Beneficial Interactive Effects Provided by an Arbuscular Mycorrhizal Fungi and Yeast on the Growth of Oenothera picensis Established on Cu Mine Tailings
Abstract
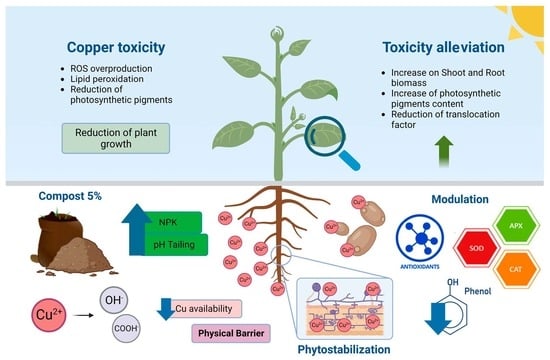
:1. Introduction
2. Results
2.1. Characterization of PGP Traits in Cu-Tolerant Yeast
2.2. AMF Determinations
2.3. Plant Biomass Production and Photosynthetic Pigments
2.4. Cu Concentrations in Tailings and Plant Tissues
2.5. Antioxidant Activities and Phenols
2.6. Multivariate Associations
3. Discussion
4. Materials and Methods
4.1. Mine Tailings, Compost, Biological Material, and Procedures for Physicochemical Characterization
4.2. Isolation and Identification of Cu-Tolerant Yeast with PGP Activities
4.3. Growth Conditions
4.4. Cu Content in Mine Tailings and Plant Tissues
4.5. Plant Photosynthetic Pigments and Antioxidant Response
4.6. Arbuscular Mycorrhizal Fungal Measurements
4.7. Experimental Design
4.8. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Aponte, H.; Meli, P.; Butler, B.; Paolini, J.; Matus, F.; Merino, C.; Cornejo, P.; Kuzyakov, Y. Meta-analysis of heavy metal effects on soil enzyme activities. Sci. Total Environ. 2020, 737, 139744. [Google Scholar] [CrossRef] [PubMed]
- Schoenberger, E. Environmentally sustainable mining: The case of tailings storage facilities. Resour. Policy 2016, 49, 119–128. [Google Scholar] [CrossRef]
- Wang, F. Occurrence of arbuscular mycorrhizal fungi in mining-impacted sites and their contribution to ecological restoration: Mechanisms and applications. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1901–1957. [Google Scholar] [CrossRef]
- Mendez, M.O.; Maier, R.M. Phytoremediation of mine tailings in temperate and arid environments. Rev. Environ. Sci. Biotechnol. 2008, 7, 47–59. [Google Scholar] [CrossRef]
- Kossoff, D.; Dubbin, W.E.; Alfredsson, M.; Edwards, S.J.; Macklin, M.G.; Hudson-Edwards, K.A. Mine tailings dams: Characteristics, failure, environmental impacts, and remediation. Appl. Geochem. 2014, 51, 229–245. [Google Scholar] [CrossRef]
- Lima, A.T.; Mitchell, K.; O’Connell, D.W.; Verhoeven, J.; Van Cappellen, P. The legacy of surface mining: Remediation, restoration, reclamation and rehabilitation. Environ. Sci. Policy 2016, 66, 227–233. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Mine Wastes: Characterization, Treatment and Environmental Impacts, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar] [CrossRef]
- Tapia, Y.; Bustos, P.; Salazar, O.; Casanova, M.; Castillo, B.; Acuña, E.; Masaguer, A. Phytostabilization of Cu in mine tailings using native plant Carpobrotus aequilaterus and the addition of potassium humates. J. Geochem. Explor. 2017, 183, 102–113. [Google Scholar] [CrossRef]
- Ye, Z.H.; Shu, W.S.; Zhang, Z.Q.; Lan, C.Y.; Wong, M.H. Evaluation of major constraints to revegetation of lead/zinc mine tailings using bioassay techniques. Chemosphere 2002, 47, 1103–1111. [Google Scholar] [CrossRef]
- Aponte, H.; Herrera, W.; Cameron, C.; Black, H.; Meier, S.; Paolini, J.; Tapia Fernández, Y.; Cornejo, P. Alteration of enzyme activities and functional diversity of a soil contaminated with copper and arsenic. Ecotoxicol. Environ. Saf. 2020, 192, 110264. [Google Scholar] [CrossRef]
- Miller, A.; Zégre, N. Mountaintop removal mining and catchment hydrology. Water 2014, 6, 472–499. [Google Scholar] [CrossRef]
- Pérez, R.; Tapia, Y.; Antilén, M.; Casanova, M.; Vidal, C.; Silambarasanm, S.; Cornejo, P. Rhizosphere Management for Phytoremediation of Copper Mine Tailings. J. Soil Sci. Plant Nutr. 2021, 21, 3091–3109. [Google Scholar] [CrossRef]
- Medina, J.; Monreal, C.; Chabot, D.; Meier, S.; González, M.E.; Morales, E.; Parillo, R.; Borie, F.; Cornejo, P. Microscopic and spectroscopic characterization of humic substances from a compost amended copper contaminated soil: Main features and their potential effects on Cu immobilization. Environ. Sci. Pollut. Res. 2017, 24, 14104–14116. [Google Scholar] [CrossRef] [PubMed]
- Tapia, Y.; Loch, B.; Castillo, B.; Acuña, E.; Casanova, M.; Salazar, O.; Cornejo, P.; Antilén, M. Accumulation of Sulphur in Atriplex nummularia Cultivated in Mine Tailings and Effect of Organic Amendments Addition. Water Air Soil Pollut. 2020, 231, 8. [Google Scholar] [CrossRef]
- Silambarasan, S.; Logeswari, P.; Valentine, A.; Cornejo, P.; Kannan, V.R. Pseudomonas citronellolis strain SLP6 enhances the phytoremediation efficiency of Helianthus annuus in copper contaminated soils under salinity stress. Plant Soil. 2020, 457, 241–253. [Google Scholar] [CrossRef]
- Pérez, R.; Tapia, Y.; Antilén, M.; Casanova, M.; Vidal, C.; Santander, C.; Aponte, H.; Cornejo, P. Interactive effect of compost application and inoculation with the fungus Claroideoglomus claroideum in Oenothera picensis plants growing in mine tailings. Ecotox. Environ. Safe. 2021, 208, 111495. [Google Scholar] [CrossRef]
- Meier, S.; Alvear, M.; Borie, F.; Aguilera, P.; Ginocchio, R.; Cornejo, P. Influence of copper on root exudate patterns in some metallophytes and agricultural plants. Ecotoxicol. Environ. Saf. 2012, 75, 8–15. [Google Scholar] [CrossRef]
- Cornejo, P.; Meier, S.; García, S.; Ferrol, N.; Durán, P.; Borie, F.; Seguel, A. Contribution of inoculation with arbuscular mycorrhizal fungi to the bioremediation of a copper contaminated soil using Oenothera picensis. J. Soil Sci. Plant Nutr. 2017, 17, 14–21. [Google Scholar] [CrossRef]
- Wang, L.; Ji, B.; Hu, Y.; Liu, R.; Sun, W. A review on in situ phytoremediation of mine tailings. Chemosphere 2017, 184, 594–600. [Google Scholar] [CrossRef]
- Xu, D.; Zhan, C.; Liu, H.; Lin, H. A critical review on environmental implications, recycling strategies, and ecological remediation for mine tailings. Environ. Sci. Pollut. Res. 2019, 26, 35657–35669. [Google Scholar] [CrossRef]
- Rodriguez, R.; Marticorena, C.; Alarcón, D.; Baeza, C.; Cavieres, L.; Finot, V.L.; Fuentes, N.; Kiessling, A.; Mihoc, M.; Pauchard, A.; et al. Catálogo de las plantas vasculares de Chile. Gayana Bot. 2018, 75, 1–430. [Google Scholar] [CrossRef]
- González, I.; Neaman, A. Evaluación de la tolerancia al cobre de dos poblaciones de Oenothera picensis Phil. subsp picensis (Onagraceae). Gayana Bot. 2015, 72, 240–249. [Google Scholar] [CrossRef]
- González-Guerrero, M.; Melville, L.H.; Ferrol, N.; Lott, J.N.A.; Azcón-Aguilar, C.; Peterson, R.L. Ultrastructural localization of heavy metals in the extraradical mycelium and spores of the arbuscular mycorrhizal fungus Glomus intraradices. Can. J. Microbiol. 2008, 54, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Meier, S.; Curaqueo, G.; Khan, N.; Bolan, N.; Rilling, J.; Vidal, C.; Fernández, N.; Acuña, J.; González, M.E.; Cornejo, P.; et al. Effects of biochar on copper inmobilization and soil microbial communities in a metal-contaminated soil. J. Soil Sediments 2017, 17, 1237–1250. [Google Scholar] [CrossRef]
- Cornejo, P.; Pérez-Tienda, J.; Meier, S.; Valderas, A.; Borie, F.; Azcón-Aguilar, C.; Ferrol, N. Copper compartmentalization in spores as a survival strategy of arbuscular mycorrhizal fungi in Cu-polluted environments. Soil Biol. Biochem. 2013, 57, 925–928. [Google Scholar] [CrossRef]
- Gujre, N.; Agnihotri, R.; Rangan, L.; Sharma, M.P.; Mitra, S. Deciphering the dynamics of glomalin and heavy metals in soils contaminated with hazardous municipal solid wastes. J. Hazard. Mater. 2021, 416, 125869. [Google Scholar] [CrossRef] [PubMed]
- Begum, N.; Qin, C.; Ahanger, M.A.; Raza, S.; Khan, M.I.; Ashraf, M.; Ahmed, N.; Zhang, L. Role of Arbuscular Mycorrhizal Fungi in Plant Growth Regulation: Implications in Abiotic Stress Tolerance. Front. Plant Sci. 2019, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Cicatelli, A.; Lingua, G.; Todeschini, V.; Biondi, S.; Torrigiani, P.; Castiglione, S. Arbuscular mycorrhizal fungi modulate the leaf transcriptome of a Populus alba L. clone grown on a zinc and copper-contaminated soil. Environ. Exp. Bot. 2012, 75, 25–35. [Google Scholar] [CrossRef]
- Degola, F.; Fattorini, L.; Bona, E.; Sprimuto, C.T.; Argese, E.; Berta, G.; Sanità di Toppi, L. The symbiosis between Nicotiana tabacum and the endomycorrhizal fungus Funneliformis mosseae increases the plant glutathione level and decreases leaf cadmium and root arsenic contents. Plant Physiol. Biochem. 2015, 92, 11–18. [Google Scholar] [CrossRef]
- Jiang, Q.Y.; Zhuo, F.; Long, S.H.; Zhao, H.D.; Yang, D.J.; Ye, Z.H.; Li, S.S.; Jing, Y.X. Can arbuscular mycorrhizal fungi reduce Cd uptake and alleviate Cd toxicity of Lonicera japonica grown in Cd-added soils? Sci. Rep. 2016, 6, 2–10. [Google Scholar] [CrossRef]
- Luo, J.; Li, X.; Jin, Y.; Traore, I.; Dong, L.; Yang, G.; Wang, Y. Effects of arbuscular mycorrhizal fungi Glomus mosseae on the growth and medicinal components of Dysosma versipellis under copper stress. Bull. Environ. Contam. Toxicol. 2020, 107, 924–930. [Google Scholar] [CrossRef]
- Luo, J.; Yan, Q.; Yang, G.; Wang, Y. Impact of the Arbuscular Mycorrhizal Fungus Funneliformis mosseae on the Physiological and Defence Responses of Canna indica to Copper Oxide Nanoparticles Stress. J. Fungi 2022, 8, 513. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.F.; Sun, P.F.; Lu, H.Y.; Wei, J.Y.; Xiao, H.S.; Fang, W.T.; Cheng, B.Y.; Chou, J.Y. Plant growth-promoting traits of yeasts isolated from the phyllosphere and rhizosphere of Drosera spatulata. Lab. Fungal Biol. 2016, 120, 433–448. [Google Scholar] [CrossRef] [PubMed]
- Nimsi, K.A.; Manjusha, K.; Kathiresan, K.; Arya, H. Plant growth-promoting yeasts (PGPY), the latest entrant for use in sustainable agriculture: A review. J. Appl. Microbiol. 2023, 134, lxac088. [Google Scholar] [CrossRef] [PubMed]
- Sarabia, M.; Cornejo, P.; Azcón, R.; Carreón-Abud, Y.; Larsen, J. Mineral phosphorus fertilization modulates interactions between maize, rhizosphere yeasts and arbuscular mycorrhizal fungi. Rhizosphere 2017, 4, 89–93. [Google Scholar] [CrossRef]
- Silambarasan, S.; Logeswari, P.; Cornejo, P.; Kannan, V.R. Evaluation of the production of exopolysaccharide by plant growth promoting yeast Rhodotorula sp. strain CAH2 under abiotic stress conditions. Int. J. Biol. Macromol. 2019, 121, 55–62. [Google Scholar] [CrossRef]
- Silambarasan, S.; Logeswari, P.; Cornejo, P.; Abraham, J.; Valentine, A. Simultaneous mitigation of aluminum, salinity and drought stress in Lactuca sativa growth via formulated plant growth promoting Rhodotorula mucilaginosa CAM4. Ecotoxicol. Environ. Saf. 2019, 180, 63–72. [Google Scholar] [CrossRef]
- Irawati, W.; Wijaya, Y.; Christian, S.; Djojo, E.S. Characterization of heavy metals resistant yeast isolated from activated sludge in Rungkut, Surabaya, Indonesia as biosorbent of mercury, copper, and lead. AIP Conf. Proc. 2016, 1744, 020061. [Google Scholar] [CrossRef]
- Radić, D.S.; Pavlović, V.P.; Lazović, M.M.; Jovičić-Petrović, J.P.; Karličić, V.M.; Lalević, B.T.; Raičević, V.B. Copper-tolerant yeasts: Raman spectroscopy in determination of bioaccumulation mechanism. Environ. Sci. Pollut. Res. 2017, 24, 21885–21893. [Google Scholar] [CrossRef]
- Fracchia, S.; Sampedro, I.; Scervino, J.M.; Garcia-Romera, I.; Ocampo, J.A.; Godeas, A. Influence of saprobe fungi and their exudates on arbuscular mycorrhizal symbioses. Symbiosis 2004, 36, 169–182. [Google Scholar]
- Gollner, M.J.; Püschel, D.; Rydlová, J.; Vosátka, M. Effect of inoculation with soil yeasts on mycorrhizal symbiosis of maize. Pedobiologia 2006, 50, 341–345. [Google Scholar] [CrossRef]
- Filho, S.; Vidor, C. Solubilização de fosfato por microrganismos na presença de fontes de carbono. Rev. Bras. Cienc. Solo 2000, 24, 311–319. [Google Scholar] [CrossRef]
- Khalid, A.; Arshad, M.; Zahir, Z.A. Screening plant growth-promoting rhizobacteria for improving growth and yield of wheat. J. Appl. Microbiol. 2004, 96, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Santibañez, C.; De La Fuente, L.M.; Bustamante, E.; Silva, S.; León-Lobos, P.; Ginocchio, R. Potential use of organic- and hard-rock mine wastes on aided phytostabilization of large-scale mine tailings under semiarid mediterranean climatic conditions: Short-Term field study. Appl. Environ. Soil Sci. 2012, 895817. [Google Scholar] [CrossRef]
- Dold, B.; Fontbote, L. Element cycling and secondary mineralogy in porphyry copper tailings as a function of climate, primary mineralogy, and mineral processing. J. Geochem. Explor. 2001, 74, 3–55. [Google Scholar] [CrossRef]
- Nayuki, K.; Chen, B.; Ohtomo, R.; Kuga, Y. Cellular imaging of cadmium in resin sections of arbuscular mycorrhizas using synchrotron micro X-ray fluorescence. Microbes Environ. 2014, 29, 60–66. [Google Scholar] [CrossRef]
- Wang, F.Y.; Lin, X.G.; Yin, R. Inoculation with arbuscular mycorrhizal fungus Acaulospora mellea decreases Cu phytoextraction by maize from Cu-contaminated soil. Pedobiologia 2007, 51, 99–109. [Google Scholar] [CrossRef]
- Hu, Q.Q.; Shu, J.Q.; Li, W.M.; Wang, G.Z. Role of Auxin and Nitrate Signaling in the Development of Root System Architecture. Front. Plant Sci. 2021, 12, 690363. [Google Scholar] [CrossRef]
- Glick, B.R. Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Ferrol, N.; Tamayo, E.; Vargas, P. The heavy metal paradox in arbuscular mycorrhizas: From mechanisms to biotechnological applications. J. Exp. Bot. 2016, 67, 6253–6265. [Google Scholar] [CrossRef]
- de Souza, L.A.; de Andrade, S.A.L.; de Souza, S.C.R.; Schiavinato, M.A. Arbuscular mycorrhiza confers Pb tolerance in Calopogonium mucunoides. Acta Physiol. Plant. 2012, 34, 523–531. [Google Scholar] [CrossRef]
- de Andrade, S.A.; da Silveira, A.P.; Jorge, R.A.; de Abreu, M.F. Cadmium accumulation in sunflower plants influenced by arbuscular mycorrhiza. Int. J. Phytoremediation 2008, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lingua, G.; Bona, E.; Todeschini, V.; Cattaneo, C.; Marsano, F.; Berta, G. Effects of Heavy Metals and Arbuscular Mycorrhiza on the Leaf Proteome of a Selected Poplar Clone: A Time Course Analysis. PLoS ONE 2012, 7, e38662. [Google Scholar] [CrossRef] [PubMed]
- Meier, S.; Cornejo, P.; Cartes, P.; Borie, F.; Medina, J.; Azcón, R. Interactive effect between Cu-adapted arbuscular mycorrhizal fungi and biotreated agrowaste residue to improve the nutritional status of Oenothera picensis growing in Cu-polluted soils. J. Plant Nutr. Soil Sci. 2015, 178, 126–135. [Google Scholar] [CrossRef]
- Raaijmakers, J.M.; Paulitz, T.C. The rhizosphere: A playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 2009, 321, 341–361. [Google Scholar] [CrossRef]
- Cruz, F.J.R.; Ferreira, R.L.C.; Conceição, S.S.; Lima, E.U.; Oliveira Neto, C.F.; Galvão, J.R.; Lopes, S.C.; Viegas, I.J.M. Copper Toxicity in Plants: Nutritional, Physiological, and Biochemical Aspects. In Advances in Plant Defense Mechanisms; IntechOpen: London, UK, 2022. [Google Scholar] [CrossRef]
- Dhalaria, R.; Kumar, D.; Kumar, H.; Nepovimova, E.; Kuča, K.; Torequl Islam, M.; Verma, R. Arbuscular Mycorrhizal Fungi as Potential Agents in Ameliorating Heavy Metal Stress in Plants. Agronomy 2020, 10, 815. [Google Scholar] [CrossRef]
- Seguel, A.; Cumming, J.R.; Klugh-stewart, K.; Cornejo, P.; Borie, F. The role of arbuscular mycorrhizas in decreasing aluminium phytotoxicity in acidic soils: A review. Mycorrhiza 2013, 23, 167–183. [Google Scholar] [CrossRef] [PubMed]
- Balíková, K.; Vojtková, H.; Duborská, E.; Kim, H.; Matúš, P.; Urík, M. Role of Exopolysaccharides of Pseudomonas in Heavy Metal Removal and Other Remediation Strategies. Polymers 2022, 14, 4253. [Google Scholar] [CrossRef]
- Sandhya, V.; Ali, S.Z. The production of exopolysaccharide by Pseudomonas putida GAP-P45 under various abiotic stress conditions and its role in soil aggregation. Microbiology 2015, 84, 512–519. [Google Scholar] [CrossRef]
- Fatima, T.; Arora, N.K. Pseudomonas entomophila PE3 and its exopolysaccharides as biostimulants for enhancing growth, yield and tolerance responses of sunflower under saline conditions. Microbiol. Res. 2021, 244, 126671. [Google Scholar] [CrossRef]
- Busnelli, M.P.; Lazzarini Behrmann, I.C.; Ferreira, M.L.; Candal, R.J.; Ramirez, S.A.; Vullo, D.L. Metal-Pseudomonas veronii 2E interactions as strategies for innovative process developments in environmental biotechnology. Front. Microbiol. 2021, 12, 622600. [Google Scholar] [CrossRef]
- Ferreira, M.L.; Ramirez, S.A.; Vullo, D.L. Chemical characterization and ligand behaviour of Pseudomonas veronii 2E siderophores. World J. Microbiol. Biotechnol. 2018, 34, 134. [Google Scholar] [CrossRef] [PubMed]
- Kabata-Pendias, A. Trace Elements in Soils and Plants, 4th ed.; CRC Press: Boca Raton, FA, USA, 2010. [Google Scholar] [CrossRef]
- Vidal, C.; Ruiz, A.; Ortiz, J.; Larama, G.; Perez, R.; Santander, C.; Ferreira, P.A.A.; Cornejo, P. Antioxidant Responses of Phenolic Compounds and Immobilization of Copper in Imperata cylindrica, a Plant with Potential Use for Bioremediation of Cu Contaminated Environments. Plants 2020, 9, 1397. [Google Scholar] [CrossRef] [PubMed]
- Vidal, C.; Larama, G.; Meneses, C.; Cornejo, P. Main Molecular Pathways Associated with Copper Tolerance Response in Imperata cylindrica by de novo Transcriptome Assembly. Plants 2021, 10, 357. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Yu, W.; Ma, Q.; Zhou, H.; Jiang, C. Toxicity of sulfadiazine and copper and their interaction to wheat (Triticum aestivum L.) seedlings. Ecotoxicol. Environ. Saf. 2017, 142, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, V.; Shamshiri, M.H.; Shirani, H.; Roosta, H.R. Effect of mycorrhizal inoculation on ecophysiological responses of pistachio plants grown under different water regimes. Photosynthetica 2011, 49, 531–538. [Google Scholar] [CrossRef]
- Kranner, I.; Beckett, R.P.; Wornik, S.; Zorn, M.; Pfeifhofer, H.W. Revival of a resurrection plant correlates with its antioxidant status. Plant J. 2002, 31, 13–24. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2017, 54, 287–293. [Google Scholar] [CrossRef]
- Tanwir, K.; Amna; Javed, M.T.; Shahid, M.; Akram, M.S.; Ali, Q. Chapter 32—Antioxidant defense systems in bioremediation of organic pollutants. In Handbook of Bioremediation, 2nd ed.; Hasanuzzaman, M., Vara Prasad, M.N., Eds.; Academic Press: San Diego, CA, USA, 2021; Volume 3, pp. 505–521. [Google Scholar] [CrossRef]
- Alarcon, S.; Tereucan, G.; Cornejo, P.; Contreras, B.; Ruiz, A. Metabolic and antioxidant effects of inoculation with arbuscular mycorrhizal fungi in crops of flesh-coloured Solanum tuberosum treated with fungicides. J. Sci. Food Agric. 2021, 102, 2270–2280. [Google Scholar] [CrossRef]
- Dehghanian, Z.; Habibi, K.; Dehghanian, M.; Aliyar, S.; Asgari Lajayer, B.; Astatkie, T.; Minkina, T.; Keswani, C. Reinforcing the bulwark: Unravelling the efficient applications of plant phenolics and tannins against environmental stresses. Heliyon 2022, 8, e09094. [Google Scholar] [CrossRef]
- Handa, N.; Kohli, S.K.; Sharma, A.; Thukral, A.K.; Bhardwaj, R.; Fathi, E.; Allah, A.; Alqarawi, A.A.; Ahmad, P. Selenium modulates dynamics of antioxidative defence expression, photosynthetic attributes and secondary metabolites to mitigate chromium toxicity in Brassica juncea L. plants. Environ. Exp. Bot. 2018, 161, 180–192. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Demirata, B.; Ozyürek, M.; Celik, S.E.; Bektaşoğlu, B.; Berker, K.I.; Ozyurt, D. Comparative evaluation of various total antioxidant capacity assays applied to phenolic compounds with the CUPRAC assay. Molecules 2007, 12, 1496–1547. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.; Bustamante, L.; Vergara, C.; Von Baer, D.; Mardones, C. Hydroxycinnamic acid and flavonols in native edible berries of South Patagonia. Food Chem. 2015, 167, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Handa, N.; Kohli, S.; Sharma, A.; Thukral, A.; Bhardwaj, R.; Alyemeni, M.; Wijaya, L.; Ahmad, P. Selenium ameliorates chromium toxicity through modifications in pigment system, antioxidative capacity, osmotic system, and metal chelators in Brassica juncea seedlings. S. Afr. J. Bot. 2018, 119, 1–10. [Google Scholar] [CrossRef]
- Almughraby, E.; Kalimullin, M.I.; Timofeeva, O.A. Phytochemical composition and antioxidant activity in Brassica oleracea var. Sabellica under the effect of plant growth regulators. EurAsian J. BioSci. 2019, 13, 1037–1043. [Google Scholar]
- Santander, C.; Ruiz, A.; García, S.; Aroca, R.; Cumming, J.; Cornejo, P. Efficiency of two arbuscular mycorrhizal fungal inocula to improve saline stress tolerance in lettuce plants by changes of antioxidant defense mechanisms. J. Sci. Food Agric. 2020, 100, 1577–1587. [Google Scholar] [CrossRef] [PubMed]
- Amprayn, K.O.; Rose, M.T.; Kecskés, M.; Pereg, L.; Nguyen, H.T.; Kennedy, I.R. Plant growth promoting characteristics of soil yeast (Candida tropicalis HY) and its effectiveness for promoting rice growth. Appl. Soil Ecol. 2012, 61, 295–299. [Google Scholar] [CrossRef]
- Mehta, S.; Nautiyal, C.S. An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr. Microbiol. 2001, 1, 51–56. [Google Scholar] [CrossRef]
- Edi-Premono, M.; Moawad, A.M.; Vleck, P.L.G. Effect of phosphate solubilizing Pseudomonas putida on the growth of maize and its survival in the rhizosphere. Indones. J. Crop Sci. 1996, 11, 13–23. [Google Scholar]
- Ahemad, M.; Khan, M.S. Evaluation of plant-growth-promoting activities of rhizobacterium Pseudomonas putida under herbicide stress. Ann. Microbiol. 2012, 62, 1531–1540. [Google Scholar] [CrossRef]
- Louden, B.C.; Haarmann, D.; Lynne, A.M. Use of Blue Agar CAS Assay for Siderophore Detection. J. Microbiol. Biol. Educ. 2011, 1, 51–53. [Google Scholar] [CrossRef] [PubMed]
- Dworkin, M.; Foster, J. Experiments with some microorganisms which utilize ethane and hydrogen. J. Bacteriol. 1958, 75, 592–603. [Google Scholar] [CrossRef] [PubMed]
- Penrose, D.M.; Glick, B.R. Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol. Plant 2003, 118, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Weber, R.P. The colorimetric estimation of indole acetic acid. Plant Physiol. 1951, 26, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Brick, J.M.; Bostock, R.M.; Silverstone, S.E. Rapid in situ assay for indole acetic acid production by bacteria immobilized on nitrocellulose membrane. Appl. Environ. Microbiol. 1991, 57, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, G.; Du, C.; Mou, H.; Cui, J.; Guan, H.; Hwang, H.; Wang, P. Characterization of high yield exopolysaccharide produced by Phyllobacterium sp. 921F exhibiting moisture preserving properties. Int. J. Biol. Macromol. 2017, 101, 562–568. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Maxwell, J.A. Rock and Mineral Analysis; Interscience Publishers: New York, NY, USA, 1968. [Google Scholar]
- Baker, D.E.; Amacher, M.C. Nickel, Copper, Zinc and Cadmium. In Methods of Soil Analysis Part 2: Chimical and Microbiological Properties, 2nd ed.; Page, A.L., Millers, R.H., Keeney, D.R., Eds.; Agronomy Monograph No. 9; American Society of Agronomy: Madison, WI, USA, 1982; pp. 323–334. [Google Scholar] [CrossRef]
- Wellburn, A.R. The Spectral Determination of Chlorophylls a and b, as well as Total Carotenoids, Using Various Solvents with Spectrophotometers of Different Resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and Carotenoids: Pigments of Photosynthetic Biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Beyer, W.F., Jr.; Fridovich, I. Assaying for superoxide dismutase activity: Some large consequences of minor changes in conditions. Anal. Biochem. 1987, 161, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen Peroxide is Scavenged by Ascorbate-specific Peroxidase in Spinach Chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Alonso, Á.M.; Guillén, D.A.; Barroso, C.G.; Puertas, B.; García, A. Determination of antioxidant activity of wine byproducts and its correlation with polyphenolic content. J. Agric. Food Chem. 2002, 50, 5832–5836. [Google Scholar] [CrossRef]
- Parada, J.; Valenzuela, T.; Gómez, F.; Tereucán, G.; García, S.; Cornejo, P.; Winterhalter, P.; Ruiz, A. Effect of fertilization and arbuscular mycorrhizal fungal inoculation on antioxidant profiles and activities in Fragaria ananassa fruit. J. Sci. Food Agric. 2019, 99, 1397–1404. [Google Scholar] [CrossRef]
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–161. [Google Scholar] [CrossRef]
- Giovanetti, M.; Mosse, B. An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytol. 1980, 84, 489–500. [Google Scholar] [CrossRef]
- Sieverding, E. Vesicular-Arbuscular Mycorrhiza Management in Tropical Agrosystems; GTZ: Eschborn, Germany, 1991; p. 371. [Google Scholar]









| Isolate | PSI | Phosphate Solubilization (mg mL−1) | EPS Production (g L−1) | IAA Production (μg mL−1) | Siderophore Production | ACC Deaminase Activity |
|---|---|---|---|---|---|---|
| AA | 2.30 ± 0.01 cd* | 0.55 ± 0.05 ab | 0.82 ± 0.15 ab | 9.75 ± 0.07 c | + | + |
| AB | 2.22 ± 0.01 d | 0.28 ± 0.03 b | 1.03 ± 0.07 ab | 8.86 ± 0.14 d | + | + |
| E4 | 2.57 ± 0.03 b | 0.69 ± 0.08 a | 0.46 ± 0.01 b | 9.24 ± 0.07 cd | − | + |
| F3 | 2.65 ± 0.02 ab | 0.43 ± 0.03 ab | 0.85 ± 0.12 ab | 8.67 ± 0.03 d | + | + |
| F4 | 2.36 ± 0.03 bcd | 0.37 ± 0.06 b | 0.66 ± 0.03 ab | 18.74 ± 0.07 a | + | + |
| RC | 2.86 ± 0.09 a | 0.48 ± 0.06 ab | 0.84 ± 0.14 ab | 8.83 ± 0.03 d | + | + |
| RD | 2.42 ± 0.01 bcd | 0.33 ± 0.05 b | 0.67 ± 0.22 ab | 10.75 ± 0.07 b | − | + |
| RG | 2.48 ± 0.01 bcd | 0.43 ± 0.02 ab | 0.89 ± 0.15 ab | 8.90 ± 0.08 d | + | + |
| OM | 2.51 ± 0.04 b | 0.27 ± 0.02 b | 1.34 ± 0.16 a | 11.01 ± 0.21 b | + | + |
| PM | 2.30 ± 0.01 cd | 0.28 ± 0.08 b | 0.64 ± 0.11 b | 11.27 ± 0.14 b | − | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez, R.; Tapia, Y.; Antilén, M.; Ruiz, A.; Pimentel, P.; Santander, C.; Aponte, H.; González, F.; Cornejo, P. Beneficial Interactive Effects Provided by an Arbuscular Mycorrhizal Fungi and Yeast on the Growth of Oenothera picensis Established on Cu Mine Tailings. Plants 2023, 12, 4012. https://doi.org/10.3390/plants12234012
Pérez R, Tapia Y, Antilén M, Ruiz A, Pimentel P, Santander C, Aponte H, González F, Cornejo P. Beneficial Interactive Effects Provided by an Arbuscular Mycorrhizal Fungi and Yeast on the Growth of Oenothera picensis Established on Cu Mine Tailings. Plants. 2023; 12(23):4012. https://doi.org/10.3390/plants12234012
Chicago/Turabian StylePérez, Rodrigo, Yasna Tapia, Mónica Antilén, Antonieta Ruiz, Paula Pimentel, Christian Santander, Humberto Aponte, Felipe González, and Pablo Cornejo. 2023. "Beneficial Interactive Effects Provided by an Arbuscular Mycorrhizal Fungi and Yeast on the Growth of Oenothera picensis Established on Cu Mine Tailings" Plants 12, no. 23: 4012. https://doi.org/10.3390/plants12234012