Surveillance of Landraces’ Seed Health in South Italy and New Evidence on Crop Diseases
Abstract
:1. Introduction
2. Results
2.1. Liliaceous Crops: Garlic (Allium sativum L.) and Onion (Allium cepa L.)
2.2. Leguminous Crops and Novel Pathogen Reports
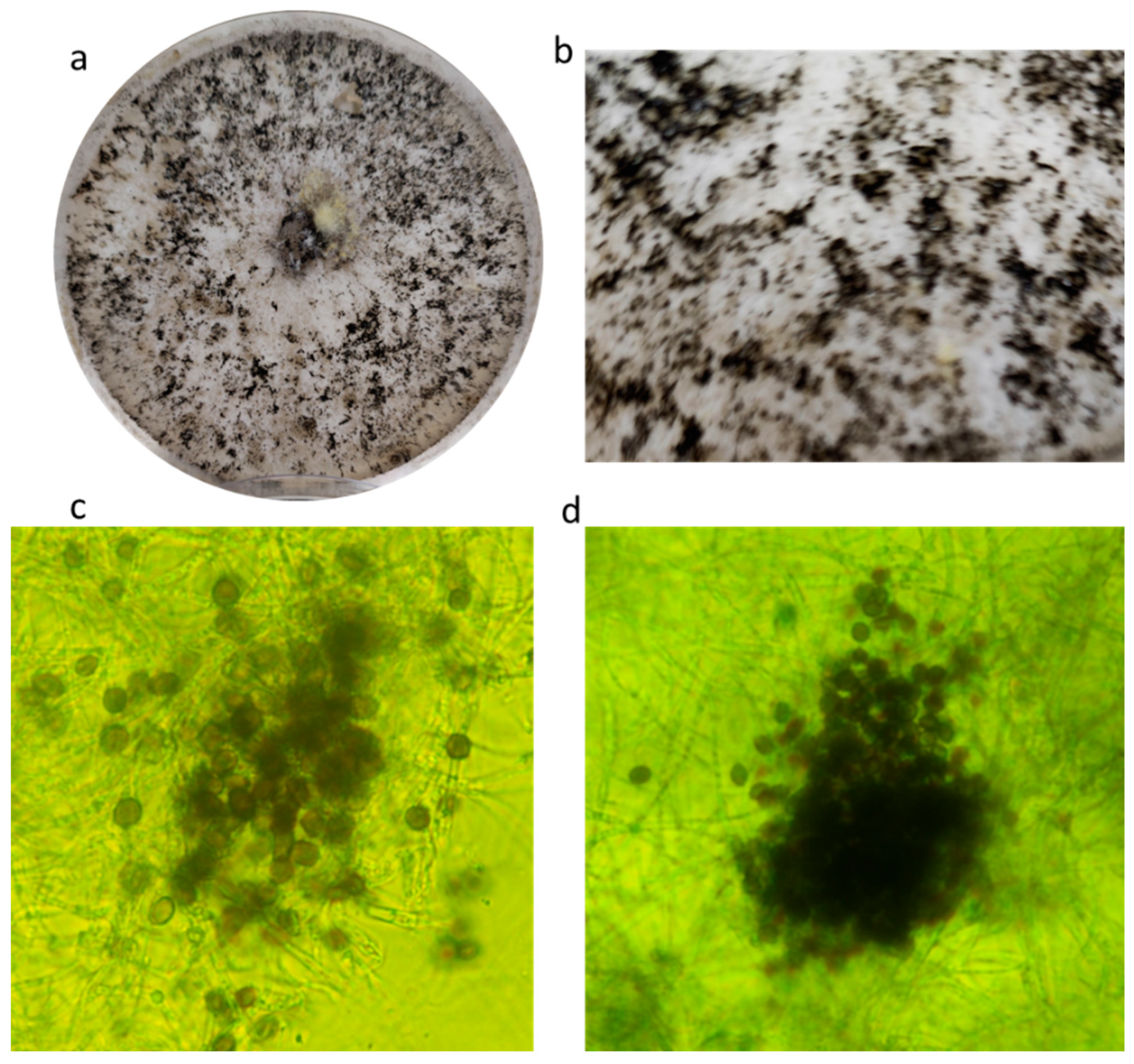
2.2.1. Cowpea (Vigna unguiculata (L.) Walp) and Common Bean (Phaseolus vulgaris L.)
2.2.2. Pea (Pisum sativum L.) and Lentils (Lens culinaris Medik.)
2.2.3. Broad Bean (Vicia faba L.) and Grass Pea (Lathyrus cicera L.)
2.3. Brassicaceae
3. Discussion
4. Materials and Methods
4.1. Sampling and Isolation of Fungal and Bacterial Strains from Seeds
4.1.1. Mycological Screening
4.1.2. Detection of Target Bacteria
4.2. Virological Analyses
4.3. Isolation from Symptomatic Mother Plants: The Case of Lentils
4.4. Morphological Characterization of Fungal Colonies
4.5. Molecular Analyses and Phylogenesis
4.6. Pathogenicity Tests
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tait, J.; Morris, D. Sustainable development of agricultural systems: Competing objectives and critical limits. Futures 2000, 32, 247–260. [Google Scholar] [CrossRef]
- Villa, T.C.C.; Maxted, N.; Scholten, M.; Ford-Lloyd, B. Defining and identifying crop landraces. Plant Genet. Resour. 2005, 3, 373–384. [Google Scholar] [CrossRef]
- Azeez, M.A.; Adubi, A.O.; Durodola, F.A.; Azeez, M.A.; Adubi, A.O.; Durodola, F.A. Landraces and Crop Genetic Improvement. In Rediscovery of Landraces as a Resource for the Future; Grillo, O., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Montesano, V.; Negro, D.; Sarli, G.; Logozzo, G.; Zeuli, P.S. Landraces in Inland areas of the Basilicata region, Italy: Monitoring and perspectives for on farm conservation. Genet. Resour. Crop Evol. 2012, 59, 701–716. [Google Scholar] [CrossRef]
- Bertacchini, E. Regional legislation in Italy for the protection of local varieties. J. Agric. Environ. Int. Dev. 2009, 103, 51–63. [Google Scholar] [CrossRef]
- Giupponi, L.; Pedrali, D.; Leoni, V.; Rodari, A.; Giorgi, A. The Analysis of Italian Plant Agrobiodiversity Databases Reveals That Hilly and Sub-Mountain Areas Are Hotspots of Herbaceous Landraces. Diversity 2021, 13, 70. [Google Scholar] [CrossRef]
- Decision adopted by the conference of the parties to the convention on biological diversity at its tenth meeting X/2. The Strategic Plan for Biodiversity 2011–2020 and the Aichi Biodiversity Targets. 2010. Available online: https://www.cbd.int/decision/cop/?id=12268 (accessed on 27 October 2022).
- Gibson, R.W. A review of perceptual distinctiveness in landraces including an analysis of how its roles have been overlooked in plant breeding for low-input farming systems. Econ. Bot. 2009, 63, 242–255. [Google Scholar] [CrossRef]
- Piergiovanni, A.R.; Margiotta, B. On farm survival of apulian legume and cereal landraces in relation to land cover/land use changes. A case study. Ital. J. Agron. 2021, 16, 1–9. [Google Scholar] [CrossRef]
- Pícha, K.; Navrátil, J.; Švec, R. Preference to Local Food vs. Preference to “National” and Regional Food. J. Food Prod. Mark. 2018, 24, 125–145. [Google Scholar] [CrossRef]
- Corrado, G.; Rao, R. Towards the Genomic Basis of Local Adaptation in Landraces. Diversity 2017, 9, 51. [Google Scholar] [CrossRef]
- EU Commission. EUROPE 2020—A Strategy for Smart, Sustainable and Inclusive Growth; EU Commission: Brussels, Belgium, 2010. [Google Scholar]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development; United Nations: New York, NY, USA, 2015. [Google Scholar]
- Piergiovanni, A.R.; Lioi, L. Italian Common Bean Landraces: History, Genetic Diversity and Seed Quality. Diversity 2010, 2, 837–862. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Plant Genetic Resources for Food and Agriculture Synthetic Account; The Second Report on The State of the World’s; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010. [Google Scholar]
- The Second Report on the State of the World’s Plant Genetic Resources for Food and Agriculture (SoWPGR-2)|Knowledge for Policy. Available online: https://knowledge4policy.ec.europa.eu/publication/second-report-state-world’s-plant-genetic-resources-food-agriculture-sowpgr-2_en (accessed on 25 January 2023).
- Denancé, N.; Grimault, V. Seed pathway for pest dissemination: The ISTA Reference Pest List, a bibliographic resource in non-vegetable crops. EPPO Bull. 2022, 52, 434–445. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Building on Gender, Agrobiodiversity and Local Knowledge | International Treaty on Plant Genetic Resources for Food and Agriculture. Available online: https://www.fao.org/plant-treaty/tools/toolbox-for-sustainable-use/details/en/c/1178627/ (accessed on 25 January 2023).
- Dell’Olmo, E.; Zaccardelli, M.; Basile, B.; Corrado, G.; Sigillo, L. Identification and Characterization of New Seedborne Pathogens in Phaseolus vulgaris Landraces of Southern Italy. Pathogens 2023, 12, 108. [Google Scholar] [CrossRef] [PubMed]
- ISF Regulated Pest List Initiative—International Seed Federation. Available online: https://worldseed.org/resources/isf-regulated-pest-list-database/isf-regulated-pest-list-initiative/ (accessed on 25 January 2023).
- Epitome: International Journal of Multidisciplinary Research. Available online: http://epitomejournals.com/ (accessed on 25 January 2023).
- Common Names of Plant Diseases. Available online: https://www.apsnet.org/edcenter/resources/commonnames/Pages/default.aspx (accessed on 25 January 2023).
- International Seed Testing Association (ISTA). Seed Health Methods—International Rules for Seed Testing. Available online: https://www.seedtest.org/en/international-rules-for-seed-testing/seed-health-methods-product-1054.html (accessed on 29 December 2022).
- Infantino, A. Malattie fungine delle leguminose minori. In LEGUMINOSE MINORI (CECE, LENTICCHIA, CICERCHIA, FAVA) MONOGRAFIA—A Cura di Massimo Zaccardelli; MiPAAF: Rome, Italy, 2008; 83p. [Google Scholar]
- Garkoti, A.; Kumar, S.; Lal, M.; Singh, V. Major diseases of lentil: Epidemiology and disease management—A Review. Agriways 2013, 1, 62–64. [Google Scholar]
- Harveson, R.M.; Pasche, J.S.; Porter, L.; Chen, W.; Burrows, M. Compendium of Pea Diseases and Pests; American Phytopathological Society: Saint Paul, MN, USA, 2021; 130p. [Google Scholar]
- Omomowo, O.I.; Babalola, O.O. Constraints and Prospects of Improving Cowpea Productivity to Ensure Food, Nutritional Security and Environmental Sustainability. Front. Plant Sci. 2021, 12, 2392. [Google Scholar] [CrossRef] [PubMed]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Alves, A.; Crous, P.W.; Correia, A.; Phillips, A.J.L.; Alves, A. Morphological and molecular data reveal cryptic speciation in Lasiodiplodia theobromae. Fungal Divers. 2008, 28, 1–13. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. AMPLIFICATION AND DIRECT SEQUENCING OF FUNGAL RIBOSOMAL RNA GENES FOR PHYLOGENETICS. In PCR Protocols; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: Cambridge, MA, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- O’Donnell, K.; Cigelnik, E. Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus Fusarium are nonorthologous. Mol. Phylogenet. Evol. 1997, 7, 103–116. [Google Scholar] [CrossRef]
- Santos, K.M.; Lima, G.S.; Barros, A.P.O.; Machado, A.R.; Souza-Motta, C.M.; Correia, K.C.; Michereff, S.J. Novel specific primers for rapid identification of Macrophomina species. Eur. J. Plant Pathol. 2020, 156, 1213–1218. [Google Scholar] [CrossRef]
- Dyko, B.J.; Sutton, B.C. New and interesting dematiaceous Hyphomycetes from Florida. Mycotaxon 1979, 8, 119–124. [Google Scholar]
- Ondřej, M.; Huňady, I. Faba bean (Vicia faba L.) breeding for resistance to anthracnose (Ascochyta fabae Speg.) in the Czech Republic. Czech J. Genet. Plant Breed. 2007, 43, 61–68. [Google Scholar] [CrossRef]
- Bolton, M.D.; Thomma, B.P.H.J.; Nelson, B.D. Sclerotinia sclerotiorum (Lib.) de Bary: Biology and molecular traits of a cosmopolitan pathogen. Mol. Plant Pathol. 2006, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Simmons, E.G. Perfect states of Stemphylium. Mycologia 1969, 61, 1–26. [Google Scholar] [CrossRef]
- Vaghefi, N.; Thompson, S.M.; Kimber, R.B.E.; Thomas, G.J.; Kant, P.; Barbetti, M.J.; van Leur, J.A.G. Multi-locus phylogeny and pathogenicity of Stemphylium species associated with legumes in Australia. Mycol. Prog. 2020, 19, 381–396. [Google Scholar] [CrossRef]
- Simmons, E.G. Alternaria themes and variations (22-26). Mycotaxon 1986, 25, 287–308. [Google Scholar]
- Tonti, S.; Prà, M.D.; Nipoti, P.; Prodi, A.; Alberti, I. First Report of Fusarium proliferatum Causing Rot of Stored Garlic Bulbs (Allium sativum L.) in Italy. J. Phytopathol. 2012, 160, 761–763. [Google Scholar] [CrossRef]
- Alberti, I.; Prodi, A.; Montanari, M.; Paglia, G.; Asioli, C.; Nipoti, P. First report of Fusarium proliferatum associated with Allium fistulosum L. in Italy. J. Plant Dis. Prot. 2018, 125, 231–233. [Google Scholar] [CrossRef]
- Gálvez, L.; Palmero, D. Fusarium Dry Rot of Garlic Bulbs Caused by Fusarium proliferatum: A Review. Horticulturae 2022, 8, 628. [Google Scholar] [CrossRef]
- Pintos, A.; Alvarado, P. Phylogenetic delimitation of Apiospora and Arthrinium. Fungal Syst. Evol. 2021, 7, 197. [Google Scholar] [CrossRef]
- Santo, A.P.; Cuzman, O.A.; Petrocchi, D.; Pinna, D.; Salvatici, T.; Perito, B. Black on white: Microbial growth darkens the external marble of florence cathedral. Appl. Sci. 2021, 11, 6163. [Google Scholar] [CrossRef]
- Micheluz, A.; Manente, S.; Prigione, V.; Tigini, V.; Varese, G.C.; Ravagnan, G. The effects of book disinfection to the airborne microbiological community in a library environment. Aerobiologia 2018, 34, 29–44. [Google Scholar] [CrossRef]
- Monkai, J.; Phookamsak, R.; Tennakoon, D.S.; Bhat, D.J.; Xu, S.; Li, Q.; Xu, J.; Mortimer, P.E.; Kumla, J.; Lumyong, S. Insight into the Taxonomic Resolution of Apiospora: Introducing Novel Species and Records from Bamboo in China and Thailand. Diversity 2022, 14, 918. [Google Scholar] [CrossRef]
- Bagherabadi, S.; Zafari, D.; Ghobadi Anvar, F. First report of leaf spot caused by Arthrinium arundinis on rosemary in Iran. J. Plant Pathol. 2014, 96, S4.126. [Google Scholar] [CrossRef]
- Liao, J.; Jiang, W.; Wu, X.; He, J.; Li, H.; Wang, T.; Cheng, L.; Chen, W.; Mo, L. First Report of Apiospora Mold on Sugarcane in China Caused by Apiospora arundinis (Arthrinium arundinis). Plant Dis. 2022, 106, 1058. [Google Scholar] [CrossRef] [PubMed]
- Marquez, N.; Giachero, M.L.; Declerck, S.; Ducasse, D.A. Macrophomina phaseolina: General Characteristics of Pathogenicity and Methods of Control. Front. Plant Sci. 2021, 12, 634397. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Venturi, V. Synergisms between microbial pathogens in plant disease complexes: A growing trend. Front. Plant Sci. 2015, 6, 385. [Google Scholar] [CrossRef]
- Cantore, P.L.; Giorgio, A.; Campion, B.; Iacobellis, N.S. Development of integrated disease control measures for the valorisation of traditional crops in Southern Italy: The case study of Fagioli di Sarconi. In The Sustainability of Agro-Food and Natural Resource Systems in the Mediterranean Basin; Vastola, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 145–158. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the pest categorisation of Xanthomonas axonopodis pv. phaseoli and Xanthomonas fuscans subsp. fuscans. EFSA J. 2014, 12, 3856. [Google Scholar] [CrossRef]
- EPPO. Xanthomonas phaseoli pv. phaseoli (XANTPH) [World distribution]—EPPO Global Database. Available online: https://gd.eppo.int/taxon/XANTPH/distribution (accessed on 27 December 2022).
- Wang, Y.; Gaba, V.; Yang, J.; Palukaitis, P.; Gal-On, A. Characterization of Synergy Between Cucumber mosaic virus and Potyviruses in Cucurbit Hosts. Phytopathology 2002, 92, 51–58. [Google Scholar] [CrossRef]
- Tarquini, G.; Martini, M.; Maestri, S.; Firrao, G.; Ermacora, P. The Virome of ‘Lamon Bean’: Application of MinION Sequencing to Investigate the Virus Population Associated with Symptomatic Beans in the Lamon Area, Italy. Plants 2022, 11, 779. [Google Scholar] [CrossRef]
- Aishwarya, P.; Rangaswamy, K.T.; Basavaraju, S.; Achari, R.; Govin, K.; Rudraswamy, K.P.; Prameela, H.A. Evaluation of the Seed-borne Nature of Bean Common Mosaic Virus (BCMV) in Cowpea. Int. J. Curr. Microbiol. Appl. Sci. 2020, 9, 239–245. [Google Scholar] [CrossRef]
- Shankar, A.C.U.; Nayaka, S.C.; Niranjan-Raj, S.; Kumar, H.B.; Reddy, M.S.; Niranjana, S.R.; Prakash, H.S. Rhizobacteria-mediated resistance against the blackeye cowpea mosaic strain of bean common mosaic virus in cowpea (Vigna unguiculata). Pest Manag. Sci. 2009, 65, 1059–1064. [Google Scholar] [CrossRef]
- Sivachandra Kumar, N.T.; Banniza, S. Assessment of the effect of seed infection with Ascochyta pisi on pea in western Canada. Front. Plant Sci. 2017, 8, 933. [Google Scholar] [CrossRef] [PubMed]
- Jiskani, A.M.; Samo, Y.; Soomro, M.A.; Leghari, Z.H.; Ghulam Nabi Gishkori, Z.; Hussain Bhutto, S.; Qayoom Majeedano, A. A destructive disease of lentil: Fusarium wilt of lentil. Plant Arch. 2021, 21, 2117–2127. [Google Scholar] [CrossRef]
- Antonopoulos, D.F.; Elena, K. Susceptibility of Greek alfalfa and clover cultivars to Fusarium oxysporum f. sp. medicaginis and potential methods of disease control. J. Plant Dis. Prot. 2008, 115, 162–166. [Google Scholar] [CrossRef]
- CABI. Distribution Maps of Plant Diseases. Available online: https://www.cabi.org/publishing-products/distribution-maps-of-plant-diseases/ (accessed on 25 January 2023).
- Omri Ben Youssef, N.; Kerdraon, L.; Mieuzet, L.; Halila, I.; Jammezi, N.; Mbazia, A.; Kharrat, M.; Le May, C. Population structure of the faba bean blight pathogen Ascochyta fabae (teleomorph, Didymella fabae) in Tunisia. Phytopathol. Mediterr. 2019, 58, 81–94. [Google Scholar] [CrossRef]
- Ahmed, S.; Abang, M.M.; Maalouf, F. Integrated management of Ascochyta blight (Didymella fabae) on faba bean under Mediterranean conditions. Crop Prot. 2016, 81, 65–69. [Google Scholar] [CrossRef]
- Antwi-Boasiako, A.; Wang, Y.; Dapaah, H.K.; Zhao, T. Mitigating against Sclerotinia Diseases in Legume Crops: A Comprehensive Review. Agronomy 2022, 12, 3140. [Google Scholar] [CrossRef]
- Njambere, E.N.; Peever, T.L.; Vandemark, G.; Chen, W. Genotypic variation and population structure of Sclerotinia trifoliorum infecting chickpea in California. Plant Pathol. 2014, 63, 994–1004. [Google Scholar] [CrossRef]
- Derbyshire, M.C.; Denton-Giles, M. The control of sclerotinia stem rot on oilseed rape (Brassica napus): Current practices and future opportunities. Plant Pathol. 2016, 65, 859–877. [Google Scholar] [CrossRef]
- Danielson, G.A.; Nelson, B.D.; Helms, T.C. Effect of Sclerotinia Stem Rot on Yield of Soybean Inoculated at Different Growth Stages. Plant Dis. 2004, 88, 297–300. [Google Scholar] [CrossRef]
- Chapara, V.; Chittem, K.; Mendoza, L.E.D.R. First Report of White Mold Caused by Sclerotinia sclerotiorum on Faba Beans in North Dakota. Plant Dis. 2018, 102, 1669. [Google Scholar] [CrossRef]
- Hanse, B.; Raaijmakers, E.E.M.; Schoone, A.H.L.; van Oorschot, P.M.S. Stemphylium sp., the cause of yellow leaf spot disease in sugar beet (Beta vulgaris L.) in the Netherlands. Eur. J. Plant Pathol. 2015, 142, 319–330. [Google Scholar] [CrossRef]
- Woudenberg, J.H.C.; Hanse, B.; van Leeuwen, G.C.M.; Groenewald, J.Z.; Crous, P.W. Stemphylium revisited. Stud. Mycol. 2017, 87, 77–103. [Google Scholar] [CrossRef] [PubMed]
- Pantidou, M.E. Fungus-Host Index for Greece; CABI: Wallingford, UK, 1973. [Google Scholar]
- El-Buni, A.M.; Rattan, S.S. Flora of Libya. Suppl. In Check List of Libyan fungi; Al Faateh University: Tripoli, Libya, 1981. [Google Scholar]
- Cook, P.R.; Dube, A.J. Host-Pathogen Index of Plant Diseases in South Australia; Field Crops Pathology Group South Australian Department of Agriculture: Adelaide, Australia, 1989. [Google Scholar]
- Mułenko, W.; Majewski, T.; Ruszkiewicz-Michalska, M. A Preliminary Checklist of Micromycetes in Poland; CABI: Wallingford, UK, 2008. [Google Scholar]
- Sheikh, F.; Dehghani, H.; Aghajani, M.A. Screening faba bean (Vicia faba L.) genotypes for resistance to Stemphylium blight in Iran. Eur. J. Plant Pathol. 2015, 143, 677–689. [Google Scholar] [CrossRef]
- Foister, C.E. The economic plant diseases of Scotland. Tech. Bull. Dep. Agric. Scot. 1961, 1, 1–210. [Google Scholar]
- Orieux, L.; Felix, S. List of Plant Diseases in Mauritius; Phytopathological Papers; Commonwealth Mycological Institute: Kew, UK, 1968; Volume 7, 48p. [Google Scholar]
- Samac, D.A.; Willbur, J.; Behnken, L.; Brietenbach, F.; Blonde, G.; Halfman, B.; Jensen, B.; Sheaffer, C. First Report of Stemphylium globuliferum Causing Stemphylium Leaf Spot on Alfalfa (Medicago sativa) in the United States. Plant Dis. 2014, 98, 993. [Google Scholar] [CrossRef]
- Omar, A.F.; Hafez, Y.M.; Emeran, A.A. Occurrence of a New Leaf Spot Disease on the Egyptian Clover (Trifolium alexandrinum L.) Caused by Stemphylium globuliferum (Vestergren) in Egypt. Egypt. J. Biol. Pest Control 2015, 25, 89–96. [Google Scholar]
- Lambein, F.; Travella, S.; Kuo, Y.H.; Van Montagu, M.; Heijde, M. Grass pea (Lathyrus sativus L.): Orphan crop, nutraceutical or just plain food? Planta 2019, 250, 821–838. [Google Scholar] [CrossRef]
- EPPO. Alternaria brassicicola (ALTEBI) [Overview]—EPPO Global Database. Available online: https://gd.eppo.int/taxon/ALTEBI (accessed on 28 December 2022).
- Macioszek, V.K.; Gapińska, M.; Zmienko, A.; Sobczak, M.; Skoczowski, A.; Oliwa, J.; Kononowicz, A.K. Complexity of Brassica oleracea–Alternaria brassicicola Susceptible Interaction Reveals Downregulation of Photosynthesis at Ultrastructural, Transcriptional, and Physiological Levels. Cells 2020, 9, 2329. [Google Scholar] [CrossRef]
- Sigillo, L.; Esposito, S.; Tripodi, P.; Serratore, G.; Pane, C. Host range and molecular typing of Xanthomonas spp. strains isolated from wild rocket (Diplotaxis tenuifolia) in Italy. Eur. J. Plant Pathol. 2021, 160, 693–705. [Google Scholar] [CrossRef]
- Scortichini, M.; Rossi, M. Recurrent infections of Xanthomonas campestris pv. campestris on crucifers in Central-Southern Italy. Inf. Fitopatol. 1994, 12, 48–50. [Google Scholar]
- Cook, A.A.; Walker, J.C.; Larson, R. Studies on the disease cycle of black rot of crucifers. Phytopathology 1952, 42, 162–167. [Google Scholar]
- Frost, K.E.; Groves, R.L.; Charkowski, A.O. Integrated Control of Potato Pathogens Through Seed Potato Certification and Provision of Clean Seed Potatoes. Plant Dis. 2013, 97, 1268–1280. [Google Scholar] [CrossRef] [PubMed]
- ISF. Detection of Pseudomonas syringae pv. pisi on Pea Seed; Developed by ISHI-Veg; ISF: Nypn, Switzerland, 2020. [Google Scholar]
- EPPO. PM 7/125 (1) ELISA tests for viruses. EPPO Bull. 2015, 45, 445–449. [Google Scholar] [CrossRef]
- Champion, R. Identifier les Champignons Transmis par les Semences—RÉMI CHAMPION—Achat Livre ou Ebook | fnac. Available online: https://livre.fnac.com/a133884/REMI-CHAMPION-Identifier-les-champignons-transmis-par-les-semences (accessed on 19 December 2022).
- Crous, P.W.; Groenewald, J.Z. A phylogenetic re-evaluation of Arthrinium. IMA Fungus 2013, 4, 133–154. [Google Scholar] [CrossRef]
- Ordóñez-Valencia, C.; Ferrera-Cerrato, R.; Quintanar-Zúñiga, R.E.; Flores-Ortiz, C.M.; Guzmán, G.J.M.; Alarcón, A.; Larsen, J.; García-Barradas, O. Morphological development of sclerotia by Sclerotinia sclerotiorum: A view from light and scanning electron microscopy. Ann. Microbiol. 2015, 65, 765–770. [Google Scholar] [CrossRef]
- Ramírez, M.; Pérez, L.M.; Montealegre, J.R. Susceptibility of different grapevine (Vitis vinifera L.) cultivars to Diplodia seriata and Diplodia mutila. Int. J. Agric. Nat. Resour. 2018, 45, 93–98. [Google Scholar] [CrossRef]









| Species | Accession | Geographic Area of Cultivation | Multiplication Field | Target Fungi (a) | Target Bacteria (b) | Target Viruses (c) | Analytical Methods (To Detect Pathogens (a, b, c)) | Literature |
|---|---|---|---|---|---|---|---|---|
| A. cepa | Agostina | Agro acerrano-mariglianese (NA) 1 | in situ | Botrytis spp. Alternaria porri Stemphylium vesicarium | / | / | (a) Mycological screening (agar plate method) | [20] |
| di Alife (alifana) | Alife (CE) 2 | ex situ | ||||||
| di Eremiti | Eremiti (SA) 3 | in situ | ||||||
| A. sativum | Bianco locale | Avellinese | in situ | Fusarium spp. Botrytis spp. Macrophomina phaseolina Sclerotinia spp. | Erwinia carotovora subsp. carotovora | / | (a) Mycological screening (agar plate method) (b) Isolation on Nutrient agar | [21,22] |
| ecotipo locale di Caposele | Caposele (AV) 4 | in situ | ||||||
| Brassica oleracea var. italica | Broccolo del Vallo di Diano | Vallo di Diano (NA) | ex situ | Alternaria brassicae, A. brassicicola, A. japonica, Phoma lingam Rizoctonia solani Sclerotinia spp. | Xanthomonas campestris pv. campestris and | / | (a) Mycological screening (agar plate method) (b) ISTA 7-019a (dilution plating method) | [20,23] |
| Broccolo dell’Olio | Agro nocerino-sarnese (SA) | ex situ | ||||||
| Broccolo di Natale | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Broccolo San Pasquale | Agro acerrano-mariglianese (NA) | ex situ | ||||||
| Brassica oleracea var. sabellica | Torzella riccia | Agro acerrano-mariglianese (NA) | ex situ | |||||
| Brassica rapa var. rapa | Catozza | Casertano, napoletano and Agro nocerino-sarnese (SA) | ex situ | Alternaria brassicae, A. brassicicola, A. japonica, Phoma lingam Rizoctonia solani Sclerotinia spp. | Xanthomonas campestris pv. campestris | / | (a) Mycological screening (agar plate method) (b) ISTA 7-019a (dilution plating method) | |
| L. sativus | Alta Irpinia | Avellinese | in situ | Ascochyta sp. Fusarium spp. Botrytis spp. | Pseudomonas spp. | / | (a) Mycological screening (agar plate method) (b) Dilution plating method | [24] |
| di Calitri | Calitri (AV) | ex situ | ||||||
| di Colliano | Colliano (SA) | ex situ | ||||||
| di Montefalcone | Montefalcone (BN) 5 | in situ | ||||||
| L. cicera | Maracuocciolo/Maracuccia | Ascea/Camerota (SA) | ex situ | Ascochyta sp. Fusarium spp. Botrytis spp. | Pseudomonas spp. | / | (a) Mycological screening (agar plate method) (b) Dilution plating method | [24] |
| L. culinaris | di Colliano | Calitri (AV) | in situ | Ascochyta sp. Fusarium spp. Botrytis spp. Sclerotinia spp. Rizoctonia solani | / | / | (a) Isolation on PDA | [24,25] |
| P. vulgaris | Bianco di Villa Santa Croce | Villa di Santa Croce (CE) | ex situ | Colletotrichum lindemuthianum Fusarium spp. Ascochyta phaseolorum Rizoctonia solani Macrophomina phaseolina Sclerotinia sclerotiorum Botrytis spp. | Pseudomonas savastanoi pv. phaseolicola Pseudomonas syringae pv. syringae Xanthomonas axonopodis pv. phaseoli | AMV CMV BCMV BCMNV | (a) Mycological 1 screening (agar plate method) (b) ISTA 7-21; ISTA 7-023 (Dilution plating method) (c) DAS ELISA (EPPO Standard PM 7/125(1)) | [23] |
| Butirro Acc. 2021 | Vico Equense (NA) | ex situ | ||||||
| Butirro Acc. 2020 | Vico Equense (NA) | ex situ | ||||||
| Cannellino Sessantino dei 30 anni | Acerra (NA) | in situ | ||||||
| Cannellino Bianco di Calitri | Calitri (AV) | in situ | ||||||
| dei 7 anni | Visciano (NA) | in situ | ||||||
| dei Signori | Agro nocerino-sarnese (SA) | ex situ | ||||||
| della Regina | Valle d’ll’Angelo (SA) | ex situ | ||||||
| della Regina acc.2 2021 Bianco | Montano Antilia (SA) | ex situ | ||||||
| della Regina Acc.2 2021 Gandolfi | Montano Antilia (SA) | ex situ | ||||||
| della Regina Acc.2 2021 Mazzamauro | Montano Antilia (SA) | ex situ | ||||||
| della Regina Acc.2 | San Lupo (BN) | ex situ | ||||||
| Della Regina di Gorga | Gorga (SA) | ex situ | ||||||
| della Regina | Montano Antilia (SA) | ex situ | ||||||
| Dente di Morto Acc. 2021 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.1 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.2 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.3 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.4 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.5 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.6 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc.7 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| Dente di Morto Acc. 2020 | Agro acerrano-mariglianese (NA) | in situ | ||||||
| di Prata Melizzano | Prata Sanni–a–Melizzano (BN) | ex situ | ||||||
| di Volturara Irpina | Volturara Irpina (NA) | in situ | ||||||
| Fasulo a tubbettiello | Ruviano (CE) | ex situ | ||||||
| Giallo del Fortore | Alto Fortore (BN) | ex situ | ||||||
| Lardari | Agerola (NA) | in situ | ||||||
| Regina | Grottaminarda (AV) | in situ | ||||||
| Rosso di Acerra | Acerra (NA) | ex situ | ||||||
| Schiacciatello | Cellole (SA) | ex situ | ||||||
| Tabaccanti | Vallo di Diano (NA) | ex situ | ||||||
| Tondino Bianco di Calitri | Calitri (AV) | in situ | ||||||
| Tondino di Castel di Sasso Acc. 2022 | Castel di Sasso (CE) | ex situ | ||||||
| Tondino di Villaricca | Villaricca (NA) | ex situ | ||||||
| Tondino di Villaricca | Villaricca (NA) | ex situ | ||||||
| Tondo Bianco | Sarno (SA) | ex situ | ||||||
| P. sativum | Centogiorni 2021 | Vesuvio/Agro nocerino-sarnese (NA) | ex situ | Ascochyta spp. Macrophomina phaseolina Rizoctonia solani Fusarium spp. | Pseudomonas syringae pv. pisi | CMV | (a) Mycological screening (agar plate method) (b) ISHI-Veg (Di-lution plating method) (c) DAS ELISA (EPPO Standard PM | [26] |
| Centogiorni Acc. 2021 | Acerra (NA) | ex situ | ||||||
| Centogiorni Acc. 2021 | Pollenatrocchia (NA) | in situ | ||||||
| Centogiorni Somma Vesuviana Acc. 2021 | Somma Vesuviana (NA) | in situ | ||||||
| Santacroce | Flegrea Area (NA) | in situ | ||||||
| Santacroce Acc. M 2021 | Flegrea Area (NA) | ex situ | ||||||
| Santacroce Acc. N 2021 | Flegrea Area (NA) | ex situ | ||||||
| Santacroce Acc. T 2021 | Napoletano | ex situ | ||||||
| V. faba | A sciabola Acc. 2021 | Agro acerrano-mariglianese (NA) | in situ | Ascochyta fabae Rizoctonia solani | Pseudomonas spp. | CMV | (a) Mycological screening (agar plate method) (b) Dilution plating method (c) DAS ELISA (EPPO Standard PM 7/125(1)) | [24] [27] |
| Di Colliano | COLLIANO (SA) | ex situ | ||||||
| lunga | Somma Vesuviana (NA) | ex situ | ||||||
| lunga Acc. 2020 | Nocerino-sarnese (NA) | ex situ | ||||||
| lunga Acc. 2021 | Nocerino-sarnese (NA) | ex situ | ||||||
| A Corna | Agro acerrano-mariglianese (NA) | ex situ | ||||||
| V. unguiculata | Corna dei Signori | Castel S. Giorgio (SA) | in situ | Colletotrichum sp., Fusarium spp. Rizoctonia solani Macrophomina phaseolina Sclerotinia sclerotiorum | Pseudomonas spp. | CMV, BCMV | (a) Mycological screening (agar plate method) (b) Dilution plating method (c) DAS ELISA (EPPO Standard PM 7/125(1)) | [27] |
| Corna dei signori (San Marzano) | San Marzano sul Sarno (SA) | ex situ | ||||||
| Fagiolino lungo San Marzano | San Marzano sul Sarno (SA) | in situ |
| Accession Number | Locus | Strain Code | Species | Host/Species–Landrace |
|---|---|---|---|---|
| OQ280977 | ITS | CREA OF 1115.1 | Alternaria alternata | Allium cepa–“Agostina” |
| OQ132873 | ITS | CREA OF 1128.1 | Apiospora arundinis | P. vulgaris–“Tondino di Castel di Sasso” |
| OQ133464 | ITS | CREA OF 1113.1 | Sclerotinia sclerotiorum | V.–faba–“Lunga (nocerino-sarnese) |
| OQ134131 | ITS | CREA OF 190.4 | Fusarium oxysporum | L. culinaris–“di Colliano” |
| OQ134394 | ITS | CREA OF 1231.3 | Alternaria brassicicola | B. oleracea var. sabellica–“Torzella riccia” |
| OQ148383 | ITS | CREA OF 1231.1 | Alternaria alternata | B. oleracea var. sabellica–Torzella riccia” |
| OQ148397 | ITS | CREA OF 1231.5 | Alternaria alternata | B. oleracea var. sabellica– “Torzella riccia” |
| OQ148398 | ITS | CREA OF 1231.4 | Alternaria alternata | B. oleracea var. sabellica– “Torzella riccia” |
| OQ148399 | ITS | CREA OF 1231.6 | Alternaria alternata | B. oleracea var. sabellica– “Torzella riccia” |
| OQ148403 | ITS | CREA OF 1235.2 | Ascochyta fabae | V. faba–“A Corna” |
| OQ148428 | ITS | CREA OF 1122.4 | Ascochyta pisi | P. sativum –“Centogiorni (Pollenatrocchia)” |
| OQ148433 | ITS | CREA OF 1122.5 | Ascochyta pisi | P. sativum–“Santacroce Acc.M” |
| OQ148577 | ITS | CREA OF 1119.1 | Fusarium proliferatum | A. sativum–“Bianco locale” |
| OQ148588 | ITS | CREA OF 355.4 | Stemphylium globuliferum | L. cicera–“Maracuoccio” |
| OQ249627 | β-tub | CREA OF 1128.1 | Apiospora arundinis | P. vulgaris–“Tondino di Castel di Sasso” |
| OQ249628 | TEF 1-α | CREA OF 1113.1 | Sclerotinia sclerotiorum | V. faba–“Lunga (nocerino-sarnese) |
| OQ249629 | TEF 1-α | CREA OF 190.4 | Fusarium oxysporum | L. culinaris–“Di Colliano” |
| OQ249630 | TEF 1-α | CREA OF 1112.1 | Stemphylium vesicarium | V. faba–“A Sciabola” |
| OQ266773 | ITS | CREA OF 1112.1 | Stemphylium vesicarium | Vicia faba -A sciabola |
| OQ302538 | ITS | CREA OF 1231.2 | Alternaria infectoria | B. oleracea var. sabellica– “Torzella riccia” |
| Locus/Target | Definition | Primers | Sequence (5′-3′) | Reference | Isolates |
|---|---|---|---|---|---|
| EF-1α | Translation elongation factor 1α | EF1-728F | CATCGAGAAGTTCGAGAAGG | [28] | CREA OF 1113.1 CREA OF 1112.1 |
| EF1-968R | TACTTGAAGGAACCCTTACC | ||||
| EF1-688F | CGGTCACTTGATCTACAAGTGC | [29] | CREA OF 190.4 | ||
| EF1-1251R | CCTCGAACTCACCAGGTACCG | ||||
| ITS1 | Internal transcribing spacer 1 | ITS1 | TCCGTAGGTGAACCTGCGG | [30] | All |
| ITS4 | TCCTCCGTCTATTGATATGC | ||||
| β-tub | β-tubulin | T1 | AACATGCGTGAGATTGTAAGT | [31] | CREA OF 1128.1 |
| β-tub-2B | ACCCTCAGTGTAGTGACCCTTGGC | ||||
| Calmodulin/Macrophomina spp. | Calmodulin | MpCalF | CAATCTCTTTCTCCCCTACAGGA | [32] | CREA OF 1128.3 |
| MpCalR | ACTGCGCAAAAGCGCCAGTAAAC | ||||
| MsCalF | CAATGTCTTTCTCCACTGCAGGA | ||||
| MsCalR | TACTGCGCCAAAGCGGCAGTAAA | ||||
| MeCalF | AGCCCGCCTTGCTCCACCCTGTTCT | ||||
| MeCalR | TGTATACTGCGCAAAAGCGGCAGT | ||||
| EF-1α | Translation elongation factor 1α | MpTefF | AAACACACTTTTCGCACTCCTGC- | ||
| MpTefR | TATGCTCGCAGAGAAGAACACGA | ||||
| MsTefF | GGCACACTTTTCGCGCTTCTGTA | ||||
| MsTefR | TGTGCTCGCTGGGAAGAACATGA | ||||
| MeTefF | AAGCATACTTTTCGTGCTCCTGC | ||||
| MeTefR | AAAGGAACATGAGTGGCCAAAAA | ||||
| Xap/Xff | p7X4c | GGCAACACCCGATCCCTAAACAGG | [23] | CREA OF 522, 539, 540, 541, 542, 543, 544, 545, 546 | |
| p7X4e | CGCCGGAAGCACGATCCTCGAAG | ||||
| Xcc | DLH120 | CCGTAGCACTTAGTGCAATG | [23] | CREA OF 1231.20, 1231.21, 1231.22 | |
| DLH125 | GCATTTCCATCGGTCACGATTG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dell’Olmo, E.; Zaccardelli, M.; Onofaro Sanaja, V.; Basile, B.; Sigillo, L. Surveillance of Landraces’ Seed Health in South Italy and New Evidence on Crop Diseases. Plants 2023, 12, 812. https://doi.org/10.3390/plants12040812
Dell’Olmo E, Zaccardelli M, Onofaro Sanaja V, Basile B, Sigillo L. Surveillance of Landraces’ Seed Health in South Italy and New Evidence on Crop Diseases. Plants. 2023; 12(4):812. https://doi.org/10.3390/plants12040812
Chicago/Turabian StyleDell’Olmo, Eliana, Massimo Zaccardelli, Vincenzo Onofaro Sanaja, Boris Basile, and Loredana Sigillo. 2023. "Surveillance of Landraces’ Seed Health in South Italy and New Evidence on Crop Diseases" Plants 12, no. 4: 812. https://doi.org/10.3390/plants12040812









