presented the greatest yield on the PGPR plus OP treatment with 14.9% (EVS) and 4.1% (TV) yield increases in comparison with the non-inoculated control. While not significant, these increases are considered “acceptable” by the farmers and by inoculant manufacturers [
12] and therefore should be further studied for commercial application. On the other hand, the other two cultivars showed no effects or even decreases in yield at both locations (
Figure 3). As significant positive effects have been observed in some of the greenhouse experiments, the lack of a significant effect in the field experiment could be attributed to: (1) Effects of the environment [
43,
51]; (2) competition with soil indigenous microbiota [
22,
54,
55,
56]; (3) influence of parasites and pathogens [
22,
57]; or (4) leaching of the inoculum and amendment due to the spray application and the occurrence of extreme precipitation events. While Bv AP193 was selected for its capability to grow and consume pectin [
27], if the pectin washed away or was diluted in the soil, this may explain the lack of significant effects observed in the field. It could also be that there may be other
Bacillus strains or pathogenic microorganisms that were able to grow more rapidly on a pectin-rich substrate and therefore may be able to survive better under field conditions. Future experiments should focus on isolating new
Bacillus strains that catabolize pectin-rich substrates rapidly, or adapting existing strains to improve their ability to grow on OP as a growth substrate, in order to achieve a better plant response. In addition, it needs to be tested whether seed coating or in-furrow seed treatment will be more effective in producing a stable growth promotion response in multiple cultivars under different environments.
In this experiment we have demonstrated that inoculation with AP193 plus orange peel increases soybean growth characteristics of a broad variety of cultivars. However, when the effect of inoculation is analyzed individually cultivar by cultivar, some cultivars showed a very positive significant effect of the inoculation, while other did not showed any effect. In order for this treatment to be usable by industry, the response of all cultivars needs to be homogeneous and therefore the variable response obtained in our experiment needs to be further investigated. Chemical analysis of root exudates of responsive and irresponsive cultivars could inform us of the root exudates that are more effective at attracting and promoting the growth of specific PGPRs. Likewise, transcriptome studies of specific cultivars and PGPR strains could give us information about the molecular mechanisms that mediate plant–microbe interactions in the context of a pectin-rich amendment. Lastly, research in new inoculation methods and synbiotic formulations could facilitate better outcomes for PGPR–plant responses across a wider diversity of cultivars and environmental conditions.