Stomatal Ratio Showing No Response to Light Intensity in Oryza
Abstract
1. Introduction
2. Results
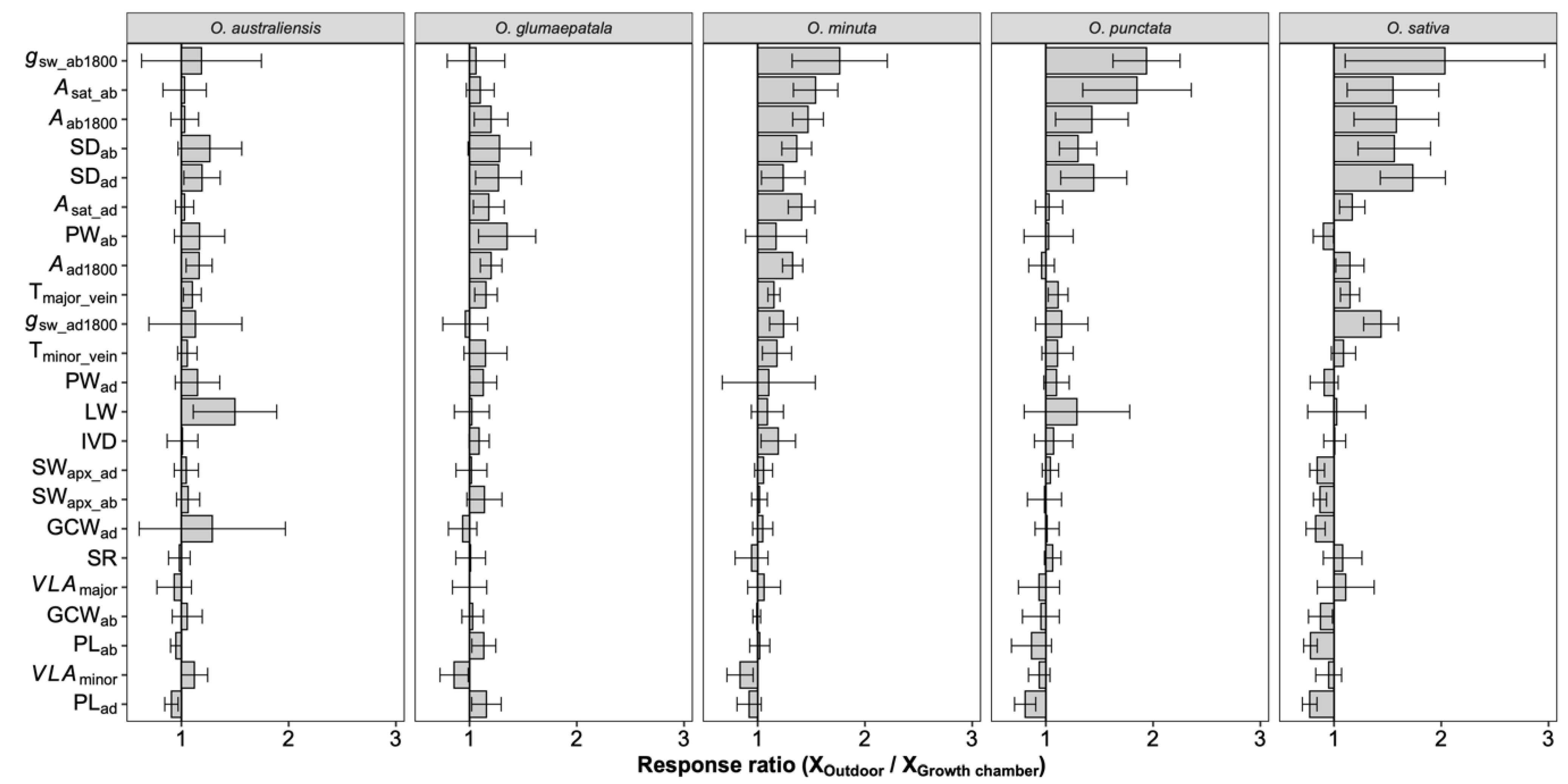
2.1. Variation across Species in Leaf Traits
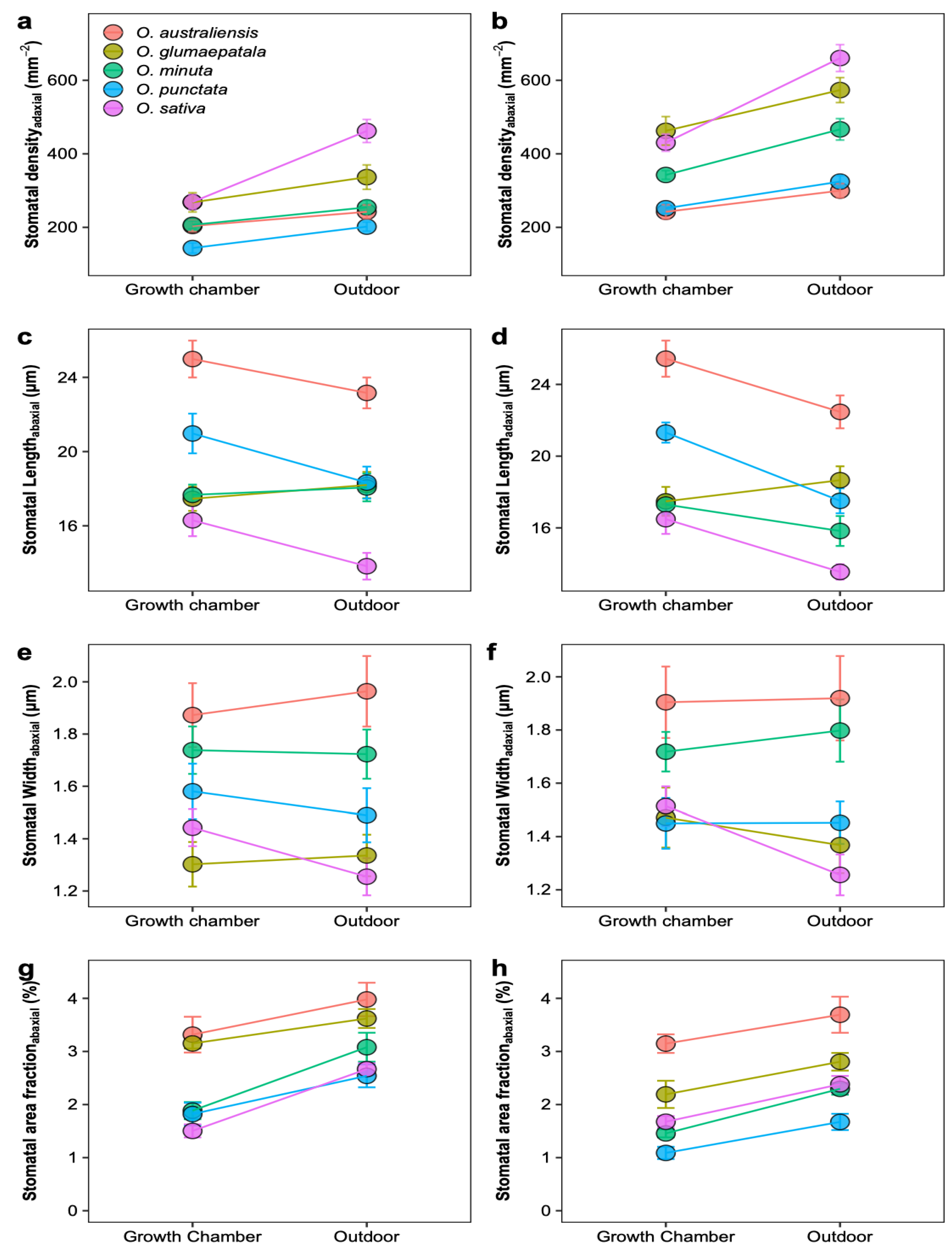
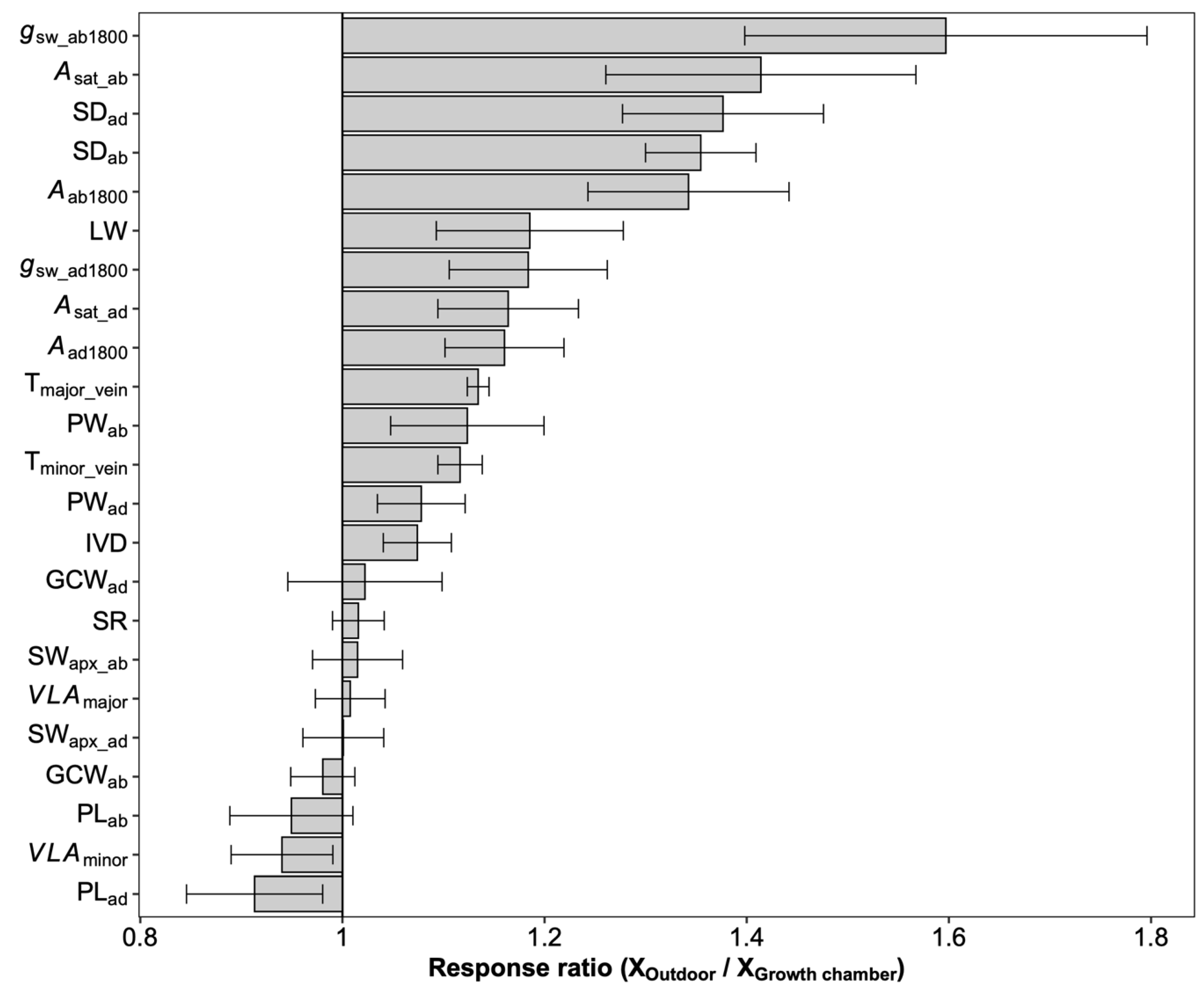
2.2. Plasticity across Growth Conditions in Leaf Traits
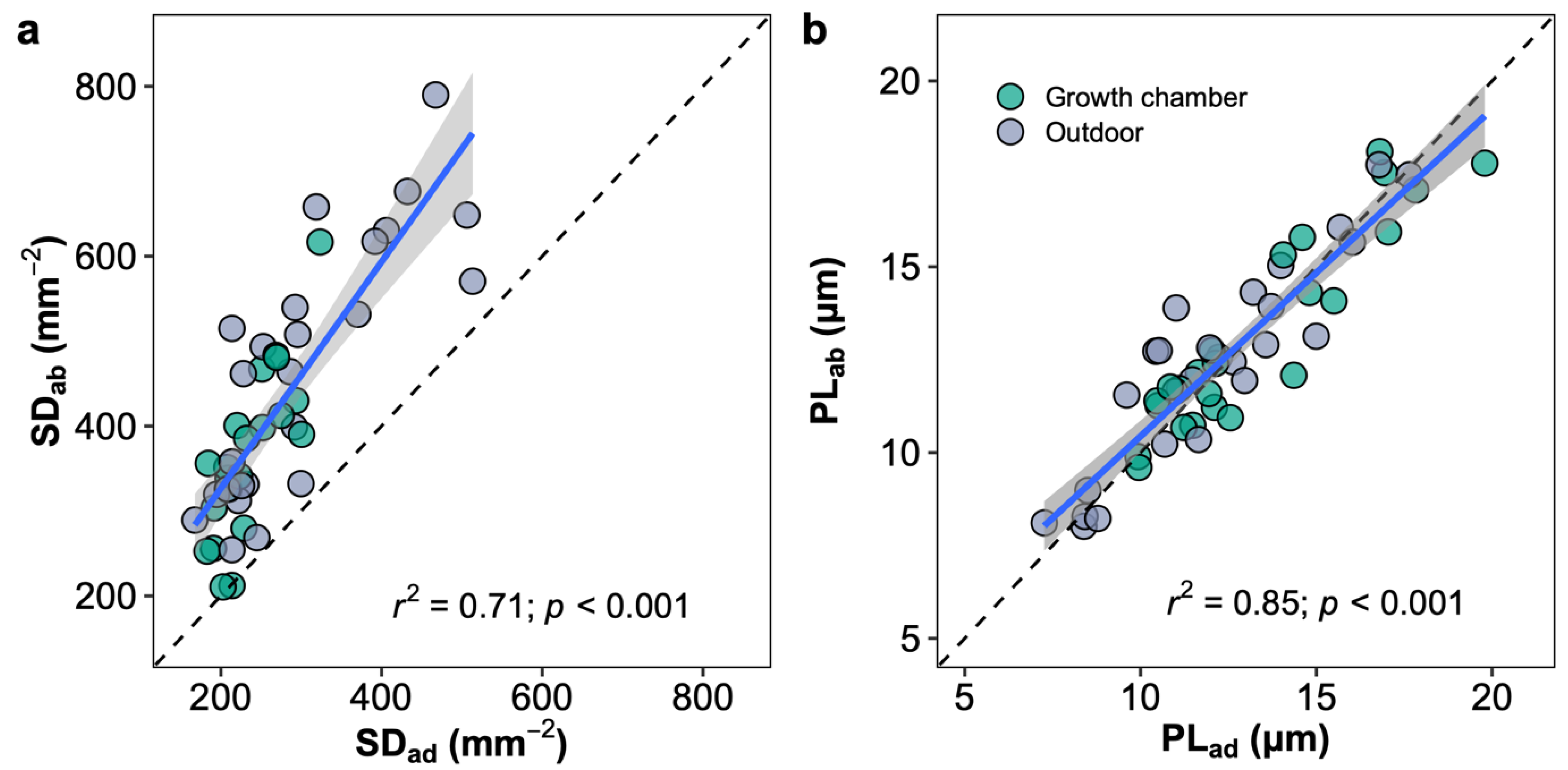
2.3. Upper versus Lower Leaf Surface
3. Discussion
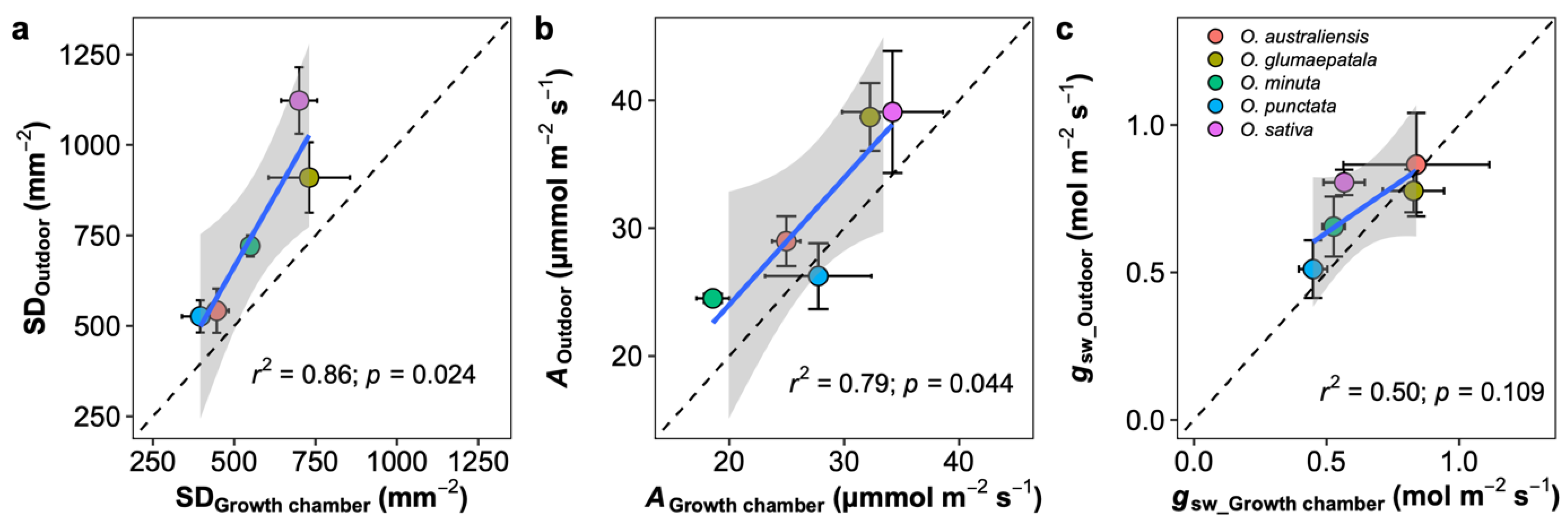
3.1. Stomatal Density Variation among Species
3.2. Responses of Stomatal Density, Size, and Ratio to Growth Conditions
3.3. Growth Chamber versus Outdoor
4. Materials and Methods
4.1. Plant Material and Growth
4.2. Light Response Curves
4.3. Stomatal Density and Size Measurements
4.4. Leaf Anatomical Traits
4.5. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Doheny-Adams, T.; Hunt, L.; Franks, P.J.; Beerling, D.J.; Gray, J.E. Genetic manipulation of stomatal density influences stomatal size, plant growth and tolerance to restricted water supply across a growth carbon dioxide gradient. Philos. Trans. R. Soc. B-Biol. Sci. 2012, 367, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Faralli, M.; Matthews, J.; Lawson, T. Exploiting natural variation and genetic manipulation of stomatal conductance for crop improvement. Curr. Opin. Plant Biol. 2019, 49, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Xiong, D.; Flexas, J. From one side to two sides: The effects of stomatal distribution on photosynthesis. New Phytol. 2020, 228, 1754–1766. [Google Scholar] [CrossRef]
- Harrison, E.L.; Arce Cubas, L.; Gray, J.E.; Hepworth, C. The influence of stomatal morphology and distribution on photosynthetic gas exchange. Plant J. 2020, 101, 768–779. [Google Scholar] [CrossRef] [PubMed]
- Franks, P.J.; Farquhar, G.D. The mechanical diversity of stomata and its significance in gas-exchange control. Plant Physiol. 2007, 143, 78–87. [Google Scholar] [CrossRef]
- Xiong, D.; Douthe, C.; Flexas, J. Differential coordination of stomatal conductance, mesophyll conductance, and leaf hydraulic conductance in response to changing light across species. Plant Cell Environ. 2018, 41, 436–450. [Google Scholar] [CrossRef]
- Baillie, A.L.; Fleming, A.J. The developmental relationship between stomata and mesophyll airspace. New Phytol. 2020, 225, 1120–1126. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Sugano, S.S.; Shimada, T.; Hara-Nishimura, I. Enhancement of leaf photosynthetic capacity through increased stomatal density in Arabidopsis. New Phytol. 2013, 198, 757–764. [Google Scholar] [CrossRef]
- Franks, P.J.; Doheny-Adams, T.W.; Britton-Harper, Z.J.; Gray, J.E. Increasing water-use efficiency directly through genetic manipulation of stomatal density. New Phytol. 2015, 207, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Buckley, C.R.; Caine, R.S.; Gray, J.E. Pores for Thought: Can Genetic Manipulation of Stomatal Density Protect Future Rice Yields? Front. Plant Sci. 2020, 10, 1783. [Google Scholar] [CrossRef]
- Drake, P.L.; Froend, R.H.; Franks, P.J. Smaller, faster stomata: Scaling of stomatal size, rate of response, and stomatal conductance. J. Exp. Bot. 2013, 64, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Milla, R.; de Diego-Vico, N.; Martin-Robles, N. Shifts in stomatal traits following the domestication of plant species. J. Exp. Bot. 2013, 64, 3137–3146. [Google Scholar] [CrossRef]
- Lawson, T.; Blatt, M.R. Stomatal Size, Speed, and Responsiveness Impact on Photosynthesis and Water Use Efficiency. Plant Physiol. 2014, 164, 1556–1570. [Google Scholar] [CrossRef] [PubMed]
- Lawson, T.; Matthews, J. Guard Cell Metabolism and Stomatal Function. Annu. Rev. Plant Biol. 2020, 71, 273–302. [Google Scholar] [CrossRef] [PubMed]
- Becklin, K.M.; Ward, J.K.; Way, D.A. Photosynthesis, Respiration, and Climate Change; Springer: Cham, Switzerland, 2021. [Google Scholar]
- Casson, S.; Gray, J.E. Influence of environmental factors on stomatal development. New Phytol. 2008, 178, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Casson, S.A.; Hetherington, A.M. Environmental regulation of stomatal development. Curr. Opin. Plant Biol. 2010, 13, 90–95. [Google Scholar] [CrossRef]
- Amitrano, C.; Arena, C.; Rouphael, Y.; De Pascale, S.; De Micco, V. Vapour pressure deficit: The hidden driver behind plant morphofunctional traits in controlled environments. Ann. Appl. Biol. 2019, 175, 313–325. [Google Scholar] [CrossRef]
- Kumar, S.; Tripathi, S.; Singh, S.P.; Prasad, A.; Akter, F.; Abu Syed, M.; Badri, J.; Das, S.P.; Bhattarai, R.; Natividad, M.A.; et al. Rice breeding for yield under drought has selected for longer flag leaves and lower stomatal density. J. Exp. Bot. 2021, 72, 4981–4992. [Google Scholar] [CrossRef]
- Konrad, W.; Roth-Nebelsick, A.; Grein, M. Modelling of stomatal density response to atmospheric CO2. J. Theor. Biol. 2008, 253, 638–658. [Google Scholar] [CrossRef]
- Ohsumi, A.; Kanemura, T.; Homma, K.; Horie, T.; Shiraiwa, T. Genotypic variation of stomatal conductance in relation to stomatal density and length in rice (Oryza sativa L.). Plant Prod. Sci. 2007, 10, 322–328. [Google Scholar] [CrossRef]
- Nunes, T.D.G.; Zhang, D.; Raissig, M.T. Form, development and function of grass stomata. Plant J. 2020, 101, 780–799. [Google Scholar] [CrossRef] [PubMed]
- Paradiso, R.; de Visser, P.H.B.; Arena, C.; Marcelis, L.F.M. Light response of photosynthesis and stomatal conductance of rose leaves in the canopy profile: The effect of lighting on the adaxial and the abaxial sides. Funct. Plant Biol. 2020, 47, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Drake, P.L.; de Boer, H.J.; Schymanski, S.J.; Veneklaas, E.J. Two sides to every leaf: Water and CO2 transport in hypostomatous and amphistomatous leaves. New Phytol. 2019, 222, 1179–1187. [Google Scholar] [CrossRef]
- Muir, C.D. Light and growth form interact to shape stomatal ratio among British angiosperms. New Phytol. 2018, 218, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Mott, K.A.; Peak, D. Effects of the mesophyll on stomatal responses in amphistomatous leaves. Plant Cell Environ. 2018, 41, 2835–2843. [Google Scholar] [CrossRef]
- Lake, J.A.; Woodward, F.I.; Quick, W.P. Long-distance CO2 signalling in plants. J. Exp. Bot. 2002, 53, 183–193. [Google Scholar] [CrossRef]
- Richardson, F.; Jordan, G.J.; Brodribb, T.J. Leaf hydraulic conductance is linked to leaf symmetry in bifacial, amphistomatic leaves of sunflower. J. Exp. Bot. 2020, 71, 2808–2816. [Google Scholar] [CrossRef]
- Wall, S.; Vialet-Chabrand, S.; Davey, P.; Van Rie, J.; Galle, A.; Cockram, J.; Lawson, T. Stomata on the abaxial and adaxial leaf surfaces contribute differently to leaf gas exchange and photosynthesis in wheat. New Phytol. 2022, 235, 1743–1756. [Google Scholar] [CrossRef]
- Nunes, T.D.; Slawinska, M.W.; Lindner, H.; Raissig, M.T. Quantitative effects of environmental variation on stomatal anatomy and gas exchange in a grass model. Cold Spring Harb. Lab. 2021, 101, 780–799. [Google Scholar] [CrossRef]
- McKown, K.H.; Bergmann, D.C. Stomatal development in the grasses: Lessons from models and crops (and crop models). New Phytol. 2020, 227, 1636–1648. [Google Scholar] [CrossRef]
- Caine, R.S.; Yin, X.; Sloan, J.; Harrison, E.L.; Mohammed, U.; Fulton, T.; Biswal, A.K.; Dionora, J.; Chater, C.C.; Coe, R.A.; et al. Rice with reduced stomatal density conserves water and has improved drought tolerance under future climate conditions. New Phytol. 2019, 221, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, J.; Thakur, V.; Nepomuceno, R.; Coe, R.A.; Dionora, J.; Elmido-Mabilangan, A.; Llave, A.D.; Delos Reyes, A.M.; Monroy, A.N.; Canicosa, I.; et al. Natural Diversity in Stomatal Features of Cultivated and Wild Oryza Species. Rice 2020, 13, 58. [Google Scholar] [CrossRef] [PubMed]
- Gago, J.; Carriqui, M.; Nadal, M.; Jose Clemente-Moreno, M.; Eduardo Coopman, R.; Fernie, A.R.; Flexas, J. Photosynthesis Optimized across Land Plant Phylogeny. Trends Plant Sci. 2019, 24, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, P.; Or, D. Effects of stomata clustering on leaf gas exchange. New Phytol. 2015, 207, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Mott, K.A.; Buckley, T.N. Patchy stomatal conductance: Emergent collective behaviour of stomata. Trends Plant Sci. 2000, 5, 258–262. [Google Scholar] [CrossRef]
- Salisbury, E.J. On the causes and ecological significance of stomatal frequency, with special reference to the woodland flora. Philos. Trans. R. Soc. Lond. Ser. B-Contain. Pap. A Biol. Character 1928, 216, 1–65. [Google Scholar] [CrossRef]
- Wang, Y.; Noguchi, K.; Terashima, I. Distinct light responses of the adaxial and abaxial stomata in intact leaves of Helianthus annuus L. Plant Cell Environ. 2008, 31, 1307–1316. [Google Scholar] [CrossRef]
- Muir, C.D. Is Amphistomy an Adaptation to High Light? Optimality Models of Stomatal Traits along Light Gradients. Integr. Comp. Biol. 2019, 59, 571–584. [Google Scholar] [CrossRef]
- Wang, S.; Zhou, Z.; Rahiman, R.; Lee, G.S.Y.; Yeo, Y.K.; Yang, X.; Lau, O.S. Light regulates stomatal development by modulating paracrine signaling from inner tissues. Nat. Commun. 2021, 12, 3403. [Google Scholar] [CrossRef]
- Sack, L.; Dietrich, E.M.; Streeter, C.M.; Holbrook, N.M. Leaf palmate venation and vascular redundancy confer tolerance of hydraulic disruption. Proc. Natl. Acad. Sci. USA 2008, 105, 1567–1572. [Google Scholar] [CrossRef]
- Brown, K.W.; Rosenberg, N.J. Effect of windbreaks and soil water potential on stomatal diffusion resistance and photosynthetic rate of sugar beets (beta-vulgaris). Agron. J. 1970, 62, 4–8. [Google Scholar] [CrossRef]
- Paradiso, R.; Marcelis, L.F.M. The Effect of Irradiating Adaxial or Abaxial Side on Photosynthesis of Rose Leaves. Acta Hortic. 2012, 956, 157–163. [Google Scholar] [CrossRef]
- Wang, C.; Du, Y.M.; Zhang, J.X.; Ren, J.T.; He, P.; Wei, T.; Xie, W.; Yang, H.K. Effects of exposure of the leaf abaxial surface to direct solar radiation on the leaf anatomical traits and photosynthesis of soybean (Glycine max L.) in dryland farming systems. Photosynthetica 2021, 59, 496–507. [Google Scholar] [CrossRef]
- Poorter, H.; Fiorani, F.; Pieruschka, R.; Wojciechowski, T.; van der Putten, W.H.; Kleyer, M.; Schurr, U.; Postma, J. Pampered inside, pestered outside? Differences and similarities between plants growing in controlled conditions and in the field. New Phytol. 2016, 212, 838–855. [Google Scholar] [CrossRef]
- Talbott, L.D.; Srivastava, A.; Zeiger, E. Stomata from growth-chamber-grown Vicia faba have an enhanced sensitivity to CO2. Plant Cell Environ. 1996, 19, 1188–1194. [Google Scholar] [CrossRef]
- Xiong, D.; Flexas, J.; Yu, T.; Peng, S.; Huang, J. Leaf anatomy mediates coordination of leaf hydraulic conductance and mesophyll conductance to CO2 in Oryza. New Phytol. 2017, 213, 572–583. [Google Scholar] [CrossRef]
- Chen, L.; Luo, W.; Huang, J.; Peng, S.; Xiong, D. Leaf photosynthetic plasticity does not predict biomass responses to growth irradiance in rice. Physiol. Plant. 2021, 173, 2155–2165. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org (accessed on 12 March 2020).


| Trait | Symbol | Unit | Species | Growth Condition | Species × Growth Condition |
|---|---|---|---|---|---|
| Adaxial stomatal density | SDad | mm−2 | *** | *** | *** |
| Abaxial stomatal density | SDab | mm−2 | *** | *** | ** |
| Stomatal density | SD | mm−2 | *** | *** | *** |
| Minor vein thickness | Tminor_vein | μm | *** | *** | ns |
| Major vein thickness | Tmajor_vein | μm | *** | *** | ns |
| Adaxial stomatal pore length | PLad | μm | *** | *** | *** |
| Abaxial stomatal pore length | PLab | μm | *** | * | *** |
| Adaxial stomatal pore width | PWad | μm | *** | ns | ns |
| Abaxial stomatal pore width | PWab | μm | *** | * | ** |
| Adaxial stomatal complex width | SWad | μm | *** | ns | * |
| Abaxial stomatal complex width | SWab | μm | *** | ns | ** |
| Adaxial guard cell width | GCWad | μm | *** | ns | ns |
| Abaxial guard cell width | GCWab | μm | *** | ns | ns |
| Leaf width | LW | μm | *** | ** | * |
| Stomatal ratio | SR | - | *** | ns | ns |
| Major vein density | VLAmajor | mm mm−2 | *** | ns | ns |
| Minor vein density | VLAminor | mm mm−2 | *** | ** | ** |
| Inter-vein distance | IVD | μm | ** | * | ns |
| Light saturated photosynthetic rate with adaxial illuminated | Asat_ad | μmol m−2 s−1 | *** | *** | *** |
| Light saturated photosynthetic rate with abaxial illuminated | Asat_ab | μmol m−2 s−1 | *** | ** | *** |
| Photosynthetic rate with adaxial illuminated | Aad1800 | μmol m−2 s−1 | *** | *** | * |
| Photosynthetic rate with abaxial illuminated | Aab1800 | μmol m−2 s−1 | *** | *** | * |
| Stomatal conductance with adaxial illuminated | gsw_ad1800 | mol m−2 s−1 | *** | * | ns |
| Stomatal conductance with abaxial illuminated | gsw_ab1800 | mol m−2 s−1 | *** | *** | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Zheng, L.; Xiong, D.; Wang, F.; Man, J.; Deng, N.; Cui, K.; Huang, J.; Peng, S.; Ling, X. Stomatal Ratio Showing No Response to Light Intensity in Oryza. Plants 2023, 12, 66. https://doi.org/10.3390/plants12010066
Wang T, Zheng L, Xiong D, Wang F, Man J, Deng N, Cui K, Huang J, Peng S, Ling X. Stomatal Ratio Showing No Response to Light Intensity in Oryza. Plants. 2023; 12(1):66. https://doi.org/10.3390/plants12010066
Chicago/Turabian StyleWang, Tiange, Linna Zheng, Dongliang Xiong, Fei Wang, Jianguo Man, Nanyan Deng, Kehui Cui, Jianliang Huang, Shaobing Peng, and Xiaoxia Ling. 2023. "Stomatal Ratio Showing No Response to Light Intensity in Oryza" Plants 12, no. 1: 66. https://doi.org/10.3390/plants12010066
APA StyleWang, T., Zheng, L., Xiong, D., Wang, F., Man, J., Deng, N., Cui, K., Huang, J., Peng, S., & Ling, X. (2023). Stomatal Ratio Showing No Response to Light Intensity in Oryza. Plants, 12(1), 66. https://doi.org/10.3390/plants12010066








