Abstract
During 2021 and 2022, eight field-collected and five laboratory Helicoverpa zea strains with varying susceptibility to different Bt proteins were evaluated for their responses against HearNPV using diet-overlay bioassays. The five laboratory strains included SS (susceptible to all Bt proteins), CRY-RR (resistant to Cry1 and Cry2), VIP-RR-70 (resistant to Vip3Aa), VIP-RR-15 (resistant to Vip3Aa), and TRE-RR (resistant to Cry1, Cry2, and Vip3Aa). Our findings showed that the susceptibility of TRE-RR, VIP-RR-70, and VIP-RR-15 strains to HearNPV was similar to that of the SS strain. However, the field and Cry-RR strains were more resistant to HearNPV compared to the SS strain. Because most feral H. zea strains in the southern U.S. have developed practical resistance to Cry Bt proteins but remain susceptible to Vip3Aa, the results suggest that the reduced susceptibility to HearNPV in H. zea may be associated with the resistance to Cry Bt proteins but not with the resistance to Vip3Aa. Correlation analysis confirmed that there was a significant positive relationship between Cry resistance and HearNPV resistance, but not between the Vip3Aa resistance and HearNPV resistance in H. zea. Our findings provide valuable insights into the relationship between susceptibility to HearNPV and resistance to Bt proteins in H. zea.
1. Introduction
The cotton bollworm (Helicoverpa zea Boddie) is a polyphagous insect pest that can attack many agricultural crops, including corn (Zea mays L.), sorghum (Sorghum bicolor (L.) Moench), cotton (Gossypium spp.), soybeans (Glycine max (L.) Merr.), tomatoes (Solanum lycopersicum L.), hemp (Cannabis sativa L.), cabbage (Brassica oleracea L.), cowpeas (Vigna unguiculata (L.) Walp), cucumber (Cucumis sativus L.), and eggplant (Solanum melongena L.) [1,2,3,4,5]. Helicoverpa zea is primarily controlled using conventional insecticides and Bt plant-incorporated protectants [6,7]. However, this pest is notorious for developing resistance to insecticides and has developed resistance to various classes of insecticides, such as organochlorines (IRAC MoA group 2A), organophosphates (IRAC MoA group 1B), and pyrethroids (IRAC MoA group 3A). In recent years, H. zea has also developed resistance to Cry1 and Cry2 Bt proteins (IRAC MoA group 11A) [6,8,9,10,11,12]. The development of resistance to insecticides and Bt proteins by H. zea poses a significant challenge for pest management.
Effective management of H. zea populations in the field can be achieved through the implementation of integrated pest management (IPM) approaches by mitigating the already established resistance and delaying the onset of resistance to new insecticide classes [13,14]. Helicoverpa armigera nucleopolyhedrovirus (HearNPV) is a type of entomopathogenic virus belonging to the family Bacculoviridae [15]. It selectively targets lepidopteran insects in the subfamily Heliothinae, including H. zea [16]. HearNPV-based insecticides are host-specific and selective with a novel mode of action (IRAC MoA group 31) [15,17]. Based on these characteristics, incorporating HearNPV into an IPM program targeting H. zea may be a highly effective strategy. In fact, HearNPV has already been adopted to a limited extent for managing H. zea in soybeans, grain sorghum, and hemp in the U.S., and researchers have investigated its potential to manage H. zea in cotton as well [18,19,20,21].
HearNPV can be applied as a foliar spray like conventional insecticides and is activated upon larval ingestion. The virus infects the host with two phases, namely primary and secondary infection. During the primary phase, the larvae ingest the virus occlusion bodies (OBs) that protect the virus from environmental conditions. These OBs then travel through the digestive tract of the larvae and reach the midgut. Once in the insect midgut, the alkaline content of the midgut activates the virus by degrading the occlusion bodies, causing the release of occlusion-derived virions. These virions then cross the peritrophic membrane, infect the midgut cells, and replicate. In the second phase of the infection, the replicated virus egresses as a budded virus and spreads throughout the host’s muscle, fat body, hemocytes, and tracheal cells, causing the host to liquefy [15,22,23]. When the host larva dies and liquefies, millions of viral particles are released into the environment, which can result in horizontal and abiotic transmissions [24].
Resistance to baculovirus in insects has been reported in several studies. For instance, H. zea and the African cotton leafworm (Spodoptera littoralis Boisduval) have shown resistance to Autographa californica multiple nucleopolyhedrovirus (AcMNPV) infections. These insects developed resistance to the virus by melanizing and encapsulating the tracheal epidermis, a mechanism that prevents secondary infection from occurring [25]. In Germany, there have been several reports of field resistance of the codling moth (Cydia pomonella L.) to Cydia pomonella Granulovirus (CpGV). In resistant C. pomonella, the virus can cross the peritrophic membrane, but the larva provokes a systemic blockade that prevents the viral DNA replication within the midgut cells [26,27]. Additionally, Baculovirus heliothis resistance has also been reported in Heliothis subflexa (Guenée), but the mechanism of this resistance is unknown [28].
Developing new insecticides for pest management is a challenging and costly process that is also subject to strict regulatory requirements [29]. As HearNPV-based insecticides become more prominent Lepidoptera management tools, it is imperative to establish proactive Insecticide Resistance Management (IRM) programs for the long-term use of HearNPV. Understanding the baseline susceptibility of insect pests to insecticides is essential for a resistance monitoring program [30,31]. Therefore, it is critically important to determine the baseline susceptibility and cross-resistance of H. zea to HearNPV before the widespread adoption of this new insecticide mode of action group. The objective of this study is to understand the efficacy and baseline susceptibility of several field and laboratory Bt-susceptible and resistant strains of H. zea to HearNPV.
2. Results
2.1. Dose-Response Bioassays in 2021
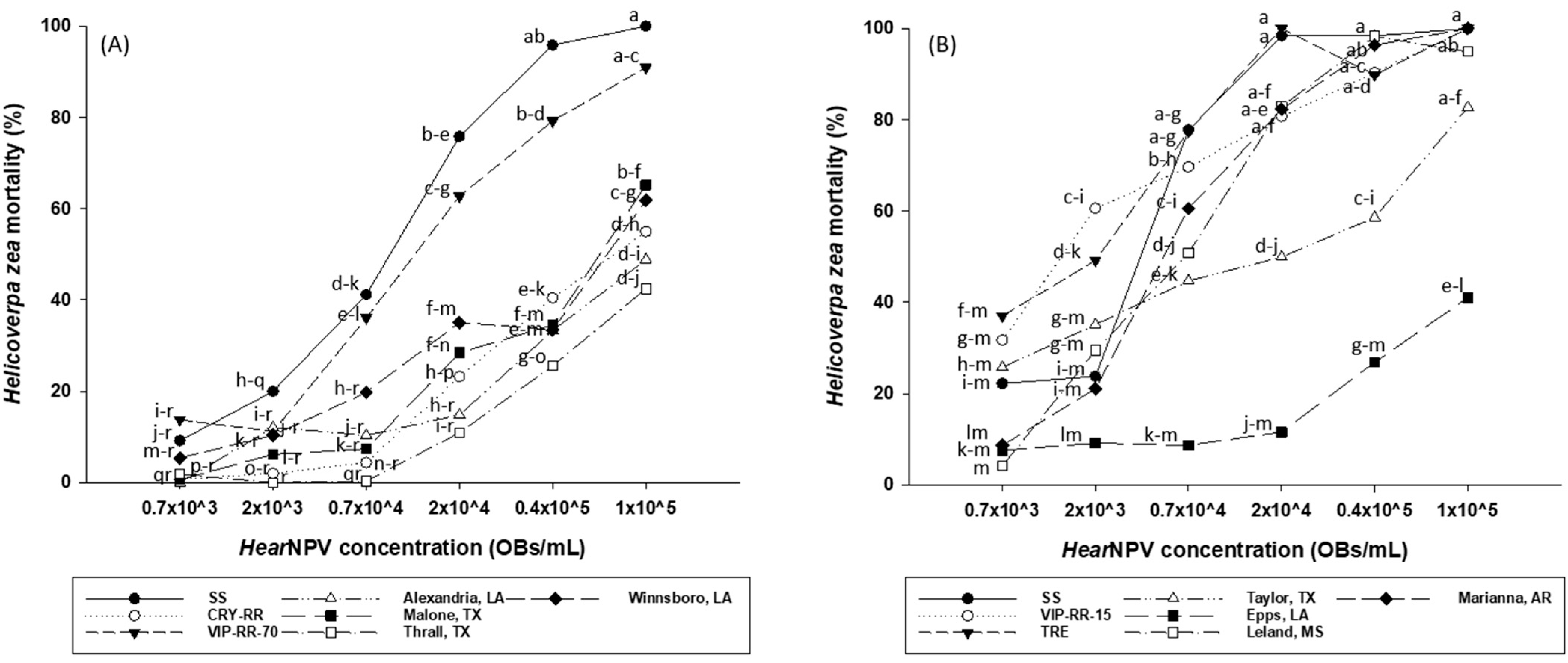
In 2021, the effects of insect strain and HearNPV concentration on larval mortality were significant (F = 52.47; df = 6, 131.3; p < 0.0001 for insect strain and F = 137.04; df = 5, 131; p < 0.0001 for HearNPV concentration). The interactions between insect strain and HearNPV concentration were also significant (F = 2.15; df = 30, 131; p = 0.0017). SS H. zea was susceptible to HearNPV with an estimated LC50 value of 6.5 × 103 OBs/mL (Table 1) and 100.0% mortality at the highest tested concentration of 1 × 105 OBs/mL (Figure 1A). VIP-RR-70 strain, which is susceptible to Cry1Ac and Cry2Ab2 (Table 2), was also susceptible to HearNPV with an estimated LC50 value of 10.5 × 103 OBs/mL and 91.0% mortality at the HearNPV concentration of 1 × 105 Obs/mL (Table 1 and Figure 1A). Based on the overlapping of 95% confidence intervals of LC50s, there were no significant differences in susceptibility against HearNPV between the SS and VIP-RR-70 strains. Relative to SS, the resistance ratio for VIP-RR-70 was 1.6-fold against HearNPV (Table 1). In contrast, the Cry-RR and all field strains that are highly resistant to Cry proteins (Table 2) exhibited significant levels of resistance against HearNPV relative to SS, based on the non-overlapping of 95% CLs (Table 1). In addition, the mortality of Cry-RR and field strains of H. zea were significantly (p < 0.05) lower than that of SS at the high HearNPV concentrations of 2 × 104–1 × 105 OBs/mL (Figure 1A). Relative to SS, the resistance ratios for Cry-RR and field strains ranged from 9.5- to 22.7-fold (Table 1).

Table 1.
Dose response of laboratory and field-collected H. zea strains against HearNPV.
Figure 1.
Mean percent mortality among Helicoverpa zea strains as affected by HearNPV concentrations in 2021 (A) and 2022 (B). Means with the same letter are not statistically different (p > 0.05). OBs/mL = occlusion bodies per milliliter.

Table 2.
Bt resistance ratio of field-collected and laboratory strains of H. zea.
2.2. Dose-Response Bioassays in 2022
In 2022, the effects of insect strain, HearNPV concentration, and the interactions between insect strain and HearNPV concentration on larval mortality were all significant (F = 48.71; df = 6, 113.4; p < 0.0001 for insect strain; F = 122.89; df = 5, 113; p < 0.0001 for HearNPV concentration; F = 4.16; df = 30, 113; p < 0.0001 for the interactions). Similarly, SS showed high susceptibility against HearNPV, with an estimated LC50 value of 2.8 × 103 OBs/mL (Table 1) and 100.0% mortality at the highest HearNPV concentration (Figure 1B). The VIP-RR-15 strain of H. zea, which is resistant to Vip3Aa Bt proteins but susceptible to Cry1Ac and Cry2Ab2 bt proteins, was also susceptible to HearNPV with an estimated LC50 value of 1.8 × 103 OBs/mL and 100.0% mortality at the HearNPV concentration of 1 × 105 OBs/mL (Table 1 and Figure 1B). Based on the overlapping of 95% confidence intervals of LC50s, there were no significant differences in susceptibility against HearNPV between the SS and VIP-RR-15 strains. Compared to SS, the resistance ratio for VIP-RR-15 was 0.7-fold against HearNPV (Table 1). By contrast, all field strains that are highly resistant to Cry proteins (Table 2), exhibited significant levels of resistance against HearNPV compared to SS based on the non-overlapping of 95% CLs (Table 1). Relative to SS, the resistance ratios for these field strains ranged from 1.8-to 175.0-fold (Table 1). The TRE-RR strain, which is resistant to Cry1Ac, Cry2Ab2, and Vip3Aa Bt proteins, was susceptible to HearNPV with an estimated LC50 value of 1.6 × 103 OBs/mL, resulting in a resistance ratio of 0.7-fold against HearNPV relative to SS (Table 1).
2.3. Correlation Analysis between HearNPV and Bt Resistance
The resistance ratio data for HearNPV and Cry1Ac, Cry2Ab2, or Vip3Aa Bt proteins in Table 1 and Table 2 were used in the correlation analysis to explore the potential relationship between HearNPV and Bt resistance in H. zea. Due to a statistical outlier (resistance ratio for Epps-LA strain) in the pair of Cry1Ac and HearNPV, two separate correlation analyses were conducted for exploring the relationship between HearNPV and Cry1Ac resistance, one using the complete data set and another with the outlier removed.
When analyzing the complete data set for HearNPV and Cry1Ac resistance in H. zea, there was no significant correlation observed between Cry1Ac and HearNPV resistance (r = 0.02, p = 0.9438). However, after removing the outlier, a highly significant positive correlation was found between HearNPV and Cry1Ac resistance (r = 0.81, p = 0.0015). Regarding Cry2Ab2 resistance, a significant positive correlation was detected between Cry2Ab2 and HearNPV resistance (r = 0.67, p = 0.0089). On the other hand, we did not observe any significant correlation between the Vip3Aa and HearNPV resistance ratios (r = −0.17, p = 0.5566).
3. Discussion
The widespread development of practical resistance to Cry Bt proteins in H. zea in the U.S. highlights the urgent need for alternative pest management strategies and novel insecticides with different modes of action [36]. To address this challenge, incorporating baculoviruses like HearNPV into Integrated Pest Management (IPM) and Insect Resistance Management (IRM) programs for H. zea is a promising approach. HearNPV offers high efficacy, specificity, and selectivity, as demonstrated by previous studies that showed its effectiveness and low variation in susceptibility across field populations and the susceptible strain of the closely related species, old world bollworm (Helicoverpa armigera Hübner) [17]. Despite its potential, the adoption of HearNPV for managing H. zea in various agricultural crops in the U.S. has been limited [18,19,20,37,38]. Expanding the use of HearNPV could significantly contribute to mitigating insecticide and Bt resistance in H. zea. In this study, we conducted the first investigation to determine the baseline susceptibility to HearNPV of multiple field and laboratory strains of H. zea, which exhibited varying susceptibility levels to different Bt proteins. Furthermore, we aimed to explore any potential relationship between HearNPV and Bt resistance in H. zea.
Based on the findings of our study, H. zea strains displaying resistance to Cry1Ac and/or Cry2Ab2 Bt proteins exhibited significant reduced susceptibility to HearNPV compared to the genetically similar SS strain based on the non-overlapping of 95% CLs. Furthermore, correlation analysis based on resistance ratios revealed positive associations between HearNPV resistance and Cry1Ac and/or Cry2Ab2 in H. zea. In contrast, H. zea strains resistant to Vip3Aa Bt protein exhibited a high level of susceptibility to HearNPV similar to the SS strain, and correlation analysis showed no significant correlation between the Vip3Aa and HearNPV resistance in H. zea. However, the TRE-RR strain, which is resistant to Cry1, Cry2, and Vip3Aa Bt proteins, remained highly susceptible to HearNPV. Although the underlying reason for this high susceptibility is unclear, it provides further evidence of the value of HearNPV as a new resistance management tool. In a similar study, Cry2Ab/Vip3A-resistant strains of H. armigera and the Australian bollworm (Helicoverpa punctigera Wallengren) exhibited LC50 values of HearNPV comparable to their respective reference-susceptible strains [39].
Due to the failures of Cry1 and Cry2 Bt proteins and the growing use of Vip3Aa expressing corn and cotton, there is now increased selection pressure for Vip3Aa resistance in H. zea in the U.S. Therefore, it is urgent to integrate alternative strategies into the IPM and IRM program to preserve the durability of the Vip3Aa protein in managing H. zea. In this current study, we found that H. zea strains susceptible to Cry Bt proteins but resistant to Vip3Aa Bt proteins, and resistant to both Cry and Vip3Aa Bt proteins, remained susceptible to HearNPV. These results suggest that the HearNPV virus has potential in preventing and delaying the evolution of resistance to Vip3Aa in H. zea. Due to the rise of resistance to Bt crops in Helicoverpa spp., biological control agents are becoming more popular. The renewed interest in alternative biological control agents is also a result of public demand as awareness grows of the potentially detrimental impacts pesticides have on beneficial insects, human health, and the environment [40,41,42]. When applied in conjunction with a holistic Integrated Pest Management (IPM) strategy, NPV-based insecticides provide an effective and environmentally friendly alternative to chemical insecticides [43,44]. Moreover, studies have previously reported the effectiveness of NPV-based insecticides against insecticide-resistant insect pests, including those resistant to Bt proteins [17,39,45,46].
Nevertheless, it is important to acknowledge that the risk for HearNPV resistance development exists, particularly if this new mode of action group is used extensively without the proper implementation of Insect Resistance Management (IRM) programs. Therefore, it is of critical importance to develop sustainable and proactive strategies to delay resistance and promote integrated pest management approaches for using HearNPV in H. zea. Several strategies can be implemented to mitigate the development of HearNPV resistance in H. zea. These approaches include a regular Insect Resistance Monitoring program and the adoption of diverse control measures including crop and insecticide rotation, utilization of biological control agents such as beneficial arthropods (predators and parasitoids), mechanical control, sanitation, and tank-mixes of HearNPV with one or more insecticides that have a distinct mode of action [47,48]. Due to its unique mode of action, HearNPV can effectively contribute to managing the insecticide resistance commonly encountered in H. zea control [17]. Additionally, HearNPV as a selective insecticide has no impact on natural enemies and other beneficial arthropods [16,49]. Consequently, the adoption of HearNPV helps preserve beneficial organisms, maximizes biological control, and mitigates the risk of insecticide-induced secondary pest outbreaks [15,17,49]. In conclusion, by characterizing the susceptibility of different H. zea strains to HearNPV and investigating its correlation with Bt resistance, our study contributes to understanding the potential of HearNPV as an effective tool in managing H. zea. The varying levels of susceptibility to HearNPV among different strains of H. zea and its correlations with specific Bt resistance traits warrant further investigation to understand the underlying mechanisms. These findings have practical implications for developing sustainable strategies to effectively combat insecticide resistance and promote integrated pest management approaches for this pest.
4. Materials and Methods
4.1. Laboratory Insect Strains
A susceptible strain (SS) was obtained from Benzon Research Inc. (Carlisle, PA, USA) and has been previously shown to be susceptible to Cry1Ac, Cry2Ab2, Cry1F, and Vip3Aa Bt proteins [50,51]. A CRY-RR strain was established using an F2 screen method with populations collected in Snook, Texas in 2018 [50]. This strain is resistant to Cry1Ac, Cry1F, and Cry2Ab2 Bt proteins, but susceptible to the Vip3Aa Bt protein [32]. A VIP-RR-70 strain was isolated through F2 screening with moths light-trapped in Snook, Texas in 2019 [50]. A VIP-RR-15 strain was isolated through F2 screening with larvae collected in Stoneville, Mississippi in 2020 [52]. Both VIP-RR-70 and VIP-RR-15 strains are highly resistant to the Vip3Aa Bt protein but are susceptible to Cry1Ac, Cry1F, and Cry2Ab2 Bt proteins [50,52]. A TRE-RR strain was derived through the F2 method by crossing CRY-RR and VIP-RR-70 strains. The TRE-RR strain has been documented to be resistant to Cry1Ac, Cry2Ab2, and Vip3Aa Bt proteins [33]. Prior to this study, all the original resistant strains of H. zea had been backcrossed with SS and reselected for resistance to produce resistant strains that were genetically similar to the SS strain.
4.2. Field-Collected Insect Strains
In 2021, field strains of H. zea were collected from Intrasect (Cry1Ab + Cry1F), DoublePro (Cry1A.105 + Cry2Ab2), and Trecepta (Cry1A.105 + Cry2Ab2 + Vip3Aa) corn (Table 2). Laboratory bioassays revealed that the Thrall-TX and Winnsboro-LA strains were resistant to both Cry1Ac and Cry2Ab2, but susceptible to Vip3Aa (Table 2). The Malone-TX strain was resistant to Cry1Ac but was susceptible to Cry2Ab2 and Vip3Aa. The Alexandria-LA strain was not tested for resistance to Cry1Ac but was susceptible to Cry2Ab2 and Vip3Aa. In 2022, the field-collected populations were obtained from non-Bt corn and crimson clover (Table 2). The Epps-LA, Leland-LA, and Taylor-TX strains were all resistant to both Cry1Ac and Cry2Ab2, but susceptible to Vip3Aa, while the Mariana-AR strain was resistant to Cry1Ac, but susceptible to Cry2Ab2 and Vip3Aa (Table 2).
4.3. Preliminary Tests for Determining HearNPV Concentrations
We used Heligen (AgBiTech, Fort Worth, TX, USA), a commercial insecticide containing occlusion bodies (OBs) of the nucleopolyhedrovirus of Helicoverpa spp. at a concentration of 7.5 × 109 OBs/mL, as the HearNPV inoculum in this study. To determine the appropriate HearNPV concentrations that could cause mortality in H. zea ranging from 0 to 100%, we conducted diet-overlay bioassays using a series of HearNPV concentrations, including 1 × 10, 2 × 10, 3 × 10, 1 × 102, 2 × 102, 3 × 102, 1 × 103, 2 × 103, 3 × 103, 1 × 104, 2 × 104, 3 × 104, 1 × 105, 2 × 105, and 3 × 105 OBs/mL, along with a non-treated control. Based on the preliminary bioassay results, we selected concentrations of 7 × 102, 2 × 103, 7 × 103, 2 × 104, 4 × 104, and 1 × 105 OBs/mL for conducting the full range dose-response bioassays discussed below.
4.4. Dose-Response Bioassays
The aforementioned six concentrations along with a non-treated control were used to determine the susceptibility of H. zea to HearNPV in the diet-overlay bioassays. To facilitate visualization of the solution on the diet, a red food dye (McCormick Culinary, Hunt Valley, MD, USA) at a concentration of 1 µL/mL was added to the treatment solutions. In 2021, we performed bioassays using 29 mL solo condiment cups (Dart Container Corporation, Mason, MI, USA), with each cup filled with 6 mL of liquid diet (Southland Product, Inc., Lake Village, AR, USA). After the diet was cooled, we overlaid 100 µL of HearNPV treatment solution onto the surface of the diet. We then used a fine paintbrush to infest each cup with a second instar H. zea larva and covered the cups with corresponding lids. Each bioassay was replicated 3 to 5 times with 14 to 30 larvae per replication. In 2022, we switched to using 128-well bioassay trays (C-D International, Pitman, NJ, USA), with each well containing 1 mL of liquid diet. We overlaid each well with 50 µL of HearNPV treatment solutions and infested it with a second instar H. zea larva. After infestation, the bioassay trays were sealed with air-vented covers (C-D International, Pitman, NJ, USA), as described in previous studies [32,53]. Each bioassay was repeated 3 to 5 times with 12 to 16 larvae per replication. Both cups and bioassay trays along with infested insects were kept in a climate-controlled room at 25 °C and a photoperiod of 16:8 (L:D). Larval mortality was recorded daily until 10 days after inoculation.
4.5. Data Analysis
Larval mortality for each HearNPV concentration was corrected based on the control mortality according to the Abbott’s formula [54]. Probit analysis [55] was utilized to determine the median lethal concentration (LC50) that caused 50% mortality and the corresponding 95% confidence limit (CL) for each H. zea strain. The resistance ratio was calculated by dividing the LC50 value of the tested strain by the LC50 value of the SS strain. Differences among LC50 values for H. zea strains were assessed by comparing the 95% confidence interval of the LC50 for each strain. Overlapping confidence intervals indicate nonsignificant differences between LC50s, whereas non-overlapping intervals indicate significant differences. In addition, the mortality response of H. zea to HearNPV was analyzed using a two-way ANOVA with insect strain and HearNPV concentration as the two main factors [55]. The means were separated using the Tukey–Kramer adjustment (α = 0.05). To account for heterogeneity, the mortality of each strain was transformed using arcsine transformation, but the non-transformed data are presented. Furthermore, a correlation analysis was performed utilizing Pearson’s correlation coefficient using GraphPad Prism 9.5.0 [56] to determine the correlation between Bt and HearNPV resistance in H. zea.
5. Conclusions
The challenge posed by the evolution of resistance in H. zea to various insecticides and Bt proteins makes pest management highly complex. The utilization of biological agents, such as HearNPV, in the management of H. zea could potentially play a crucial role in addressing this challenge effectively and sustainably. Understanding the baseline susceptibility of insect pests to insecticides is a crucial component of a proactive resistance monitoring program. In this study, we assessed the response of several field and laboratory strains of H. zea to HearNPV and investigated the potential relationship between HearNPV and Bt resistance in H zea. We found that H. zea strains resistant to Cry Bt proteins demonstrated significantly reduced susceptibility to HearNPV, whereas strains resistant to the Vip3Aa Bt protein exhibited high susceptibility to the virus. Correlation analysis indicated a significant positive relationship between Cry resistance and HearNPV resistance in H. zea, whereas no significant correlation was observed between Vip3Aa resistance and HearNPV resistance. Overall, these results suggest that HearNPV has the potential to help manage Vip3Aa resistance in H. zea. Therefore, it is beneficial to integrate HearNPV as an alternative management tool in IPM and IRM programs for H. zea, given the existing widespread Cry resistance in this insect pest. The adoption of HearNPV for H. zea management not only mitigates resistance development in the pest but also offers a reduced-risk insecticide alternative, aligning with the public demand for more sustainable agriculture and a healthier environment. Further investigation is essential to understand the underlying mechanisms responsible for varying levels of susceptibility to HearNPV among the different strains of H. zea, as well as the associations between HearNPV susceptibility and Bt resistance.
Author Contributions
Conceptualization, W.C., D.L.K., and F.Y.; methodology, W.C., D.L.K. and F.Y.; validation, W.C., D.L.K. and F.Y.; formal analysis, W.C., D.L.K. and F.Y.; investigation, W.C. and H.K.; resources, D.L.K. and P.G.M.; data curation, W.C., D.L.K. and F.Y.; writing—original draft preparation, W.C.; writing—review and editing, W.C., D.L.K., F.Y. and P.G.M.; visualization, W.C.; supervision, D.L.K.; project administration, W.C.; funding acquisition, D.L.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by AgBiTech, USDA National Institute of Food and Agriculture: Biotechnology Risk Assessment Research Grants Program 2020-33522-32318/project accession no. 2003504, and Cotton Incorporated.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Acknowledgments
We thank Tyler Towles (Louisiana State University), James Villegas (Louisiana State University), Ben Thrash (University of Arkansas), and Don Cook (Mississippi State University) for assisting in the H. zea collections.
Conflicts of Interest
Author Paula G. Marçon was employed by the company AgBiTech. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Delahaut, K.A.; Cullen, E.M.; Wedberg, J.L. Corn Earworm; Extension Publication A3655; University of Wisconsin: Madison, WI, USA, 2004; Available online: http://corn.agronomy.wisc.edu/Management/pdfs/A3655.pdf. (accessed on 16 February 2021).
- Capinera, J.L. Corn Earworm, Helicoverpa zea; Extension Publication EENY-145; University of Florida: Gainesville, FL, USA, 2020; Available online: https://entnemdept.ufl.edu/creatures/veg/corn_earworm.htm. (accessed on 12 February 2021).
- Foster, R.E. Corn Earworm; Extension Publication E-31-W; Purdue University: West Lafayette, IN, USA, 2017; Available online: https://extension.entm.purdue.edu/publications/E-31/E-31.html. (accessed on 18 February 2021).
- Reay-Jones, F.P.F. Pest status and management of corn earworm (Lepidoptera: Noctuidae) in field corn in the United States. J. Integr. Pest Manag. 2019, 10, 19. [Google Scholar] [CrossRef]
- Ajayi, O.S.; Samuel-Foo, M. Hemp pest spectrum and potential relationship between Helicoverpa zea infestation and hemp production in the United States in the face of climate change. Insects 2021, 12, 940. [Google Scholar] [CrossRef] [PubMed]
- Vemula, S.R.; Porter, P.; Schuster, G.L.; Lewis, B.E. Susceptibility of Helicoverpa zea to commercial insecticides used in green bean production on Texas High Plains. Tex. J. Agric. Nat. Resour. 2012, 25, 1–12. [Google Scholar]
- Towles, T.; Brown, S.; Huang, F. The Cotton Bollworm (Helicoverpa zea): A Pest of Louisiana Row Crops; PUB3777; Louisiana Cooperative Extension Service: Baton Rouge, LA, USA, 2021; Available online: https://www.lsuagcenter.com/profiles/aiverson/articles/page1614272395040 (accessed on 20 March 2023).
- Adams, A.; Gore, J.; Catchot, A.; Musser, F.; Cook, D.; Krishnan, N.; Irby, T. Susceptibility of Helicoverpa zea (Lepidoptera: Noctuidae) neonates to diamide insecticides in the Midsouthern and Southeastern United States. J. Econ. Entomol. 2016, 109, 2205–2209. [Google Scholar] [CrossRef] [PubMed]
- Olmstead, D.L.; Nault, B.A.; Shelton, A.M. Biology, ecology, and evolving management of Helicoverpa zea (Lepidoptera: Noctuidae) in sweet corn in the United States. J. Econ. Entomol. 2016, 109, 1667–1676. [Google Scholar] [CrossRef] [PubMed]
- Reisig, D.D.; Huseth, A.S.; Bacheler, J.S.; Aghaee, M.; Braswell, L.; Burrack, H.J.; Flanders, K.; Greene, J.K.; Herbert, D.A.; Jacobson, A.; et al. Long-term empirical and observational evidence of practical Helicoverpa zea resistance to cotton with pyramided Bt toxins. J. Econ. Entomol. 2018, 111, 1824–1833. [Google Scholar] [CrossRef]
- Reisig, D.D.; Kurtz, R. Bt resistance implications for Helicoverpa zea (Lepidoptera: Noctuidae) insecticide resistance management in the United States. Environ. Entomol. 2018, 47, 1357–1364. [Google Scholar] [CrossRef]
- Reisig, D.; Kerns, D.; Gore, J.; Musser, F. Managing pyrethroid- and Bt-resistant bollworm in southern U.S. cotton. Crops Soils 2019, 52, 30–35. [Google Scholar] [CrossRef]
- Hoy, M.A. Myths, Models, and mitigation of resistance to pesticides. Phil. Trans. R. Soc. Lond. B 1998, 353, 1787–1795. [Google Scholar] [CrossRef]
- Martinez, J.C. Theoretically Tested Remediation in Response to Insect Resistance to Bt Corn and Bt Cotton: A New Paradigm. Ph.D. Thesis, Mississippi State University, Starkville, MS, USA, 2015. Available online: https://scholarsjunction.msstate.edu/td/4705 (accessed on 12 May 2023).
- Blissard, G.W.; Theilmann, D.A. Baculovirus entry and egress from insect cells. Annu. Rev. Virol. 2018, 5, 113–139. [Google Scholar] [CrossRef]
- Gettig, R.R.; McCarthy, W.J. Genotypic variation among wild isolates of Heliothis spp nuclear polyhedrosis viruses from different geographical regions. Virology 1982, 117, 245–252. [Google Scholar] [CrossRef]
- Muraro, D.S.; Gonçalves, T.M.; Amado, D.; Lima, M.F.; Popham, H.J.R.; Marçon, P.G.; Omoto, C. Baseline susceptibility and cross-resistance of HearNPV in Helicoverpa armigera (Lepidoptera: Noctuidae) in Brazil. Insects 2022, 13, 820. [Google Scholar] [CrossRef] [PubMed]
- OSU Extension Service. Corn Earworm Management in Hemp. Available online: https://extension.oregonstate.edu/crop-production/hemp/corn-earworm-management-hemp. (accessed on 17 January 2023).
- Musser, F.R.; Catchot, A.L.; Davis, J.A.; Herbert, D.A.; Lorenz, G.M.; Reed, T.; Reisig, D.D.; Stewart, S.D. 2015 Soybean insect losses in the southern US. Midsouth Entomol. 2016, 9, 5–17. [Google Scholar]
- Stewart, S.; Brown, S.; Steckel, S.; Williams, M.; McClure, A. Insect Control Recommendations for Field Crops: Cotton, Soybean, Field Corn, Sorghum, Wheat, and Pasture; Publication PB 1768; UT Extension: Knoxville, TN, USA, 2022; Available online: http://extension.msstate.edu/sites/default/files/publications/publications/P2471_2022_web_reduced.pdf (accessed on 15 March 2023).
- Calvin, W.; Kerns, D.L.; Green, J.; Gore, J.; Perkin, L.; Vyavhare, S.; Parajulee, M.N.; Schnell, R. Investigating the potential for utilizing grain sorghum as a nursery crop for HearNPV dissemination into cotton. In Proceedings of the Beltwide Cotton Conference, San Antonio, TX, USA, 4–6 January 2022. [Google Scholar]
- O’Reilly, D.R.; Miller, L.K.; Luckow, V.A. Bacculovirus Expression Vectors: A Laboratory Manual; W. H. Freeman and Company: New York, NY, USA, 1992. [Google Scholar]
- Hunter-Fujita, F.R.; Entwistle, P.F.; Evans, H.F.; Crook, N.E. Characteristics of insect pathogenic viruses. In Insect Viruses and Pest Management; Hunter-Fugita, F.R., Entwistle, P.F., Evans, H.F., Crook, N.E., Eds.; John Wiley and Sons: New York, NY, USA, 1998; pp. 7–26. [Google Scholar]
- Boucias, D.G.; Pendland, J.C. General features of viral disease agents. In Principles of Insect Pathology; Boucias, D.G., Pendland, J.C., Eds.; Kluwer Academic Publishers: Norwell, MA, USA, 1998; pp. 31–64. [Google Scholar]
- Rohrmann, G.F. Baculovirus Molecular Biology, 4th ed.; National Center for Biotechnology Information: Bethesda, MD, USA, 2019. Available online: https://www.ncbi.nlm.nih.gov/books/NBK543458/ (accessed on 15 February 2021).
- Asser-Kaiser, S.; Fritsch, E.; Undorf-Spahn, K.; Kienzle, J.; Eberle, K.E.; Gund, N.A.; Reineke, A.; Zebitz, C.P.W.; Heckel, D.G.; Huber, J.; et al. Rapid emergence of baculovirus resistance in codling moth due to dominant, sex-linked inheritance. Science 2007, 317, 1916–1918. [Google Scholar] [CrossRef] [PubMed]
- Asser-Kaiser, S.; Radtke, P.; El-Salamouny, S.; Winstanley, D.; Jehle, J.A. Baculovirus resistance in codling moth (Cydia pomonella L.) caused by early block of virus replication. Virology 2011, 410, 360–367. [Google Scholar] [CrossRef]
- Ignoffo, C.M.; Huettel, M.D.; McIntosh, A.H.; Garcia, C.; Wilkening, P. Genetics of resistance of Heliothis subflexa (Lepidoptera: Noctuidae) to Baculovirus heliothis. Ann. Entomol. Soc. Am. 1985, 78, 468–473. [Google Scholar] [CrossRef]
- Sparks, T.C. Insecticide discovery: An evaluation and analysis. Pestic. Biochem. Phys. 2013, 107, 8–17. [Google Scholar] [CrossRef]
- Miller, A.L.E.; Tindall, K.; Leonard, B.R. Bioassays for monitoring insecticide resistance. J. Vis. Exp. 2010, 46, e2129. [Google Scholar] [CrossRef]
- Pereira, R.M.; Neto, D.A.; Amado, D.; Durigan, M.R.; Franciscatti, R.A.; Mocheti, M.; Omoto, C. Baseline susceptibility and frequency of resistance to diamide insecticides in Helicoverpa armigera (Lepidoptera: Noctuidae) populations in Brazil. Crop Prot. 2020, 137, 105266. [Google Scholar] [CrossRef]
- Yang, F.; González, J.C.S.; Head, G.P.; Price, P.A.; Kerns, D.L. Multiple and non-recessive resistance to Bt proteins in a Cry2Ab2-resistant population of Helicoverpa zea. Crop Prot. 2021, 145, 105650. [Google Scholar] [CrossRef]
- Yang, F.; Kerns, D.L. Stability of Helicoverpa zea strains possessing resistance to Cry1A, Cry2A, and Vip3Aa proteins, and their ability to survive on different blended Bt proteins. In Proceedings of the Beltwide Cotton Conference, New Orleans, LA, USA, 10–12 January 2023. [Google Scholar]
- Kerns, D.; Brown, S.; Catchot, A.; Cook, D.R.; Gonzales, J.; Gore, J.; Crow, W.; Lorenz, G.M.; Stewart, S.D.; Studebaker, G.; et al. Update on Bt resistance in Helicoverpa zea and the value of treating Bt cotton with insecticides. In Proceedings of the Beltwide Cotton Conference, San Antonio, TX, USA, 4–6 January 2022. [Google Scholar]
- Kerns, D.L.; Brown, S.; Cook, D.; Crow, W.; Kennedy, H.D.; Studebaker, G.; Thrash, B.C.; Towles, T.B.; Yang, F. Monitoring Bt resistance and Bt cotton trait performance in 2022. In Proceedings of the Beltwide Cotton Conference, New Orleans, LA, USA, 10–12 January 2023. [Google Scholar]
- Jurat-Fuentes, J.L.; Heckel, D.G.; Ferré, J. Mechanisms of resistance to insecticidal proteins from Bacillus thuringiensis. Annu. Rev. Entomol. 2021, 66, 121–140. [Google Scholar] [CrossRef] [PubMed]
- Popham, H.J.R.; Nusawardani, T.; Bonning, B.C. Introduction to the use of baculoviruses as biological insecticides. In Baculovirus and Insect Cell Expression Protocols. Methods in Molecular Biology; Murhammer, D., Ed.; Humana Press: New York, NY, USA, 2016; Volume 1350, Available online: https://doi.org/10.1007/978-1-4939-3043-2_19 (accessed on 5 May 2022).
- Black, J.L.; Lorenz, G.M.; Cato, A.J.; Bateman, N.R.; Seiter, N.J. Efficacy of Helicoverpa armigera Nucleopolyhedrovirus on soybean for control of Helicoverpa zea (Boddie) (Lepidoptera: Noctuidae) in Arkansas agriculture. Insects 2022, 13, 91. [Google Scholar] [CrossRef]
- Windus, L.C.E.; Jones, A.M.; Downes, S.; Walsh, T.; Knight, K.; Kinkema, M. HearNPV susceptibility in Helicoverpa armigera and Helicoverpa punctigera strains resistant to Bt toxins Cry1Ac, Cry2Ab, and Vip3Aa. J. Invertebr. Pathol. 2021, 183, 107598. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Bayo, F. Indirect effect of pesticides on insects and other arthropods. Toxics 2021, 9, 177. [Google Scholar] [CrossRef]
- Huynh, H.T.; Chan, L.C.L.; Tran, T.T.B.; Nielsen, L.K.; Reid, S. Improving the robustness of a low-cost insect cell medium for baculovirus biopesticides production, via hydrolysate streamlining using a tube bioreactor-based statistical optimization routine. Biotechnol. Prog. 2012, 28, 788–802. [Google Scholar] [CrossRef] [PubMed]
- Pathak, V.M.; Verma, V.K.; Rawat, B.S.; Kaur, B.; Babu, N.; Sharma, A.; Dewali, S.; Yadav, M.; Kumari, R.; Singh, S.; et al. Current status of pesticide effects on environment, human health and it’s eco-friendly management as bioremediation: A comprehensive review. Front. Microbiol. 2022, 13, 962619. [Google Scholar] [CrossRef]
- Duraimurug, P.; Regupathy, A. Push-pull strategy with trap crops, neem and nuclear polyhedrosis virus for insecticide resistance management in Helicoverpa armigera (Hübner) in cotton. Am. J. Appl. Sci. 2005, 2, 1042–1048. [Google Scholar] [CrossRef]
- Ahmed, K.; Awan, M.S. Integrated management of insect pests of chickpea Cicer arietinum (L. Walp) in South Asian countries: Present status and future strategies—A review. Pak. J. Zool. 2013, 45, 1125–1145. [Google Scholar]
- McCutchen, W.F.; Flexner, L. Joint Actions of Baculoviruses and Other Control Agents. In Biopesticides: Use and Delivery. Methods in Biotechnology; Hall, F.R., Menn, J.J., Eds.; Humana Press: New York, NY, USA, 1999; Volume 5. [Google Scholar] [CrossRef]
- Bentivenha, J.P.F.; Rodrigues, J.G.; Lima, M.F.; Marcon, P.; Popham, H.J.R.; Omoto, C. Baseline susceptibility of Spodoptera frugiperda (Lepidoptera: Noctuidae) to SfMNPV and evaluation of cross-resistance to major insecticides and Bt proteins. J. Econ. Entomol. 2019, 112, 91–98. [Google Scholar] [CrossRef]
- PES: Pesticide Environmental Stewardship. Take Steps to Avoid Insecticide Resistance. Available online: https://pesticidestewardship.org/resistance/insecticide-resistance/take-steps-to-avoid-insecticide-resistance/ (accessed on 17 January 2023).
- Wilen, C.A.; Koike, S.T.; Ploeg, A.T.; Tjosvold, S.A.; Bethke, J.A.; Mathews, D.M.; Stapleton, J.J. UC IPM Pest Management Guidelines: Floriculture and Ornamental Nurseries; Publication 3392; UC ANR: Davis, CA, USA, 2013. [Google Scholar]
- Godfrey, L.D.; Goodell, P.B.; Natwick, E.T.; Haviland, D.R.; Barlow, V.M. Selectivity of Insecticides and Miticides; Publication 3444; University of California Agriculture and Natural Resources: Davis, CA, USA, 2013; Available online: https://ipm.ucanr.edu/PMG/r114900711.html#ipmpagetop (accessed on 17 January 2023).
- Yang, F.; González, J.C.S.; Little, N.; Reisig, D.; Payne, G.; Dos Santos, R.F.; Jurat-Fuentes, J.L.; Kurtz, R.; Kerns, D.L. First documentation of major Vip3Aa resistance alleles in field populations of Helicoverpa zea (Boddie) (Lepidoptera: Noctuidae) in Texas, USA. Sci. Rep. 2020, 10, 5867. [Google Scholar] [CrossRef]
- Yang, F.; Kerns, D.L.; Little, N.S.; González, J.C.S.; Tabashnik, B.E. Early warning of resistance to Bt toxin Vip3Aa in Helicoverpa zea. Toxins 2021, 13, 618. [Google Scholar] [CrossRef]
- Santiago-Gonzalez, J.C.; Kerns, D.L.; Head, G.P.; Yang, F. A modified F2 screen for estimating Cry1Ac and Cry2Ab resistance allele frequencies in Helicoverpa zea (Lepidoptera: Noctuidae). J. Econom. Entomol. 2023, 116, 289–296. [Google Scholar] [CrossRef]
- Santiago-Gonzalez, J.C.; Kerns, D.L.; Yang, F. Resistance Allele Frequency of Helicoverpa zea to Vip3Aa Bacillus thuringiensis protein in the southeastern U.S. Insects 2023, 14, 61. [Google Scholar] [CrossRef]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econom. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- SAS Institute. User’s Manual, version 9.4; SAS Institute Inc.: Cary, NC, USA, 2011.
- GraphPad Prism. GraphPad Software, version 9.5.0; GraphPad Prism: San Diego, CA, USA, 2022. Available online: www.graphpad.com (accessed on 3 January 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).