1. Introduction
Roses belong to the genus
Rosa, where considerable diversity exists within and between species. They are characterized by their ability to germinate everywhere and are perfectly adapted to pioneer conditions, where there are almost always extreme growing conditions. They also grow in climatic vegetation communities, except they occupy different places and may not be as lush there. As an ornamental plant, roses have been common throughout the world since time immemorial. Rosehips, a rich source of bioactive compounds beneficial to humans, are becoming increasingly prominent [
1,
2].
Functional foods or dietary supplements that protect humans from oxidative stress and many diseases have recently become increasingly popular. Roses (rosehips and petals) are used for various purposes, including protecting health and treating influenza, infections, inflammatory diseases, and chronic pain. In addition, they have beneficial effects on skincare and healing ulcers. They are also used in foods and beverages, such as tea, jams, and jellies. They have recently been used as an ingredient in probiotic drinks, yoghurts and soups as dietary supplements. They are known to be antioxidant, anti-inflammatory and antibacterial, to improve the immune system, and assist against respiratory, gastric, and intestinal problems. In relation to these properties, several main species of roses are essential. Among them,
R. gallica is very interesting since it is the most widespread European species and the parent species for several cultivars [
3,
4].
Song et al. [
5] demonstrated that
R. gallica petal extract promoted skin whitening and anti-wrinkle effects by regulating intracellular signaling, supporting its usefulness in cosmetic products for skin whitening. In addition, Jo et al. [
3] concluded that
R. gallica is promising for use as a functional ingredient in developing antiaging nutraceuticals. Ueno et al. [
6] studied the neuropsychological effects of a water-soluble extract of
R. gallica in male mice exposed to chronic stress and those under normal conditions. When the mice were exposed to stress,
R. gallica had an anti-stress effect. They concluded that
R. gallica has the potential as a medicinal plant that prevents stress. The medicinal effects of
R. subcanina are also widely known and confirmed. Their effects are similar to those of
R. canina, about which we found several studies and which is accepted as a control species in the
Rosa genus. Tumbas Šaponjac et al. [
7] confirmed that vitamin C and flavonoids are responsible for the antioxidant effect of rosehip tea, while only polyphenolics contribute to its antiproliferative effect. Daels-Rakotoarison et al. [
8] reported that
R. canina extract positively affects the respiratory activity of neutrophils. Ashtiyani et al. [
9] concluded that
R. canina fruit extract protects against renal dysfunction, oxidative stress and histological damage.
Uggla et al. [
10] found that the two main sugars in
R. dumalis and
R. rubiginosa were glucose and fructose. Demir et al. [
11] studied the organic acid and sugar compositions of
R. gallica,
R. canina,
R. dumalis,
R. gallica,
R. dumalis subsp.
boissieri and
R. hirtissima. They also determined glucose and fructose as the main sugars. The highest content was determined in
R. canina and
R. gallica. They concluded that there were no great differences in the content of analyzed sugars. Rosus et al. [
12] reported the total sugar content in various genotypes of roses, including
R. subcanina. They found great qualitative and quantitative variability among the analyzed genotypes. Adamczak et al. [
13] studied the bioactive composition of
R. canina,
R. dumalis,
R. glauca,
R. inodora,
R. jundzillii,
R. rubiginosa,
R. sherardii,
R. tomentosa,
R. villosa and
R. zalana. They found that the citric acid content was, on average higher than the ascorbic acid content. They also confirmed that there is great variability among genotypes. Javanmard et al. [
14] reported the ascorbic acid content of five wild native
R. canina species from Iran. Demir et al. [
11] determined the contents of citric, malic, and ascorbic acid, in addition to the mentioned sugars. Ascorbic acid was present in the highest concentrations in the hips and malic acid in the lowest concentrations. Statistically significant differences were observed among the samples. High content of carotenoids was noted by Hornero-Mendez and Minquez-Mosquera [
15], who listed β-carotene and lycopene as the main carotenoids. This is also consistent with other reports [
16,
17]. Olsson et al. [
18] concluded that rosehips have a high content of total carotenoids compared to other berries and small fruits. This was found by researching ten fruit species whose carotenoid content differed almost 150 times.
Kunc et al. [
19] studied the phenolic profile of
R. pendulina,
R. spinosissima and their hybrid
R. pendulina ×
spinosissima (
R. reversa). They found that out of 28 different phenolic compounds identified, quercetin-3-glucuronide was only present in the petals of the hybrid. The highest content of total phenolics was found in
R. spinosissima. Cunja et al. [
20] reported the phenolic profile of petals of
Rosa canina,
Rosa glauca,
Rosa rubuginosa and
Rosa sempervirens, as well as three modern cultivars ‘Rosarium Uetersen’, ‘Ulrich Brunner Fils’ and ‘Schwansse’. They found seven different anthocyanins and thirty-one flavonols, as well as 14 phenolic acids and their derivatives, 15 flavonols and 20 tannins. Cendrovski et al. [
21] reported that
R. rugosa petals are a rich source of phenolic compounds, which determine their antioxidant properties. The main polyphenolics were ellagitannins, accounting for 69 to 74% of all petal polyphenolics. Four other anthocyanins were identified: cyanidin 3,5-di-O-glucoside, peonidin 3-O-sophoroside, peonidin 3,5-di-O-glucoside and peonidin 3-O-glucoside, of which peonidin 3,5-di-O-glucoside accounted for approximately 85% of all anthocyanin compounds identified. Cunja et al. [
20] determined thirty-one flavonols in rose petals; their content varied widely among the species and cultivars studied. Shameh et al. [
22] reported eight different phenolic acids in the petals of six
Rosa species. They listed gallic acid, caffeic acid, chlorogenic acid,
p-coumaric acid, rutin, apigenin, cinnamic acid and quercetin.
Our research aimed to investigate the primary and secondary metabolites of rosehips and petals of
R. gallica compared to
R. subcanina, which, although not as common as some other wild roses, is also found in Slovenia.
R. gallica was chosen because it is still unexplored in various bioactive substances and is strongly present in Slovenia. Our analyses are based on the research of Kunc et al. [
1], who analyzed a broad spectrum of different bioactive compounds in roses. Considering that roses, in general, are highly variable and unexplored, our research will contribute to greater knowledge of the bioactive composition of rosehip fruits and flowers and encourage researchers to analyze the mentioned plants further. By comparing our results with those of other research, we can predict whether the petals and hips of
R. gallica, compared to
R. subcanina, are a rich source of bioactive compounds that could potentially be used in the food industry, alternative treatments, and cosmetic preparations.
3. Discussion
We determined the content of primary and secondary metabolites in the hips and petals of the not yet well-studied rose genotype
R. gallica, which grows naturally in the southwestern part of Slovenia, in Podgorje. We compared the data with the results measured in the control genotype
R. subcanina, which grows in the same area, close to
R. gallica. Five different types of sugars were determined in the hips: sucrose, glucose, fructose and the sugar alcohols sorbitol and mannitol. Sorbitol was present only in
R. gallica. The total sugar content in the hips of
R. gallica was 9.49 g/kg FW, whereas, in
R. subcanina, it was 6.12 g/kg FW. Yoruk et al. [
23] determined the sugar content of
R. iberica,
R. canina,
R. villosa,
R. dumalis and
R. pisiformis. Fructose content ranged from 13.58 to 18.44 g/kg FW, glucose from 6.89 to 10.04 g/kg FW and sucrose from 0.57 to 5.61 g/kg FW. The content of all sugars in our experiment was much lower than in the experiment of Yoruk et al. [
23]. The sucrose content of our samples was within the range reported by Yoruk et al. [
23]. It was the sugar with the lowest content. As Yoruk et al. [
23] noted, such differences in content are due to environmental conditions such as climate, soil structure and plant genetics. When plants are exposed to drought stress, solid compounds, especially sugars, are synthesised to regulate osmotic potential [
24]. Similar results were also reported by Abaci et al. [
25], who studied the sugar content of
Rosa iberica Stev. The total content was 267.4 g/kg FW, with the major sugar being glucose, followed by fructose, sorbitol, and sucrose. Their results also showed extremely high values of total sugar content compared to ours. The rosehips investigated by Yoruk et al. [
23] and Abaci et al. [
25] were grown in Turkey, in the area of Lake Van [
23] and are widespread in the north and east Anatolia [
25], where there are completely different conditions for growth compared to the rosehips that we included in the research. We assume that rosehips in Turkey were extremely more exposed to stress conditions and that the lack of water in 2021 [
1] in our area was so small compared to the Turkish stress conditions that it did not cause such an intense increase in sugars. Similarly, high values were also reported by Demir et al. [
11] and Rosu et al. [
12]. If the results are compared with those of the research conducted by Cunja et al. [
26], who analyzed samples of
R. canina, which also grew in Slovenia, it can be seen that the results are quite similar. For easier comparison, we converted our FW values to dry weight (DW), which is 37% FW in our case. The total sugar content was thus 2.26 and 3.5 g/kg DW. Cunja et al. [
26] reported that the total sugar contents of
R. canina were between 0.26 and 0.48 g/kg DW. Our samples thus had higher sugar contents. The increase in sugar content may be due to higher altitude and greater drought stress than the samples studied by Cunja et al. [
26]. It should be noted that the mentioned research involved different genotypes, which additionally contributes to the different sugar contents.
Organic acids were citric, malic, quinic, shikimic and fumaric. It can be seen that the total acid content in our samples was higher than that in the literature. Cunja et al. [
26] reported that citric acid was the most important organic acid detected in rosehips (up to 58% of the total organic acids). In our samples, we found a higher content of quinic acid and a lower one of citric acid. However, citric acid was immediately after quinic acid. Peña et al. [
27] reported that the total organic acid content of
R. canina and
R. rubiginosa ranged from 46.2 g/kg to 73.2 g/kg FW. Okatan et al. [
28] reported that the citric acid content in their samples of
R. canina was between 15.9 and 22 g/kg FW. Quinic acid, the most available acid in our experiment (44.07 and 55.11 g/kg FW), ranged from 48 to 72 g/kg FW in their samples. Quinic acid was also the most available organic acid in the hybrid
R. pendulina ×
spinosissima (48.8 g/kg FW), while the content of this acid in the original species
R. pendulina was only 0.29 g/kg FW in the experiment mentioned by Kunc et al. [
29]. As reported by Wang et al. [
30], the main indicator of fruit quality is the content and ratio between soluble sugars and organic acids. Akagić et al. [
31] reported that the ratio between sugars and acids strongly influences the fruit’s taste, shelf life and nutritional properties and are reliable indicators of consumer acceptability. In addition, changes in the content and composition of sugars and organic acids are also reflected in changes in the quality of fresh fruit and its products. Sugars also participate in the biosynthesis of polyphenolics, which means that the more sugar some fruit contains, the higher the polyphenol content is essential because of the nutrients that polyphenolics add to food. For an easier idea of how sweet or sour the fruits we analyzed were, we compared the results with those reported by Mahmood et al. [
32]. Namely, they found that the total sugar content (sum of sucrose, glucose, and fructose) for ripe strawberries was from 48 to 54 g/kg FW, and for mulberries, the values ranged from 79.3 g/kg to 143.9 g/kg FW. The total content of organic acids in the studied strawberries varied between 13 and 16 g/kg. Mikulic-Petkovsek et al. [
33] reported that the organic acid content of 20 grape varieties varied between 5.8 and 10.8 g/kg FW. The total sugar content of blueberries ranged from 49.99 to 111.67 g/kg FW, and the organic acid content ranged from 2.50 to 14.23 g/kg FW [
34].
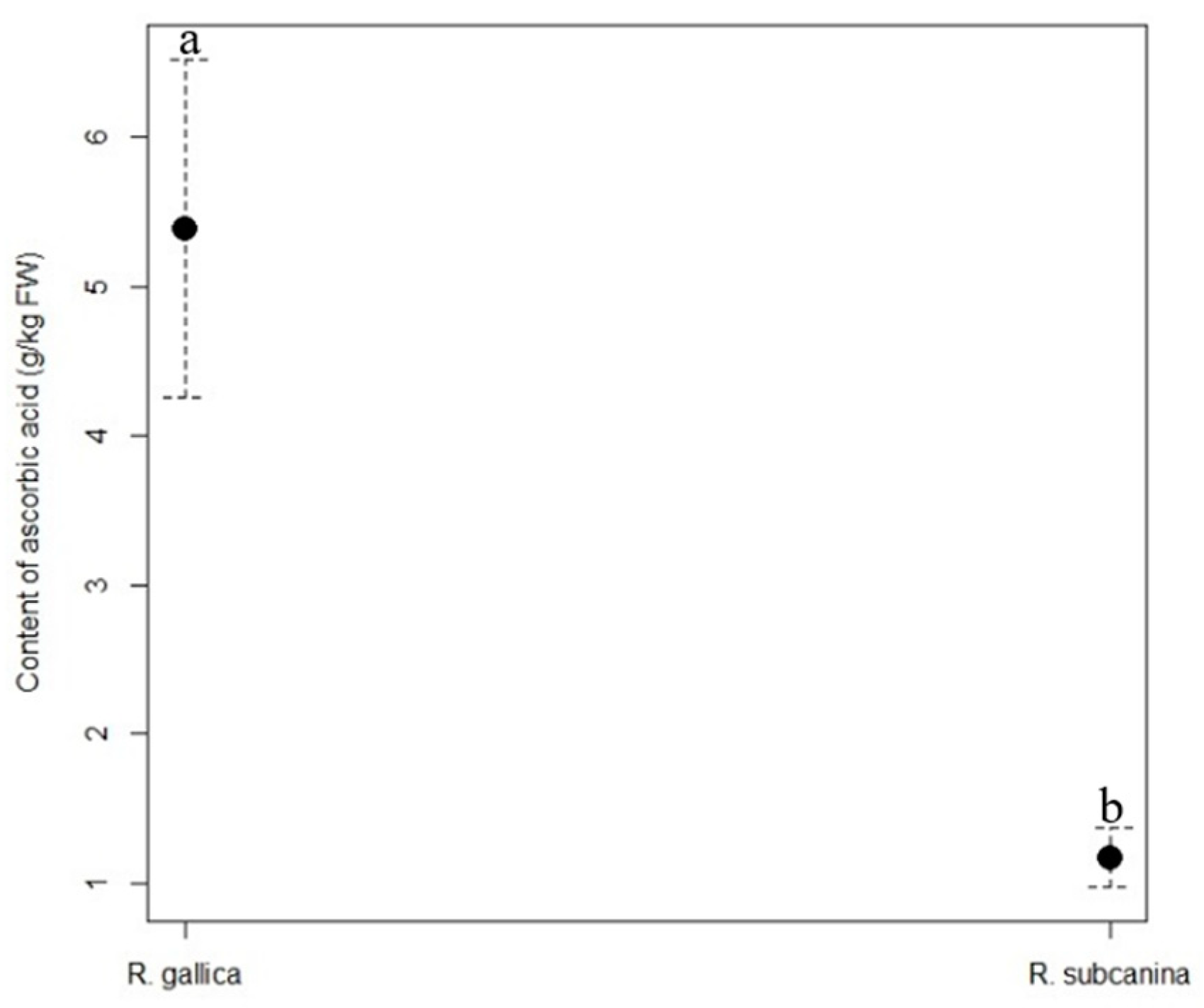
The content of ascorbic acid Is highly dependent on a variety of factors. The most Important are cultivar, stage of ripeness, altitude etc. The content of ascorbic acid in our samples was 5.39 g/kg FW for
R. gallica and 1.17 g/kg FW for
R. subcanina. Comparing our results with the results of an experiment conducted by Rosus et al. [
12], we note that the mentioned authors determined higher contents of ascorbic acid, from 8.7 g/kg FW in
R. rubiginosa to 6.2 g/kg FW in
R. caesia. Alp et al. [
35] determined the ascorbic acid content of rosehips from 10 different wild
R. dumalis genotypes. The content ranged from 4.02 to 5.11 g/kg FW. In previous studies, the ascorbic acid content in rosehip fruits has been quite variable, with a range of 1.80 to 9.65 g/kg FW [
36,
37,
38,
39,
40]. Krzaczek et al. [
41] reported that the taxonomic assignment level below species plays a major role in ascorbic acid accumulation. Roman et al. [
39] also studied ascorbic acid in samples of
R. canina from Transylvania. They found that the ascorbic acid content of frozen samples grown at 1250 m above sea level was 3.6 g/kg, and of those grown at 440 m was 1.12 g/kg of frozen pulp. They found a good correlation between ascorbic acid content in rosehips and altitude. The site’s altitude in our experiment is about 500 m above sea level, and the ascorbic acid content is even lower than that found by Roman et al. [
39]. Kunc et al. [
29] reported that the content of ascorbic acid in the rosehips of
R. pendulina ×
spinosissima was 10.45 g/kg FW, which is almost twice that in
R. penduline, with 5.30 g/kg FW. Our experiment found that the ascorbic acid content of the two genotypes we studied was very similar to the content measured in
R. pendulina in an experiment described by Kunc et al. [
29]. Medveckiene et al. [
42] studied five different rosehips, including
R. canina, and found that the content of the above acid was 3.85 g/kg FW, the lowest of all the rosehips studied. The highest content (7.4 g/kg FW) was determined in
R. rugosa ‘Rubra’.
Five carotenoids (lutein, zeaxanthin, lycopene, α-carotene, and ẞ-carotene) were determined in
R. gallica and
R. subcanina samples. Total carotenoid content was higher in
R. gallica than in
R. subcanina. Alp et al. [
35] found that the total carotenoid content of 10
R. dumalis genotypes ranged from 47 to 85 mg/100 g FW. Rosus et al. [
12] reported that the total carotenoid content of
R. subcanina was 34.95 mg/100 g FW. In our samples, ẞ-carotene was present in the largest proportions (23.37 and 27.56 mg/100 g FW). Zhong et al. [
43] studied the carotenoid content of
R. rubiginosa, R. multiflora, R. virginiana and
R. rugosa. They found a large difference in carotenoid composition among the roses.
R. virginiana had the most diverse carotenoid composition and the highest carotenoid content. It was found that the group of lycopene together with ẞ-carotene accounted for 28 to 54% of the content of all carotenoids. Medveckiene et al. [
42] reported that the content of ẞ-carotene ranged from 3.95 mg/100 g to 31.4 mg/100 g. In our samples, lycopene content was found to be 2.19 mg/100 g FW to 2.21 mg/100 g FW. The carotenoid with the lowest content in our samples was zeaxanthin, 0.29 to 0.33 mg/100 g FW. Similar contents were also reported by Medveckiene et al. [
42], 0.23 to 0.32 mg/100 g. As can be seen from the above data, rosehips are a rich source of carotenoids, but there are differences in the content, which is the result of genetic variations, maturity level, agro-meteorological conditions, growing conditions, storage, and analysis method [
44]. Because of all these factors, we assume that the content in our samples is also in the lower range of the average carotenoid content compared to data from the literature.
Total phenolics content in pulp with skin was extremely high in
R. gallica (15.8 g/kg FW) compared to
R. subcanina (5.31 g/kg FW). The total content of phenolic compounds in seeds was 1.27 g/kg FW in
R. subcanina and 1.71 g/kg FW in
R. gallica. Considering how low the sugar content determined in the samples was, the lower content of phenolic substances in the samples of
R. subcanina, compared to
R. gallica, is also logical. Flavones were determined only in the fruit pulp with skin and dihydrochalcones in the seeds. Najda and Buczkowska [
45] investigated the phenolic content of 5 different rosehips. The phenolic content was between 1.1 g/kg FW and 2.2 g/kg FW. Pena et al. [
27] reported the total phenolic content in
Rosa spp. Varied between 0.3 g/kg and 14 g/kg. Unlike the aforementioned studies, much higher values are reported by Demir et al. [
11], who found that the total content of phenolic compounds in
R. dumalis subs.
Boissieri was 52.94 g/kg and in
R. canina 31.08 g/kg.
We determined 53 phenolic compounds in the petals of
R. gallica compared to only 31 in
R. subcanina. Cunja et al. [
20], who studied
R. canina, also determined 31 different phenolic compounds in this species, morphologically very similar to
R. subcanina. Cendrowski et al. [
21] determined 20 phenolic compounds in
R. rugosa petals identified by UPLC-ESI-MS. Ellagitannins represented the major fraction, accounting for 69 to 74% of petal polyphenolics. In our samples, gallotannins dominated in
R. subcanina, accounting for up to 81% of the phenolic compounds identified. In
R. gallica, flavonols dominated, with a proportion of 47%, immediately followed by gallotannins with 40%. In contrast to Cendrowski et al. [
21], Kumar et al. [
46], Velioglu and Mazza [
47] and Ochir et al. [
48], we determined quercetin and kaempferol derivatives in our samples. Total phenolic compound content ranged from 22.08 g/kg FW (2012 season) to 25.90 g/kg FW (2013 season) in Cendrowski et al. [
21]. In our studied samples, these values were 35.5 g/kg FW for
R. subcanina and 46.9 g/kg FW for
R. gallica. In comparison with the research results of Kunc et al. [
1,
19], who studied the phenolic composition of the flowers of
R. pendulina,
R. spinosissima and their hybrids, we found that the petals of
R. gallica and
R. subcanina had much lower values than those of the aforementioned roses. Of the flavonols, Cunja et al. [
20] determined quercetin acetyl hexoside rhamnoside, kaempferol 3-galactoside and kaempferol 3-glucoside, which were not present in our samples. Catechin, epicatechin and procyanidins were present only in the leaves but not in the petals, as in the samples of
R. gallica that we analyzed. In
R. subcanina, we could not determine catechin and procyanidin dimer 3 and procyanidin trimer 1. Cunja et al. [
20] found that quercetins accounted for 63.7% of flavonols. In our samples, these values were a little higher. In
R. gallica, they accounted for 82% and in
R. subcanina, 73% of the total flavonols. Schieber et al. [
49] described that quercetin-3-rhamnoside was present in trace amounts in the petals of
R. damascena. In our samples, it was the most abundant of the flavonols. Its contents were 0.36 g/kg FW in
R. gallica and 0.25 g/kg FW in
R. subcanina. Wan et al. [
50] showed that kaempferol-3-O-rhamnoside was the major flavonol in the petals of
R. damascena cultivars. Comparing the results of our two genotypes, it can be seen that we also identified the mentioned compound in our samples, but its content was not dominant. The content of the anthocyanin cyanidin-3-glucoside in our two genotypes was 0.18 g/kg FW (
R. gallica) and 0.04 g/kg FW (
R. subcanina). Considering the flower color, the low anthocyanin content in the petals of
R. subcanina was expected. Kunc et al. [
19] reported slightly higher values, 0.19 and 0.24 g/kg FW. Considering the anthocyanin content of other plants for comparison, Zannou et al. [
51], for example, studied orange blossom (
Echium amoenum), an annual herb native to the Mediterranean region, an excellent source of anthocyanins and used in various forms because of its biological activity. The dominant anthocyanin was cyanidin-3-glucoside, ranging from 0.03 to 1.13 g/kg. Li et al. [
52] investigated the content of anthocyanins in 51 edible wildflowers.
Lilium brownii, with a cyanidin-3-glucoside content of 0.106 mg/100 g, and
Ipomoea cairica 0.109 g/kg, were highlighted as those with lower contents. The highest contents were recorded for
Jatropha integerrima 6.41 g/kg and
Pelargonium hortorum 4.97 g/kg.