Advances in Understanding the Genetic Basis of Fatty Acids Biosynthesis in Perilla: An Update
Abstract
:1. Introduction
2. Earlier Identification and Cloning of Fatty Acid Encoding Gene in Perilla
3. Transcriptomics Sheds Lights into Key Master Player Enzymes of Perilla Fatty Acid Biosynthesis
4. Whole-Genome-Driven Fatty Acid Genes Discovery
5. Concluding Remarks and Outlook
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Nitta, M.; Lee, J.K.; Kang, C.W.; Katsuta, M.; Yasumoto, S.; Liu, D.; Nagamine, T.; Ohnishi, O. The Distribution of Perilla Species. Genet. Resour. Crop Evol. 2005, 52, 797–804. [Google Scholar] [CrossRef]
- Nitta, M.; Lee, J.K.; Ohnishi, O. Asian Perilla crops and their weedy forms: Their cultivation, utilization and genetic relationships. Econ. Bot. 2003, 57, 245–253. [Google Scholar] [CrossRef]
- Zhang, Y.; Shen, Q.; Leng, L.; Zhang, D.; Chen, S.; Shi, Y.; Ning, Z.; Chen, S. Incipient diploidization of the medicinal plant Perilla within 10,000 years. Nat. Commun. 2021, 12, 5508. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.M. Ethnomedicinal, phytochemical and pharmacological investigations of Perilla frutescens (L.) Britt. Molecules 2019, 24, 102. [Google Scholar] [CrossRef] [Green Version]
- Banno, N.; Akihisa, T.; Tokuda, H.; Yasukawa, K.; Higashihara, H.; Ukiya, M.; Watanabe, K.; Kimura, Y.; Hasegawa, J.I.; Nishino, H. Triterpene acids from the leaves of Perilla frutescens and their anti-inflammatory and antitumor-promoting effects. Biosci. Biotechnol. Biochem. 2004, 68, 85–90. [Google Scholar] [CrossRef] [Green Version]
- Narisawa, T.; Takahashi, M.; Kotanagi, H.; Kusaka, H.; Yamazaki, Y.; Koyama, H.; Fukaura, Y.; Nishizawa, Y.; Kotsugai, M.; Isoda, Y.; et al. Inhibitory Effect of Dietary Perilla Oil Rich in the n-3 Polyunsaturated Fatty Acid α-Linolenic Acid on Colon Carcinogenesis in Rats. Jpn. J. Cancer Res. 1991, 82, 1089–1096. [Google Scholar] [CrossRef]
- Lin, C.S.; Kuo, C.L.; Wang, J.P.; Cheng, J.S.; Huang, Z.W.; Chen, C.F. Growth inhibitory and apoptosis inducing effect of Perilla frutescens extract on human hepatoma HepG2 cells. J. Ethnopharmacol. 2007, 112, 557–567. [Google Scholar] [CrossRef]
- Wijendran, V.; Hayes, K.C. Dietary n-6 and n-3 fatty acid balance and cardiovascular health. Annu. Rev. Nutr. 2004, 24, 597–615. [Google Scholar] [CrossRef]
- Wang, F.; Zhu, H.; Hu, M.; Wang, J.; Xia, H.; Yang, X.; Yang, L.; Sun, G. Perilla Oil Supplementation Improves Hypertriglyceridemia and Gut Dysbiosis in Diabetic KKAy Mice. Mol. Nutr. Food Res. 2018, 62, 1800299. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Zhao, S.; Li, W.; Ma, L.; Ding, M.; Li, R.; Liu, Y. High-fat diet from perilla oil induces insulin resistance despite lower serum lipids and increases hepatic fatty acid oxidation in rats. Lipids Health Dis. 2014, 13, 15. [Google Scholar] [CrossRef] [Green Version]
- Paradee, N.; Utama-ang, N.; Uthaipibull, C.; Porter, J.B.; Garbowski, M.W.; Srichairatanakool, S. Extracts of Thai Perilla frutescens nutlets attenuate tumour necrosis factor-α-activated generation of microparticles, ICAM-1 and IL-6 in human endothelial cells. Biosci. Rep. 2020, 40, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, T.; Yasuda, T.; Ueda, J.; Ohsawa, K. Antidepressant-like effects of apigenin and 2,4,5-trimethoxycinnamic acid from Perilla frutescens in the forced swimming test. Biol. Pharm. Bull. 2003, 26, 474–480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takeda, H.; Tsuji, M.; Inazu, M.; Egashira, T.; Matsumiya, T. Rosmarinic acid and caffeic acid produce antidepressive-like effect in the forced swimming test in mice. Eur. J. Pharmacol. 2002, 449, 261–267. [Google Scholar] [CrossRef]
- Takeda, H.; Tsuji, M.; Miyamoto, J.; Matsumiya, T. Rosmarinic acid and caffeic acid reduce the defensive freezing behavior of mice exposed to conditioned fear stress. Psychopharmacology 2002, 164, 233–235. [Google Scholar] [CrossRef] [PubMed]
- Kamalashiran, C.; Pattaraarchachai, J.; Muengtaweepongsa, S. Feasibility and Safety of Perilla Seed Oil as an Additional Antioxidative Therapy in Patients with Mild to Moderate Dementia. J. Aging Res. 2018, 2018, 5302105. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Gou, Z.; Fan, Q.; Li, L.; Lin, X.; Wang, Y.; Jiang, S.; Jiang, Z. Effects of dietary perilla seed oil supplementation on lipid metabolism, meat quality, and fatty acid profiles in Yellow-feathered chickens. Poult. Sci. 2019, 98, 5714–5723. [Google Scholar] [CrossRef] [PubMed]
- Peiretti, P.G.; Gasco, L.; Brugiapaglia, A.; Gai, F. Effects of perilla (Perilla frutescens L.) seeds supplementation on performance, carcass characteristics, meat quality and fatty acid composition of rabbits. Livest. Sci. 2011, 138, 118–124. [Google Scholar] [CrossRef]
- Chung, C.-H.; Kim, J.-L.; Lee, Y.-C.; Choi, Y.-L. Cloning and Characterization of a Seed-Specific -3 Fatty Acid Desaturase cDNA from Perilla frutescens. Plant Cell Physiol. 1999, 40, 114–118. [Google Scholar] [CrossRef] [Green Version]
- Hwang, S.K.; Hwang, Y.S. Molecular cloning and functional expression of Perilla frutescens 3-ketoacyl-(acyl carrier protein) synthase III. Mol. Cells 2000, 10, 375–381. [Google Scholar] [CrossRef]
- Hwang, S.K.; Kim, K.H.; Hwang, Y.S. Molecular cloning and expression analysis of 3-ketoacyl-ACP synthases in the immature seeds of Perilla frutescens. Mol. Cells 2000, 10, 533–539. [Google Scholar] [CrossRef]
- Lee, K.-R.; Lee, Y.; Kim, E.-H.; Lee, S.-B.; Roh, K.H.; Kim, J.-B.; Kang, H.-C.; Kim, H.U. Functional identification of oleate 12-desaturase and ω-3 fatty acid desaturase genes from Perilla frutescens var. frutescens. Plant Cell Rep. 2016, 35, 2523–2537. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Chen, B.; Win, A.N.; Fu, C.; Lian, J.; Liu, X.; Wang, R.; Zhang, X.; Chai, Y. Omega-3 fatty acid desaturase gene family from two ω-3 sources, Salvia hispanica and Perilla frutescens: Cloning, characterization and expression. PLoS ONE 2018, 13, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Liao, B.N.; Hao, Y.J.; Lu, J.X.; Bai, H.Y.; Guan, L.; Zhang, T. Transcriptomic analysis of Perilla frutescens seed to insight into the biosynthesis and metabolic of unsaturated fatty acids. BMC Genom. 2018, 19, 213. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Song, C.; Song, L.; Shang, Z.; Yang, S.; Zhang, D.; Sun, W.; Shen, Q.; Zhao, D. RNA sequencing and coexpression analysis reveal key genes involved in α-linolenic acid biosynthesis in Perilla frutescens seed. Int. J. Mol. Sci. 2017, 18, 2433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.U.; Lee, K.R.; Shim, D.; Lee, J.H.; Chen, G.Q.; Hwang, S. Transcriptome analysis and identification of genes associated with ω-3 fatty acid biosynthesis in Perilla frutescens (L.) var. frutescens. BMC Genom. 2016, 17, 474. [Google Scholar] [CrossRef] [Green Version]
- Fukushima, A.; Nakamura, M.; Suzuki, H.; Saito, K.; Yamazaki, M. High-throughput sequencing and de novo assembly of red and green forms of the Perilla frutescens var. crispa transcriptome. PLoS ONE 2015, 10, e0129154. [Google Scholar] [CrossRef] [Green Version]
- Duan, W.; Shi-Mei, Y.; Zhi-Wei, S.; Jing, X.; De-Gang, Z.; Hong-Bin, W.; Qi, S. Genome-Wide Analysis of the Fatty Acid Desaturase Gene Family Reveals the Key Role of PfFAD3 in α-Linolenic Acid Biosynthesis in Perilla Seeds. Front. Genet. 2021, 12, 735862. [Google Scholar] [CrossRef]
- Liping, W.; Shen, W.; Kazachkov, M.; Chen, G.; Chen, Q.; Carlsson, A.S.; Stymne, S.; Weselake, R.J.; Zou, J. Metabolic interactions between the lands cycle and the kennedy pathway of glycerolipid synthesis in arabidopsis developing seeds. Plant Cell 2012, 24, 4652–4669. [Google Scholar] [CrossRef] [Green Version]
- Bates, P.D.; Fatihi, A.; Snapp, A.R.; Carlsson, A.S.; Browse, J.; Lu, C. Acyl editing and headgroup exchange are the major mechanisms that direct polyunsaturated fatty acid flux into triacylglycerols. Plant Physiol. 2012, 160, 1530–1539. [Google Scholar] [CrossRef] [Green Version]
- Konishi, T.; Shinohara, K.; Yamada, K.; Sasaki, Y. Acetyl-CoA Carboxylase in Higher Plants: Most Plants Other Than Gramineae Have Both the Prokaryotic and the Eukaryotic Forms of This Enzyme. Plant Cell Physiol. 1996, 37, 117–122. [Google Scholar] [CrossRef] [Green Version]
- Jung, S.H.; Kim, R.J.; Kim, K.J.; Lee, D.H.; Suh, M.C. Plastidial and mitochondrial malonyl CoA-ACP malonyltransferase is essential for cell division and its overexpression increases storage oil content. Plant Cell Physiol. 2019, 60, 1239–1249. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Munz, J.; Cass, C.; Zienkiewicz, A.; Kong, Q.; Ma, W.; Sanjaya, S.; Sedbrook, J.C.; Benning, C. Ectopic expression of WRI1 affects fatty acid homeostasis in Brachypodium distachyon vegetative tissues. Plant Physiol. 2015, 169, 1836–1847. [Google Scholar] [CrossRef] [Green Version]
- An, D.; Kim, H.; Ju, S.; Go, Y.S.; Kim, H.U.; Suh, M.C. Expression of Camelina WRINKLED1 Isoforms Rescue the Seed Phenotype of the Arabidopsis wri1 Mutant and Increase the Triacylglycerol Content in Tobacco Leaves. Front. Plant Sci. 2017, 8, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grimberg, Å.; Carlsson, A.S.; Marttila, S.; Bhalerao, R.; Hofvander, P. Transcriptional transitions in Nicotiana benthamiana leaves upon induction of oil synthesis by WRINKLED1 homologs from diverse species and tissues. BMC Plant Biol. 2015, 15, 192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, R.; Ye, R.; Gao, L.; Zhang, L.; Wang, R.; Mao, T.; Zheng, Y.; Li, D.; Lin, Y. Characterization and Ectopic Expression of CoWRI1, an AP2/EREBP Domain-Containing Transcription Factor from Coconut (Cocos nucifera L.) Endosperm, Changes the Seeds Oil Content in Transgenic Arabidopsis thaliana and Rice (Oryza sativa L.). Front. Plant Sci. 2017, 8, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Hua, W.; Zhan, G.; Wei, F.; Wang, X.; Liu, G.; Wang, H. Increasing seed mass and oil content in transgenic Arabidopsis by the overexpression of wri1-like gene from Brassica napus. Plant Physiol. Biochem. 2010, 48, 9–15. [Google Scholar] [CrossRef]
- Ma, W.; Kong, Q.; Arondel, V.; Kilaru, A.; Bates, P.D.; Thrower, N.A.; Benning, C.; Ohlrogge, J.B. WRINKLED1, A Ubiquitous Regulator in Oil Accumulating Tissues from Arabidopsis Embryos to Oil Palm Mesocarp. PLoS ONE 2013, 8, e68887. [Google Scholar] [CrossRef]
- Yang, H.; Yu, C.; Yan, J.; Wang, X.; Chen, F.; Zhao, Y.; Wei, W. Overexpression of the Jatropha curcas JcERF1 gene coding an AP2/ERF-Type transcription factor increases tolerance to salt in transgenic tobacco. Biochemistry 2014, 79, 1226–1236. [Google Scholar] [CrossRef]
- Baud, S.; Mendoza, M.S.; To, A.; Harscoët, E.; Lepiniec, L.; Dubreucq, B. WRINKLED1 specifies the regulatory action of LEAFY COTYLEDON2 towards fatty acid metabolism during seed maturation in Arabidopsis. Plant J. 2007, 50, 825–838. [Google Scholar] [CrossRef]
- Fukuda, N.; Ikawa, Y.; Aoyagi, T.; Kozaki, A. Expression of the genes coding for plastidic acetyl-CoA carboxylase subunits is regulated by a location-sensitive transcription factor binding site. Plant Mol. Biol. 2013, 82, 473–483. [Google Scholar] [CrossRef] [Green Version]
- Kazaz, S.; Barthole, G.; Domergue, F.; Ettaki, H.; To, A.; Vasselon, D.; de Vos, D.; Belcram, K.; Lepiniec, L.; Baud, S. Differential activation of partially redundant Δ9 stearoyl-ACP desaturase genes is critical for omega-9 monounsaturated fatty acid biosynthesis during seed development in arabidopsis. Plant Cell 2020, 32, 3613–3637. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Shao, J.; Tang, S.; Shen, Q.; Wang, T.; Chen, W.; Hong, Y. Wrinkled1 accelerates flowering and regulates lipid homeostasis between oil accumulation and membrane lipid anabolism in Brassica napus. Front. Plant Sci. 2015, 6, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Zhai, Z.; Kuczynski, K.; Keereetaweep, J.; Schwender, J.; Shanklin, J. Wrinkled1 regulates biotin attachment domain-containing proteins that inhibit fatty acid synthesis. Plant Physiol. 2019, 181, 55–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maeo, K.; Tokuda, T.; Ayame, A.; Mitsui, N.; Kawai, T.; Tsukagoshi, H.; Ishiguro, S.; Nakamura, K. An AP2-type transcription factor, WRINKLED1, of Arabidopsis thaliana binds to the AW-box sequence conserved among proximal upstream regions of genes involved in fatty acid synthesis. Plant J. 2009, 60, 476–487. [Google Scholar] [CrossRef]
- Pouvreau, B.; Baud, S.; Vernoud, V.; Morin, V.; Py, C.; Gendrot, G.; Pichon, J.P.; Rouster, J.; Paul, W.; Rogowsky, P.M. Duplicate maize wrinkled1 transcription factors activate target genes involved in seed oil biosynthesis. Plant Physiol. 2011, 156, 674–686. [Google Scholar] [CrossRef] [Green Version]
- Ruuska, S.A.; Girke, T.; Benning, C.; Ohlrogge, J.B. Contrapuntal networks of gene expression during Arabidopsis seed fillingW. Plant Cell 2002, 14, 1191–1206. [Google Scholar] [CrossRef] [Green Version]
- Shen, B.; Allen, W.B.; Zheng, P.; Li, C.; Glassman, K.; Ranch, J.; Nubel, D.; Tarczynski, M.C. Expression of ZmLEC1 and ZmWRI1 increases seed oil production in maize. Plant Physiol. 2010, 153, 980–987. [Google Scholar] [CrossRef] [Green Version]
- Browse, J.; McConn, M.; James, D.; Miquel, M. Mutants of Arabidopsis deficient in the synthesis of α-linolenate. Biochemical and genetic characterization of the endoplasmic reticulum linoleoyl desaturase. J. Biol. Chem. 1993, 268, 16345–16351. [Google Scholar] [CrossRef]
- Okuley, J.; Lightner, J.; Feldmann, K.; Yadav, N.; Lark, E.; Browse, J. Arabidopsis FAD2 gene encodes the enzyme that is essential for polyunsaturated lipid synthesis. Plant Cell 1994, 6, 147–158. [Google Scholar] [CrossRef]
- Speerling, P.; Heinz, E. Isomeric sn-1-octadecenyl and sn-2-octadecenyl analogues of lysophosphatidylcholine as substrates for acylation and desaturation by plant microsomal membranes. Eur. J. Biochem. 1993, 213, 965–971. [Google Scholar] [CrossRef]
- Chen, J.; Tan, R.K.; Guo, X.J.; Fu, Z.L.; Wang, Z.; Zhang, Z.Y.; Tan, X.L. Transcriptome analysis comparison of lipid biosynthesis in the leaves and developing seeds of Brassica napus. PLoS ONE 2015, 10, e0130067. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Kazachkov, M.; Shen, W.; Bai, M.; Wu, H.; Zou, J. Deciphering the roles of Arabidopsis LPCAT and PAH in phosphatidylcholine homeostasis and pathway coordination for chloroplast lipid synthesis. Plant J. 2014, 80, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Pérez, A.J.; Santos-Pereira, J.M.; Martins-Noguerol, R.; DeAndrés-Gil, C.; Troncoso-Ponce, M.A.; Venegas-Calerón, M.; Sánchez, R.; Garcés, R.; Salas, J.J.; Tena, J.J.; et al. Genome-Wide Mapping of Histone H3 Lysine 4 Trimethylation (H3K4me3) and Its Involvement in Fatty Acid Biosynthesis in Sunflower Developing Seeds. Plants 2021, 10, 706. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, C.; Liu, X.; Cheng, J.; Li, S.; Zhu, J.K.; Gong, Z. Peroxisomal β-oxidation regulates histone acetylation and DNA methylation in Arabidopsis. Proc. Natl. Acad. Sci. USA 2019, 116, 10576–10585. [Google Scholar] [CrossRef] [Green Version]
- Yamazaki, M.; Kobayashi, M.; Saito, K. Transformation of Perill frutescens var. crispa Using an Agrobacterium-Ri Binary Vector System. Plant Biotechnol. 1997, 14, 169–173. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.-H.; Lee, Y.-H.; Kim, D.; Park, Y.-H.; Lee, J.-Y.; Hwang, Y.-S.; Kim, Y.-H. Agrobacterium-mediated genetic transformation of Perilla frutescens. Plant Cell Rep. 2004, 23, 386–390. [Google Scholar] [CrossRef]
- Dolgin, E. T-cell vaccines could top up immunity to COVID, as variants loom large. Nat. Biotechnol. 2022, 40, 3–4. [Google Scholar] [CrossRef]
- Schwartz, C.; Lenderts, B.; Feigenbutz, L.; Barone, P.; Llaca, V.; Fengler, K.; Svitashev, S. CRISPR–Cas9-mediated 75.5-Mb inversion in maize. Nat. Plants 2020, 6, 1427–1431. [Google Scholar] [CrossRef]
- Kelliher, T.; Starr, D.; Su, X.; Tang, G.; Chen, Z.; Carter, J.; Wittich, P.E.; Dong, S.; Green, J.; Burch, E.; et al. One-step genome editing of elite crop germplasm during haploid induction. Nat. Biotechnol. 2019, 37, 287–292. [Google Scholar] [CrossRef]
- Svitashev, S.; Schwartz, C.; Lenderts, B.; Young, J.K.; Mark Cigan, A. Genome editing in maize directed by CRISPR-Cas9 ribonucleoprotein complexes. Nat. Commun. 2016, 7, 13274. [Google Scholar] [CrossRef]
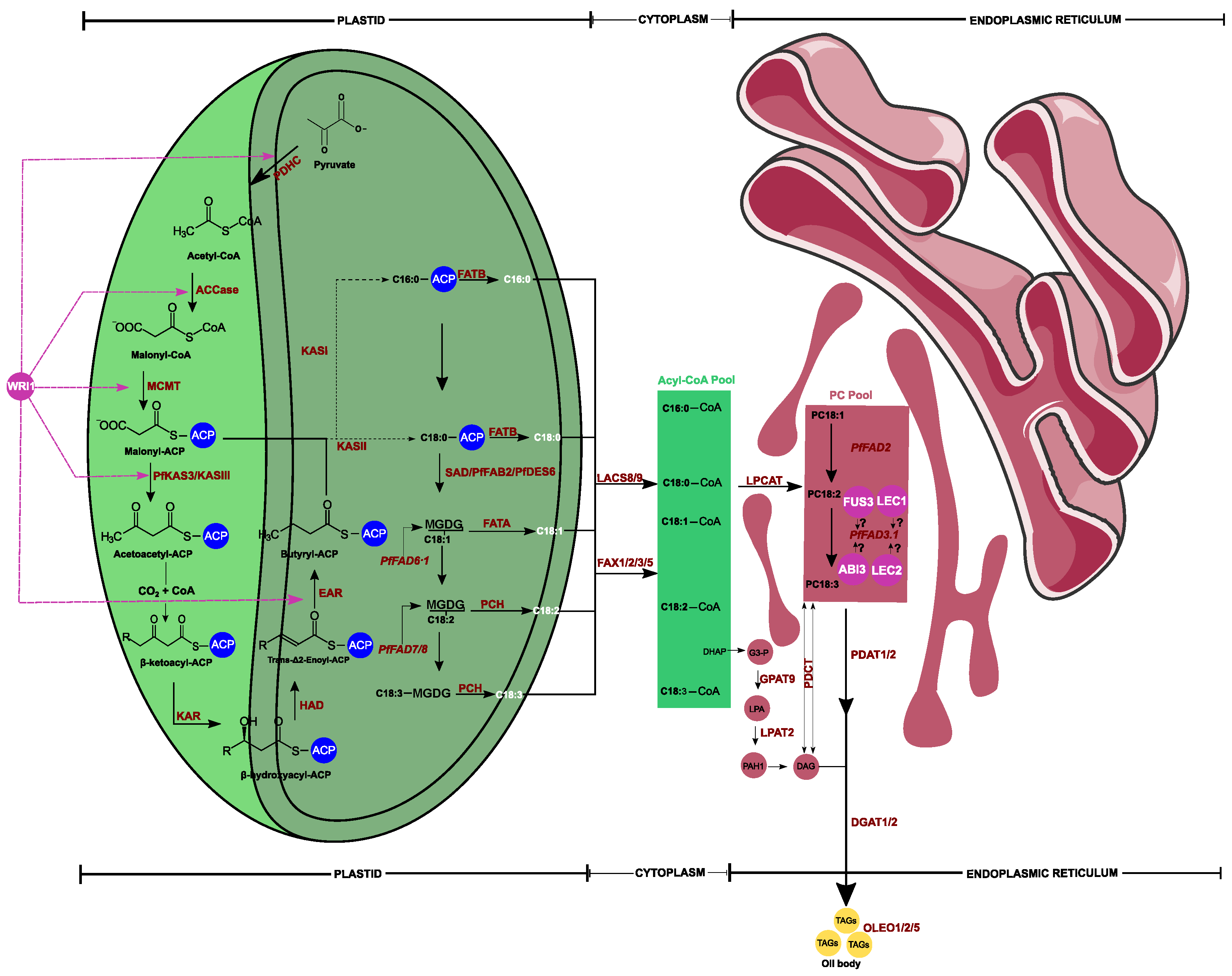
| Enzyme ID | Enzyme Name | GeneID | Homologous | Pathways Involved | Field of Study | References | ||
|---|---|---|---|---|---|---|---|---|
| PF40 * | Dayudeulkkae ** | PC *** | A. Thaliana | |||||
| PDH(E1α) | Pyruvate Dehydrogenase E1 Subunit Alpha 1 | Locus_2112 | AT1G01090.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| PDH(E1ß) | Pyruvate Dehydrogenase E1 Subunit beta 1 | Locus_25208 | AT2G34590.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| EMB3003(E2) | Pyruvate dehydrogenase e2 component (dihydrolipoamide acetyltransferase) | Locus_33306 | AT1G34430.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| LTA2 (E2) | Plastid E2 Subunit of Pyruvate Decarboxylase, PLE2 | Locus_5104 | AT3G25860.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| LPD1 (E3) | Lipoamide dehydrogenase | Locus_7407 | AT3G16950.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| α-CTa | Alpha-carboxyltransferase Isoform a | Locus_8492 | AT2G38040.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| α-CTb | Apha-carboxyltransferase Isoform b | Locus_2178 | AT2G38040.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| ß-CT | Beta-carboxyltransferase | Locus_53041 | ATCG00500.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| BC | Biotin carboxylase | Locus_22078 | AT5G35360.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| BCCP1 | Biotin carboxyl carrier protein of acetyl-CoA carboxylase 1 | Locus_29162 | AT5G16390.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| BCCP2 | Biotin carboxyl carrier protein of acetyl-CoA carboxylase 2 | Locus_17340 | AT5G15530.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| MCMT | Malonyl-CoA ACP transacylase | Locus_14579 | AT2G30200.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| KASIII | 3-Ketoacyl-ACP synthase | Locus_10821 | AT1G62640.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| KAR | 3-ketoacyl-ACP reductase | Locus_1445 | AT1G24360.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| HAD | 3-hydroxyacyl-ACP dyhydratase | Locus_19332 | AT5G10160.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| EAR | 2-enoyl-ACP reductase | Locus_25443 | AT2G05990.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| FATA | Fatty acyl-ACP thioesterase A | Locus_29919 | AT3G25110.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| FATB | Fatty acyl-ACP thioesterase B | Locus_6603 | AT1G08510.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| FAB2 | Fatty acid biosynthesis2 | Locus_13564 | AT2G43710.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| DES6 | Stearoyl-acyl carrier protein desaturase | Locus_9486 | AT1G43800.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| KASI | Ketoacyl-ACP Synthase I | Locus_26341 | AT5G46290.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| KASII | Ketoacyl-ACP Synthase II | Locus_1373 | AT1G74960.1 | FA de novo biosynthesis and export from plastid | Transcriptomics | [25] | ||
| LACS8 | Long-chain acyl-CoA synthetase 8 | chr07_36292788_36299197 chr19_22302145_22308533 | Locus_3838 | chr06_37084362_37090768 | AT2G04350.1 | FA de novo biosynthesis and export from plastid | Genome Assembly, Transcriptomics | [3,25] |
| LACS9 | Long-chain acyl-CoA synthetase 9 | chr03_70622879_70627324 chr09_58852417_58856892 chr01_02424545_02428997 | Locus_23636 | chr01_02424545_02428997 | AT1G77590.1 | FA de novo biosynthesis and export from plastid | Genome Assembly, Transcriptomics | [3,25] |
| FAX1 | Fatty acid export 1 | chr05_24282740_24284950 chr01_71691539_71693779 | chr02_42552603_42554830 | FA de novo biosynthesis and export from plastid | [25] | |||
| FAX2 | Fatty acid export 2 | chr07_10626150_10628000 | chr06_11381976_11383822 | FA de novo biosynthesis and export from plastid | [25] | |||
| FAX3 | Fatty acid export 3 | chr04_00857340_00859552 | chr03_67540865_67543081 | FA de novo biosynthesis and export from plastid | [25] | |||
| FAX5 | Fatty acid export 5 | chr04_65527957_65529911 chr07_22534802_22537586 chr06_00746938_00748860 chr19_10735560_10738363 | chr03_02347871_02349825 chr06_23562111_23564893 | FA de novo biosynthesis and export from plastid | [25] | |||
| FAD2 | Omega-6 fatty acid desaturase | chr12_56933298_56934446 chr11_05592060_05593208 chr11_05575254_05576393 | Locus_733 | chr08_55538081_55539229 | AT3G12120.1 | Acyl editing of phospatidylcholine | Genome Assembly, Transcriptomics | [3,25] |
| chr12_56948107_56949167 | chr08_55558209_55559348 | |||||||
| FAD3 | Omega-3 fatty acid desaturase | chr12_04645208_04647776 chr11_54194712_54197265 | Locus_22029 | chr08_04030082_04032640 | AT2G29980.1 | Acyl editing of phospatidylcholine | Genome Assembly, Transcriptomics | [3,25] |
| FAD8 | Omega-8 fatty acid desaturase | Locus_5107 | AT5G05580.2 | Acyl editing of phospatidylcholine | Transcriptomics | [25] | ||
| GPAT9 | Glycerol-3-phosphate acyltransferase 9 | chr12_33733527_33737891 chr11_26255533_26259881 | Locus_10180 | chr08_33038421_33042132 | AT5G60620.1 | Acyl-CoA-dependent TAG synthesis in Kennedy pathway | Genome Assembly, Transcriptomics | [3,25] |
| LPAT2 | 1-acyl-sn-glycerol-3-phosphate acyltransferase 2 | chr05_23583386_23588593 chr05_34400913_34404444 chr01_72114246_72119454 | Locus_6587 | chr02_43313059_43318262 chr02_32585727_32589258 | AT3G57650.1 | Acyl-CoA-dependent TAG synthesis in Kennedy pathway | Genome Assembly, Transcriptomics | [3,25] |
| PAH1 | Phenylalanine hydrolase 1 | chr01_61567423_61570965 chr14_08597119_08602056 chr15_37103964_37108907 chr03_61656532_61661875 chr18_09154357_09159306 chr17_34575710_34580664 chr09_50343045_50349360 | chr10_43830659_43835596 chr01_11516392_11522733 | Acyl-CoA-dependent TAG synthesis in Kennedy pathway | ||||
| DGAT1 | Diacylglycerol O-acyltransferase 1 | chr01_09730655_09741367 chr01_48275733_48286173 | Locus_14696 | chr05_08797620_08808333 | AT2G19450.1 | Acyl-CoA-dependent TAG synthesis in Kennedy pathway | Genome Assembly, Transcriptomics | [3,25] |
| DGAT2 | Diacylglycerol O-acyltransferase 2 | chr14_26782964_26787941 chr18_25811826_25816791 | Locus_12629 | chr10_25785382_25790335 | AT3G51520.1 | Acyl-CoA-dependent TAG synthesis in Kennedy pathway | Genome Assembly, Transcriptomics | [3,25] |
| DGAT3 | Diacylglycerol O-acyltransferase 3 | Locus_1560 | AT1G48300.1 | Acyl-CoA-dependent TAG synthesis in Kennedy pathway | Transcriptomics | [25] | ||
| LPCAT | Lysophosphatidylcholine acyltransferase | chr01_06996630_07001595 chr05_56678891_56685081 chr01_03079195_03084058 chr07_53028425_53034567 chr01_43224061_43229071 chr02_66141068_66147271 chr02_04634020_04638876 chr19_35211932_35217537 | Locus_43749 | PC00000058_00436672_00441634 chr02_10454190_10460391 chr05_03185967_03190829 chr06_54113419_54119561 | AT1G12640.1 | PC-mediated TAG synthesis | Transcriptomics | [3,25] |
| CPT1 | Diacylglycerol cholinephosphotransferase | Locus_7821 | AT1G13560.1 | PC-mediated TAG synthesis | Transcriptomics | [25] | ||
| CPT2 | Diacylglycerol cholinephosphotransferase | Locus_22567 | AT3G25585.1 | PC-mediated TAG synthesis | Transcriptomics | [25] | ||
| PDAT1 | Phospholipid:diacylglycerol acyltransferase 1 | chr05_44104376_44108847 | Locus_7255 | chr02_22969948_22974420 | AT5G13640.1 | Acyl-CoA independent pathway | Transcriptomics | [3,25] |
| chr03_00447151_00451507 | PC00002899_00154872_00159184 | |||||||
| chr02_52135886_52140327 | ||||||||
| chr09_00376677_00380564 | ||||||||
| PDAT2 | Phospholipid:diacylglycerol acyltransferase 2 | chr05_38922115_38924735 | Locus_29208 | chr02_28050267_28052887 | AT3G44830.1 | Acyl-CoA independent pathway | Transcriptomics | [3,25] |
| chr02_45992086_45994691 | ||||||||
| PDCT | Phosphatidylcholine:diacylglycerol cholinephosphotransferase | chr03_46291224_46293449 chr09_37050943_37053194 | Locus_15867 | chr01_27228085_27230144 | AT3G15820.1 | Acyl-CoA independent pathway | Genome Assembly, Transcriptomics | [3,25] |
| OLEO2 | Oleosin2 | chr15_52133834_52134256 | Locus_31790 | AT5G40420.1 | TAG assembly | Transcriptomics | [3,25] | |
| chr17_50355018_50355440 | chr09_02008310_02008732 | |||||||
| OLEO | Oleosin | chr14_08347244_08347714 | Locus_31788 | chr10_44101965_44102435 | AT3G18570.1 | TAG assembly | Transcriptomics | [3,25] |
| chr18_08871500_08871970 | ||||||||
| OLEO1 | Oleosin1 | chr05_05196095_05196523 | Locus_29266 | chr02_64426568_64426996 | AT4G25140.1 | TAG assembly | Transcriptomics | [3,25] |
| chr01_30156121_30156549 | ||||||||
| OLEO5 | Oleosin5 | chr05_59989345_59989911 | Locus_29276 | AT3G01570.1 | TAG assembly | Transcriptomics | [3,25] | |
| chr05_59997449_59997976 | chr02_07157257_07157823 | |||||||
| chr02_69562819_69563393 | chr02_07149192_07149719 | |||||||
| chr02_69577662_69578195 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bae, S.-H.; Zoclanclounon, Y.A.B.; Kumar, T.S.; Oh, J.-H.; Lee, J.; Kim, T.-H.; Park, K.Y. Advances in Understanding the Genetic Basis of Fatty Acids Biosynthesis in Perilla: An Update. Plants 2022, 11, 1207. https://doi.org/10.3390/plants11091207
Bae S-H, Zoclanclounon YAB, Kumar TS, Oh J-H, Lee J, Kim T-H, Park KY. Advances in Understanding the Genetic Basis of Fatty Acids Biosynthesis in Perilla: An Update. Plants. 2022; 11(9):1207. https://doi.org/10.3390/plants11091207
Chicago/Turabian StyleBae, Seon-Hwa, Yedomon Ange Bovys Zoclanclounon, Thamilarasan Senthil Kumar, Jae-Hyeon Oh, Jundae Lee, Tae-Ho Kim, and Ki Young Park. 2022. "Advances in Understanding the Genetic Basis of Fatty Acids Biosynthesis in Perilla: An Update" Plants 11, no. 9: 1207. https://doi.org/10.3390/plants11091207









