Age Determination and Growth Characteristics of the Potentilla griffithii: A Comparison of Two Different Habitats in Western Sichuan Plateau, China
Abstract
:1. Introduction
- (1)
- Can the age of P. griffithii plants be determined by leaf scars? If yes, what is the age structure character of P. griffithii in two typical habitats in the western Sichuan Plateau?
- (2)
- What survival strategies does P. griffithii adopt in different habitats in the western Sichuan Plateau?
- (3)
- Are age–trait relationships consistent enough to allow a general model to accurately predict plant growth conditions, based on plant age?
2. Results
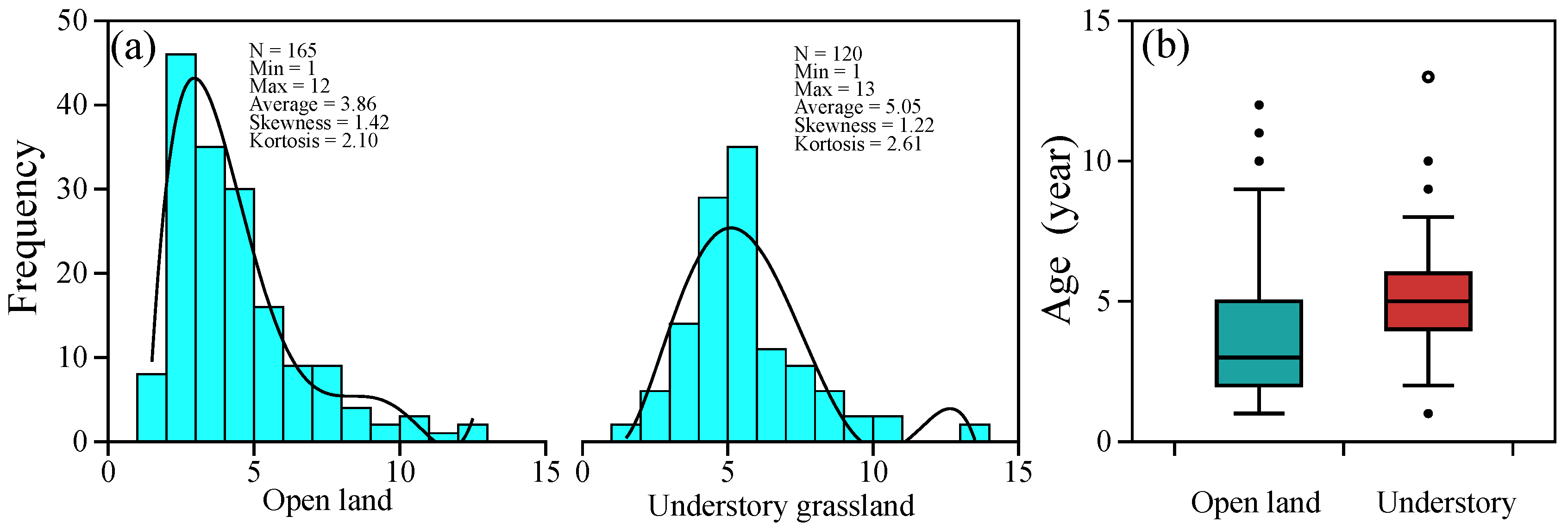
2.1. Age Determination of P. griffithii in Common Garden Individuals
2.2. Age Distribution
2.3. Trait Comparison
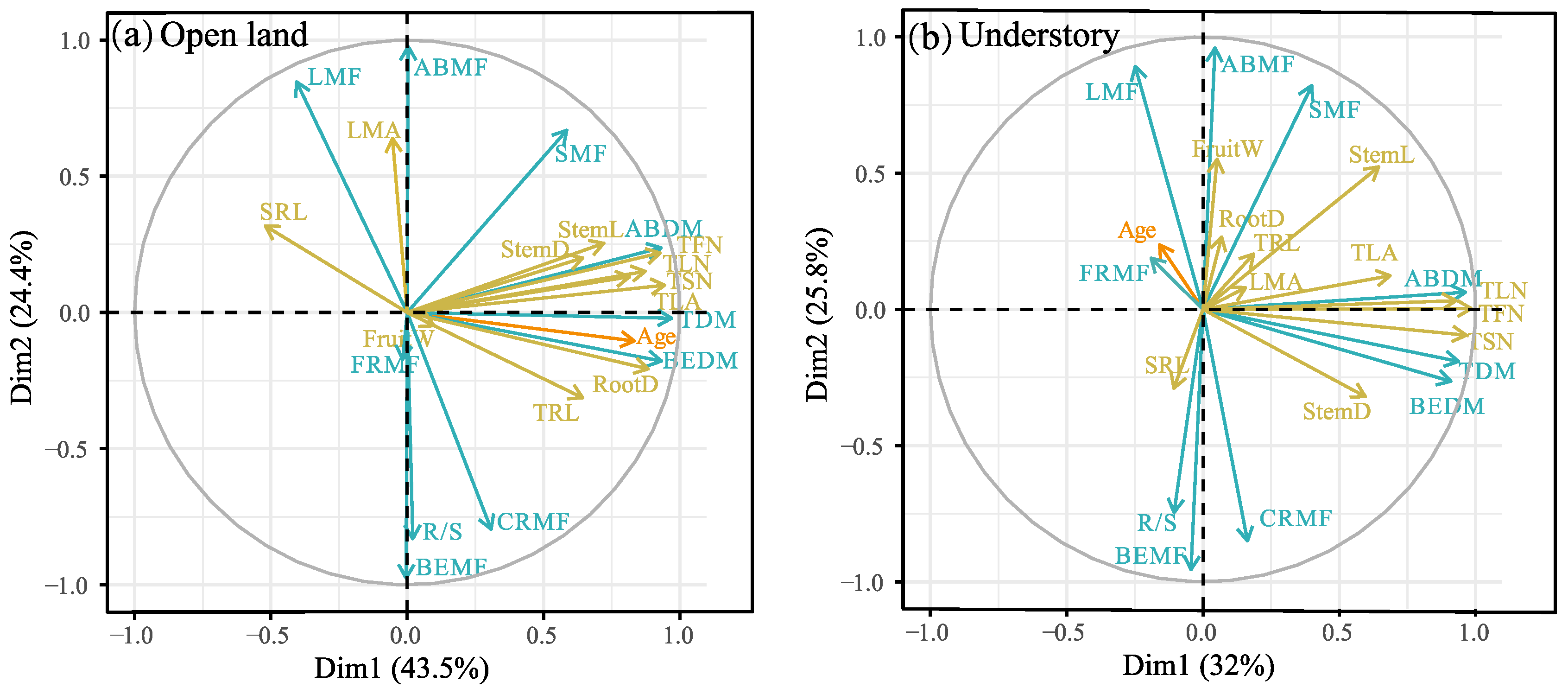
2.4. Relationships between Age and Plant Traits
3. Discussion
3.1. Leaf Scars Can Be Used for Aging P. griffithii
3.2. Growth Strategies of P. griffithii in Two Habitats
3.3. Do Age–Trait Relationships Allow a General Model to Accurately Predict Plant Growth Conditions from Plant Age?
3.4. Future Implications
4. Materials and Methods
4.1. Plant Species
4.2. Environmental Design
4.2.1. Common Garden Experiment
4.2.2. Field Experiment
4.3. Measurements
4.4. Trait Measurements
- Leaf mass per area (LMA) (g cm−2): LMA = leaf dry mass (g)/leaf area (cm2);
- Mean fruit weight (FruitW) (mg): FruitW = total fruit dry mass (g)/fruit number;
- Specific root length (SRL) (cm g−1): SRL = root length (cm)/below-ground dry mass (g);
- Leaf mass fraction (LMF) (g g−1): LMF = leaf dry mass (g)/total plant dry mass (g);
- Stem mass fraction (SMF) (g g−1): SMF = stem dry mass (g)/total plant dry mass (g);
- Fine root mass fraction (FRMF) (g g−1): FRMF = fine root dry mass (g)/total plant dry mass (g);
- Coarse root mass fraction (FRMF) (g g−1): FRMF = coarse root dry mass (g)/total plant dry mass (g);
- Below-ground biomass mass fraction (FRMF) (g g−1): BEMF = below-ground dry mass (g)/total plant dry mass (g);
- Above-ground biomass mass fraction (ABMF) (g g−1): ABMF = above-ground dry mass (g)/total plant dry mass (g);
- Root/shoot ratio (R/S ratio) (g g−1): R/S ratio = below-ground dry mass (g)/above-ground dry mass (g).
| Cate. | No. | Trait Code | Full Name | Explanations (Unit) |
|---|---|---|---|---|
| Biomass and allocation | 1 | TDM | Total dry mass | The total dry mass below and above the ground (g) |
| 2 | ABDM | Above-ground dry mass | The total dry mass of leaves and stems (g) | |
| 3 | BEDM | Below-ground dry mass | The total dry mass of roots (g) | |
| 4 | R/S | Root-shoot ratio | The ratio of the below-ground and above-ground dry mass | |
| 5 | ABMF | Above-ground mass fraction | Above-ground dry mass/total plant dry mass (g g−1) | |
| 6 | BEMF | Below-ground biomass mass fraction | Root dry mass/total plant dry mass (g g−1) | |
| 7 | LMF | Leaf mass fraction | Leaf dry mass/total plant dry mass (g g−1) | |
| 8 | SMF | Stem mass fraction | Stem dry mass/total plant dry mass (g g−1) | |
| 9 | FRMF | Fine root mass fraction | Fine root dry mass/total plant dry mass (g g−1) | |
| 10 | CRMF | Coarse root mass fraction | Coarse root dry mass/total plant dry mass (g g−1) | |
| The whole-plant morphology | 11 | TLN | Total leaf number | Total leaves per individual (No.) |
| 12 | TFN | Total fruit number | Total aggregate fruits per individual (No.) | |
| 13 | TSN | Total stem number | Total stems per individual (No.) | |
| 14 | TLA | Total leaf area | The sum of all the leaf area (cm2) | |
| 15 | StemD | Stem diameter | Mean stem diameter of two dimensions per stem (mm) | |
| 16 | StemL | Total stem length | Mean stem length per plant (cm) | |
| 17 | RootD | Root diameter | Mean coarse root diameter of two dimensions (mm) | |
| 18 | TRL | Root length | The sum of all the root lengths (cm) | |
| 19 | FruitW | Mean fruit weight | Mean aggregate fruit weight (mg) | |
| 20 | SRL | Specific root length | Root length per mass (cm/g) | |
| 21 | LMA | Leaf mass per area | Leaf mass per unit leaf area (g/cm2) |
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tomczyk, M.; Latté, K.P. Potentilla—A review of its phytochemical and pharmacological profile. J. Ethnopharm. 2009, 122, 184–204. [Google Scholar] [CrossRef] [PubMed]
- Qiu, R.; Fang, X.; Tang, Y.; Du, S.; Zeng, X.; Brewer, E. Zinc hyperaccumulation and uptake by Potentilla griffithii Hook. Int. J. Phytoremediat. 2006, 8, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.J.; Qiu, R.L.; Senthilkumar, P.; Jiang, D.; Chen, Z.W.; Tang, Y.T.; Liu, F.J. Tolerance, accumulation and distribution of zinc and cadmium in hyperaccumulator Potentilla griffithii. Environ. Exp. Bot. 2009, 66, 317–325. [Google Scholar] [CrossRef]
- Field, C.; Mooney, H.A. Leaf age and seasonal effects on light, water, and nitrogen use efficiency in a California shrub. Oecologia 1983, 56, 348–355. [Google Scholar] [CrossRef]
- Reich, P.B.; Walters, M.B.; Ellsworth, D.S. Leaf life-span in relation to leaf, plant, and stand characteristics among diverse ecosystems. Ecol. Monogr. 1992, 62, 365–392. [Google Scholar] [CrossRef]
- Reich, P.B.; Uhl, C.; Walters, M.B.; Prugh, L.; Ellsworth, D.S. Leaf demography and phenology in Amazonian rain forest: A census of 40,000 leaves of 23 tree species. Ecol. Monogr. 2004, 74, 3–23. [Google Scholar] [CrossRef] [Green Version]
- Reich, P.B.; Walters, M.B.; Ellsworth, D.S. Leaf age and season influence the relationships between leaf nitrogen, leaf mass per area and photosynthesis in maple and oak trees. Plant Cell Environ. 1991, 14, 251–259. [Google Scholar] [CrossRef]
- Wilson, K.B.; Baldocchi, D.D.; Hanson, P.J. Leaf age affects the seasonal pattern of photosynthetic capacity and net ecosystem exchange of carbon in a deciduous forest. Plant Cell Environ. 2001, 24, 571–583. [Google Scholar] [CrossRef]
- Kitajima, K.; Mulkey, S.S.; Samaniego, M.; Joseph Wright, S. Decline of photosynthetic capacity with leaf age and position in two tropical pioneer tree species. Am. J. Bot. 2002, 89, 1925–1932. [Google Scholar] [CrossRef] [Green Version]
- Mauromicale, G.; Ierna, A.; Marchese, M. Chlorophyll fluorescence and chlorophyll content in field-grown potato as affected by nitrogen supply, genotype, and plant age. Photosynthetica 2006, 44, 76–82. [Google Scholar] [CrossRef]
- Pantin, F.; Simonneau, T.; Muller, B. Coming of leaf age: Control of growth by hydraulics and metabolics during leaf ontogeny. New Phytol. 2012, 196, 349–366. [Google Scholar] [CrossRef]
- Shipley, B.; Meziane, D. The balanced-growth hypothesis and the allometry of leaf and root biomass allocation. Funct. Ecol. 2002, 16, 326–331. [Google Scholar] [CrossRef]
- Hódar, J.A.; Zamora, R.; Castro, J.; Gómez, J.M.; García, D. Biomass allocation and growth responses of Scots pine saplings to simulated herbivory depend on plant age and light availability. Plant Ecol. 2008, 197, 229–238. [Google Scholar] [CrossRef]
- Maksymowych, R. Analysis of Leaf Development; Cambridge University Press Archive: Cambridge, UK, 1973; Volume 1. [Google Scholar]
- Oyama, K. Are age and height correlated in Chamaedorea tepejilote (Palmae)? J. Trop. Ecol. 1993, 9, 381–385. [Google Scholar] [CrossRef]
- Zahedi, S.M.; Sarikhani, H. Effect of far-red light, temperature, and plant age on morphological changes and induction of flowering of a ‘June-bearing’ strawberry. Hortic. Environ. Biotechnol. 2016, 57, 340–347. [Google Scholar] [CrossRef]
- Punja, Z.K.; Sutton, D.B.; Kim, T. Glandular trichome development, morphology, and maturation are influenced by plant age and genotype in high THC-containing cannabis (Cannabis sativa L.) inflorescences. J. Cannabis Res. 2023, 5, 12. [Google Scholar] [CrossRef]
- Hanzawa, F.M.; Kalisz, S. The relationship between age, size, and reproduction in Trillium grandiflorum (liliaceae). Am. J. Bot. 1993, 80, 405–410. [Google Scholar] [CrossRef]
- Ehlers, B.K.; Olesen, J.M. Flower production in relation to individual plant age and leaf production among different patches of Corydalis intermedia. Plant Ecol. 2004, 174, 71–78. [Google Scholar] [CrossRef]
- Burd, M.; Read, J.; Sanson, G.D.; Jaffré, T. Age–size plasticity for reproduction in monocarpic plants. Ecology 2006, 87, 2755–2764. [Google Scholar] [CrossRef]
- Duque, L.; Poelman, E.H.; Steffan-Dewenter, I. Plant age at the time of ozone exposure affects flowering patterns, biotic interactions and reproduction of wild mustard. Sci. Rep. 2021, 11, 23448. [Google Scholar] [CrossRef]
- Gatsuk, L.E.; Smirnova, O.V.; Vorontzova, L.I.; Zaugolnova, L.B.; Zhukova, L.A. Age states of plants of various growth forms: A review. J. Ecol. 1980, 68, 675–696. [Google Scholar] [CrossRef]
- Bowers, M.D.; Stamp, N.E. Effects of plant age, genotype and herbivory on Plantago performance and chemistry. Ecology 1993, 74, 1778–1791. [Google Scholar] [CrossRef]
- Rixen, C.; Casteller, A.; Schweingruber, F.H.; Stoeckli, V. Age analysis helps to estimate plant performance on ski pistes. Bot. Helv. 2004, 114, 127–138. [Google Scholar]
- Sharabani, G.; Shtienberg, D.; Borenstein, M.; Shulhani, R.; Lofthouse, M.; Sofer, M.; Chalupowicz, L.; Barel, V.; Manulis-Sasson, S. Effects of plant age on disease development and virulence of Clavibacter michiganensis subsp. michiganensis on tomato. Plant Pathol. 2013, 62, 1114–1122. [Google Scholar] [CrossRef]
- Case, R.A.; MacDonald, G.M. A dendroclimatic reconstruction of annual precipitation on the western Canadian prairies since AD 1505 from Pinus flexilis James. Quat. Res. 1995, 44, 267–275. [Google Scholar] [CrossRef]
- Martinelli, N. Climate from dendrochronology: Latest developments and results. Glob. Planet. Chang. 2004, 40, 129–139. [Google Scholar] [CrossRef]
- Brandes, A.F.D.N.; Rizzieri, Y.C.; Tamaio, N.; Pace, M.R.; Barros, C.F. A global review on wood growth rings in lianas. Dendrochronologia 2022, 71, 125920. [Google Scholar] [CrossRef]
- Callado, C.; da Silva Neto, S.; Scarano, F.; Costa, C. Periodicity of growth rings in some flood-prone trees of the Atlantic Rain Forest in Rio de Janeiro, Brazil. Trees 2001, 15, 492–497. [Google Scholar] [CrossRef]
- Menezes, M.; Berger, U.; Worbes, M. Annual growth rings and long-term growth patterns of mangrove trees from the Bragança peninsula, North Brazil. Wetl. Ecol. Manag. 2003, 11, 233–242. [Google Scholar] [CrossRef]
- Verheyden, A.; Kairo, J.G.; Beeckman, H.; Koedam, N. Growth rings, growth ring formation and age determination in the mangrove Rhizophora mucronata. Ann. Bot. 2004, 94, 59–66. [Google Scholar] [CrossRef] [Green Version]
- Dietz, H.; Fattorini, M. Comparative analysis of growth rings in perennial forbs grown in an alpine restoration experiment. Ann. Bot. 2002, 90, 663–668. [Google Scholar] [CrossRef] [Green Version]
- Dietz, H.; Schweingruber, F.H. Annual rings in native and introduced forbs of lower Michigan, U.S.A. Can. J. Bot. 2002, 80, 642–649. [Google Scholar] [CrossRef]
- Dietz, H.; von Arx, G. Climatic fluctuation causes large- scale synchronous variation in radial root increments of perennial forbs. Ecology 2005, 86, 327–333. [Google Scholar] [CrossRef]
- Schweingruber, F.H.; Dietz, H. Annual rings in the xylem of dwarf shrubs and perennial dicotyledonous herbs. Dendrochronologia 2001, 19, 115–126. [Google Scholar]
- Schweingruber, F.H.; Poschlod, P. Growth rings in herbs and shrubs: Life span, age determination and stem anatomy. For. Snow Landsc. Res. 2005, 79, 195–415. [Google Scholar]
- Swart, E.D.; Groenwold, R.; Kanne, H.J.; Stam, P.; Marcelis, L.F.; Voorrips, R.E. Non-destructive estimation of leaf area for different plant ages and accessions of Capsicum annuum L. J. Hortic. Sci. Biotechnol. 2004, 79, 764–770. [Google Scholar] [CrossRef]
- Duke, N.C.; Zuleika, S.; Pinzon, M. Aging Rhizophora seedlings from leaf scar nodes: A technique for studying recruitment and growth in mangrove forests. Biotropica 1992, 24, 173–186. [Google Scholar] [CrossRef]
- Clark, S.L.; Hallgren, S.W. Can oaks be aged from bud scars? Southwest. Nat. 2004, 49, 243–246. [Google Scholar] [CrossRef]
- Zhang, X.; Li, H.; Hu, X.; Zheng, P.; Hirota, M.; Kamijo, T. Photosynthetic Properties of Miscanthus condensatus at Volcanically Devastated Sites on Miyake-jima Island. Plants 2020, 9, 1212. [Google Scholar] [CrossRef]
- Freschet, G.T.; Swart, E.M.; Cornelissen, J.H. Integrated plant phenotypic responses to contrasting above-and below-ground resources: Key roles of specific leaf area and root mass fraction. New Phytol. 2015, 206, 1247–1260. [Google Scholar] [CrossRef]
- Hu, H.; Li, F.L.; McCormack, M.L.; Huang, L.; Bao, W.K. Functionally divergent growth, biomass allocation and root distribution of two xerophytic species in response to varying soil rock fragment content. Plant Soil 2021, 463, 265–277. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Ackerly, D.D. Community assembly and shifts in plant trait distributions across an environmental gradient in coastal California. Ecol. Monogr. 2009, 79, 109–126. [Google Scholar] [CrossRef] [Green Version]
- Dwyer, J.M.; Hobbs, R.J.; Mayfield, M.M. Specific leaf area responses to environmental gradients through space and time. Ecology 2014, 95, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Lusk, C.H.; Warton, D.I. Global meta-analysis shows that relationships of leaf mass per area with species shade tolerance depend on leaf habit and ontogeny. New Phytol. 2007, 176, 764–774. [Google Scholar] [CrossRef] [PubMed]
- Damián, X.; Fornoni, J.; Domínguez, C.A.; Boege, K. Ontogenetic changes in the phenotypic integration and modularity of leaf functional traits. Funct. Ecol. 2018, 32, 234–246. [Google Scholar] [CrossRef] [Green Version]
- Dayrell, R.L.; Arruda, A.J.; Pierce, S.; Negreiros, D.; Meyer, P.B.; Lambers, H.; Silveira, F.A. Ontogenetic shifts in plant ecological strategies. Funct. Ecol. 2018, 32, 2730–2741. [Google Scholar] [CrossRef] [Green Version]
- Cavender-Bares, J.; Bazzaz, F.A. Changes in drought response strategies with ontogeny in Quercus rubra: Implications for scaling from seedlings to mature trees. Oecologia 2000, 124, 8–18. [Google Scholar] [CrossRef]
- Boege, K.; Marquis, R.J. Facing herbivory as you grow up: The ontogeny of resistance in plants. Trends Ecol. Evol. 2005, 20, 441–448. [Google Scholar] [CrossRef]
- Niinemets, Ü. Adaptive adjustments to light in foliage and whole-plant characteristics depend on relative age in the perennial herb Leontodon hispidus. New Phytol. 2004, 162, 683–696. [Google Scholar] [CrossRef]
- Niinemets, U. Key plant structural and allocation traits depend on relative age in the perennial herb Pimpinella saxifraga. Ann. Bot. 2005, 96, 323–330. [Google Scholar] [CrossRef] [Green Version]
- Moriuchi, K.S.; Winn, A.A. Relationships among growth, development and plastic response to environment quality in a perennial plant. New Phytol. 2005, 166, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Wright, I.J.; Reich, P.B.; Westoby, M.; Ackerly, D.D.; Baruch, Z.; Bongers, F.; Cavender-Bares, J.; Chapin, T.; Cornelissen, J.H.; Diemer, M.; et al. The worldwide leaf economics spectrum. Nature 2004, 428, 821–827. [Google Scholar] [CrossRef]
- Mason, C.M.; McGaughey, S.E.; Donovan, L.A. Ontogeny strongly and differentially alters leaf economic and other key traits in three diverse Helianthus species. J. Exp. Bot. 2013, 64, 4089–4099. [Google Scholar] [CrossRef] [Green Version]
- Reich, P.B. The world-wide ‘fast–slow’plant economics spectrum: A traits manifesto. J. Ecol. 2014, 102, 275–301. [Google Scholar] [CrossRef]
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: Meta-analyses of interspecific variation and environmental control. New Phytol. 2012, 193, 30–50. [Google Scholar] [CrossRef]
- Maherali, H.; Caruso, C.M.; Sherrard, M.E. The adaptive significance of ontogenetic changes in physiology: A test in Avena barbata. New Phytol. 2009, 183, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.M.; Donovan, L.A. Does investment in leaf defenses drive changes in leaf economic strategy? A focus on whole-plant ontogeny. Oecologia 2015, 177, 1053–1066. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Adler, P.B. When should plant population models include age structure? J. Ecol. 2014, 102, 531–543. [Google Scholar] [CrossRef] [Green Version]
- Whitmore, T. Canopy gaps and the two major groups of forest trees. Ecology 1989, 70, 536–538. [Google Scholar] [CrossRef]
- Fiorucci, A.S.; Fankhauser, C. Plant strategies for enhancing access to sunlight. Curr. Biol. 2017, 27, R931–R940. [Google Scholar] [CrossRef] [Green Version]
- Johnstone, J. Alpine plant life: Functional plant ecology of high mountain ecosystems. Mt. Res. Dev. 2021, 41, M1–M2. [Google Scholar] [CrossRef]
- Tilman, D. Plant Strategies and the Dynamics and Structure of Plant Communities. (MPB-26), Volume 26; Princeton University Press: Princeton, NJ, USA, 2020; Volume 26. [Google Scholar]
- Wang, Q.W.; Liu, C.; Robson, T.M.; Hikosaka, K.; Kurokawa, H. Leaf density and chemical composition explain variation in leaf mass area with spectral composition among 11 widespread forbs in a common garden. Physiol. Plant. 2021, 173, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Bloom, A.J.; Chapin, F.S., III; Mooney, H.A. Resource limitation in plants-an economic analogy. Annu. Rev. Ecol. Syst. 1985, 16, 363–392. [Google Scholar] [CrossRef]
- Freschet, G.T.; Violle, C.; Bourget, M.Y.; Scherer-Lorenzen, M.; Fort, F. Allocation, morphology, physiology, architecture: The multiple facets of plant above-and below-ground responses to resource stress. New Phytol. 2018, 219, 1338–1352. [Google Scholar] [CrossRef]
- Wang, X.; Taub, D.R. Interactive effects of elevated carbon dioxide and environmental stresses on root mass fraction in plants: A meta-analytical synthesis using pairwise techniques. Oecologia 2010, 163, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Henn, J.J.; Damschen, E.I. Plant age affects intraspecific variation in functional traits. Plant Ecol. 2021, 222, 669–680. [Google Scholar] [CrossRef]
- Ying, R.R.; Du, S.J.; Hu, P.J.; Zhao, Z.H.; Zhou, X.Y.; Tang, Y.T.; Qiu, R.L. Kinetic characteristics of Zn uptake by Potentilla griffithii Hook. f. var. velutina Card. J. Appl. Ecol. 2008, 19, 1349–1354. [Google Scholar]
- Qiu, R.L.; Thangavel, P.; Hu, P.J.; Senthilkumar, P.; Ying, R.R.; Tang, Y.T. Interaction of cadmium and zinc on accumulation and sub-cellular distribution in leaves of hyperaccumulator Potentilla griffithii. J. Hazard. Mater. 2011, 186, 1425–1430. [Google Scholar] [CrossRef] [PubMed]
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef] [Green Version]



| Cate. | Traits | Open Land | Understory Grassland | p-Value | ||
|---|---|---|---|---|---|---|
| No. of Samplings | Mean + SE | No. of Samplings | Mean + SE | |||
| Biomass and allocation | TDM (g) | 171 | 3.21 ± 0.27 | 126 | 2.93 ± 0.27 | NS |
| ABDM (g) | 171 | 1.13 ± 0.11 | 124 | 0.62 ± 0.065 | <0.001 | |
| BEDM (g) | 171 | 2.07 ± 0.17 | 126 | 2.31 ± 0.21 | NS | |
| R/S | 171 | 2.11 ± 0.097 | 124 | 4.41 ± 0.29 | <0.001 | |
| ABMF (g g−1) | 171 | 0.36 ± 0.008 | 124 | 0.22 ± 0.0076 | <0.001 | |
| BEMF (g g−1) | 171 | 0.64 ± 0.008 | 126 | 0.78 ± 0.0076 | <0.001 | |
| LMF (g g−1) | 171 | 0.31 ± 0.0077 | 126 | 0.21 ± 0.0074 | <0.001 | |
| SMF (g g−1) | 90 | 0.077 ± 0.0056 | 37 | 0.059 ± 0.0074 | NS | |
| FRMF (g g−1) | 165 | 0.26 ± 0.014 | 124 | 0.21 ± 0.017 | <0.01 | |
| CRMF (g g−1) | 171 | 0.55 ± 0.0094 | 126 | 0.69 ± 0.0095 | <0.001 | |
| The whole-plant morphology | TLN (No.) | 170 | 21.73 ± 2.13 | 124 | 11.93 ± 1.29 | <0.001 |
| TFM (No.) | 90 | 19.47 ± 2.35 | 37 | 14.99 ± 3.44 | NS | |
| TSN (No.) | 90 | 2.98 ± 0.27 | 39 | 2.01 ± 0.31 | <0.001 | |
| TLA (cm2) | 122 | 73.34 ± 4.53 | 126 | 64.77 ± 3.74 | NS | |
| StemD (mm) | 91 | 2.24 ± 0.062 | 39 | 2.11 ± 0.091 | NS | |
| StemL (cm) | 91 | 17.65 ± 0.78 | 37 | 22.74 ± 0.79 | <0.001 | |
| RootD (mm) | 171 | 7.55 ± 0.26 | 126 | 8.49 ± 0.27 | <0.01 | |
| TRL (cm) | 171 | 25.18 ± 1.28 | 126 | 24.41 ± 0.95 | NS | |
| FruitW (mg) | 90 | 10.72 ± 0.91 | 37 | 8.1 ± 0.32 | <0.05 | |
| SRL (cm g−1) | 171 | 21.98 ± 1.61 | 125 | 20.41 ± 2.77 | NS | |
| LMA (mg cm−2) | 121 | 10.89 ± 0.44 | 126 | 7.65 ± 0.14 | <0.001 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Lin, X.; Wei, D.; Bao, W.; Hu, B. Age Determination and Growth Characteristics of the Potentilla griffithii: A Comparison of Two Different Habitats in Western Sichuan Plateau, China. Plants 2023, 12, 2920. https://doi.org/10.3390/plants12162920
Zhang X, Lin X, Wei D, Bao W, Hu B. Age Determination and Growth Characteristics of the Potentilla griffithii: A Comparison of Two Different Habitats in Western Sichuan Plateau, China. Plants. 2023; 12(16):2920. https://doi.org/10.3390/plants12162920
Chicago/Turabian StyleZhang, Xiulong, Xingxing Lin, Dandan Wei, Weikai Bao, and Bin Hu. 2023. "Age Determination and Growth Characteristics of the Potentilla griffithii: A Comparison of Two Different Habitats in Western Sichuan Plateau, China" Plants 12, no. 16: 2920. https://doi.org/10.3390/plants12162920





