Isolation of Volatile Compounds by Microwave-Assisted Extraction from Six Veronica Species and Testing of Their Antiproliferative and Apoptotic Activities
Abstract
:1. Introduction
2. Results
2.1. Extraction of Volatile Components from Six Veronica Species
2.1.1. Composition of Essential Oil
2.1.2. Composition of Hydrosols
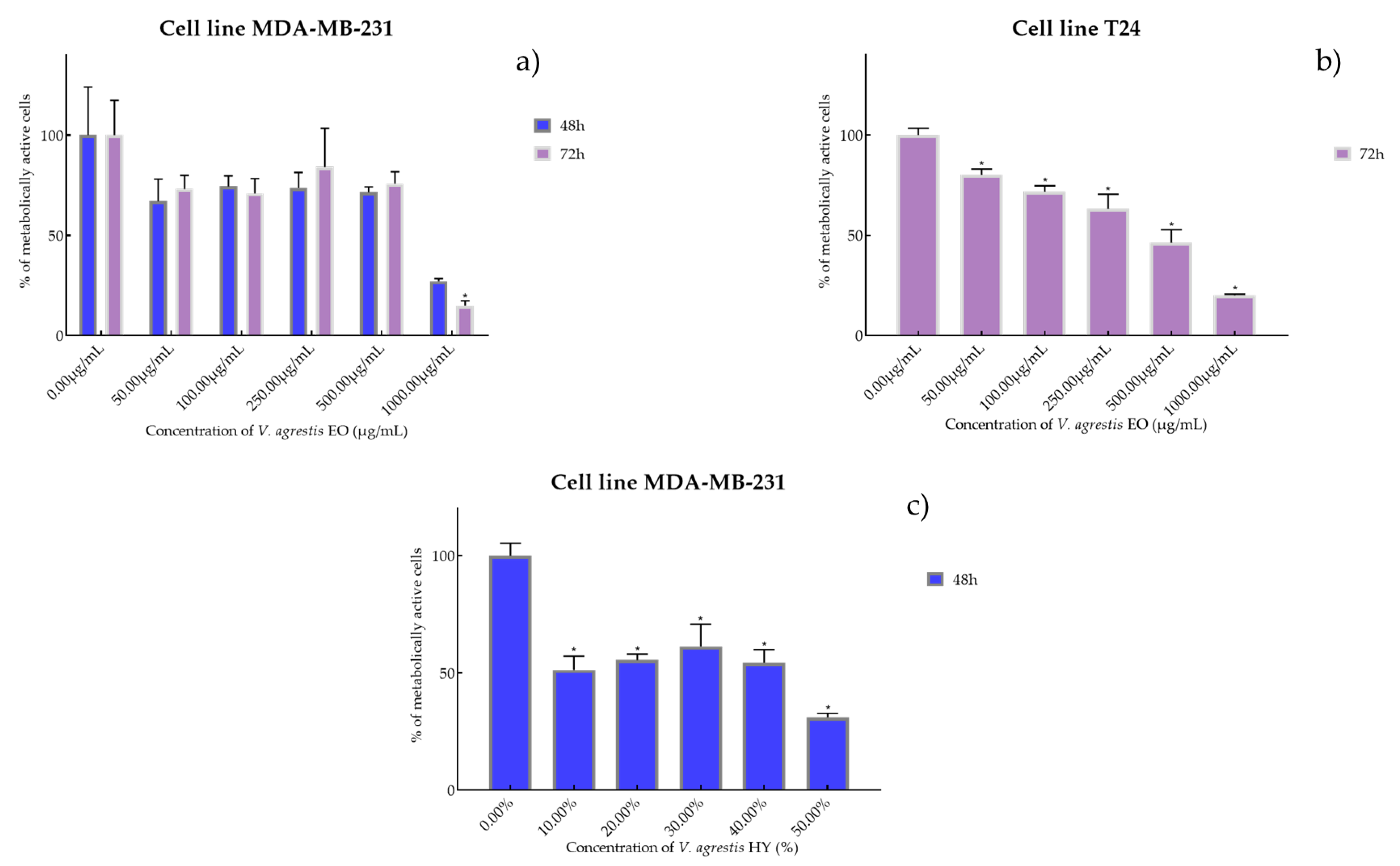
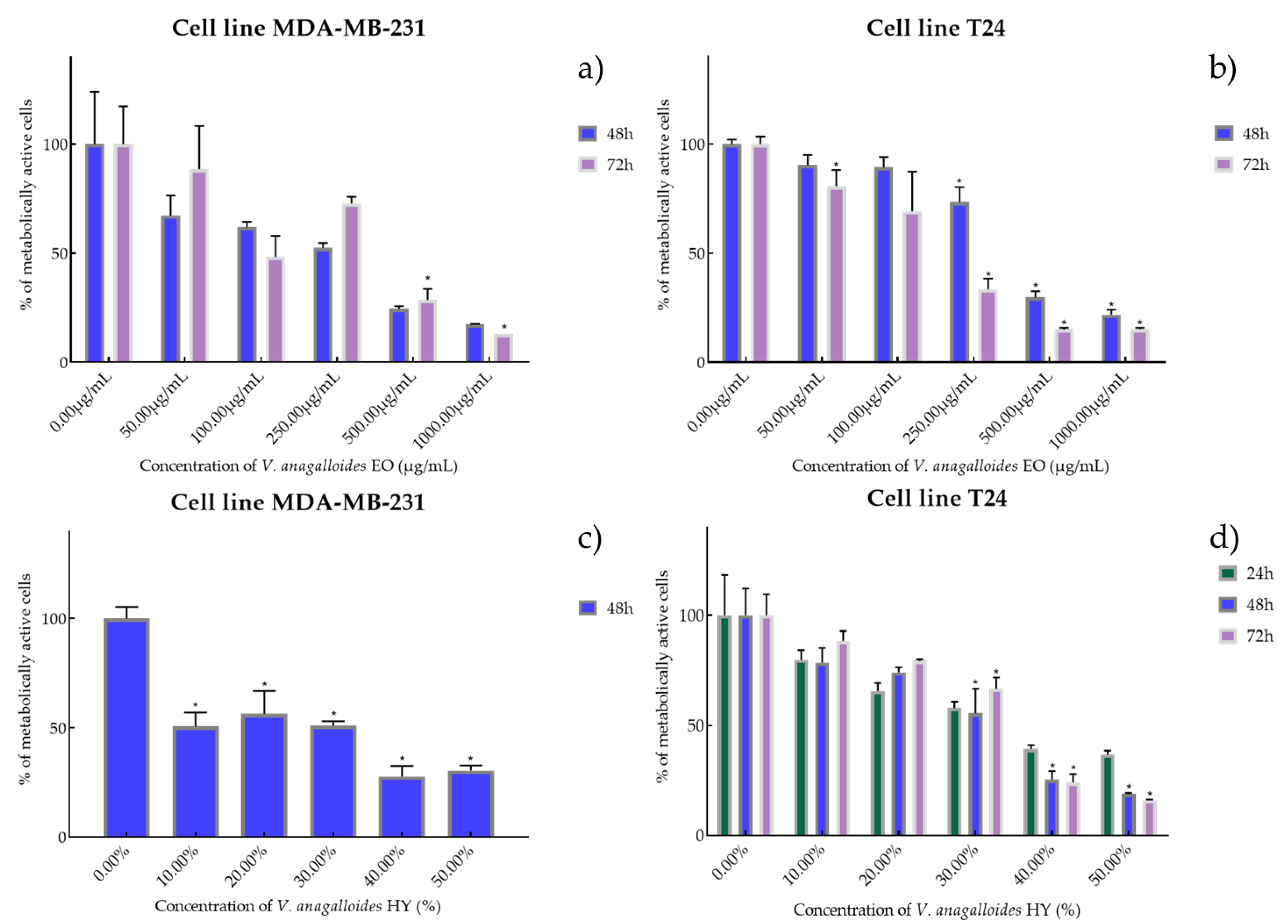
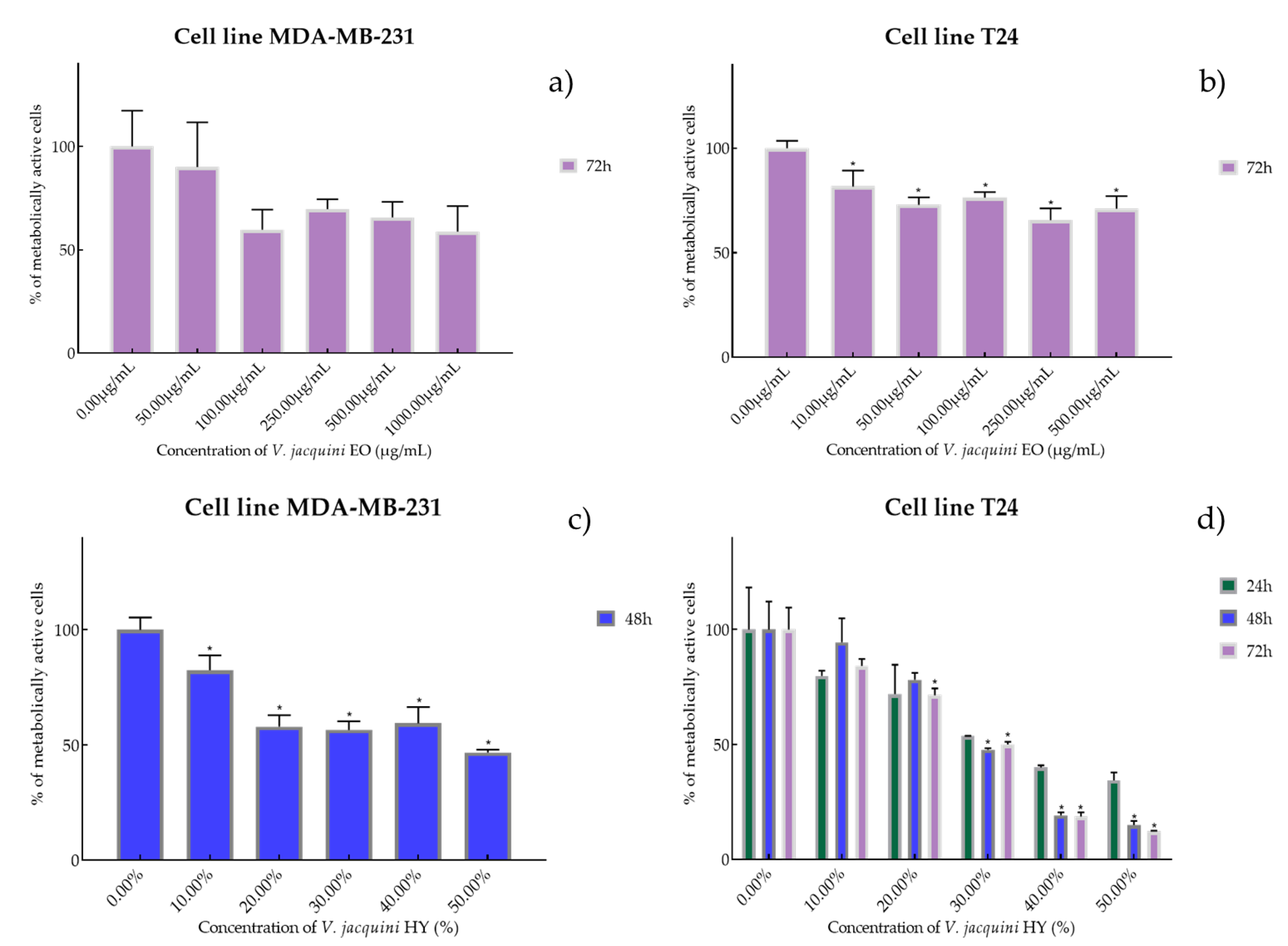
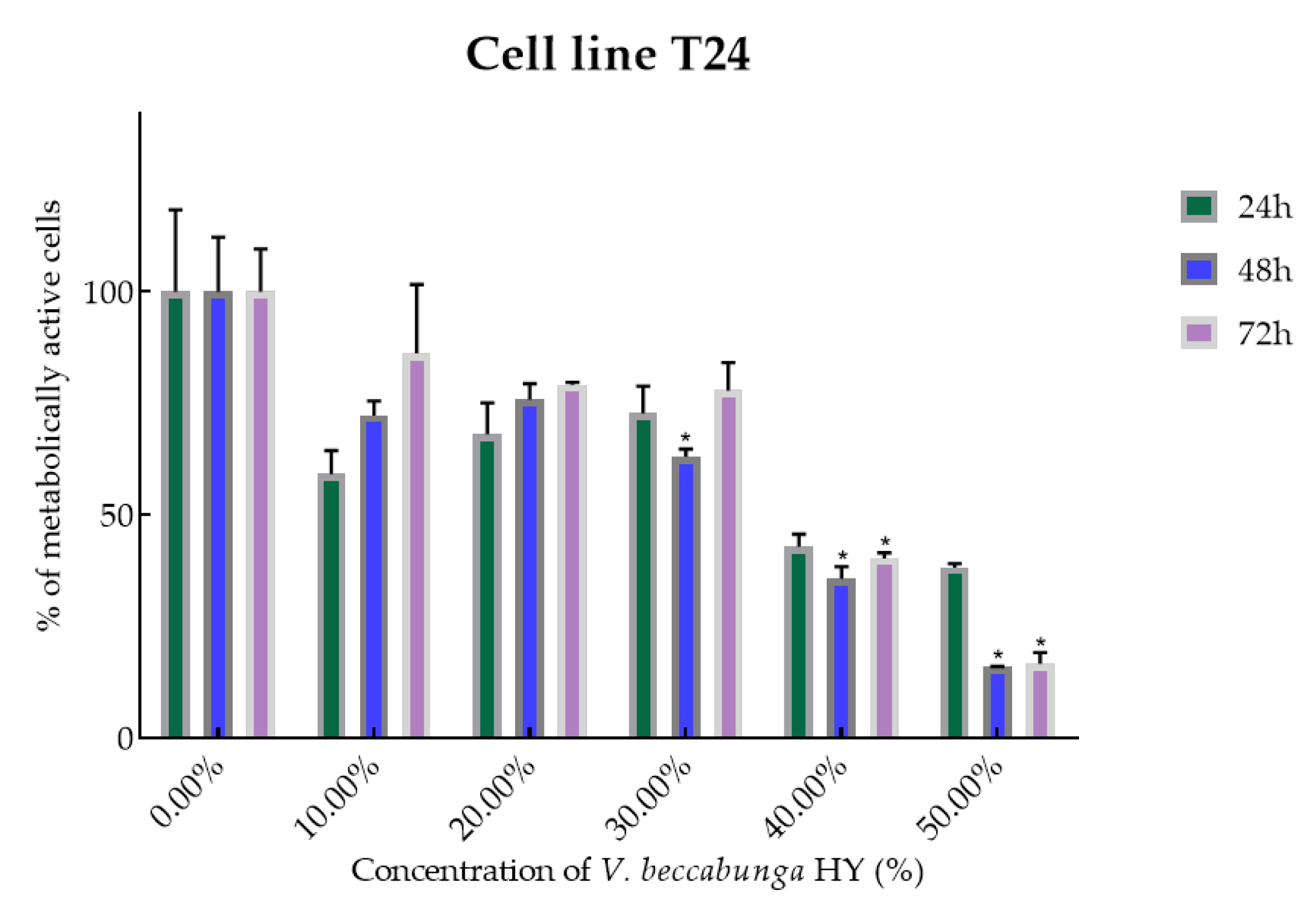
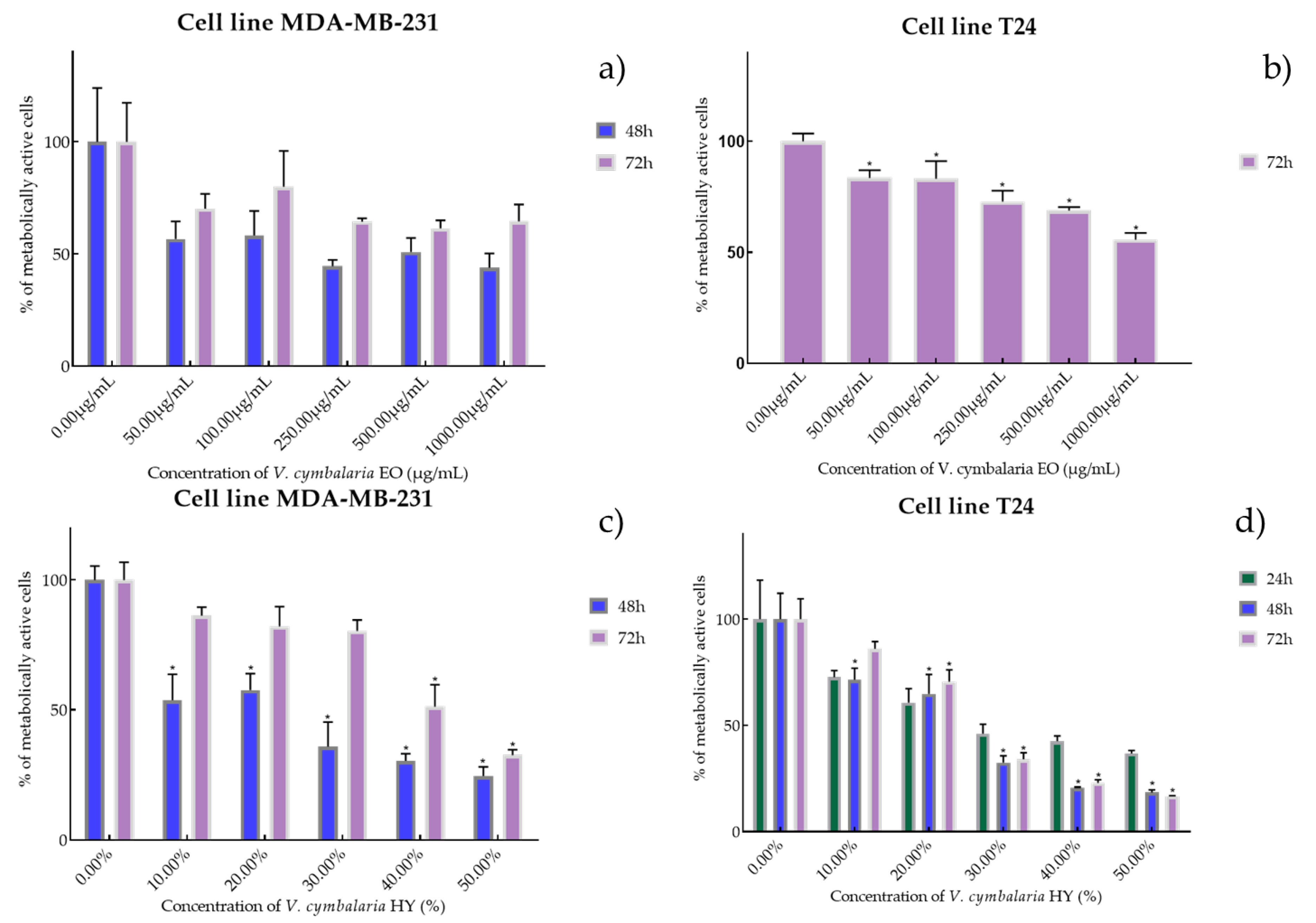
2.2. Cell Viability and Proliferation Using MTT Assay
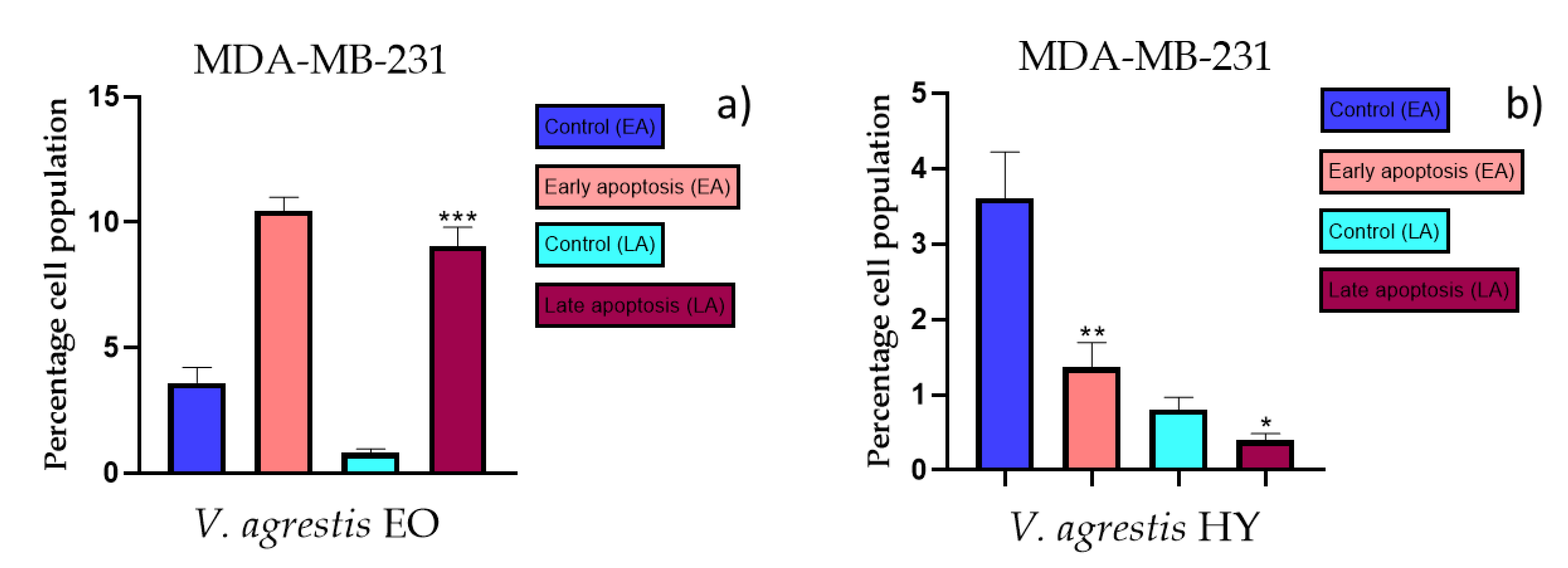
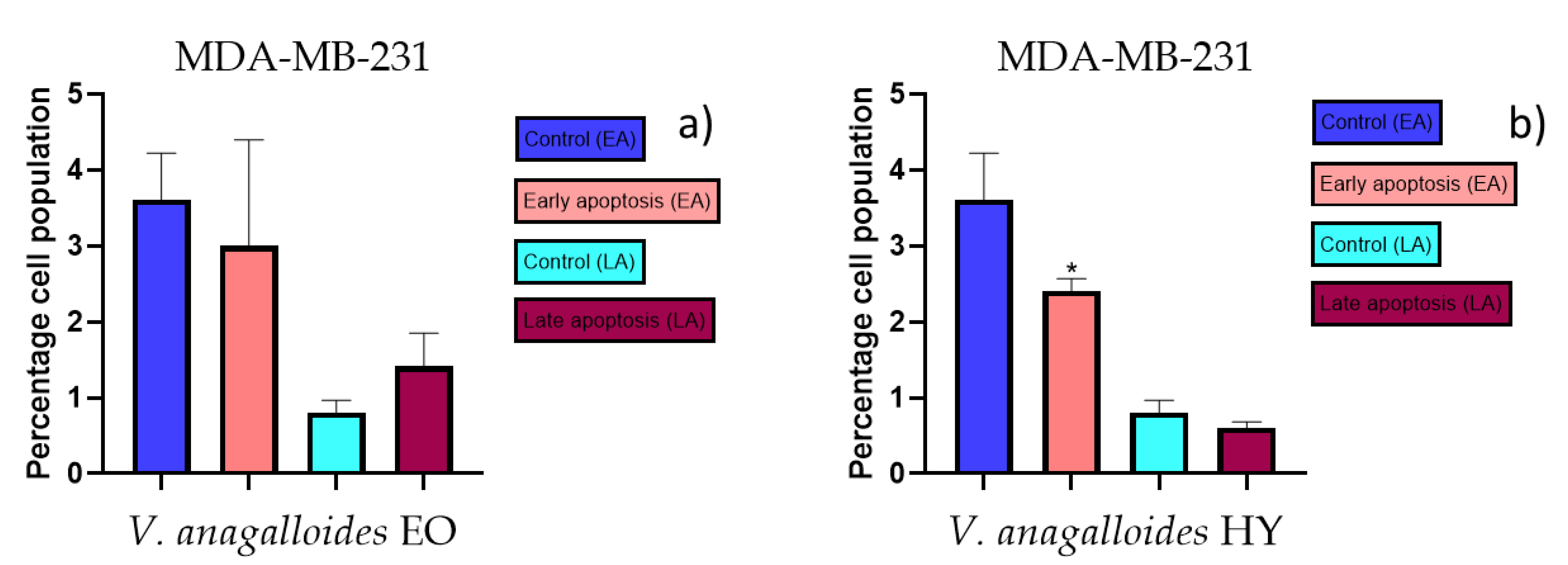
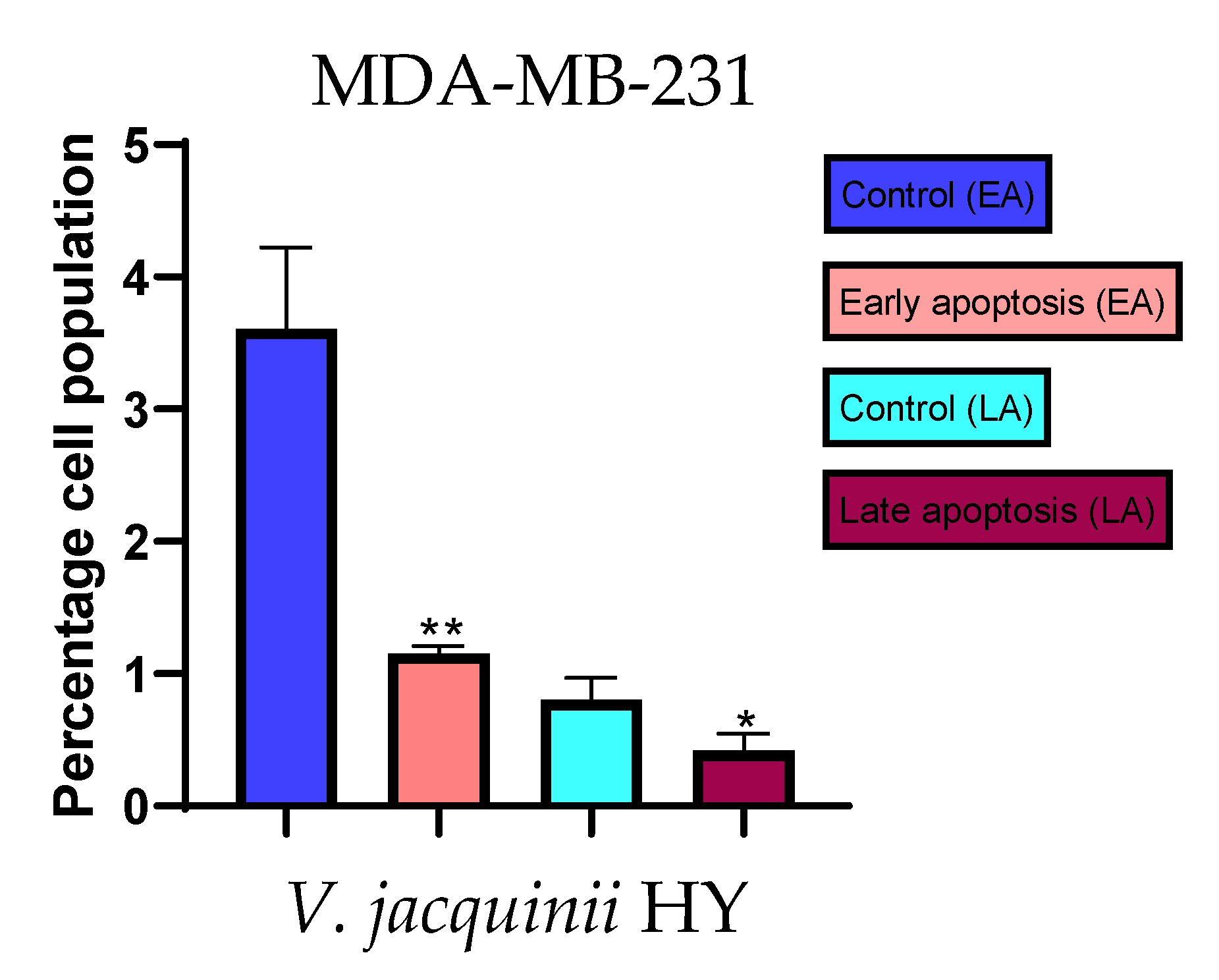
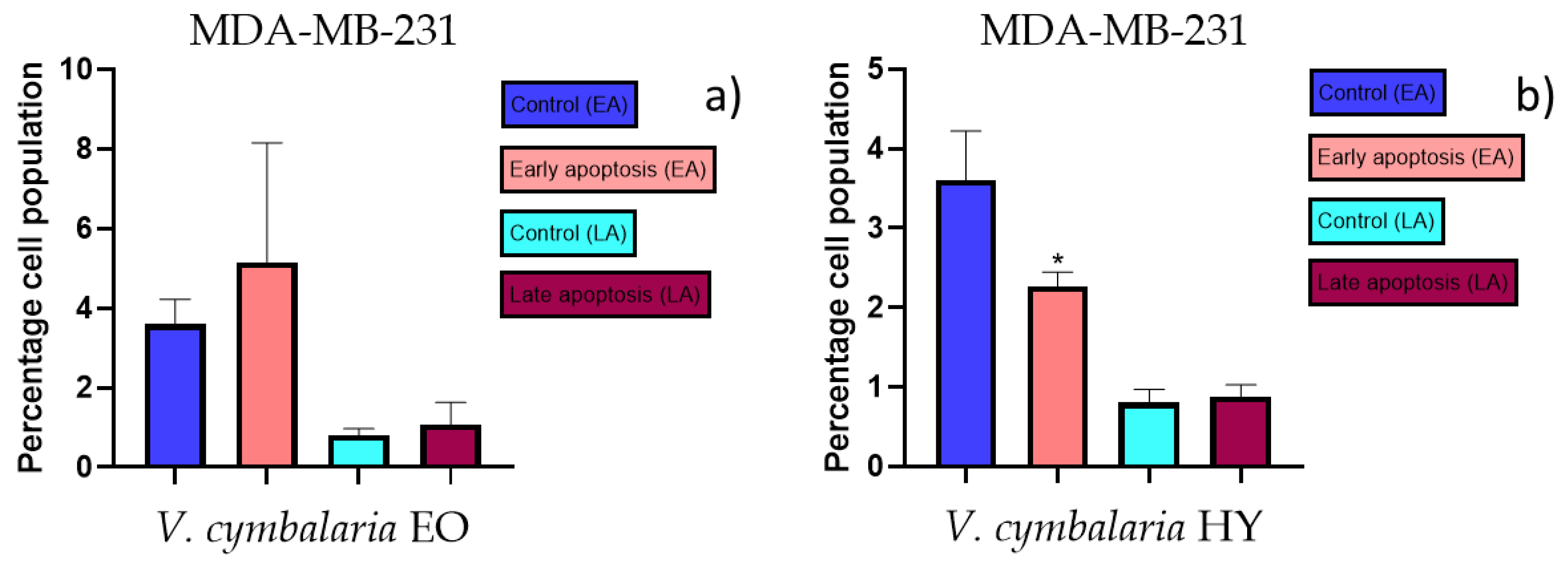
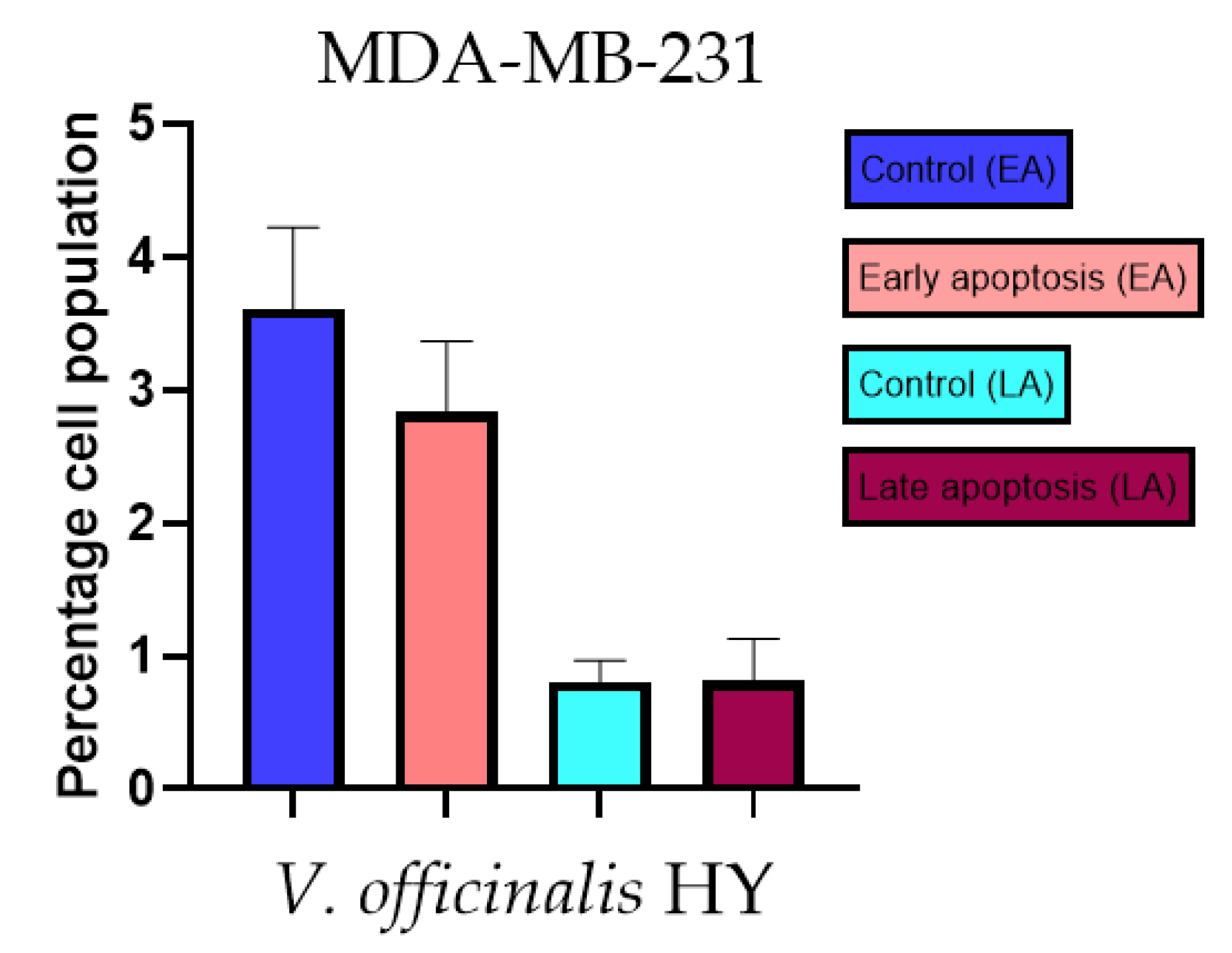
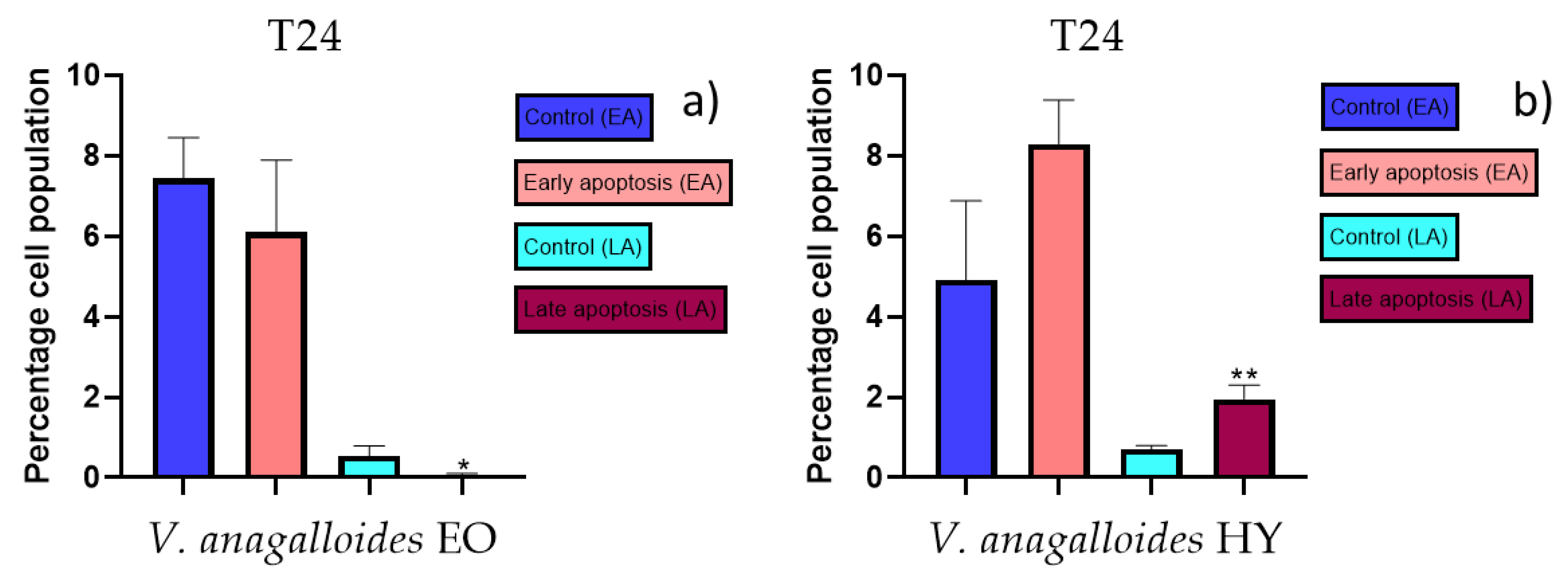
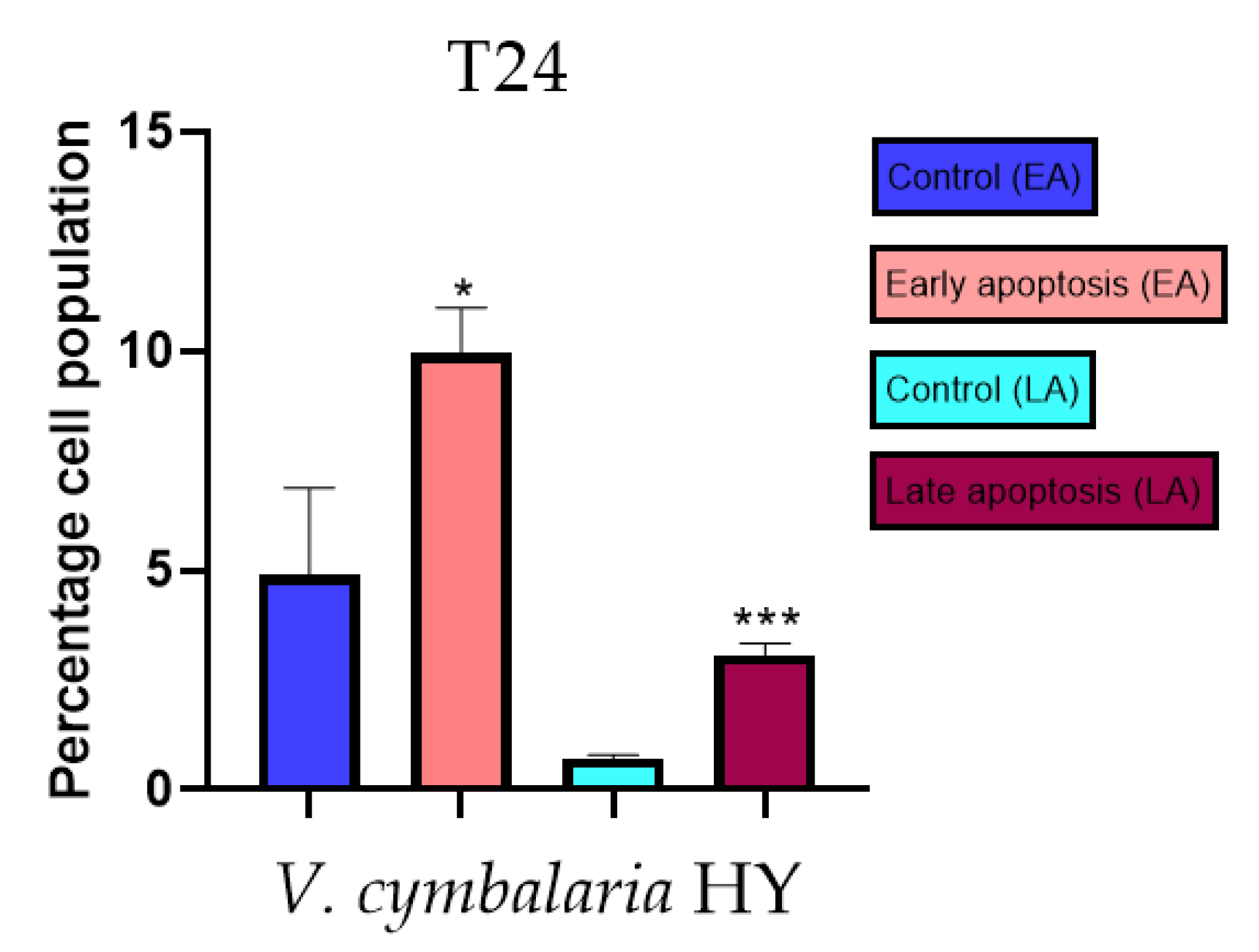
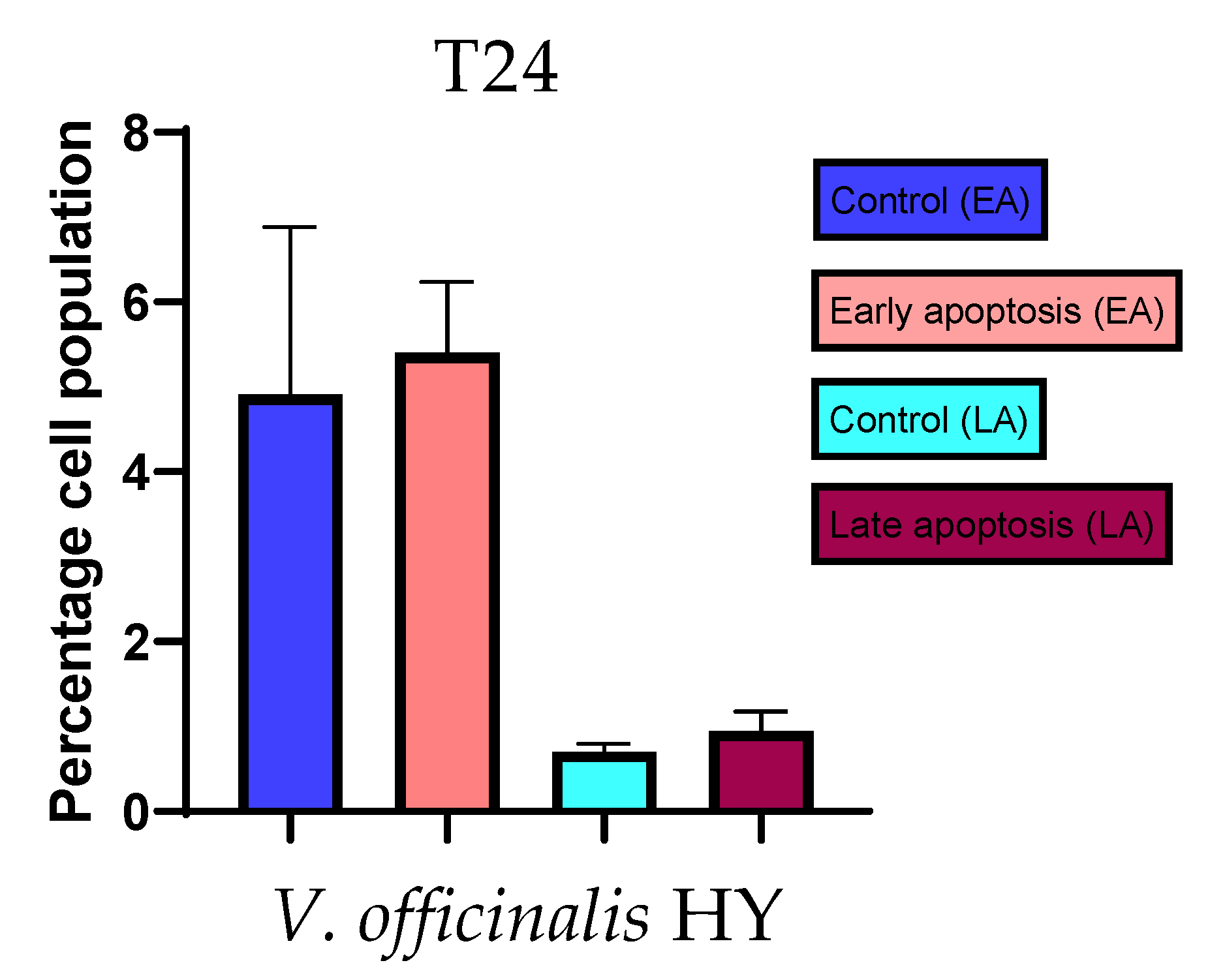
2.3. Apoptotic Activity
3. Discussion
4. Materials and Methods
4.1. Preparation, Extraction, and Identification of Volatile Compounds from Six Veronica Species
4.1.1. Preparation of Plant Material from Six Veronica Species
4.1.2. Extraction of Volatile Compounds
4.1.3. Identification of Volatile Compounds
4.2. Cell Viability and Proliferation Were Determined by Measuring Cellular Metabolism Using MTT Assay
4.3. Apoptotic Activity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fitzgerald, M.; Heinrich, M.; Booker, A. Medicinal Plant Analysis: A Historical and Regional Discussion of Emergent Complex Techniques. Front. Pharmacol. 2019, 10, 1480. [Google Scholar] [CrossRef] [PubMed]
- Dunkić, V.; Nazlić, M.; Ruščić, M.; Vuko, E.; Akrap, K.; Topić, S.; Milović, M.; Vuletić, N.; Puizina, J.; Jurišić Grubešić, R.; et al. Hydrodistillation and Microwave Extraction of Volatile Compounds: Comparing Data for Twenty-One Veronica Species from Different Habitats. Plants 2022, 11, 902. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Wang, F.; Liu, J.; Zou, Y.; Chen, X. A Comparison of Volatile Fractions Obtained from Lonicera macranthoides via Different Extraction Processes: Ultrasound, Microwave, Soxhlet Extraction, Hydrodistillation, and Cold Maceration. Integr. Med. Res. 2015, 4, 171–177. [Google Scholar] [CrossRef]
- Vrca, I.; Šćurla, J.; Kević, N.; Burčul, F.; Čulić, V.Č.; Bočina, I.; Blažević, I.; Bratanić, A.; Bilušić, T. Influence of Isolation Techniques on the Composition of Glucosinolate Breakdown Products, Their Antiproliferative Activity and Gastrointestinal Stability of Allyl Isothiocyanate. Eur. Food Res. Technol. 2022, 248, 567–576. [Google Scholar] [CrossRef]
- Vrca, I.; Ramić, D.; Fredotović, Ž.; Možina, S.S.; Blažević, I.; Bilušić, T. Chemical Composition and Biological Activity of Essential Oil and Extract from the Seeds of Tropaeolum majus L. var. altum. Food Technol. Biotechnol. 2022, 60, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Ríos, J.L. Essential Oils. In Essential Oils in Food Preservation, Flavor and Safety; Elsevier: Amsterdam, The Netherlands, 2015; pp. 3–10. ISBN 978-0-12416-641-7. [Google Scholar]
- Nazlić, M.; Akrap, K.; Kremer, D.; Dunkić, V. Hydrosols of Veronica Species—Natural Source of Free Volatile Compounds with Potential Pharmacological Interest. Pharmaceuticals 2022, 15, 1378. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, S.; Serio, A.; López, C.C.; Paparella, A. Hydrosols: Biological Activity and Potential as Antimicrobials for Food Applications. Food Control 2018, 86, 126–137. [Google Scholar] [CrossRef]
- Hamedi, A.; Moheimani, S.M.; Sakhteman, A.; Etemadfard, H.; Moein, M. An Overview on Indications and Chemical Composition of Aromatic Waters (Hydrosols) as Functional Beverages in Persian Nutrition Culture and Folk Medicine for Hyperlipidemia and Cardiovascular Conditions. J. Evid.-Based Complement. Altern. Med. 2017, 22, 544–561. [Google Scholar] [CrossRef]
- Twaij, B.M.; Hasan, M.N. Bioactive Secondary Metabolites from Plant Sources: Types, Synthesis, and Their Therapeutic Uses. Int. J. Plant Biol. 2022, 13, 4–14. [Google Scholar] [CrossRef]
- Doğan, Z.; Genç, Y.; Harput, Ü.; Karadeniz Pekgöz, A.; Saraçoğlu, İ. Chemical Profiling and Cytotoxic Activity of Aqueous Extract of Veronica peduncularis M.Bieb.: A Chemotaxonomical Approach. İstanbul J. Pharm. 2021, 51, 372–377. [Google Scholar] [CrossRef]
- Xue, H.; Chen, K.X.; Zhang, L.Q.; Li, Y.M. Review of the Ethnopharmacology, Phytochemistry, and Pharmacology of the Genus Veronica. Am. J. Chin. Med. 2019, 47, 1193–1221. [Google Scholar] [CrossRef] [PubMed]
- Kostadinova, E.P.; Alipieva, K.I.; Kokubun, T.; Taskova, R.M.; Handjieva, N.V. Phenylethanoids, Iridoids and a Spirostanol Saponin from Veronica turrilliana. Phytochemistry 2007, 68, 1321–1326. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Shetty, M.S.; Anil Kumar, N.V.; Živković, J.; Calina, D.; Docea, A.O.; Emamzadeh-Yazdi, S.; Kılıç, C.S.; Goloshvili, T.; Nicola, S.; et al. Veronica Plants—Drifting from Farm to Traditional Healing, Food Application, and Phytopharmacology. Molecules 2019, 24, 2454. [Google Scholar] [CrossRef] [PubMed]
- Mocan, A.; Vodnar, D.C.; Vlase, L.; Crișan, O.; Gheldiu, A.M.; Crișan, G. Phytochemical Characterization of Veronica officinalis L., V. teucrium L. and V. orchidea Crantz from Romania and Their Antioxidant and Antimicrobial Properties. Int. J. Mol. Sci. 2015, 16, 21109–21127. [Google Scholar] [CrossRef] [PubMed]
- Jun, Y.; Lee, S.M.; Ju, H.K.; Lee, H.J.; Choi, H.K.; Jo, G.S.; Kim, Y.S. Comparison of the Profile and Composition of Volatiles in Coniferous Needles According to Extraction Methods. Molecules 2016, 21, 363. [Google Scholar] [CrossRef] [PubMed]
- Kovaleva, A.; Osmachko, A.; Ilina, T.; Goryacha, O.; Omelyanchik, L.; Grytsyk, A.; Koshovyi, O. Chemical Composition of Essential Oils from Flowers of Veronica longifolia L., Veronica incana L. and Veronica spicata L. Sci. Pharm. Sci. 2022, 38, 69–79. [Google Scholar] [CrossRef]
- Küpeli, E.; Harput, U.S.; Varel, M.; Yesilada, E.; Saracoglu, I. Bioassay-Guided Isolation of Iridoid Glucosides with Antinociceptive and Anti-Inflammatory Activities from Veronica anagallis-aquatica L. J. Ethnopharmacol. 2005, 102, 170–176. [Google Scholar] [CrossRef]
- Nazlić, M.; Fredotović, Ž.; Vuko, E.; Vuletić, N.; Ljubenkov, I.; Kremer, D.; Jurišić Grubešić, R.; Stabentheiner, E.; Randić, M.; Dunkić, V. Free Volatile Compounds of Veronica austriaca ssp. jacquinii (Baumg.) Eb. Fisch. and Their Biological Activity. Plants 2021, 10, 2529. [Google Scholar] [CrossRef]
- Živković, J.; Barreira, J.C.M.; Stojković, D.; Ćebović, T.; Santos-Buelga, C.; Maksimović, Z.; Ferreira, I.C.F.R. Phenolic Profile, Antibacterial, Antimutagenic and Antitumour Evaluation of Veronica urticifolia Jacq. J. Funct. Foods 2014, 9, 192–201. [Google Scholar] [CrossRef]
- Yin, L.; Lu, Q.; Tan, S.; Ding, L.; Guo, Y.; Chen, F.; Tang, L. Bioactivity-Guided Isolation of Antioxidant and Anti-Hepatocarcinoma Constituents from Veronica ciliata. Chem. Cent. J. 2016, 10, 27. [Google Scholar] [CrossRef]
- Harput, U.S.; Saracoglu, I.; Inoue, M.; Ogihara, Y. Anti-Inflammatory and Cytotoxic Activities of Five Veronica Species. Biol. Pharm. Bull. 2002, 25, 483–486. [Google Scholar] [CrossRef]
- Blowman, K.; Magalhães, M.; Lemos, M.F.L.; Cabral, C.; Pires, I.M. Anticancer Properties of Essential Oils and Other Natural Products. eCAM 2018, 2018, 3149362. [Google Scholar] [CrossRef] [PubMed]
- Gautam, N.; Mantha, A.K.; Mittal, S. Essential Oils and Their Constituents as Anticancer Agents: A Mechanistic View. Biomed. Res. Int. 2014, 2014, 154106. [Google Scholar] [CrossRef] [PubMed]
- Lesgards, J.F.; Baldovini, N.; Vidal, N.; Pietri, S. Anticancer Activities of Essential Oils Constituents and Synergy with Conventional Therapies: A Review. Phytother. Res. 2014, 28, 1423–1446. [Google Scholar] [CrossRef] [PubMed]
- Hussar, P. Apoptosis Regulators Bcl-2 and Caspase-3. Encyclopedia 2022, 2, 1624–1636. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2017; ISBN 978-1-932633-21-4. [Google Scholar]
- Nazlić, M.; Kremer, D.; Akrap, K.; Topić, S.; Vuletić, N.; Dunkić, V. Extraction, Composition and Comparisons–Free Volatile Compounds from Hydrosols of Nine Veronica Taxa. Horticulturae 2023, 9, 16. [Google Scholar] [CrossRef]
- Baptiste, J.; Fokou, H.; Michel, P.; Dongmo, J.; Boyom, F.F. Essential Oil’s Chemical Composition and Pharmacological Properties. In Essential Oils—Oils of Nature; Intech Open: London, UK, 2020. [Google Scholar]
- Nazlić, M.; Fredotović, Ž.; Vuko, E.; Fabijanić, L.; Kremer, D.; Stabentheiner, E.; Ruščić, M.; Dunkić, V. Wild Species Veronica officinalis L. and Veronica saturejoides Vis. ssp. saturejoides—Biological Potential of Free Volatiles. Horticulturae 2021, 7, 295. [Google Scholar] [CrossRef]
- Sakthivel, R.; Malar, D.S.; Devi, K.P. Phytol Shows Anti-Angiogenic Activity and Induces Apoptosis in A549 Cells by Depolarizing the Mitochondrial Membrane Potential. Biomed. Pharmacother. 2018, 105, 742–752. [Google Scholar] [CrossRef]
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. β-Caryophyllene and β-Caryophyllene Oxide-Natural Compounds of Anticancer and Analgesic Properties. Cancer Med. 2016, 5, 3007–3017. [Google Scholar] [CrossRef]
- Dahham, S.; Tabana, Y.; Iqbal, M.; Ahamed, M.; Ezzat, M.; Majid, A.; Majid, A. The Anticancer, Antioxidant and Antimicrobial Properties of the Sesquiterpene β-Caryophyllene from the Essential Oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef]
- Wu, C.-S.; Chen, Y.-J.; Chen, J.J.W.; Shieh, J.-J.; Huang, C.-H.; Lin, P.-S.; Chang, G.-C.; Chang, J.-T.; Lin, C.-C. Terpinen-4-Ol Induces Apoptosis in Human Nonsmall Cell Lung Cancer In Vitro and In Vivo. eCAM 2012, 2012, 818261. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.; Emami, S. β-Ionone and Its Analogs as Promising Anticancer Agents. Eur. J. Med. Chem. 2016, 123, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Kim, Y.S.; Jang, W.J.; Rakib, A.M.; Oh, T.W.; Kim, B.H.; Kim, S.Y.; Kim, J.O.; Ha, Y.L. Anti-Proliferative Effects of β-Ionone on Human Lung Cancer A-549 Cells. J. Life Sci. 2013, 23, 1351–1359. [Google Scholar] [CrossRef]
- Yin, L.; Sun, Z.; Ren, Q.; Su, X.; Zhang, D. Methyl Eugenol Induces Potent Anticancer Effects in RB355 Human Retinoblastoma Cells by Inducing Autophagy, Cell Cycle Arrest and Inhibition of PI3K/MTOR/Akt Signalling Pathway. JBUON 2018, 23, 1174–1178. [Google Scholar] [PubMed]
- Yi, J.-L.; Shi, S.; Shen, Y.-L.; Wang, L.; Chen, H.-Y.; Zhu, J.; Ding, Y. Myricetin and Methyl Eugenol Combination Enhances the Anticancer Activity, Cell Cycle Arrest and Apoptosis Induction of Cis-Platin against HeLa Cervical Cancer Cell Lines. Int. J. Clin. Exp. Pathol. 2015, 8, 1116–1127. [Google Scholar] [PubMed]
- Cerchiara, T.; Straface, S.; Brunelli, E.; Tripepi, S.; Gallucci, M.C.; Chidichimo, G. Antiproliferative Effect of Linalool on RPMI 7932 Human Melanoma Cell Line: Ultrastructural Studies. NPC 2015, 10, 547–549. [Google Scholar] [CrossRef]
- Moreno-Escobar, J.A.; Bazalda, S.; Villarreal, M.L.; Bonilla-Barbosa, J.R.; Mendoza, S.; Rodríguez-López, V. Cytotoxic and Antioxidant Activities of Selected Lamiales Species from Mexico. Pharm. Biol. 2011, 49, 1243–1248. [Google Scholar] [CrossRef]
- Saracoglu, I.; Harput, U.S. In Vitro Cytotoxic Activity and Structure Activity Relationships of Iridoid Glucosides Derived from Veronica Species. Phytother. Res. 2012, 26, 148–152. [Google Scholar] [CrossRef]
- Saracoglu, I.; Oztunca, F.H.; Nagatsu, A.; Harput, U.S. Iridoid Content and Biological Activities of Veronica cuneifolia subsp. cuneifolia and V. cymbalaria. Pharm. Biol. 2011, 49, 1150–1157. [Google Scholar] [CrossRef]
- Okun, I.; Balakin, K.V.; Tkachenko, S.E.; Ivachtchenko, A.V. Caspase Activity Modulators as Anticancer Agents. Med. Chem. 2008, 8, 322–341. [Google Scholar]
- Feng, K.; Jiang, R.; Sun, L.W. Studies on the Anti-Tumor Activity in Vitro of the Flavonoid Extract in Round Leaf Speedwell. In Proceedings of the First International Conference on Cellular, Molecular Biology, Biophysics and Bioengineering, Lushan, China, 17–18 September 2010. [Google Scholar]
- Mastelić, A.; Čulić, V.Č.; Mužinić, N.R.; Vuica-Ross, M.; Barker, D.; Leung, E.Y.; Reynisson, J.; Markotić, A. Glycophenotype of Breast and Prostate Cancer Stem Cells Treated with Thieno[2,3-b]Pyridine Anticancer Compound. Drug Des. Dev. Ther. 2017, 11, 759–769. [Google Scholar] [CrossRef]
- Bilušić, T.; Šola, I.; Rusak, G.; Poljuha, D.; Čikeš Čulić, V. Antiproliferative and Pro-Apoptotic Activities of Wild Asparagus (Asparagus acutifolius L.), Black Bryony (Tamus communis L.) and Butcher’s Broom (Ruscus aculeatus L.) Aqueous Extracts against T24 and A549 Cancer Cell Lines. J. Food Biochem. 2019, 43, e12781. [Google Scholar] [CrossRef]
- Ahmadov, I.S.; Bandaliyeva, A.A.; Nasibova, A.N.; Hasanova, F.V.; Khalilov, R.I. The Synthesis of the Silver Nanodrugs in the Medicinal Plant Baikal Skullcap (Scutellaria baicalensis Georgi) and Their Antioxidant, Antibacterial Activity. Adv. Biol. Earth Sci. 2020, 5, 103–118. [Google Scholar]
- NIST Chemistry WebBook. Available online: https://webbook.nist.gov/ (accessed on 12 March 2021).
- Ertas, A.; Boga, M.; Kizil, M.; Ceken, B.; Goren, A.C.; Hasimi, N.; Demirci, S.; Topcu, G.; Kolak, U. Chemical Profile and Biological Activities of Veronica thymoides subsp. pseudocinerea. Pharm. Biol. 2015, 53, 334–339. [Google Scholar] [CrossRef]
- Li, F. Analysis of Chemical Constituents of Essential Oil in Veronica linariifolia by Gas Chromatography-Mass Spectrometry. Chin. J. Anal. Chem. 2002, 30, 822–825. [Google Scholar]
- Valyova, M.; Hadjimitova, V.; Stoyanov, S.; Ganeva, Y.; Petkov, I. Free Radical Scavenging Activity of Extracts from Bulgarian Veronica officinalis L. and GC-MS Analysis of Ethanol Extract. Internet J. Aesthetic Antiaging Med. 2008, 2, 2–6. [Google Scholar] [CrossRef]



| Species | Locality | Latitude | Longitude | Altitude a.s.l. (m) | Voucher No. |
|---|---|---|---|---|---|
| V. agrestis | Duba Stonska | 42° 52′27.9″ N | 17° 38′36.7″ E | 12 | CROVeS-22-2022 |
| V. anagalloides | Njivice, Krk Island | 45° 10′18.3″ N | 14° 34′10.1″ E | 6 | CROVeS-07-2022 |
| V. austriaca ssp. jacquinii | Korčula Island | 42° 56′52.8″ N | 16° 59′39.7″ E | 441 | CROVeS-02-2022 |
| V. beccabunga | Podevčevo | 46° 12′05.5″ N | 16° 17′58.2″ E | 215 | CROVeS-08-2022 |
| V. cymbalaria | Žrnovo, Korčula Island | 42° 56′59.6″ N | 17° 06′45.1″ E | 153 | CROVeS-03-2022 |
| V. officinalis | Krasno, Velebit Mt | 44° 49′52.4″ N | 15° 01′06.9″ E | 868 | CROVeS-16-2022 |
| V. agrestis | V. anagalloides | V. austriaca ssp. jacquinii | V. beccabunga | V. cymbalaria | V. officinalis | |||
|---|---|---|---|---|---|---|---|---|
| Component | RIa | RIb | EO ± SD | EO ± SD | EO ± SD | EO ± SD | EO ± SD | EO ± SD |
| Monoterpene hydrocarbons | 1.38 | 1.17 | 0.88 | - | - | - | ||
| α-Thujene | 924 | 1012 | 0.65 ± 0.01 | - | - | - | - | - |
| α-Pinene * | 935 | 1017 | 0.73 ± 0.01 | 1.17 ± 0.01 | 0.88 ± 0.01 | - | - | - |
| β-Phellandrene | 1002 | 1195 | - | - | - | - | - | - |
| Oxygenated monoterpenes | 0.88 | 3.88 | 0.46 | 3.87 | 0.58 | 0.77 | ||
| γ-Terpinene | 1057 | 1225 | - | 0.33 ± 0.01 | - | - | - | - |
| Linalool | 1095 | 1506 | 0.34 ± 0.01 | 2.79 ± 0.01 | 0.46 ± 0.01 | 0.53 ± 0.01 | 0.58 ± 0.01 | 0.77 ± 0.01 |
| Terpinen-4-ol | 1174 | 1686 | 0.54 ± 0.01 | 0.76 ± 0.01 | - | 0.88 ± 0.01 | - | - |
| trans-p-Mentha-1(7),8-dien-2-ol | 1187 | 1803 | - | - | - | - | - | - |
| Piperitone | 1250 | 1719 | - | - | - | 2.46 ± 0.01 | - | - |
| Sesquiterpene hydrocarbons | 4.43 | 12.59 | 4.31 | 7.21 | 3.86 | 2.92 | ||
| (E)-Caryophyllene * | 1424 | 1585 | 3.25 ± 0.01 | 7.55 ± 0.01 | 3.08 ± 0.01 | 5.75 ± 0.01 | 2.53 ± 0.01 | 1.15 ± 0.01 |
| allo-Aromadendrene | 1465 | 1662 | 0.18 ± 0.03 | 1.21 ± 0.1 | 0.72 ± 0.05 | 0.81 ± 0.01 | 0.56 ± 0.01 | 1.77 ± 0.01 |
| Germacrene D | 1481 | 1692 | 0.41 ± 0.03 | 3.83 ± 0.01 | 0.81 ± 0.1 | 0.22 ± 0.01 | 0.77 ± 0.1 | - |
| δ-Selinene | 1492 | 1756 | 0.26 ± 0.01 | - | - | 0.43 ± 0.01 | - | - |
| δ-Cadinene | 1517 | 1745 | 0.33 ± 0.07 | - | - | - | - | - |
| Oxygenated sesquiterpenes | 7.58 | 25.43 | 18.64 | 12.08 | 34.9 | 16.07 | ||
| Spathulenol | 1577 | 2101 | - | 1.28 ± 0.05 | - | - | - | - |
| Caryophyllene oxide * | 1581 | 1955 | 2.11 ± 0.01 | 5.22 ± 0.01 | 7.35 ± 0.01 | 6.21 ± 0.01 | 23.83 ± 0.01 | 2.23 ± 0.01 |
| Viridiflorol | 1592 | 2099 | - | - | - | 0.45 ± 0.05 | - | - |
| γ-Eudesmol | 1632 | 2175 | 0.32 ± 0.01 | 0.56 ± 0.01 | 0.88 ± 0.01 | 0.73 ± 0.01 | 0.54 ± 0.01 | - |
| α-Muurolol | 1645 | 2181 | - | - | - | - | - | 3.93 ± 0.01 |
| α-Bisabolol | 1685 | 2210 | - | 1.43 ± 0.01 | 0.77 ± 0.01 | - | - | 0.32 ± 0.01 |
| α-Bisabolol oxide | 1748 | 2511 | - | 0.77 ± 0.01 | - | - | - | 0.75 ± 0.01 |
| Hexahydrofarnesyl acetone * | 1839 | 2113 | 5.15 ± 0.03 | 16.17 ± 0.01 | 9.64 ± 0.01 | 4.69 ± 0.01 | 10.53 ± 0.01 | 8.84 ± 0.01 |
| Oxygenated diterpene | 56.57 | 3.56 | 13.02 | 28.08 | 16.66 | 6.17 | ||
| Phytol * | 1942 | 2610 | 56.57 ± 0.01 | 3.56 ± 0.01 | 13.02 ± 0.01 | 28.08 ± 0.01 | 16.66 ± 0.01 | 6.17 ± 0.01 |
| Phenolic compounds | 0.77 | 2.02 | - | - | 1.38 | 5.06 | ||
| Thymol * | 1289 | 2154 | 0.77 ± 0.1 | - | - | - | 0.65 ± 0.01 | - |
| Methyl eugenol | 1403 | 2005 | - | 2.02 ± 0.01 | - | - | 0.73 ± 0.01 | 3.61 ± 0.01 |
| (Z)-Methyl isoeugenol | 1451 | 2070 | - | - | - | - | - | 1.45 ± 0.01 |
| Acids, alcohols and esters | 12.98 | 29.68 | 34.53 | 22.47 | 34.22 | 37.65 | ||
| 3-Hexen-1-ol | 873 | 1383 | - | - | - | - | - | 0.46 ± 0.07 |
| Benzaldehyde | 952 | 1508 | - | - | - | - | - | 1.15 ± 0.01 |
| Benzene acetaldehyde | 1036 | 1633 | 0.26 ± 0.01 | 2.03 ± 0.01 | 0.86 ± 0.01 | 0.44 ± 0.01 | 8.61 ± 0.01 | 6.08 ± 0.01 |
| n-Nonanal | 1100 | 1389 | - | 0.72 ± 0.01 | - | -- | - | 0.72 ± 0.05 |
| (E)-β-Damascenone | 1384 | 1819 | 4.82 ± 0.01 | 5.55 ± 0.01 | - | 0.86 ± 0.01 | 1.05 ± 0.1 | 5.34 ± 0.01 |
| β-Ionone | 1487 | 1935 | 2.29 ± 0.01 | 13.13 ± 0.01 | 6.01 ± 0.01 | 3.75 ± 0.01 | 6.83 ± 0.01 | 11.33 ± 0.01 |
| Hexadecanoic acid * | 1959 | 2912 | 5.61 ± 0.01 | 8.25 ± 0.01 | 27.66 ± 0.01 | 17.86 ± 0.01 | 17.73 ± 0.01 | 12.57 ± 0.01 |
| Hydrocarbons | 11.2 | 17.11 | 23.02 | 21.13 | 4.04 | 28.16 | ||
| Heneicosane * | 2100 | 2100 | 0.81 ± 0.05 | 2.21 ± 0.01 | - | 0.45 ± 0.02 | 0.30 ± 0.01 | - |
| Docosane * | 2200 | 2200 | 0.28 ± 0.01 | 1.47 ± 0.01 | 0.32 ± 0.01 | 1.42 ± 0.01 | 0.13 ± 0.01 | 1.92 ± 0.01 |
| Tricosane * | 2300 | 2300 | 0.25 ± 0.07 | 3.42 ± 0.01 | 9.11 ± 0.01 | 0.65 ± 0.02 | 0.15 ± 0.01 | 0.53 ± 0.01 |
| Tetracosane * | 2400 | 2400 | 8.48 ± 0.01 | 1.17 ± 0.01 | 5.63 ± 0.01 | 0.57 ± 0.01 | 0.26 ± 0.01 | 0.56 ± 0.01 |
| Pentacosane * | 2500 | 2500 | - | 6.75 ± 0.01 | 2.27 ± 0.01 | 0.36 ± 0.01 | 0.28 ± 0.01 | 0.21 ± 0.01 |
| Hexacosane * | 2600 | 2600 | - | 0.65 ± 0.01 | 0.17 ± 0.01 | 0.33 ± 0.05 | 0.46 ± 0.01 | 6.57 ± 0.01 |
| Heptacosane * | 2700 | 2700 | - | 0.32 ± 0.01 | 0.32 ± 0.05 | 11.82 ± 0.01 | 0.53 ± 0.01 | 17.21 ± 0.01 |
| Octacosane * | 2800 | 2800 | 1.38 ± 0.01 | 1.12 ± 0.1 | 5.2 ± 0.01 | 5.53 ± 0.01 | 1.93 ± 0.01 | 1.16 ± 0.01 |
| Total identification (%) | 95.79 | 95.44 | 95.86 | 94.87 | 95.64 | 96.8 |
| V. agrestis | V. anagalloides | V. austriaca ssp. jacquinii | V. beccabunga | V. cymbalaria | V. officinalis | |||
|---|---|---|---|---|---|---|---|---|
| Component | RIa | RIb | HY ± SD | HY ± SD | HY ± SD | HY ± SD | HY ± SD | HY ± SD |
| Monoterpene hydrocarbons | 6.38 | 3.81 | - | 17.11 | 1.28 | 0.54 | ||
| α-Thujene | 924 | 1012 | 4.32 ± 0.01 | - | - | - | - | - |
| α-Pinene * | 935 | 1017 | 2.06 ± 0.01 | 3.81 ± 0.01 | - | 17.11 ± 0.01 | 1.28 ± 0.01 | 0.54 ± 0.01 |
| Oxygenated monoterpenes | 10.16 | 6.37 | 8.2 | 23.85 | 2.18 | 13.1 | ||
| γ-Terpinene | 1057 | 1225 | - | - | 0.53 ± 0.01 | - | - | - |
| Linalool | 1095 | 1506 | 5.83 ± 0.01 | 4.27 ± 0.01 | - | 3.11 ± 0.01 | 1.73 ± 0.01 | 6.61 ± 0.01 |
| Terpinen-4-ol | 1174 | 1686 | 0.34 ± 0.01 | 1.45 ± 0.01 | 0.66 ± 0.01 | 0.34 ± 0.01 | 0.45 ± 0.01 | 6.49 ± 0.01 |
| α-Terpineol | 1184 | 1660 | 0.72 ± 0.01 | 0.65 ± 0.01 | - | - | - | - |
| trans-p-Mentha-1(7),8-dien-2-ol | 1187 | 1803 | 0.66 ± 0.01 | - | 7.01 ± 0.01 | 0.86 ± 0.01 | - | - |
| Piperitone | 1250 | 1719 | 2.61 ± 0.01 | - | - | 19.54 ± 0.01 | - | - |
| Sesquiterpene hydrocarbons | 9.77 | 8.86 | 12.07 | 7.73 | 2.45 | 2.15 | ||
| (E)-Caryophyllene * | 1424 | 1585 | 5.37 ± 0.01 | 4.32 ± 0.01 | 6.31 ± 0.01 | 6.21 ± 0.01 | 0.97 ± 0.07 | 1.44 ± 0.01 |
| allo-Aromadendrene | 1465 | 1662 | 1.01 ± 0.01 | 2.21 ± 0.05 | 0.75 ± 0.03 | 0.67 ± 0.01 | 0.41 ± 0.01 | 0.71 ± 0.01 |
| Germacrene D | 1481 | 1692 | 2.34 ± 0.01 | 0.80 ± 0.01 | 2.15 ± 0.01 | 0.85 ± 0.01 | 0.26 ± 0.01 | - |
| δ-Selinene | 1492 | 1756 | 0.73 ± 0.01 | 1.53 ± 0.01 | 2.86 ± 0.1 | - | 0.81 ± 0.01 | - |
| δ-Cadinene | 1517 | 1745 | 0.32 ± 0.01 | - | - | - | - | - |
| Oxygenated sesquiterpenes | 17.34 | 7.98 | 9.46 | 5.08 | 7.58 | 3.99 | ||
| Spathulenol | 1577 | 2101 | 0.36 ± 0.1 | - | 0.46 ± 0.01 | - | 0.55 ± 0.01 | - |
| Caryophyllene oxide * | 1581 | 1955 | 14.01 ± 0.01 | 5.15 ± 0.01 | 5.35 ± 0.01 | 2.19 ± 0.01 | 6.26 ± 0.01 | 1.42 ± 0.1 |
| Viridiflorol | 1592 | 2099 | 0.85 ± 0.01 | 0.43 ± 0.01 | 0.86 ± 0.03 | 1.18 ± 0.01 | - | - |
| γ-Eudesmol | 1632 | 2175 | 2.12 ± 0.05 | 1.65 ± 0.01 | - | 0.69 ± 0.01 | - | - |
| α-Muurolol | 1645 | 2181 | - | - | 1.77 ± 0.01 | 0.44 ± 0.07 | 0.65 ± 0.01 | 1.64 ± 0.01 |
| α-Bisabolol | 1685 | 2210 | - | - | 0.45 ± 0.01 | 0.58 ± 0.01 | 0.12 ± 0.01 | 0.61 ± 0.01 |
| α-Bisabolol oxide | 1748 | 2511 | - | 0.75 ± 0.01 | - | - | - | 0.32 ± 0.01 |
| Hexahydrofarnesyl acetone * | 1839 | 2113 | - | - | 0.57 ± 0.01 | - | - | - |
| Phenolic compounds | 11.37 | 13.57 | 42.58 | 1.15 | 69.93 | 30.92 | ||
| Thymol * | 1289 | 2154 | 2.32 ± 0.1 | - | 5.57 ± 0.01 | 1.15 ± 0.01 | - | 1.81 ± 0.03 |
| p-Vinyl guaicol | 1313 | 2156 | - | - | - | - | - | - |
| Methyl eugenol | 1403 | 2005 | 9.05 ± 0.01 | 13.57 ± 0.01 | 37.01 ± 0.01 | - | 38.61 ± 0.01 | 22.01 ± 0.01 |
| (Z)-Methyl isoeugenol | 1451 | 2070 | - | - | - | - | 31.32 ± 0.01 | 7.12 ± 0.01 |
| Acids, alcohols and esters | 42.94 | 55.13 | 23.55 | 38.8 | 12.08 | 44.24 | ||
| Isopentyl acetate | 863 | 1127 | - | - | 0.22 ± 0.01 | - | - | - |
| Benzaldehyde | 952 | 1508 | 6.29 ± 0.01 | 13.56 ± 0.01 | 6.33 ± 0.01 | 4.57 ± 0.01 | 0.13 ± 0.03 | 10.36 ± 0.01 |
| Benzene acetaldehyde | 1036 | 1633 | 11.56 ± 0.01 | 9.67 ± 0.01 | 13.34 ± 0.01 | 10.73 ± 0.01 | 10.81 ± 0.01 | 3.77 ± 0.03 |
| n-Nonanal | 1100 | 1389 | 2.35 ± 0.1 | 3.82 ± 0.01 | 1.57 ± 0.01 | - | - | 1.26 ± 0.01 |
| (E)-β-Damascenone | 1384 | 1819 | 12.42 ± 0.01 | 11.55 ± 0.01 | 1.01 ± 0.01 | 8.32 ± 0.01 | - | 14.01 ± 0.01 |
| β-Ionone | 1487 | 1935 | 10.32 ± 0.01 | 16.53 ± 0.01 | 1.08 ± 0.01 | 15.18 ± 0.01 | 1.14 ± 0.01 | 14.84 ± 0.01 |
| Total identification (%) | 97.96 | 95.72 | 95.86 | 93.72 | 95.5 | 94.94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vrca, I.; Čikeš Čulić, V.; Lozić, M.; Dunkić, N.; Kremer, D.; Ruščić, M.; Nazlić, M.; Dunkić, V. Isolation of Volatile Compounds by Microwave-Assisted Extraction from Six Veronica Species and Testing of Their Antiproliferative and Apoptotic Activities. Plants 2023, 12, 3244. https://doi.org/10.3390/plants12183244
Vrca I, Čikeš Čulić V, Lozić M, Dunkić N, Kremer D, Ruščić M, Nazlić M, Dunkić V. Isolation of Volatile Compounds by Microwave-Assisted Extraction from Six Veronica Species and Testing of Their Antiproliferative and Apoptotic Activities. Plants. 2023; 12(18):3244. https://doi.org/10.3390/plants12183244
Chicago/Turabian StyleVrca, Ivana, Vedrana Čikeš Čulić, Mirela Lozić, Niko Dunkić, Dario Kremer, Mirko Ruščić, Marija Nazlić, and Valerija Dunkić. 2023. "Isolation of Volatile Compounds by Microwave-Assisted Extraction from Six Veronica Species and Testing of Their Antiproliferative and Apoptotic Activities" Plants 12, no. 18: 3244. https://doi.org/10.3390/plants12183244
APA StyleVrca, I., Čikeš Čulić, V., Lozić, M., Dunkić, N., Kremer, D., Ruščić, M., Nazlić, M., & Dunkić, V. (2023). Isolation of Volatile Compounds by Microwave-Assisted Extraction from Six Veronica Species and Testing of Their Antiproliferative and Apoptotic Activities. Plants, 12(18), 3244. https://doi.org/10.3390/plants12183244










