In Vitro Morphogenesis of Tobacco: Modulation of Endogenous Growth Regulators by Tulsi (Holy Basil)
Abstract
1. Introduction
2. Results
2.1. Effects of Tulsi Leaf Extracts on Organogenesis
2.2. Differences in the Mode of Regeneration of N. tabacum Explants
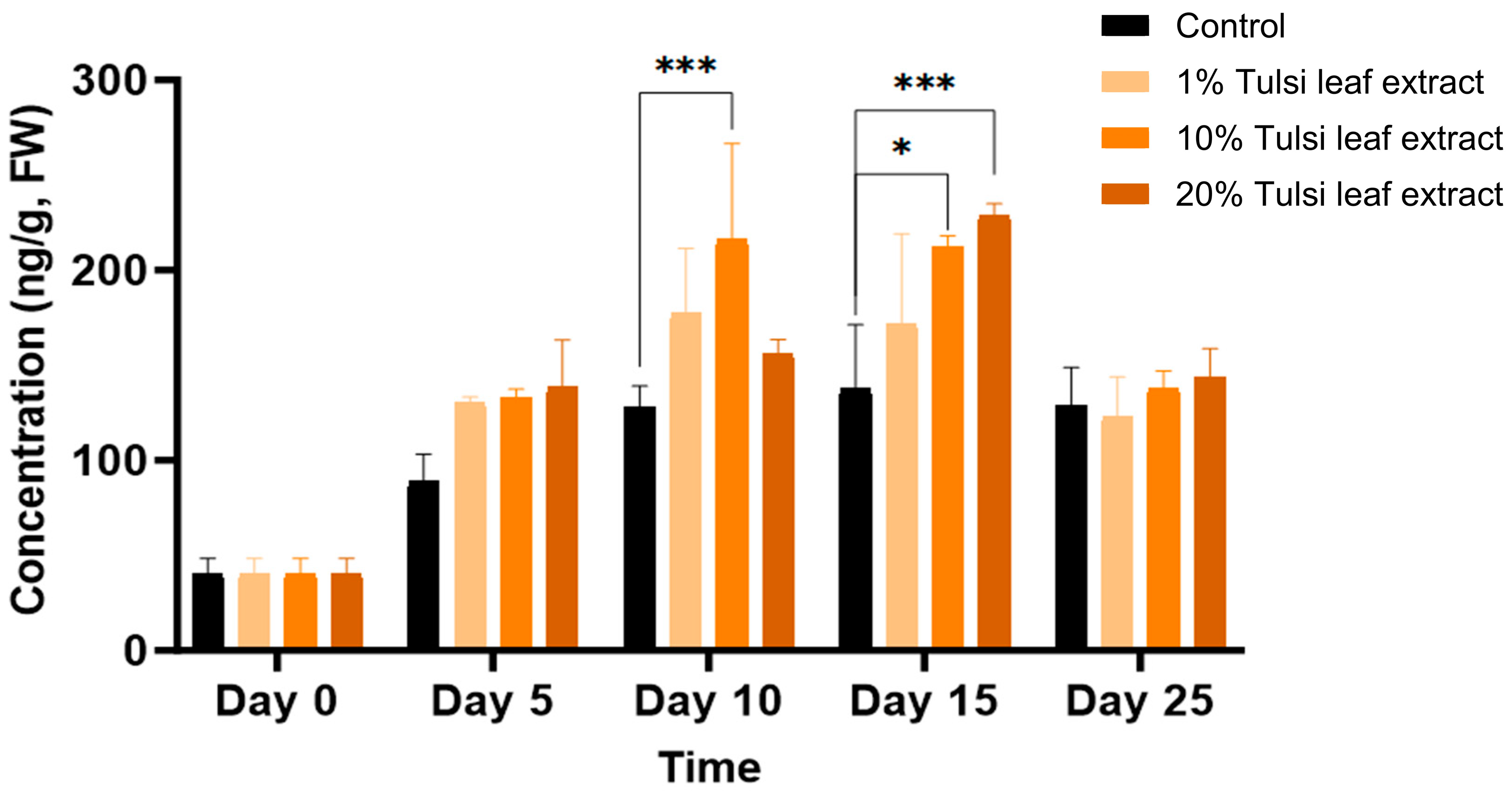
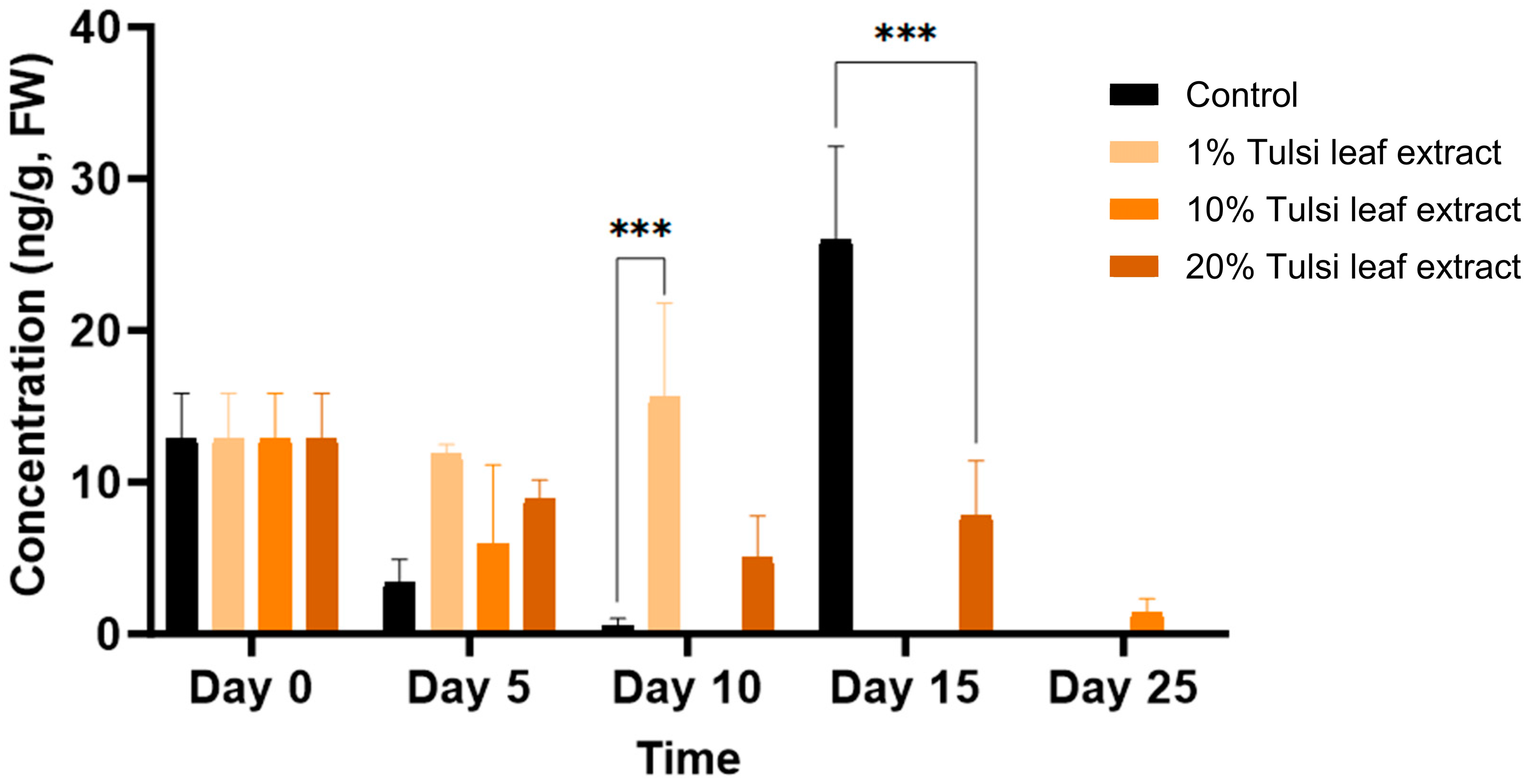
2.3. Effect of the Extracts on PGRs
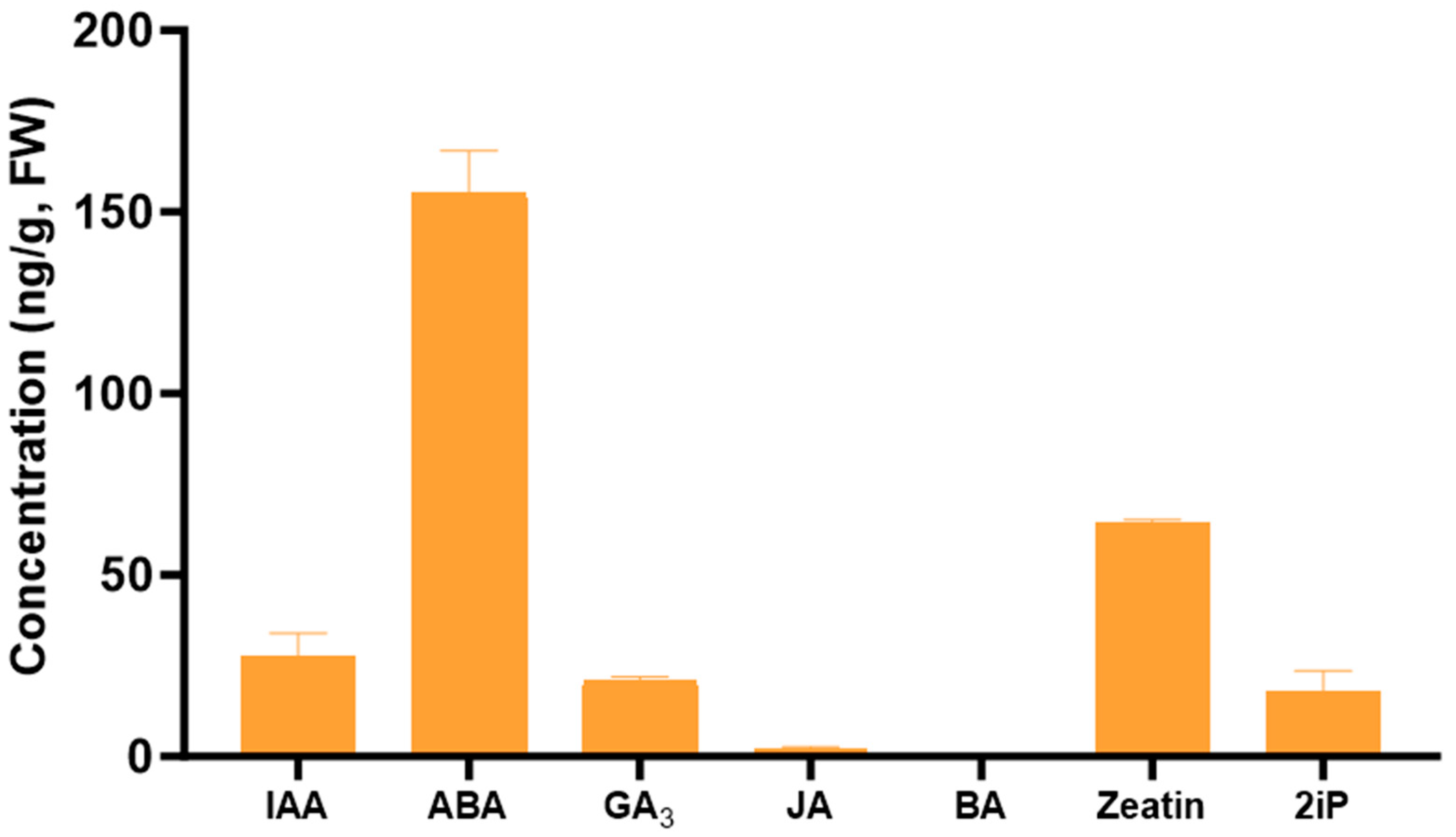
2.3.1. Endogenous Levels of PGRs in Tulsi Leaf Extracts
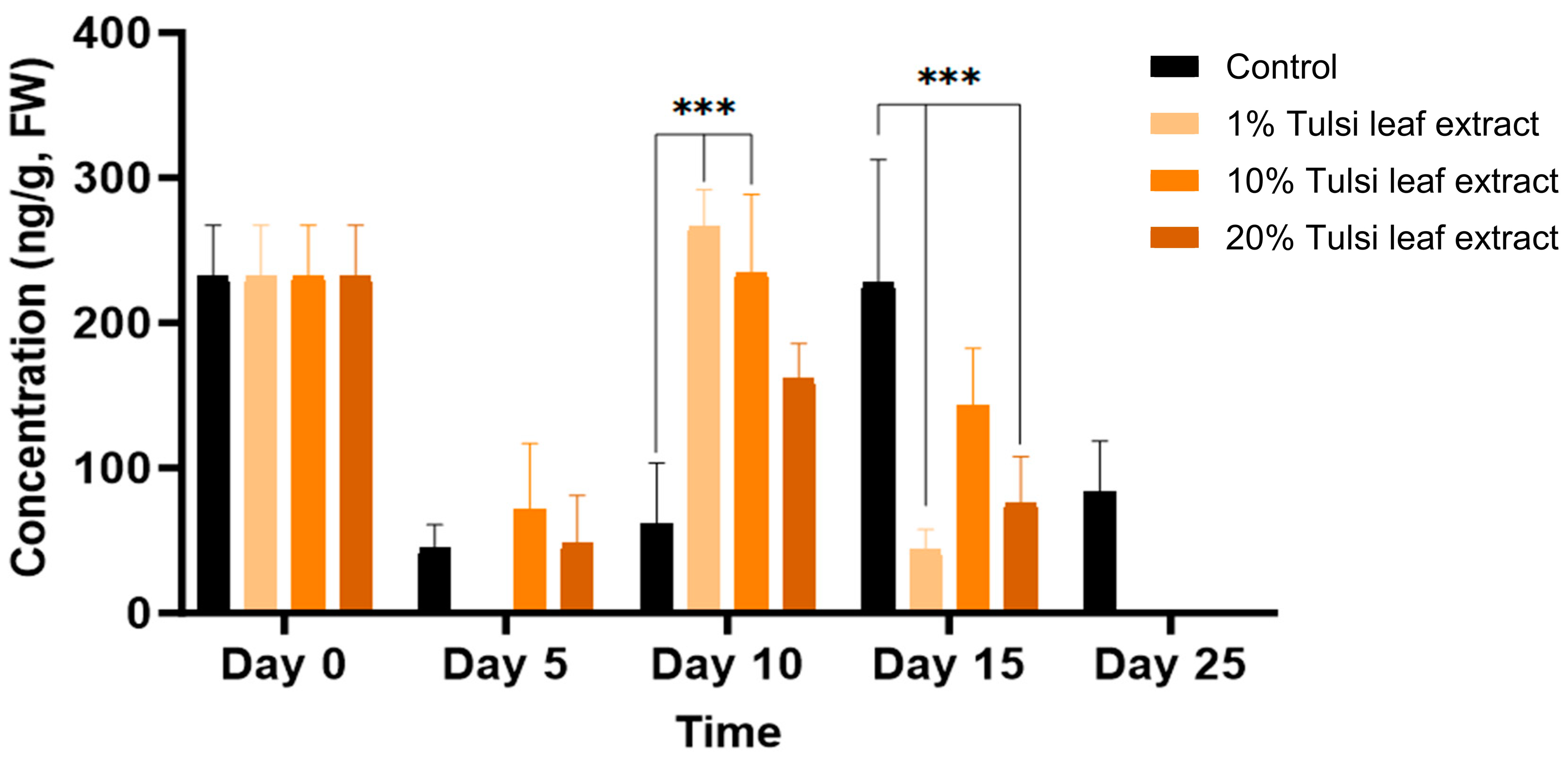
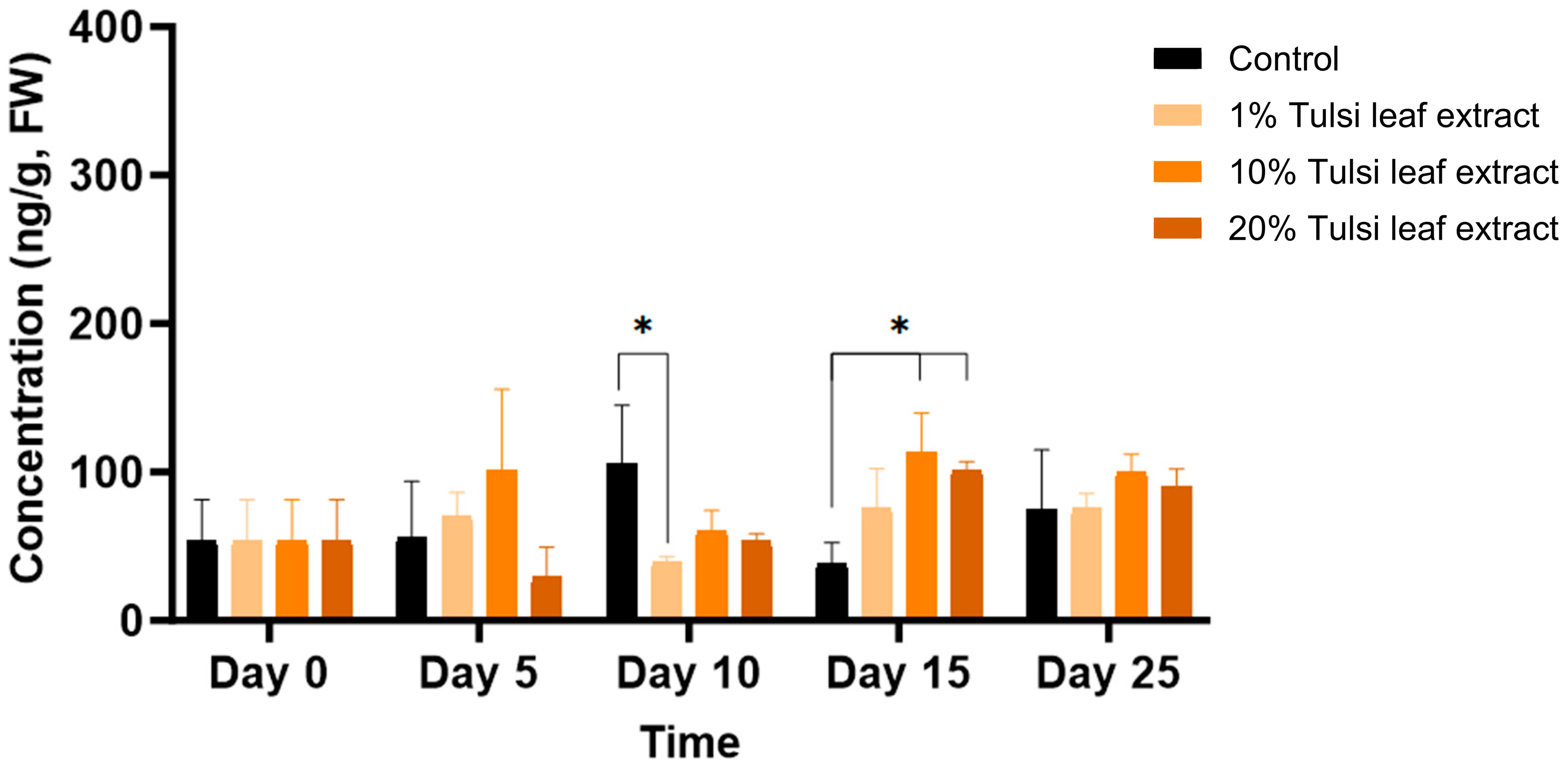
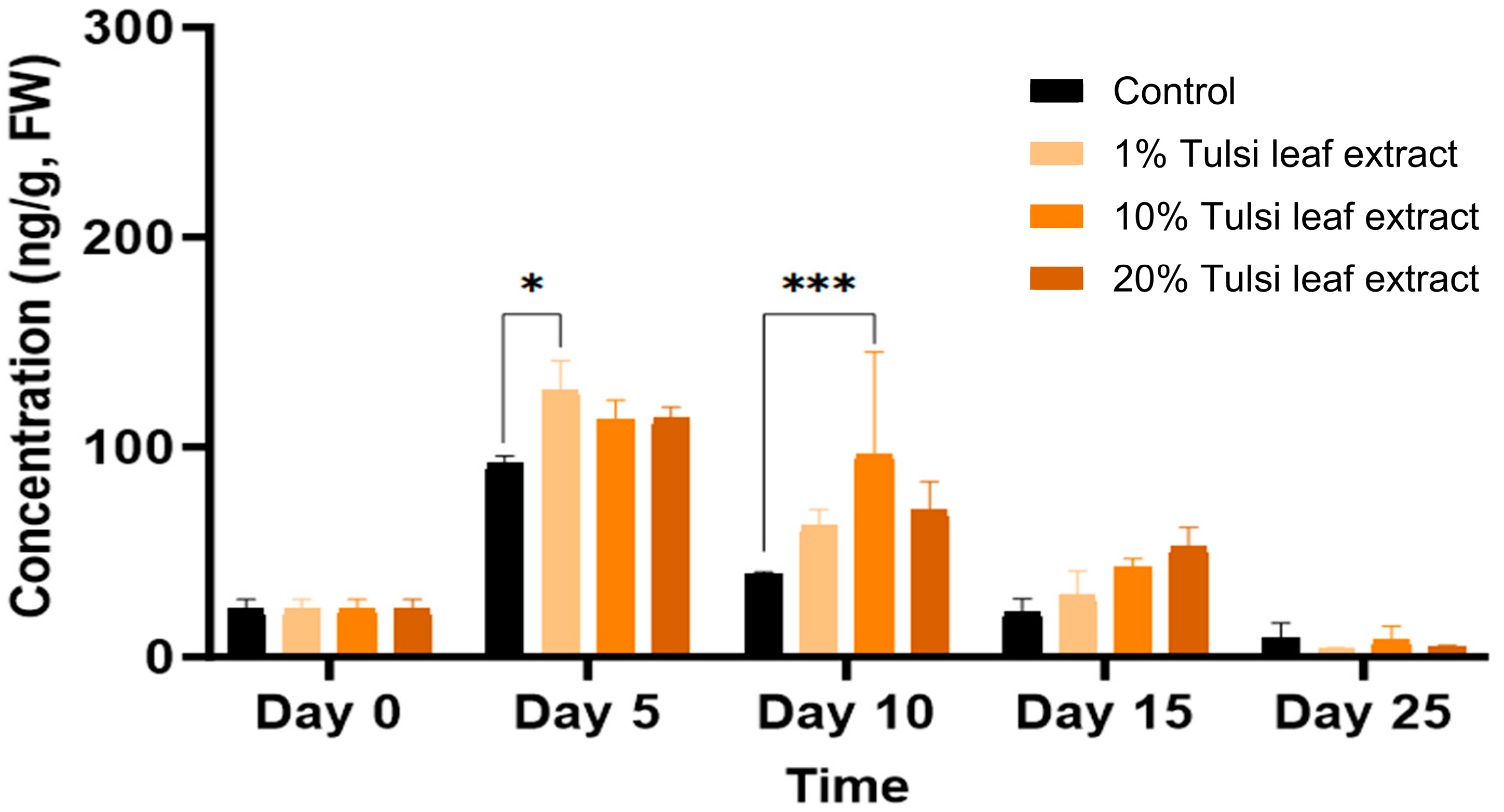
2.3.2. Interplay among PGRs
Indole-3-Acetic Acid (IAA)
Abscisic Acid (ABA)
Gibberellic Acid (GA3)
6-Benzylaminopurine (BA)
Zeatin
6-(γ, γ-Dimethylallylamino)Purine (2iP)
Jasmonic Acid (JA)
2.3.3. Interaction between Zeatin and Abscisic Acid in Tobacco Leaf Explants
3. Discussion
4. Materials and Methods
4.1. Growth of Plant Materials and Preparation of Extracts
4.2. Nodal Culture Establishment of Tobacco Explants
Tobacco Leaf Disc Explants Culture and Sample Collection
4.3. Detection and Quantification of Hormones
4.3.1. Sample Collection and Extraction
4.3.2. Separation and Quantification
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Asghar, S.; Ghori, N.; Hyat, F.; Li, Y.; Chen, C. Use of Auxin and Cytokinin for Somatic Embryogenesis in Plant: A Story from Competence towards Completion. Plant Growth Regul. 2023, 99, 413–428. [Google Scholar] [CrossRef]
- Chen, B.; Fiers, M.; Dekkers, B.J.W.; Maas, L.; Van Esse, G.W.; Angenent, G.C.; Zhao, Y.; Boutilier, K. ABA Signalling Promotes Cell Totipotency in the Shoot Apex of Germinating Embryos. J. Exp. Bot. 2021, 72, 6418–6436. [Google Scholar] [CrossRef]
- Gaspar, T.; Keveks, C.; Penel, C.; Greppin, H.; Reid, D.M.; Thorpe, T.A. Plant Hormones and Plant Growth Regulators in Plant Tissue Culture. In Vitro Cell. Dev. Biol.-Plant 1996, 32, 272–289. [Google Scholar] [CrossRef]
- Hangarter, R.P.; Peterson, M.D.; Good, N.E. Biological Activities of Indoleacetylamino Acids and Their Use as Auxins in Tissue Culture. Plant Physiol. 1980, 65, 761–767. [Google Scholar] [CrossRef]
- Wójcik, A.M.; Wójcikowska, B.; Gaj, M.D. Current Perspectives on the Auxin-Mediated Genetic Network That Controls the Induction of Somatic Embryogenesis in Plants. Int. J. Mol. Sci. 2020, 21, 1333. [Google Scholar] [CrossRef] [PubMed]
- Magyar-Tábori, K.; Dobránszki, J.; da Silva, J.A.T.; Bulley, S.M.; Hudák, I. The Role of Cytokinins in Shoot Organogenesis in Apple. Plant Cell Tiss Organ Cult. 2010, 101, 251–267. [Google Scholar] [CrossRef]
- D’Angeli, S.; Lauri, P.; Dewitte, W.; Van Onckelen, H.; Caboni, E. Factors Affecting In Vitro Shoot Formation from Vegetative Shoot Apices of Apple and Relationship between Organogenic Response and Cytokinin Localisation. Plant Biosyst. 2001, 135, 95–100. [Google Scholar] [CrossRef]
- McGaw, B.A.; Burch, L.R. Cytokinin Biosynthesis and Metabolism. In Plant Hormones: Physiology, Biochemistry and Molecular Biology; Davies, P.J., Ed.; Springer: Dordrecht, The Netherlands, 1995; pp. 98–117. [Google Scholar] [CrossRef]
- Zhu, Y.; Ouyang, W.; Li, Y.; Chen, Z. The Effects of 2ip and 2, 4-D on Rice Calli Differentiation. Plant Growth Regul. 1996, 19, 19–24. [Google Scholar]
- Zhu, Y.; Shari, X.; Chen, Z. Elevated Endogenous Isopentenyl Adenine Content Is Correlated with an Extremely Shooty Rice Phenotype. Plant Growth Regul. 1995, 17, 1–5. [Google Scholar]
- Haberer, G.; Kieber, J.J. Cytokinins. New Insights into a Classic Phytohormone. Plant Physiol. 2002, 128, 354–362. [Google Scholar] [CrossRef]
- Skoog, F.; Miller, C.O. Chemical Regulation of Growth and Organ Formation in Plant Tissues Cultured In Vitro. Symp. Soc. Exp. Biol. 1957, 11, 118–130. [Google Scholar] [PubMed]
- Gupta, R.; Chakrabarty, S.K. Gibberellic Acid in Plant. Plant Signal. Behav. 2013, 8, e25504. [Google Scholar] [CrossRef] [PubMed]
- Nolan, K.E.; Song, Y.; Liao, S.; Saeed, N.A.; Zhang, X.; Rose, R.J. An Unusual Abscisic Acid and Gibberellic Acid Synergism Increases Somatic Embryogenesis, Facilitates Its Genetic Analysis and Improves Transformation in Medicago truncatula. PLoS ONE 2014, 9, e99908. [Google Scholar] [CrossRef][Green Version]
- Rai, M.K.; Shekhawat, N.S.; Harish; Gupta, A.K.; Phulwaria, M.; Ram, K.; Jaiswal, U. The Role of Abscisic Acid in Plant Tissue Culture: A Review of Recent Progress. Plant Cell Tissue Organ Cult. 2011, 106, 179–190. [Google Scholar] [CrossRef]
- Kikuchi, A.; Sanuki, N.; Higashi, K.; Koshiba, T.; Kamada, H. Abscisic Acid and Stress Treatment Are Essential for the Acquisition of Embryogenic Competence by Carrot Somatic Cells. Planta 2006, 223, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Liu, W.; Gu, Z.; Wu, S.; Yilan, E.; Zhou, W.; Lin, J. Roles of the Wound Hormone Jasmonate in Plant Regeneration. J. Exp. Bot. 2023, 74, 1198–1206. [Google Scholar] [CrossRef]
- Han, G. Evolution of Jasmonate Biosynthesis and Signaling Mechanisms. J. Exp. Bot. 2017, 68, 1323–1331. [Google Scholar] [CrossRef] [PubMed]
- Molnár, Z.; Virág, E.; Ördög, V. Natural Substances in Tissue Culture Media of Higher Plants. Acta Biol. Szeged. 2011, 55, 123–127. [Google Scholar]
- Gajdošová, S.; Spíchal, L.; Kamínek, M.; Hoyerová, K.; Novák, O.; Dobrev, P.I.; Galuszka, P.; Klíma, P.; Gaudinová, A.; Žižková, E.; et al. Distribution, Biological Activities, Metabolism, and the Conceivable Function of Cis-Zeatin-Type Cytokinins in Plants. J. Exp. Bot. 2011, 62, 2827–2840. [Google Scholar] [CrossRef]
- Crouch, I.; van Staden, J. Evidence for the Presence of Plant Growth Regulators in Commercial Seaweed Products. Plant Growth Regul. 1993, 31, 21–29. [Google Scholar]
- Swamy, M.K.; Mohanty, S.K.; Anuradha, M. The Effect of Plant Growth Regulators and Natural Supplements on In Vitro Propagation of Pogostemon cablin Benth. J. Crop Sci. Biotechnol. 2014, 17, 71–78. [Google Scholar]
- Prando, M.A.S.; Chiavazza, P.; Faggio, A.; Contessa, C. Effect of Coconut Water and Growth Regulator Supplements on In Vitro Propagation of Corylus avellana L. Sci. Hortic. 2014, 171, 91–94. [Google Scholar] [CrossRef]
- Yadav, A.; Singh, S.; Yadav, V. Screening Herbal Extracts as Biostimulant to Increase Germination, Plant Growth and Secondary Metabolite Production in Wheatgrass. Sci. Rep. 2024, 14, 607. [Google Scholar] [CrossRef] [PubMed]
- Vongnhay, V.; Shukla, M.R.; Ayyanath, M.M.; Sriskantharajah, K.; Saxena, P.K. Enhanced In Vitro Plant Morphogenesis of Tobacco: Unveiling Indoleamine-Modulated Adaptogenic Properties of Tulsi (Ocimum sanctum L.). Plants 2024, 13, 1370. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.M. Tulsi—Ocimum sanctum: A Herb for All Reasons. J. Ayurveda Integr. Med. 2014, 5, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Bohn-Courseau, I. Auxin: A Major Regulator of Organogenesis L’ Auxine: Un régulateur majeur de l’organogenèse. Comptes Rendus-Biol. 2010, 333, 290–296. [Google Scholar] [CrossRef]
- Cassells, A.C.; Long, R.D.; Mousdsiie, D.M.A. Endogenous IAA and Morphogenesis in Tobacco Petiole Cultures. Physiol. Plant. 1982, 56, 507–512. [Google Scholar] [CrossRef]
- Seldimirova, O.A.; Kudoyarova, G.R.; Kruglova, N.N. Changes in Distribution of Zeatin and Indole-3-Acetic Acid in Cells during Callus Induction and Organogenesis In Vitro in Immature Embryo Culture of Wheat. In Vitro Cell. Dev. Biol.-Plant 2016, 52, 251–264. [Google Scholar] [CrossRef]
- Scarpella, E. Pattern Formation in the Vascular System of Monocot and Dicot Plant Species. New Phytol. 2004, 164, 209–242. [Google Scholar] [CrossRef]
- Guo, H.; Kang, X.; Yuan, M.; Wu, R.; Du, L. Relationship between Somatic Embryogenesis and Endogenous Hormones of Cinnamomum camphora L. Plant Cell Tiss Organ Cult. 2024, 156, 53. [Google Scholar]
- Shi, J.; Chen, J. Desiccation Treatment and Endogenous IAA Levels Are Key Factors Influencing High Frequency Somatic Embryogenesis in Cunninghamia lanceolata (Lamb.) Hook. Front. Plant Sci. 2017, 8, 2054. [Google Scholar] [CrossRef]
- Ben Ali, N.; Benkaddour, R.; Rahmouni, S.; Boussaoudi, I.; Hamdoun, O.; Hassoun, M.; Azaroual, L.; Badoc, A.; Lamarti, A.; Ben, N.; et al. Secondary Somatic Embryogenesis in Cork Oak: Influence of Plant Growth Regulators Regulators. Forest Sci. Technol. 2023, 19, 78–88. [Google Scholar] [CrossRef]
- Kamada, H.; Harada, H. Changes in the Endogenous Level and Effects of Abscisic Acid during Somatic Embryogenesis of Daucus carota L. Plant Cell Physiol. 1981, 22, 1423–1429. [Google Scholar] [CrossRef]
- Acanda, Y.; Martínez, Ó.; Prado, M.J.; González, M.V.; Rey, M. Changes in Abscisic Acid Metabolism in Relation to the Maturation of Grapevine (Vitis vinifera L., Cv. Mencía) Somatic Embryos. BMC Plant Biol. 2020, 20, 487. [Google Scholar] [CrossRef] [PubMed]
- Kiyosue, T.; Nakayama, J.; Satoh, S.; Isogai, A.; Suzuki, A.; Kamada, H.; Harada, H. Partial amino-acid sequence of ECP31, a carrot embryogenic-cell protein, and enhancement of its accumulation by abscisic acid in somatic embryos. Planta. 1992, 186, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Karami, O.; Saidi, A. The Molecular Basis for Stress-Induced Acquisition of Somatic Embryogenesis. Mol. Biol. Rep. 2010, 37, 2493–2507. [Google Scholar] [CrossRef] [PubMed]
- Ribnicky, D.M.; Ilić, N.; Cohen, J.D.; Cooke, T.J. The Effects of Exogenous Auxins on Endogenous Indole-3-Acetic Acid Metabolism: The Implications for Carrot Somatic Embryogenesis. Plant Physiol. 1996, 112, 549–558. [Google Scholar] [CrossRef]
- Ochatt, S.J.; Revilla, M.A. From Stress to Embryos: Some of the Problems for Induction and Maturation of Somatic Embryos. Methods Mol. Biol. 2016, 1359, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Braybrook, S.A.; Harada, J.J. LECs Go Crazy in Embryo Development. Trends Plant Sci. 2008, 13, 624–630. [Google Scholar] [CrossRef]
- Asthana, P.; Rai, M.K.; Jaiswal, U.; Kinetin, K. 6-Benzylaminopurine Mediated Indirect Organogenesis in Sapindus trifoliatus L. through Internodal Segments. Vegetos 2024. [Google Scholar] [CrossRef]
- Bhanupriya, C.; Kar, S. Callus-Mediated Organogenesis and Regeneration of Sorghum bicolor under the Influence of Natural and Synthetic Growth Regulators. In Vitro Cell. Dev. Biol.-Plant 2024, 60, 355–364. [Google Scholar] [CrossRef]
- Ramage, C.M.; Williams, R.R. Cytokinin-Induced Abnormal Shoot Organogenesis Is Associated with Elevated Knotted1-Type Homeobox Gene Expression in Tobacco. Plant Cell Rep. 2004, 22, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Tran Thanh Van, M.; Dien, N.T.; Chlyah, A. Regulation of Organogenesis in Small Explants of Superficial Tissue of Nicotiana tabacum L. Planta 1974, 119, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Avilez-Montalvo, J.R.; Quintana-Escobar, A.O.; Hugo, A.M.; Brito-Arg, L.; Galaz-Ávalos, R.M.; Uc-Chuc, M.A.; Loyola-Vargas, V.M. Auxin-Cytokinin cross Talk in Somatic Embryogenesis of Coffea canephora. Plants 2022, 11, 2013. [Google Scholar] [CrossRef] [PubMed]
- Phillips, G.C.; Garda, M. Plant Tissue Culture Media and Practices: An Overview. In Vitro Cell. Dev. Biol.-Plant 2019, 55, 242–257. [Google Scholar] [CrossRef]
- Khan, M.I.; Ahmad, N.; Anis, M. The Role of Cytokinins on In Vitro Shoot Production in Salix tetrasperma Roxb.: A Tree of Ecological Importance. Trees-Struct. Funct. 2011, 25, 577–584. [Google Scholar] [CrossRef]
- Goebel-Tourand, I.; Mauro, M.C.; Sossountzov, L.; Miginiac, E.; Deloire, A. Arrest of Somatic Embryo Development in Grapevine: Histological Characterization and the Effect of ABA, BAP and Zeatin in Stimulating Plantlet Development. Plant Cell. Tissue Organ Cult. 1993, 33, 91–103. [Google Scholar] [CrossRef]
- Wang, Y.; Pang, D.; Ruan, L.; Liang, J.; Zhang, Q.; Qian, Y.; Zhang, Y.; Bai, P.; Wu, L.; Cheng, H.; et al. Integrated Transcriptome and Hormonal Analysis of Naphthalene Acetic Acid-Induced Adventitious Root Formation of Tea Cuttings (Camellia sinensis). BMC Plant Biol. 2022, 22, 319. [Google Scholar] [CrossRef]
- Cao, X.; Hammerschlag, F.A. Improved Shoot Organogenesis from Leaf Explants of Highbush Blueberry. HortScience 2000, 35, 945–947. [Google Scholar] [CrossRef]
- Zhao, X.M.; Lian, Y.J.; Jin, Z.L.; Zhang, X.J.; Yan, Y.; Fan, S.J. Shoot Organogenesis and Somatic Embryogenesis in Leaf Tissue of Pulsatilla Tongkangensis Y.N. Lee & T.C. Lee. Plant Biotechnol. Rep. 2022, 16, 389–400. [Google Scholar] [CrossRef]
- Chandler, C.K.; Draper, A.D. Effect of Zeatin and 2iP on Shoot Proliferation of Three Highbush Blueberry Clones In Vitro. HortScience 1986, 21, 1065–1066. [Google Scholar]
- Eccher, T.; Noe, N. Comparison Between 2iP and Zeatin in the Micropropagation of Highbush Blueberry (Vaccinium corymbosum). Acta Hortic. 1989, 241, 185–190. [Google Scholar]
- Ostrolucká, M.G.; Libiaková, G.; Ondrußková, E.; Gajdoßová, A. In Vitro Propagation of Vaccinium Species. Acta Univ. Latv. 2004, 676, 207–676. [Google Scholar]
- Murvanidze, N.; Nisler, J.; Leroux, O.; Werbrouck, S.P.O. Cytokinin Oxidase/Dehydrogenase Inhibitors Stimulate 2iP to Induce Direct Somatic Embryogenesis in Coffea arabica. Plant Growth Regul. 2021, 94, 195–200. [Google Scholar] [CrossRef]
- Hatanaka, T.; Arakawa, O.; Yasuda, T.; Uchida, N.; Yamaguchi, T. Effect of plant growth regulators on somatic embryogenesis in leaf cultures of Coffea canephora. Plant Cell Rep. 1991, 10, 179–182. [Google Scholar] [PubMed]
- Kahia, J.; Mantell, S. High-Frequency Direct Somatic Embryogenesis and Plantlet Regeneration High-Frequency Direct Somatic Embryogenesis and Plantlet Regeneration from Leaves Derived from In Vitro-Germinated Seedlings of a Coffea arabica Hybrid Cultivar. HortScience 2016, 51, 1148–1152. [Google Scholar] [CrossRef]
- Alves, S.; Cristina, V.; Carmazini, B.; Damião, C. 2-Isopentenyladenine in the Induction of Direct Somatic Embryogenesis Capacity of Coffea arabica L. Ciência Rural 2018, 48, e20180001. [Google Scholar]
- Khan, T.; Singh, A.K. Regeneration via Somatic Embryogenesis and Organogenesis in Different Cultivars of Cotton (Gossypium spp.). In Vitro Cell. Dev. Biol.-Plant 2006, 42, 498–501. [Google Scholar] [CrossRef]
- Xie, C.H.; Huang, W.; Li, B. Effects of Benzylaminopurine, 6-(δ, δ-Dimethylallyamino)-purine and Thidiazuron on Shoot Organogenesis of Nicotiana Species. Tob. Sci. 2004, 46, 17–23. [Google Scholar]
- Hassan, M.M.; Allam, M.A.; El Din, I.M.S.; Malhat, M.H.; Taha, R.A. High-Frequency Direct Somatic Embryogenesis and Plantlet Regeneration from Date Palm Immature Inflorescences Using Picloram. J. Genet. Eng. Biotechnol. 2021, 19, 33. [Google Scholar]
- Lehminger-mertens, R.; Jacobsen, H.J. Protoplast Regeneration and Organogenesis from Pea Protoplasts. In Vitro Cell. Dev. Biol. 1989, 25, 571–574. [Google Scholar] [CrossRef]
- Nabi, N.; Singh, S.; Saffeullah, P. Responses of In Vitro Cell Cultures to Elicitation: Regulatory Role of Jasmonic Acid and Methyl Jasmonate: A Review. In Vitro Cell. Dev. Biol.-Plant 2021, 57, 341–355. [Google Scholar] [CrossRef]
- Koo, A.J.K.; Gao, X.; Jones, A.D.; Howe, G.A.; Lansing, E.; Lansing, E.; Lansing, E. A Rapid Wound Signal Activates the Systemic Synthesis of Bioactive Jasmonates in Arabidopsis. Plant J. 2009, 59, 974–986. [Google Scholar] [CrossRef] [PubMed]
- Park, O.; Bae, S.H.; Kim, S.; Seo, P.J. Novo Shoot Regeneration in Arabidopsis. Plant Signal. Behav. 2019, 14, 1–3. [Google Scholar] [CrossRef]
- Ammirato, P.V. Hormonal Control of Somatic Embryo Development from Cultured Cells of Caraway. Plant Physiol. 1977, 59, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Shukla, M.R.; Kibler, A.; Turi, C.E.; Erland, L.A.E.; Alan Sullivan, J.; Murch, S.J.; Saxena, P.K. Selection and Micropropagation of an Elite Melatonin Rich Tulsi (Ocimum sanctum L.) Germplasm Line. Agronomy 2021, 11, 207. [Google Scholar] [CrossRef]
- Ayyanath, M.M.; Shukla, M.R.; Saxena, P.K. Indoleamines Impart Abiotic Stress Tolerance and Improve Reproductive Traits in Hazelnuts. Plants 2023, 12, 1233. [Google Scholar] [CrossRef]
- Yalçın, S.; Şükran Okudan, E.; Karakaş, Ö.; Önem, A.N.; Sözgen Başkan, K. Identification and Quantification of Some Phytohormones in Seaweeds Using UPLC-MS/MS. J. Liq. Chromatogr. Relat. Technol. 2019, 42, 475–484. [Google Scholar] [CrossRef]




| Treatment | Average Number of Regenerants/Explant | Average Number of Shoots Greater than 1 cm/Explant | Average Number of Embryo-Like Structures |
|---|---|---|---|
| Control | 6.14 ± 0.5 c | 6.14 ± 0.5 c | 0 c |
| 1% Tulsi leaf extract | 9.60 ± 0.5 b | 8.75 ± 0.5 b | 0.98 ± 0.5 cb |
| 10% Tulsi leaf extract | 12.58 ± 0.5 a | 10.58 ± 0.5 a | 2.50 ± 0.5 a |
| 20% Tulsi leaf extract | 13.07 ± 0.5 a | 12.05 ± 0.5 a | 1.53 ± 0.5 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vongnhay, V.; Shukla, M.R.; Ayyanath, M.-M.; Sriskantharajah, K.; Saxena, P.K. In Vitro Morphogenesis of Tobacco: Modulation of Endogenous Growth Regulators by Tulsi (Holy Basil). Plants 2024, 13, 2002. https://doi.org/10.3390/plants13142002
Vongnhay V, Shukla MR, Ayyanath M-M, Sriskantharajah K, Saxena PK. In Vitro Morphogenesis of Tobacco: Modulation of Endogenous Growth Regulators by Tulsi (Holy Basil). Plants. 2024; 13(14):2002. https://doi.org/10.3390/plants13142002
Chicago/Turabian StyleVongnhay, Vanessa, Mukund R. Shukla, Murali-Mohan Ayyanath, Karthika Sriskantharajah, and Praveen K. Saxena. 2024. "In Vitro Morphogenesis of Tobacco: Modulation of Endogenous Growth Regulators by Tulsi (Holy Basil)" Plants 13, no. 14: 2002. https://doi.org/10.3390/plants13142002
APA StyleVongnhay, V., Shukla, M. R., Ayyanath, M.-M., Sriskantharajah, K., & Saxena, P. K. (2024). In Vitro Morphogenesis of Tobacco: Modulation of Endogenous Growth Regulators by Tulsi (Holy Basil). Plants, 13(14), 2002. https://doi.org/10.3390/plants13142002






