On the Possible Trade-Off between Shoot and Root Biomass in Wheat
Abstract
:1. Introduction
2. Results
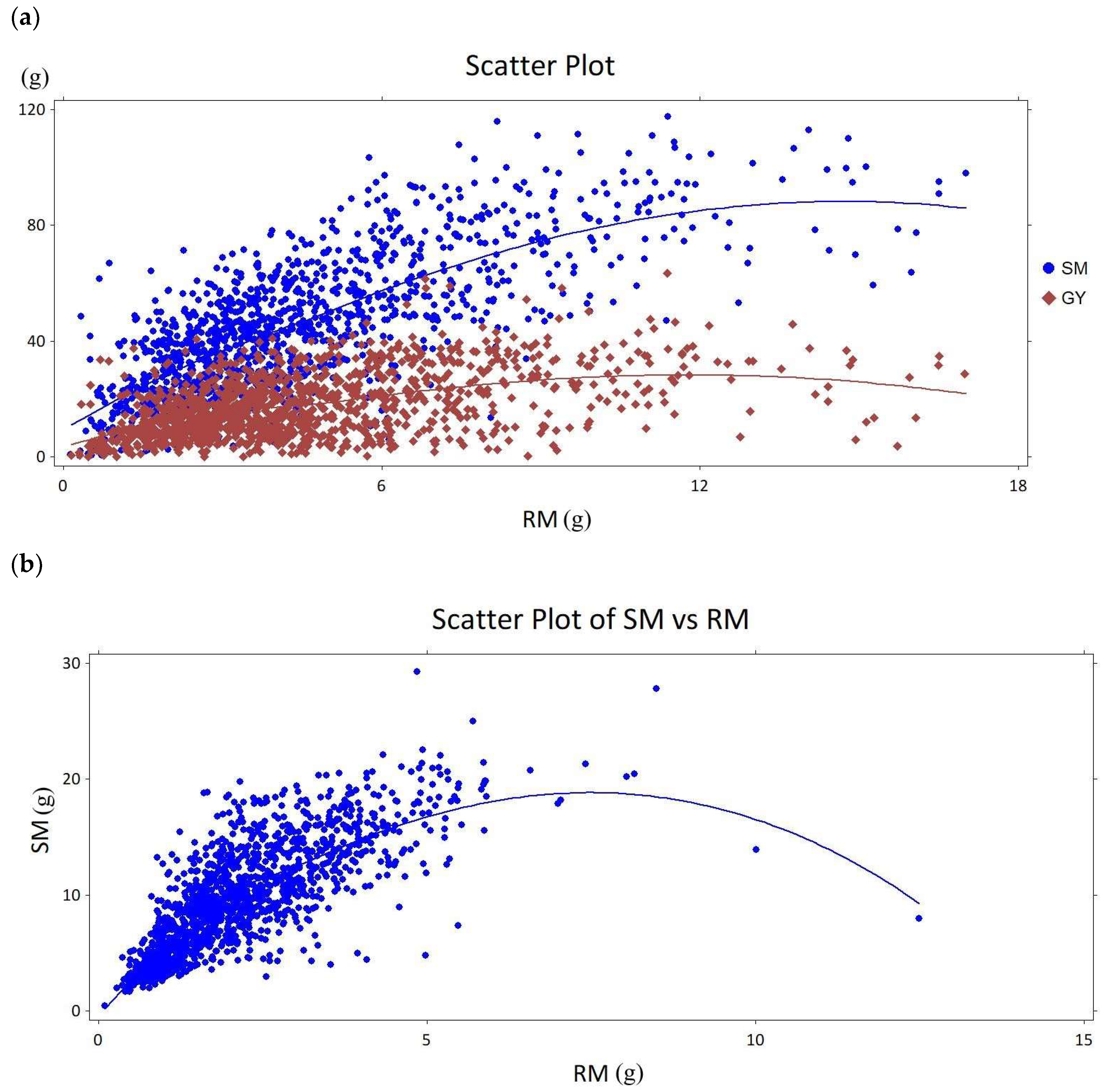
2.1. Combined Data Analysis
2.2. Trade-Off Pot Experiments
2.3. Trade-Off Tube Experiments
3. Discussion
3.1. Genetic Variation for Root Biomass
3.2. Trade-Offs in Root and Shoot Biomass
4. Materials and Methods
4.1. Plant Material
4.2. Experimental Design for Combined Data
4.3. Experimental Design for Trade-off Study
4.4. Growth Conditions and Irrigation Regimes on Trade-off Experiments
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Den Herder, G.; Van Isterdael, G.; Beeckman, T.; De Smet, I. The roots of a new green revolution. Trends Plant Sci. 2010, 15, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Manske, G.G.B.; Vlek, P.L.G. Root architecture-wheat as a model plant. In Plant Roots: The Hidden Half, 3rd ed.; Waisel, Y., Eshel, A., Beeckman, T., Kafkafi, U., Eds.; Routledge: Milton Park, UK, 2002; p. 249. [Google Scholar]
- Manschadi, A.M.; Christopher, J.; deVoil, P.; Hammer, G.L. The role of root architectural traits in adaptation of wheat to water-limited environments. Funct. Plant Biol. 2006, 33, 823–837. [Google Scholar] [CrossRef] [Green Version]
- Richards, R.A. Genetic Opportunities to Improve Cereal Root Systems for Dryland Agriculture. Plant Prod. Sci. 2008, 11, 12–16. [Google Scholar] [CrossRef]
- Comas, L.; Becker, S.; Cruz, V.M.V.; Byrne, P.F.; Dierig, D.A. Root traits contributing to plant productivity under drought. Front. Plant Sci. 2013, 4, 442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watt, M.; Wasson, A.P.; Chochois, V. Root-based solutions to increasing crop productivity. In Plant Roots: The Hidden Half; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Mac Key, J. The wheat root. In Proceedings of the 4th International Wheat Genetics Symposium, Columbia, MO, USA, 6–11 August 1973; Sears, E.R., Sears, L.M.S., Eds.; Agricultural Experiment Station, College of Agriculture, University of Missouri: Columbia, MO, USA, 1973; pp. 827–842. [Google Scholar]
- Richards, R.A.; Passioura, J.B. Seminal Root Morphology and Water Use of Wheat I. Environmental Effects1. Crop Sci. 1981, 21, 249–252. [Google Scholar] [CrossRef]
- Richards, R.; Passioura, J. A breeding program to reduce the diameter of the major xylem vessel in the seminal roots of wheat and its effect on grain yield in rain-fed environments. Aust. J. Agric. Res. 1989, 40, 943–950. [Google Scholar] [CrossRef]
- Ehdaie, B.; Waines, J.G. Chromosomal location of genes influencing plant characters and evapotranspiration efficiency in bread wheat. Euphytica 1997, 96, 363–375. [Google Scholar] [CrossRef]
- Richards, R.A. Physiological traits used in the breeding of new cultivars for water-scarce environments. Agric. Water Manag. 2006, 80, 197–211. [Google Scholar] [CrossRef]
- Sharma, S.; Xu, S.; Ehdaie, B.; Hoops, A.; Close, T.J.; Lukaszewski, A.J.; Waines, J.G. Dissection of QTL effects for root traits using a chromosome arm-specific mapping population in bread wheat. TAG Theor. Appl. Genet. Theor. Angew. Genet. 2011, 122, 759–769. [Google Scholar] [CrossRef] [Green Version]
- Bektas, H.; Waines, J.G. Root and shoot traits in parental, early and late generation Green Revolution wheats (Triticum spp.) under glasshouse conditions. Genet. Resour. Crop Evol. 2018, 65, 2003–2012. [Google Scholar] [CrossRef]
- Bektas, H.; Hohn, C.E.; Waines, J.G. Root and shoot traits of bread wheat (Triticum aestivum L.) landraces and cultivars. Euphytica 2016, 212, 297–311. [Google Scholar] [CrossRef]
- Hochholdinger, F.; Yu, P.; Marcon, C. Genetic Control of Root System Development in Maize. Trends Plant Sci. 2018, 23, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Sanguineti, M.C.; Li, S.; Maccaferri, M.; Corneti, S.; Rotondo, F.; Chiari, T.; Tuberosa, R. Genetic dissection of seminal root architecture in elite durum wheat germplasm. Ann. Appl. Biol. 2007, 151, 291–305. [Google Scholar] [CrossRef]
- Landjeva, S.; Neumann, K.; Lohwasser, U.; Börner, A. Molecular mapping of genomic regions associated with wheat seedling growth under osmotic stress. Biol. Plant. 2008, 52, 259–266. [Google Scholar] [CrossRef]
- Li, P.; Chen, J.; Wu, P.; Zhang, J.; Chu, C.; See, D.; Brown-Guedira, G.; Zemetra, R.; Souza, E. Quantitative Trait Loci Analysis for the Effect of Rht-B1 Dwarfing Gene on Coleoptile Length and Seedling Root Length and Number of Bread Wheat. Crop Sci. 2011, 51, 2561–2568. [Google Scholar] [CrossRef]
- Hamada, A.; Nitta, M.; Nasuda, S.; Kato, K.; Fujita, M.; Matsunaka, H.; Okumoto, Y. Novel QTLs for growth angle of seminal roots in wheat (Triticum aestivum L.). Plant Soil 2012, 354, 395–405. [Google Scholar] [CrossRef] [Green Version]
- Bai, C.H.; Liang, Y.L.; Hawkesford, M.J. Identification of QTLs associated with seedling root traits and their correlation with plant height in wheat. J. Exp. Bot. 2013, 64, 1745–1753. [Google Scholar] [CrossRef] [Green Version]
- Christopher, J.; Christopher, M.; Jennings, R.; Jones, S.; Fletcher, S.; Borrell, A.; Manschadi, A.M.; Jordan, D.; Mace, E.; Hammer, G. QTL for root angle and number in a population developed from bread wheats (Triticum aestivum) with contrasting adaptation to water-limited environments. Theor. Appl. Genet. 2013, 126, 1563–1574. [Google Scholar] [CrossRef]
- Liu, X.L.; Li, R.Z.; Chang, X.P.; Jing, R.L. Mapping QTLs for seedling root traits in a doubled haploid wheat population under different water regimes. Euphytica 2013, 189, 51–66. [Google Scholar] [CrossRef]
- Zhang, H.; Cui, F.; Wang, H. Detection of quantitative trait loci (QTLs) for seedling traits and drought tolerance in wheat using three related recombinant inbred line (RIL) populations. Euphytica 2013, 196, 313–330. [Google Scholar] [CrossRef]
- Botwright Acuña, T.L.; Rebetzke, G.J.; He, X.; Maynol, E.; Wade, L.J. Mapping quantitative trait loci associated with root penetration ability of wheat in contrasting environments. Mol. Breed. 2014, 34, 631–642. [Google Scholar] [CrossRef]
- Canè, M.A.; Maccaferri, M.; Nazemi, G.; Salvi, S.; Francia, R.; Colalongo, C.; Tuberosa, R. Association mapping for root architectural traits in durum wheat seedlings as related to agronomic performance. Mol. Breed. New Strateg. Plant Improv. 2014, 34, 1629–1645. [Google Scholar] [CrossRef] [Green Version]
- Chesnokov, Y.V.; Goncharova, E.A.; Sitnikov, M.N.; Kocherina, N.V.; Lohwasser, U.; Borner, A. Mapping QTL for water regime in spring bread wheat. Russ. J. Plant Physiol. 2014, 61, 834–841. [Google Scholar] [CrossRef]
- Kabir, M.R.; Liu, G.; Guan, P.; Wang, F.; Khan, A.A.; Ni, Z.; Yao, Y.; Hu, Z.; Xin, M.; Peng, H.; et al. Mapping QTLs associated with root traits using two different populations in wheat (Triticum aestivum L.). Euphytica 2015, 206, 175–190. [Google Scholar] [CrossRef]
- Petrarulo, M.; Marone, D.; Ferragonio, P.; Cattivelli, L.; Rubiales, D.; De Vita, P.; Mastrangelo, A.M. Genetic analysis of root morphological traits in wheat. Mol. Genet. Genom. 2015, 290, 785–806. [Google Scholar] [CrossRef]
- Gabay, G.; Zhang, J.; Burguener, G.F.; Howell, T.; Wang, H.; Fahima, T.; Lukaszewski, A.; Moriconi, J.I.; Santa Maria, G.E.; Dubcovsky, J. Structural rearrangements in wheat (1BS)–rye (1RS) recombinant chromosomes affect gene dosage and root length. Plant Genome 2021, 14, e20079. [Google Scholar] [CrossRef]
- Gabay, G.; Wang, H.; Zhang, J.; Moriconi, J.I.; Burguener, G.F.; Gualano, L.D.; Howell, T.; Lukaszewski, A.; Staskawicz, B.; Cho, M.-J.; et al. Dosage differences in 12-OXOPHYTODIENOATE REDUCTASE genes modulate wheat root growth. Nat. Commun. 2023, 14, 539. [Google Scholar] [CrossRef] [PubMed]
- Mäkelä, A.; Sievänen, R. Comparison of two shoot—Root partitioning models with respect to substrate utilization and functional balance. Ann. Bot. 1987, 59, 129–140. [Google Scholar] [CrossRef]
- Davidson, R. Effect of root/leaf temperature differentials on root/shoot ratios in some pasture grasses and clover. Ann. Bot. 1969, 33, 561–569. [Google Scholar] [CrossRef]
- Evans, L. Aspects of the comparative physilogy of grain yield in cereals. Adv. Agron. 1976, 28, 301–359. [Google Scholar]
- Gregory, P.; Boote, K.; Bennett, J.; Sinclair, T.; Paulsen, G. Root growth and activity. In Physiology and Determination of Crop Yield; ASA: Oak Park, IL, USA; CSSA: Madison, WI, USA; SSSA: Madison, WI, USA, 1994; p. 65. [Google Scholar]
- Fageria, N.K. Maximizing Crop Yields; CRC Press: Boca Raton, FL, USA, 1992. [Google Scholar]
- Waines, G.; Ehdaie, B. Domestication and Crop Physiology: Roots of Green-Revolution Wheat. Ann. Bot. 2007, 100, 991–998. [Google Scholar] [CrossRef] [Green Version]
- Yaseen, M.; Malhi, S.S. Exploitation of genetic variability among wheat genotypes for tolerance to phosphorus deficiency stress. J. Plant Nutr. 2011, 34, 665–699. [Google Scholar] [CrossRef]
- Lopes, M.S.; Reynolds, M.P. Partitioning of assimilates to deeper roots is associated with cooler canopies and increased yield under drought in wheat. Funct. Plant Biol. 2010, 37, 147–156. [Google Scholar] [CrossRef]
- Uga, Y.; Sugimoto, K.; Ogawa, S.; Rane, J.; Ishitani, M.; Hara, N.; Kitomi, Y.; Inukai, Y.; Ono, K.; Kanno, N.; et al. Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nat. Genet. 2013, 45, 1097–1102. [Google Scholar] [CrossRef]
- Blum, A. Drought resistance, water-use efficiency, and yield potential—Are they compatible, dissonant, or mutually exclusive? Aust. J. Agric. Res. 2005, 56, 1159–1168. [Google Scholar] [CrossRef] [Green Version]
- Palta, J.A.; Chen, X.; Milroy, S.P.; Rebetzke, G.J.; Dreccer, M.F.; Watt, M. Large root systems: Are they useful in adapting wheat to dry environments? Funct. Plant Biol. 2011, 38, 347–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waines, J.G.; Ehdaie, B. Optimizing root characters and grain yield in wheat. Czech J. Genet. Plant Breed. 2005, 41, 326–330. [Google Scholar] [CrossRef] [Green Version]
- Kahlown, M.A.; Ashraf, M.; Zia Ul, H. Effect of shallow groundwater table on crop water requirements and crop yields. Agric. Water Manag. 2005, 76, 24–35. [Google Scholar] [CrossRef]
- Hurd, E.A. Root Study of Three Wheat Varieties and Their Resistance to Drought and Damage by Soil Cracking. Can. J. Plant Sci. 1964, 44, 240–248. [Google Scholar] [CrossRef]
- Hurd, E.A. Phenotype and drought tolerance in wheat. Agric. Meteorol. 1974, 14, 39–55. [Google Scholar] [CrossRef]
- Passioura, J.B. Roots and drought resistance. Agric. Water Manag. 1983, 7, 265–280. [Google Scholar] [CrossRef]
- Ehdaie, B.; Whitkus, R.W.; Waines, J.G. Root Biomass, Water-Use Efficiency, and Performance of Wheat–Rye Translocations of Chromosomes 1 and 2 in Spring Bread Wheat ‘Pavon’. Crop Sci. 2003, 43, 710–717. [Google Scholar] [CrossRef]
- de Dorlodot, S.; Forster, B.; Pages, L.; Price, A.; Tuberosa, R.; Draye, X. Root system architecture: Opportunities and constraints for genetic improvement of crops. Trends Plant Sci. 2007, 12, 474–481. [Google Scholar] [CrossRef]
- Bengough, A.G.; McKenzie, B.M.; Hallett, P.D.; Valentine, T.A. Root elongation, water stress, and mechanical impedance: A review of limiting stresses and beneficial root tip traits. J. Exp. Bot. 2011, 62, 59–68. [Google Scholar] [CrossRef] [Green Version]
- Maheepala, D.C.; Ehdaie, B.; Waines, J.G. Yield performance of wheat isolines with different dosages of the short arm of rye chromosome 1. J. Agron. Crop Sci. 2015, 201, 152–160. [Google Scholar] [CrossRef]
- Ehdaie, B.; Bektas, H.; Hohn, C.E.; Waines, J.G. Optimum root biomass for maximum grain yield in bread wheat under well watered and droughted field conditions. In Proceedings of the CSSA Annual Meetings, Minneapolis, MN, USA, 15–18 November 2015. [Google Scholar]
- Bektas, H. Determination of Root Traits in Wild, Landrace and Modern Wheats and Dissection of Quantitative Trait Loci (QTL) for Root Characters in Bread Wheat; University of California: Riverside, CA, USA, 2015. [Google Scholar]
- Bektas, H.; Hohn, C.E.; Waines, J.G. Characteristics of the root system in the diploid genome donors of hexaploid wheat (Triticum aestivum L.). Genet. Resour. Crop Evol. 2017, 64, 1641–1650. [Google Scholar] [CrossRef]
- Bektas, H.; Hohn, C.E.; Waines, J.G. Dissection of quantitative trait loci for root characters and day length sensitivity in SynOpDH wheat (Triticum aestivum L.) bi-parental mapping population. Plant Genet. Resour. Charact. Util. 2020, 18, 130–142. [Google Scholar] [CrossRef]
- Hohn, C.E. Discovery and Verification of Quantitative Trait Loci (QTLs) for Seminal Root Traits and Insights Into Root to Shoot Tradeoffs in Hexaploid Wheat (Triticum aestivum L.). Ph.D. Thesis, University of California, Riverside, CA, USA, 2016. [Google Scholar]
- Hohn, C.E.; Bektas, H. Preliminary tests indicate the absence of polymorphism among 1RS. 1BL translocations in wheat for root biomass enhancement. Euphytica 2021, 217, 76. [Google Scholar] [CrossRef]
- Hohn, C.E.; Bektas, H. Genetic Mapping of Quantitative Trait Loci (QTLs) Associated with Seminal Root Angle and Number in Three Populations of Bread Wheat (Triticum aestivum L.) with Common Parents. Plant Mol. Biol. Rep. 2020, 38, 572–585. [Google Scholar] [CrossRef]
- van der Bom, F.J.T.; Williams, A.; Bell, M.J. Root architecture for improved resource capture: Trade-offs in complex environments. J. Exp. Bot. 2020, 71, 5752–5763. [Google Scholar] [CrossRef]
- Robinson, D.; Peterkin, J.H. Clothing the Emperor: Dynamic Root-Shoot Allocation Trajectories in Relation to Whole-Plant Growth Rate and in Response to Temperature. Plants 2019, 8, 212. [Google Scholar] [CrossRef] [Green Version]
- Kiaer, L.P.; Weisbach, A.N.; Weiner, J. Root and shoot competition: A meta-analysis. J. Ecol. 2013, 101, 1298–1312. [Google Scholar] [CrossRef]
- Qi, Y.; Wei, W.; Chen, C.; Chen, L. Plant root-shoot biomass allocation over diverse biomes: A global synthesis. Glob. Ecol. Conserv. 2019, 18, e00606. [Google Scholar] [CrossRef]
- Chen, Y.; Palta, J.; Prasad, P.V.V.; Siddique, K.H.M. Phenotypic variability in bread wheat root systems at the early vegetative stage. BMC Plant Biol. 2020, 20, 185. [Google Scholar] [CrossRef]
- Ehdaie, B.; Waines, J.G. Sowing date and nitrogen rate effects on dry matter and nitrogen partitioning in bread and durum wheat. Field Crops Res. 2001, 73, 47–61. [Google Scholar] [CrossRef]
- Ehdaie, B.; Alloush, G.A.; Waines, J.G. Genotypic variation in linear rate of grain growth and contribution of stem reserves to grain yield in wheat. Field Crops Res. 2008, 106, 34–43. [Google Scholar] [CrossRef]
- Sharma, S.; Bhat, P.R.; Ehdaie, B.; Close, T.J.; Lukaszewski, A.J.; Waines, J.G. Integrated genetic map and genetic analysis of a region associated with root traits on the short arm of rye chromosome 1 in bread wheat. Theor. Appl. Genet. 2009, 119, 783–793. [Google Scholar] [CrossRef] [Green Version]
- Ehdaie, B.; Layne, A.; Waines, J.G. Root system plasticity to drought influences grain yield in bread wheat. Euphytica 2012, 186, 219–232. [Google Scholar] [CrossRef]
- Lynch, J.P. Steep, cheap and deep: An ideotype to optimize water and N acquisition by maize root systems. Ann. Bot. 2013, 112, 347–357. [Google Scholar] [CrossRef] [Green Version]
- Figueroa-Bustos, V.; Palta, J.A.; Chen, Y.; Stefanova, K.; Siddique, K.H.M. Wheat Cultivars with Contrasting Root System Size Responded Differently to Terminal Drought. Front. Plant Sci. 2020, 11, 1258. [Google Scholar] [CrossRef]
- Figueroa-Bustos, V.; Palta, J.; Chen, Y.; Siddique, K. Characterization of Root and Shoot Traits in Wheat Cultivars with Putative Differences in Root System Size. Agronomy 2018, 8, 109. [Google Scholar] [CrossRef] [Green Version]
- Figueroa-Bustos, V.; Palta, J.A.; Chen, Y.; Siddique, K.H.M. Early Season Drought Largely Reduces Grain Yield in Wheat Cultivars with Smaller Root Systems. Plants 2019, 8, 305. [Google Scholar] [CrossRef] [Green Version]
- Cabal, C.; Martínez-García, R.; de Castro Aguilar, A.; Valladares, F.; Pacala, S.W. The exploitative segregation of plant roots. Science 2020, 370, 1197–1199. [Google Scholar] [CrossRef] [PubMed]
- Ehdaie, B.; Waines, J. Determination of a chromosome segment influencing rooting ability in wheat-rye 1BS-1RS recombinant lines [Triticum aestivum L.; Secale cereale L.]. J. Genet. Breed. 2006, 60, 71–76. [Google Scholar]
- Steel, R.G.D.; Torrie, J.H.; Dickey, D.A. Principles and Procedures of Statistics: A Biometrical Approach; McGraw-Hill: New York, NY, USA, 1997. [Google Scholar]









| Mapping Populations | Data from Diverse Sets of Lines | Trade-Off Experiments | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Population Name | Sonora × Foisy (SF) | Sonora × CBdeM (SC) | Chiddam Blanc de Mars (CBdeM) × Foisy (CF) | Synthetic W7984 × Opata M85 (SynOpDH) | Wheat Wild Relatives | Historical CIMMYT Accessions | Turkish Wheat Accessions | 1RS.1BL Translocation and 1B Substitution Lines | Pavon 76 and Yecora Rojo |
| Number of Accessions evaluated | 141 | 146 | 128 | 147 | 15 | 9 | 19 | 32 | 2 |
| Growth conditions | PVC tubes and 3.8 l pots | PVC tubes | PVC tubes | PVC tubes and 3.8 l pots | |||||
| Growth duration | 21–28 days in 2014–2015; 30–60 days in 2013; 40 days in 2016 | Maturity in 2012–2014 | 40–70 days between 2012–2015 | Maturity in 2015–2016 | |||||
| Reference | [55,57] | [13,14,52,53,54] | [55,56] | ||||||
| Trait | Duration/Growth Stage | N | Mean (g) | SD | SE Mean | C.V. | Minimum (g) | Median (g) | Maximum (g) |
|---|---|---|---|---|---|---|---|---|---|
| Shoot Biomass | 21–28 | 3768 | 0.81 | 0.51 | 0.01 | 62.47 | 0.05 | 0.66 | 3.00 |
| 40–70 | 1342 | 10.98 | 9.68 | 0.27 | 88.19 | 1.54 | 8.78 | 70.19 | |
| Maturity | 1243 | 46.33 | 22.43 | 0.61 | 48.41 | 0.36 | 44.07 | 117.11 | |
| Root Biomass | 21–28 | 3768 | 0.30 | 0.22 | 0.00 | 73.50 | 0.01 | 0.26 | 2.29 |
| 40–70 | 1342 | 2.14 | 1.27 | 0.04 | 59.38 | 0.27 | 1.88 | 12.49 | |
| Maturity | 1243 | 4.76 | 2.98 | 0.08 | 62.54 | 0.15 | 3.96 | 17.68 |
| Tubes | Pots | |||||
|---|---|---|---|---|---|---|
| Pavon 76 | Treatment | RM/SM | RM < 30/RM > 30 | RM < 30/SM | Treatment | RM/SM |
| Control | 0.26 | 1.11 | 0.14 | Control | 0.20 | |
| Deep | 0.49 | 0.50 | 0.16 | Anthesis | 0.40 | |
| Shallow | 0.22 | 3.15 | 0.16 | Heading | 0.43 | |
| Booting | 0.30 | |||||
| Yecora Rojo | Treatment | RM/SM | RM < 30/RM > 30 | RM < 30/SM | Treatment | RM/SM |
| Control | 0.86 | 0.41 | 0.25 | Control | 0.55 | |
| Deep | 0.87 | 0.54 | 0.30 | Anthesis | 0.55 | |
| Shallow | 0.39 | 8.54 | 0.35 | Heading | 0.44 | |
| Booting | 0.45 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bektas, H.; Hohn, C.E.; Lukaszewski, A.J.; Waines, J.G. On the Possible Trade-Off between Shoot and Root Biomass in Wheat. Plants 2023, 12, 2513. https://doi.org/10.3390/plants12132513
Bektas H, Hohn CE, Lukaszewski AJ, Waines JG. On the Possible Trade-Off between Shoot and Root Biomass in Wheat. Plants. 2023; 12(13):2513. https://doi.org/10.3390/plants12132513
Chicago/Turabian StyleBektas, Harun, Christopher E. Hohn, Adam J. Lukaszewski, and John Giles Waines. 2023. "On the Possible Trade-Off between Shoot and Root Biomass in Wheat" Plants 12, no. 13: 2513. https://doi.org/10.3390/plants12132513







